Abstract
We here present evidence-based guidelines for the pharmacological treatment of migraine. These guidelines, created by the Italian Society for the Study of Headache and the International Headache Society, aim to offer clear, actionable recommendations to healthcare professionals. They incorporate evidence-based recommendations from randomized controlled trials and expert-based opinions. The guidelines follow the GRADE approach for assessing the quality of evidence. The guideline development involved a systematic review of literature across multiple databases, adherence to Cochrane review methods, and a structured framework for data extraction and interpretation. Although the guidelines provide a robust foundation for migraine treatment, they also highlight gaps in current research, such as the paucity of head-to-head drug comparisons and the need for long-term outcome studies. These guidelines serve as a resource to standardize migraine treatment and promote high-quality care across different healthcare settings.
The full document of the SISC-IHS guideline is available here: https://doi.org/10.1177/03331024241305381
Introduction
Evidence-based guidelines are pivotal tools to guide treatment. These guidelines distill vast amounts of research into practical recommendations, ensuring that healthcare professionals can deliver the most effective treatments to patients. By systematically reviewing the latest evidence, guidelines help bridge the gap between research findings and clinical practice, promoting consistency and quality in healthcare delivery. The strength of evidence-based guidelines lies in their ability to synthesize diverse sources of evidence into actionable recommendations. By prioritizing interventions with demonstrated efficacy, guidelines empower clinicians to make informed decisions that optimize patient outcomes.
In the migraine field, numerous guidelines exist. Notably, guidelines from the European Headache Federation (1,2) have been meticulously developed utilizing a robust methodology, such as the GRADE approach (3). This methodology includes systematic evaluation of available evidence, quantitative pooled statistical synthesis of data, and rigorous rating of evidence quality (3). However, it is noteworthy that these guidelines primarily focus on a subset of available treatments, notably monoclonal antibodies targeting the calcitonin gene-related peptide (CGRP) pathway and onabotulinumtoxinA (1,2).
Conversely, guidelines from the American Headache Society provide comprehensive coverage of all available pharmacological treatments for migraine (4–6). However, they are not grounded in quantitative analyses of the available data.
In pursuit of promoting optimal care for individuals suffering from migraine, the Italian Society for the Study of Headache (SISC) and the International Headache Society (IHS) have embarked on a collaborative endeavor. This initiative aims to develop a joint evidence-based guideline on the pharmacological treatment of migraine.
The overarching goal of this joint guideline is to furnish healthcare practitioners with evidence-based recommendations for the pharmacological management of migraine to provide clear, actionable guidance for the treatment of migraine. The presentation of this guideline is organized as a summary document to facilitate consultation, the main document with all the information to derive evidence-based recommendations, and a supplementary file with additional information. For each of the considered drug categories, the main document contains quantitative summary of the evidence, the evaluation of the quality of evidence, a summary on safety and tolerability, a summary of the evidence-based guideline, and expert-based opinions to support clinical use.
Methods
Details of the Methods are available in section 2 of the main document available at: https://doi.org/10.1177/03331024241305381.
The guideline working group was appointed by the ‘Italian Society for the Study of Headaches’ ('Società Italiana per lo Studio delle Cefalee’ – SISC) and by the ‘International Headache Society’ (IHS) and is described in Table 1.
Guideline working group.

The working group included a chair, a coordination supporting group, external reviewers, administrative supporting persons, and module working subgroups, each assigned to a specific drug class (Table 1). Each subgroup had both SISC and IHS components.
This guideline is based on the best available evidence from randomized controlled trials (RCTs) and a rigorous evaluation of the quality of evidence for each intervention and outcome. The considered interventions were drugs commonly used for the acute treatment of migraine attacks or for migraine prevention.
For the acute treatment of migraine attacks, the following drug classes were considered:
− Non-steroidal anti-inflammatory drugs (NSAIDs) and cicloxygenase 2 (COX2) inhibitors (Section 3.1) − Triptans (Chapter 3.2) − Paracetamol/acetaminophen (Chapter 3.3) − Combination analgesics (Chapter 3.4) − Antiemetics (Chapter 3.5) − Opioids (Chapter 3.6) − Ditans (Chapter 3.7) − Gepants for acute treatment (Chapter 3.8) − Antidepressants (Chapter 4.1) − Anti-seizure medications (Chapter 4.2) − Beta-blockers (Chapter 4.3) − Calcium channel blockers and blood-pressure lowering agents (Chapter 4.4) − Botulinum toxin (Chapter 4.5) − Gepants for acute treatment (Chapter 4.6) − Monoclonal antibodies targeting the CGRP pathway (Chapter 4.7)
For migraine prevention, the following classes were considered:
Those chapters addressed the available evidence of treatments compared with placebo. Head-to-head comparisons between different active drugs were reported in Chapter 3.9 for the acute treatment of migraine attacks and in Chapter 4.8 for migraine prevention.
