Abstract
Background
To evaluate the benefit-risk assessment of atogepant and calcitonin gene–related peptide (CGRP) monoclonal antibodies (mAbs) vs. placebo based on the number needed to treat (NNT) and the number needed to harm (NNH) in a blended episodic migraine and chronic migraine (EM + CM) population.
Methods
The NNT was calculated based on achievement of a ≥ 50% reduction in mean monthly migraine days (MMDs) from baseline across 12 weeks. The NNH was calculated using the proportion of participants reporting a discontinuation due to adverse events (AEs). The primary analysis included data from studies of atogepant, erenumab, galcanezumab, eptinezumab and fremanezumab.
Results
In the primary analysis, the calculated NNT for atogepant 60 mg vs. placebo was 4.2 (95% credible interval (CrI) = 3.1–6.7), which was the lowest relative to the CGRP mAbs in the blended EM + CM population. Participants who received atogepant 60 mg or fremanezumab showed the most favorable NNH values (−1010 (95% Crl = 44 to ∞ to number needed to benefit 80) for atogepant) resulting from lower rates of discontinuation due to AEs compared with those receiving placebo.
Conclusions
Atogepant demonstrated a favorable benefit-risk profile, with NNT and NNH values comparable (not statistically significant) with those of CGRP mAbs across all analyses.
This is a visual representation of the abstract.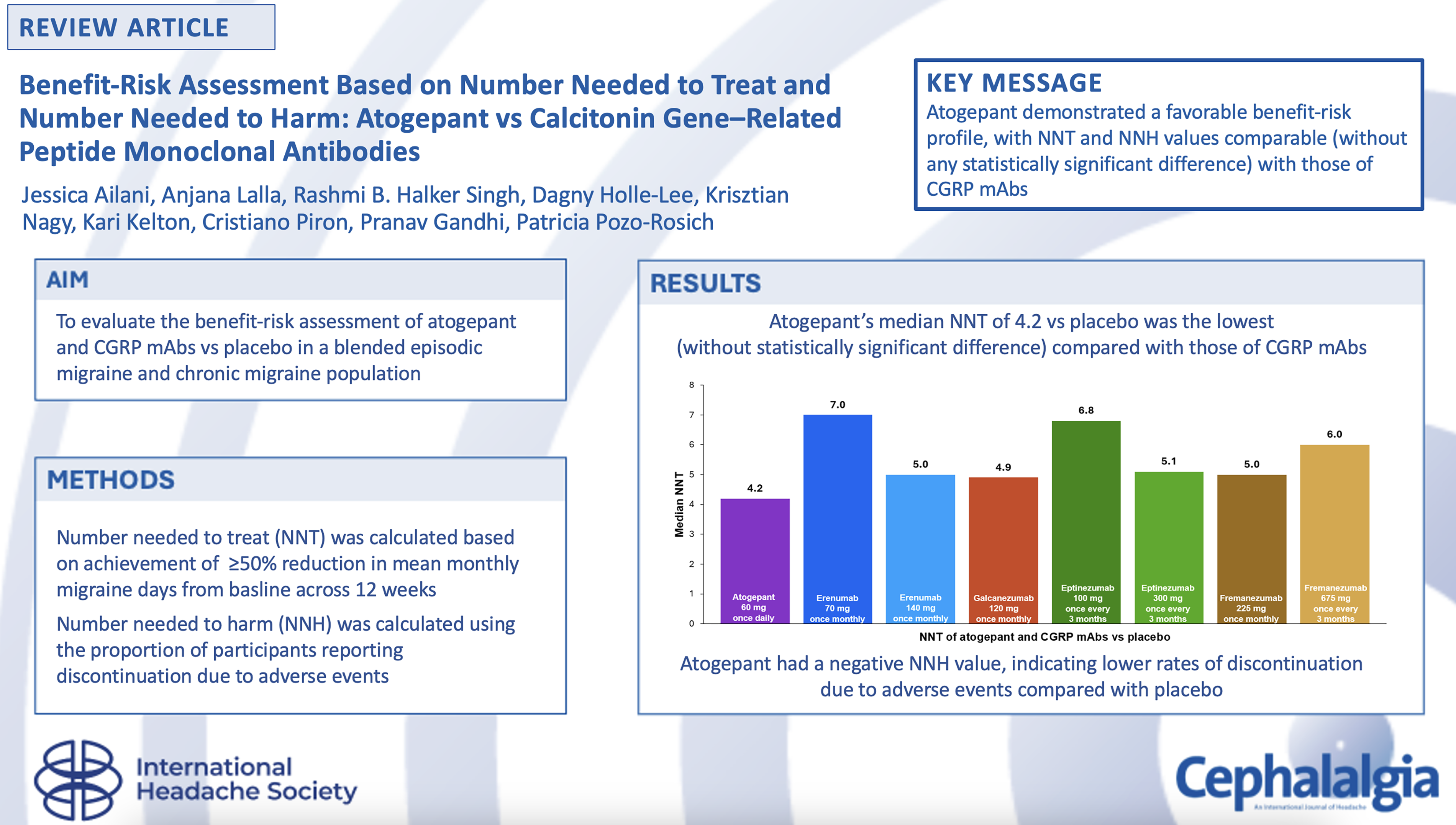
Introduction
Migraine is a debilitating neurologic disease that affects more than 1 billion people worldwide (1,2). A migraine attack is characterized by intense headache often accompanied by photophobia, phonophobia and nausea (1). People who have up to 14 monthly headache days (MHDs) are classified as having episodic migraine (EM) and those experiencing ≥15 MHDs, at least eight of which are migraine days, for >3 months are classified as having chronic migraine (CM) (1,3). Functional impairment because of migraine includes substantial negative impacts on professional and financial well-being, relationships, and overall health (4,5).
