Abstract
Background/Hypothesis:
Calcitonin gene-related peptide monoclonal antibodies (CGRP mabs) are relatively new preventive treatments in adult migraine. Co-morbid medical conditions such as autoimmune or other neurologic/oncologic disorders are not uncommon in migraine patients and some exhibit notable co-morbidity such as asthma. These clinical conditions may necessitate concomitant treatment with another monoclonal antibody with a different mechanism of action other than the CGRP pathway in some individuals with migraine.
Methods:
We report a retrospective case series in 23 patients identified from our headache clinic treated concurrently with a CGRP monoclonal antibody and other monoclonal antibody for another medical condition. These other medical conditions were neurologic, oncologic, or autoimmune conditions. These patients were evaluated for tolerability, safety, and stability of disease processes including their migraine response.
Results/Conclusion:
We did not find evidence of new adverse or serious adverse side effects with coadministration of CGRP and non-CGRP monoclonal antibodies during our study time period of 13 months, for the duration of overlap between treatments (between 3 and 12 months).
Keywords
Introduction
Current research elucidates that calcitonin-gene related peptide (CGRP) plays a pivotal role in migraine genesis. 1 Recent development of the CGRP monoclonal antibodies is an important addition to the treatment paradigm for managing migraine preventive therapy in adults. 2 There are four current CGRP monoclonal antibodies used in migraine prevention for the past several years including erenumab, fremenazumab, galcanezumab, and eptinezumab. They have demonstrated efficacy, tolerability, and safety when used in episodic and chronic migraine. More specific mechanism of action includes CGRP receptor binding in erenumab and binding to the CGRP ligand in the other CGRP monoclonal antibodies. 3
There has been utilization of monoclonal antibodies across all medical specialties including autoimmune disorders, asthma, neurology, and oncology. These have included asthma, eczema, rheumatoid arthritis, psoriasis, and gastrointestinal disease processes such as Crohn’s disease. Many of these conditions occur co-morbidly with migraine. The monoclonal antibodies have specific targeted mechanisms of action such as CGRP in migraine and others such as interleukin 23 and 12, tumor necrosis factor, or anti-myelin oligodendrocyte glycoprotein in other monoclonal antibodies. 4,5
Further investigation is needed in using multiple monoclonal antibodies chronically in the same patient for different clinical conditions due to continued frequent use of these therapies. Evaluating tolerability in these patients is useful information with ever increasing use of the monoclonal antibodies for multiple disease processes. It can be assumed that monoclonal antibodies are appropriate for combination therapy due to their targeted mechanisms of action and lack of pharmacokinetic interactions; however, there is little data in the literature reporting these findings. 6,7
Methods
We utilized a retrospective case series headache clinic chart review from our medical records (EPIC), identifying adult migraine patients from 18 to 70 years of age from our headache clinic. These patients presented between February 1, 2020 and March 31, 2021. Sample size was based on available data. Patients identified were undergoing treatment with a CGRP monoclonal antibody with concurrent diagnosis of cancer or autoimmune disease and also receiving treatment with a monoclonal antibody for their other disease process. The diagnosis of migraine was made using the International Classification of Headache Disorders-ICHD-3. 8 Approval for this study was obtained from our Institutional Review Board.
Demographic information obtained included age, sex, diagnosis of episodic versus chronic migraine, migraine frequency, monoclonal antibodies being used, and other disease condition treated. Tolerability of the combined therapy was obtained from the medical record visits.
Results
We identified 23 patients who were on both CGRP monoclonal antibodies for episodic or chronic migraine and one other monoclonal antibody for another medical condition (see Table 1). The patients’ ages were between 19 years old and 69 years old. Nineteen patients were female with four being male. Three of the patients were on fremanezumab, eight were on erenumab, eleven were on galcanezumab, and one was on eptinezumab. The other medical conditions than migraine requiring another monoclonal antibody were variable and included: ulcerative colitis (1), asthma (1), aortitis (1), hidradenitis suppurativa (1), rheumatoid arthritis (3), hyperlipidemia (1), stiff person syndrome (1), sarcoidosis (1), psoriatic arthritis (4), anti-myelin oligodendrocyte glycoprotein syndrome (1), Crohn’s disease (4), lymphoplasmacytic lymphoma (1), multiple sclerosis (2), and osteoporosis (1). The other monoclonal antibodies that were used in other medical conditions than migraine included: vedolizumab (1), omalizumab (1), tocilizumab (1), adalimumab (2), evolocumab (1), infliximab (5), rituximab (3), abatacept (2), etanercept (1), golimumab (1), ustekinumab (2), natalizumab (1), denosumab (1), and ocrelizumab (1). At the time of our investigation, the patients involved were on overlapping therapy with a CGRP monoclonal antibody and another monoclonal antibody between 3 months and 12 months. Prior to CGRP monoclonal antibody therapy, the patients, where the data was available, had an average total headache days per month of 22.3, and an average migraine days per month of 16.6. Following CGRP monoclonal antibody therapy, the patients had an average total headache days per month of 16.6, and an average migraine days per month of 9.8. Among all the patients, the only side effects reported were constipation, hair loss, and injection site reactions. There were no serious adverse events reported. In all 23 patients, they remained on the other non-CGRP monoclonal antibodies for the duration of overlap of treatment with CGRP monoclonal antibodies without adjustments in dosage or discontinuation of the non-CGRP antibody.