Clinically relevant questions were framed using the PICO format (Population, Intervention, Comparator, and Outcomes) by members of the guideline panel. To provide clinical guidance, we also used expert-based opinions. The text clearly distinguishes between evidence-based recommendations and expert-based opinions to inform the reader where guidance is based solely on evidence data or where it considers experience and opinions.
For the acute treatment of migraine attacks, the following outcomes were considered:
− pain freedom at 2 h from intake; − pain relief at 2 h from intake. − persisting monthly headache/migraine days, defined as the residual days reported by patients at the end of the treatment (as reported in headache diaries); − change in monthly headache/migraine days, defined as the variation in days reported by patients from baseline to the end of follow-up (as reported in headache diaries); − ≥50% responder rate, defined as the proportions of patients reporting a ≥ 50% reduction in monthly headache/migraine days compared with baseline. The ≥50% reduction of monthly attacks was also considered for ≥50% responder rate whenever the reduction in monthly headache/migraine days was not available.
For preventive treatment, the following outcomes were considered:
The chosen outcomes were more extensive than those issued by the International Headache Society guidelines for RCTs of migraine prevention (7,8), in order to include the highest possible number of RCTs. Patient-reported outcomes were not included because of substantial heterogeneity across instruments used. Given the expected minimal impact of serious adverse events, we did not consider safety as an important or critical outcome to derive evidence-based recommendations. We addressed tolerability and used this information to draft expert-based opinion sections.
The final guideline report includes patient populations, interventions, comparators, and outcomes for which the systematic literature search showed the presence of available RCTs.
Search of available evidence was performed according to the Cochrane guidelines for systematic reviews of interventions (9) and overviews of reviews (10). Cochrane guidelines were also followed for study selection, data extraction and synthesis. Reporting was performed according to relevant items of the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement (11).
Literature search was performed between 10 and 11 February 2022 for all topics except for antiemetics, which were included in the search plan in September 2022; related search strings were launched on 8 September 2022. As the process of literature search and analysis took more than 12 months, search strings were re-launched in May 2023 and November 2023 to update the search to the RCTs published from February 2022. Two researchers (VC and RO) performed a literature search for each pharmacological class of acute treatments and migraine preventive drugs. Three scientific databases were searched, namely PubMed, Scopus, and Cochrane Database, since the beginning of indexing, utilizing the PICOM (Patients – Intervention – Comparison – Outcome – Methods) methodology. To ensure a broad coverage of available literature, when building search strings, only Participants (i.e., migraine patients) and Interventions (i.e., drugs) were considered for each topic.
Study selection was performed by each module subgroup.
The inclusion and exclusion criteria for eligibility and inclusion phase are presented in Table 2.
Inclusion and exclusion criteria for evaluation of references in eligibility and inclusion phases for stage 2.

Utilizing an .xlsx spreadsheet template, module subgroups extracted data for each included study.
For each PICO question, all extracted data about outcomes were classified, analyzed, and presented as pooled analyses of the available RCTs for each selected outcome. Results were presented as pooled analyses even when only one RCT was available.
The quality of available evidence was then rated according to the Grading of recommendations, Assessment, Development and Evaluation (GRADE) (12) system, that also drove the development of evidence-based recommendations. For each outcome, we rated the quality of evidence as either high, moderate, low, or very low based on: risk of bias (study limitations), inconsistency (differences between the results of trials), indirectness (differences between the questions investigated in trials and the question of interest), imprecision (random error), and other considerations (other conditions that might affect risk of bias such as conflicting results between two outcomes described in the same study, availability of only one outcome for a comparison). In the presence of different quality of evidence across different outcomes for the same PICO, the overall quality of evidence was rated as the lowest among them. High quality of evidence indicates situations in which there is high certainty that the true effect lies close to the estimated effect; low or very low quality of evidence indicates situations in which the true effect may be substantially different from the estimated effect.
For each PICO question, the coordination supporting group and the chair derived evidence-based recommendations – if possible – according to the available evidence. Each recommendation was rated as strong or weak. The strength of a recommendation indicates the extent to which one can be confident that adherence to the recommendation will give more benefit than harm. For strong recommendations we used the term “we recommend”; for weak recommendations we used the term “we suggest”. Recommendations can be either for or against an intervention, depending on the results of meta-analyses of RCTs.