Circulating calcitonin gene–related peptide (CGRP) in the blood and activation of the neuronal CGRP receptor have long been implicated in migraine pathogenesis (6–10). Several different preventive therapies targeting the CGRP pathway are available to alleviate migraine. Atogepant is an oral, small-molecule CGRP receptor antagonist approved for the preventive treatment of episodic migraine (EM) and chronic migraine (CM) (11). There are also several monoclonal antibodies (mAbs) that target the CGRP pathway to reduce migraine frequency (12,13). Erenumab is an approved mAb that targets the CGRP receptor (14); galcanezumab (15), eptinezumab (16) and fremanezumab (17) are approved mAbs that target the CGRP ligand. Atogepant is an oral tablet taken daily, whereas the CGRP mAbs are administered via injection, either intravenously (eptinezumab) or subcutaneously (erenumab, fremanezumab, galcanezumab) monthly or quarterly, depending on the antibody and dosage (11,14–17).
The recently updated American Headache Society position statement recommends CGRP-targeted therapies, including gepants, as first-line options for the preventive treatment of migraine. The statement points to the efficacy, safety and tolerability of CGRP-targeted therapies demonstrated in clinical trials and real-world studies (18). However, there is limited evidence comparing the efficacy of these preventive migraine therapies (19–23). The disease burden of migraine on the population and the disability experienced by each individual with migraine generate a considerable need for healthcare professionals to make informed decisions when selecting a therapeutic regimen. This clinical choice is largely driven by the overall risk-benefit profile of treatments, which encompasses the appropriate efficacy, as well as safety and tolerability outcomes. Number needed to treat (NNT) and number needed to harm (NNH) are metrics that have been used to quantify the effect sizes of clinically relevant benefits and harms, respectively (24). However, randomized clinical trials simultaneously comparing these treatments are rare, and therefore, indirect comparisons can provide useful estimates for differences in treatments if each has been compared against a common comparator. The goal of this meta-analysis was to evaluate the benefit-risk profiles of atogepant and CGRP mAbs (erenumab, galcanezumab, eptinezumab and fremanezumab) vs. placebo in a blended episodic migraine and chronic migraine (EM + CM) population. The results from the various clinical trials allowed for the determination of NNT and NNH as an indirect treatment comparison to aid in clinical decision-making.
The NNT reflects the number of participants that would need to be treated with a medication for one additional participant to reach a favorable outcome such as a treatment response (24,25). In this meta-analysis, the desired outcome was at least a 50% reduction in monthly migraine days (MMDs) across 12 weeks. Although MMD reduction is a commonly reported endpoint in clinical trials, a 50% response rate is more informative in clinical practice to guide treatment decisions. Thus, a smaller NNT indicates greater efficacy and a more powerful medication (24). The NNH, on the other hand, represents the number of participants that would need to be treated with a medication for one additional patient to reach an unfavorable outcome (24,25). In this meta-analysis, the NNH outcome of interest was an adverse event (AE) leading to treatment discontinuation because this outcome allows for comparison of tolerability across different treatments. A larger NNH value indicates greater tolerability (24). These NNT and NNH values are clinically intuitive and relevant assessments of effect size that can inform real-world treatment management.
Methods
Study design
This study was a post hoc analysis of existing randomized clinical trial data and published, peer-reviewed literature. The analysis was performed on a blended EM + CM population. The study characteristics are outlined in the supplemental material (Table S1). Of the 12 key registrational studies included in the primary analysis, 11 were phase 3, one was phase 2, and all were double-blind, placebo-controlled, randomized clinical trials that lasted 12–48 weeks. Sensitivity analyses were also conducted to encompass a broader scope of data. Each of these sensitivity analyses were performed in conjunction with the primary analysis. The first sensitivity analysis included five dose-relationship studies, which were phase 2, 2b, 2b/3 or 3 double-blind, placebo-controlled, randomized clinical trials that spanned 12–49 weeks. The second sensitivity analysis incorporated seven Asian studies (conducted in China, Japan, India, Russia and other countries), which were phase 2 and 3 double-blind, placebo-controlled, randomized clinical trials that lasted 12–24 weeks. Detailed methodologies have been published previously (see supplemental material, Table S1).
Data sources and outcomes
A comprehensive literature search was performed on 1 September 2022 and relevant studies were identified. The studies meeting the following criteria were included in this analysis: randomized clinical trial design of migraine preventive treatment for EM or CM in a non-treatment failure-specific population; use of atogepant or a CGRP mAb licensed dose approved by the UK/European Medicines Agency or United States Food and Drug Administration; use of placebo as one of the comparators; and evaluation of the outcomes outlined below (19–23,26–44). An overview of the studies included in each analysis is presented in Table 1.
Episodic and chronic migraine preventive treatment clinical trials included in the NNT/NNH analysis.

CM = chronic migraine; EM = episodic migraine; NNH = number needed to harm; NNT = number needed to treat.
Outcomes
The efficacy outcome for NNT calculation was 50% response rate derived from MMDs; that is the percentage of participants who achieved at least a 50% reduction in their MMDs across 12 weeks from baseline. The NNT represents the number of participants over a given time that need to be treated to achieve one additional 50% clinical response vs. placebo.
The safety outcome for NNH calculation was discontinuation due to AEs. NNH represents the number of participants that would need to be treated before observing one additional safety outcome of interest relative to the placebo cohort. It should be noted that the data for discontinuation due to AE from the different studies were not based on the same time point; study duration is outlined in the supplemental material (Table S1). The approach to comparing these data is described below. For treatments with higher discontinuation due to AE rate than with placebo, NNH was positive, with higher NNH values being more favorable. Treatments with a lower incidence of discontinuation due to AEs relative to placebo resulted in negative NNH values, suggesting that additional discontinuation due to AEs are less likely to occur when participants are treated with treatment of interest relative to placebo. Negative values favor the treatment of interest relative to placebo.
