Abstract
We here present evidence-based guidelines for the pharmacological treatment of migraine. These guidelines, created by the Italian Society for the Study of Headache and the International Headache Society, aim to offer clear, actionable recommendations to healthcare professionals. They incorporate evidence-based recommendations from randomized controlled trials and expert-based opinions. The guidelines follow the Grading of Recommendations, Assessment, Development and Evaluation approach for assessing the quality of evidence. The guideline development involved a systematic review of literature across multiple databases, adherence to Cochrane review methods, and a structured framework for data extraction and interpretation. Although the guidelines provide a robust foundation for migraine treatment, they also highlight gaps in current research, such as the paucity of head-to-head drug comparisons and the need for long-term outcome studies. These guidelines serve as a resource to standardize migraine treatment and promote high-quality care across different healthcare settings.
Summary of recommendations
Table 1.1 reports the contents of the guideline. Tables 1.2–1.5 report the summary of all the evidence-based recommendations.
Contents of the guideline
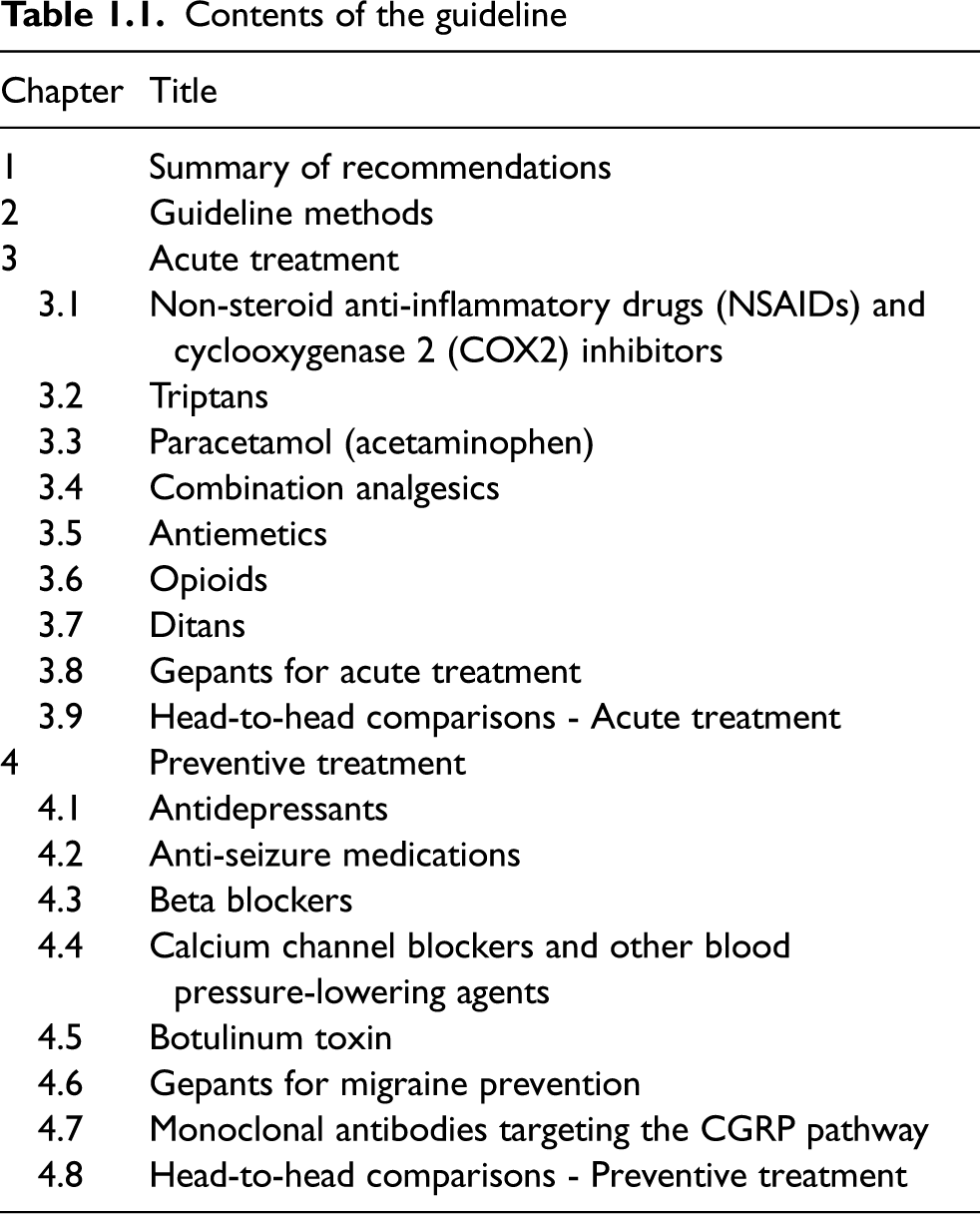
Contents of the guideline

Summary of recommendations for acute migraine treatments. Rows report strength of recommendations; columns report quality of evidence

Summary of recommendations for the prevention of any migraine (no distinction between episodic and chronic). Rows report strength of recommendations; columns report quality of evidence

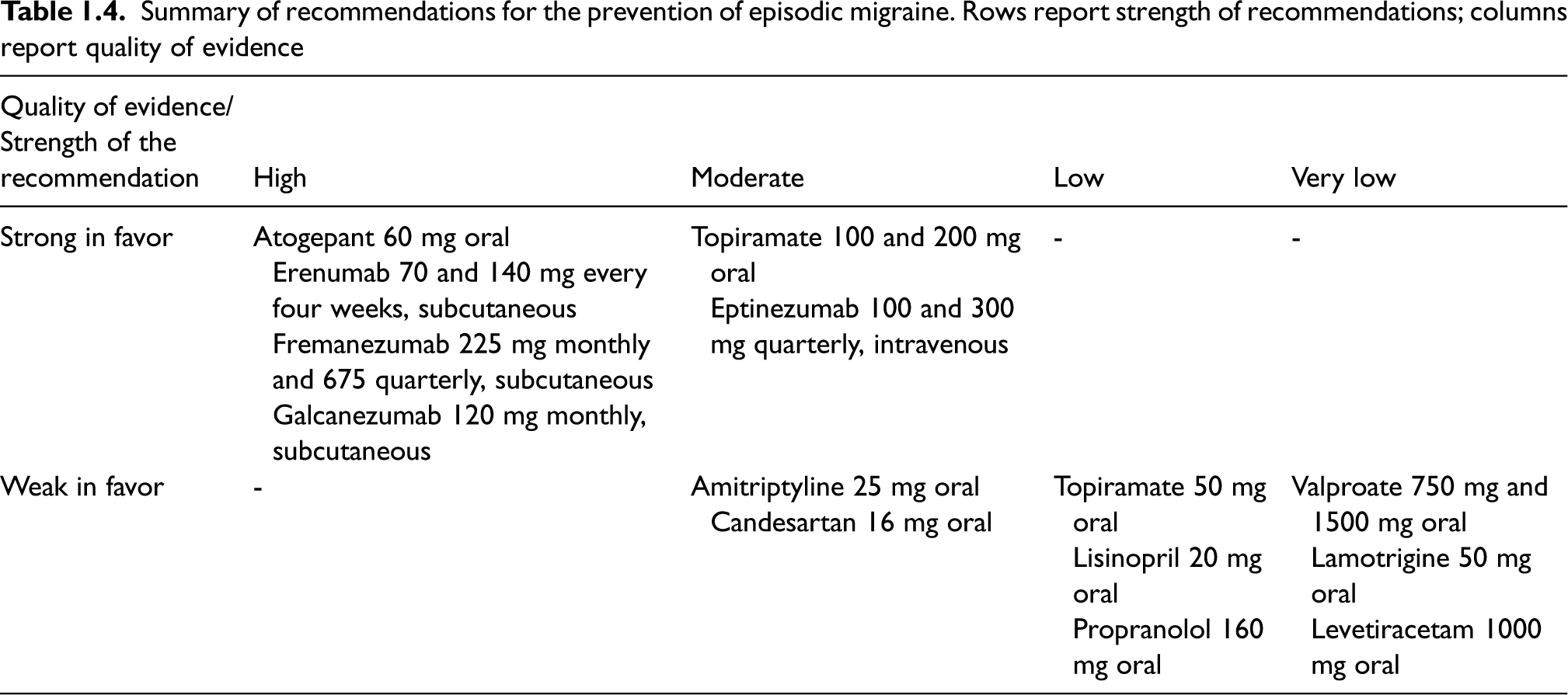
Summary of recommendations for the prevention of episodic migraine. Rows report strength of recommendations; columns report quality of evidence
Summary of recommendations for the prevention of chronic migraine. Rows report strength of recommendations; columns report quality of evidence

Details on available evidence can be found in each specific section.
Guideline working groups and process
This Guideline was initiated by the Italian Society for the Study of Headaches (‘Società Italiana per lo Studio delle Cefalee’ – SISC) and later continued as a joint guideline with the International Headache Society (IHS). The SISC Board appointed initial members to form the Guideline Working Group and the IHS added further members. The Guideline Working Group consisted of a chair, a coordination supporting group, internal reviewers, and 19 module subgroups responsible for addressing the different sections of the guideline. One subgroup evaluated the methods, eight the acute treatment of migraine attacks, and seven the preventive treatments. Additionally, the coordination supporting group evaluated head-to-head comparisons of acute and preventive treatments. All subgroups included at least two senior experts in migraine management, defined as physicians with more than 15 years of clinical and research experience in migraine treatment, as verified by the chair and coordination supporting group. The SISC and the IHS Boards were responsible for the selection of senior experts from their respective Societies.
To encompass all the possible pharmacological treatments of migraine, we kept acute and preventive treatments together in one Guideline. Acute and preventive treatments were examined in two different Sections, Section 3 and Section 4, respectively. The final Guideline consists of three parts:
A full document (the present document); A short version for rapid consultation; Appendices containing details about literature searches.
To ensure consistency and homogeneity of approach across the groups, a web-based meeting was organized to share the objectives and methodology of the guidelines and to train all researchers involved in the guideline’s development. Small groups meetings were held as needed to monitor progress, verify homogeneity of approach across groups, and address any problems encountered during the development process.
The responsibility for study selection, accuracy of extracted data, and data analysis for both placebo-controlled and head-to-head comparative studies was assigned to each group dealing with specific drug classes. The placebo-controlled studies comparisons were assessed in sections dedicated to each specific drug class for the final report. Two additional sections were included to report head-to-head comparisons in acute treatment and prevention, respectively.
The IHS contribution to the guidelines consisted in the addition of an IHS chair to the SISC chair, two IHS members to the internal reviewers’ group, and one or two members for each of the 19 SISC subgroups. The IHS members initially revised the literature search performed by the coordination support group and by the SISC groups; afterwards, they performed two updates on the search; finally, they revised the updated manuscripts.
This guideline is based on the best available evidence from randomized controlled trials (RCTs) and a rigorous evaluation of the quality of evidence for each intervention and outcome. Clinically relevant questions were framed using the PICO format (Population, Intervention, Comparator, and Outcomes). An initial set of questions was developed by the SISC chair and coordinating supporting group according to clinical experience and previous guidelines. Additional questions were added based on available evidence. To provide clinical guidance, we also used expert-based opinions. The text clearly distinguishes between evidence-based recommendations and expert-based opinions to inform the reader where guidance is based solely on evidence data or where it considers experience and opinions.
PICO questions
Clinical questions for evidence-based recommendations were developed according to the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) system as PICO. The PICO format is valuable to clearly define the Population (P), Intervention (I), Comparator (C), and Outcomes (O).
The Population (P) of interest is represented by subjects with migraine. We developed separate PICO questions for episodic and chronic migraine; the group ‘any migraine’ (no distinction between episodic and chronic) was also considered depending on available data. Intervention (I) was represented by any drug belonging to a class used for the acute or preventive treatment of migraine attacks. Comparators (C) were placebo or active drug(s). Outcomes (O) were different for treatments used for acute and for preventive treatment of migraine attacks and were chosen based on the IHS guidelines for clinical trials for migraine (1–3) because they were rated as important or critical by members of the Guideline Working Groups. For the treatment of acute migraine attack, the following outcomes were considered:
pain freedom at 2 h from intake; pain relief at 2 h from intake.
Other outcomes such as the absence of the most bothersome symptom at 2 h were not considered as they were measured only in the most recent RCTs.
For preventive treatments, the following outcomes were considered:
persisting monthly headache/migraine days, defined as the residual days reported by subjects in the final period of the treatment (as reported in headache diaries); change in monthly headache/migraine days, defined as the variation in days reported by patients from baseline to the end of follow-up (as reported in headache diaries); ≥50% responder rate, defined as the proportions of subjects reporting a ≥50% reduction in monthly headache/migraine days compared with baseline. The ≥50% reduction of monthly attacks was also considered for ≥50% responder rate whenever the reduction in monthly headache/migraine days was not available.
The chosen outcomes were more extensive than those issued by the International Headache Society guidelines for RCTs of migraine prevention (2,3), in order to include the highest possible number of RCTs. Patient-reported outcomes were not included because of substantial heterogeneity across instruments used. Given the expected minimal impact of serious adverse events, we did not consider safety as an important or critical outcome to derive evidence-based recommendations. We addressed tolerability and used this information to draft expert-based opinion sections.
The final guideline report includes patient groups, interventions, comparators, and outcomes for which the systematic literature search showed the presence of available RCTs.
Literature search
Search of available evidence was performed according to the Cochrane guidelines for systematic reviews of interventions (4) and overviews of reviews (5). Cochrane guidelines were also followed for study selection, data extraction and synthesis. Reporting was performed according to relevant items of the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement (6).
A literature search was performed between 10 and 11 February 2022 for all topics except from antiemetics, which was included in the search plan in September 2022; related search strings were launched on 8 September 2022. As the process of literature search and analysis took more than 12 months, search strings were re-launched in May 2023 and November 2023 to update the search to the RCTs published from February 2022. Two researchers (VC and RO) performed a literature search for each pharmacological class of acute treatments – namely, non-steroidal anti-inflammatory drugs, triptans, paracetamol, combination analgesics, opioids, ditans, and gepants – and migraine preventive drugs – namely, antidepressants, anti-seizure medications, beta-blockers, calcium channel blockers, blood pressure-lowering medications, botulinum toxin, gepants, and monoclonal antibodies targeting the CGRP pathway. Three scientific databases were searched, namely PubMed, Scopus, and Cochrane Database, since the beginning of indexing, utilizing the PICOM (Patients – Intervention – Comparison – Outcome – Methods) methodology. To ensure a broad coverage of available literature, when building search strings, only Participants (i.e., migraine patients) and Interventions (i.e., drugs) were considered for each topic. The same search strings were launched in two separate searches. In Search 1, we looked for systematic reviews and meta-analyses, while in Search 2 we looked for RCTs published after the reviews and the meta-analyses retrieved in Search 1. If Search 1 did not allow the retrieval of any systematic reviews or meta-analysis, Search 2 was considered for RCT inclusion since the beginning of indexing of each database. Search 1 was performed at the beginning of the activity, while Search 2 was performed at the beginning and repeated in May 2023 and November 2023. Only published literature was considered for searches. The full search strategies are included in each section of this guideline. Reference management and duplicate removal were performed with EndNote X6®.
Study selection
Study selection was performed by each module subgroup. The selection process was performed in two stages. In Stage 1, systematic reviews and meta-analysis covering the topic of interest were screened to identify eligible studies. In Stage 2, additional RCTs, published after the selected systematic review and meta-analyses were considered for inclusion. In case no systematic reviews and meta-analyses were available, only RCTs were selected.
Inclusion and exclusion criteria for evaluation of references in eligibility and inclusion phases for stage 1

Inclusion and exclusion criteria for evaluation of references in eligibility and inclusion phases for stage 2

If duplicates were identified during study selection, they were considered and accounted for in the study selection flow-chart. Full texts of all RCTs identified in all systematic reviews and meta-analyses included in Stage 1 were evaluated according to the same criteria. Therefore, module subgroups selected the final number of RCTs included in the review. This final number was revised if needed after the literature search updates performed in May 2023 and November 2023.
Data extraction
Utilizing an Excel spreadsheet template, module subgroups extracted the following data for each included study:
Type of migraine: episodic or chronic or any migraine (no distinction between episodic and chronic) according to the inclusion criteria described in evaluated studies and considering ICHD criteria (7). We considered as separate groups subjects with episodic and chronic migraine when selected studies were specifically aimed to address those groups individually in their primary analyses. In the absence of primary analyses considering episodic and chronic migraine or if it was not possible to individuate those groups according to ICHD criteria, we included the data in the ‘any migraine’ section; Comparison(s): compared drugs, doses, and administration routes; Number of subjects: total number and number in each treatment group; For continuous outcomes: mean and standard deviation or standard error or confidence interval (either 95% or 99%) in each treatment group. Standard deviation = √ Standard error Standard deviation = Standard deviation = For categorical outcomes: number of subjects reporting the outcome in each treatment group. If the required data was not available, the outcome was extracted as reported in the studies. For the outcomes ‘persisting monthly migraine days’ and ‘change in monthly migraine days’, assessment time points were retrieved, according to what was reported in the included studies; if these outcomes were reported at different time points, we selected them according to the following priority: 12 weeks, 24 weeks, and others. For the outcomes pain freedom at 2 h and pain relief at 2 h, if outcome data were reported for more than one headache attack, we considered the outcome of the treatment of the first attack described.
If needed, standard deviations were calculated starting from standard errors or form confidence intervals, using one of the following formulas:
If the required data were not available, the outcome was extracted as reported in studies (e.g., outcome of interest expressed as median and not mean).
Data analysis
For each PICO question, all extracted data about outcomes were classified, analyzed, and presented as main evidence or additional evidence as reported below.
Meta-analyses were performed using RevMan®, version 5.3. Computed effect sizes were Standardized Mean Difference (SMD) for continuous outcomes and Relative Risk (RR) for categorical outcomes. Pooled effect sizes were computed using the random effect model and expressed with a 95% Confidence Interval (95% CI).
Risk of bias evaluation
The risk of bias of all RCTs was evaluated according to the Cochrane tool (4) and included in the forest plots. This evaluation was performed twice from two different raters of each module working subgroup after a wash-out period of at least 15 days. Disagreements and indecisions were solved by discussion with another member of the module working subgroup. Items included in the risk of bias assessment were the following:
Random sequence generation (selection bias) Allocation concealment (selection bias) Blinding of participants and personnel (performance bias) Blinding of outcome assessment (detection bias) Incomplete outcome data (attrition bias) Selective reporting (reporting bias) Other bias
Evidence quality rating
The quality of available evidence was then rated according to the Grading of recommendations, Assessment, Development and Evaluation (GRADE) (8) system, which also drove the development of evidence-based recommendations.
The overall quality of evidence for outcomes derived from studies considered in the main evidence was evaluated according to the GRADE methodology and reported in evidence profile tables (5). For each outcome, we rated the quality of evidence as either high, moderate, low, or very low based on: risk of bias (study limitations), inconsistency (differences between the results of trials), indirectness (differences between the questions investigated in trials and the question of interest), imprecision (random error), and other considerations (e.g., conflicting results between two outcomes described in the same study, availability of only one outcome for a comparison). For the assessment of imprecision, the clinical decision threshold was established as a risk ratio limit of 0.5 to 2.0 (9); thus, if the confidence interval of a RCT was not significant but included those thresholds, the results were considered imprecise. Imprecision was also considered serious if the numbers of participants included in the RCT were <50 for each arm.
To rate the overall risk of bias in the GRADE evidence profile tables, we used adapted Cochrane criteria as reported in Table 2.3. GRADE tables were built using GradePro®. In the presence of different quality of evidence across different outcomes for the same PICO, the overall quality of evidence was rated as the lowest among them. High quality of evidence indicates situations in which there is high certainty that the true effect lies close to the estimated effect; low or very low quality of evidence indicates situations in which the true effect may be substantially different from the estimated effect.
Approach for summary assessments of the risk of bias for each important outcome (across domains) within and across studies

For data included in additional evidence, the quality of evidence was considered by default low and downgraded to very low in the presence of additional limitations. According to the GRADE methodology, we also considered a potential upgrade of the quality of evidence in the case of evidence of large magnitude of the therapeutic effect.
Drafting recommendations
For each PICO question, the coordination supporting group and the chair derived evidence-based recommendations from the main evidence, if available. The additional evidence was used to develop evidence-based recommendations in the absence of the main evidence. For PICO with both main and additional evidence, the overall quality of evidence was derived from the main evidence, but additional evidence was considered to conceptualize the recommendation. Recommendations were issued for each question with available main evidence. For those with only additional evidence, recommendations were issued only if an adequate number of participants (≥50 per each arm of RCTs) were included.
For each recommendation, we provided the quality of evidence and the strength of the recommendation.
Quality of evidence reflects confidence in the estimate as reported above. The strength of a recommendation indicates the extent to which one can be confident that adherence to the recommendation will provide more benefit than harm. Recommendations were rated as “strong” if the addition of further evidence was unlikely to determine a change in the overall result or “weak” if the addition of further evidence was deemed likely to change the overall results. For strong recommendations we used the term “we recommend”; for weak recommendations we used the term “we suggest”. Recommendations can be either for or against an intervention, depending on the results of meta-analyses of RCTs. In case of conflicting results (e.g., superiority to placebo in one outcome and neutrality in another outcome), the most precise result – i.e., that with the narrowest confidence interval – was considered.
Expert-based opinions
To provide clinical guidance, expert-based opinions were also incorporated in addition to evidence-based recommendations. The text clearly distinguishes between the two to inform the reader about the sources of guidance. This section is intended to provide practical suggestions for the management of migraine patients, with a rationale based on the available literature.
Topics and suggestions for expert-based opinions were identified by each module working subgroup and by two experts who were responsible for harmonizing this part across all the sections (AA, CDL). Additionally, these experts developed an introductory section to provide general concepts on acute treatment and prevention.
Internal review
All data synthesis, evidence-based recommendations, and expert-based opinions were reviewed by four experts (CT, IR, H-CD, ML). The resulting evidence-based recommendations and expert-based opinions were then sent to the entire working group (SISC and IHS) for approval. To be included in the guidelines, statements needed to receive at least 70% approval from the working group. Topics that did not meet the approval threshold were still considered in the document synthesis and discussed in the text.
Non-steroidal anti-inflammatory drugs (NSAIDs) and cycloxygenase 2 (COX2) inhibitors
Introduction
Nonsteroidal anti-inflammatory drugs (NSAIDs) are a class of heterogeneous compounds that largely share therapeutic and adverse effects. The class includes acetylated salicylates (aspirin), non-acetylated salicylates (diflunisal, salsalate), propionic acids (naproxen, ibuprofen), acetic acids (diclofenac, indomethacin), enolic acids (meloxicam, piroxicam), anthranilic acids (meclofenamate, mefenamic acid), naphthylalanine (nabumetone), and diaryl heterocyclic compounds (celecoxib, rofecoxib, parecoxib, etoricoxib, and lumiracoxib) (10).
NSAIDs are approved for use as antipyretic, anti-inflammatory, and analgesic agents.
The main therapeutic effects of NSAIDs derive from their ability to inhibit the prostaglandin synthase enzymes known as cycloxygenases (COXs) (10). There are two forms of COXs, COX1, expressed constitutively in most cells and COX2, which is induced by cytokines, shear stress and tumor promoters (10). COX1 is expressed as the dominant constitutive form in gastric epithelial cells and is considered as the major source of cytoprotective prostaglandin production (10,11).
Traditional NSAIDs inhibit both COX types and are believed to interfere with the homeostatic functions of the constitutively expressed COX1. NSAIDs gastrointestinal (GI) toxicity is related to the inhibition of COX1 and subsequent decrease in GI protection (11).
Aspirin irreversibly inhibits the activity of COXs and the duration of its effects is related to the turnover of COXs in different tissues. For example, platelets are anucleate and have a reduced ability to synthesize proteins, therefore COX1 inhibition lasts the platelet lifetime (10).
When used as general analgesics in non-migraine pain, both unselective (NSAIDs) and selective COX-2 inhibitors (Coxibs) may be less effective than opioids, but lack the opioid adverse effect of depressing the respiratory centers and have less potential for physical dependence (10). These drugs are particularly effective when inflammation has caused peripheral and or central sensitization of pain perception and this explains their usefulness in migraine attack therapy (10).
It is probable that the analgesic and anti-inflammatory actions of NSAIDs are not limited to COX inhibition. At higher concentrations NSAIDS are known to reduce the production of superoxide radicals, decrease NO synthase and pro-inflammatory cytokines, and modify lymphocyte activities in vitro (10).
Experimental findings suggest that celecoxib-mediated COX2 inhibition reduces the intensity of migraine headache and potentially terminates an attack via the attenuation of dural macrophages’ activation and arterial dilatation outside the blood-brain barrier, and pial macrophages’ activation inside the blood-brain barrier (12).
Compared to other NSAIDs, indomethacin may be a more potent vasoconstrictor, that more consistently reduces cerebral blood flow (CBF) and inhibits carbon dioxide (CO2) reactivity (13). In addition, indomethacin reduces cerebrospinal fluid pressure (14).
NSAIDs side effects consist mainly in gastrointestinal adverse events (from gastric pain to gastric or duodenal ulcers), which are mostly due to COX1 inhibition and the Coxibs were designed to reduce the risk of GI side effects, due to their selective inhibition of COX2 (10).
Since the late nineteenth century with the introduction of synthetic acetylsalicylic acid, NSAIDs have been largely used for the acute treatment of migraine – at least in attacks of mild-to-moderate intensity. NSAIDs have been recommended as first-line drugs, particularly for attacks of mild-to-moderate intensity, being the most commonly used analgesic agents worldwide (15–18).
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for the guideline on NSAIDs for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 3.1.1.
Results
Overall, we retrieved 1346 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included seven systematic reviews and meta-analyses (19–25) that were considered as sources of RCTs. After analyzing the full texts of these RCTs, we included 17 of them in the quantitative synthesis (26–42) (Figure 3.1.1).

Meta-analysis selection flow-chart - non-steroidal anti-inflammatory drugs and COX2 inhibitors.
Overall, we retrieved 7973 references from searching for RCTs and after removing duplicates we had 7689 references to analyze. However, considering the most recent included meta-analysis on the topic, the analysis of additional RCTs was performed for studies published since 2010 (31 references). We finally included two additional RCTs (43,44). From the literature search update performed in May 2023, we retrieved 534 references. Further 38 references were retrieved from the November 2023 update. We included one study from this updated search (45) (Figure 3.1.2).

RCTs selection flow-chart. *trials published since 2010 were screened.
Overall, 20 studies were included in the data synthesis to develop the evidence-based guideline. All the studies reported a comparison between an active drug and placebo and were presented in this section. Eight RCTs also included head-to-head comparisons (26,27,29,30,37,38,41,44) and were included in Section 3.9.
Acetylsalicylic acid (Aspirin)
Main evidence
We found three RCTs (29,33,34) addressing oral acetylsalicylic acid 1000 mg compared to placebo in the treatment of acute migraine attack that met the criteria to be included in main evidence (Figures 3.1.3 and 3.1.4).

Forest plot showing the comparison between oral acetylsalicylic acid 1000 mg and oral placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between oral acetylsalicylic acid 1000 mg and oral placebo for the outcome pain relief at 2 h.
The pooled analysis showed the superiority of oral acetylsalicylic acid over placebo considering the outcomes pain freedom at 2 h (Figure 3.1.3) and pain relief at 2 h (Figure 3.1.4). The quality of evidence for both outcomes was considered moderate (Table 3.1.1).
GRADE evidence profile table for oral acetylsalicylic acid 1000mg versus oral placebo in patients with migraine

Additional evidence
We found three RCTs (26,30,36) addressing oral acetylsalicylic acid 900 mg and 1000 mg compared to oral placebo in the acute treatment of migraine that did not meet the pre-defined criteria to be included in main evidence. The quality of evidence was considered very low (Figures 3.1.5 and 3.1.6).

Forest plot showing the comparison between oral acetylsalicylic acid 900 mg or 1000 mg and oral placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between oral acetylsalicylic acid 900 mg or 1000 mg and oral placebo for the outcome pain relief at 2 h.
The pooled analysis showed the superiority of oral acetylsalicylic acid 1000 mg over oral placebo considering the outcomes pain freedom at 2 h (Figure 3.1.5) and pain relief at 2 h (Figure 3.1.6). The 900 mg oral acetylsalicylic acid dose reached statistical significance only for pain relief.
Evidence-based recommendation for PICO 3.1.1 In subjects with migraine, we recommend oral acetylsalicylic acid 1000 mg for the acute treatment of migraine attacks. Quality of evidence: Moderate ⊕⊕⊕⊖ Strength of the recommendation: Strong (↑↑).
Safety and tolerability of NSAIDs
NSAIDs mechanism of action (COX1 and COX2 inhibition - see introduction section) is responsible for both effectiveness and adverse effects of this pharmaceutical class. Here we report a general discussion about NSAIDs adverse effects (AE). We will also report some AEs specific to each drug.
Gastrointestinal complaints are the most frequent AEs to NSAIDs and include nausea, vomiting, and dyspepsia. Hypersensitivity to NSAIDs represents one of the major concerns in the use of those drugs, also because allergy to one of these drugs compromises the use of the others by possibly leading to anaphylactic reactions. Other important NSAIDs adverse effects (often therapeutically used) are a major risk of bleeding, especially gastrointestinal bleeding, due to their antiplatelet activity. Renal adverse effects have also been reported in patients with pre-existing renal dysfunction, as have cardiovascular adverse effects (thromboembolic disease, atrial fibrillation) and, less commonly, hepatic damages.
Safety and tolerability of acetylsalicylic acid
Dexketoprofen trometamol
Main evidence
None.
Additional evidence
We found one RCT (42) addressing oral dexketoprofen trometamol 25–50 mg compared to oral placebo in the acute treatment of migraine that did not meet the criteria to be included in the main evidence. The quality of evidence was considered very low (Figures 3.1.7 and 3.1.8).