To provide clinical guidance, expert-based opinions were also incorporated in addition to evidence-based recommendations. The text clearly distinguishes between the two to inform the reader about the sources of guidance. This section is intended to provide practical suggestions for the management of migraine patients, with a rationale based on the available literature.
Topics and suggestions for expert-based opinions were identified by each module working subgroup and by two experts who were responsible for harmonizing this part across all the sections (AA, CDL). Additionally, these experts developed an introductory chapter to provide general concepts on acute treatment and prevention.
Results
Full results of this guideline are presented in the main document and summarized in Table 3 for the acute treatment, in Table 4 for the prevention of episodic migraine, in Table 5 for the prevention of chronic migraine, and in Table 6 for any migraine (no distinction between episodic and chronic).
Summary of recommendations for acute migraine treatments. Rows report strength of recommendations; columns report quality of evidence.

Summary of recommendations for the prevention of any migraine (no distinction between episodic and chronic). Rows report strength of recommendations; columns report quality of evidence.
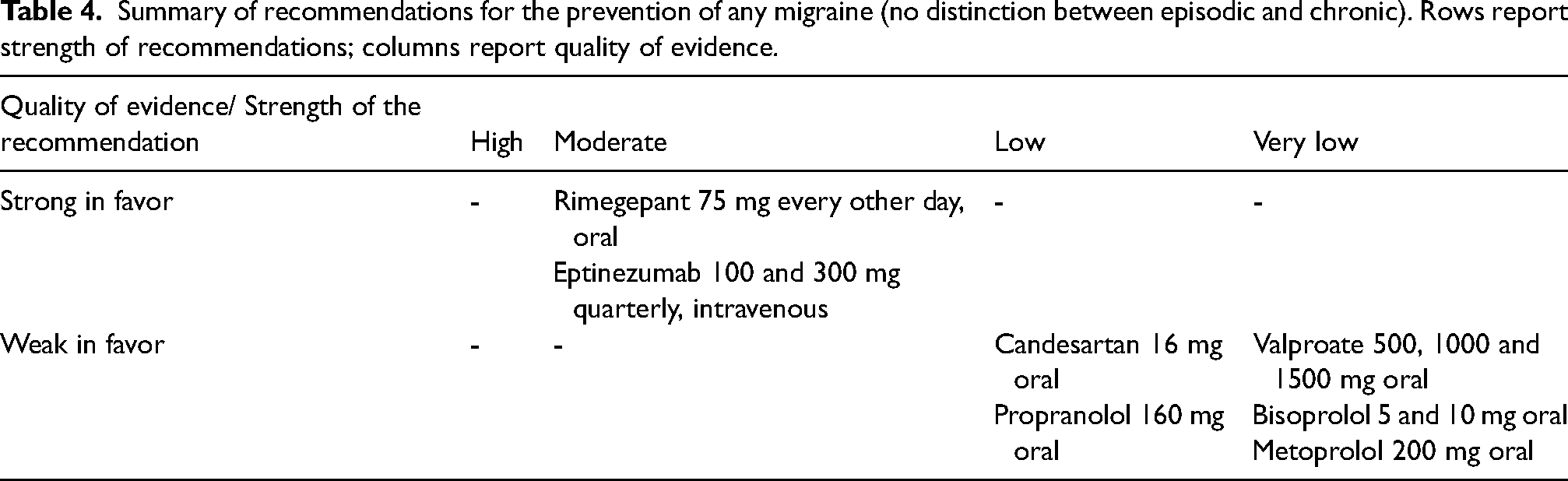
Summary of recommendations for the prevention of episodic migraine. Rows report strength of recommendations; columns report quality of evidence.

Summary of recommendations for the prevention of chronic migraine. Rows report strength of recommendations; columns report quality of evidence.

It is worth mentioning that for calcium-channel blockers there are no trials meeting the eligibility criteria for providing evidence-based recommendations. Considering the long-standing use of those drugs in the prevention of migraine an expert-based opinion is provided in chapter 4.4 to support their use.
Table 7 reports a summary of guidance for the acute treatment of migraine derived from head-to-head studies. Details for this part can be found in chapter 3.9. The following comparisons were found: NSAIDs vs NSAIDs, triptans vs NSAIDs, triptans vs triptans, triptans vs other analgesics, combination analgesics vs NSAIDs, combination analgesics vs triptans, combination analgesics vs paracetamol, combination analgesics vs combination analgesics, combination analgesics vs other analgesics.
