Abstract
Background:
The majority of CGRP monoclonal antibodies for migraine prevention are administered subcutaneously. Therefore, we attempted to calculate the pooled placebo response with subcutaneous placebo injections in this systematic review and meta-analysis.
Methods:
We identified 16 randomized controlled trials that met our inclusion and exclusion criteria through a comprehensive search in five electronic databases (PubMed Central, EMBASE, MEDLINE, Cochrane library and clinicaltrials.gov). The risk of bias was assessed for all included studies. Random effects model was used to calculate pooled mean monthly migraine days and 50% response rates.
Results:
A total of 4240 subjects were included from 16 studies in this meta-analysis. The pooled mean monthly migraine day reduction with subcutaneous placebo injections was 2.15 (95% CI: 1.60–2.69). The pooled proportion of patients achieving a 50% reduction in mean monthly headache days was 26% (95% CI: 20%–31%). Placebo response accounted for more than 50% of therapeutic gain in our study.
Conclusion:
A substantial placebo response was noted with subcutaneous injections in migraine CGRP monoclonal antibody clinical trials. This meta-analysis may serve as a reference point to calculate sample size in clinical trials using subcutaneous interventions for migraine prevention. We registered our study at PROSPERO (CRD42020185300).
Introduction
Placebo effect is a complex neurobiological phenomenon that may either mimic or enhance the therapeutic response. 1,2 Conditioning, desire, expectations, beliefs and emotions are the key psychological factors that drive the placebo effect. 2 –4 Headache, like many other pain disorders, can invoke multiple behavioral components that constitute a placebo response. 5 –8 Migraine headache, a common and debilitating primary headache disorder, is an excellent model to quantify placebo analgesia.
Several meta-analyses estimated the magnitude of placebo response with migraine medications in the past. 9 –14 Previous meta-analyses reported a mean placebo response of 24–30% with acute abortive therapy. 5 –9 Placebo response is higher with acute abortive treatments than preventative therapy. 9 The placebo effect in oral migraine preventative therapy clinical trials was shown to vary from 14% to 31%. 10 Among the preventative interventions, acupuncture showed the highest placebo effect at 50%. 11,12 A meta-analysis of 22 oral migraine prophylaxis randomized controlled trials (RCT) showed a 50% responder rate (50% RR) of 23.5 ± 8.0% and a 16.8 ± 12.7% reduction in migraine attack frequency with placebos. 13 Macedo et al. conducted a meta-analysis of 32 RCTs and reported a 50% response rate with placebo in 21% [13–28%] of patients; 18% reduction in mean monthly migraine attacks was noted with oral placebos when compared to oral prophylactic medications. 14
The route of placebo administration may alter the magnitude of the overall therapeutic response. 15 Subcutaneous (subQ) placebos were 6.7% (95% CI 2.4–11.0%) more effective than oral placebos in reducing headache intensity 2 hours after the treatment. 8 However, a network meta-analysis from 2013 reported contradictory findings with no significant advantage of parenteral medication delivery over the enteral route. 16 Invasive procedures, such as sham surgery and sham acupuncture exert a greater placebo effect than oral placebos. 16
Oral medications have been the mainstay of migraine preventative treatment for decades. The recent approval of calcitonin gene-related peptide monoclonal antibodies (CGRP MAB) – erenumab, 17 –21 galcanezumab, 22 –26 and fremanezumab 27 –29 – transformed the landscape of migraine preventative treatment. 30 CGRP MAB have a unique dosage frequency and route of administration: all three biologics are administered monthly via subQ injections, with fremanezumab also having an option for quarterly administration. 31 In preapproval clinical studies of all three CGRP MAB, the percentage of patients reporting >50% reduction in migraine headache days with placebo was comparable with previous studies for other preventative treatments. 17 –29 Considering the novel route and frequency of CGRP MAB administration, we hypothesized that subQ injections might exert a distinct placebo response than oral medications. A better understanding of this placebo response will aid in designing future research studies and clinical trials with optimal sample size and scientific rigor. Therefore, we aimed to conduct this meta-analysis and systematic review to re-evaluate the pooled placebo response with subQ injections in migraine prevention.
Methods
We conducted a systematic review and meta-analysis of RCTs. We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist in our review. 28 –32 We registered our study protocol at the prospective register of systematic review (PROSPERO) prior to the synthesis of the data to avoid unintentional duplication. The registration number is CRD42020185300. Institutional review board approval is not required for meta-analysis at our institution.