Forest plot showing the comparison between oral dexketoprofen trometamol 25 mg or dexketoprofen trometamol 50 mg and oral placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between oral dexketoprofen trometamol 25 mg or dexketoprofen trometamol 50 mg and oral placebo for the outcome pain relief at 2 h.
The study showed superiority of oral dexketoprofen trometamol 50 mg over oral placebo considering the outcomes pain freedom at 2 h (Figure 3.1.7) and pain relief at 2 h (Figure 3.1.8). The benefits of the 25 mg dosing were not proven for the outcome pain freedom at 2 h.
We found one RCT (42) addressing oral dexketoprofen trometamol 25 mg compared to oral dexketoprofen trometamol 50 mg in the acute treatment of migraine that did not meet the criteria to be included in main evidence. The overall risk of bias was considered unclear and the quality of evidence very low (Figures 3.1.9 and 3.1.10).
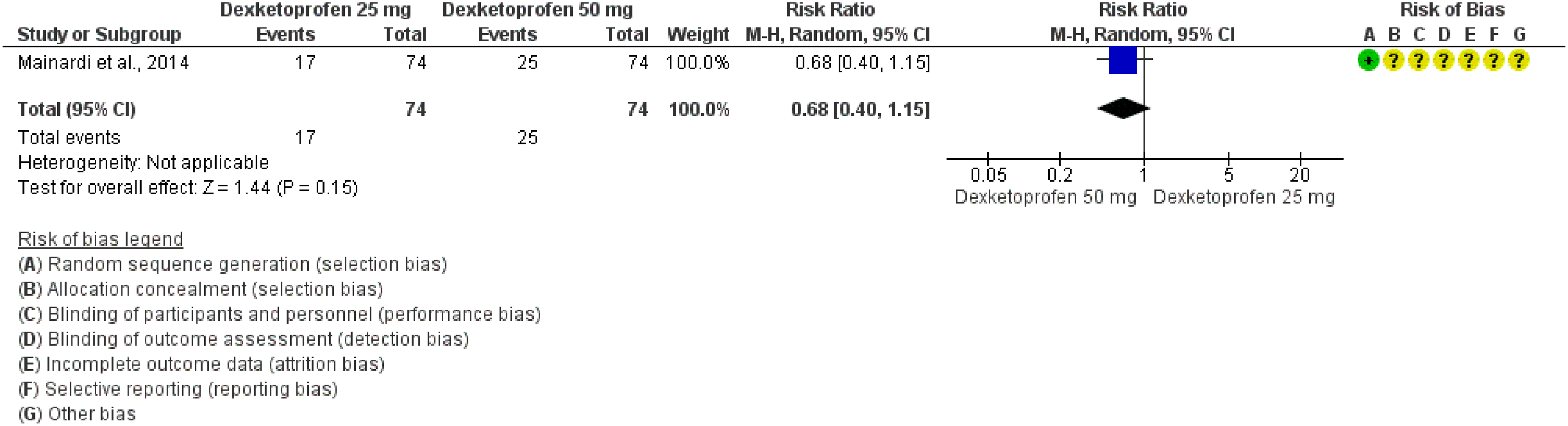
Forest plot showing the comparison between oral dexketoprofen trometamol 25 mg and oral dexketoprofen trometamol 50 mg for the outcome pain free at 2 h.

Forest plot showing the comparison between oral dexketoprofen trometamol 25 mg and oral dexketoprofen trometamol 50 mg for the outcome pain relief at 2 h.
The study did not show a significant difference between oral dexketoprofen trometamol 50 mg and dexketoprofen 25 mg considering the outcomes pain freedom at 2 h (Figure 3.1.9) and pain relief at 2 h (Figure 3.1.10).
Evidence-based recommendation for PICO 3.1.2 In subjects with migraine, we suggest oral dexketoprofen trometamol 50 mg for the acute treatment of migraine attacks. There is uncertain evidence of the efficacy of the 25 mg dose of oral dexketoprofen. Quality of evidence: Very low (⊕⊖⊖⊖). Strength of the recommendation: Weak (↑)
Safety and tolerability of dexketoprofen trometamol
Diclofenac
Main evidence
We found two RCTs (31,35) addressing diclofenac potassium 50 mg orally dispersible tablets (ODTs) compared to placebo and one RCT (45) addressing subcutaneous diclofenac sodium compared to subcutaneous placebo in the acute treatment of migraine. All the RCTs met the criteria for main evidence.
The pooled analysis showed benefits of oral diclofenac potassium 50 mg over placebo considering the outcomes pain freedom at 2 h (Figure 3.1.11) and pain relief at 2 h (Figure 3.1.12). Subcutaneous diclofenac sodium was superior to subcutaneous placebo considering the outcome pain relief at 2 h (Figure 3.1.12). The quality of evidence for both outcomes was considered moderate (Table 3.1.2).

Forest plot showing the comparison between diclofenac and placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between diclofenac and placebo for the outcome pain relief at 2 h.
GRADE evidence profile table for diclofenac versus placebo in patients with migraine

Only one trial; blow numbers of patients.
Additional evidence
None.
Evidence-based recommendations for PICO 3.1.3 In subjects with migraine, we recommend oral diclofenac potassium 50 mg (tablets or solution) for the acute treatment of migraine attacks.
Quality of evidence: Moderate ⊕⊕⊕⊖
Strength of the recommendation: Strong (↑↑)
In subjects with migraine, we recommend subcutaneous diclofenac sodium 50 mg for the acute treatment of migraine attacks.
Quality of evidence: Low ⊕⊕⊖⊖
Strength of the recommendation: Weak (↑)
Safety and tolerability of diclofenac
Ibuprofen
Main evidence
We found six RCTs (28,30,32,37–39) addressing oral ibuprofen 200 mg, 400 mg or 600 mg as compared to placebo in the treatment of acute migraine attack that met the criteria to be included in main evidence.
The pooled analysis showed benefits of ibuprofen 200 mg, 400 mg and 600 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.1.13) and pain relief at 2 h (Figure 3.1.14). The quality of evidence for both outcomes was considered moderate (Table 3.1.3).

Forest plot showing the comparison between oral ibuprofen 200–400–600 mg and placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between oral ibuprofen 200–400–600 mg and placebo for the outcome pain relief at 2 h.
GRADE evidence profile table for oral ibuprofen 200, 400, and 600 mg vs placebo in migraine

Only one trial.
We found two RCTs (28,32) comparing different dosages of ibuprofen (200 mg, 400 mg, and 600 mg) that met the criteria for main evidence. The pooled analyses did not show significant differences among the different dosages of the drug when considering the outcomes of pain freedom at 2 h and pain relief at 2 h (Figure 3.1.15–Figure 3.1.20). The quality of evidence for all comparisons and both outcomes was considered moderate or low (Table 3.1.4).

Forest plot showing the comparison between oral ibuprofen 200 mg and oral ibuprofen 400 mg for the outcome pain freedom at 2 h.

Forest plot showing the comparison between oral ibuprofen 200 mg and oral ibuprofen 400 mg for the outcome pain relief at 2 h.

Forest plot showing the comparison between oral ibuprofen 400 mg and oral ibuprofen 600 mg for the outcome pain freedom at 2 h.

Forest plot showing the comparison between oral ibuprofen 400 mg and oral ibuprofen 600 mg for the outcome pain relief at 2 h.

Forest plot showing the comparison between oral ibuprofen 200 mg and oral ibuprofen 600 mg for the outcome pain freedom at 2 h.

Forest plot showing the comparison between oral ibuprofen 200 mg and oral ibuprofen 600 mg for the outcome pain relief at 2 h.
GRADE evidence profile table for oral ibuprofen 200, 400, or 600 mg versus placebo in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.1.4 In subjects with migraine, we recommend oral ibuprofen 200–400–600 mg for the acute treatment of migraine attacks.
Quality of evidence: Low ⊕⊕⊖⊖
Strength of the recommendation: Strong (↑↑).
Safety and tolerability of ibuprofen
Ketorolac
Main evidence
We found one RCT (44) addressing ketorolac 31.5 mg nasal spray as compared to placebo in the acute treatment of migraine that met the criteria for main evidence.
The study showed benefits of ketorolac 31.5 mg nasal spray over placebo considering the outcomes of pain freedom at 2 h (Figure 3.1.21) and pain relief at 2 h (Figure 3.1.22). The quality of evidence for both outcomes was considered moderate (Table 3.1.5).

Forest plot showing the comparison between ketorolac 31.5 mg nasal spray and placebo nasal spray for the outcome pain freedom at 2 h.
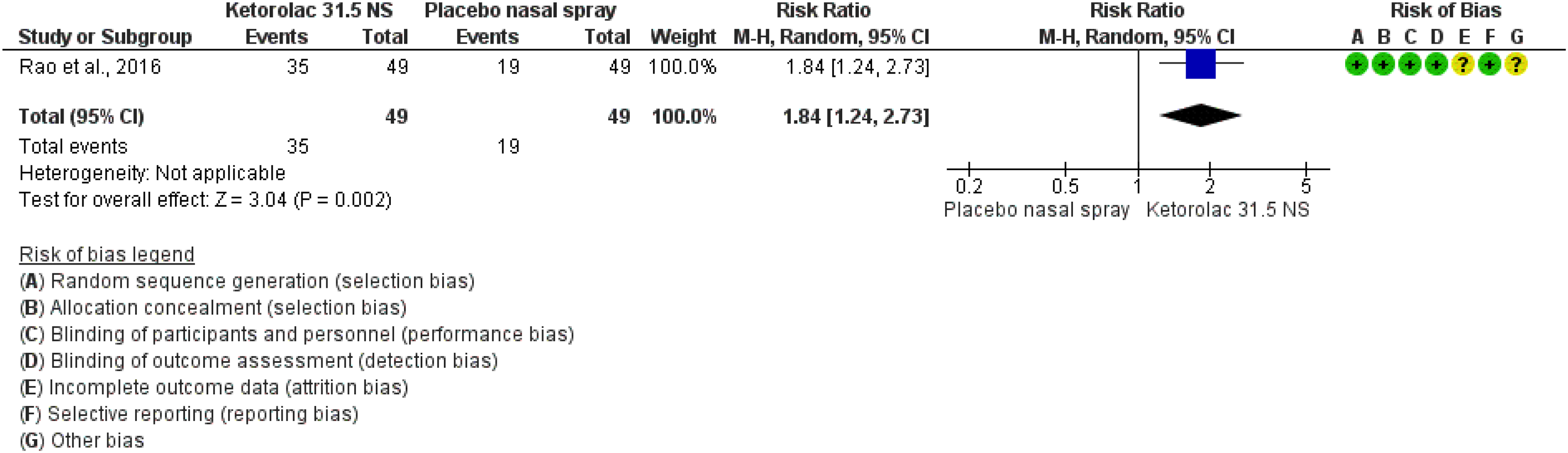
Forest plot showing the comparison between ketorolac 31.5 mg nasal spray and placebo nasal spray for the outcome pain relief at 2 h.
GRADE evidence profile table for ketorolac nasal spray versus placebo in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.1.5 In subjects with migraine, we suggest ketorolac 31.5 mg nasal spray for the acute treatment of migraine attacks.
Quality of evidence: Low ⊕⊕⊖⊖
Strength of the recommendation: Weak (↑).
Safety and tolerability of ketorolac
Naproxen
Main evidence
None.
Additional evidence
We found three RCTs (27,41) addressing oral naproxen 500–825 mg as compared to placebo in the treatment of acute migraine attack that did not meet the criteria to be included in main evidence. We point out that one RCT (27) reported data from two different studies. The overall risk of bias was considered unclear and the quality of evidence very low (Figures 3.1.23 and 3.1.24).

Forest plot showing the comparison between naproxen 500 mg or naproxen 825 mg and placebo for the outcome pain freedom at 2 h. One trial (27) reported results of two studies (a and b).

Forest plot showing the comparison between naproxen 500 mg or naproxen 825 mg and placebo for the outcome pain relief at 2 h. One trial (27) reported results of two studies (a and b).
The pooled analysis showed benefits of naproxen 500 mg and 825 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.1.23) and pain relief at 2 h (Figure 3.1.24).
Evidence-based recommendation for PICO 3.1.6 In subjects with migraine, we recommend oral naproxen 500 mg or 825 mg for the acute treatment of migraine attacks. Quality of evidence: Very low (⊕⊖⊖⊖).
Strength of the recommendation: Strong (↑↑).
Safety and tolerability of naproxen
Celecoxib
Main evidence
We found one RCT (43) addressing oral celecoxib 120 mg compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in main evidence.
The study showed benefits of celecoxib 120 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.1.25) and pain relief at 2 h (Figure 3.1.26). The quality of evidence for both outcomes was considered moderate (Table 3.1.6).

Forest plot showing the comparison between celecoxib 120 mg and placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between celecoxib 120 mg and placebo for the outcome pain relief at 2 h.
GRADE evidence profile table for oral celecoxib 120 mg versus placebo in patients with migraine
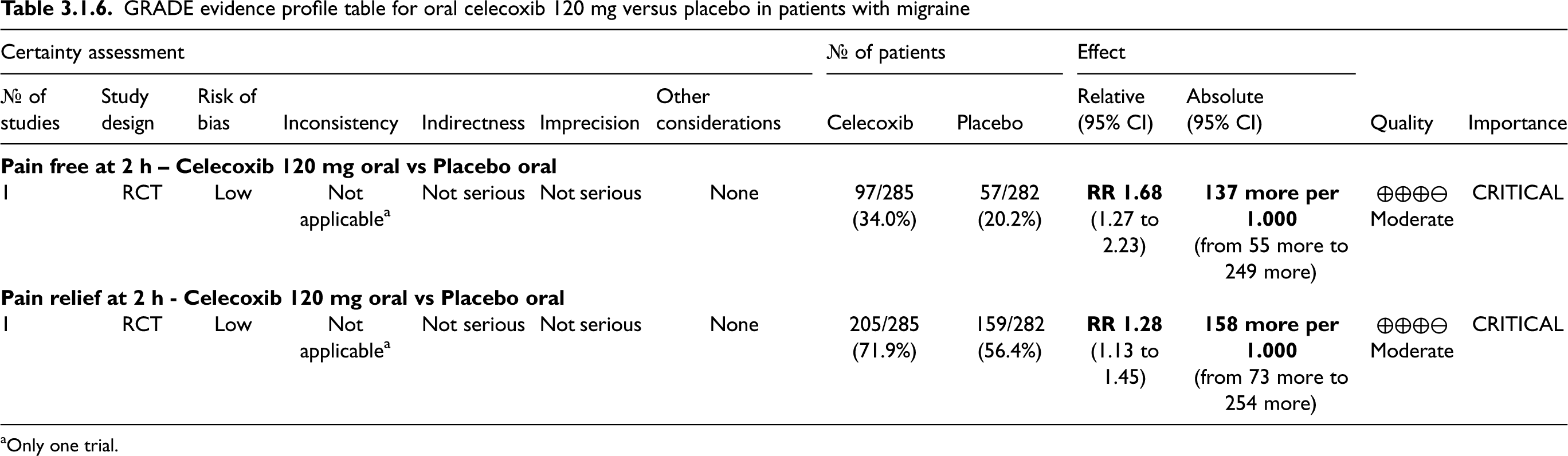
Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.1.7 In subjects with migraine, we recommend oral celecoxib 120 mg for the acute treatment of migraine attacks.
Quality of evidence: Moderate ⊕⊕⊕⊖
Strength of the recommendation: Weak (↑)
Safety and tolerability of coxibs
Coxibs mechanism of action, which is different to common NSAIDs, does not involve COX1 blockage, thus is not responsible for antiplatelet effect. Like non-selective NSAIDs, coxibs bear a cardiovascular risk (including the increased risk of heart attacks and strokes – for these concerns rofecoxib was withdrawn from the market) and of gastrointestinal effects (bleeding and ulceration). In addition, coxibs may worsen hypertension, renal and liver failure. Allergic reactions, up to and including anaphylaxis, are described. It should be mentioned that cardiovascular adverse effects of coxibs were reported when the drugs were taken daily for joint or back pain (51,52); it is reasonable to expect that the cardiovascular safety of coxibs is better if they are taken occasionally to treat episodic migraine.
Safety and tolerability of celecoxib
Evidence-based guideline summary
We found studies on aspirin, dexketoprofen trometamol, diclofenac potassium, ibuprofen, ketorolac, naproxen, and celecoxib to be included for deriving evidence-based recommendations for this guideline. Evidence-based recommendations were possible for several NSAIDs and for celecoxib (Figure 3.1.27).

Summary of evidence-based recommendations on NSAIDs for the acute treatment of migraine attacks.
Expert-based opinions
Topic: Low versus high dosage
Topic: Timing to take medication
Topic: NSAIDs versus coxibs
Topic: Which NSAID to use
Topic: Indomethacin
Introduction
Triptans have represented a significant advancement in the acute treatment of migraine (57). They were originally developed to address the acute treatment of migraine by emulating ergot-like properties while avoiding its well-known adverse effects (58). Belonging to the tryptamine family and structurally similar to serotonin (5-HT) with substitutions in 3 and 5 positions, triptans act as selective agonist of 5-HT1B/1D receptors (59). Although they were initially chosen based on their vasoactive properties, recent research has challenged the vascular mechanisms of action associated with them. While the precise mechanisms of action remain unknown, triptans are believed to exert their effects at various levels of the nervous system (60,61). Specifically, their activity on the 5-HT1B receptors induces vasoconstriction of meningeal vessels, while their action on the 5-HT1D receptors inhibits the release of vasoactive peptides involved in neurogenic inflammation (62) and pain signaling (63–65).
Since the introduction of sumatriptan, several other triptans have been developed, all administered orally but with some available in different formulations (nasal spray, oral disintegrating tablets, etc.), and marketed (zolmitriptan, rizatriptan, naratriptan, almotriptan, eletriptan and frovatriptan), to date. Due to subtle differences in their chemical structure, triptans demonstrate unique pharmacokinetic characteristics peculiarities leading to differences in efficacy and tolerability profile (66). Herein, we conducted a systematic review and meta-analysis of randomized controlled trials (RCTs) investigating the efficacy and safety of triptans when compared with placebo. Efficacy and tolerability were evaluated across all recommended doses in the meta-analysis of placebo-controlled trials. In addition, this analysis includes comparisons among different doses of the same triptan, while head-to-head comparisons among different triptans are presented in Section 3.9. Combinations of triptans and other acute drugs are presented in Section 3.4.
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for the guideline on triptans for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 3.2.1.
Results
Overall, we retrieved 2445 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included 21 systematic reviews and meta-analyses (58, 67–86) that were considered as source of randomized controlled trials (RCTs). After analyzing full texts of these RCTs, we included 119 of them in the quantitative synthesis (27, 29, 38, 41, 44, 87–199) (Figure 3.2.1).

Meta-analysis selection flow-chart - triptans.
Overall, we retrieved 4616 references from searching for RCTs and after removing duplicates we had 3653 references to analyze. However, considering the most recent included systematic review and meta-analysis (77) on the topic, the analysis of additional RCTs was performed for papers published since 2016 (1114 references). We finally included two additional RCTs (200,201) (Figure 3.2.2).

RCTs selection flow-chart - triptans. *trials published since 2016 were screened.
From the literature search update performed in May 2023, we retrieved 270 references, while 161 further references were retrieved from the update performed in November 2023. We did not include further studies from this additional search.
Overall, 121 studies were included in data synthesis to develop the evidence-based guideline. Among these, 46 reported a comparison between two active drugs and their results are included in Section 3.9.
Almotriptan
Main evidence
We found five RCTs (87–91) addressing oral almotriptan compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in the main evidence.
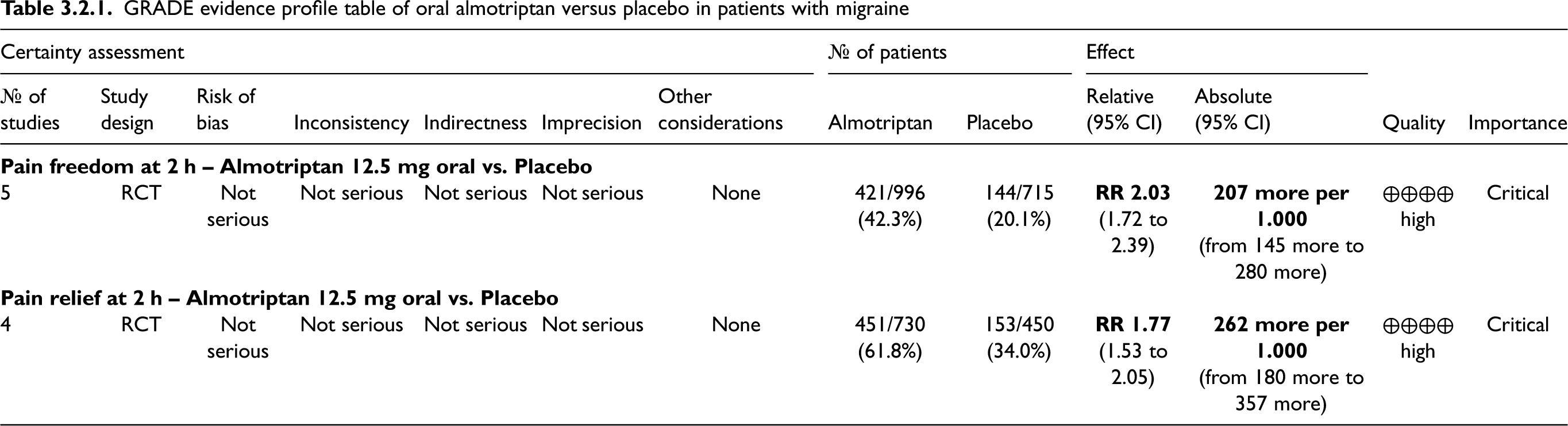
The pooled analysis showed benefits of oral almotriptan 12.5 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.2.3) and pain relief at 2 h (Figure 3.2.4). The quality of evidence for both outcomes was considered high (Table 3.2.1).

Forest plot showing the comparison between almotriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between almotriptan 12.5 mg and placebo for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of oral almotriptan versus placebo in patients with migraine
Additional evidence
None.
Evidence-based recommendation for PICO 3.2.1 In subjects with migraine, we recommend oral almotriptan 12.5 mg for the acute treatment of migraine attacks. Quality of evidence: High (⊕⊕⊕⊕).
Strength of the recommendation: Strong (↑↑).
Eletriptan
Main evidence
We found 11 RCTs (92–101,167) addressing eletriptan as compared to placebo in the acute treatment of migraine attacks that met the criteria for main evidence.
The pooled analysis showed benefits of oral eletriptan 20 and 40 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.2.5) and pain relief at 2 h (Figure 3.2.6). The quality of evidence for both outcomes was considered high (Table 3.2.2).
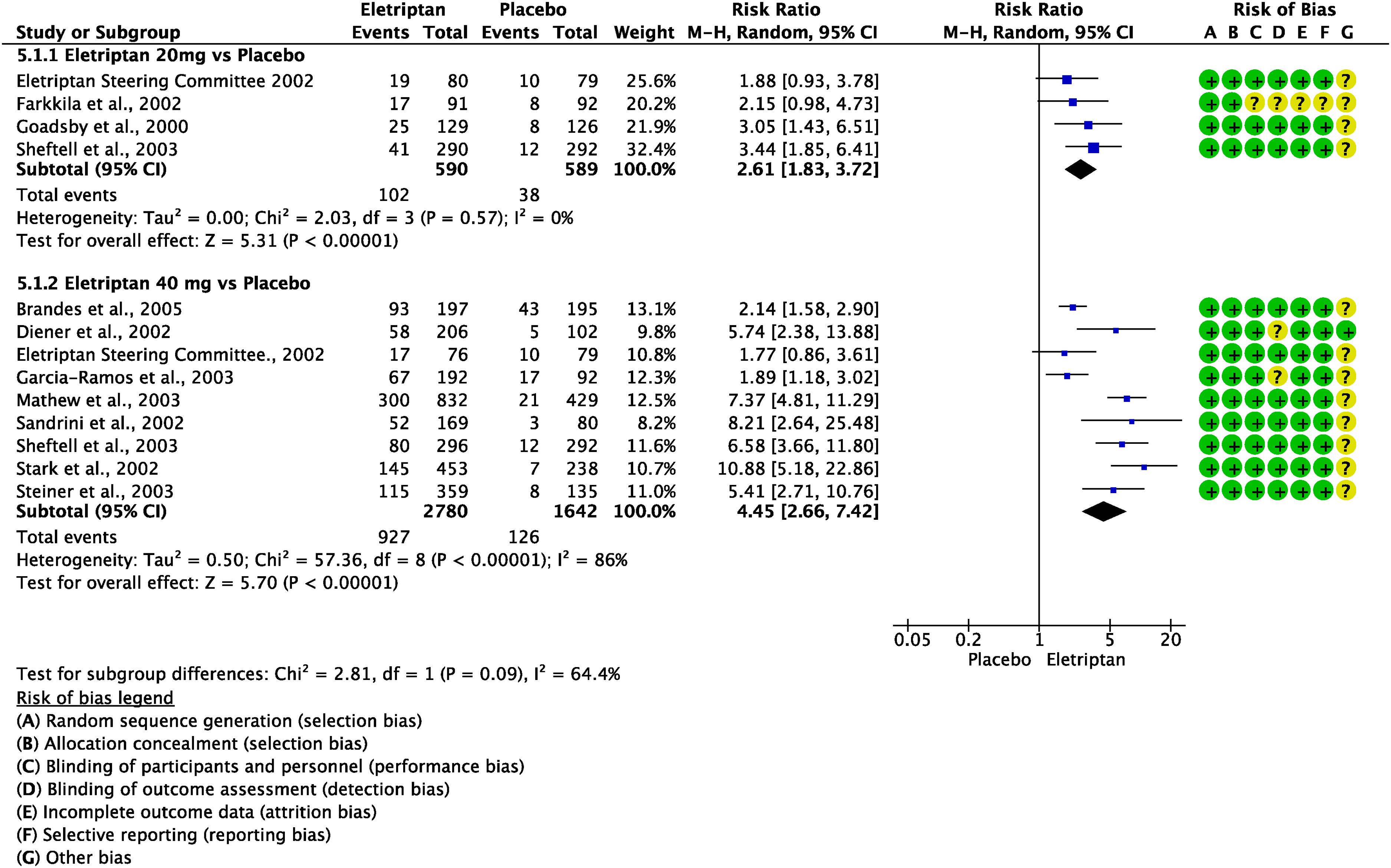
Forest plot showing the comparison between eletriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral eletriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of oral eletriptan versus placebo in patients with migraine

Additional evidence
We found one RCT (101) addressing oral eletriptan 40 mg as compared to placebo in the acute treatment of migraine attack that did not meet the criteria for main evidence. The overall risk of bias was considered low (Figures 3.2.7 and 3.2.8).

Forest plot showing the comparison between oral eletriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral eletriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
The pooled analysis showed benefits of oral eletriptan 40 mg over placebo considering the outcomes pain freedom at 2 h (Figure 3.2.7) and pain relief at 2 h (Figure 3.2.8).
Evidence-based recommendation for PICO 3.2.2 In subjects with migraine, we recommend oral eletriptan 20 and 40 mg for the acute treatment of migraine attacks. Quality of evidence: High (⊕⊕⊕⊕).
Strength of the recommendation: Strong (↑↑).
Frovatriptan
Main evidence
We found three RCTs (102–104) addressing oral frovatriptan compared to placebo in the acute treatment of migraine attacks that met the pre-defined criteria to be included in the main evidence.
The pooled analysis showed benefits of oral frovatriptan 2.5 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.2.9) and pain relief at 2 h (Figure 3.2.10). The quality of evidence for pain freedom was considered moderate (due to wide confidence interval) while it was considered high for pain relief (Table 3.2.3).

Forest plot showing the comparison between frovatriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral frovatriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of oral frovatriptan versus placebo in patients with migraine

Additional evidence
We found two RCTs (105,106) addressing oral frovatriptan 2.5 mg compared to placebo in the acute treatment of migraine attack that did not meet the pre-defined criteria to be included in the main evidence. The overall risk of bias was considered low. The pooled analysis showed benefits of frovatriptan over placebo considering both the outcomes of pain freedom at 2 h (Figure 3.2.11) and pain relief at 2 h (Figure 3.2.12).
Evidence-based recommendation for PICO 3.2.3 In subjects with migraine, we recommend oral frovatriptan 2.5 mg for the acute treatment of migraine attacks. Quality of evidence: High (⊕⊕⊕⊕).
Strength of the recommendation: Strong (↑↑).

Forest plot showing the comparison between frovatriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between frovatriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
Naratriptan
Main evidence
We found four RCTs (96,107–109) addressing oral naratriptan as compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of oral naratriptan 2.5 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.2.13) and pain relief at 2 h (Figure 3.2.14) whereas data from two studies showed benefits of naratriptan 1 mg over placebo considering the outcomes pain-free at 2 h (Figure 3.2.13) and pain relief at 2 h (Figure 3.2.14). The quality of evidence for both outcomes was considered high for naratriptan 2.5 mg and moderate for naratriptan 1 mg (Table 3.2.4).

Forest plot showing the comparison between oral naratriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral naratriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of oral naratriptan versus placebo in patients with migraine

Only one trial.
Additional evidence
We found one RCT (110) addressing oral naratriptan 2.5 mg as compared to placebo in the acute treatment of migraine attacks that did not meet the pre-defined criteria to be included in the main evidence. The overall risk of bias was considered moderate, and the quality of evidence was considered very low (Figures 3.2.15 and 3.2.16).

Forest plot showing the comparison between oral naratriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral naratriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
The results did not confirm benefits of oral naratriptan 2.5 mg over placebo considering the outcomes pain freedom at 2 h (Figure 3.2.15), whereas they showed benefit of naratriptan 2.5 mg considering the outcome of pain relief at 2 h (Figure 3.2.16).
Evidence-based recommendation for PICO 3.2.4 In subjects with migraine, we recommend oral naratriptan 1 mg or 2.5 mg for the acute treatment of migraine attacks. Quality of evidence: High (⊕⊕⊕⊕).
Strength of the recommendation: Strong (↑↑).
Rizatriptan
Main evidence
We found 12 RCTs (38,107,111–120) addressing oral rizatriptan as compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of oral rizatriptan 5 and 10 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.2.17) and pain relief at 2 h (Figure 3.2.18). The quality of evidence for both outcomes was considered high (Table 3.2.5).

Forest plot showing the comparison between oral rizatriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral rizatriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of oral rizatriptan versus placebo in the acute treatment of migraine attacks

Additional evidence
We found one RCT (121) addressing oral rizatriptan as compared to placebo in the acute treatment of migraine attacks that did not meet the pre-defined criteria to be included in the main evidence. The overall risk of bias was considered high, and the quality of evidence was considered very low (Figures 3.2.19 and 3.2.20).

Forest plot showing the comparison between rizatriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between rizatriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
The results showed benefits of oral rizatriptan over placebo considering the outcomes of freedom at 2 h (Figure 3.2.19) and pain relief at 2 h (Figure 3.2.20).
We found one RCT (115) addressing oral rizatriptan 5 mg as compared to oral rizatriptan 10 mg in the acute treatment of migraine attacks that met the criteria to be included in the main evidence. The overall risk of bias was considered low and the quality of evidence was considered high. The study did not show a significant difference between oral rizatriptan 5 mg and oral rizatriptan 10 mg considering the outcomes of pain freedom at 2 h (Figure 3.2.21). The quality of evidence was considered moderate (Table 3.2.6).
Evidence-based recommendation for PICO 3.2.5 In subjects with migraine, we recommend oral rizatriptan 5 or 10 mg for the acute treatment of migraine attacks. Quality of evidence: High (⊕⊕⊕⊕).
Strength of the recommendation: Strong (↑↑).

Forest plot showing the comparison between rizatriptan 5 mg and rizatriptan 10 mg placebo for the outcome pain freedom at 2 h in patients with migraine.
GRADE evidence profile table of oral rizatriptan 5 mg versus oral rizatriptan 10 mg in the acute treatment of migraine attacks

Only one trial.
Sumatriptan
Main evidence
We found 27 RCTs (30,41,44,93,97,98,112,114,122–136,176,182,194,198) addressing sumatriptan as compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of oral (50 and 100 mg), subcutaneous (6 mg/mL) and nasal spray (10 and 20 mg) sumatriptan over placebo considering the outcomes pain freedom at 2 h (Figure 3.2.22) and pain relief at 2 h (Figure 3.2.23). The quality of evidence for both outcomes was considered high except for sumatriptan 10 mg nasal spray, for which it was considered moderate (Table 3.2.7).