Statistical analysis
This meta-analysis calculated the NNT and NNH of atogepant and CGRP mAbs vs. placebo. Bayesian network meta-analysis methodology was used to produce a single pooled estimate of the placebo rate used to calculate the NNTs or NNHs for atogepant and CGRP mAbs. This approach is consistent with the recommendations from the National Institute for Health and Care Excellence (NICE) Technical Support Document (45). The Bayesian method considers the relative precision of different studies using the exact binomial likelihood with different sizes of Ns. In other words, the relative weighting of each study is dependent on the number of participants in that study, not the classification of EM or CM. Using the binomial likelihood, studies with a larger sample size were weighted more because it is less likely there will be significant outliers in those studies. A random effects (RE) model was preferred over the fixed effect (FE) model to calculate the median 50% response rates, NNTs and 95% credible intervals (CrIs) vs. placebo across 12 weeks. Measure of heterogeneity to compare FE and RE models was performed based on residual deviance and deviance information criterion statistics (45,46). If there were truly significant differences in relative treatment effects in separate EM and CM subgroups, we would expect notable heterogeneity expressed in the RE models’ superior fit to the FE model. However, based on the heterogeneity measures, there was no significantly superior fit for RE models relative to FE models, suggesting that the treatment effects across EM and CM were comparable.
NNT and NNH were calculated as the inverse of the absolute risk reduction or ARR [1/absolute risk reduction]. For NNT, the ARR was the difference between the proportion of participants who achieved a 50% response rate with the active treatment and the proportion of participants who achieved a 50% response rate with placebo. For NNH, the ARR was the difference between the proportion of participants who discontinued due to AEs with the active comparator and the proportion of participants who discontinued due to AEs with placebo. Data for discontinuation due to AEs were reported at different time points and discontinuation can differ depending on which time point it is measured. To account for this, a cloglog link function model was used to estimate median discontinuation rates due to AEs and calculate NNH and 95% CrIs. The cloglog model takes time point of the data into consideration and assumes a constant event rate over time, with estimates reported as hazard ratios. When 95% CrIs for NNH included infinity, the difference was deemed not significant.
Results
Of the 12 studies comprising the primary analysis, seven enrolled participants diagnosed with EM and five enrolled participants with CM. The demographic and baseline clinical characteristics were similar across trials (see supplemental material, Table S2). In the EM trials, the mean MMDs at baseline was 7.4–9.2, the mean age was 39.8–42.9 years and 83.7%–88.8% of participants were female. In the CM trials, the mean MMDs at baseline was 16.0–19.6, the mean age was 39.7–42.9 years and 82.8%–88.2% of participants were female.
In the blended EM + CM primary analysis, all treatments had comparable calculated NNT values (Figure 1), with a median NNT of 4.2 (95% CrI = 3.1–6.7) for atogepant 60 mg once daily vs. placebo, which was numerically lower than the NNTs of all CGRP mAbs. This NNT value indicates that, if approximately four participants were to be treated with atogepant 60 mg once daily across 12 weeks, one extra would reach a 50% reduction in MMDs as a result of the treatment. Among the CGRP mAbs, erenumab 140 mg once monthly, galcanezumab 120 mg once monthly, eptinezumab 300 mg once every 3 months and fremanezumab 225 mg once monthly clustered with NNTs of 4.9–5.1. Across all the sensitivity analyses, fremanezumab 225 mg once monthly had a numerically slightly lower NNT than all other treatments (Table 2). Fremanezumab 225 mg had an NNT of 4.6 (95% CrI = 3.2–7.6) compared with 5.0 (95% CrI = 3.7–7.6) for atogepant 60 mg once daily upon inclusion of the dose-response studies. With inclusion of the Asian studies, fremanezumab 225 mg once monthly had an NNT of 3.9 (95% CrI = 2.8–6.1), followed closely by fremanezumab 675 mg once every 3 months with an NNT of 4.0 (95% CrI = 2.8–6.4) and atogepant 60 mg once daily with an NNT of 4.2 (95% CrI = 3.1–6.7). Across all analyses, atogepant 60 mg once daily NNTs ranged from 4.2–5.0. The NNT range was 4.9–7.8 for erenumab (both doses), 4.8–4.9 for galcanezumab 120 mg once monthly, 5.1–6.8 for eptinezumab (both doses) and 3.9–6.0 for fremanezumab (both doses). None of these results were statistically significant and the CrIs overlapped, indicating comparability between atogepant and the CGRPs. Overall, across all analyses, the NNT for atogepant 60 mg once daily vs. placebo was comparable with the NNTs for CGRP mAbs in the blended EM + CM population.