Summary of recommendations for the treatment of acute migraine attacks derived from head-to-head comparison trials.

Table 8 reports a summary of guidance for the preventive treatment of migraine derived from head-to-head studies. Details for this part can be found in chapter 4.8. The following comparisons were found: NSAIDs vs NSAIDs, triptans vs NSAIDs, triptans vs triptans, triptans vs other analgesics, combination analgesics vs NSAIDs, combination analgesics vs triptans, combination analgesics vs paracetamol, combination analgesics vs combination analgesics, combination analgesics vs other analgesics.
Summary of recommendations for the prevention of migraine attacks derived from head-to-head comparison trials.

Discussion
This comprehensive guideline represents the first detailed framework for the acute treatment and prevention of migraines, providing evidence-based recommendations grounded in a systematic and quantitative assessment of the available randomized controlled trials (RCTs) on pharmacological treatments. The main strength of this guideline is the use of the well-established GRADE methodology offering a rating of the quality of evidence for each migraine treatment.
Notably, many migraine preventive drugs that have been widely used in clinical practice were investigated in trials conducted many years ago, often not meeting the rigorous quality criteria of modern evidence-based medicine (7,13). While this does not negate the potential effectiveness of these drugs, it does indicate that the existing evidence may not be robust enough to strongly support their use. This consideration should be applied particularly to the oldest established preventive agents for migraine, including tricyclic antidepressants, beta blockers, and calcium channel blockers. As a general rule, more recent treatments have stronger evidence than the older ones, not only because of their efficacy and safety profile, but also because of improvements over time in the design of RCTs.
Additionally, the field of migraine research suffers from a paucity of head-to-head studies with conclusive results, which hampers the ability to recommend one drug over another, with few exceptions. Therefore, it is crucial for the migraine treatment field to advance using the highest standards to derive unbiased evidence that can guide patient care effectively. There is a strong need to compare the long-term outcomes of different drugs and to understand how these medications can prevent the progression of migraine into difficult-to-treat conditions, such as high-frequency episodic migraine, chronic migraine, medication overuse headache, or refractory migraine.
Although strongly grounded in evidence-based principles, this guideline cannot be considered as a complete guide to migraine treatment. Safety outcomes were excluded from evidence-based recommendations because it is established that the considered drugs are not associated with serious risks for the patients. Besides, several topics that are relevant to clinical practice, such as drug titration, duration of treatment, and treatment combinations, were excluded from evidence-based recommendations and could only be addressed by expert-based opinions.
This guideline provides a solid foundation for deriving best practices at the national level, considering the available pharmacological treatments and the unique aspects of health systems and resident populations. Ideally, drugs supported by the best evidence regarding efficacy and tolerability profiles should be broadly accessible.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
R.O.: Consulting fees, Allergan-AbbVie, Eli Lilly. Honoraria: Eli Lilly, Novartis, Pfizer, Teva. Support for attending meetings, Allergan-AbbVie, Eli Lilly, Lundbeck, Novartis, Pfizer, Teva. Participating, Allergan-AbbVie, Eli Lilly. Leadership, Jr. Editorial Board member J Headache Pain. Receipt of equipment, Novartis, Eli Lilly. Other financial or non-financial, Eli Lilly, Novartis, Teva
V.C.: Honoraria, Teva. Support for attending meetings, Teva
F.A.: Received honorarium to be on the ad board and lecturing from AbbVie, Pfizer, Eli Lilly, TEVA, Lundbeck. Leadership, Treasurer, IHS.
H.M.A.-K.: Support for present manuscript, H.M.A.-K. reports personal fees from Pfizer, outside of the submitted work.
A.A.: Honoraria, Speakers by Novartis, Teva, Eli-Lilly, Pfizer. Congress attendance supported by Teva. Leadership, Secretary for Italian Headache Society.
S.A.: Consulting: Allergan/AbbVie, Eli Lilly, Impel NeuroPharma, Linpharma, Lundbeck, Pfizer, Satsuma, Teva, Theranica. Honoraria, Lectures presentations: AbbVie, Eli Lilly, Teva, Pfizer, Lundbeck. Leadership: Associate Editor for Cephalalgia, BMC Neurology, Frontiers in Neurology, Headache and Pain Research, Neurology Reviews, and Trustee of the International Headache Society Board.