Forest plot showing the comparison between sumatriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between sumatriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of sumatriptan versus placebo in patients with migraine
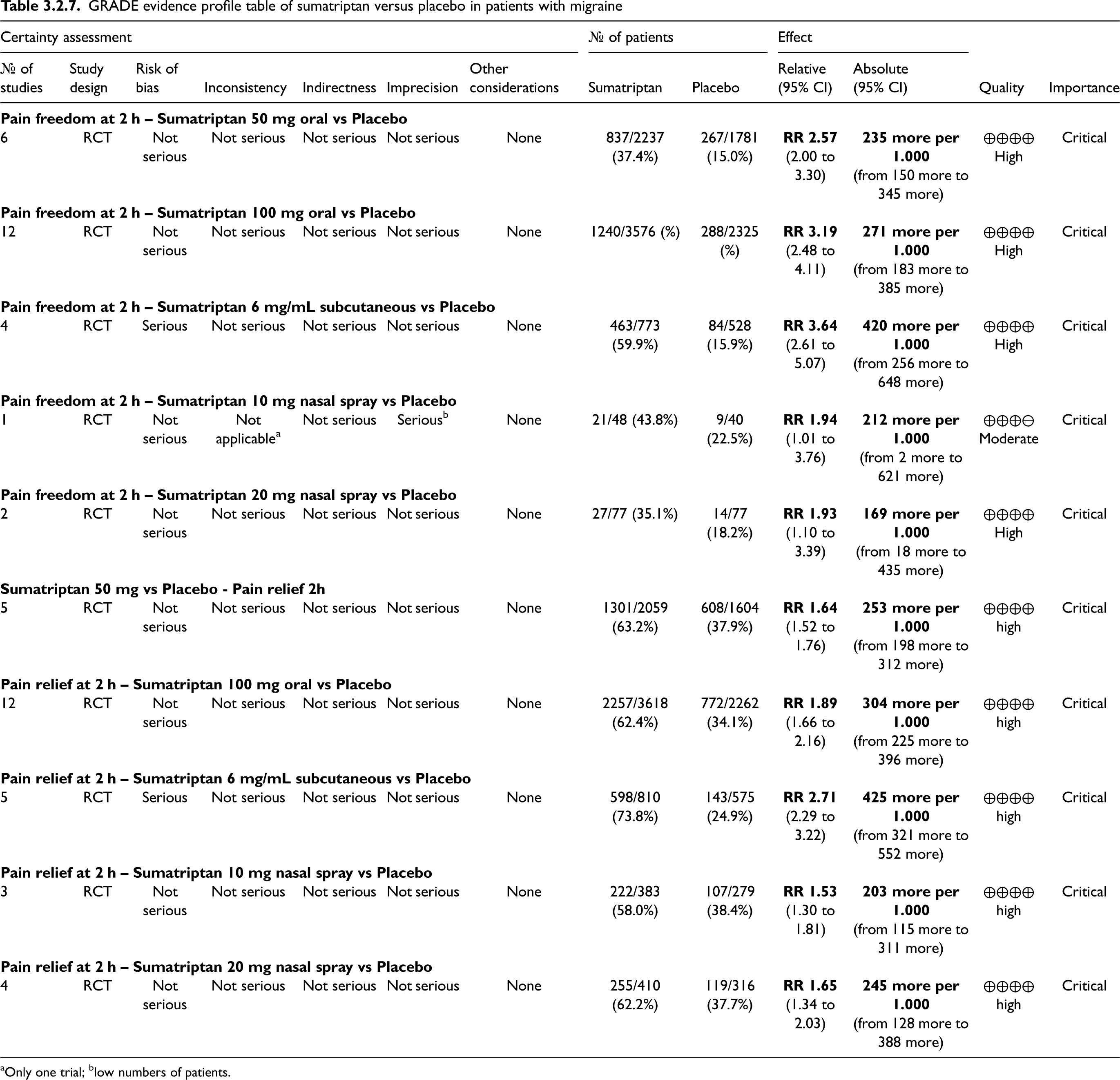
Only one trial; blow numbers of patients.
Additional evidence
We found 12 RCTs (29,121,137,139–145,199,202) addressing oral or subcutaneous sumatriptan compared to placebo in the acute treatment of migraine attacks that did not meet the criteria to be included in the main evidence. The pooled analysis showed benefits of oral (50 and 100 mg) and subcutaneous (6 mg/mL) sumatriptan over placebo considering the outcomes of pain freedom at 2 h (Figure 3.2.24) and pain relief at 2 h (Figure 3.2.25).

Forest plot showing the comparison between sumatriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between sumatriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
We found two RCTs (146,147) addressing oral sumatriptan 50 mg compared to oral sumatriptan 100 mg in the acute treatment of migraine attacks that met the criteria to be included in the main evidence. The overall risk of bias was considered low and the quality of evidence was considered high (Figure 3.2.26). The studies showed a significant difference between oral sumatriptan 50 mg and oral sumatriptan 100 mg considering the outcome pain freedom at 2 h (Figure 3.2.26). The pooled analysis showed no benefit of oral sumatriptan 100 mg over oral sumatriptan 50 mg considering the outcome pain freedom at 2 h. The quality of evidence was considered moderate (Table 3.2.8).

Forest plot showing the comparison between sumatriptan 50 mg and sumatriptan 100 mg for the outcome pain freedom at 2 h in patients with migraine.
GRADE evidence profile table of sumatriptan 50 mg versus sumatriptan 100 mg in the acute treatment of migraine attacks

*Wide confidence interval.
We found one RCT (148) addressing oral sumatriptan 100 mg as compared to oral sumatriptan 50 mg in the acute treatment of migraine attacks that did not meet the criteria to be included in the main evidence. The study did not show a significant difference between oral sumatriptan 50 mg and oral sumatriptan 100 mg considering the outcomes pain freedom at 2 h (Figure 3.2.27) and pain relief at 2 h (Figure 3.2.28).
Evidence-based recommendations for PICO 3.2.6 1) In subjects with migraine, we recommend oral sumatriptan 50 and 100 mg for the acute treatment of migraine attacks. Strength of the recommendation: Strong (↑↑).
2) In subjects with migraine, we recommend subcutaneous sumatriptan 6 mg/mL for the acute treatment of migraine attacks. Strength of the recommendation: Strong (↑↑).
3) In subjects with migraine, we recommend sumatriptan nasal spray 10 and 20 mg for the acute treatment of migraine attacks.
Strength of the recommendation: Strong (↑↑)
Quality of evidence: High (⊕⊕⊕⊕).
Quality of evidence: High (⊕⊕⊕⊕).
Quality of evidence: High (⊕⊕⊕⊕).

Forest plot showing the comparison between sumatriptan 50 mg and sumatriptan 100 mg for the outcome pain freedom6 at 2 h in patients with migraine.

Forest plot showing the comparison between sumatriptan 50 mg and sumatriptan 100 mg for the outcome pain relief at 2 h in patients with migraine.
Zolmitriptan
Main evidence
We found 11 RCTs (100,120,150–158) addressing zolmitriptan as compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of oral zolmitriptan 2.5 mg over placebo considering the outcomes of freedom at 2 h (Figure 3.2.29) and pain relief at 2 h (Figure 3.2.30). The quality of evidence for both outcomes was considered high. Additionally, pooled analysis did not show benefits from zolmitriptan 5 mg nasal spray over placebo considering the outcome pain freedom at 2 h. The outcome pain relief was not available; quality of evidence was considered low (Table 3.2.9).
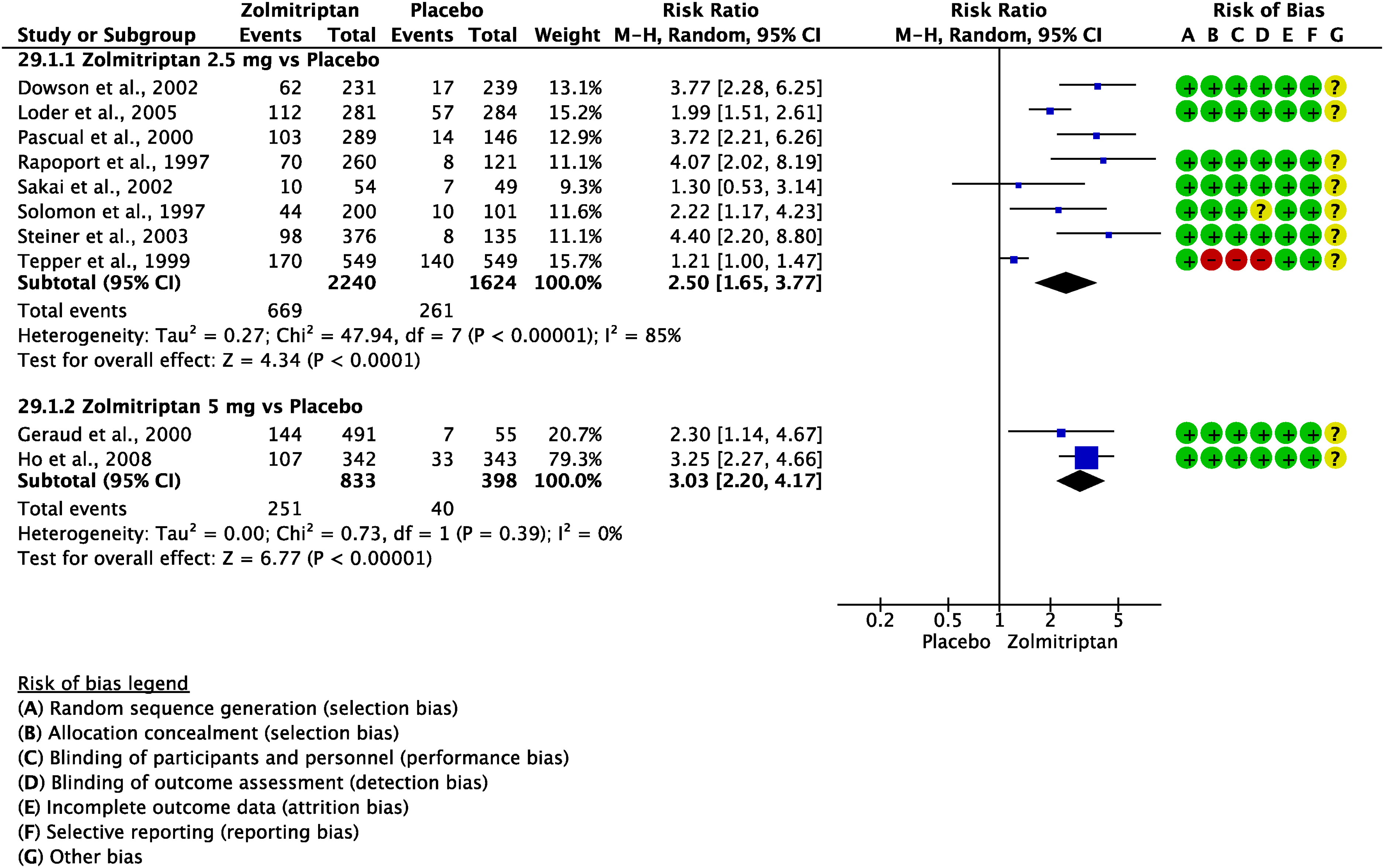
Forest plot showing the comparison between oral zolmitriptan and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral zolmitriptan and placebo for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of oral zolmitriptan versus placebo in the acute treatment of migraine attacks

Wide confidence intervals.
We found one RCT (154) addressing oral zolmitriptan 2.5 mg as compared to oral zolmitriptan 5 mg in the acute treatment of migraine attacks that met the criteria to be included in the main evidence. The overall risk of bias was considered high and the quality of evidence was considered very low. The study did not show a significant difference between oral zolmitriptan 2.5 mg and oral zolmitriptan 5 mg considering the outcomes of pain freedom at 2 h (Figure 3.2.31) and pain relief at 2 h (Figure 3.2.32). The pooled analysis did not show benefits of oral zolmitriptan 5 mg over oral zolmitriptan 2.5 mg considering the outcomes of pain freedom at 2 h and pain relief at 2 h (Figures 3.2.31 and 3.2.32). The quality of evidence for both outcomes was considered very low (Table 3.2.10).

Forest plot showing the comparison between oral zolmitriptan 5 mg and oral zolmitriptan 2.5 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral zolmitriptan 5 mg and oral zolmitriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of oral zolmitriptan 2.5 mg versus oral zolmitriptan 5 mg in patients with migraine

Only one trial.
We found one RCT (157) addressing oral zolmitriptan 2.5 mg compared to zolmitriptan 5 mg nasal spray in the acute treatment of migraine attacks that met the criteria to be included in the main evidence. The overall risk of bias was considered low. The study did not show a significant difference between oral zolmitriptan 2.5 mg and zolmitriptan 5 mg nasal spray considering the outcome of pain relief at 2 h (Figure 3.2.33). The quality of evidence was considered moderate (Table 3.2.11).
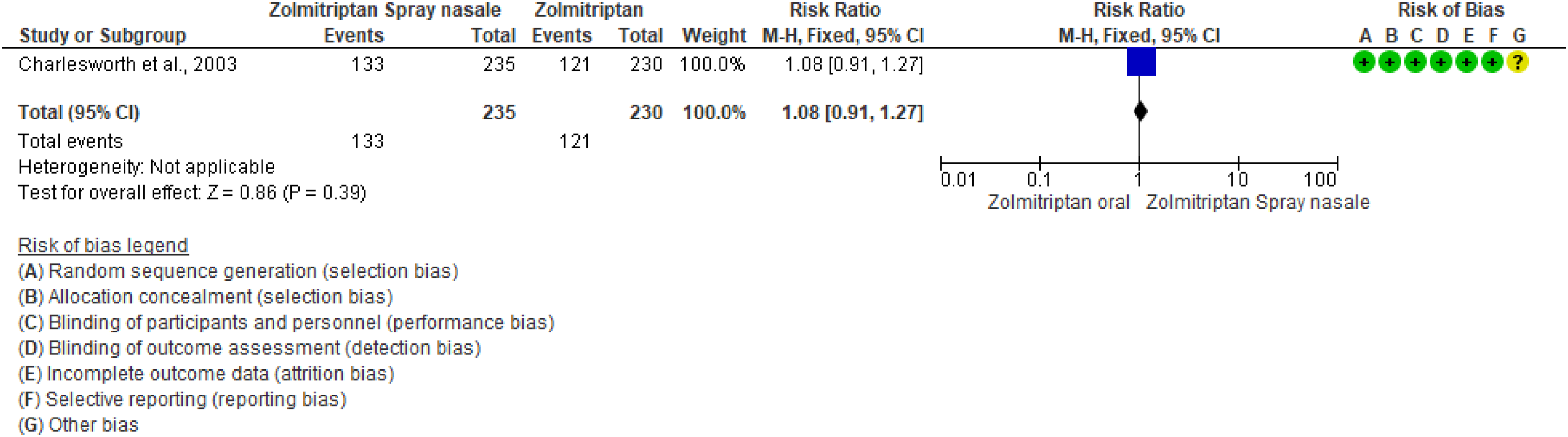
Forest plot showing the comparison between zolmitriptan 5 mg nasal spray and oral zolmitriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table of oral zolmitriptan 2.5 mg versus zolmitriptan 5 mg nasal spray in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendations for PICO 3.2.7 1) In subjects with migraine, we recommend oral zolmitriptan 2.5 mg for the acute treatment of migraine attacks. Strength of the recommendation: Strong (↑↑).
2) In subjects with migraine, we suggest against zolmitriptan 5 mg nasal spray for the acute treatment of migraine attacks. Strength of the recommendation: Weak (̄).
Quality of evidence: High (⊕⊕⊕⊕).
Quality of evidence: Moderate (⊕⊕⊕⊖).
Safety and tolerability of triptans
“Chest-related AEs”, also known as “triptan sensation”, are not related to myocardial ischemia according to aggregated data from trials, real world experiences and pharmacodynamic instrumental assessments (by electrocardiogram, myocardial scintigraphy and angiography) (210). Therefore, “chest-related AEs” characterized by chest pressure, chest pain, shortness of breath, palpitations, and anxiety, should be considered non-serious AEs. Among different triptans, almotriptan 12.5 mg showed the lowest incidence of ‘chest symptoms’ (58,59).
Although rare, it is worth mentioning ‘central nervous system AEs’ (asthenia, abnormal dreams, agitation, aphasia, ataxia, confusion, dizziness, somnolence, headache, speech disorder, thinking abnormal, tremor, vertigo, and other focal neurological symptoms) (211). The “central nervous system AEs” rates of incidence largely overlap among triptans with higher values for eletriptan 80 mg and lower values for almotriptan 12.5 mg (58,59).
Evidence-based guideline summary
We found several studies on triptans to be included for deriving evidence-based recommendations for this guideline. There is high quality of evidence to recommend the use of oral almotriptan 12.5 mg, eletriptan 20 and 40 mg, frovatriptan 2.5 mg, naratriptan 2.5 mg, rizatriptan 5 and 10 mg, sumatriptan 50 and 100 mg, zolmitriptan 2.5 mg and subcutaneous sumatriptan 6 mg for the acute treatment of migraine attacks. (Figure 3.2.34). Additionally, there was moderate quality of evidence to recommend oral naratriptan 1 mg and sumatriptan nasal spray 10 and 20 mg (Figure 3.2.34). On the other hand, evidence did not show benefits from zolmitriptan nasal spray and thus we suggest against its use (Figure 3.2.34).

Summary of evidence-based recommendations on triptans for the acute treatment of migraine attacks.
The decision on the triptan to choose may be guided by several factors including attack characteristics (i.e. rapid onset, severity, duration, presence of recurrence), associated symptoms, such as nausea or vomiting, drug availability and costs, or patients’ preference.
Expert-based opinion
Topic: When should triptans be taken during a migraine attack?
Topic: When should triptans be given during an attack of migraine with aura?
Several reports showing cerebral vasoconstriction characterizing hemiplegic migraine, migraine with brainstem aura (i.e., former basilar migraine or basilar-type migraine), and migraine with prolonged aura contraindicate triptans for these phenotypes based on the potential vasoconstrictive effects (219,223). However, recent case reports seem to suggest triptans effectiveness and safety when administered during attacks of hemiplegic migraine, migraine with brainstem aura and migraine with prolonged aura. Furthermore, although not specifically studied in RCTs, there is no evidence to date that triptans are unsafe for treatment of brainstem aura or hemiplegic migraine in clinical practice (219,224). However, more data are required to elucidate the efficacy, tolerability, and safety issues of triptans when administered in patients experiencing these peculiar aura phenomena.
Topic: Which formulation to choose?
Based on the fast absorption property, nasal sprays formulation of zolmitriptan or sumatriptan should also be considered in patients with rapid pain escalation during attacks or full-blown attack upon awakening (230). However, based on the fastest absorption as well as the highest bioavailability resulting in both more rapid action and greater efficacy compared to other triptans formulations (231).
Topic: What to do in case of headache relapse after initial response?
If relapses occur frequently, triptans with longer plasma half-lives (T1/2) such as eletriptan, naratriptan, or frovatriptan as well as combination of triptan and NSAID (due to synergistic/additive interaction) should be considered (85,234).
Topic: How many administrations of a triptan are needed to define whether or not a patient is a responder?
Topic: If a patient does not respond to a triptan, does it make sense to try a different triptan?
According to the Consensus of the EHF on the definition of effective treatment of a migraine attack and of triptan failure, patients are defined triptan-resistant when they do not respond to at least two different triptans, while patients not responding to at least three different triptans including sumatriptan subcutaneous formulation are defined triptan refractory. Patients can be considered non-responders to a specific triptan when effective treatment is observed in less than three out of four consecutive attacks (235).
Topic: If a triptan is not tolerated, is it reasonable to try another triptan?
Topic: How many triptans per month could be administered?
Topic: Can pharmacokinetic differences affect triptan choice?
Frovatriptan, naratriptan, and eletriptan are characterized by longer plasma half-lives (T1/2) compared to the other triptans, with longer duration of action leading to lower migraine relapse rate (59). Rizatriptan is characterized by a shorter time to peak plasma level (Tmax) compared to other triptans, suggesting a faster onset of action. Almotriptan and naratriptan are characterized by a higher oral bioavailability compared to other triptans, suggesting a more consistent efficacy over multiple attacks. Eletriptan is characterized by higher lipophilicity compared to other triptans, with higher potential to cross the blood-brain-barrier, which suggests central sites of action able to putatively increase the treatment efficacy as well as the risk of central nervous system adverse events (211).
Topic: Do triptans show relevant drug-to-drug interactions?
Topic: How should triptans be used to treat menstrual migraine?
Topic: Can triptans be used during pregnancy?
Topic: Can triptans be used during lactation?
Topic: Triptans and renal impairment
Topic: Triptans and hepatic failure
Topic: When should triptans be considered for the acute treatment of migraine attacks?
Introduction
Paracetamol, also known as acetaminophen, is one of the most frequently used over-the-counter medications for the treatment of acute pain, including headaches (256). It has a lower risk of gastric bleeding compared to other analgesics, even if at the expense of a risk for liver toxicity (257,258). Paracetamol is often recommended for pediatric migraine. Some clinicians also suggest the use of paracetamol for the treatment of acute migraine attacks during pregnancy, although the evidence on the safety of fetal exposure to the drug is conflicting (259–262).
Paracetamol has a weak anti-inflammatory action and the mechanisms by which it causes analgesia are not fully understood. Various proposed mechanisms include the inhibition of cyclooxygenases (24,263,264), the involvement of serotonergic pathways, and interactions with the endocannabinoid system (265).
This section systematically analyzes existing data on the efficacy of paracetamol, critically evaluating controlled studies that have explored its role in treating acute migraine attacks. The section is concerned with paracetamol alone, while combinations of paracetamol with other acute medication are reviewed in Section 3.4.
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 3.3.1.
Results
In total, we retrieved 1152 references for systematic reviews and meta-analyses. Following the removal of duplicates and completion of screening stages, we included 35 articles.
Eventually, we identified two meta-analyses as source of randomized controlled trials (RCTs) (24,264). After thorough analysis of the full texts of these RCTs, we included three of them in the qualitative synthesis (118,266,267) (Figure 3.3.1).

Meta-analysis selection flow chart - paracetamol.
In our search for RCTs, we initially retrieved 1134 references. After removing duplicates, 1026 references remained for analysis. Considering that the most recent included systematic review/meta-analysis on the topic was published in 2014, the analysis of additional RCTs was performed for papers published after that year, ultimately including no additional RCTs. From the literature search update performed in May 2023 and November 2023, we retrieved 115 references. No additional studies were included from this subsequent search (Figure 3.3.2).

RCTs selection flow chart - paracetamol. *trials published since 2014 were screened.
In total, three studies were included in the literature review and subsequent quantitative synthesis (meta-analyses) for the development of the evidence-based guideline. All the studies provided a comparison between an active drug and placebo, and all are discussed below.
Main evidence
We identified three RCTs (118,266,267) comparing oral paracetamol 1000 mg to placebo in subjects with migraine that met the criteria for main evidence, though they reported an unclear risk of bias (Figures 3.3.3 and 3.3.4).

Forest plot showing the comparison between oral paracetamol 1000 mg and placebo for the outcome pain freedom at 2 h in patients with migraine.
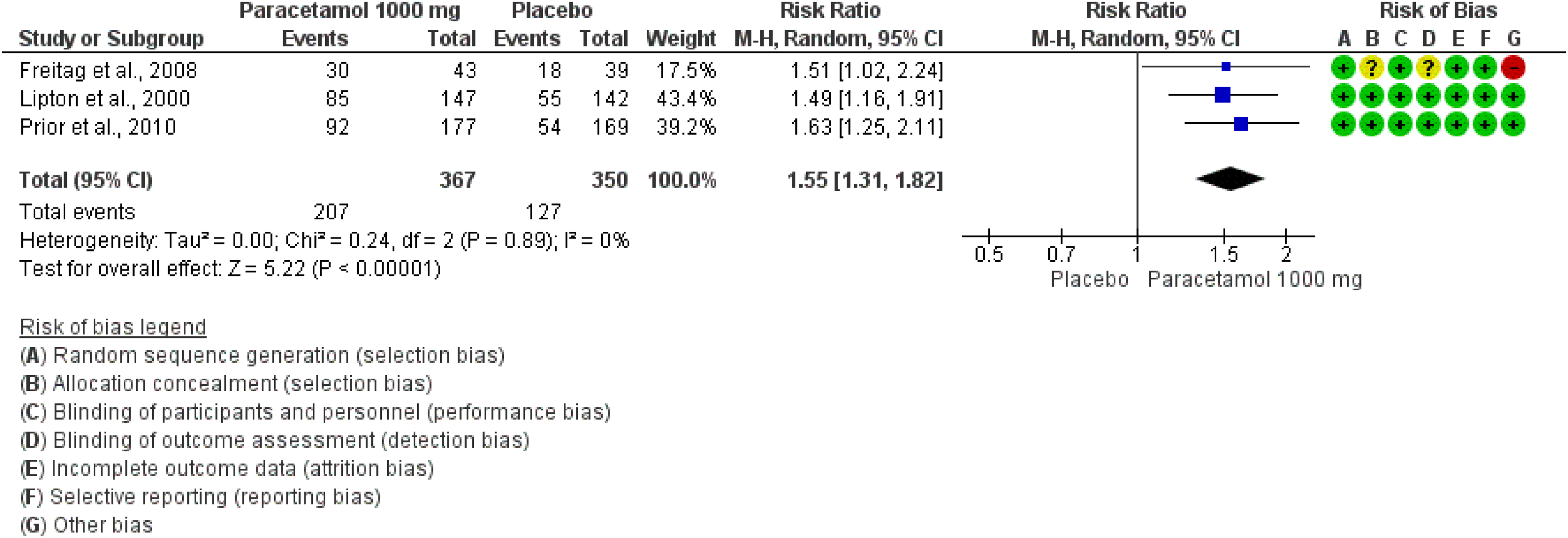
Forest plot showing the comparison between oral paracetamol 1000 mg and placebo for the outcome pain relief at 2 h in patients with migraine.
The pooled analysis of those three studies revealed a significant benefit of orally administered paracetamol (1000 mg) over placebo for both outcomes - pain freedom at 2 h and pain relief at 2 h. The quality of evidence for both outcomes was considered high (Table 3.3.1).
GRADE evidence profile table for oral paracetamol 1000 mg versus placebo

Additional evidence
None.
Evidence-based recommendation for PICO 3.3.1 In subjects with migraine, we recommend oral paracetamol 1000 mg for the acute treatment of migraine attacks.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Safety and tolerability of paracetamol
Paracetamol is generally considered safe for managing migraine in children despite the lack of specifically designed controlled trials. Regarding the use of paracetamol during pregnancy, literature is not concord in affirming its safety (270). Some – but not all – studies warn that paracetamol exposure during pregnancy might be associated with the risk of some neurodevelopmental and endocrine alterations in the newborn (262,271).
Expert-based opinions
Optimization of paracetamol use
Topic: Paracetamol 1000 mg vs Placebo
Introduction
The complex pathophysiological mechanisms of migraine underpin the variety of migraine symptoms including not only pain, but also nausea, photophobia, phonophobia and other autonomic symptoms. This may explain why none of the currently available monotherapies provides broad coverage of the multiple pathogenic processes in migraine (27,61,63,64,81,275–280). Combining drugs with different mechanisms of action and targeting different peripheral or central migraine mechanisms has a theoretical synergistic therapeutic effect. Furthermore, combining differently acting analgesics could lead to the use of a lower dosage of the individual drugs, with a lower expected incidence of side effects (281–284). Several drug combinations are commercially available, although their availability varies greatly from country to country in both composition and dosage. They include two-drug or three-drug combinations, such as aspirin, paracetamol, and caffeine (APC), oral metoclopramide plus aspirin, sumatriptan/naproxen and rizatriptan/paracetamol. The three-drug combination APC is the first over-the-counter (OTC) medication registered for acute migraine treatment in the United States (US) in 1998. As for the individual molecules, acetylsalicylic acid mechanisms of action in migraine are based on the inhibition of cyclooxygenase activity resulting in the platelet aggregation and plasma extravasation in the context of neurogenic inflammation, involving meningeal vessels, and its central action on the modulation of pain transmission (285–290). The analgesic property of the non-opioid, non-salicylate medication paracetamol may involve spinal and supraspinal actions, although the exact mechanism of action is unknown (291). On the other hand, caffeine is a methylxanthine used in several analgesic preparations because of its central cholinergic analgesia (292). Its antinociceptive effect is attributed to an inhibition of cyclooxygenase activity and adenosine receptor antagonism both at central and peripheral levels (293). It is used as an adjuvant in the treatment of both pain and headache (294,295). Caffeine alone shows analgesic properties in several clinical pain conditions (296). The observed synergism of acetylsalicylic acid, paracetamol, and caffeine on the inhibition of PGE 2 synthesis in microglial cells, a common model for the COX-2 inhibiting activity of non-steroidal anti-inflammatory drugs, may partly explain these effects (297). Furthermore, coadministration of caffeine and acetylsalicylic acid or other analgesics has been demonstrated to increase plasma acetylsalicylic acid concentration and the mechanism of this increase may involve plasma protein binding displacement (298). The dopamine receptor antagonist metoclopramide is indicated in many pathological conditions to treat nausea and vomiting including migraine (299). Metoclopramide in oral, suppository or parenteral form has been shown to improve gastrointestinal motility enhancing the absorption of other drugs and is effective in relieving nausea and vomiting frequently associated with migraine (300,301). Combinations containing oral metoclopramide plus an analgesic, in particular acetylsalicylic acid, may be a valid therapeutic option for patients when triptans are contraindicated or not tolerated or for those patients who frequently experience nausea and vomiting as associated symptoms (301). Notably, there is evidence suggesting that intravenous metoclopramide alone might be effective for migraine treatment in emergency department settings (302–304). Opioids in combination with analgesics are commonly prescribed for the management of acute migraine pain especially in the US (305–307). As far as the mechanism of action is concerned, tramadol specifically appears to reduce pain centrally binding weakly to μ-opioid receptors and inhibiting serotonin and norepinephrine reuptake, and codeine is considered a weak narcotic central analgesic (308).
The combination of ergotamine/caffeine is indicated to treat migraine and cluster headache attacks (309), in patients with severe symptoms or not responding to NSAIDs or other combination-analgesics (310). The main component of the combination is ergotamine tartrate, first used for migraine therapy in the 1920s. Its vasoconstrictor effect is due to its action on the smooth muscle in the arterial walls. It selectively binds to alpha-adrenergic receptors thereby stimulating vascular smooth muscle and causing vasoconstriction in both arteries and veins. Ergotamine also blocks serotonin 5-HT1B receptors, causing vasoconstriction in cerebral blood vessels thereby relieving the pain. It has high affinity for other serotonin receptors, including 5-HT1A, 5-HT2A, 5-HT2B and dopamine D2, but it has a central emetic effect (311). The theoretical basis for using ergotamine tartrate in combination with caffeine consists in the putative enhancement of its absorption (312). Combining drugs containing a triptan and a NSAID/analgesic agent is also an attractive option for the abortive treatment of migraine based on the potential synergy of these drugs both at the central and peripheral levels in the trigeminovascular system. Specifically, NSAIDs reduce meningeal inflammation potentially by inhibiting prostaglandin production. They also have been shown to block neurogenic dural plasma extravasation and trigeminal sensitization caused by CGRP-mediated dural vasodilatation (287). NSAIDs may also be beneficial in treating migraine with allodynia by preventing central sensitization (313,314). Among the NSAIDs, naproxen sodium has a superior safety profile with respect to cardiovascular risk and is an excellent NSAID for the treatment of migraine attacks. It suppresses sensitization of central trigeminovascular neurons in the spinal trigeminal nucleus in an animal model of intracranial pain (315). It has slower absorption but a much longer half-life than ibuprofen and diclofenac (27). Triptans act at the level of trigeminovascular system as agonists of the serotonin receptors (5-HT receptors) 1B/1D by blocking the transmission of signals to the trigeminal nucleus, thereby preventing peripheral sensitization which is involved in head pain maintenance (241). The combination of naproxen with triptans has been proven effective and well tolerated, also in migraine patients with cutaneous allodynia (316,317).
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for the Guideline on migraine drug combination for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 3.4.1.
We considered here as “analgesic” any drug that was intended for the acute treatment of migraine attacks, without selecting any specific classes of medication. As per the Methods (Section 2), we excluded intravenous formulations.
Results
Overall, we retrieved 249 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening, we included 19 systematic reviews and meta-analyses (19,20,57,84,283,284,305,318–329) as source of randomized controlled trials (RCTs) (Figure 3.4.1).