Search strategy
Two authors (AB and LA) conducted a systematic database literature searches in PubMed Central, EMBASE, MEDLINE, Cochrane library and clinicaltrials.gov with keywords “Migraine Headache,” “Migraine,” “Randomized Controlled Trial,” “Placebo,” “Calcitonin Gene-Related Peptide,” “Erenumab,” “Galcanezumab,” and “Fremanezumab” from January 2010 through July 2020. We did literature search and data entry in August 2020. In the first step, free text searching of the keywords and their synonyms was performed using appropriate truncations, wildcards and proximity searching. We also searched for key concepts using corresponding subject headings in each database. We conducted the final search by combining the individual results using appropriate Boolean operators. The searches were complemented by screening the references of selected articles to find those that did not appear in the search databases. Additional verification was performed by a simple Google search.
Initial screening was conducted on Mendeley’s reference manager. A blinded full-text screening of the eligible studies was done on a web-based platform (https://www.rayyan.ai/) by 2 authors (SG, MG) and further verified by AB, who served as a third reviewer.
Eligibility criteria
Data collection
Data were extracted manually from the included studies using a structured data extraction form, developed and pilot tested using the “Cochrane Consumers and Communication Review Group” Data Extraction Template. 34 General information was collected about the study, such as author, year of publication, title, information related to methods section (such as study design), sample sizes, study participants, inclusion and exclusion criteria, quality-related information, and outcome measures.
Risk of bias
The risk of bias was assessed using the Cochrane Risk of Bias tool for RCTs. 35 The assessment was done and reported according to the Cochrane Consumers and Communication Review Group guidelines, which recommend the detailed reporting of the following individual elements for RCTs: “random sequence generation, allocation sequence concealment, blinding (participants, personnel), blinding (outcome assessment), completeness of outcome data, selective outcome reporting and other sources of bias.” The risk of bias was assessed at the study level.
Statistical analysis
We performed a meta-analysis with the final set of selected studies using the R software (R Foundation for Statistical Computing, Vienna, Austria). Random effects model was applied because of the anticipated heterogeneity and final data was reported as pooled prevalence with 95% confidence interval (CI) for outcomes such as the proportion of patients with a 50% reduction in MMD, and pooled mean with 95% CI for continuous outcomes such as placebo and interventional drugs response in MMD reduction. Visual representation of these pooled estimates was done by forest plot.
Heterogeneity was evaluated by chi-square of heterogeneity and I2 statistic. A p-value less than 0.10 in the chi-square test indicates significant heterogeneity, whereas the I2 value was used to quantify the heterogeneity using the following criteria: less than 25% = mild heterogeneity, 25–75% = moderate heterogeneity and >75% = substantial heterogeneity. 35 We performed subgroup analyses based on types of migraine and follow-up period due to the high heterogeneity of the study characteristics. Post hoc study design-based subgroup analysis was conducted to analyze the influence of unequal allocation ratio. RCTs included in this meta-analysis were divided into studies with near equal medication to placebo allocation ratio and higher medication to placebo ratio (unequal allocation ratio, i.e. > 1:1). Publication bias was evaluated and visually represented through a funnel plot. We also assessed the asymmetry of the plot using Egger’s test. P-value <0.10 was considered a statistically significant publication bias.