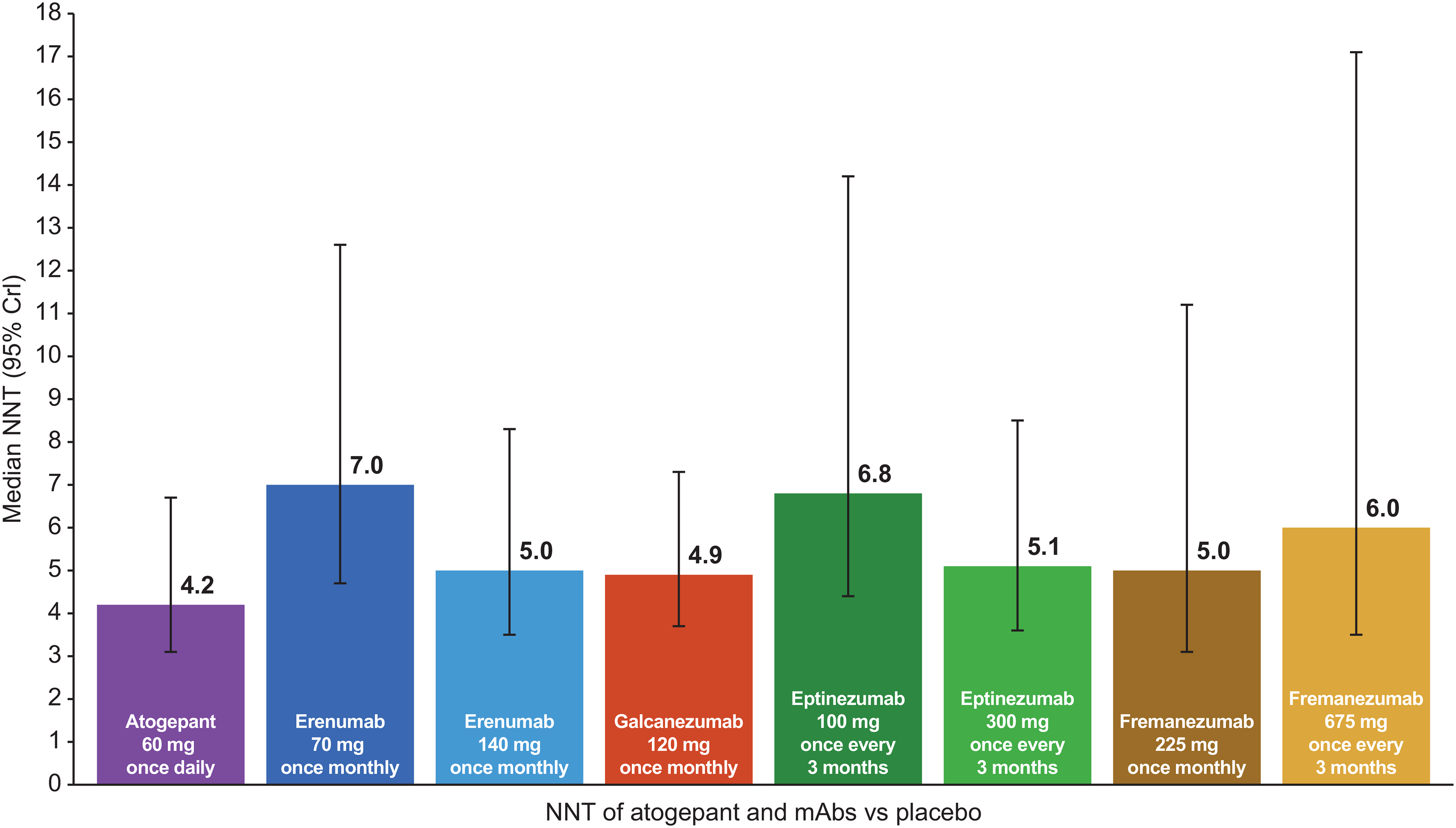
Median number needed to treat (NNT) for atogepant and calcitonin gene–related peptide monoclonal antibodies in the primary analysis.a Response rate outcome is defined as percent of participants who achieved at least a 50% reduction in their monthly migraine days across 12 weeks. aPrimary analysis includes ADVANCE (19), PROGRESS (26), EVOLVE 1 and 2 (20,29), REGAIN (30), ARISE (27), STRIVE (21), Tepper et al. (28), HALO EM (22) and PROMISE 1 and 2 (23,31). Crl = credible interval.
NNT for atogepant and CGRP mAbs (blended EM + CM a ) for primary and sensitivity analyses.
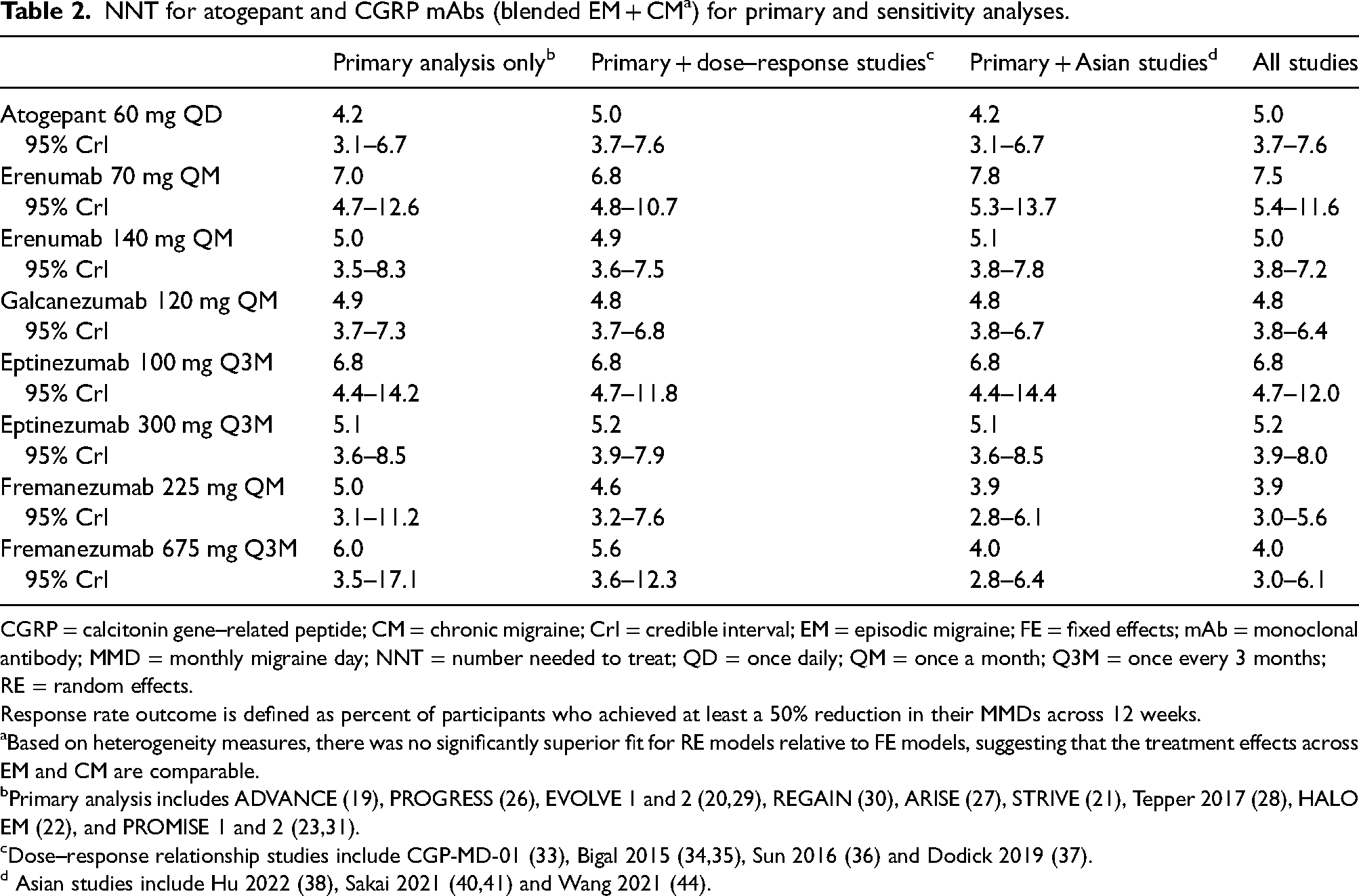
CGRP = calcitonin gene–related peptide; CM = chronic migraine; CrI = credible interval; EM = episodic migraine; FE = fixed effects; mAb = monoclonal antibody; MMD = monthly migraine day; NNT = number needed to treat; QD = once daily; QM = once a month; Q3M = once every 3 months; RE = random effects.