Meta-analysis selection flow chart - combination analgesics.
Overall, we retrieved 655 references from searching for RCTs and after removing duplicates we had 607 references to analyze. After screening stages and full text analysis, 33 studies were included in the literature synthesis to develop the evidence-based guideline (26,27,41,118,166,167,177,186,189,194,300,327,330–350). From the literature search update performed in May 2023 and November 2023, we retrieved 62 and 23 additional references, respectively. No further studies were included from the search updates (Figure 3.4.2).

RCTs selection flow chart - combination analgesics.
Among RCTs, 19 reported a comparison between an active combination analgesic and placebo and are presented in this section. The remaining 14 RCTs reported head-to-head comparisons and were included in Section 3.9.
Acetylsalicylic Acid + Paracetamol + Caffeine
Main evidence
We found two RCTs (335,340) addressing oral acetylsalicylic acid + paracetamol + caffeine as compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in main evidence (Figure 3.4.3).

Forest plot showing the comparison between oral acetylsalicylic acid 500 mg + paracetamol 500 mg + caffeine 130 mg and placebo for the outcome pain relief at 2h in patients with migraine.
The pooled analysis showed benefits of oral acetylsalicylic acid 500 mg + paracetamol 500 mg + caffeine 130 mg over placebo considering the outcome of pain relief at 2 h (Figure 3.4.3). The quality of evidence was considered high (Table 3.4.1).
GRADE evidence profile table for oral acetylsalicylic acid 500 mg + paracetamol 500 mg + caffeine 130 mg vs placebo in patients with migraine
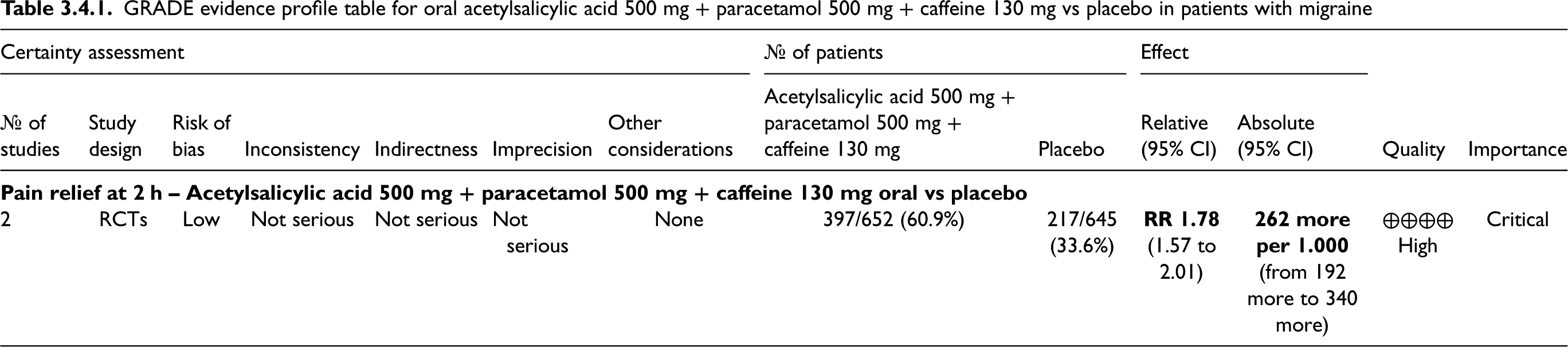
Additional evidence
We found one RCT addressing oral acetylsalicylic acid + paracetamol + caffeine as compared to placebo in the acute treatment of migraine attacks that did not meet the criteria to be included in main evidence (336).
The study showed benefits of oral acetylsalicylic acid 500 mg + paracetamol 500 mg + caffeine 130 mg over placebo considering the outcome of pain relief at 2 h but not the outcome of pain freedom at 2 h (Figure 3.4.4 and 3.4.5).

Forest plot showing the comparison between oral acetylsalicylic acid 500 mg + paracetamol 500 mg + caffeine 130 mg and placebo for the outcome pain freedom at 2h in patients with migraine.

Forest plot showing the comparison between oral acetylsalicylic acid 500 mg + paracetamol 500 mg + caffeine 130 mg and placebo for the outcomes pain relief at 2h in patients with migraine.
We found one RCT (350) addressing the efficacy of oral acetylsalicylic acid 250 mg + paracetamol 250 mg + caffeine 65 mg compared to placebo in the acute treatment of migraine attacks that did not meet the criteria to be included in the quantitative analysis as it was primarily meant to be a head-to-head study against ibuprofen 200 mg and did not consider the outcomes of interest for the present guideline.
The RCT showed benefits of acetylsalicylic acid 250 mg + paracetamol 250 mg + caffeine 65 mg over placebo considering pain intensity scores at 2, 3, and 4 h post-dose.
Evidence-based recommendation for PICO 3.4.1 In subjects with migraine, we recommend the oral combination of acetylsalicylic acid 500 mg + paracetamol 500 mg + caffeine 130 mg for the acute treatment of migraine attacks.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Safety and tolerability of acetylsalicylic acid + paracetamol + caffeine
Acetylsalicylic acid + metoclopramide
Main evidence
We found two RCTs addressing oral acetylsalicylic acid + metoclopramide as compared to placebo in the treatment of acute migraine attack (194,300).
The pooled analysis showed benefits of oral acetylsalicylic acid 900 mg + metoclopramide 10 mg over placebo considering the outcomes of pain freedom at 2 h and pain relief at 2 h (Figures 3.4.6 and 3.4.7). The quality of evidence for both outcomes was considered moderate (Table 3.4.2).

Forest plot showing the comparison between oral acetylsalicylic acid 900 mg + metoclopramide 10 mg and placebo for the outcome pain freedom at 2h in patients with migraine.

Forest plot showing the comparison between oral acetylsalicylic acid 900 mg + metoclopramide 10 mg and placebo for the outcome pain relief at 2h in patients with migraine.
GRADE evidence profile table for oral acetylsalicylic acid 900 mg + metoclopramide 10 mg versus placebo in patients with migraine

Additional evidence
None.
Evidence-based recommendation for PICO 3.4.2 In subjects with migraine, we recommend the oral combination of acetylsalicylic acid 900 mg + metoclopramide 10 mg for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Strong (↑↑)
Safety and tolerability of acetylsalicylic acid + metoclopramide
Paracetamol + tramadol
Main evidence
We found one RCT addressing oral paracetamol + tramadol as compared to placebo in the acute treatment of migraine attack (347). The study showed benefits of oral paracetamol 650 mg + tramadol 75 mg over placebo considering the outcomes of pain freedom at 2 h and pain relief at 2 h (Figures 3.4.8 and 3.4.9). The quality of evidence for both outcomes was considered low (Table 3.4.3).
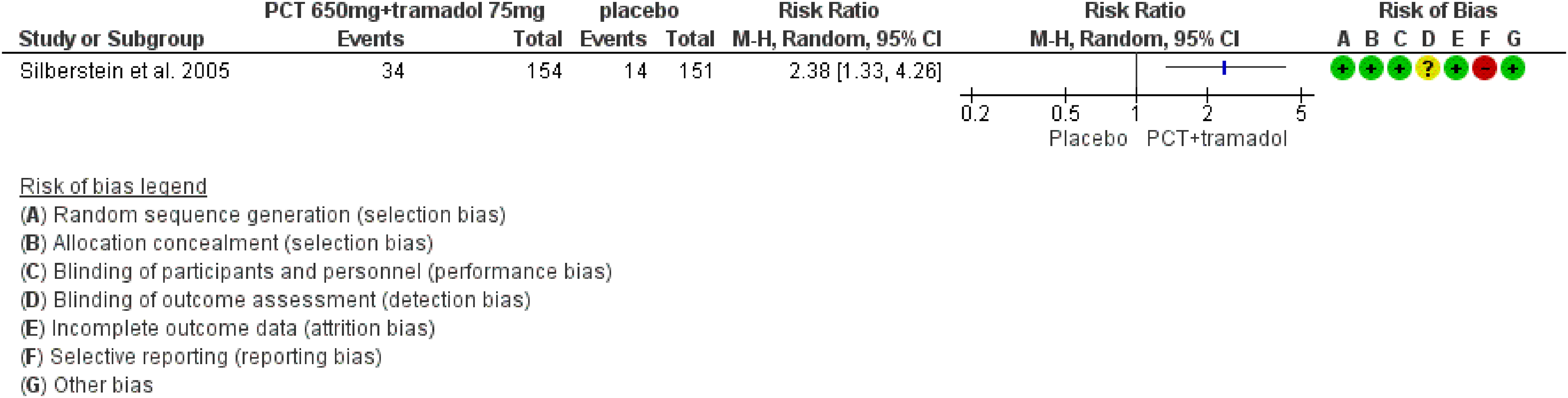
Forest plot showing the comparison between oral paracetamol 650 mg + tramadol 75 mg and placebo for the outcome pain freedom at 2h in patients with migraine.

Forest plot showing the comparison between oral paracetamol 650 mg + tramadol 75 mg and placebo for the outcome pain relief at 2h in patients with migraine.
GRADE evidence profile table for oral paracetamol 650 mg + tramadol 75 mg versus placebo in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.4.3 In subjects with migraine, we recommend the oral combination of paracetamol 650 mg + tramadol 75 mg for the acute treatment of migraine attacks. Quality of evidence: Moderate (⊕⊕⊕⊖). Strength of the recommendation: Weak (↑).
[NOTE: Considering the risk associated with the class of opioids, their use in the acute treatment of migraine should be considered only when all the other available options have proved ineffective.]
Safety and tolerability of paracetamol + tramadol
Paracetamol + codeine
Main evidence
None.
Additional evidence
We found one RCT comparing oral paracetamol + codeine to placebo in the acute treatment of migraine attacks (26). This RCT did not meet the criteria for main evidence. The quality of evidence was considered very low.
The study showed benefits of oral paracetamol 400 mg + codeine 25 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.4.10) and pain relief at 2 h (Figure 3.4.11).
Evidence-based recommendation for PICO 3.4.4 In subjects with migraine, we suggest the oral combination of paracetamol 400 mg + codeine 25 mg for the acute treatment of migraine attacks. Quality of evidence: Very low (⊕⊖⊖⊖). Strength of the recommendation: Weak (↑).
[NOTE: Considering the risk associated to the class of opioids, their use in the acute treatment of migraine should be considered only when all the other available options have proved ineffective.]

Forest plot showing the comparison between oral paracetamol 400 mg + codeine 25 mg and placebo for the outcome pain freedom at 2h in patients with migraine.

Forest plot showing the comparison between oral paracetamol 400 mg + codeine 25 mg and placebo for the outcome pain relief at 2h in patients with migraine.
Safety and tolerability of paracetamol + codeine
Sumatriptan + naproxen
Main evidence
We found four RCTs (327,332,341,346) addressing oral sumatriptan + naproxen as compared to placebo in the acute treatment of migraine attacks.
The pooled analysis showed benefits of oral sumatriptan 85 mg + naproxen 500 mg versus placebo considering the outcome of pain freedom at 2 h; more limited evidence coming from two RCTs did not show benefits considering the outcome of pain relief at 2 h (Figure 3.4.12 and 3.4.13). The quality of evidence for both outcomes was considered low (Table 3.4.5).

Forest plot showing the comparison between oral sumatriptan 85 mg + naproxen 500 mg and placebo for the outcome pain freedom at 2h in patients with migraine.

Forest plot showing the comparison between oral sumatriptan 85 mg + naproxen 500 mg and placebo for the outcome pain relief at 2h in patients with migraine.
GRADE evidence profile table for oral sumatriptan 85 mg + naproxen 500 mg versus placebo in migraine

Very different results across trials; bconfidence interval included the threshold for clinical decision.
Additional evidence
We found three RCTs (27,41,186) addressing oral sumatriptan + naproxen as compared to placebo in the acute treatment of migraine attacks that did not meet the criteria to be included in main evidence. The overall risk of bias was considered unclear, and the quality of evidence was considered very low.
One RCT (41) showed benefits of oral sumatriptan 50 mg + naproxen 500 mg over placebo considering the outcome pain freedom and pain relief at 2 h; the other two RCTs (27,186) showed benefits of oral sumatriptan 85 mg + naproxen 500 mg versus placebo in the outcomes pain freedom and pain relief at 2 h (Figures 3.4.14 and 3.4.15).
Evidence-based recommendation for PICO 3.4.5 In subjects with migraine, we recommend the oral combination of sumatriptan 85 mg + naproxen 500 mg for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Strong (↑↑)

Forest plot showing the comparison between oral sumatriptan 50 mg + naproxen 500 mg and placebo, or sumatriptan 85 mg + naproxen 500 mg and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral sumatriptan 50 mg + naproxen 500 mg versus placebo, or sumatriptan 85 mg + naproxen 500 mg versus placebo for the outcome pain relief at 2 h in patients with migraine.
Safety and tolerability of sumatriptan + naproxen
Rizatriptan + paracetamol
Main evidence
We found one RCT (118) addressing oral rizatriptan + paracetamol as compared to placebo in the acute treatment of migraine attacks that met the criteria for main evidence.
The study showed benefits of oral rizatriptan 10 mg + paracetamol 1000 mg over placebo considering the outcomes of pain freedom at 2 h and pain relief at 2 h (Figure 3.4.16 and 3.4.17). The quality of evidence for both outcomes was considered low (Table 3.4.6).

Forest plot showing the comparison between oral rizatriptan 10 mg + paracetamol 1000 mg and placebo for the outcome pain freedom at 2h in patients with migraine.

Forest plot showing the comparison between oral rizatriptan 10 mg + paracetamol 1000 mg and placebo for the outcome pain relief at 2h in patients with migraine.
GRADE evidence profile table for oral rizatriptan 10 mg + paracetamol 1000 mg versus placebo in patients with migraine
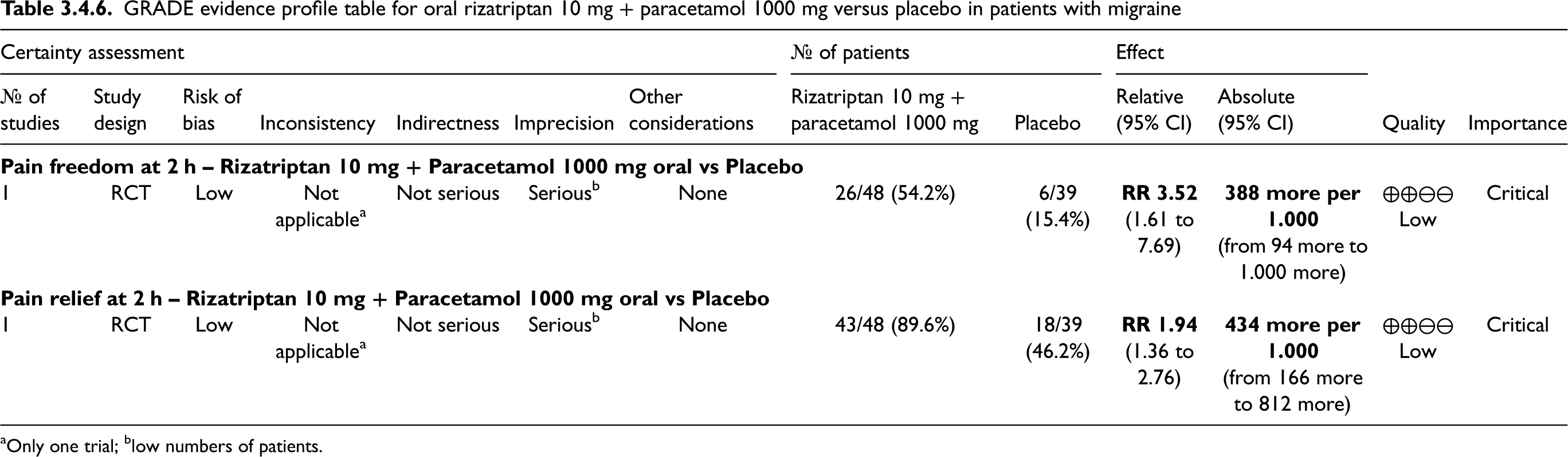
Only one trial; blow numbers of patients.
Despite the low quality of evidence and the low number (<100) of subjects included in the trial, we nevertheless provided a strong recommendation for the use of rizatriptan 10 mg + paracetamol 1000 mg, as both drugs alone were associated with high efficacy over placebo (see Sections 3.2 and 3.3).
Additional evidence
None.
Evidence-based recommendation for PICO 3.4.6 In subjects with migraine, we recommend the oral combination of rizatriptan 10 mg + paracetamol 1000 mg for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Strong (↑↑)
Safety and tolerability of rizatriptan + paracetamol
Ergotamine + caffeine
Main evidence
None.
Additional evidence
We found one RCT comparing ergotamine + caffeine to placebo in the acute treatment of migraine attacks (167).
The study did not show benefits of oral ergotamine 2 mg + caffeine 200 mg over placebo considering the outcome pain freedom at 2 h, whereas it showed significant benefit in the outcome pain relief at 2 h (Figure 3.4.18 and 3.4.19). The quality of evidence was downgraded to very low due to the imprecision of results (the clinical decision threshold was included in the confidence interval).
Evidence-based recommendation for PICO 3.4.7 In subjects with migraine, we suggest the oral combination of ergotamine 2 mg + caffeine 200 mg for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)

Forest plot showing the comparison between oral ergotamine 2 mg + caffeine 200 mg and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral ergotamine 2 mg + caffeine 200 mg and placebo for the outcome pain relief at 2 h in patients with migraine.
Safety and tolerability of ergotamine + caffeine
Common side effects of the combination of ergotamine + caffeine are nausea and vomiting after both oral and parenteral administration, due to the effect on emetic centers. Insomnia, dizziness cramps and transient lower limb muscle pain have also been described (378). A broad range of side effects can also be attributed to ergotamine because this drug is not specific for the 5HT-1B/1D serotonin receptors. They include chest pain, discomfort, or tightness, cold hands/feet, muscle pain/weakness, numbness/tingling or a bluish color skin of the fingers or toes, continuing loss of appetite, increased frequency of urination, irregular heartbeat, and back pain (379).
Considering the poor safety and tolerability profile of ergotamine and the availability of other safer vasoconstrictive agents for the acute treatment of migraine, combinations containing ergotamine should be proposed only when other options have been tried and failed.
Butalbital + paracetamol + caffeine
Main evidence
We found one RCT (332) addressing oral butalbital 50 mg + paracetamol 325 mg + caffeine 40 mg as compared to placebo in the acute treatment of migraine attacks. The analysis showed no benefit of oral butalbital + paracetamol + caffeine versus placebo considering the outcome pain freedom at 2 h (Figure 3.4.20). The quality of evidence was considered moderate due to the availability of only one RCT (Table 3.4.7).

Forest plot showing the comparison between oral butalbital 50 mg + paracetamol 325 mg + caffeine 40 mg and placebo for the outcome pain freedom at 2 h in patients with migraine.
GRADE evidence profile table for oral butalbital 50 mg + paracetamol 325 mg + caffeine 40 mg versus placebo in migraine
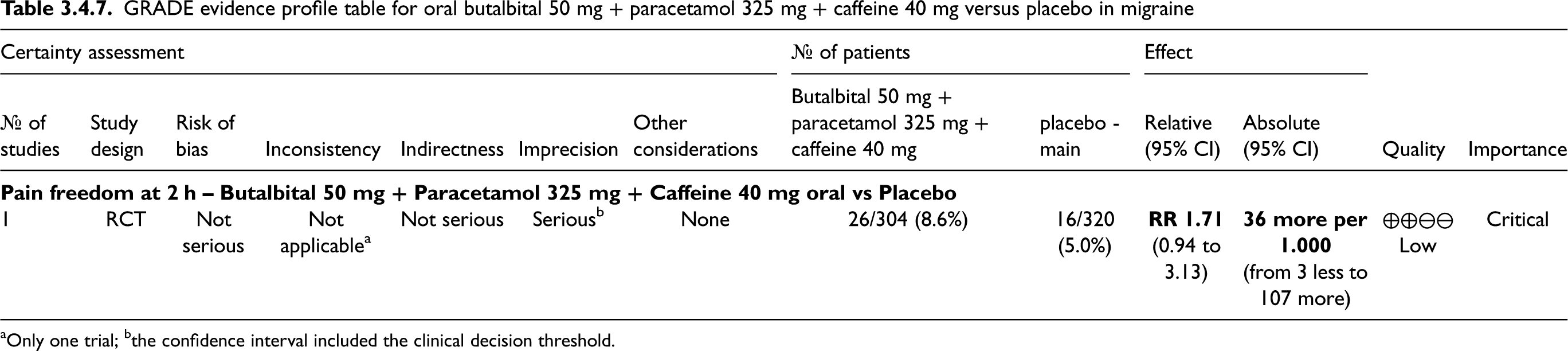
Only one trial; bthe confidence interval included the clinical decision threshold.

Summary of evidence-based recommendations on combinations drugs for the treatment of the acute migraine attack.
Additional evidence
None.
Evidence-based recommendation for PICO 3.4.8 In subjects with migraine, we suggest against the oral combination of butalbital 50 mg + paracetamol 325 mg + caffeine 40 mg for the acute treatment of migraine attacks. Quality of evidence: Low (⊕⊕⊖⊖).
Strength of the recommendation: Weak (↓)
Safety and tolerability of butalbital + paracetamol + caffeine
Evidence-based guideline summary
We found two RCTs (335,340) addressing oral acetylsalicylic acid + paracetamol + caffeine as compared to placebo in the acute treatment of migraine attacks that met the pre-defined criteria to be included in main evidence. The pooled analysis showed benefits of oral acetylsalicylic acid 500 mg + paracetamol 500 mg + caffeine 130 mg over placebo considering the outcome of pain relief at 2 h (Figure 3.4.3). The quality of evidence was considered moderate (Table 3.4.1). The strength of the recommendation was considered strong in favor.
We found two RCTs addressing oral acetylsalicylic acid + metoclopramide as compared to placebo in the acute treatment of migraine attacks (194,300). The pooled analysis showed benefits of oral acetylsalicylic acid 900 mg + metoclopramide 10 mg over placebo considering the outcomes of pain freedom at 2 h and pain relief at 2 h (Figures 3.4.6 and 3.4.7). The quality of evidence for both outcomes was considered moderate (Table 3.4.3). The strength of the recommendation was considered strong in favor.
We found one RCT addressing oral paracetamol + tramadol as compared to placebo in the acute treatment of migraine attacks (347). The study showed benefits of oral paracetamol 650 mg + tramadol 75 mg over placebo considering the outcomes of pain freedom at 2 h and pain relief at 2 h (Figures 3.4.8 and 3.4.9). The quality of evidence for both outcomes was considered low. The strength of the recommendation was considered strong in favor.
We found one RCT comparing oral paracetamol + codeine to placebo in the acute treatment of migraine attacks (26). This RCT did not meet most of the pre-defined quality criteria and for this reason the quality of evidence was considered very low. The study showed benefits of oral paracetamol 400 mg + codeine 25 mg over placebo considering the outcomes of pain freedom at 2 h and pain relief at 2 h (Figure 3.4.10). The quality of evidence was considered very low. The strength of the recommendation was considered weak in favor. The combination should not be considered as a first choice as we need to consider the potential risk associated with the use of medications containing opioids.
We found four RCTs (327,332,341,346) addressing oral sumatriptan + naproxen as compared to placebo in the acute treatment of migraine attacks. The pooled analysis showed benefits of oral sumatriptan 85 mg + naproxen 500 mg versus placebo considering the outcome pain freedom at 2 h; more limited evidence coming from two RCTs (27,327) did not show benefits considering the outcome pain relief at 2 h (Figure 3.4.12 and 3.4.13). The quality of evidence for both outcomes was considered high (Table 3.4.5). The strength of the recommendation was considered weak in favor.
We found one RCTaddressing oral rizatriptan + paracetamol as compared to placebo in the acute treatment of migraine attacks. This RCT did not meet all the pre-defined quality criteria. The study showed benefits of oral rizatriptan 10 mg + paracetamol 1000 mg over placebo considering the outcomes of pain relief at 2 h and pain freedom at 2 h (Figure 3.4.14 and 3.4.15). The quality of evidence for both outcomes was considered low (Table 3.4.6). The strength of the recommendation was considered strong in favor.
We found one RCTcomparing ergotamine + caffeine to placebo in the treatment of acute migraine attack. The study did not show benefits of oral ergotamine 2 mg + caffeine 200 mg over placebo considering the outcome pain freedom at 2 h, whereas it showed significant benefit in the outcome of pain relief at 2 h (Figure 3.4.18 and 3.4.19). The quality of evidence for both outcomes was considered low. The strength of the recommendation was considered weak in favor.
We found one RCTcomparing butalbital + paracetamol + caffeine to placebo in the acute treatment of migraine attacks. The study did not show benefits of butalbital 50 mg + paracetamol 325 mg + caffeine 40 mg over placebo considering the outcome of pain freedom at 2 h (Figure 3.4.20). The quality of evidence was considered moderate (Table 3.4.7). The strength of the recommendation was considered weak against.
Expert-based opinion
Topic: When should a combination analgesic be taken during a migraine attack?
Oral combination of ergotamine + caffeine can be used when rizatriptan or sumatriptan alone or in combination with naproxen or paracetamol are ineffective or only partially effective (309).
Topic: When should a combination analgesic drug be taken during an attack of migraine with aura?
Topic: What to do in case of headache relapse after initial response to a combination analgesic?
Topic: How many doses of combination analgesics could be administered per month?
Codeine/tramadol-containing analgesics have a great potential of misuse/overuse or physical and psychological dependence. In particular, codeine is metabolized in the liver like morphine, at a rate which varies consistently among individuals, with quick metabolizers having a higher risk of overuse and addiction (388). Combination drugs containing codeine have been found to increase the risk of MOH in comparison with single analgesics (389). Drugs containing codeine are therefore not recommended for chronic migraine because of the limited clinically relevant improvement in terms of sustained pain reduction and functioning recovery due to codeine in addition to paracetamol compared to paracetamol alone, and in the long run, the risk of dependence outweighs benefits. This is also true for combination drugs containing tramadol (390).
Ergot derivatives and caffeine-containing products increase the risk of transition from episodic to chronic migraine, particularly for those subjects with high headache frequency. It is generally accepted that ergot derivatives—which are marketed in combination with caffeine—exert a relevant influence on the development of MOH, and in recent years, their use has been restricted to those migraine patients with severe but low-frequency attacks (391).
Topic: Can pharmacokinetic and pharmacodynamic differences affect the choice of combination analgesics?
Codeine, in combination with other analgesics such as paracetamol, has a greater analgesic effect than the individual components and a longer lasting efficacy. For combination drugs containing codeine and tramadol, weak metabolizers and ultra-fast metabolizers have been identified, the former having a reduced analgesic effect, the latter an increased risk of side effects due to the two weak opioids. The combination of paracetamol and tramadol demonstrated genuine synergy and integrates paracetamol's rapid onset of efficacy with tramadol's prolonged analgesic effect. Combinations containing acetylsalicylic acid, paracetamol and caffeine have the potential of a greater efficacy than the single components with caffeine improving the paracetamol enteric absorption. Paracetamol plus tramadol has comparable efficacy to paracetamol plus codeine, but with reduced somnolence and constipation compared with the codeine combination (392,393).
Caffeine combined with ergotamine can increase the enteric absorption of ergotamine and contribute synergistically to the constriction of cerebral vasculature, thereby providing better efficacy than ergotamine alone and a valid alternative for triptans when they are ineffective (309).
Metoclopramide with his prokinetic action favors acetylsalicylic acid absorption and this is particularly useful for migraine attacks with prevalent nausea and vomiting.
The combination of rizatriptan with paracetamol or of sumatriptan with naproxen did not imply differences in the pharmacodynamic and pharmacokinetic properties of the single drugs in the combination.
Acetylsalicylic acid + paracetamol + caffeine combination
In primary microglial cells both paracetamol and caffeine dose-dependently inhibited microglial PGE-2 synthesis. In combination with acetylsalicylic acid both substances augmented the inhibitory effect of acetylsalicylic acid on LPS-induced PGE-2 synthesis. Whereas paracetamol inhibited only COX enzyme activity, caffeine also inhibited COX-2 protein synthesis. These results suggest that the clinical activity of paracetamol and caffeine is due to the inhibition of COX and support the clinical experience of an adjuvant analgesic effect of caffeine and paracetamol when combined with acetylsalicylic acid (297).
Additionally, in a pain-induced functional impairment model in rats, caffeine significantly increased the analgesic effect of aspirin at all doses, without modifying aspirin, salicylic acid, or gentisic acid plasma levels. It is concluded that caffeine potentiates the analgesic effect of aspirin by a pharmacodynamic, but not by a pharmacokinetic mechanism (395).
Caffeine did not affect the pharmacokinetics of ASA and paracetamol when used as an adjuvant in ASA/paracetamol fixed-dose combination under fasting conditions, suggesting that caffeine enhances the analgesic efficacy of these drugs by pharmacodynamic rather than pharmacokinetic interactions (399).
Paracetamol + codeine combination
More recent findings showed that codeine activity seems to be related not only to its conversion to morphine but also by itself and through its metabolites, such as norcodeine (NORC) and codeine-6-glucuronide (C-6-G). The addition of codeine to paracetamol significantly improves the analgesic action and reduces the number needed to treat (NNT) from 5 to 2.3–3.1 (401).
Paracetamol + tramadol combination
Acetylsalicilic acid + Metoclopramide combination
Metoclopramide stimulates and coordinates the motility of the upper gastrointestinal tract, without changing gastric, pancreatic and biliary secretion and has an antiemetic effect. Its mechanism of action is complex, being a competitive antagonist of D1 and D2 dopamine receptors and 5-HT3 serotonin receptors, as well as a nonspecific agonist of 5-HT4 receptors involved in the stimulation of enteric cholinergic neurons. It therefore has intestinal prokinetic activity which results in accelerated gastric emptying and decreased reflux from the duodenum into the stomach and esophagus (299). In an animal model metoclopramide was unable to affect the peripheral response to trigeminovascular activation (neurogenic inflammation), but it did suppress the central response (the trigeminal cervical complex neuronal firing) after dural electrical stimulation, which suggests a possible anti-migraine action of the drug (415).
Sumatriptan + naproxen combination
Rizatriptan + paracetamol combination
Ergotamine + caffeine combination
Topic: Do combination analgesics show relevant drug interactions?
Acetylsalicylic acid
Naproxen
Paracetamol
Caffeine
Metoclopramide
Codeine and tramadol
Sumatriptan and rizatriptan
Ergotamine
Topic: Can combination analgesics be used during pregnancy?
Aspirin, as well as NSAIDs, usually are not recommended during pregnancy. These drugs should be avoided during pregnancy due to the fetal risks such as renal injury, oligohydramnios, constriction of the ductus arteriosus (with potential for persistent pulmonary hypertension in the newborn), necrotizing enterocolitis, and intracranial hemorrhage (461).
A limited use of caffeine is recommended during pregnancy; therefore, compounds containing caffeine should preferentially be avoided. Indeed, observational studies and meta-analyses suggest an increased risk from maternal caffeine consumption for miscarriage, stillbirth, lower birth weight and/or small for gestational age (462).
The use of triptans and other drugs exhibiting vasoconstrictive properties in pregnant women requires a careful benefits/risks evaluation. There is no data to suggest teratogenicity for any of the triptans, although the most common adverse events recorded with available registries are spontaneous abortion, preterm delivery and low birth weight (463,464). These findings underline the need for a careful evaluation of the potential serious adverse effects of triptans and their potential impact on pregnancy outcome (463). This observation should be extended to the combination drugs containing triptans. Ergotamine is contraindicated in women who are or may become pregnant, since the drug may cause fetal harm. Its use during pregnancy is associated with low birthweight and/or preterm birth which may relate to ergotamine-induced vasoconstriction in the placenta of pregnant women. Ergotamine may cause acute fetal hypoxia and myocardial ischemia. Based on these observations, ergotamine-containing compounds should be avoided (465,466).
Topic: Can combination analgesics be used during lactation?
Caffeine is not harmful for breastfed babies but can make them irritable and may increase the baby's heart and breathing rates.
Therefore, many combination drugs can be used in limited doses during breast-feeding with the exclusion of combinations containing codeine or tramadol.
Compounds containing ergotamine are contraindicated during lactation.
Topic: combination-analgesics and renal impairment
Topic: Combination-analgesics and hepatic failure
Introduction
Antiemetics are drugs currently used in several diseases in order to treat nausea and vomiting, belonging to different classes including antagonists of dopamine, serotonin, histamine, muscarinic and neurokinin systems, benzodiazepines and corticosteroids (486). During the acute phase of a migraine attack, antiemetics can relieve nausea and vomiting, and lead to a reduction in pain (24,486). Antiemetics are often considered the treatment of choice in the emergency room setting, where parenteral routes of administration are often used (304).
Dopamine antagonists, such as metoclopramide, prochlorperazine, domperidone, and chlorpromazine are the mainstay of migraine-related nausea treatment, although 5-HT3 receptor antagonists (e.g., ondansetron) are also used in the emergency room, especially in the pediatric population. Within the class of antiemetics, metoclopramide and prochlorperazine are dopamine D2 receptor blockers that have the greatest evidence for efficacy in migraine. Metoclopramide exhibits both peripheral and central actions, demonstrating efficacy in reducing nausea and migraine pain, while also enhancing the impact of concurrent analgesics (487,488). Although domperidone and chlorpromazine are good antiemetics, the evidence for their effect in migraine treatment is weaker (489–491).
Antiemetic agents are generally safe and well-tolerated, but they might be associated with adverse events, including extrapyramidal symptoms – such as dystonia and akathisia – and QT interval prolongation (486).
We performed a systematic review and meta-analysis of randomized controlled trials (RCTs) to evaluate the efficacy of antiemetics compared with placebo for the acute treatment of migraine in adults. The primary endpoints in most studies were the number of patients with headache resolution or with reduced headache severity, while the secondary outcome was improvement in quality of life.
Section-specific methods
This section followed the general procedure to develop this guideline (see Section 2).
Search strings for the guideline on antiemetics for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 3.5.1.
Results
The database search for systematic reviews and meta-analyses resulted in a total of 1042 hits (Figure 3.5.1). After duplicate removal and screening 990 titles and abstracts, one systematic review and meta-analysis was considered as source of RCTs (24) after analyzing full texts of these RCTs, we included no RCTs in the quantitative synthesis (Figure 3.5.1).