Results
Study selection process
We identified 16 eligible studies after screening 236 results through a systematic literature search, as illustrated in the PRISMA flow diagram. During the second screening stage, 16 studies met the eligibility criteria and were included in our review (Figure 1). 17 –29,36,37
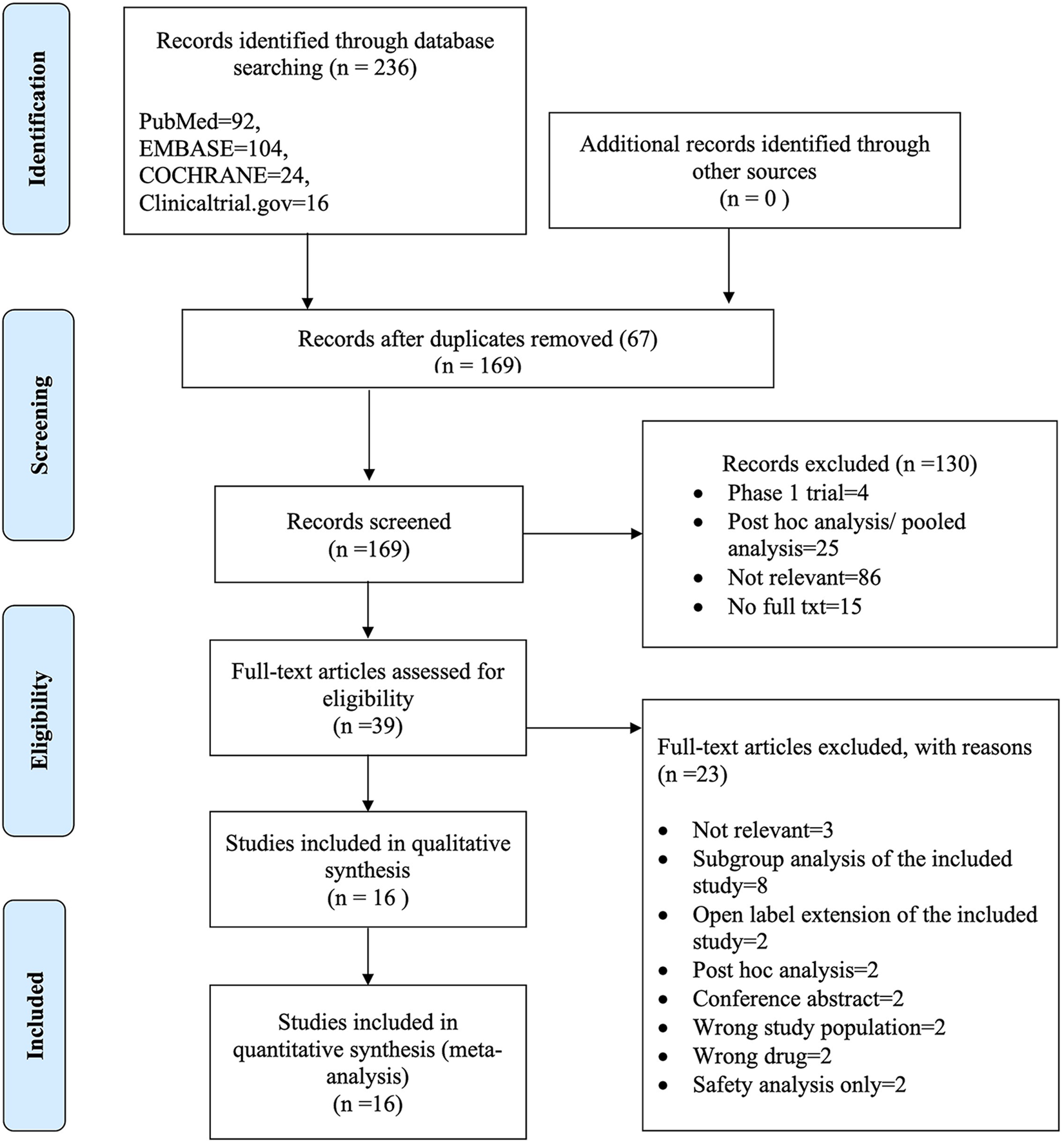
PRISMA flow diagram.
Characteristics of included studies
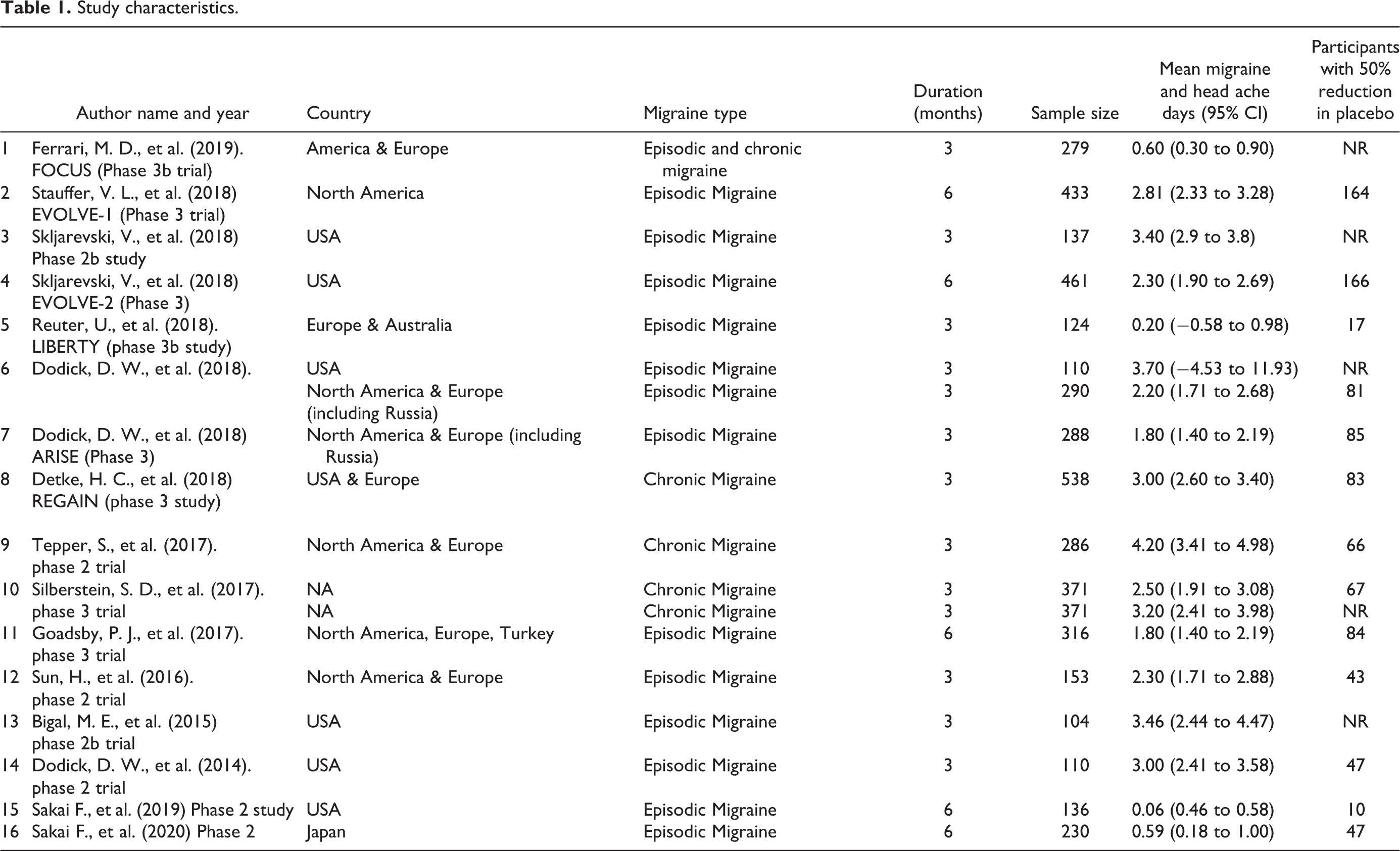
Characteristics of the studies are described in Table 1. 16 RCTs-Erenumab (6), Fremanezumab (5) and Galcanezumab (5)-were included in this meta-analysis and systematic review. The sample size varied from 104 to 538 in the included RCTs. The reduction in MMD and 50% RR were reported in 16 and 12 studies; 5 RCTs reported a reduction in monthly headache days (MHD) as an outcome measure. Out of 16 RCTs, 12 included patients with EM, 3 had patients with CM, and one study included both EM and CM patients. Most studies were conducted in North America or Europe. Subjects were enrolled in Australia in one study and 2 studies were done in Japan. Out of 16 studies, 11 studies were conducted for 3 months and 5 studies for 6 months. All analyzed studies had a low to unclear risk of bias with respect to all the domains in the Cochrane risk of bias tool and all studies satisfied the criteria to be a high-quality study (Figure 2).
Study characteristics.

Forest plot demonstrating pooled placebo response of subcutaneous injections as measured by mean monthly migraine day reduction of all 16 studies included in this meta-analysis. Risk of bias assessment was also illustrated. A = Random sequence generation, B = Allocation concealment, C = Blinding of participants and personnel, D = Blinding of outcome assessment, E = Incomplete outcome data assessment, F = Selective reporting, G = Other bias.
Pooled placebo response of subcutaneous injections used for migraine prevention
Reduction in MMD and 50% response rate (Figures 2 and 3)

Forest plot demonstrating the pooled placebo response of subcutaneous injections as measured by the proportion of subjects with a 50% reduction in mean monthly migraine days.
