Abstract
Background:
Trigeminal neuralgia can be classical or idiopathic. While trigeminal neuralgia (TN) due to space-occupying lesions is atypical, such lesions rarely cause severe TN secondary to trigeminal nerve irritation. Mass effect from these lesions has been shown to correlate with symptom burden, due to direct or indirect compressive effects. A tethering effect, provoked by an abnormal root-stretching force, theoretically plays a role in trigeminal nerve hyperexcitability.
Case:
The likely etiology in this case presentation is a large glomus tumor invading the middle and posterior cranial fossa. Glomus tumors are uncommon benign tumors of the head and neck derived from neural crest cells. Even more strikingly, a large glomus tumor causes bilateral TN due to direct compression on one side and indirect compression on the contralateral side.
Conclusion:
Although the gold standard in TN management is carbamazepine, other anti-epileptic drugs (AEDs) have been used in the treatment of patients unable to take carbamazepine. A few studies suggest levetiracetam alleviates central and neuropathic pain, supporting the hypothesis that it may be effective in management of TN.
Background
While TN due to space-occupying lesions is relatively rare, such lesions can cause severe TN secondary to trigeminal nerve irritation. 1 –4 Although the gold standard in TN management is carbamazepine, other AEDs have been used in the treatment of patients unable to take carbamazepine. 5,6 Levetiracetam has been studied as an alternative treatment of trigeminal neuralgia. 7
Case presentation
The patient is a 34-year-old female with bilateral TN, right-sided pain more severe than on the left, in the setting of a large right-sided jugular glomus tumor pressing on her brainstem (Figures 1 and 2). Her pain was triggered by touching of the face, brushing her teeth, talking, and chewing, corroborated by her cranial nerve exam. She also reported pain with swallowing more on the right side, which indicated a possible component of glossopharyngeal neuralgia. At baseline her pain was managed with levetiracetam 750 mg BID and carbamazepine 200 mg. Her recent history was significant for an admission 4 months prior with similar symptoms. At that time, she was still able to tolerate oral medications.
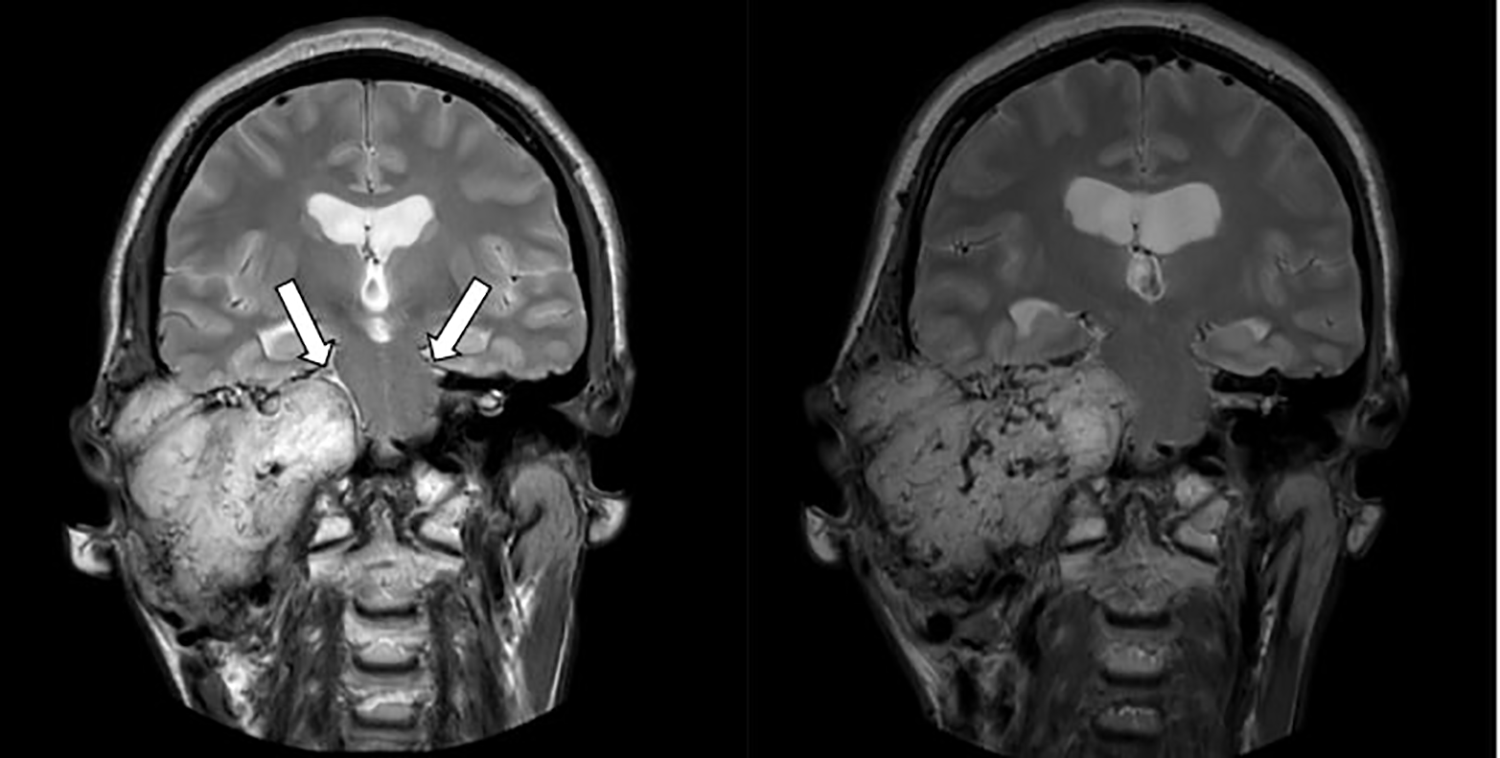
Coronal T2-weighted images demonstrate a large mass in the central skull base on the right, with predominantly high signal intensity and large flow voids. The mass replaces all the structures of the right middle and inner ear, extending to the petrous apex. It causes superior and medial displacement of the cisternal segment of the right trigeminal nerve (arrow), along with effacement of the CSF around the left trigeminal nerve (arrow).

Coronal and axial post-contrast T1-weighted images show the intense heterogeneous enhancement of the mass in the central skull base on the right, which has mass effect on the pons and right middle cerebellar peduncle. Areas of necrosis are present within the mass.