Meta-analysis selection flow chart - antiemetics.
Overall, we retrieved 1315 references from searching for RCTs in the context of antiemetics and migraine, and after removing duplicates, we had 1100 references to screen. However, considering the most recent included systematic review and meta-analysis on the topic (24), the analysis of additional RCTs was performed for papers published since 2021 (101 references) (Figure 3.5.2).

RCTs selection flow chart. *trials published since 2021 were screened.
Overall, no studies were included in the qualitative or quantitative synthesis (meta-analyses) to develop the evidence-based guideline.
Expert-based opinions
There is a lack of good quality RCTs on acute pharmacological treatment of migraine with antiemetics. Available evidence mostly focused on dopaminergic antagonists, like metoclopramide and prochlorperazine. Therefore, while antiemetics are commonly utilized in the emergency room or at home to manage nausea associated with migraine attacks (487), their use as a standalone acute migraine treatment is discouraged due to the poor quality of published studies (488,489). Of note, a comparative study conducted on a small population suggested that a buccal formulation of prochlorperazine 3 mg produced a faster and more marked efficacy than oral or buccal placebo, or oral ergotamine tartrate 1 mg + caffeine 100 mg (489). In another study metoclopramide was more effective than placebo in relieving nausea, but not migraine pain (488).
Though some reports suggest that antiemetics may alleviate migraine pain, they are indicated for the symptomatic treatment of nausea and vomiting in association to standard acute therapy for migraine attacks (see Section 3.4 on combination analgesics). They can be used alone only when acetaminophen, NSAIDs and migraine-specific drugs for the acute treatment of attacks are ineffective or contraindicated (488).
It is important to keep in mind that antiemetics can also result in side effects such as extrapyramidal symptoms (e.g., akathisia), hypotension, and arrhythmias, including Torsade de Pointes and QT interval prolongation (486).
Overall, antiemetics may thus be a viable alternative for patients who experience intense nausea/vomiting during migraine attacks and are resistant to or have contraindications to acetaminophen, NSAIDs and migraine specific acute treatments (488,489).
Introduction
Opioids are semi-synthetic and synthetic substances that act on opioid receptors to produce morphine-like effects. These receptors are found principally in the central and peripheral nervous system, in the gastrointestinal tract. Their stimulation induces somatic and psychoactive effects, reduces pain, and suppresses cough and diarrhea (492). In the headache field, opioids are potentially useful thanks to their ability to promote immediate relief of moderate to severe acute pain (493). On the other hand, their persistent use is associated with long-term opioid consequences such as dependence and need of increasing dosage to obtain pain relief; this may also lead to life-threatening overdose (494). Over the last decades, the increased prescription of this class of drugs has led to widespread misuse of both prescription and non-prescription opioids. The soaring increase in opioid prescriptions and misuse in the United States (US) contributed to the so-called “opioid crisis” in North America (495) and was a driver for the onset of the first wave of the epidemic of drug overdose deaths. These alarming phenomena led to an update of the Canadian guideline on opioids for chronic non-cancer pain (CNCP) (496) and to a guideline from the United States (US) Centers for Disease Control (CDC) (497), which focused on harm reduction in relation to opioid prescribing for CNCP.
A large number of randomized placebo-controlled trials (RCTs) were conducted to test the efficacy of opioids in the treatment of CNCP. These have provided evidence of a modest efficacy in pain relief and on the improvement of physical functioning, while underlining clinically relevant adverse events (498).
In the migraine field, the extent of use of opioids has been recently assessed by a web-based survey conducted in a large representative US population (ObserVational survey of the Epidemiology, tReatment and Care of MigrainE [OVERCOME]) (499). The outcome of the survey shows that among the 12,299 patients with 0–3 monthly headache days (MHDs), 59.0% never used opioids, 26.0% formerly used them, and 15.0% were current opioid users. When considering the 8844 patients with ≥4 MHDs, 44.9% never used opioids, 31.2% formerly used them, and 23.9% were current opioid users.
Although the situation in European countries is far less critical than the opioid crisis observed in North America (500), there are increasing concerns about the safety of long-term opioid therapy for patients with CNCP (501). Therefore, the European Pain Federation released the European clinical practice recommendations on opioids for CNCP, which were endorsed by several other European scientific associations, including the European Academy of Neurology (EAN) and the European Headache Federation (EHF) (502). Notably, in several chronic pain conditions, opioids are considered neither a universal cure nor a universally dangerous threat. They can only be used for some selected chronic noncancer pain syndromes, but it is strongly recommended to consider them only if established non-pharmacological and pharmacological treatment options have failed. In the acute setting, opioids are often prescribed for immediate postoperative pain and other painful conditions for patients in the emergency department and primary care settings. In this framework, opioids should be prescribed only when necessary, at the lowest effective dose, and for the shortest period required (497).
Section-specific methods
Search strings for the Guideline on opiates for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 3.6.1.
Results
Overall, we retrieved 1096 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included one systematic review with meta-analysis (24) that was considered as the source of randomized controlled trials (RCTs). After analyzing full texts of these RCTs, we included one of them in the quantitative synthesis (503) (Figure 3.6.1).

Meta-analysis selection flow chart - opioids.
Overall, we retrieved 719 references from searching for RCTs and after removing duplicates we had 693 references to analyze. However, considering the most recent included systematic review with meta-analysis on the topic (24), the analyses of additional RCTs were performed for papers published since 24 February 2021 and did not result in the inclusion of any additional RCTs (Figure 3.6.2).
RCTs selection flow chart - opioids. *trials published since February 24, 2021 were screened.
Overall, one study was included in the quantitative synthesis (meta-analyses) to develop the evidence-based guideline (503). It reported a comparison between an active drug and placebo and was presented in this paper.
Butorphanol
Main evidence
None.
Additional evidence
We found one RCT comparing butorphanol to placebo in the acute treatment of migraine attacks (503) that did not meet the criteria to be included in the main evidence. The risk of bias of this study was very serious and the overall quality of evidence was further downgraded to very low (Figures 3.6.3 and 3.6.4).

Forest plot showing the comparison between butorphanol 1 mg intranasal vs placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between butorphanol 1 mg intranasal vs placebo for the outcome pain relief at 2 h in patients with episodic migraine.
The study showed benefits of intranasal butorphanol 1 mg over placebo considering the outcomes pain freedom at 2 h (Figure 3.6.3) and pain relief at 2 h (Figure 3.6.4).
Evidence-based recommendation for PICO 3.6.1 In patients with migraine, we suggest intranasal butorphanol 1 mg for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)
[NOTE: Considering the risk associated with the class of opioids, their use in the acute treatment of migraine should be considered only when all the other available options have proved ineffective.]
Safety and tolerability of opioids
Evidence-based guideline summary
We found only one study on butorphanol for deriving evidence-based recommendations for this guideline. There is low quality of evidence to suggest the use of intranasal butorphanol for the acute treatment of migraine attacks. (Figure 3.6.5).

Summary of evidence-based recommendations on opioids for the acute treatment of migraine attacks.
Expert-based opinions
Topic: Opioids in the acute treatment of migraine attacks
Before considering a trial with opioids, we suggest optimization of nonopioid pharmacotherapy and non-pharmacologic therapy. By a trial with opioids, we mean initiation, titration, and monitoring of response, with discontinuation of opioids if a clinically meaningful improvement in pain or function is not achieved. Opioids should never be used in patients with migraine with concurrent or past substance use disorder or active psychiatric disorders and in patients with MOH.
Introduction
Lasmiditan is a first-in-class ditan developed as an acute treatment of migraine. It is a high-affinity, highly selective 5-hydroxytriptamine (serotonin)-1F (5-HT1F) receptor agonist. Lasmiditan has a unique chemical structure based on a pyridinoyl-piperidine scaffold that replaces the indole group of triptans (504).
5-HT1F receptors are expressed at various levels within the CNS and the trigeminovascular system, essential for the pathophysiology of migraine. Lasmiditan exerts its therapeutic effect through the agonistic effects at the 5-HT1F receptor, blocking pain transmission through the trigeminal ganglion and nucleus trigeminalis caudalis and inhibiting CGRP and glutamate release (505,506).
Lasmiditan is highly lipophilic and capable of crossing the blood-brain barrier, resulting in the occurrence of CNS-related adverse events (507).
Unlike triptans, agonists of the 5-HT1F receptor are devoid of vasoconstrictor properties, as demonstrated in preclinical animal models and in vitro human models (508,509). They were demonstrated to be safe and effective also in patients with concomitant cardiovascular risk factors (504,510,511).
Therefore, lasmiditan may address important unmet needs in patients with cardiovascular risk factors, cardiovascular diseases, or patients who respond poorly to other acute treatments.
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for the guideline on ditans for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 3.7.1.
Results
Overall, we retrieved 170 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included six systematic reviews and meta-analyses (24,507,512–515) that were considered as sources of RCTs. After analyzing full texts of these RCTs, we included four RCTs in the quantitative synthesis (516–519) (Figure 3.7.1).

Meta-analysis selection flow chart - ditans.
Overall, we retrieved 320 references from searching for RCTs and, after removing duplicates, we had 237 references to analyze. However, considering the most recent included systematic review and meta-analysis on the topic, the analysis of additional RCTs was performed for papers published since 2021 (49 references). We finally included one additional RCT (520). No further studies were included from the literature search updates performed in May 2023 and November 2023 (Figure 3.7.2).

RCTs selection flow chart - ditans. *trials published since 2021 were screened.
Overall, five studies were included in the quantitative synthesis (meta-analyses) to develop the evidence-based guideline. All of them reported a comparison between lasmiditan and placebo and are presented in this section.
Lasmiditan
Main evidence
We found five RCTs (516–520) addressing lasmiditan as compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of oral lasmiditan 50, 100 and 200 mg over placebo considering the outcomes of pain freedom at 2 h (Figure 3.7.3) and pain relief at 2 h (Figure 3.7.4). The quality of evidence for both outcomes was high (Table 3.7.1)
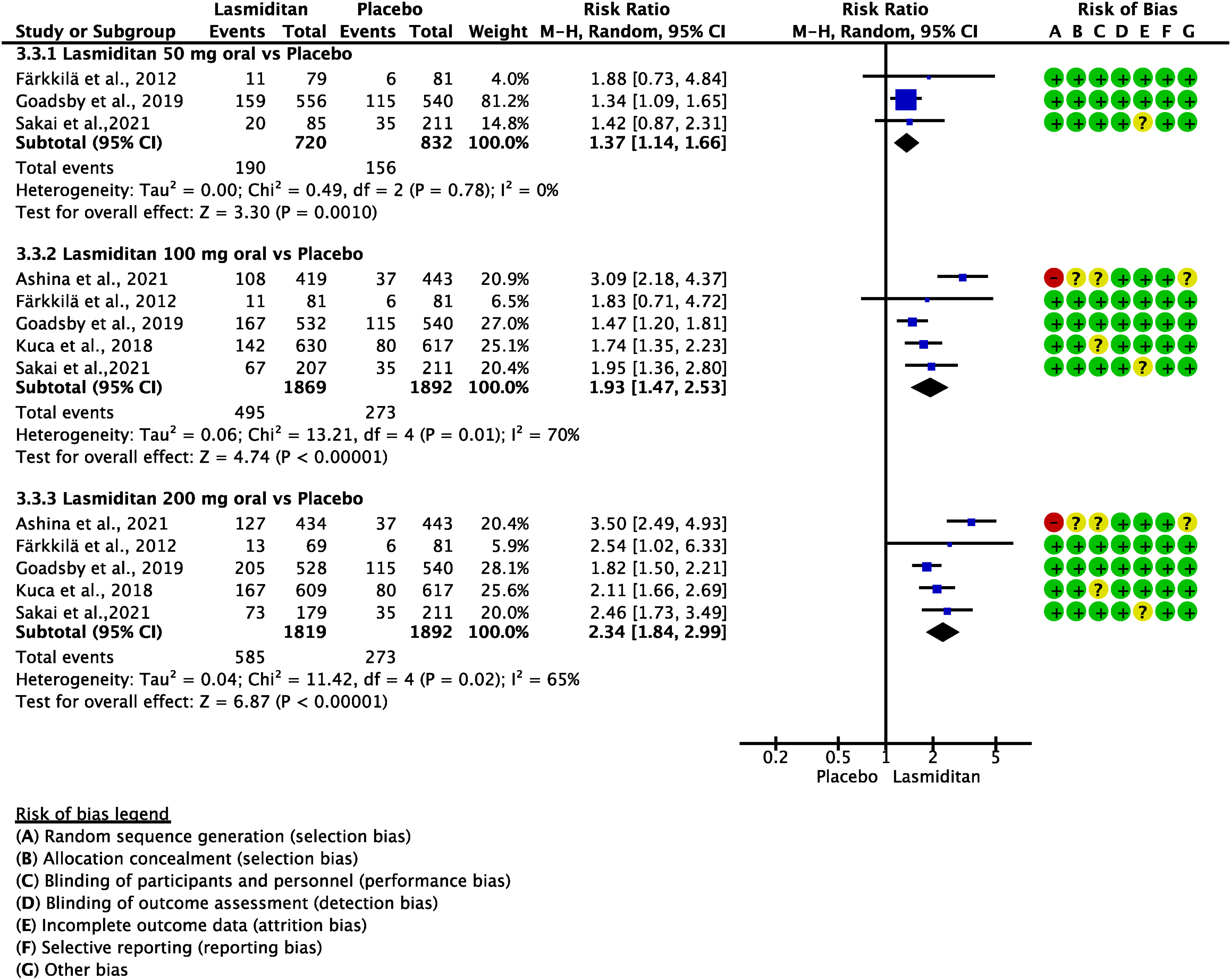
Forest plot showing the comparison between oral lasmiditan (50, 100, and 200 mg) and placebo for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral lasmiditan (50, 100, and 200 mg) and placebo for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral lasmiditan versus placebo in migraine

Additional evidence
None.
Evidence-based recommendation for PICO 3.7.1 In patients with migraine, we recommend lasmiditan 50 mg, 100 mg, or 200 mg for the acute treatment of migraine attacks.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Safety and tolerability of lasmiditan
Evidence-based guideline summary
We found studies on lasmiditan to be included for deriving evidence-based recommendations for this guideline. There is high quality of evidence to recommend the use of lasmiditan for the acute treatment of migraine attacks (Figure 3.7.5). Notably, lasmiditan lacks head-to-head comparisons with other acute treatments for migraine, such as triptans (please refer to Section 3.9 for other comparisons).

Summary of evidence-based recommendations on lasmiditan for the acute treatment of the migraine attacks.
Expert-based opinions
Topic: Lasmiditan dosing
Topic: Tolerability
Topic: Use in patients with cardiovascular risk factors and disease
Introduction
Gepants were developed in the early 2000s as a new symptomatic treatment for migraine. However, their development was soon interrupted due to liver toxicity: telcagepant, a first generation gepant, determined indeed a significant increase in the serum levels of liver aminotransferase levels compared to placebo (523). Hence, research focused on the chemical manipulation of the first generation gepants led to the second generation gepants, devoid of liver toxicity (524). The Food and Drug Administration (FDA) approved rimegepant 75 mg in 2020 and ubrogepant 50 and 100 mg in 2019 for the acute treatment of migraine attacks (524). The European Medicine Agency (EMA) approved the use of rimegepant in Europe in 2022. More recently, another gepant has been studied: zavegepant, a third generation gepant, characterized by the intranasal administration route (525).
Gepants are small molecules that bind with high affinity to the CGRP receptor, thus inducing an allosteric modulation that prevents CGRP binding. Notably, their specific mechanism of action justifies their good safety profile (526). Gepants do not constrict or tighten blood vessels, although it must be noted that they inhibit vasoconstriction and the opening of collaterals in conditions of ischemia (526).
Section-specific methods
This section followed the general procedure to develop this guideline with the exception of the literature search update, which was performed directly in November 2023 overcoming the May 2023 update.
Search strings for the Guideline on the gepants (acute treatment) for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 3.8.1.
Results
Overall, we retrieved 173 references from searching for systematic reviews and meta-analyses. After duplicate removal and selection phases, we included eight systematic reviews and meta-analyses (24,237,527–532) as a source of randomized controlled trials (RCTs). After analyzing full texts of these RCTs, we included seven of them in the quantitative synthesis (533–539) (Figure 3.8.1).

Meta-analysis selection flow chart - gepants for acute treatment.
Overall, we retrieved 365 references from searching for RCTs and, after removing duplicates, we had 304 references to analyze. However, considering the most recent included systematic review/meta-analysis on the topic, the analysis of additional RCTs was performed for papers published since May 2022 (31 references). We finally included one additional RCT on rimegepant (540) and two RCTs on zavegepant (541,542) (Figure 3.8.2). Notably, the selection of RCTs on zavegepant was performed via a specific search string (see Online Appendix 3.8.1 and Online Appendix 3.8.2).

RCTs selection flow chart - gepants for acute treatment. *trials published since 01 May 2022 were screened **Trials on zavegepant assessed with a further search string (see Online Appendix 3.8.1) and flow chart (see Online Appendix 3.8.2).
Overall, 12 studies were included in the quantitative synthesis (meta-analyses) to develop the evidence-based guideline. All the included studies reported a comparison between a gepant and placebo and are presented in this paper.
Rimegepant
Main evidence
We found five RCTs (533–536,540) addressing rimegepant oral-disintegrating tablets compared to placebo in the acute treatment of migraine attacks that met the criteria to be included in the main evidence. In particular, the studies by Yu et al. (540) and Croop et al. (543) tested rimegepant oral-disintegrating tablets, whilst the others rimegepant tablets (534–536). Since the two formulations were found similarly effective, we discussed them together (544). The pooled analysis showed benefits of oral rimegepant 75 mg daily over placebo considering the outcomes of pain freedom at 2 h (Figure 3.8.3) and pain relief at 2 h (Figure 3.8.4). The quality of evidence for both outcomes was high (Table 3.8.1).
Forest plot showing the comparison between rimegepant 75 mg oral-disintegrating tablets and placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between rimegepant 75 mg oral-disintegrating tablets and placebo for the outcome pain relief at 2 h.
GRADE evidence profile table for rimegepant 75mg oral-disintegrating tablets versus placebo for the acute treatment of migraine attacks

Additional evidence
None.
Evidence-based recommendation for PICO 3.8.1 In patients with migraine, we recommend rimegepant 75 mg oral-disintegrating tablets for the acute treatment of the attacks.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Ubrogepant
Main evidence
We found five RCTs (537–539,545,546) addressing ubrogepant compared to placebo for the acute treatment of migraine attacks that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of oral ubrogepant 50 and 100 mg over placebo considering the outcomes pain freedom at 2 h (Figure 3.8.5) and pain relief at 2 h (Figure 3.8.6). The quality of evidence for both outcomes was high (Table 3.8.2).

Forest plot showing the comparison between oral ubrogepant 50 mg or 100 mg and placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between oral ubrogepant 50 mg or 100 mg and placebo for the outcome pain relief at 2 h.
GRADE evidence profile table for oral ubrogepant versus placebo in the acute treatment of migraine attacks

Additional evidence
None.
Evidence-based recommendation for PICO 3.8.2 In patients with migraine, we recommend oral ubrogepant 50 or 100 mg for the acute treatment of attacks.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Zavegepant
The study selection for zavegepant was added following the literature search update. The related flowchart is provided in Online Appendix 3.8.2.
Main evidence
We found two RCTs (541,542) addressing zavegepant compared to placebo in the acute treatment of migraine attacks that met the predefined criteria to be included in the main evidence.
The pooled analysis showed benefits of zavegepant 5, 10, and 20 mg nasal spray over placebo considering the outcomes of pain freedom at 2 h (Figure 3.8.7) and pain relief at 2 h (Figure 3.8.8). The quality of evidence for both outcomes was high for the 10 mg dosage – due to the presence of two RCTs – and moderate for the 5 mg and 20 mg doses of zavegepant (Table 3.8.3) due to the presence of only one RCT.

Forest plot showing the comparison between zavegepant 5 mg, 10 mg, or 20 mg nasal spray and placebo for the outcome pain freedom at 2 h.

Forest plot showing the comparison between zavegepant 5 mg, 10 mg, or 20 mg nasal spray and placebo for the outcome pain relief at 2 h.
GRADE evidence profile table for zavegepant nasal spray versus placebo in the acute treatment of migraine attacks

Only one trial.
Additional evidence
None.
Evidence-based recommendations for PICO 3.8.3
1) In patients with migraine, we recommend intranasal zavegepant 10 mg for the acute treatment of migraine attacks.
Strength of the recommendation: Strong (↑↑)
2) In patients with migraine, we suggest against zavegepant 5 mg nasal spray for the acute treatment of migraine attacks. Strength of the recommendation: Weak (↓)
3) In patients with migraine, we suggest zavegepant 20 mg nasal spray for the acute treatment of migraine attacks.
Strength of the recommendation: Weak (↑).
Quality of evidence: High (⊕⊕⊕⊕)
Quality of evidence: Moderate (⊕⊕⊕⊖)
Quality of evidence: Moderate (⊕⊕⊕⊖).
Safety and tolerability of rimegepant, ubrogepant, and zavegepant
The safety of rimegepant and zavegepant in geriatric patients has not been explored, so they are not recommended in elderly patients with migraine. Ubrogepant has been tested up to 75 years of age. Children and adolescents have not been studied yet, therefore gepants are at present contraindicated below the age of 18 years. Pregnant or lactating women were excluded from RCTs too, hence, gepants are not recommended in these categories. It ought to be taken into consideration that ubrogepant induces fetal toxicity in rats and it passes in the maternal milk in rats, where it has similar concentrations than in the plasma (550). For rimegepant, it has been demonstrated that a single dose can result in its presence in maternal milk (551), even if less than 1%. Hence, rimegepant should be avoided in breastfeeding women (551). Since rimegepant and ubrogepant are metabolized by the liver, they should not be administered in patients with liver impairment (550,552). Finally, as rimegepant and ubrogepant are substrates for the P glycoprotein (P-gp), their use is contraindicated in the presence of severe renal impairment (550,552). Zavegepant excretion is mainly biliary and fecal (525); the drug is mainly metabolized by CYP3A4 and CYP2D6 (525). Mild or moderate liver impairment does not seem to alter the metabolism of zavegepant, while the drug has not been tested in patients with severe liver impairment (525,553). Zavegepant has also not been tested in patients with renal impairment, which however should not be considered a problem given the very small proportion of renal metabolism of the drug (553). The use of zavegepant during pregnancy has not been evaluated in humans and is therefore contraindicated, although animal studies showed no significant increase in birth defects (525,554).
Evidence-based guideline summary
We found studies on rimegepant, ubrogepant, and zavegepant to be included for deriving evidence-based recommendations for this guideline. There is high quality of evidence to recommend the use of rimegepant, ubrogepant, and zavegepant for the acute treatment of migraine attacks (Figure 3.8.9).
Summary of evidence-based recommendations on gepants for the acute treatment of migraine attacks.
Expert-based opinion
Optimization of ubrogepant use
Topic: Ubrogepant dosing
Ubrogepant and anti-CGRP monoclonal antibodies
Topic: Ubrogepant co-administered with an anti-CGRP monoclonal antibody
Zavegepant and oral gepants
Topic: Choice of zavegepant in special cases
Introduction
Several acute treatments for migraine have been assessed comparatively to test their efficacy and tolerability. In clinical practice, it is important to know those comparisons to have an idea of the possible efficacy of different approaches to treat migraine attacks. However, there is a need for an orderly and rational presentation of those studies and of their results to provide clinicians with evidence-based tools for clinical judgment.
For those reasons, the present section reports a systematic and critical evaluation of the efficacy results of head-to-head studies comparing the different acute treatments for migraine.
Section-specific methods
All RCTs included in this section were retrieved from the search strings of the other sections dedicated to RCTs of acute treatments. We considered only PICOs including at least one drug as intervention or comparator that had an independent comparison against placebo.
At first, we collected all the head-to-head RCTs retrieved from the literature search. Duplicates were removed as an initial step.
All RCTs were considered only once, even if eligible for more than one comparison. Comparisons were structured to be read in either direction, without the concept of a main intervention and its comparator. The terms “intervention” and “comparator” were replaced by “Drug 1” and “Drug 2” in the PICO structure (Table 3.9.1). Nevertheless, given the number of available trials, we classified them into the following groups of comparisons:
Non-steroidal anti-inflammatory drugs (NSAIDs) vs NSAIDs Triptans vs NSAIDs Triptans vs triptans Triptans vs other analgesics Combination analgesics vs NSAIDs Combination analgesics vs triptans Combination analgesics vs paracetamol Combination analgesics vs combination analgesics Combination analgesics vs other analgesics A hypothesis of non-inferiority or superiority of one drug over the other was clearly stated The study sample size was justified.
RCTs were included in the main evidence if they met all the following criteria:
All trials not meeting these criteria were excluded from the main evidence and considered for additional evidence.
PICO structure of randomized controlled trials included in the present chapter. ASA, acetylsalicylic acid; CAs, combination analgesics

PICO structure of randomized controlled trials included in the present chapter. ASA, acetylsalicylic acid; CAs, combination analgesics

Only head-to-head comparisons between different drugs were included in the present section.
We restricted comparisons to oral and intranasal drugs, excluding studies comparing parenteral routes of administration of drugs. This choice was made to decrease the heterogeneity of comparisons and to adhere to the outpatient settings in which oral and intranasal drugs are the most used.
Figure 3.9.1 reports the results of the literature search. Table 3.9.1 reports the PICO question structure of the present section.

Flowchart of study selection for the present chapter.
It is important to note that PICO questions referring to the present section were structured after the literature search, as there were no expected results prior to the search.
Table 3.9.1 reports the comparisons that were included in the present section, while Figure 3.9.87 reports a summary of each recommendation.
Comparison between different non-steroidal anti-inflammatory drugs
Main evidence
We found one RCT comparing oral acetylsalicylic acid 1000 mg to oral ibuprofen 400 mg in patients with migraine (30) that met the criteria to be included in the main evidence. The risk of bias was unclear (Figures 3.9.2 and 3.9.3). The RCT showed no difference between oral acetylsalicylic acid 1000 mg and oral ibuprofen 400 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.2) and pain relief at 2 h (Figure 3.9.3). The quality of evidence for both outcomes was considered low (Table 3.9.2).