C.B.: Honoraria, AbbVie. Support for meeting attendance, AbbVie, Lilly, Pfizer, Lundbeck
F.B.: Received honoraria for lectures, presentations from: Pfizer, Lilly, TEVA, Alnylam, AbbVie, Lundbeck, Alexion. Support for attending meetings, TEVA, Alnylam, Roche, Sanofi, Biogen, AbbVie, Alexion, Lundbeck, Lilly.
P.C.: Received speaker honoraria from: AbbVie, Bayer Schering, Bial, Biogen-Dompè, Biogen-Idec, Eisai, Genzyme, Lundbeck, Lusofarmaco, Merck- Serono, Novartis, Prexton, Teva, UCB Pharma, Zambon. PC received support to attend national and international conferences from: AbbVie, Bayer Schering, Bial, Biogen-Dompè, Biogen-Idec, Eisai, Genzyme, Lundbeck, Lusofarmaco, Merck-Serono, Novartis, Prexton, Teva, UCB Pharma, Zambon. Participation on Advisory Board for Lilly. Participation in Data Safety, Roche, Therapy for Parkinson's Disease. PC is Past President of the Società Italiana per lo Studio delle Cefalee (SISC)
S.C.: Honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events for Angelini, Teva, Novartis, AbbVie, Lundbeck, Pfizer. Support for attending meetings and/or travel by AbbVie, Lilly. Participation on Advisory Board for Lilly. Advisor of ANIRCEF. PI for trial of Lilly, Novartis, Teva, Lundbeck.
S.C.: Headache Australia website medical reviewer. AbbVie to attend 2022 Australia New Zealand Neurologist Association ASM. ordinary committee member on the Australian New Zealand Headache Society- current.
C.C.C.: Research support from the American Heart Association with funds paid to her institution. (Unrelated to this manuscript). Consulting, Satsuma, eNeura. Honoraria, HMP Global for migraine education lectures. Leadership, American Migraine Foundation Editorial Board.
A.C.: Filed patents on novel uses of anti-CGRP biologics and gepants.
R.H.C.: IHEAD support from IHS. Travel support from the Augustinus Foundation.
M.K.C.: Honoraria, Allergan Korea, Handok-Teva, Eli Lilly and Company, and Yuyu Pharmaceutical Company.
G.C.: Consulting fees, Pfizer, AbbVie. Honoraria, Pfizer, Lilly. Meeting support, TEVA, Lilly, Pfizer. Safety boards, Pfizer, AbbVie. Leadership, Trustee and co-chair of the membership committee for the International Headache Society, member of the board at large of the European Headache Federation, Secretary and member of the study center of the Italian Society for the Study of Headaches, member of the Special Interest Group on Brainstem of the International Federation of Clinical Neurophysiology.
S.C.: Funding for travel, accommodation and congress registration provided by Pfizer.
R.D.I.: Honoraria from Eli-Lilly; TEVA; Lundbeck. Funding support for meeting attendance, TEVA; Lundbeck. Advisory boards, Pfizer; AbbVie.
M.D.T: Honoraria, presentations in national conferences supported by TEVA, Lundbeck, Pfizer, Lilly. Support from TEVA for EHF last meeting in Barcelona. Leadership, President Elected Italian Society for Headache study (SISC).
C.D.L.: Consulting, Pfizer. Honoraria, Pfizer, Eli Lilly.
V.D.S.: Support for travels for attending meetings from Alexion, Alnylam, Argenx. V.D.S. is member of Italian Society of Neurology (SIN), Italian Society for the Study of Headache (SISC), Italian Society of Peripheral Nervous System (ASNP), Italian Society of Clinical Neurophysiology (SINC). V.D.S. received compensation for speaking from Alexion, and Alnylam; he is Sub-Investigator in clinical trials for Alexion, Alnylam, Argenx, Dianthus, and Sanofi.
H.-C.D.: Grants from German Research Council. Honoraria, Lundbeck, Novartis, Teva. Leadership, Clinical trials committee of the IHS NorHead Norway.
E.E.: Received honoraria for the preparation of educational materials from Allergan-AbbVie.
V.F.: Grants from AbbVie, Eli-Lilly, Lundbeck, Pfizer and Teva. Consulting fees from AbbVie, Eli-Lilly and Teva. Honoraria from Eli-Lilly, Lundbeck, Pfizer and Teva. Support for attending meetings, AbbVie and Teva. Advisory board, AbbVie.