Response rate outcome is defined as percent of participants who achieved at least a 50% reduction in their MMDs across 12 weeks.
Based on heterogeneity measures, there was no significantly superior fit for RE models relative to FE models, suggesting that the treatment effects across EM and CM are comparable.
Primary analysis includes ADVANCE (19), PROGRESS (26), EVOLVE 1 and 2 (20,29), REGAIN (30), ARISE (27), STRIVE (21), Tepper 2017 (28), HALO EM (22), and PROMISE 1 and 2 (23,31).
The NNH for atogepant 60 mg once daily was −1010 (95% CrI = 44 to ∞ to number needed to benefit [NNB] 80) in the primary analysis (Figure 2), with a negative value indicating lower discontinuation due to AEs among participants who received atogepant compared with those who received placebo. An NNH value of −1010 indicates that for approximately every 1000 participants treated with placebo, one would discontinue due to a treatment-related adverse event. The proportion of participants that discontinued treatment with atogepant in these pooled populations was even lower. Similar findings were observed among participants who received fremanezumab, with lower discontinuation due to AEs compared with those who received placebo, resulting in negative NNH values (fremanezumab 225 mg once monthly: −1124 (95% CrI = 40 to ∞ to NNB 78); fremanezumab 675 mg once every 3 months: −346 (95% CrI = 52 to ∞ to NNB 65)). For the majority of treatments across all NNH analyses, the 95% CrIs indicated nonsignificant values as they included infinity. Atogepant 60 mg once daily and eptinezumab 100 mg once every 