Forest plot showing the comparison between oral acetylsalicylic acid 1000 mg and oral ibuprofen 400 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral acetylsalicylic acid 1000 mg and oral ibuprofen 400 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral acetylsalicylic acid 1000 mg versus oral ibuprofen 400 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.1 In patients with migraine, we suggest acetylsalicylic acid 1000 mg and ibuprofen 400 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: =
Comparisons between triptans and non-steroidal anti-inflammatory drugs
Main evidence
We found one RCT comparing rizatriptan 10 mg to ibuprofen 400 mg in patients with migraine (38) that met the criteria to be included in the main evidence. The risk of bias was low (Figures 3.9.4 and 3.9.5). The RCT showed no difference between oral rizatriptan 10 mg and oral ibuprofen 400 mg considering the outcome pain freedom at 2 h (Figure 3.9.4) while oral rizatriptan 10 mg was slightly superior to oral ibuprofen 400 mg considering the outcome pain relief at 2 h (Figure 3.9.5). The quality of evidence for both outcomes was considered low to moderate (Table 3.9.3).

Forest plot showing the comparison between oral rizatriptan 10 mg and oral ibuprofen 400 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral rizatriptan 10 mg and oral ibuprofen 400 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral rizatriptan 10 mg versus oral ibuprofen 400 mg in patients with migraine

Only one trial; bthe confidence interval included the threshold for clinical decision.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.2 In patients with migraine, we suggest oral rizatriptan 10 mg over oral ibuprofen 400 mg for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Weak (↑)
Main evidence
We found two RCTs comparing sumatriptan 50 mg to acetylsalicylic acid 1000 mg in patients with migraine (29,30) that met the criteria to be included in the main evidence. The risk of bias was unclear (Figures 3.9.6 and 3.9.7). The RCTs showed no difference between sumatriptan 50 mg and acetylsalicylic acid 1000 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.6) and pain relief at 2 h (Figure 3.9.7). The quality of evidence for both outcomes was considered moderate (Table 3.9.4).
Forest plot showing the comparison between oral sumatriptan 50 mg and oral acetylsalicylic 1000 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral sumatriptan 50 mg and oral acetylsalicylic 1000 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral sumatriptan 50 mg versus oral acetylsalicylic acid 1000 mg in patients with migraine

Additional evidence
None.
Evidence-based recommendation for PICO 3.9.3 In patients with migraine, we suggest oral sumatriptan 50 mg and oral acetylsalicylic acid 1000 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
None.
Additional evidence
We found one RCT comparing oral sumatriptan 50 mg and ibuprofen 400 mg (30) that did not meet the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was considered unclear (Figures 3.9.8 and 3.9.9). The RCT found no difference between oral sumatriptan 50 mg and oral ibuprofen 400 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.8) and pain relief at 2 h (Figure 3.9.9). The quality of evidence was considered very low due to the availability of only one RCT with unclear risk of bias.
Evidence-based recommendation for PICO 3.9.4 In patients with migraine, we suggest sumatriptan 50 mg and ibuprofen 400 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =

Forest plot showing the comparison between oral sumatriptan 50 mg and oral ibuprofen 400 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral sumatriptan 50 mg and oral ibuprofen 400 mg for the outcome pain relief at 2 h in patients with migraine.
Main evidence
We found one RCT comparing sumatriptan 20 mg nasal spray to ketorolac 31.5 mg nasal spray in patients with migraine (44) that met the criteria to be included in the main evidence. The risk of bias was high (Figures 3.9.10 and 3.9.11). The RCT showed no difference between sumatriptan 20 mg nasal spray and ketorolac 31.5 mg nasal spray considering the outcomes of pain freedom at 2 h (Figure 3.9.10) and pain relief at 2 h (Figure 3.9.11). The quality of evidence for both outcomes was considered very low (Table 3.9.5) due to the availability of only one RCT with high risk of bias and low numbers of patients (<50 per group).

Forest plot showing the comparison between sumatriptan 20 mg nasal spray and ketorolac 31.5 mg nasal spray for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between sumatriptan 20 mg nasal spray and ketorolac 31.5 mg nasal spray for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for sumatriptan 20 mg nasal spray versus ketorolac 31.5 mg nasal spray in patients with migraine
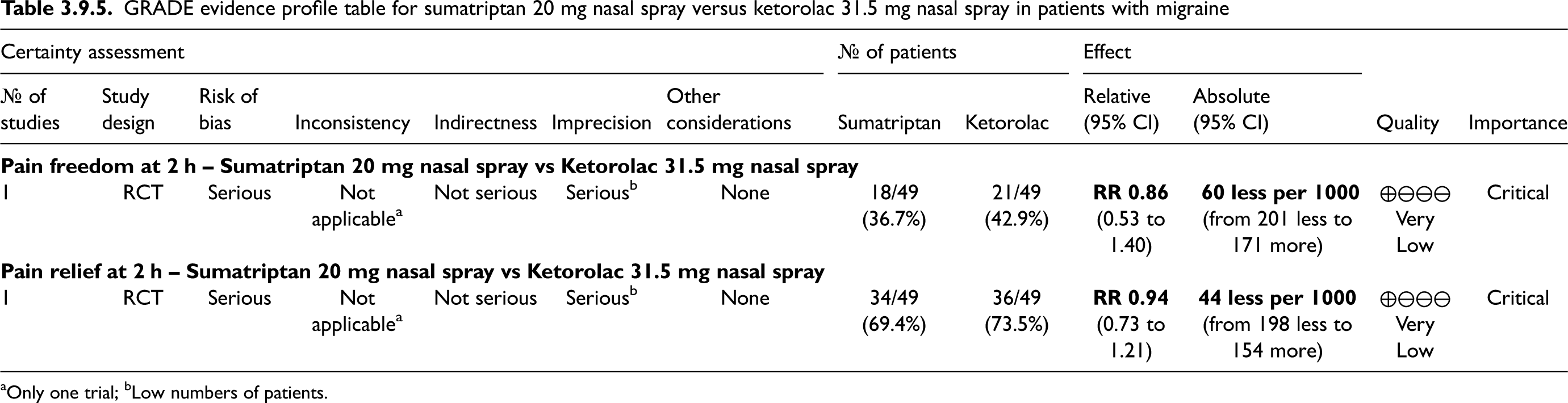
Only one trial; bLow numbers of patients.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.5 In patients with migraine, we suggest sumatriptan 20 mg nasal spray and ketorolac 31.5 mg nasal spray as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =
Main evidence
We found one RCT comparing oral sumatriptan 100 mg with oral naproxen 500 mg (174) that met the criteria for main evidence. The risk of bias was unclear (Figure 3.9.12). The RCT showed no difference between oral sumatriptan 100 mg and oral naproxen 500 mg considering the outcome pain freedom at 2 h (Figure 3.9.12). The quality of evidence was considered low (Table 3.9.6).
Forest plot showing the comparison between oral sumatriptan 100 mg and oral naproxen 500 mg for the outcome pain freedom at 2 h in patients with migraine.
GRADE evidence profile table for oral sumatriptan 100 mg versus oral naproxen 500 mg in patients with migraine
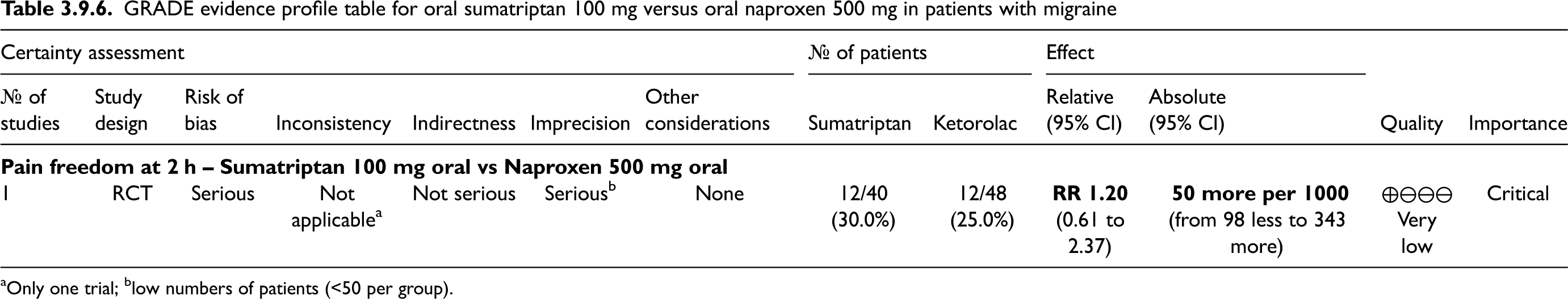
Only one trial; blow numbers of patients (<50 per group).
Additional evidence
We found two RCTs comparing oral sumatriptan 50 mg or 85 mg with oral naproxen 500 mg (27,41). Those RCTs did not meet the criteria for main evidence as they did not report criteria for sample size calculation. The risk of bias was unclear (Figures 3.9.13 and 3.9.14). Overall, the RCTs showed that the 85 mg dose of oral sumatriptan was superior to oral naproxen 500 mg considering the outcomes pain freedom at 2 h (Figure 3.9.13) and pain relief at 2 h (Figure 3.9.14), while the 50 mg dose of oral sumatriptan was not more effective than oral naproxen 500 mg. The quality of evidence was considered very low due to the availability of RCTs with unclear risk of bias.
Evidence-based recommendation for PICO 3.9.6 In patients with migraine, we suggest oral sumatriptan 50 mg or 100 mg and oral naproxen 500 mg as equivalent options for the acute treatment of migraine attacks; there is very low evidence to suggest sumatriptan 85 mg over naproxen 500 mg.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =

Forest plots showing the comparison between oral sumatriptan (50 mg or 85 mg) and oral naproxen 500 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral sumatriptan (50 mg or 85 mg) and oral naproxen 500 mg for the outcome pain relief at 2 h in patients with migraine.
Comparisons between two different triptans
Main evidence
We found one RCT comparing oral almotriptan 12.5 mg with oral frovatriptan 2.5 mg (160) that met the criteria for inclusion in main evidence. The risk of bias was low (Figures 3.9.15 and 3.9.16). The RCT found no difference between oral almotriptan and oral frovatriptan considering the outcomes of pain freedom at 2 h (Figure 3.9.15) and pain relief at 2 h (Figure 3.9.16). The quality of evidence was considered moderate (Table 3.9.7) due to the availability of only one RCT.

Forest plot showing the comparison between oral almotriptan 12.5 mg and oral frovatriptan 2.5 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral almotriptan 12.5 mg and oral frovatriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for almotriptan 12.5 mg versus frovatriptan 2.5 mg for the acute treatment of migraine attacks

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.7 In patients with migraine, we suggest almotriptan 12.5 mg and frovatriptan 2.5 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
We found two RCTs comparing oral almotriptan 12.5 mg with oral sumatriptan 50 mg or 100 mg (150,193) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.17 and 3.9.18). The two RCTs found that oral almotriptan was inferior to oral sumatriptan 50 mg and equivalent to oral sumatriptan 100 mg considering the outcome pain freedom at 2 h (Figure 3.9.17), while oral almotriptan and oral sumatriptan were equivalent considering the outcome pain relief at 2 h (Figure 3.9.18). The quality of evidence was considered moderate (Table 3.9.8).

Forest plot showing the comparison between oral almotriptan 12.5 mg and oral sumatriptan 50 mg or 100 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral almotriptan 12.5 mg and oral sumatriptan 50 mg or 100 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral almotriptan 12.5 mg versus oral sumatriptan 50 mg or 100 mg in patients with migraine
Only one trial.
Additional evidence
We found one RCT comparing oral almotriptan 12.5 mg to oral sumatriptan 100 mg for the acute treatment of migraine attacks (173) that did not meet the criteria for main evidence as it did not specify a sample size calculation for pain freedom at 2 h. The risk of bias was low (Figure 3.9.19). The RCT showed no difference between oral almotriptan 12.5 mg and oral sumatriptan 100 mg considering the outcome pain freedom at 2 h (Figure 3.9.19).
Evidence-based recommendation for PICO 3.9.8 In patients with migraine, we suggest oral almotriptan 12.5 mg and oral sumatriptan – 50 mg or 100 mg – as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =

Forest plot showing the comparison between oral almotriptan 12.5 mg and oral sumatriptan 100 mg for the outcome pain freedom at 2 h in patients with migraine.
Main evidence
We found one RCT comparing oral almotriptan 12.5 mg with oral zolmitriptan 2.5 mg (178) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.20 and 3.9.21). The RCT showed no difference between oral almotriptan 12.5 mg and oral zolmitriptan 2.5 mg considering the outcome of pain freedom at 2 h (Figure 3.9.20) and pain relief at 2 h (Figure 3.9.21). The quality of evidence was considered moderate (Table 3.9.9).

Forest plot showing the comparison between oral almotriptan 12.5 mg and oral zolmitriptan 2.5 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral almotriptan 12.5 mg and oral zolmitriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral almotriptan 12.5 mg versus oral zolmitriptan 2.5 mg in patients with migraine
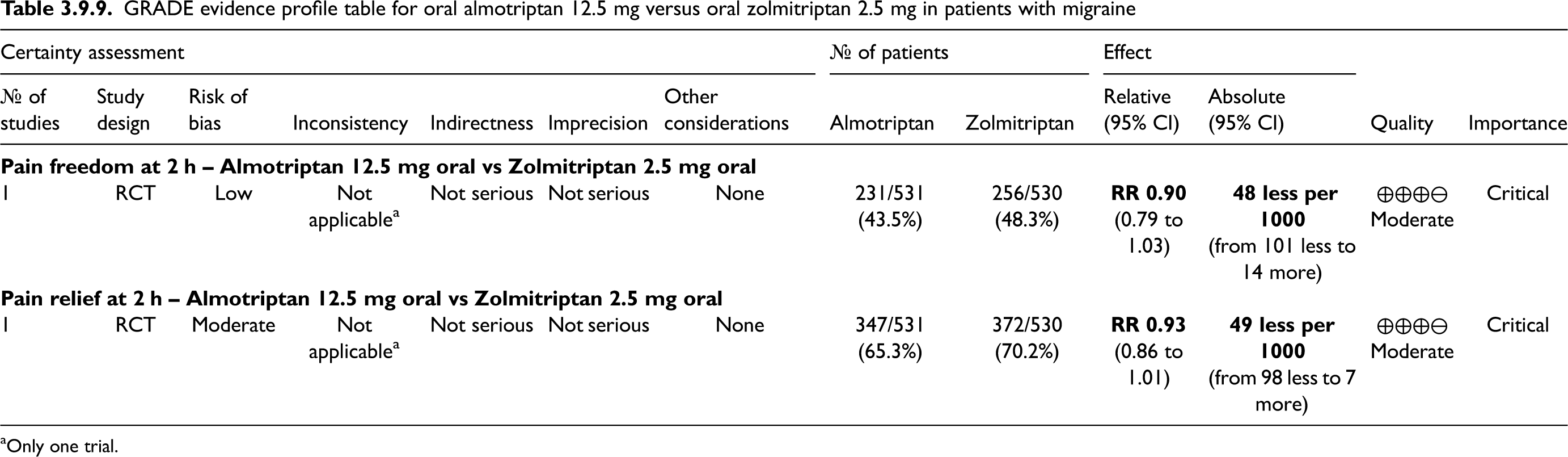
Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.9 In patients with migraine, we suggest oral almotriptan 12.5 mg and oral zolmitriptan 2.5 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: weak (=)
Main evidence
We found one RCT comparing oral eletriptan 40 mg to oral naratriptan 2.5 mg (96) that met the criteria for main evidence. The risk of bias was considered unclear (Figures 3.9.22 and 3.9.23). The RCT found that oral eletriptan 40 mg was superior to oral naratriptan 2.5 mg considering the outcomes pain freedom at 2 h (Figure 3.9.22) and pain relief at 2 h (Figure 3.9.23). The quality of evidence was considered low (Table 3.9.10).

Forest plot showing the comparison between oral eletriptan 40 mg and oral naratriptan 2.5 mg for the outcome pain freedom at 2 h in patients with migraine.
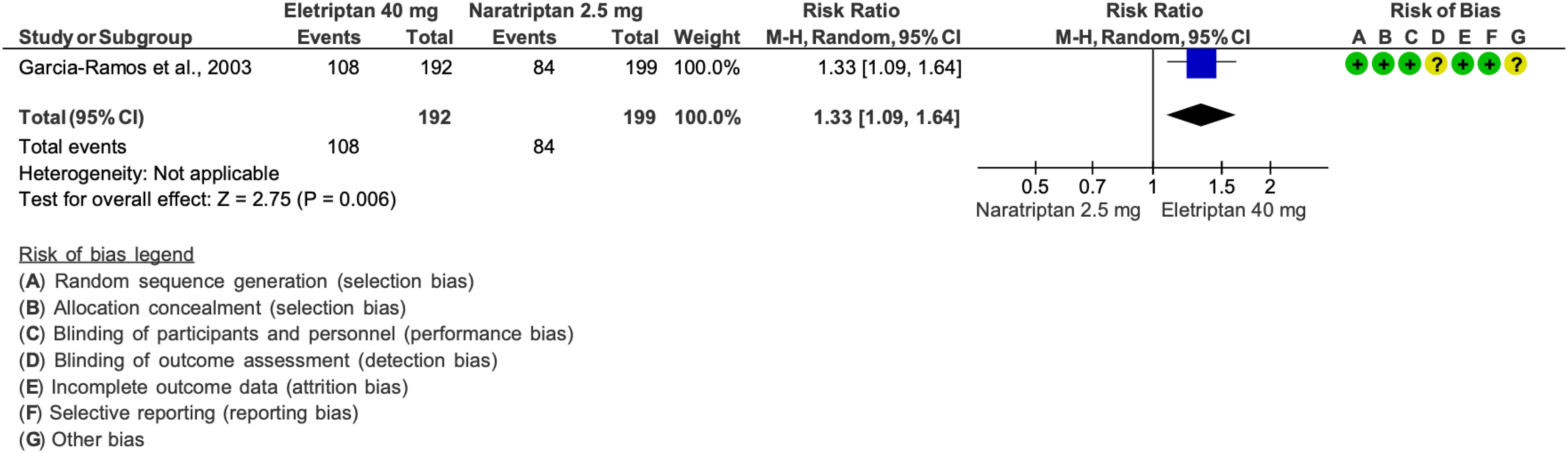
Forest plot showing the comparison between oral eletriptan 40 mg and oral naratriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral eletriptan 40 mg vs oral naratriptan 2.5 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.10 In patients with migraine, we suggest eletriptan 40 mg over naratriptan 2.5 mg for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Weak (↑)
Main evidence
None.
Additional evidence
We found one RCT comparing oral eletriptan 40 mg with oral rizatriptan 10 mg (195) that did not meet the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was considered high (Figures 3.9.24 and 3.9.25). The RCT showed that oral eletriptan 40 mg was equivalent to oral rizatriptan 10 mg considering the outcome pain freedom at 2 h (Figure 3.9.24) and superior to oral rizatriptan 10 mg considering the outcome pain relief at 2 h (Figure 3.9.25). Due to the small number of patients (<50 in each treatment arm), no recommendation was issued.
Evidence-based recommendation for PICO 3.9.11 None. Quality of evidence: - Strength of the recommendation: -

Forest plot showing the comparison between eletriptan 40 mg and rizatriptan 10 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between eletriptan 40 mg and rizatriptan 10 mg for the outcome pain relief at 2 h in patients with migraine.
Main evidence
We found three RCTs comparing oral eletriptan 40 mg with oral sumatriptan 100 mg (93,97,98) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.26 and 3.9.27). The RCTs found that oral eletriptan 40 mg was superior to oral sumatriptan 100 mg considering the outcomes pain freedom at 2 h (Figure 3.9.26) and pain relief at 2 h (Figure 3.9.27). The quality of evidence was considered high (Table 3.9.11).
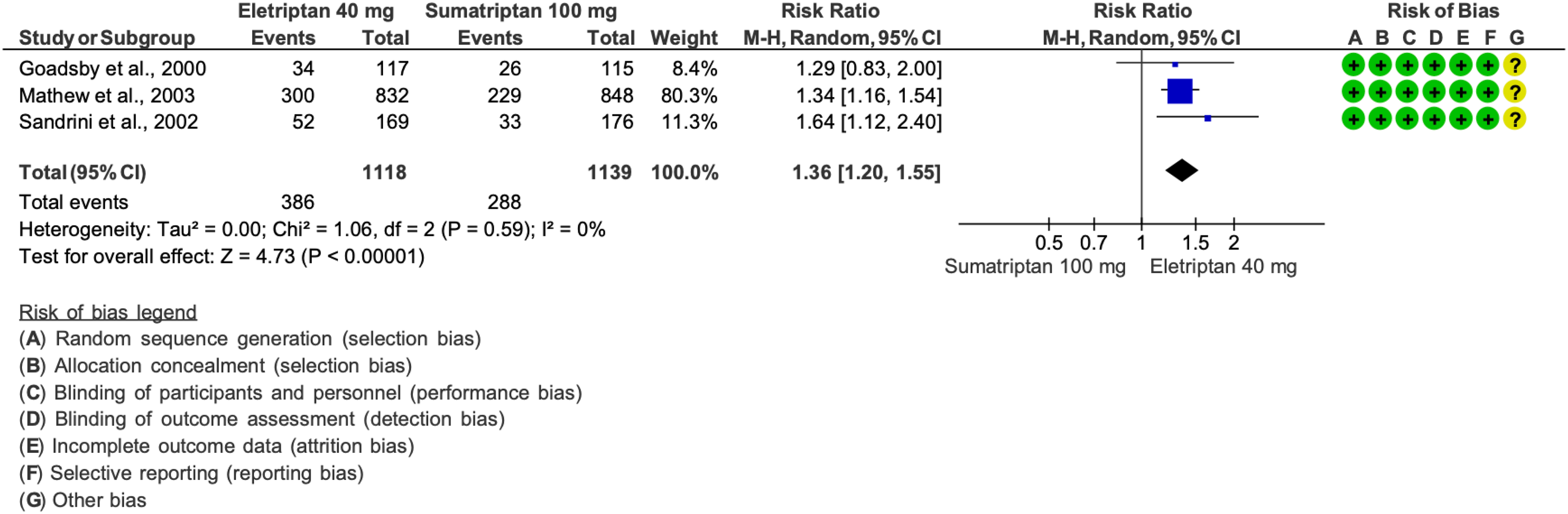
Forest plot showing the comparison between oral eletriptan 40 mg and oral sumatriptan 100 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral eletriptan 40 mg and oral sumatriptan 100 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for eletriptan 40 mg vs sumatriptan 100 mg in patients with migraine

Additional evidence
None.
Evidence-based recommendation for PICO 3.9.12 In patients with migraine, we recommend eletriptan 40 mg over sumatriptan 100 mg for the acute treatment of migraine attacks.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Main evidence
We found one RCT comparing oral eletriptan 40 mg with oral zolmitriptan 2.5 mg (100) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.28 and 3.9.29). The RCT showed that oral eletriptan 40 mg and oral zolmitriptan 2.5 mg were equivalent considering the outcomes of pain freedom at 2 h (Figure 3.9.28) and pain relief at 2 h (Figure 3.9.29). The quality of evidence was considered moderate (Table 3.9.12).

Forest plot showing the comparison between oral eletriptan 40 mg and oral zolmitriptan 2.5 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral eletriptan 40 mg and oral zolmitriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral eletriptan 40 mg vs oral zolmitriptan 2.5 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.13 In patients with migraine, we suggest eletriptan 40 mg and zolmitriptan 2.5 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
We found one RCT comparing oral frovatriptan 2.5 mg and oral rizatriptan 10 mg (190) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.30 and 3.9.31). The RCT showed no difference between oral frovatriptan 2.5 mg and oral rizatriptan 10 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.30) and pain relief at 2 h (Figure 3.9.31). The quality of evidence was considered moderate (Table 3.9.13).

Forest plot showing the comparison between oral frovatriptan 2.5 mg and oral rizatriptan 10 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral frovatriptan 2.5 and oral rizatriptan 10 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for frovatriptan 2.5 mg vs rizatriptan 10 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.14 In patients with migraine, we suggest frovatriptan 2.5 mg and rizatriptan 10 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
We found one RCT comparing oral frovatriptan 2.5 mg with oral zolmitriptan 2.5 mg (200) that met the criteria for main evidence. The risk of bias was low (Figures 3.9.32 and 3.9.33). The RCT showed no difference between oral frovatriptan 2.5 mg and oral zolmitriptan 2.5 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.32) and pain relief at 2 h (Figure 3.9.33). The quality of evidence was considered moderate (Table 3.9.14) due to the availability of only one RCT.

Forest plot showing the comparison between oral frovatriptan 2.5 mg and oral zolmitriptan 2.5 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral frovatriptan 2.5 mg and oral zolmitriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for frovatriptan 2.5 mg vs zolmitriptan 2.5 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.15 In patients with migraine, we suggest frovatriptan 2.5 mg and zolmitriptan 2.5 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
We found one RCT comparing oral naratriptan 2.5 mg with oral rizatriptan 10 mg (107) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.34 and 3.9.35). The RCT showed that oral rizatriptan 10 mg was superior to oral naratriptan 2.5 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.34) and pain relief at 2 h (Figure 3.9.35). The quality of evidence was considered moderate (Table 3.9.15).

Forest plot showing the comparison between oral naratriptan 2.5 mg and oral rizatriptan 10 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral naratriptan 2.5 mg and oral rizatriptan 10 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral naratriptan 2.5 mg vs oral rizatriptan 10 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.16 In patients with migraine, we suggest rizatriptan 10 mg over naratriptan 2.5 mg for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: weak (↑)
Main evidence
We found four RCTs comparing oral rizatriptan 5 or 10 mg to oral sumatriptan 50 or 100 mg (112,114,121,184) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.36 and 3.9.37). Overall, the RCTs showed no difference between rizatriptan and sumatriptan considering the outcomes of pain freedom at 2 h (Figure 3.9.36) and pain relief at 2 h (Figure 3.9.37). The quality of evidence was considered moderate-to-high (Table 3.9.16).

Forest plot showing the comparison between oral rizatriptan (5 or 10 mg) and oral sumatriptan (50 or 100 mg) for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral rizatriptan (5 or 10 mg) and oral sumatriptan (50 or 100 mg) for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for rizatriptan (5 or 10 mg) versus sumatriptan (50 or 100 mg) in patients with migraine

Additional evidence
We found one RCT comparing oral rizatriptan 10 mg with oral sumatriptan 50 mg (558) that did not meet the criteria for main evidence as it did not report a sample size calculation. The risk of bias was considered unclear (Figure 3.9.38). The RCT showed no difference between oral rizatriptan 10 mg and oral sumatriptan 50 mg considering the outcome pain freedom at 2 h (Figure 3.9.38).
Evidence-based recommendation for PICO 3.9.17 In patients with migraine, we suggest rizatriptan (5 mg or 10 mg) and sumatriptan (50 mg or 100 mg) as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
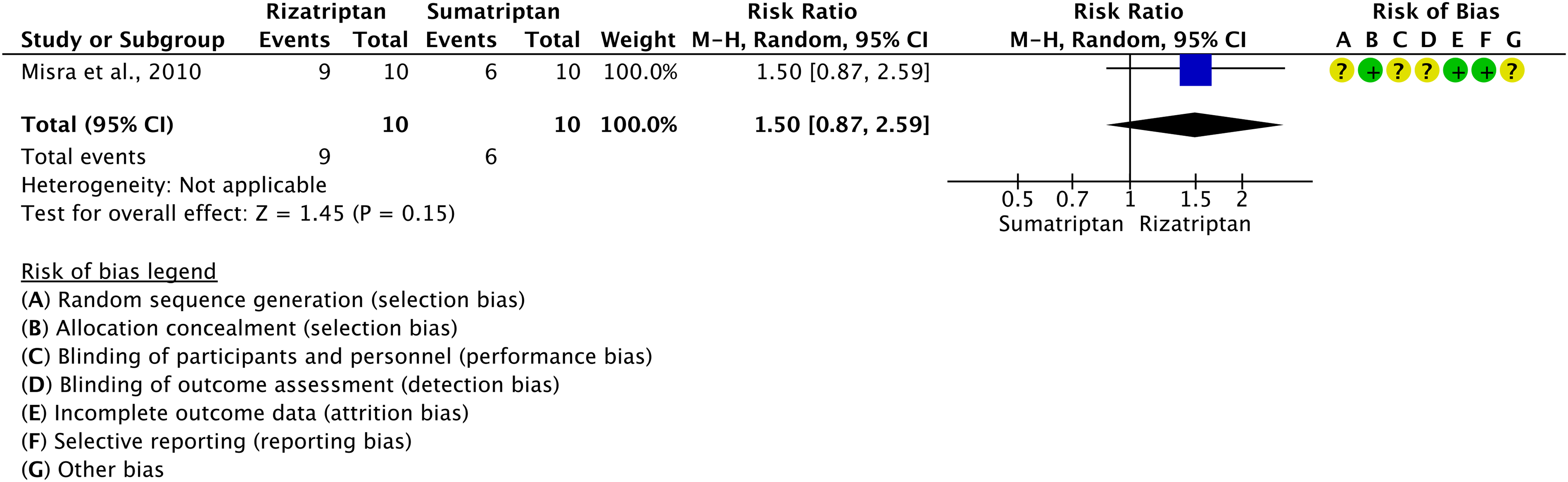
Forest plot showing the comparison between oral rizatriptan (5 or 10 mg) and oral sumatriptan (50 or 100 mg) for the outcome pain relief at 2 h in patients with migraine.
Main evidence
We found one RCT comparing oral rizatriptan 10 mg with oral zolmitriptan 2.5 (120) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.39 and 3.9.40). The RCT showed no difference between oral rizatriptan 10 mg and oral zolmitriptan 2.5 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.39) and pain relief at 2 h (Figure 3.9.40). The quality of evidence was considered moderate (Table 3.9.17).
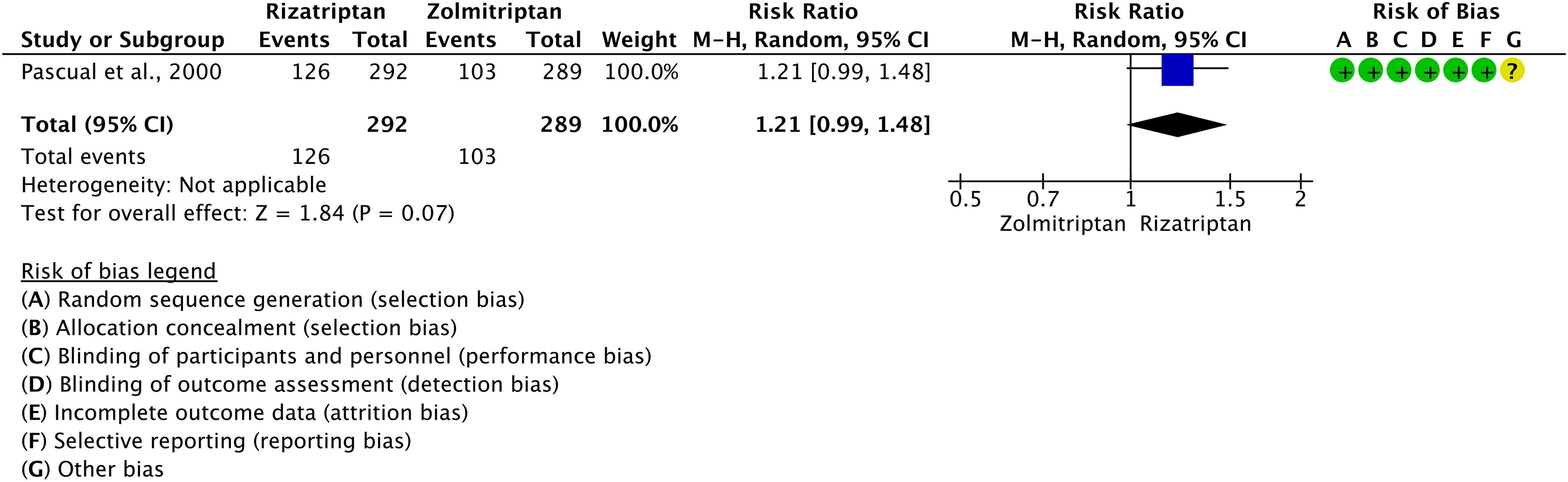
Forest plot showing the comparison between oral rizatriptan 10 mg and oral zolmitriptan 2.5 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral rizatriptan 10 mg and oral zolmitriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral rizatriptan 10 mg versus oral zolmitriptan 2.5 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.18 In patients with migraine, we suggest oral rizatriptan 10 mg and oral zolmitriptan 2.5 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
We found two RCTs comparing oral zolmitriptan 2.5 or 5 mg with oral sumatriptan 50 or 100 mg (176,179) that met the criteria for main evidence. The risk of bias was considered low or unclear (Figures 3.9.41 and 3.9.42). The RCTs showed no difference between oral zolmitriptan and oral sumatriptan considering the outcomes of pain freedom at 2 h (Figure 3.9.41) and pain relief at 2 h (Figure 3.9.42). The quality of evidence was considered low or moderate (Table 3.9.18).