P.G.: European Research Council - Advanced grant € 2,185,921; European research Council - Proof of Concept grant € 150,000; Royalties for books of the series Handbook of Experimental Pharmacology, Springer. Compensations varied between € 300 to 2100 per year; Pfizer; Eli Lilly; AbbVie; Lundbeck; Allergan; TEVA; Novartis: compensations varied between € 800 and € 4500. Recipient of 3 Patents (102021000015095, 102021000015098, 102021000029894) own by FloNext Srl. Current value € 0. President, Italian Society for the Study of Headache. Compensation € 0. Founder and shareholder of the Academic Spinoff (University of Florence) FloNext Srl; Current value € 0.
M.T.G.: Independent Medical Education Grant from Pfizer. Speaker or advisory board from, Teva, AbbVie, Pfizer. Support for attending meetings AbbVie, Pfizer.
A.G.: Lectures supported by Lilly, TEVA, Novartis, AbbVie. Educational meeting supported by Lilly, TEVA, AbbVie.
F.G.: Honoraria from Lundbeck and Pfizer. Leadership, President of the Italian Society for the Study of Headaches (SISC).
S.G.: Consulting fees, AbbVie, Lilly, Angelini, Pfizer, Lundbeck. Honoraria, AbbVie, Lilly, Angelini, Pfizer, Lundbeck. Meeting attendance, AbbVie, Lilly, Pfizer, Lundbeck. Advisory boards, AbbVie, Lilly, Angelini, Pfizer, Lundbeck,
W.-S.H.: Lecture on online education program for nursing patients with epilepsy in request of Korean Nurse association (2024-01-17).
A.H.: Consulting fees, Novartis, Sanofi Genzyme, Biologix, Merck, Hikma Pharma, Janssen, Inspire Pharma, Future Pharma, Elixir pharma. Honoraria for lectures/ presentations: Novartis, Allergan, Merck, Biologix, Janssen, Roche, Sanofi Genzyme, Bayer, Hikma Pharma, Al Andalus, Chemipharm, Lundbeck, Inspire Pharma, Future Pharma and Habib Scientific Office, and Everpharma. Support for attending meetings, congresses, symposia and/or travel: Novartis, Allergan, Merck, Biologix, Roche, Sanofi Genzyme, Bayer, Hikma Pharma, Chemipharm, and Al Andalus and Clavita pharm. Advisory boards, Clavita pharm Advisory board: Novartis, Sanofi Genzyme, Biologix, Merck, Hikma Pharma, Janssen, Inspire Pharma, Future Pharma, Elixir pharma. Leadership, Member of Education committee of IHS Member of Membership committee of IHS Member of regional societies committee of IHS Vice President of MENAA headache society Secretary General of headache chapter of Egyptian society of Neurology.
K.H.: Consulting, Otsuka Pharmaceutical Co., Ltd. Honoraria, Amgen Astellas BioPharma K.K., Daiichi Sankyo Company, Limited, Eisai Co., Ltd., Eli Lilly Japan K.K., MSD Co., Ltd., Otsuka Pharmaceutical Co., Ltd., Pfizer Japan Inc. Support for meeting attendance, Amgen Astellas BioPharma K.K., Daiichi Sankyo Company, Limited, Eisai Co., Ltd., Eli Lilly Japan K.K., MSD Co., Ltd., Otsuka Pharmaceutical Co., Ltd., Pfizer Japan Inc. Advisory boards, Amgen Astellas BioPharma K.K., Eli Lilly Japan K.K., MSD Co., Ltd., Otsuka Pharmaceutical Co., Ltd Leadership, Japan Patient Advocacy Coalition
J.H.: In the past 36 months I held or still hold research grants from the following entities (unrelated to this manuscript): Bristol Myers Squibb Migraine Trust International Headache Society National Institute for Health and Care Research (NIHR) Medical Research Council (MRC). Consulting and/or advisory boards for AbbVie, Cannovex, Chordate Medical, Eli Lilly, Lundbeck, Sanofi, Teva. Lectures/speaking/educational events: Chordate Medical, MD-Horizonte, Lundbeck, Teva. Manuscript writing/associate editor work: NEJM Journal Watch, Oxford University Press, Quintessence Publishing, Sage Publishing, Springer Healthcare. Data Monitoring Committee: Chordate Medical. Advisory Board: AbbVie, Chordate Medical, Eli Lilly, Lundbeck, Teva.
Board of Trustees of the International Headache Society. Council Member and Treasurer of the British Association for the Study of Headache. Associate Editor for Cephalalgia, Cephalalgia Reports, J Headache Pain, Frontiers in Pain Research, Journal of Oral & Facial Pain and Headache. Stock options for Chordate Medical.