The pooled MMD reduction was 2.15 (95% CI: 1.60–2.69). However, the heterogeneity was substantial (I2 = 94%; chi-square test for heterogeneity = p < 0.001). Twelve studies reported 50% RR and the pooled proportion of patients achieving a 50% reduction in MMD was 26% (95% CI: 20%–31%). Substantial heterogeneity was also noted with 50% RR (I2 = 94%; chi-square test for heterogeneity p < 0.001). The funnel plot did not show evidence of publication bias. (Eggers test for studies that reported MMD and 50% RR showed a p-value of 0.38 and 0.19, respectively).
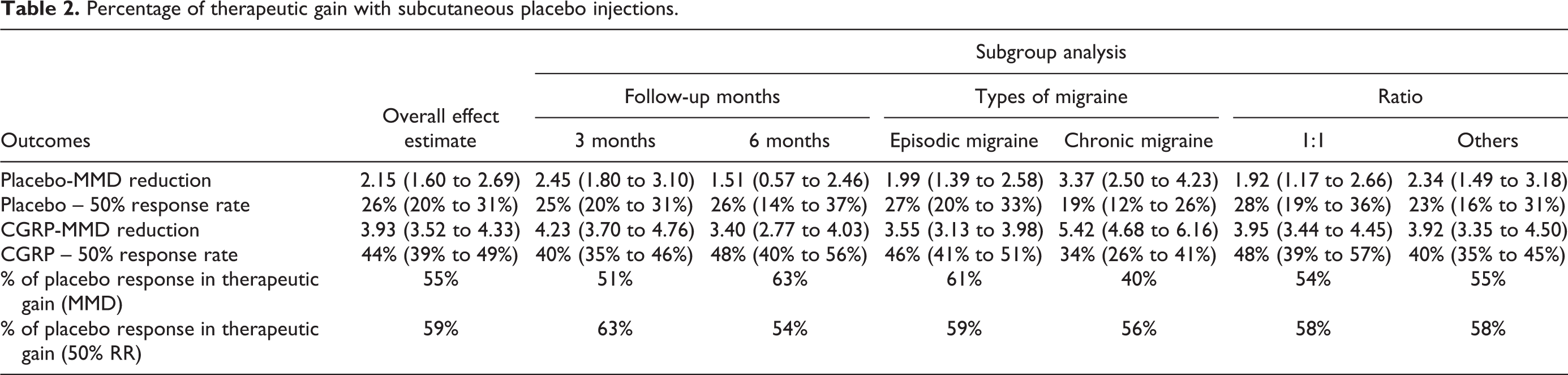
Subgroup analysis (Table 2 and supplementary figures A, B, C and D)
We performed subgroup analysis based on the duration of the study (11 studies lasted 3 months and 5 studies were completed after 6 months). The pooled mean reduction in MMD was higher in the 3-month group (2.45; 95% CI 1.80–3.10; I2 = 92%) compared with the 6-month group (1.51; 95% CI: 0.57–2.46; I2 = 96%). The pooled proportion of 50% reduction in MMD was higher in the 6-months group (26%; 95% CI: 14% -37%; I2 = 97%) compared with 3-month group (25%; 95% CI 20% -31%; I2 = 89%).
We also analyzed the variation in placebo response between patients with EM and CM. The pooled mean reduction in MMD was higher in RCTs that included patients with CM (3.37; 95% CI: 2.50–4.23; I2 = 73%) compared to EM (1.99; 95% CI: 1.39–2.58; I2 = 94%). The pooled 50% RR was higher in EM RCTs (27%; 95% CI: 20%–33%; I2 = 93%) compared to CM (19%; 95% CI: 12%–26%; I2 = 85%).
A posteriori subgroup analysis was conducted based on the medication to placebo allocation ratio. Equal allocation was noted in 7 studies and unequal allocation was identified in 9 studies. Higher pooled mean MMD reduction was noted in the unequal/greater medication to placebo ratio group (2.34; 95% CI 1.49–3.18; I2 = 95%) compared to the studies with the equal allocation (1.92; 95% CI 1.17–2.66; I2 = 94%). The pooled proportion of 50% reduction in MMD was higher in RCTs with equal medication to placebo allocation ratio (28%; 95% CI: 19% -36%; I2 = 95%) compared to unequal/greater medication to placebo allocation (23%; 95% CI 16%–31%; I2 = 91%).
Placebo response accounted for more than 50% of therapeutic gain due to CGRP MAB sub Q injections; 55% of MMD reduction and 59% of 50% RR due to subQ therapeutic interventions were found to be due to placebo response (Table 2).
Percentage of therapeutic gain with subcutaneous placebo injections.