3 months had the only negative NNH values upon inclusion of the dose-response studies (both −6667 (95% CrI = 58 to ∞ to NNB 107 for atogepant and 33 to ∞ to NNB 80 for eptinezumab 100 mg) (Table 3). Atogepant 60 mg once daily also had the lowest NNH upon inclusion of the Asian studies (−1261 (95% CrI = 39 to ∞ to NNB 78)) followed by erenumab 140 mg once monthly (−593 (95% CrI = 47 to ∞ to NNB 65)).
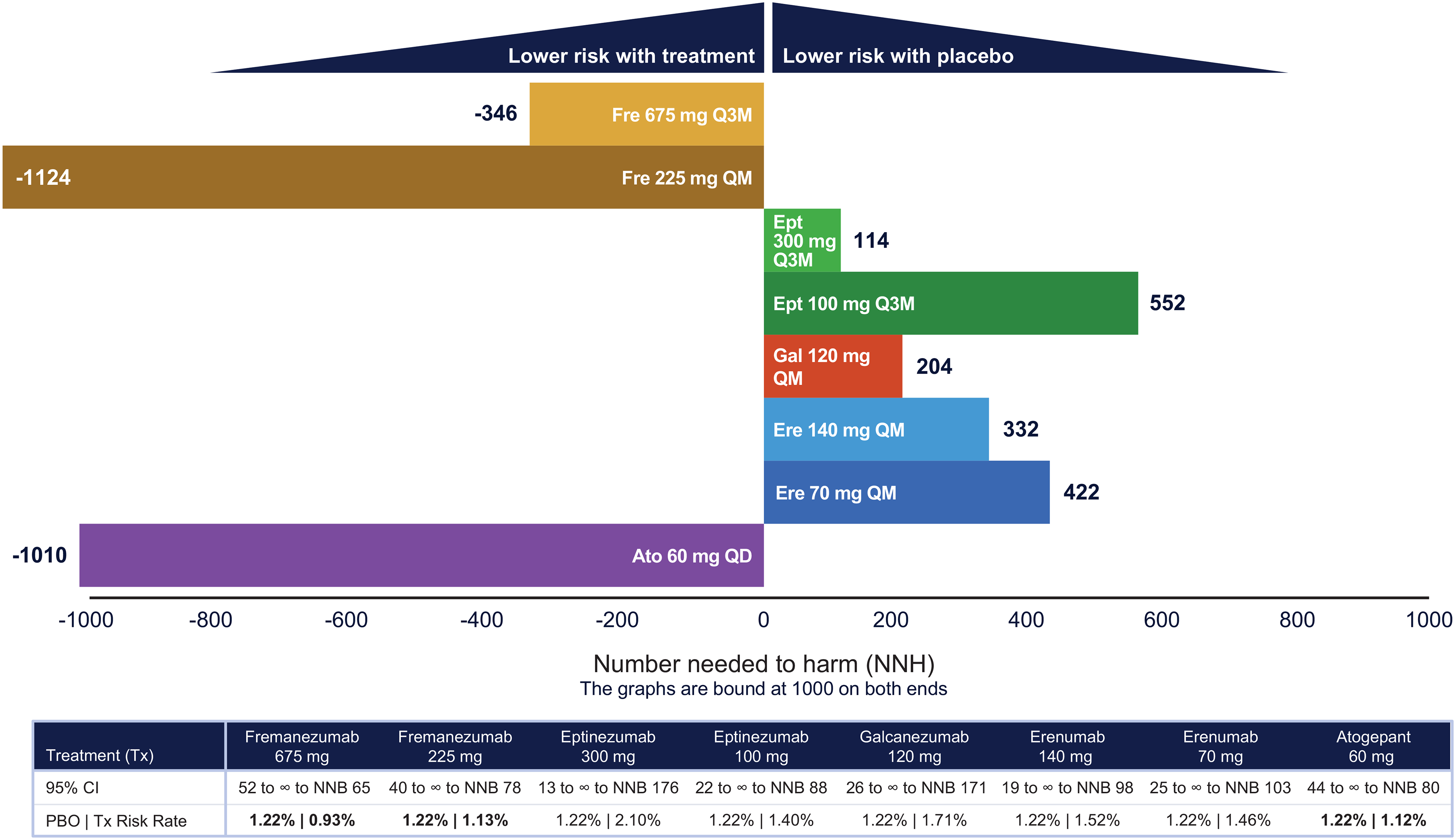
Number needed to harm (NNH) for atogepant and calcitonin gene–related peptide monoclonal antibodies in the primary analysis.a Bold values indicate that rates are higher for PBO relative to active treatment. aPrimary analysis includes ADVANCE (19), PROGRESS (26), EVOLVE 1 and 2 (20,29), REGAIN (30), ARISE (27), STRIVE (21), Tepper et al. (28), HALO EM (22), PROMISE 1 and 2 (23,31). b95% confidence interval (CI) indicates nonsignificant values because it includes infinity. Ato = atogepant; Ept = eptinezumab; Ere = erenumab; Fre = fremanezumab; Gal = galcanezumab; NNB = number needed to benefit; PBO = placebo; QD = once daily; QM = once a month; Q3M = once every 3 months; Tx = treatment.
NNH for atogepant and CGRP mAbs (blended EM + CM a ) for primary and sensitivity analyses.
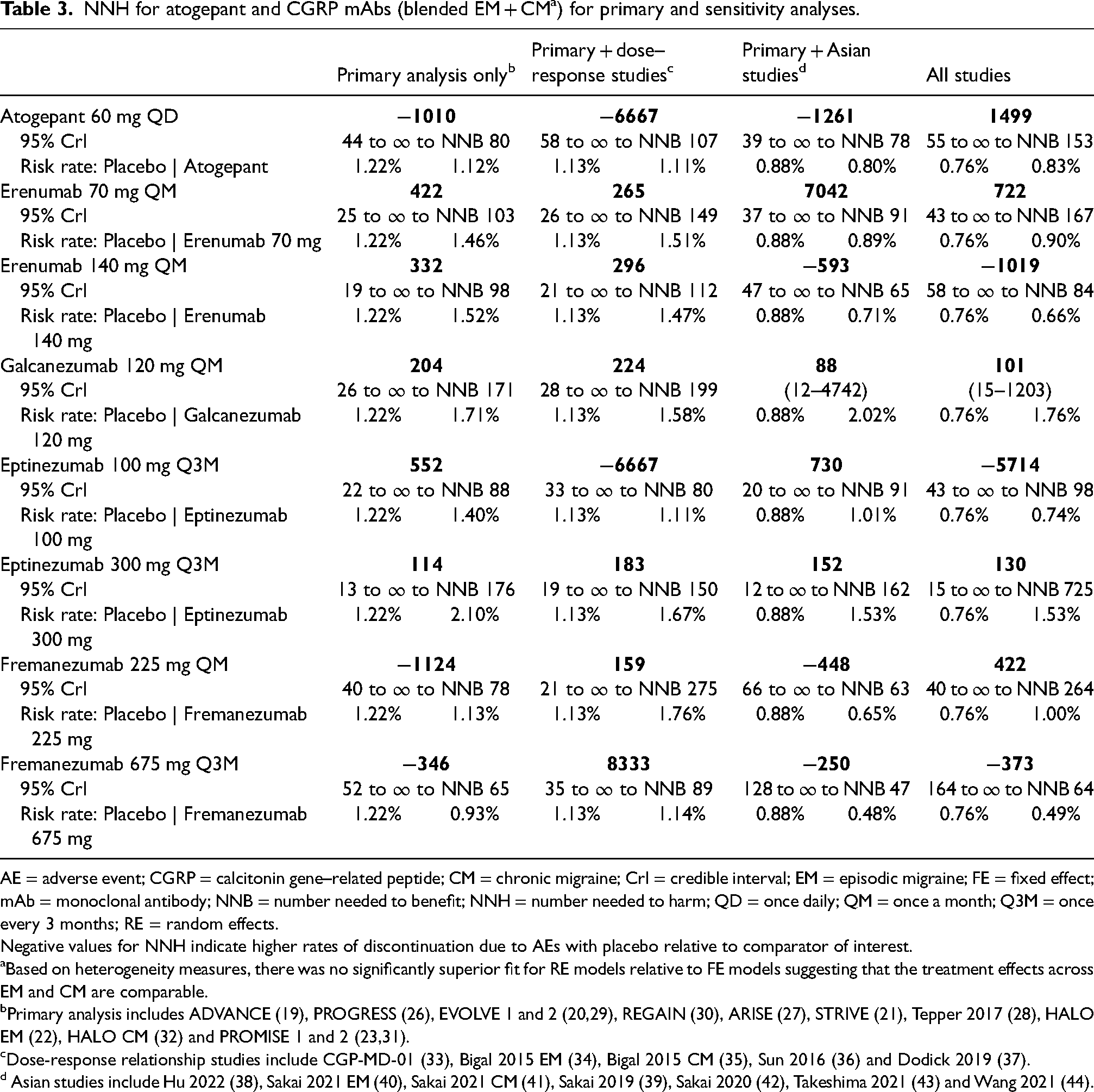
AE = adverse event; CGRP = calcitonin gene–related peptide; CM = chronic migraine; CrI = credible interval; EM = episodic migraine; FE = fixed effect; mAb = monoclonal antibody; NNB = number needed to benefit; NNH = number needed to harm; QD = once daily; QM = once a month; Q3M = once every 3 months; RE = random effects.