L.F.I.: IHS junior grant. Consulting fees, Eli-Lilly, AbbVie. Honoraria, Eli-Lilly, AbbVie, Pfizer, TEVA. Meeting attendance support, TEVA, Lundbeck.
B.J.: Honoraria, Allergan/AbbVie, Eli Lilly, GPCE, HealthEd, Lundbeck, Pfizer, Teva. Leadership, President of the Australian and New Zealand Headache Society.
A.L.H.: Grants, ALR was supported by a fellowship from the Migraine Trust.
M.L.: Royalties, Oxford University Press $600. Advisory boards $6000. Expert testimony, $5000. Bd of Directors Headache Cooperative of the Pacific Unpaid.
E.M.: Honoraria, AbbVie, Elli-Lilly, TEVA, Pfizer, Lundbeck. Meeting attendance support, AbbVie, Elli-Lilly, TEVA, Pfizer, Lundbeck
D.M.: LSC LifeSciences Consultants. Honoraria, AbbVie, Lundbeck.
T.S.M.: Grants from AbbVie, Amgen Site PI: Rehaler, AbbVie, Eli Lilly. Consulting fees, Pfizer, AbbVie, Merz. Honoraria, Medscape, American Headache Society, American Academy of Neurology. Novartis, Massachusetts Medical Society. Advisory boards, Teva, Pfizer, AbbVie, eNeura, LinPharma. Leadership: IHS Board of Trustee, Florida Society of Neurology, American Headache Society, American Academy of Neurology, American Neurological Association, Editorial Boards: Cephalalgia, Neurology, Brain and Life Magazine, American Migraine Foundation, Continuum Audio.
A.Ö.: Consulting fees, AbbVie, Drogsan, ABdi İbrahim. Honoraria, AbbVie, Drogsan, ABdi İbrahim. President of Global Migraine and Pain Society, President of Mersin Alzheimer Society, Board member of IHS.
M.F.P.P.: Consulting fees, AbbVie, Pfizer, Lundbeck, Eurofarma, Libbs, Teva, Lilly. Honoraria, AbbVie, Pfizer, Lundbeck, Eurofarma, Libbs, Teva, Lilly. Patents, BR 10 2020 020706-7 US 11,826,177 B2. Advisory boards, AbbVie, Pfizer, Lundbeck, Eurofarma, Teva. Leadership, IHS, ABRACES.
M.N.P.S.: Honoraria, Libbs, TEVA, Pfizer, Allergan/AbbVie, Lundbeck. Funding for traveling to the IHC 2023, by TEVA. Advisory boards, Pfizer, TEVA, Libbs.
P.P.-R.: Her research group has received research grants from AbbVie, Novartis and Teva; as well as, Instituto Salud Carlos III, EraNet Neuron, European Regional Development Fund (001-P-001682) under the framework of the FEDER Operative Programme for Catalunya 2014-2020 - RIS3CAT; has received funding for clinical trials from AbbVie, Amgen, Biohaven, Eli Lilly, Novartis, Teva. Received, in the last 36 months, honoraria as a consultant for: AbbVie, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer and Teva. Received, in the last three years, honoraria as a speaker for: AbbVie, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva. Norwegian Clinical Trial driven by the investigator. She is the Honorary Secretary of the International Headache Society. She is on the editorial board of Revista de Neurologia. She is an associate editor for Cephalalgia, Headache, Neurologia, The Journal of Headache and Pain and Frontiers of Neurology. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society.
M.P.P.: Consulting fees, Pfizer, Teva. Honoraria, AbbVie, Allergan, Eli-Lilly, Idorsia, Italfarmaco, Lundbeck, Pfizer, Teva, Valeas. Meeting attendance support, Idorsia, Lilly, Lundbeck, Teva. Advisory boards, Allergan, Lilly, Lundbeck, Pfizer, Teva. Leadership, Italian Society for the Study of Headache (SISC), Italian Society of Neurology,
I.R.: Honoraria, Pfizer, Eli Lilly Italy, Teva. Support for meeting attendance, AbbVie. Advisory board, Pfizer,
V.R.: Ukrainian Headache Research Society, President
A.R.: Honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events (in past 36 months) from Ely-Lilly, AbbVie, Teva Pharmaceuticals, Pfizer. Support for attending meetings and/or travel (past 36 months) from Ely-Lilly, AbbVie, Teva Pharmaceuticals, Pfizer. Participation on a Data Safety Monitoring Board or Advisory Board (past 36 months) from Ely-Lilly, AbbVie, Teva Pharmaceuticals, Pfizer. Member of executive board of the Italian Headache Society (S.I.S.C.)