Discussion
Our meta-analysis and systematic review demonstrated a substantial placebo response in migraine prophylaxis with subQ injections (pooled mean MMD reduction of 2.15 and a pooled 50% RR of 26%). The 50% RR with subQ placebos identified in this meta-analysis (26%) was higher than the previous meta-analysis by Macedo et al. with oral placebos (21%). 6
Invasive placebo interventions evoke unpleasant, painful sensations. Subjects involved in invasive clinical trials have to be more motivated to participate and it transforms into higher anticipation of therapeutic benefits because of their greater emotional investment. 38 Moreover, pain during the procedure triggers an instantaneous expectation of symptom relief from the placebo. 38 Botox PREEMPT trials in which 31 sites around the head were injected with either placebo or Botox demonstrated higher placebo responses, attributed to the enhanced invasiveness of the procedure. 15
Several neuroimaging studies supported the neurocognitive hypothesis of placebo analgesia. In 2021, the placebo imaging consortium published a “mega-analysis,” a meta-analysis of whole-brain analysis including 603 “healthy subjects” from 20 studies, showing the neuroimaging (functional MRI) correlates of placebo analgesia. 39 As expected, subjects who reported higher placebo analgesia showed a greater reduction in pain construction area activity (insula, thalamus, midcingulate, habenula, and supplementary motor areas). 39 The neural correlates of placebo analgesia did not differ significantly between migraine patients and healthy subjects, as illustrated by Linnman et al. 39 The preliminary neuroimaging study (opioid ligand 11C-diprenorphine PET scan and functional MRI) with 9 controls and 9 migraine patients did not reveal any difference in brain activation related to placebo responses between the two groups. 40
We hypothesized that migraine headache characteristics influence the placebo effect. Hence, we investigated the impact of migraine-type on placebo response in subgroup analysis. Given the high prevalence of associated psychiatric comorbidities and increased headache burden in CM, a higher placebo response was expected. 41,42 A recent meta-analysis that analyzed the placebo response of 5672 CM patients from 7 migraine prevention RCTs showed a greater placebo response with injections to the head in Botox trials followed by intravenous injections; subQ route and oral placebos were inferior to head injections. 15 75% of the therapeutic gain was explained by the placebo response in that study. 15 In contrast with that assumption, therapeutic nihilism is common in chronic migraine patients and may decrease the overall response to pharmacological interventions, including placebo. This phenomenon was demonstrated in a recent mindfulness-based cognitive therapy clinical trial that showed higher efficacy in patients with EM than CM. 43 In our study, a higher MMD reduction with subQ placebos in chronic migraine was noted. However, 50% response rate was lower in CM group when compared to EM and it is because a CM patient with higher baseline migraine days needs a substantially higher reduction in the number of migraine days per month than a EM patient with lower baseline migraine frequency, to accomplish 50% reduction. In our meta-analysis, the average baseline MMD of placebo arms in RCTs with CM is 18 and EM is 8.5. This finding emphasizes the fact that 50% RR may not serve as an optimal primary outcome measure in CM trials as smaller effect sizes can be missed.
RCT duration has the potential to modify the placebo effect. However, the association between study duration and the placebo effect has been inconsistent. A higher placebo effect was reported in RCTs with shorter follow-up times, especially in psychiatry. 44,45 A long-lasting placebo effect is also not an uncommon phenomenon. A sustained placebo effect lasting 6 months was noted in a pooled analysis that investigated the effect of topiramate on migraine headaches. 46 A prophylactic study examining the impact of botulinum toxin A treatment showed a lasting placebo effect for 9 months. 47 A meta-analysis of 47 RCTs evaluated the impact of sham surgery and showed sustained and consistent effect for 12 months. 48 In our study, we observed superior MMD reduction with a shorter study duration i.e. for 3 months; this finding indicates that the placebo effect on migraine headaches weans over time with subQ injections. However, studies conducted for 6 months showed a slightly higher pooled 50% RR when compared with 3 month RCTs (26% vs 25%). It is interesting to note that the presence of CM RCTs in the 3 month group negatively influenced the 50% response rate as expected; it is supported by the higher average baseline MMD in 3 month RCT placebo group (11) vs 6 month RCT group (8). The 1% difference between 3 month and 6 months groups does not represent a meaningful clinical significance.