Negative values for NNH indicate higher rates of discontinuation due to AEs with placebo relative to comparator of interest.
Based on heterogeneity measures, there was no significantly superior fit for RE models relative to FE models suggesting that the treatment effects across EM and CM are comparable.
Primary analysis includes ADVANCE (19), PROGRESS (26), EVOLVE 1 and 2 (20,29), REGAIN (30), ARISE (27), STRIVE (21), Tepper 2017 (28), HALO EM (22), HALO CM (32) and PROMISE 1 and 2 (23,31).
Discussion
This study provides an indirect comparison of the efficacy (NNT) and safety (NNH) of atogepant and CGRP mAbs vs. placebo in the preventive treatment of migraine for a blended EM + CM population. Atogepant 60 mg once daily demonstrated the lowest NNT to achieve a 50% reduction in MMDs compared with CGRP mAbs in the primary analysis. All of the treatments had single-digit NNT values, which indicates effective treatments with at least one in 10 participants experiencing clinically relevant improvement over placebo (24). Notably, in the primary analysis, the negative NNH value of atogepant relative to placebo means that participants receiving atogepant 60 mg once daily had even lower discontinuation rates due to AEs than participants receiving placebo. Fremanezumab 225 mg once monthly and 675 mg once every 3 months also had negative NNH values; however, none of the results were statistically significant. All treatments (atogepant and mAbs) displayed a similar efficacy and higher likelihood to help (low NNT numbers) than harm (higher or negative NNH values).
Previous studies have derived NNT and NNH values for the CGRP mAbs with the 50% response rate and discontinuation due to AEs, respectively. One such study compared CGRP mAbs, topiramate and propranolol, for the prevention of EM and CGRP mAbs, topiramate and onabotulinumtoxinA, for the prevention of CM (47). Overall, the results of their NNT analyses are consistent with the CGRP mAb results of our NNT analysis; however, the results of the NNH analysis were mixed. Another meta-analysis examining topiramate and CGRP mAbs for the preventive treatment of EM found similar NNT efficacies across all treatments. The NNH safety profiles, on the other hand, favored CGRP mAbs over traditional migraine treatments such as topiramate, which had lower NNH values (range 5–50) illuminating higher discontinuation due to AEs (48). A summary of the clinical trial populations included along with each result for our analyses and previous meta-analyses can be found in the supplemental material (Table S3). Differences between previous meta-analyses and the one presented here may be due to the analysis of different studies (e.g. our inclusion of Tepper et al. (28) on erenumab for CM) or differences in treatment center practices or placebo rates. Most notably, the previous analyses focused on separate EM and CM populations, with each treatment NNT/NNH calculated from a single trial, whereas our analyses pooled data from multiple clinical trials into a blended EM + CM population, eliminating biases from individual trials.
The blended EM + CM population reflects the reality of migraine disease, in which MMDs and thus EM/CM definition fluctuates over time (49). A potential limitation of our analyses is that the clinical trials included had different protocols with varying definitions of migraine and primary outcomes and studied different oral versus injectable treatments. Additionally, it is possible that the blended EM + CM analysis introduces differences among study participants. However, based on heterogeneity measures, there was no significantly superior fit for RE models relative to FE models, suggesting that the treatment effects across EM and CM were comparable. To investigate potential differences in treatment efficacy across EM and CM populations, we also performed meta-regressions on baseline MMDs across the included outcomes because EM and CM populations are distinguished by differences in baseline MMDs and MMD definitions can be different in different trials. The meta-regression models confirmed that there are no differences in treatment efficacy because the model did not demonstrate significantly superior fit. Thus, evaluation of the overall migraine population allowed for a comprehensive summary of each of the available therapies.
There are also several limitations inherent to a study of this nature. Predominantly, NNT and NNH calculations are indirect metrics for comparison across distinct clinical trials, each with their own criteria. In terms of safety, injection-site reactions that may occur with injectable treatments are not relevant for oral treatments. Additionally, treating physicians in the later studies benefited from adverse event findings in the earlier studies and were more aware and better prepared to address them, which may have led to a reduced discontinuation rate in later studies. Extrapolation from clinical trial participants to the general population may also be limited. For all analyses, the treatment period was limited to 12 weeks, preventing the evaluation of long-term efficacy and safety of atogepant and CGRP mAbs vs. placebo. As the data available from different studies for discontinuation were not all based on the same time point and it is generally thought that time period affects this outcome, it was necessary to account for this difference in these analyses. A model using the cloglog link function was implemented for the discontinuation endpoint. The cloglog model considers the time point of the data and assumes a constant event rate over time for the treatments; the relative effects estimated from this analysis are given as a hazard ratio. The constant event rate is a strong assumption for the cloglog model. In addition, the cloglog model assumes that all patients will experience the event with sufficient follow-up.