G.S.: Royalties, Novartis, Eli Lilly, Teva, Lundbeck, Pfizer. Consulting, Novartis, Eli Lilly, Teva, Lundbeck, Pfizer. Honoraria, Novartis, Eli Lilly, Lundbeck, Pfizer. Expert testimony, Novartis, Eli Lilly, Teva, Lundbeck, Pfizer. Stock options, Eli Lilly, Pfizer.
P.S.: Presentation and moderations for Eli-Lilly and Lundbeck. Annual congress of SISC and for Florence Symposium on CGRP (2022, 2023). Leadership, Didactic Leader for High School of Headache of SISC Italian society.
T.J.S.: American Heart Association, American Migraine Foundation, Amgen, Henry Jackson Foundation, Mayo Clinic, National Headache Foundation, National Institutes of Health, Patient Centered Outcomes Research Institute, Pfizer, Spark Neuro, United States Department of Defense. Royalties licenses, AbbVie, Allergan, Amgen, Axsome, Biodelivery Science, Biohaven, Click Therapeutics, Collegium, Eli Lilly, Ipsen, Linpharma, Lundbeck, Novartis, Satsuma, Scilex, Theranica, Tonix. Support for meeting attendance, Speech analyses for predicting migraine attacks. Predicting recovery patterns for TBI. Methods and processes for treatment migraine and post-traumatic headache. Patents, Investigator-initiated medication overuse headache clinical trial. Leadership, American Headache Society American Migraine Foundation. Stock Options: Aural Analytics, Nocira
M.S.: Speaker honoraria from Novartis, Pfizer, Allergan, Teva and Lilly.
C.T.: For this manuscript, Institutional fees for conducting clinical trials for AbbVie, Dompé, Eli Lilly, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals. Consulting fees, AbbVie, Dompé, Eli Lilly, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals. Patents, AbbVie, Dompé, Eli Lilly, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals.
A.T.: Received honoraria for lectures and speakers bureaus from AbbVie, Everpharma, Bial, Lusofarmaco, Zambon. Expert testimony, A.T. received support from Bial, AbbVie, Zambon. A.T. serves as associate Editor for the European Journal of Neurology.
M.T.: received support from the European Headache Federation for accommodation and travel tickets to attend EHC 2022 as a scientific committee member. M.T. is the president of the Iranian Headache Association.
G.V.: Honoraria from Lundbeck. Expert testimony, Teva and Lundbeck.
S.J.W: For this manuscript received research grants from the National Council of Technology and Science of Taiwan, Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, Taipei Veterans General Hospital, Taiwan Headache Society and Taiwan branches of Eli Lilly and Novartis. Received honoraria as a moderator from AbbVie, Pfizer and Biogen. Served on the advisory boards of Taiwan Pfizer, AbbVie, Hava-Biopharma.
M.A.: For this manuscript, Institutional grants from Lundbeck Foundation, Novo Nordisk Foundation, Lundbeck, and Novartis. In the past 24 months, MA has received personal compensation for consulting with AbbVie, AstraZeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals. Associate Editor of The Journal of Headache and Pain, Associate Editor of Brain.
S.S.: For this manuscript, Novartis, Uriach. Royalties, Abbott, Allergan-AbbVie, AstraZeneca, Boheringer, Eli Lilly, Lundbeck, Novartis, NovoNordisk, Pfizer, Teva. Consulting fees, Abbott, Allergan-AbbVie, AstraZeneca, Boheringer, Eli Lilly, Lundbeck, MedScape, Novartis, NovoNordisk, Pfizer, Teva. Expert testimony, Abbott, Allergan-AbbVie, AstraZeneca, Boheringer, Eli Lilly, Lundbeck, Novartis, NovoNordisk, Pfizer, Teva, Bayer, Medtronic, Starmed, Bristol-Myers-Squibb, Daiichi-Sankyo. Patents, Allergan-AbbVie, AstraZeneca, Eli Lilly, Lundbeck, Novartis, Pfizer, Teva. Leadership, President-elect European Stroke Organization, Editor-in-Chief Cephalalgia and Cephalalgia Reports, assistant editor for Stroke.
A.B., F.C., I.C., A.F., K.N.G., E.-M.H., M.H., A.Laporta, A.Lupica, I.O., L.-L.H.P., L.L.P., S.Q., M.R., D.B.S. have no conflict of interest to disclose
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