We investigated the effect of medication to placebo allocation ratio on the placebo effect as the therapeutic expectation changes with the knowledge of allocation probability. 10,49 Studies with a 1:1 medication to placebo allocation ratio or near-equal allocation ratio were shown to produce lesser placebo response when compared to RCTs with unbalanced randomization that increases the probability of receiving the medication (medication to placebo allocation ratio > 1:1). 44,45,50 Out of 16 RCTs, 7 clinical trials used 1:1 randomization design (41%). All galcanezumab RCTs used a 1:1 design, whereas all fremanezumab trials utilized 1:2 randomization protocols. 2 RCTs in the erenumab group also utilized a 1:1 design. As expected, pooled MMD reduction was lower with studies that used 1:1 randomization (1.92) compared to RCTs with a higher probability of receiving medication (2.34). The pooled 50% RR was higher in 1:1 group (28%) compared to the unequal randomization group (23%) and it is likely due to higher baseline migraine headache frequency in unequally randomized studies (11.1 vs 10.1).
Varied results observed in our subgroup analysis – as measured by the two conventional outcomes, MMD, and 50% RR – were explained by the baseline migraine headache frequency. MMD and 50% RR represent unique migraine characteristics and may not be used interchangeably. Furthermore, the differences reported in sub-group analyses are not statistically significant as the 95% confidence intervals overlapped. The results were reported as it is to avoid selective reporting of statistically significant findings.
All sixteen studies in our meta-analysis used MMD as an outcome measure; 12 studies reported 50% RR and only 5 studies utilized monthly headache days. This is a concerning finding and it is in line with the results of a recent systematic review of 268 preventative clinical trials that identified a high degree of variability and lack of standardization. 51,52 The discrepancy was profound in preventative trials compared to acute migraine trials. 51 Migraine-specific outcomes such as MMD and 50% RR were used in 69% of studies, headache-specific outcomes in 40% of studies and acute rescue treatment days in 51% of studies, independently or concomitantly. 51,52 The choice of outcome measure in a clinical trial is predominantly based on the nature of the research question and resource availability. Furthermore, a high degree of inconsistency in outcome measures may negatively influence the results of a meta-analysis. Also, the discrepancy in the outcome measures in this subgroup analyses could be due to unexplored variations in the sample characteristics.
There are some notable limitations in this study. The true placebo effect is defined as the mean difference in effect size between the placebo and non-intervention arms. Hence, we did not “technically” investigate the “true placebo effect.” 53 Hawthorne effect – a behavioral phenomenon where individuals report changes exclusively due to study participation – has the potential to alter the outcomes both in clinical trials and routine practice. 54 It is not feasible to control all the potential confounding factors, such as physiological disease fluctuations, report/recall bias, inaccurate over-the-counter pain medication usage data, and under or undiagnosed psychiatric comorbidities. Despite the extensive analysis, we could not identify the reason for the high prevalence of heterogeneity in our meta-analysis. Some of the included studies reported patient-related outcome measures, but we did not analyze them in our meta-analysis.
Conclusion
Our meta-analysis and systematic review re-demonstrated the significance of placebo response in migraine headache preventative treatments. Subcutaneous injections revealed a greater placebo response when compared with the results from oral prophylactic medication meta-analyses. Further studies to emulate and harness placebo analgesia may increase the efficacy of preventative migraine medications in clinical practice.
Supplemental material
Supplemental Material, sj-pdf-1-rep-10.1177_25158163221120103 - Placebo response with subcutaneous injections in calcitonin gene-related peptide receptor monoclonal antibody migraine preventative trials – A systematic review and meta-analysis
Supplemental Material, sj-pdf-1-rep-10.1177_25158163221120103 for Placebo response with subcutaneous injections in calcitonin gene-related peptide receptor monoclonal antibody migraine preventative trials – A systematic review and meta-analysis by Sasikanth Gorantla, Murali Mohan Reddy Gopireddy, Archana Bhat, Lavanya Ayyasamy, Sarath Kumar Jaganathan Jaishankar, Bassil Kherallah and Hrachya Nersesyan in Cephalalgia Reports
Footnotes
Abbreviations
Article highlights
In our study, the placebo response with subcutaneous injections is higher than that of oral medications reported in previous meta-analyses. Mean monthly migraine day (MMD) reduction of 2.15 (1.60–2.69) was noted with subcutaneous (subQ) placebos. 26% (20–31%) of study participants had a 50% reduction in MMD with subQ placebos. More than half of therapeutic gain was due to subQ placebos in our study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this manuscript is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