The true contribution of this study lies in its application to clinical decision-making. Atogepant is an oral medication option and can be a preferred route for some individuals with migraine, whereas CGRP mAbs are administered as either a subcutaneous or intravenous therapy (50,51). Understanding how treatments compare from an NNT and NNH perspective and being able to explain this to patients is helpful at the point of care (52). A 50% reduction in MMDs is widely regarded as a clinically significant benchmark for response to a preventive migraine treatment (53). Discontinuation due to AEs is especially relevant, as it reflects the patient experience underlying adherence and persistence rates, which are known to be the major obstacles in treating migraine (54–56). The markedly favorable NNH value of atogepant indicates the potential for greater treatment adherence. In the absence of direct clinical comparison of preventive migraine treatments in EM and CM, these NNT and NNH derivations provide risk-benefit profiles for clinicians to interpret and apply.
Conclusions
The NNT for atogepant 60 mg once daily vs. placebo was comparable with CGRP mAbs in the blended EM + CM population. Atogepant demonstrated a numerically lower NNT compared with CGRP mAbs in the primary analysis. Participants who received atogepant 60 mg once daily demonstrated lower rates of discontinuation due to AEs compared with those receiving placebo in most of the analyses in this study, resulting in negative NNH values. These strong (but not statistically significant) data demonstrate that atogepant is efficacious and well-tolerated, in line with other CGRP-targeted medications and the American Headache Society's recommendation of these treatments as first-line options for the preventive treatment of migraine.
The NNT for atogepant 60 mg once daily vs. placebo was comparable with CGRP mAbs. Atogepant demonstrated a numerically lower NNT compared with CGRP mAbs in the primary analysis. Atogepant demonstrated a favorable benefit-risk profile, supporting the American Headache Society's recommendation of using CGRP-targeted therapies, including gepants, as first-line options for the preventive treatment of migraine
Supplemental Material
sj-docx-1-cep-10.1177_03331024241299377 - Supplemental material for Benefit-risk assessment based on number needed to treat and number needed to harm: Atogepant vs. calcitonin gene–related peptide monoclonal antibodies
Supplemental material, sj-docx-1-cep-10.1177_03331024241299377 for Benefit-risk assessment based on number needed to treat and number needed to harm: Atogepant vs. calcitonin gene–related peptide monoclonal antibodies by Jessica Ailani, Anjana Lalla, Rashmi B Halker Singh, Dagny Holle-Lee, Krisztian Nagy, Kari Kelton, Cristiano Piron, Pranav Gandhi and Patricia Pozo-Rosich in Cephalalgia
Footnotes
Acknowledgments
AbbVie funded this study and participated in the study design; the collection, analysis and interpretation of data; and the review and approval of the final manuscript for publication. All authors had access to relevant data and participated in the drafting, review and approval of this publication. No honoraria or payments were made for authorship. Medical writing support was provided to the authors by Sara Nathan, PhD, of Peloton Advantage, LLC, an OPEN Health company, and was funded by AbbVie. Support for the study design and implementation was provided by James Gahn and David Rowe of Medical Decision Modeling Inc., and was funded by AbbVie.
Author contributions
AL, KN, KK, CP and PG were responsible for the study design. KK and CP were responsible for collection and assembly of data. AL, KK, CP and PG were responsible for data analysis. All authors were responsible for data interpretation. All authors were responsible for manuscript preparation. All authors were responsible for manuscript review and revisions. All authors approved the final version of the manuscript submitted for publication.
Data availability
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual and trial-level data (analysis data sets), as well as other information (eg, protocols, clinical study reports or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications. These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, Statistical Analysis Plan (SAP), and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the USA and Europe and after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, see: ![]() .
.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Jessica Ailani, MD, has served as a consultant for AbbVie, Aeon, Amgen, Axsome, BioDelivery Sciences International, Biohaven, Eli Lilly and Company, GlaxoSmithKline, Gore, Impel, Ipsen, Linpharma, Lundbeck, Merz, Miravo, Nesos, Neurolief, Pfizer, Satsuma, Teva, and Theranica. She has owned stock shares in Ctrl M, has provided editorial services for SELF magazine, and has received clinical trial grants from AbbVie, Biohaven, Eli Lilly and Company, Ipsen, Parema, Satsuma and Zosano. Anjana Lalla, MS, Pranav Gandhi, PhD, and Krisztian Nagy, MD, are employees of AbbVie and may hold AbbVie stock. Rashmi B. Halker Singh, MD, serves on the editorial board of Current Neurology and Neuroscience Reports, serves as deputy editor for Headache, and previously received grants for research support from Amgen and Eli Lilly. In the past 3 years, she has received consulting fees from AbbVie and Pfizer. Dagny Holle-Lee, MD, PhD, has received honoraria for consulting from AbbVie/Allergan, Amgen, Eli Lilly, Hormosan, Lundbeck, Novartis, Teva and Zuellig Pharma. Patricia Pozo-Rosich, MD, PhD, has received, in the past 3 years, personal fees for advisory boards and speaker panels from AbbVie, Amgen, Chiesi, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer and Teva Pharmaceuticals, and for serving on a scientific advisory or data safety monitoring board for Lilly Foundation Spain; is the principal investigator for clinical trials sponsored by AbbVie, Eli Lilly, Lundbeck, Novartis and Teva; has received (through her group) grants from AbbVie, EraNet Neuron, Instituto de Salud Carlos III, Novartis and Teva; and serves as an associate editor for Cephalalgia, Headache, Neurologia, The Journal of Headache and Pain, and Revista de Neurologia. Kari Kelton, PhD, and Cristiano Piron, MPH, are employees of Medical Decision Modeling Inc., which received payment from AbbVie for the analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This study was sponsored and funded by AbbVie. Medical writing support was provided to the authors by Sara Nathan, PhD, of Peloton Advantage, LLC, an OPEN Health company, and was funded by AbbVie. Support for the study design and implementation was provided by James Gahn and David Rowe of Medical Decision Modeling Inc., and was funded by AbbVie.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