Patients on Dual Monoclonal Antibodies.
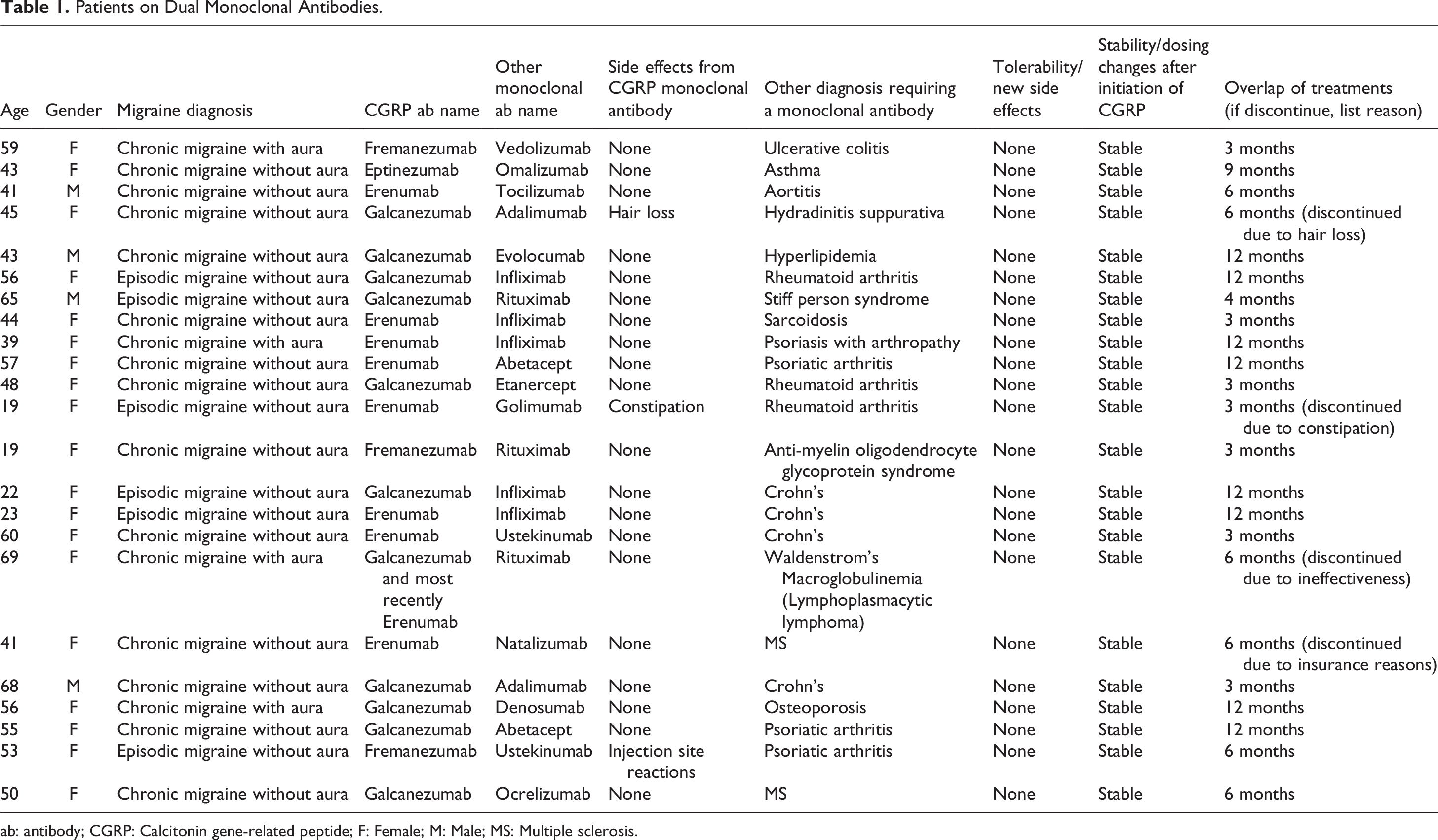
ab: antibody; CGRP: Calcitonin gene-related peptide; F: Female; M: Male; MS: Multiple sclerosis.