Upon this admission, she was started on intravenous fluids as she was not able to tolerate any PO intake. Acute pain management service (APMS) was consulted. She was initially placed on ketamine (around 0.5 mg/kg) and lidocaine (around 1 mg/kg) infusions. These were discontinued after 24 hours due to hallucinations. At that point, she was still unable to tolerate PO medications. Within 2 days, hydromorphone PCA (started from 0.1 mg/hr) and ketorolac 15 mg IV TID PRN were discontinued. She was given a single dose of phenytoin 250 mg IV and methylprednisolone 1 g IV. The medications were ceased for lack of benefit and intolerable side effects. Methocarbamol 1 g IV q4 hours was continued.
On the third day of admission, IV levetiracetam was started, initially at 1000 mg BID then increased to 2000 mg BID after she reported improvement. Her pain significantly improved on levetiracetam 2000 mg IV BID. Her glossopharyngeal neuralgia also improved, helping enable her to orally take gabapentin solution and subsequently carbamazepine tablets. Her pain was well-controlled with this regimen and she was discharged on carbamazepine, levetiracetam, and gabapentin. She was, however, readmitted for the same pain within one month. Fortunately, she was able to tolerate PO medications, so her pain was easily controlled on increased carbamazepine and gabapentin with levetiracetam discontinued.
Conclusions
Trigeminal neuralgia may be classical or idiopathic, with a frequently cited culprit being superior cerebellar artery anomalies causing irritation of the trigeminal ganglion. 7,8 Causes of TN include demyelination, compression by solid tumors or vascular lesions, or a combination of a focal lesion and its treatment. 3 These lesions include trigeminal neuromas in the middle or posterior cranial fossa, vestibular schwannomas or acoustic neuromas, tentorial dural arteriovenous fistulas, unruptured posterior communicating aneurysms, intracranial arteriovenous malformations, squamous cell carcinoma of the head and neck, epidermoid tumors, and cholesteatomas. 8 Tumor size may be correlated with symptom burden, due to direct or indirect compressive effects. 9 The symptomatic lesions are commonly found in the cerebellopontine angle. 10
Vestibular schwannomas and meningiomas account for approximately 85–90% of all cerebellopontine angle tumors. 11 A tethering effect, theorized to be provoked by an abnormal root-stretching force, has been reported to play a role in trigeminal nerve hyperexcitability. 12 Our patient’s TN occurred in the setting of a large glomus tumor invading the middle and posterior cranial fossa. Glomus tumors are rare benign tumors of the head and neck derived from neural crest cells. They arise from extra-adrenal autonomic ganglia and can secrete catecholamines.