Forest plot showing the comparison between oral zolmitriptan (2.5 or 5 mg) and oral sumatriptan (50 or 100 mg) for the outcome pain freedom at 2 h in patients with migraine.
Forest plot showing the comparison between oral zolmitriptan (2.5 or 5 mg) and oral sumatriptan (50 or 100 mg) for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral zolmitriptan (2.5 or 5 mg) versus oral sumatriptan (50 or 100 mg) in patients with migraine

Only one trial.
Additional evidence
We found one RCT comparing oral zolmitriptan (2.5 or 5 mg) with oral sumatriptan 50 mg (175) that did not meet the criteria for main evidence due to the lack of a formal sample size calculation. The risk of bias was considered low (Figure 3.9.43). The RCT showed no difference between oral zolmitriptan (2.5 or 5 mg) and oral sumatriptan 50 mg considering the outcome of pain relief at 2 h (Figure 3.9.43). The quality of evidence was considered moderate due to the availability of only one RCT.
Evidence-based recommendation for PICO 3.9.19 In patients with migraine, we suggest zolmitriptan (2.5 or 5 mg) and sumatriptan (50 mg or 100 mg) as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: =
Forest plot showing the comparison between oral zolmitriptan (2.5 or 5 mg) and oral sumatriptan 50 mg for the outcome pain relief at 2 h in patients with migraine.
Comparisons between triptans and other analgesics
Main evidence
None.
Additional evidence
We found one RCT comparing oral sumatriptan 100 mg with oral tolfenamic acid 200 mg (125) that did not meet the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was considered unclear (Figures 3.9.44 and 3.9.45). The RCT showed no difference between oral sumatriptan 100 mg and oral tolfenamic acid 200 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.44) and pain relief at 2 h (Figure 3.9.45). No recommendation was issued due to low numbers of patients (<50 per group).
Evidence-based recommendation for PICO 3.9.20 None. Quality of evidence: - Strength of the recommendation: -

Forest plot showing the comparison between oral sumatriptan 100 mg and oral tolfenamic acid 200 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral sumatriptan 100 mg and oral tolfenamic acid 200 mg for the outcome pain relief at 2 h in patients with migraine.
Comparisons between combination analgesics and non-steroidal anti-inflammatory agents
Main evidence
None.
Additional evidence
We found one RCT comparing oral paracetamol 400 mg + codeine 25 mg with oral acetylsalicylic acid 1000 mg (26) that did not meet the criteria for main evidence as it did not report a sample size calculation. The risk of bias was considered high (Figures 3.9.46 and 3.9.47). The RCT showed no difference between oral paracetamol 400 mg + codeine 25 mg and oral acetylsalicylic acid 1000 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.46) and pain relief at 2 h (Figure 3.9.47). The quality of evidence was considered very low due to the inclusion of only one RCT with a high risk of bias.
Evidence-based recommendation for PICO 3.9.21 In patients with migraine, we suggest oral paracetamol 400 mg + codeine 25 mg and oral acetylsalicylic acid 1000 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =
[NOTE: Opioids such as codeine present safety risks and should be used with caution. Please refer to Sections 3.4 and 3.6].

Forest plot showing the comparison between oral paracetamol 400 mg + codeine 25 mg and oral acetylsalicylic acid 1000 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral paracetamol 400 mg + codeine 25 mg and oral acetylsalicylic acid 1000 mg for the outcome pain relief at 2 h in patients with migraine.
Main evidence
None.
Additional evidence
We found two RCTs comparing oral sumatriptan (50 or 85 mg) + naproxen 500 mg with naproxen 500 mg alone (27,41) that did not meet the criteria for main evidence as they did not report a sample size calculation. The risk of bias was considered unclear (Figures 3.9.48 and 3.9.49). The RCT showed that oral sumatriptan (50 or 85 mg) + naproxen 500 mg was superior to oral naproxen 500 mg alone considering the outcomes of pain freedom at 2 h (Figure 3.9.48) and pain relief at 2 h (Figure 3.9.49). The quality of evidence was considered very low due to the inclusion of RCTs with unclear risk of bias.
Evidence-based recommendation for PICO 3.9.22 In patients with migraine, we suggest oral sumatriptan (50 or 85 mg) + naproxen 500 mg over oral naproxen 500 mg alone for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)

Forest plot showing the comparison between oral sumatriptan (50 or 85 mg) + naproxen 500 mg and oral naproxen 500 mg alone for the outcome pain freedom at 2 h in patients with migraine. NOTE: One study (27) is reported twice because it included two different populations.
Forest plot showing the comparison between oral sumatriptan (50 or 85 mg) + naproxen 500 mg and oral naproxen 500 mg alone for the outcome pain relief at 2 h in patients with migraine. NOTE: One study (27) is reported twice because it included two different populations.
Comparisons between combination analgesics and triptans
Main evidence
We found one RCT comparing oral paracetamol 500 mg + acetylsalicylic acid 500 mg + caffeine 130 mg with oral sumatriptan 50 mg (335) that met the criteria for main evidence. The risk of bias was considered high (Figure 3.9.50). The RCT showed that oral paracetamol 500 mg + acetylsalicylic acid 500 mg + caffeine 130 mg was superior to oral sumatriptan 50 mg considering the outcome of pain freedom at 2 h (Figure 3.9.50). The quality of evidence was considered very low (Table 3.9.19) due to the availability of only one RCT with a high risk of bias and low numbers of patients (<50 in one of the groups).

Forest plot showing the comparison between oral paracetamol 500 mg + acetylsalicylic acid 500 mg + caffeine 130 mg and oral sumatriptan 50 mg for the outcome pain freedom at 2 h in patients with migraine.
GRADE evidence profile for the comparison between oral paracetamol 500 mg + acetylsalicylic acid 500 mg + caffeine and oral sumatriptan 50 mg in patients with migraine
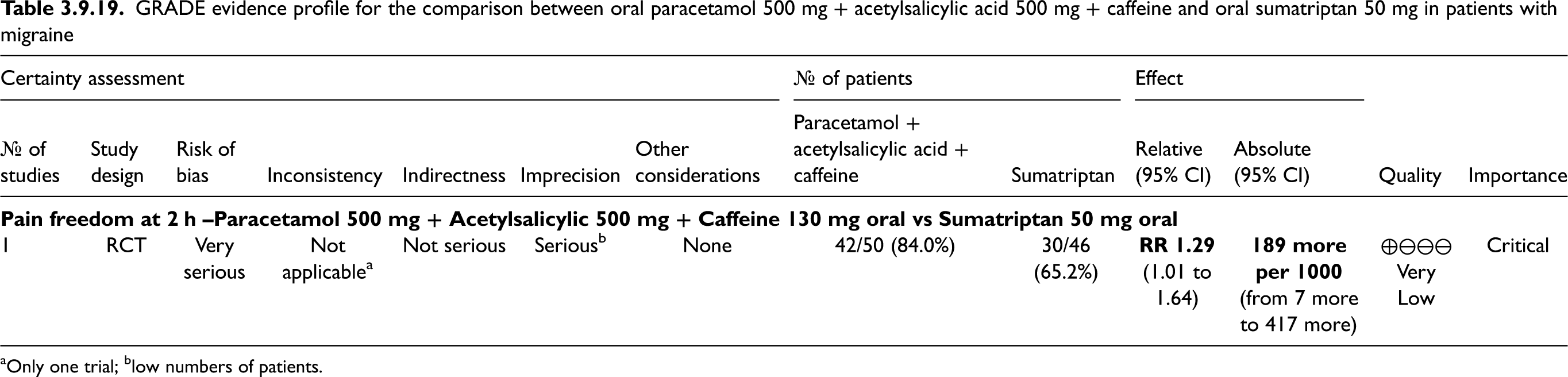
Only one trial; blow numbers of patients.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.23 In patients with migraine, we suggest oral paracetamol 500 mg + acetylsalicylic acid 500 mg + caffeine 130 mg over oral sumatriptan 50 mg for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)
Main evidence
None.
Additional evidence
We found one RCT comparing oral paracetamol 1000 mg + domperidone 20 mg with oral sumatriptan 50 mg (333) that did not meet the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was considered low (Figure 3.9.51). The RCT showed no difference between paracetamol 1000 mg + domperidone 20 mg and sumatriptan 50 mg considering the outcome of pain relief at 2 h (Figure 3.9.51). The quality of evidence was considered low (Figure 3.9.51).
Evidence-based recommendation for PICO 3.9.24 In patients with migraine, we suggest oral paracetamol 1000 mg + domperidone 20 mg and oral sumatriptan 50 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: =

Forest plot showing the comparison between oral paracetamol 1000 mg + domperidone 20 mg and oral sumatriptan 50 mg for the outcome pain relief at 2 h in patients with migraine.
Main evidence
None.
Additional evidence
We found one RCT comparing oral paracetamol 1000 mg + rizatriptan 10 mg with oral rizatriptan 10 mg alone (118) that did not meet the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was considered low (Figures 3.9.52 and 3.9.53). The RCT showed no difference between oral paracetamol 1000 mg + rizatriptan 10 mg and oral rizatriptan 10 mg alone considering the outcomes of pain freedom at 2 h (Figure 3.9.52) and pain relief at 2 h (Figure 3.9.53). The quality of evidence was considered very low due to the availability of only one RCT with low numbers of patients (<50 patients per group).
Evidence-based recommendation for PICO 3.9.25 In patients with migraine, we suggest oral paracetamol 1000 mg + rizatriptan 10 mg and oral rizatriptan 10 mg alone as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =

Forest plot showing the comparison between oral paracetamol 1000 mg + rizatriptan 10 mg and oral rizatriptan 10 mg alone for the outcome pain freedom at 2 h in patients with migraine.
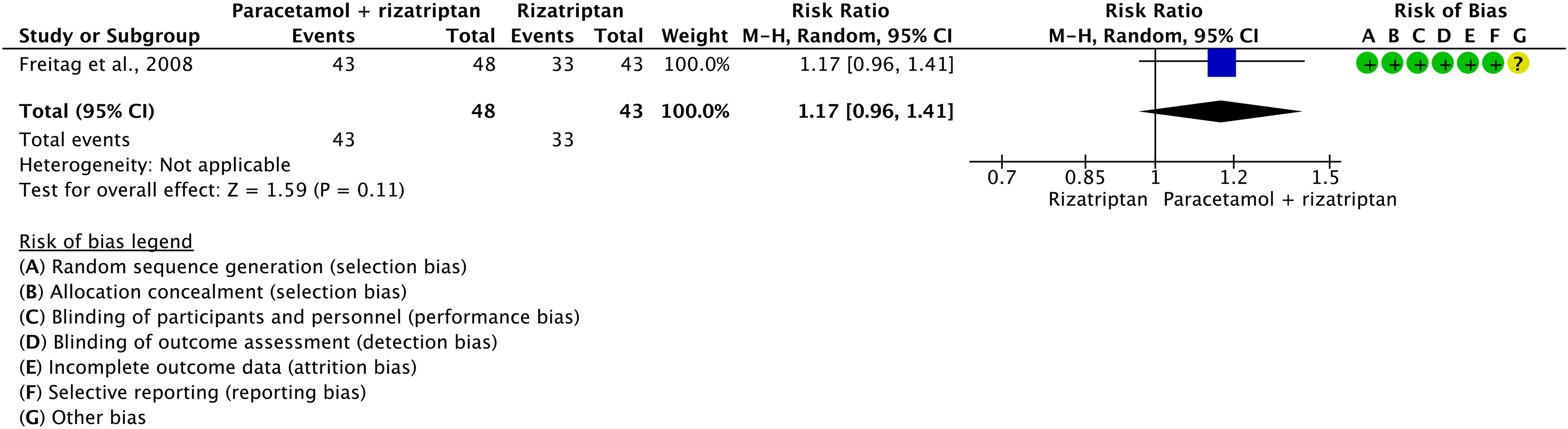
Forest plot showing the comparison between oral paracetamol 1000 mg + rizatriptan 10 mg and oral rizatriptan 10 mg alone for the outcome pain relief at 2 h in patients with migraine.
Main evidence
None.
Additional evidence
We found one RCT comparing oral almotriptan 12.5 mg + aceclofenac 100 mg with oral almotriptan 12.5 mg alone (344) that did not meet the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was considered unclear (Figure 3.9.54). The RCT showed no difference between oral almotriptan 12.5 mg + aceclofenac 100 mg and oral almotriptan 12.5 mg alone considering the outcome of pain freedom at 2 h (Figure 3.9.54). The quality of evidence was considered very low due to the availability of only one RCT with an unclear risk of bias and low numbers of patients (<50 in one of the groups).
Evidence-based recommendation for PICO 3.9.26 In patients with migraine, we suggest either oral almotriptan 12.5 mg + aceclofenac 10 mg or oral almotriptan 12.5 mg alone as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =
Forest plot showing the comparison between oral almotriptan 12.5 mg + aceclofenac 100 mg and oral almotriptan 12.5 mg alone for the outcome pain freedom at 2 h in patients with migraine.
Main evidence
We found two RCTs comparing oral acetylsalicylic acid 900 mg + metoclopramide 10 mg with oral sumatriptan 100 mg (187,194) that met the criteria for main evidence. The risk of bias was considered unclear (Figures 3.9.55 and 3.9.56). The RCTs showed no difference between oral acetylsalicylic acid 900 mg + metoclopramide 10 mg and oral sumatriptan 100 mg considering the outcome of pain freedom at 2 h (Figure 3.9.55) and pain relief at 2 h (Figure 3.9.56). The quality of evidence was considered moderate (Table 3.9.20) due the availability of RCTs with an unclear risk of bias.

Forest plot showing the comparison between oral acetylsalicylic acid 900 mg + metoclopramide 10 mg and oral sumatriptan 100 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral acetylsalicylic acid 900 mg + metoclopramide 10 mg and oral sumatriptan 100 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile for oral acetylsalicylic acid 900 mg + metoclopramide 10 mg versus oral sumatriptan 100 mg in patients with migraine

Additional evidence
None.
Evidence-based recommendation for PICO 3.9.27 In patients with migraine, we suggest oral acetylsalicylic acid + metoclopramide 10 mg and oral sumatriptan 100 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
We found one RCT comparing oral acetylsalicylic acid 900 mg + metoclopramide 10 mg with oral zolmitriptan 2.5 mg (177) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.57 and 3.9.58). The RCT showed no difference between oral acetylsalicylic acid 900 mg + metoclopramide 10 mg and oral zolmitriptan 2.5 mg considering the outcomes pain freedom at 2 h (Figure 3.9.57) and pain relief at 2 h (Figure 3.9.58). The quality of evidence was considered moderate (Table 3.9.21) due to the availability of only one RCT.
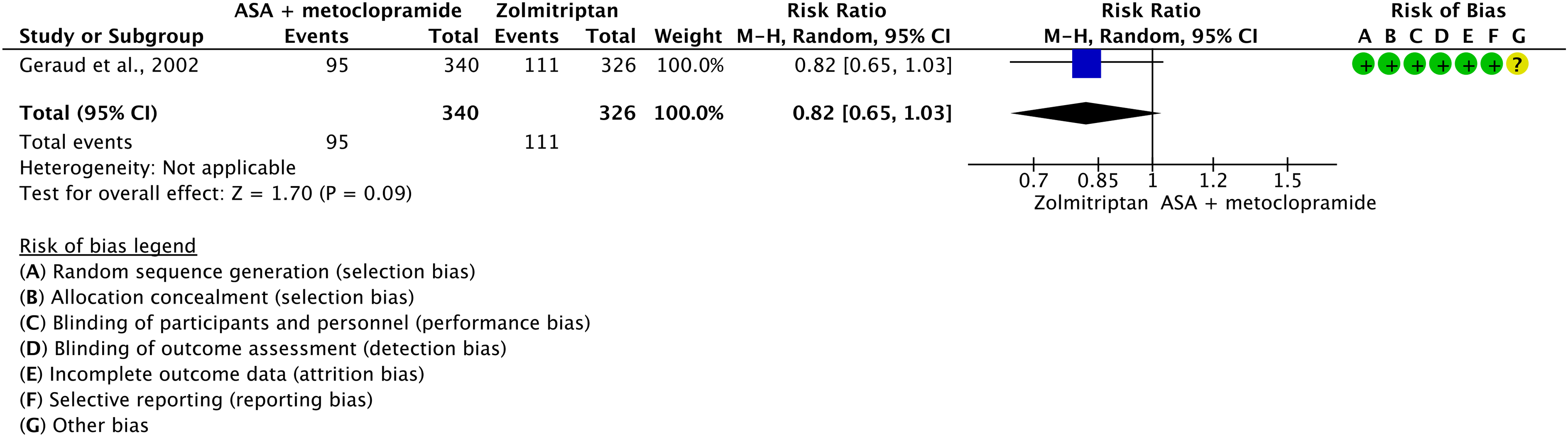
Forest plot showing the comparison between oral acetylsalicylic acid 900 mg + metoclopramide 10 mg and oral zolmitriptan 2.5 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral acetylsalicylic acid 900 mg + metoclopramide 10 mg and oral zolmitriptan 2.5 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile for oral acetylsalicylic acid 900 mg + metoclopramide 10 mg versus oral zolmitriptan 2.5 mg in patients with migraine
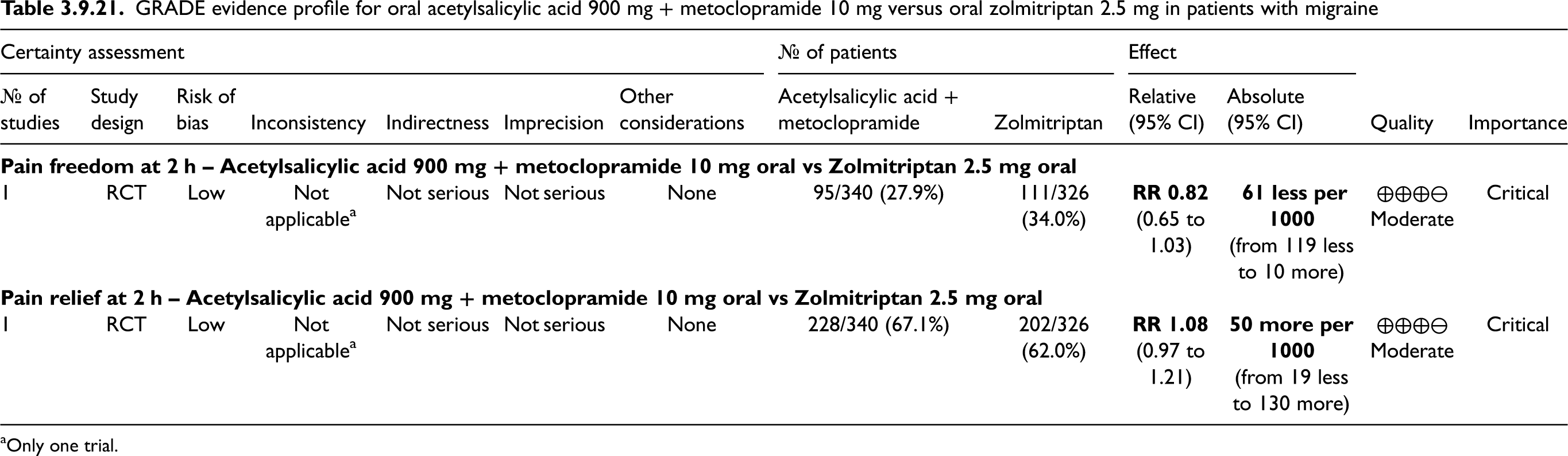
Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.28 In patients with migraine, we suggest acetylsalicylic acid + metoclopramide 10 mg and zolmitriptan 2.5 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
We found one RCT comparing oral ergotamine 2 mg + caffeine 200 mg with oral eletriptan (40 mg or 80 mg) (167) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.59 and 3.9.60). The RCT found that both dosages of oral eletriptan were superior to oral ergotamine 2 mg + caffeine 200 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.59) and pain relief at 2 h (Figure 3.9.60). The quality of evidence was considered moderate (Table 3.9.22) due to the availability of only one RCT.

Forest plot showing the comparison between oral ergotamine 2 mg + caffeine 200 mg and oral eletriptan (40 mg or 80 mg) for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between ergotamine 2 mg + caffeine 200 mg and eletriptan (40 mg or 80 mg) for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile for oral ergotamine 2 mg + caffeine 200 mg versus oral eletriptan (40 mg or 80 mg) in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.29 In patients with migraine, we recommend oral eletriptan (40 mg or 80 mg) over oral ergotamine 2 mg + caffeine 200 mg for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: strong (↑↑)
Main evidence
We found one RCT comparing oral ergotamine 2 mg + caffeine 200 mg with oral rizatriptan 10 mg (331) that met the criteria for main evidence. The risk of bias was considered high (Figures 3.9.61 and 3.9.62). The RCT found that oral rizatriptan 10 mg was superior to oral ergotamine 2 mg + caffeine 200 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.61) and pain relief at 2 h (Figure 3.9.62). The quality of evidence was considered very low (Table 3.9.23) due to the availability of only one RCT with a high risk of bias.
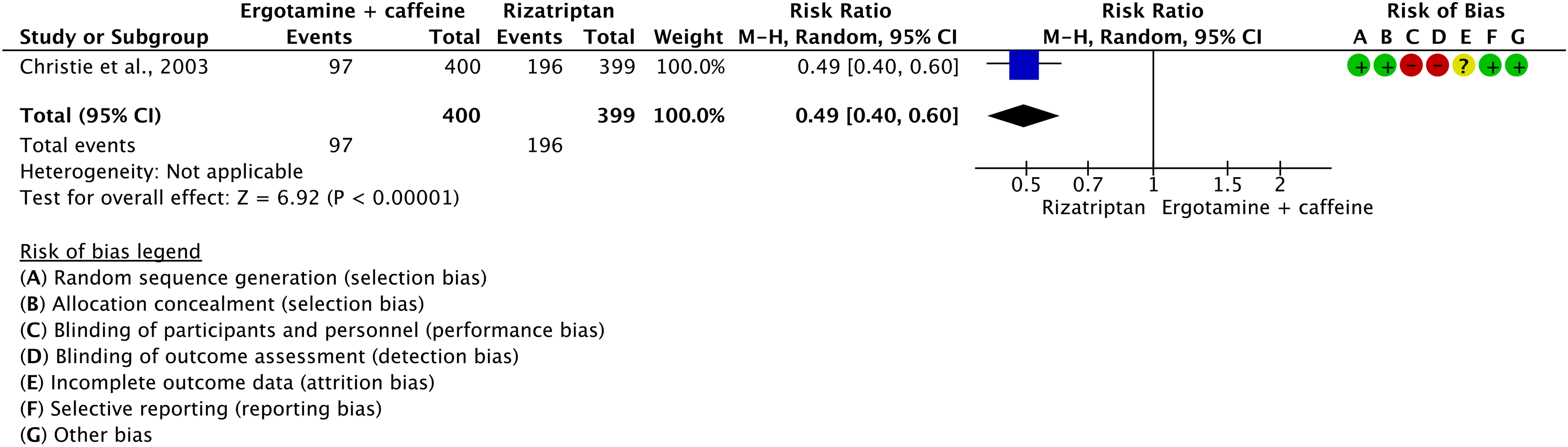
Forest plot showing the comparison between oral ergotamine 2 mg + caffeine 200 mg and oral rizatriptan 10 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral ergotamine 2 mg + caffeine 200 mg and oral rizatriptan 10 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile for oral ergotamine 2 mg + caffeine 200 mg versus oral rizatriptan 10 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.30 In patients with migraine, we recommend rizatriptan 10 mg over ergotamine 2 mg + caffeine 200 mg for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: strong (↑↑)
Main evidence
We found one RCT comparing oral ergotamine 2 mg + caffeine 200 mg with oral sumatriptan 100 mg (349) that met the criteria for main evidence. The risk of bias was considered high (Figures 3.9.63 and 3.9.64). The RCT found that oral sumatriptan 100 mg was superior to oral ergotamine 2 mg + caffeine 200 mg considering the outcomes of pain freedom at 2 h (Figure 3.9.63) and pain relief at 2 h (Figure 3.9.64). The quality of evidence was considered very low (Table 3.9.24) due to the availability of only one RCT with a high risk of bias.
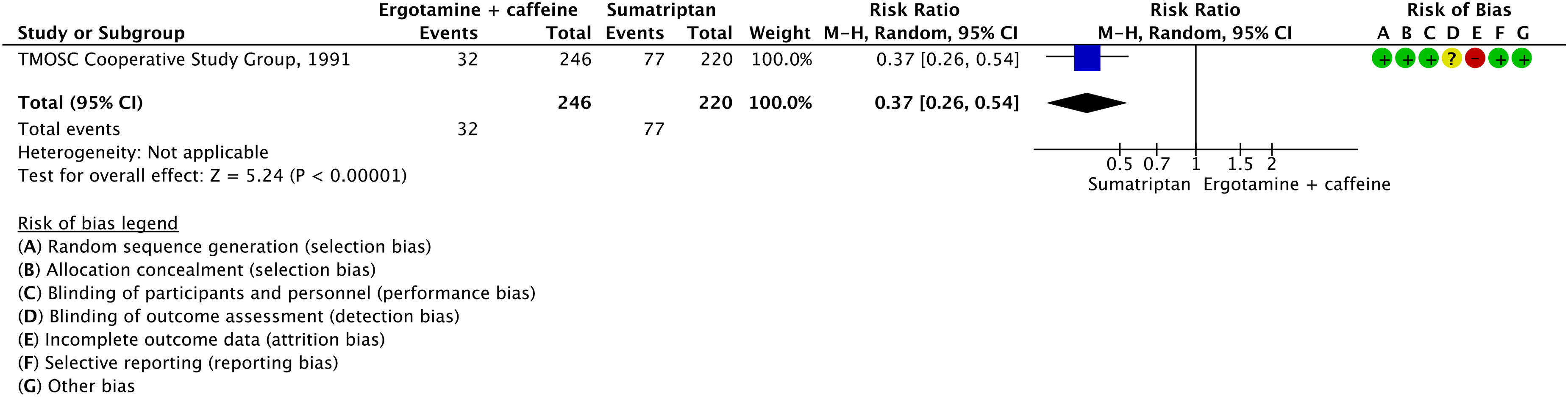
Forest plot showing the comparison between oral ergotamine 2 mg + caffeine 200 mg and oral sumatriptan 100 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral ergotamine 2 mg + caffeine 200 mg and oral sumatriptan 100 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile for oral ergotamine 2 mg + caffeine 200 mg versus oral sumatriptan 100 mg in patients with migraine
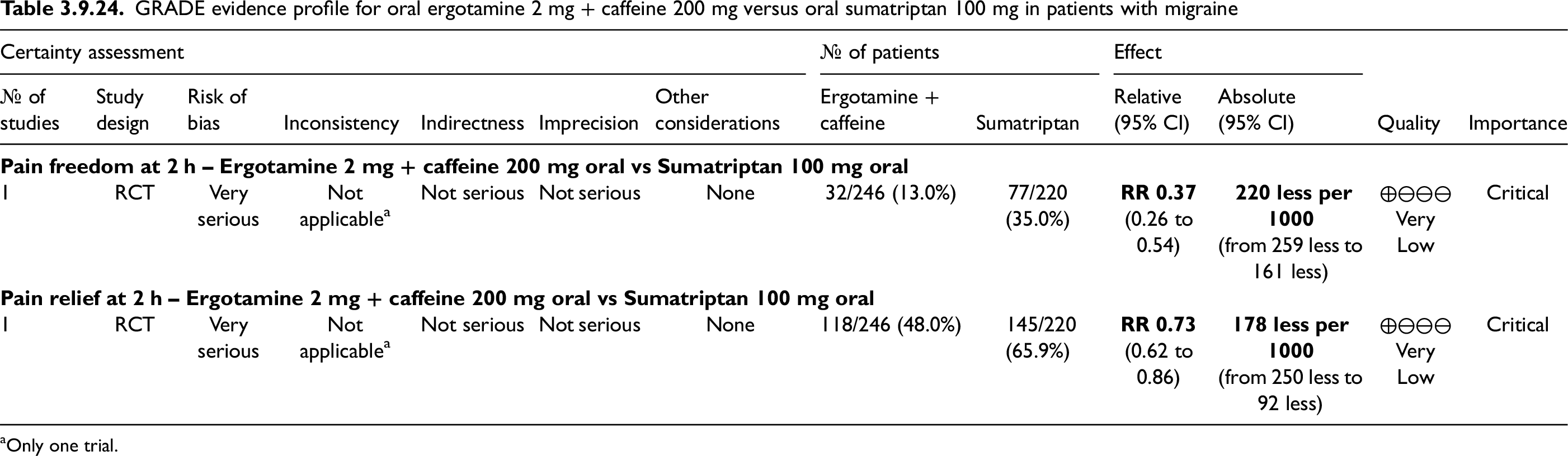
Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.31 In patients with migraine, we suggest oral sumatriptan 100 mg over oral ergotamine 2 mg + caffeine 200 mg for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: strong (↑↑)
Main evidence
None.
Additional evidence
We found one RCT comparing oral ergotamine + sumatriptan (unspecified dose) with oral sumatriptan (unspecified dose) (342) that did not meet the criteria for main evidence due to the lack of a clear hypothesis of superiority. The risk of bias was considered unclear (Figure 3.9.65). The RCT found that oral ergotamine + sumatriptan was superior to oral sumatriptan alone considering the outcome of pain freedom at 2 h (Figure 3.9.65). The quality of evidence was considered very low due to the availability of only one RCT with an unclear risk of bias.
Evidence-based recommendation for PICO 3.9.32 In patients with migraine, we suggest oral ergotamine + sumatriptan (unspecified dose) over sumatriptan alone (unspecified dose) for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊕⊖⊖)
Strength of the recommendation: Weak (↑)
[NOTE. This recommendation is based upon comparative efficacy data. Given the poor overall quality of evidence and the small number of subjects and the limited exposure they had to the combination ergotamine + sumatriptan (which does not allow to draw definite conclusion about the safety of use of the combination), the Guideline Committee considers sumatriptan alone a better choice than the combination with ergotamine.]