Discussion
Many migraine patients have other underlying medical conditions that may necessitate the use of monoclonal antibodies. Our data supports that CGRP monoclonal antibodies can be safe and well-tolerated when used in conjunction with other monoclonal antibodies. The only reported side effects found during our chart review were constipation, hair loss, and injection site reaction. These three side effects have been well described and widely reported with CGRP monoclonal antibodies alone, so it is unlikely that the reported side effects are related to the combination of monoclonal antibodies. Furthermore, no patients had adjustments in dosage or discontinuation of their other non-CGRP monoclonal antibodies for the duration of overlap of treatment.
Limitations of our study are that each side effect was counted quantitatively as opposed to any measure of qualitative severity, a limitation of the data interpretation. Qualitative metrics of the medication side effects in terms of frequency, intensity, and duration could be included in future studies. Also, we did not have access to all of the records from specialists/providers prescribing the other non-CGRP monoclonal antibody medications. It is possible that the patients did not report to our headache team side effects or lack of efficacy with the other monoclonal antibody medications, thinking it to be unrelated or irrelevant to their headache clinic visits. Future prospective studies could ask specific questions about side effects and efficacy of both monoclonal antibody medications. Our study patient number is small, heavily populated with women patients, and only within one institution, which could limit the generalizability of study. Our retrospective chart review design could easily be reproduced across multiple institutions to increase the sample size of the study. We also did not evaluate for race/ethnicity, limiting the generalizability of our study. This could be evaluated in further studies. Finally, headache and migraine day frequency was not collected on every single patient, nor was the duration of CGRP monoclonal antibody treatment consistent among patients, limiting the generalizability of our study in the context of CGRP monoclonal antibody efficacy. Future studies could monitor CGRP monoclonal antibody therapy for the standard 3-month period seen in previous clinical trials. We may have been more likely to choose monoclonal antibodies in patients already tolerating them due to familiarity with administration/patient preference/patient tolerability, creating selection bias. Also, the headache providers may have been biased to ask more about side effects on patients taking two monoclonal antibodies.
Theoretically, given CGRP monoclonal antibodies’ unique targeted mechanism of action targeting either CGRP itself or the CGRP receptor, it can be inferred that CGRP monoclonal antibodies would likely have minimal interaction with other monoclonal antibodies with different mechanisms of action. However, from our literature review, there was limited data reporting these findings. Further, ideally prospective investigation would be needed to establish statistical significance of any clinically significant crossover effects (or lack thereof) of combination monoclonal antibody therapy. The power of our data is obviously limited due to small sample size of 23, and further investigations are needed regarding the safety and tolerability of using multiple monoclonal antibodies for a single patient. Our data, however, is a good starting point for future investigations.
Clinical implications
Many patients take both CGRP antibodies along with other monoclonal antibodies for other medical conditions. Our limited study found there to be tolerability of coadministration of these different antibodies.
Future prospective studies could ask specific questions about side effects and efficacy of both monoclonal antibody medications.
Footnotes
Author contributions
The idea of the study was conceived by Kevin Weber and Ann Pakalnis. Acquisition of data was performed by Tanner Ferderer. Kevin Weber, Ann Pakalnis, and Meghan Hubert drafted the manuscript and revised it critically for important intellectual content. Kevin Weber, Tanner Ferderer, Meghan Hubert, and Ann Pakalnis contributed to final approval of the version submitted.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Weber: grants for research: Lundbeck (no personal compensation), Lilly (no personal compensation). Dr Pakalnis: grants for research: Amgen (no personal compensation; consultant (Honoraria) Abbvie. Dr Ferderer and Dr Hubert have nothing to disclose.
Ethical approval and patient consent
This study, as it was a retrospective case-series review with de-identified data, was considered exempt by the Ohio State University Institutional Review Board (2021E0396). Patient consent was not obtained due to this study’s stats as a retrospective case-series review with de-identified data. The risk to patients was considered minimal.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