The most common cranial nerves affected by head and neck tumors are facial, glossopharyngeal, vagus, accessory, and hypoglossal. 13 Encroachment on the trigeminal nerve, with involvement of the glossopharyngeal nerve, is the likely cause of our patient’s inability to tolerate carbamazepine tablets as she complained of difficulty with both chewing and swallowing.
Bilateral TN is an unusual finding and the etiology is often unclear. However, a study by Harris indicated an association between bilateral TN and MS. 14 We theorize that our patient’s bilateral TN is a function of ipsilateral compression of the right trigeminal nerve with contralateral compression of the left trigeminal nerve from mass effect of the large glomus tumor, which would be confirmed by resection (Figures 1 and 2). A similar mechanism of contralateral TN has been described in case reports by Snow, Eftekhar, and Chamadoira. 15
Various treatment options, such as radiofrequency ablation, microvascular decompression, and surgical clipping, exist for the management of TN due to space-occupying lesions. However, procedural treatments themselves can produce neuropathic pain in the trigeminal distribution due to their effects on the trigeminal ganglion, nerve roots, or branches. 16 Oral treatments such as carbamazepine, oxcarbazepine, baclofen, lamotrigine, levetiracetam, topiramate, gabapentin, pregabalin, clonazepam, amitriptyline, nortriptyline, duloxetine, venlafaxine, or even non-invasive procedures including trigeminal nerve block and botulinum toxin administration, may improve symptomatic prognosis sans need for invasive treatments. 4
Levetiracetam, along with other relatively new AEDs, has been studied for TN management with some promising results in open label clinical studies and animal models. 17,18 A few studies suggest levetiracetam may be used in management of central and neuropathic pain, supporting the hypothesis that this medication may be effective in management of TN. 19,20 Levetiracetam was also reported to be successfully used in cases of facial pain and seizure disorder. 21,22
Our case demonstrates a clinical challenge in the management of TN, when a patient is unable to tolerate oral carbamazepine tablets during an exacerbation. Multiple pain management modalities were applied, including non-steroidal anti-inflammatory drugs (NSAIDs), opioids, steroids, ketamine infusion, lidocaine, and phenytoin, without significant improvement. We observed a gradual improvement in pain with increasing doses of levetiracetam and thus kept this as the mainstay of treatment during inpatient pain management. In a prospective open label pilot study, Jorns et al. reported 4 out of 10 patients have less pain compared to baseline with 4000 mg/day dosing of levetiracetam. 6 These findings corroborate with our clinical observation since our patient improved significantly when the 4000 mg/day dose of levetiracetam was reached.
Overall, large space-occupying lesions, such as a glomus tumor in our case, can cause refractory bilateral TN. Levetiracetam may improve trigeminal neuralgia when first-line treatments are ineffective or intolerable. Availability of intravenous formulations and improved tolerability (with side effects including drowsiness, dizziness, and decreased appetite) are advantages of levetiracetam during exacerbations. Further studies are needed to establish the role of levetiracetam in trigeminal neuralgia treatment.
Footnotes
Acknowledgments
We would like to acknowledge Mark J. Burish, MD for his clinical expertise during the writing of this manuscript.
Clinical implications
Large space-occupying lesions, such as a glomus tumor in our case, can cause refractory bilateral TN. Levetiracetam may improve trigeminal neuralgia when first-line treatments are ineffective or intolerable. Availability of intravenous formulations and improved tolerability are advantages of levetiracetam during exacerbations.
Author contributions
Billie Hsieh wrote and edited the manuscript and created the figure, Barlas Benkli co-wrote and edited the manuscript, George N. Ansoanuur co-wrote and edited the manuscript, Eliana E. Bonfante-Mejia helped create the figure, Shaun O. Smart edited the manuscript.
Consent for publication
Written informed consent was obtained from the patients for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