Forest plot showing the comparison between ergotamine + sumatriptan and sumatriptan for the outcome pain freedom at 2 h in patients with migraine.
Main evidence
We found one RCT comparing oral frovatriptan 2.5 mg + dexketoprofen 37.5 mg with frovatriptan 2.5 mg alone (559) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.66 and 3.9.67). The RCT found that oral frovatriptan 2.5 mg + dexketoprofen 37.5 mg was superior to oral frovatriptan 2.5 mg alone considering the outcomes of pain freedom at 2 h (Figure 3.9.66) and pain relief at 2 h (Figure 3.9.67). The quality of evidence was considered moderate (Table 3.9.25) due to the availability of only one RCT.

Forest plot showing the comparison between oral frovatriptan 2.5 mg + dexketoprofen 37.5 mg and oral frovatriptan 2.5 mg alone for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral frovatriptan 2.5 mg + dexketoprofen 37.5 mg and oral frovatriptan 2.5 mg alone for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for frovatriptan 2.5 mg + dexketoprofen 37.5 mg versus frovatriptan 2.5 mg alone in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.33 In patients with migraine, we suggest oral frovatriptan 2.5 mg + dexketoprofen 37.5 mg over oral frovatriptan 2.5 mg alone for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of recommendation:
Main evidence
We found one RCT comparing indomethacin 25 mg + prochlorperazine 4 mg + caffeine 75 mg suppositories and oral sumatriptan 25 mg (166) that met the criteria for main evidence. The risk of bias was high (Figures 3.9.68 and 3.9.69). The RCT showed that the suppositories of indomethacin 25 mg + prochlorperazine 4 mg + caffeine 75 mg were equivalent to oral sumatriptan 25 mg considering the outcomes pain freedom at 2 h (Figure 3.9.68) and pain relief at 2 h (Figure 3.9.69). The quality of evidence was considered very low (Table 3.9.26).
Forest plot showing the comparison between indomethacin 25 mg + prochlorperazine 4 mg + caffeine 75 mg suppositories and oral sumatriptan 25 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between indomethacin 25 mg + prochlorperazine 4 mg + caffeine 75 mg suppositories and oral sumatriptan 25 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for indomethacin 25 mg + prochlorperazine 4 mg + caffeine 75 mg suppositories versus oral sumatriptan 25 mg in patients with migraine

Only one trial.
We found a further RCT comparing oral indomethacin 25 mg + prochlorperazine 2 mg + caffeine 75 mg and oral sumatriptan 50 mg (189) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.70 and 3.9.71). The RCT showed no difference between oral indomethacin 25 mg + prochlorperazine 2 mg + caffeine 75 mg and oral sumatriptan 50 mg for the outcomes pain freedom at 2 h (Figure 3.9.70) and pain relief at 2 h (Figure 3.9.71). The quality of evidence was considered moderate (Table 3.9.27).

Forest plot showing the comparison between oral indomethacin 25 mg + prochlorperazine 2 mg + caffeine 75 mg and oral sumatriptan 50 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral indomethacin 25 mg + prochlorperazine 2 mg + caffeine 75 mg and oral sumatriptan 50 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral indomethacin 25 mg + prochlorperazine 2 mg + caffeine 75 mg versus oral sumatriptan 50 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendations for PICO 3.9.34 In patients with migraine, we suggest indomethacin + prochlorperazine + caffeine suppositories and oral sumatriptan (25 mg or 50 mg) as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =
In patients with migraine, we suggest oral indomethacin + prochlorperazine + caffeine and oral sumatriptan (25 mg or 50 mg) as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Main evidence
None.
Additional evidence
We found one RCT comparing oral isometheptene 65 mg + dichloralphenazone 100 mg + paracetamol 325 mg with oral sumatriptan 25 mg (334) that did not meet the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was considered low (Figure 3.9.72). The RCT found no difference between oral isometheptene 65 mg + dichoralphenazone 100 mg + paracetamol 325 mg and oral sumatriptan 25 mg considering the outcome of pain relief at 2 h (Figure 3.9.72). The quality of evidence was considered low.
Evidence-based recommendation for PICO 3.9.35 In patients with migraine, we suggest isometheptene 65 mg + dichloralphenazone 100 mg + paracetamol 325 mg and sumatriptan 25 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: =

Forest plot showing the comparison between oral isometheptene 65 mg + dichloralphenazone 100 mg + paracetamol 325 mg and oral sumatriptan 25 mg for the outcome pain relief at 2 h in patients with migraine.
Main evidence
We found one RCT comparing oral promethazine 25 mg + sumatriptan 50 mg with oral sumatriptan 50 mg alone (330) that met the criteria for main evidence. The risk of bias was considered low (Figures 3.9.73 and 3.9.74). The RCT showed that oral promethazine 25 mg + sumatriptan 50 mg was superior to oral sumatriptan 50 mg alone considering the outcomes pain freedom at 2 h (Figures 3.9.73) and pain relief at 2 h (Figure 3.9.74). The quality of evidence was considered moderate (Table 3.9.28) due to the availability of only one RCT.

Forest plot showing the comparison between oral promethazine 25 mg + sumatriptan 50 mg and oral sumatriptan 50 mg alone for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral promethazine 25 mg + sumatriptan 50 mg and oral sumatriptan 50 mg alone for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral promethazine 25 mg + sumatriptan 50 mg versus oral sumatriptan 50 mg alone in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.36 In patients with migraine, we suggest promethazine 25 mg + sumatriptan 50 mg over sumatriptan 50 mg alone for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Weak (↑)
Main evidence
None.
Additional evidence
We found one RCT comparing oral metoclopramide 10 mg + sumatriptan 50 mg with oral sumatriptan 50 mg alone (345) that did not meet the criteria for main evidence. The risk of bias was considered unclear (Figure 3.9.75). The RCT showed no difference between oral metoclopramide 10 mg + sumatriptan 50 mg and oral sumatriptan 50 mg alone considering the outcome pain relief at 2 h (Figure 3.9.75). The quality of evidence was considered very low due to the availability of only one RCT with an unclear risk of bias and low numbers of patients (<50 per group).
Evidence-based recommendation for PICO 3.9.37 In patients with migraine, we suggest oral metoclopramide 10 mg + sumatriptan 50 mg and oral sumatriptan 50 mg alone as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (=)

Forest plot showing the comparison between oral metoclopramide 10 mg + sumatriptan 50 mg and oral sumatriptan 50 mg alone for the outcome pain relief at 2 h in patients with migraine.
Main evidence
None.
Additional evidence
We found two RCTs comparing oral sumatriptan (50 mg or 85 mg) + naproxen 500 mg to oral sumatriptan (50 mg or 85 mg) alone (27,41) that did not meet the criteria for main evidence due to the lack of a clear hypothesis of superiority. The risk of bias was considered unclear or high (Figures 3.9.76 and 3.9.77). The RCTs showed that oral sumatriptan (50 mg or 85 mg) + naproxen 500 mg was superior to oral sumatriptan (50 mg or 85 mg) alone considering the outcomes pain freedom at 2 h (Figure 3.9.76) and pain relief at 2 h (Figure 3.9.77). The quality of evidence was considered low.
Evidence-based recommendation for PICO 3.9.38 In patients with migraine, we suggest oral sumatriptan (50 mg or 85) + naproxen 500 mg over oral sumatriptan (50 mg or 85 mg) alone for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Weak (↑)

Forest plot showing the comparison between oral sumatriptan (50 mg or 85 mg) + naproxen 500 mg and oral sumatriptan (50 mg or 85 mg) alone for the outcome pain freedom at 2 h in patients with migraine.
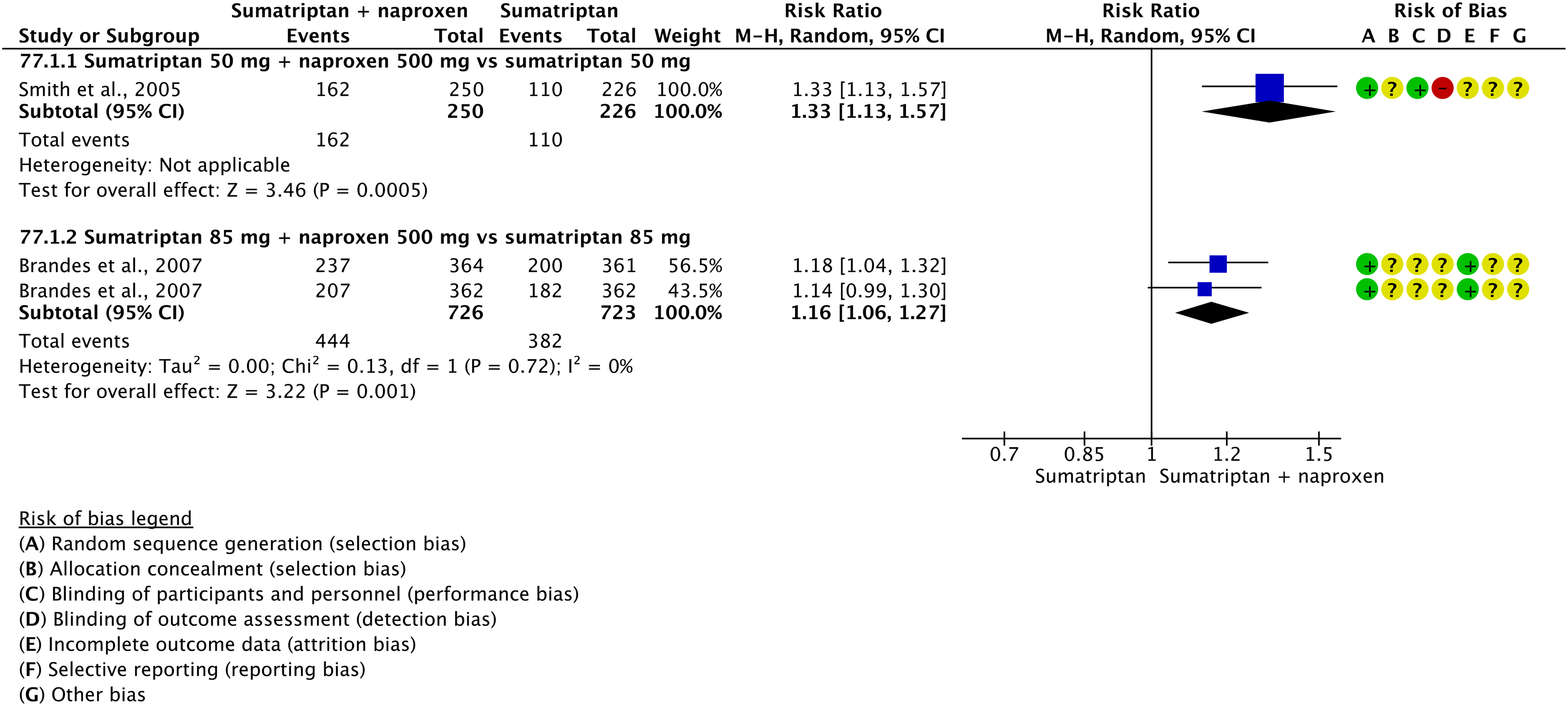
Forest plot showing the comparison between oral sumatriptan (50 mg or 85 mg) + naproxen 500 mg and oral sumatriptan (50 mg or 85 mg) alone for the outcome pain relief at 2 h in patients with migraine.
Main evidence
None.
Additional evidence
We found one RCT comparing oral trimebutine 200 mg + rizatriptan 10 mg with oral rizatriptan 10 mg alone (338) that did not meet the criteria for main evidence due to the lack of sample size calculation. The risk of bias was considered low (Figure 3.9.78). The RCT showed that oral trimebutine 200 mg + rizatriptan 10 mg was superior to oral rizatriptan 10 mg alone considering the outcome pain freedom at 2 h (Figure 3.9.78). The quality of evidence was considered low.
Evidence-based recommendation for PICO 3.9.39 In patients with migraine, we suggest oral trimebutine 200 mg + rizatriptan 10 mg over oral rizatriptan 10 mg alone for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Weak (↑)

Forest plot showing the comparison between oral trimebutine 200 mg + rizatriptan 10 mg and oral rizatriptan 10 mg alone for the outcome pain freedom at 2 h in patients with migraine.
Comparisons between combination analgesics and paracetamol
Main evidence
None.
Additional evidence
We found one RCT comparing oral rizatriptan 10 mg + paracetamol 1000 mg with oral paracetamol 1000 mg alone (118) that did not meet the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was considered low (Figures 3.9.79 and 3.9.80). The RCT showed that oral paracetamol 1000 mg + rizatriptan 10 mg was superior to oral paracetamol 1000 mg alone considering the outcomes pain freedom at 2 h (Figure 3.9.79) and pain relief at 2 h (Figure 3.9.80). The quality of evidence was considered very low due to the availability of only one RCT with low numbers of patients (<50 per group).
Evidence-based recommendation for PICO 3.9.40 In patients with migraine, we suggest oral paracetamol 1000 mg + rizatriptan 10 mg over oral paracetamol 1000 mg alone for the acute treatment of migraine attacks.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Weak (↑)

Forest plot showing the comparison between oral rizatriptan 10 mg + paracetamol 1000 mg and oral paracetamol 1000 mg alone for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral rizatriptan 10 mg + paracetamol 1000 mg and oral paracetamol 1000 mg alone for the outcome pain relief at 2 h in patients with migraine.
Comparisons between two combination analgesics
Main evidence
We found one RCT comparing oral calcium carbasalate (equivalent to acetylsalicylic acid 900 mg) + metoclopramide 10 mg and oral ergotamine 1 mg + caffeine 100 mg (339) that met the criteria for main evidence. The risk of bias was considered high (Figures 3.9.81 and 3.9.82). The RCT showed that oral calcium carbasalate + metoclopramide 10 mg was superior to oral ergotamine 1 mg + caffeine 100 mg considering the outcomes pain freedom at 2 h (Figure 3.9.81) and pain relief at 2 h (Figure 3.9.82). The quality of evidence was considered very low (Table 3.9.29) due to the availability of only one RCT with a high risk of bias.

Forest plot showing the comparison between oral calcium carbasalate + metoclopramide 10 mg and oral ergotamine 1 mg + caffeine 100 mg for the outcome pain freedom at 2 h in patients with migraine.

Forest plot showing the comparison between oral calcium carbasalate + metoclopramide 10 mg and oral ergotamine 1 mg + caffeine 100 mg for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral calcium carbasalate + metoclopramide 10 mg versus oral ergotamine 1 mg + caffeine 100 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.41 In patients with migraine, we suggest oral calcium carbasalate 900 mg + metoclopramide 10 mg over oral ergotamine 1 mg + caffeine 100 mg for the acute treatment of migraine attacks.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)
Main evidence
We found one RCT comparing oral sumatriptan 85 mg + naproxen 500 mg with oral paracetamol 325 mg + butalbital 50 mg + caffeine 40 mg (332) that met the criteria for main evidence. The risk of bias was considered low (Figure 3.9.83). The RCT showed no difference between oral sumatriptan 85 mg + naproxen 500 mg and oral paracetamol 325 mg + butalbital 50 mg + caffeine 40 mg considering the outcome of pain freedom at 2 h (Figure 3.9.83). The quality of evidence was considered moderate (Table 3.9.30) due to the availability of only one RCT.

Forest plot showing the comparison between oral sumatriptan 85 mg + naproxen 500 mg and oral paracetamol 325 mg + butalbital 50 mg + caffeine 40 mg for the outcome pain freedom at 2 h in patients with migraine.
GRADE evidence profile table for oral sumatriptan 85 mg + naproxen 500 mg versus oral paracetamol 325 mg + butalbital 50 mg + caffeine 40 mg in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.42 In patients with migraine, we suggest oral sumatriptan 85 mg + naproxen 500 mg and oral paracetamol 325 mg + butalbital 50 mg + caffeine 40 mg as equivalent options for the acute treatment of migraine attacks.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: =
Comparisons between combination analgesics and other analgesics
Main evidence
We found one RCT comparing oral acetylsalicylic acid 325 mg + butalbital 50 mg + codeine 30 mg + caffeine 40 mg with butorphanol 2 mg nasal spray (337) that met the criteria for main evidence due to the lack of a sample size calculation. The risk of bias was low (Figure 3.9.84). The RCT showed no difference between oral acetylsalicylic acid 325 mg + butalbital 50 mg + codeine 30 mg + caffeine 40 mg and butorphanol 2 mg nasal spray considering the outcome of pain relief at 2 h (Figure 3.9.84). The quality of evidence was considered moderate (Table 3.9.31) due to the availability of only one RCT.

Forest plot showing the comparison between oral acetylsalicylic acid 325 mg + butalbital 50 mg + codeine 30 mg + caffeine 40 mg and butorphanol 2 mg nasal spray for the outcome pain relief at 2 h in patients with migraine.
GRADE evidence profile table for oral acetylsalicylic acid 325 mg + butalbital 50 mg + codeine 30 mg + caffeine 40 mg versus butorphanol 2 mg nasal spray in patients with migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 3.9.43 In patients with migraine, efficacy data suggest the superiority of butorphanol 2 mg nasal spray over oral acetylsalicylic acid 325 mg + butalbital 50 mg + codeine 30 mg + caffeine 40 mg.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Weak (↑)
[NOTE: This recommendation is based on comparative efficacy data. However, given the safety risks of opioids and of barbiturates, the Guidelines Committee suggests caution in the use of both drugs.]
Summary of evidence for head-to-head comparisons of acute treatments for migraine
Figure 3.9.85 shows the summary of evidence for head-to-head comparisons of acute treatments for migraine attacks.

Summary of evidence for head-to-head comparisons of preventive treatments.
Expert-based opinion
Oral combination of paracetamol + acetylsalicylic acid + caffeine
Topic: Is the oral combination of paracetamol + acetylsalicylic acid + caffeine superior to each of the drugs alone for the acute treatment of migraine attacks?
We found one RCT comparing paracetamol 500 mg + acetylsalicylic acid 500 mg + caffeine 130 mg with ibuprofen 200 mg (350). The RCT did not meet the criteria for main evidence as it did not assess the outcomes of interest for the evidence-based guideline. In the RCT, oral paracetamol 500 mg + acetylsalicylic acid 500 mg + caffeine 130 mg showed a higher weighted sum of pain relief scores than oral ibuprofen 200 mg (p = 0.03) and an onset of pain relief 20 min earlier (p = 0.036).
Choice of acute treatments for migraine
Topic: Are there differences among NSAIDs used for the acute treatment of migraine attacks?
Topic: Which class of acute medications should be advised as a first approach in patients with migraine?
Topic: Should different acute medications be used in the same patient for the acute treatment of migraine attacks?
Topic: In patients with poor response to a triptan, is switching to a combination analgesic containing a triptan an effective option?
Topic: In patients with poor response to a triptan, is switching to a combination analgesic not containing a triptan an effective option?
Antidepressants
Introduction
Since the 1970s, antidepressants have been widely prescribed for migraine prevention, and still represent one of the most frequently used drug classes (561). Antidepressants share a complex mechanism of action, primarily involving the serotoninergic and noradrenergic pathways (562), and modulate the descending pain control system irrespective of their antidepressant effect (563).
A possible role in migraine prevention was first based on serendipity, considering their positive effects for treating several forms of chronic pain.
Tricyclics have been available since the 1950s and share a common three-ringed chemical structure associated with a secondary or tertiary amine. Their main mechanism of action involves the reuptake inhibition of serotonin and norepinephrine in the presynaptic terminals. They also act in the postsynaptic terminals as competitive antagonists of histaminergic, muscarinic cholinergic and adrenergic receptors (564). Reflecting their multifaceted actions, their clinical role in migraine prevention is probably linked to synergistic effects more than to a key single action (565).
Selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) have been available since the 1980s and have a more defined mechanism of action.
SSRIs inhibit the presynaptic serotonin transporter with different degrees of specificity and probably determine up and/or downregulation of serotonin transporters in different brain areas (566).
SNRIs exert a dual action inhibiting reuptake of both serotonin and norepinephrine, leading to an increased availability in the synaptic cleft (567). They consist of a group of different structurally unrelated molecules with a similar mechanism of action and slight differences in pharmacodynamic and pharmacokinetic properties (568).
Previous pre-clinical and clinical evidence supported the hypothesis of a complex monoamine neurotransmitters’ interplay in migraine pathophysiology (562).
The serotoninergic system involves several areas strictly related to migraine pathophysiology such as the raphe nuclei, the trigeminal nucleus caudalis, the thalamus, and the sensory cortex (569).
The same is true for the noradrenergic system, predominantly originating from the locus coeruleus in the dorsal rostral pons. It modulates different networks with both descending (as main source of noradrenergic innervation to the spinal dorsal horn, regulating nociception) and ascending projections (regulating nociception, stress response, arousal, and cognition) (570).
Serotonin fluctuates across the migraine cycle, leading to the hypothesis of an interictal low serotonin availability. Low plasma serotonin levels were described between attacks with a plasma peak during the ictal phase (571,572). In the ictal phase, an increased urinary excretion of 5-hydroxyindoleacetic acid (5-HIAA), the main serotonin metabolite, was also detected (573). The 5-HIAA metabolite was found to be increased in the cerebrospinal fluid of migraine patients, too (572).
Low monoaminergic brainstem activity was considered one of the pivotal mechanisms involved in the migraine-related phenomenon of lack of habituation (574). Noteworthy, habituation normalizes during attacks (575) and after administration of 5-HT reuptake inhibitors (576).
Finally, the role of serotonin is supported by the efficacy of acute and preventive drugs specifically acting on the serotoninergic system, namely triptans (5-HT1B/1D receptor agonists), ditans (5-HT1F agonists) and pizotifen (5-HT2A/2C antagonist), respectively.
The noradrenergic system showed an important repercussion on pain regulation and migraine, as well. Indeed, the antinociceptive actions of antidepressants are suppressed by the blockade of alpha-2 adrenoreceptor in animal models (577). In addition, detection of cerebral hypoperfusion after locus coeruleus stimulation in animal models could be a possible link to cortical spreading depression (578).
A role for serotonergic and noradrenergic pathways during the migraine cycle is also confirmed by neuroimaging evidence: the raphe nuclei was activated during spontaneous attacks and an altered connectivity in the dorsal rostral pons was recorded during pre-ictal and ictal phases (579–581).
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for the guideline on antidepressants for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 4.1.1.
Results
Overall, we retrieved 3416 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included seven systematic reviews and meta-analyses (561,582–587) that were considered as sources of RCTs. After analyzing full texts of these RCTs, we included eight of them in the quantitative synthesis (588–595) and four of them in the narrative synthesis (596–599) (Figure 4.1.1).

Meta-analysis selection flow chart - antidepressants.
Overall, we retrieved 2691 references from searching for RCTs and after removing duplicates we had 2550 references to analyze. However, considering the most recent included systematic review/meta-analysis on the topic, the analysis of additional RCTs was performed for papers published since 2016 for tricyclic antidepressants and since 2018 for SSRI/SNRI (1075 references). We finally included two additional RCTs (600,601) (Figure 4.1.2).

RCT selection flow chart - antidepressants. *trials published since 2018 (for SSRIs/SNRIs) and since 2016 (for tricyclics) were screened.
Overall, 14 RCTs (588–601) were included in the literature synthesis to develop the evidence-based guideline. Among these, 10 were included in quantitative synthesis (meta-analysis) and four in qualitative synthesis. Six RCTs (592–595,597,598) reported a comparison between an active drug and placebo and were presented in this section. Nine RCTs reported head-to-head comparisons that were included in Section 4.8.
Amitriptyline
Main evidence
None.
Additional evidence
We found two RCTs (592,593) comparing oral amitriptyline to placebo in subjects with migraine. These trials did not meet the criteria to be included in the main evidence. The RCTs did not show benefits for migraine prevention from oral amitriptyline administered with a gradual titration up to 100 mg daily. Overall, the risk of bias of included studies was high (Figure 4.1.3) and the quality of evidence was downgraded to very low.
Evidence-based recommendation for PICO 4.1.1 In subjects with migraine (no distinction between episodic and chronic), we suggest against oral amitriptyline 25–100 mg daily for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↓)
Forest plot showing the comparison between oral amitriptyline and placebo for the outcome ≥50% responder rate in patients with migraine (no distinction between episodic and chronic).
Main evidence
We found one RCT comparing oral amitriptyline 25 mg daily to placebo (595) in subjects with episodic migraine that met the criteria to be included in the main evidence.
This single study showed a significant improvement considering the outcome persisting monthly migraine days in patients treated with oral amitriptyline; no other outcomes were available. The study had a low risk of bias (Figure 4.1.4). The quality of evidence was considered moderate (Table 4.1.1).

Forest plot showing the comparison between oral amitriptyline and placebo for the outcome persisting monthly migraine days in patients with episodic migraine.
GRADE evidence profile table for oral amitriptyline versus placebo in patients with episodic migraine

Only one RCT.
Additional evidence
None.
Evidence-based recommendation for PICO 4.1.2 In subjects with episodic migraine, we suggest oral amitriptyline 25 mg daily for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of recommendation: Weak (↑)
[NOTE. The present recommendation, together with that of PICO 1, points out that amitriptyline is superior to placebo in subjects with episodic migraine, while its superiority to placebo is uncertain in subjects with chronic migraine.]
Safety and tolerability of amitriptyline
Its use should be avoided in pregnant and breastfeeding women, as these populations are not included in RCTs. Moreover, clinical data indicate that amitriptyline could cross the placenta and can be excreted in the milk with higher risk of congenital malformations (especially in the first trimester), lethargy and withdrawal symptoms (in the third trimester and during lactation) (603,604). Preventive therapies during pregnancy and breastfeeding should be limited to special situations. In cases of strong indication to continue its use during pregnancy, a careful evaluation of the risk/benefit ratio is mandatory to decide whether to continue or interrupt treatment. In case of its continuation during pregnancy, amitriptyline should be interrupted at least seven days before delivery for the above-mentioned risk of withdrawal symptoms (15). Extreme caution should be used in patients with a history of seizures because amitriptyline lowers the seizure threshold. Considering the risk of QT interval at electrocardiography prolongation and arrhythmias, attention is needed in subjects with long QT interval, arrhythmias and uncontrolled hypertension.
Duloxetine
Main evidence
None.
Additional evidence
We found one RCT comparing oral duloxetine 60 mg once a day to placebo (594) in subjects with episodic migraine that did not meet the criteria to be included in the main evidence. Overall, the risk of bias of the included RCT was not serious (Figure 4.1.5); however, the low numbers of participants included (<50 subjects per group) led to downgrading the quality of evidence to very low. This single study did not show benefits of duloxetine over placebo.
Evidence-based recommendation for PICO 4.1.3 In subjects with episodic migraine, we suggest against duloxetine 60 mg for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↓)

Forest plot showing the comparison between oral duloxetine 60 mg daily and placebo for the outcome changing in monthly headache days in patients with episodic migraine.
Evidence-based guideline summary
We found studies on amitriptyline and duloxetine to be included for deriving evidence-based recommendations for this guideline. No studies on other antidepressants met eligibility criteria.
For amitriptyline, evidence covered any migraine and episodic migraine. No studies were included for chronic migraine. For duloxetine, evidence covered episodic migraine. No studies were included for any migraine or chronic migraine.
There is moderate quality of evidence to suggest the use of amitriptyline for the prevention of episodic migraine. Data do not indicate benefits from duloxetine use (Figure 4.1.6). This evidence-based recommendation refers to subjects with migraine independently from the presence of comorbid depression or other conditions that may need treatment with antidepressants.

Summary of evidence-based recommendations on antidepressants for migraine prevention.
Expert-based opinion
Topic: Antidepressants in subjects with migraine and psychiatric or other pain comorbidities
Topic: Antidepressants other than amitriptyline
Optimization of amitriptyline use
Topic: Amitriptyline titration and dosing
In the real-world setting, dosages are generally lower than the ones tested in RCTs and should be gradually increased to reduce adverse events. Indeed, in clinical practice our experience is that doses higher than 25–50 mg per day are associated with a poor efficacy-to-tolerability ratio. Amitriptyline should be given at the lowest effective dose. Dose modifications should be accurately personalized balancing efficacy and adverse events (610). In subjects with comorbid depression, compared with those without, it might be reasonable to prescribe higher doses of amitriptyline, which are adjusted to treat comorbid depression. It is estimated that 40 mg daily of fluoxetine are equivalent to 122.3 mg daily of amitriptyline (611), which is a much higher dose than the 25 mg daily dose tested in subjects with migraine.
Introduction
Anti-seizure medications (ASMs) are the mainstay of therapy for epilepsy, and approximately 30 different ASMs with various mechanisms of action (MoA) are available to date (612,613). Several ASMs are approved treatments for non-epileptic conditions, including migraine, neuropathic pain, and psychiatric disorders (612,614). ASMs are usually characterized by a wide spectrum of therapeutic activities and different tolerability profiles.
The use of ASM in the treatment of migraine is based on their potential actions on the general modulation of pain systems or more specifically to systems involved in the pathophysiology of migraine. The specific mechanism(s) responsible for their anti-migraine efficacy is still unknown. Generally, ASMs act by stabilizing neuronal membrane potential through their effect on voltage and receptor-gated ion channels and reducing the release of pro-algesic neuropeptides (615).
Recent evidence suggests that ASMs might also exert their antimigraine activity by attenuating neurogenic inflammation in the trigeminal vascular system or directly in the release of CGRP, or by several additional MoAs in the central and peripheral nervous system (616). Numerous ASMs have been tested for migraine prevention by RCTs and open-label studies, with very different and sometimes inconsistent results. Only two ASMs, sodium valproate/divalproex sodium and topiramate, are currently approved for migraine prevention. Indeed, although several other ASMs were specifically investigated for migraine in different clinical trial phases (lamotrigine, zonisamide, gabapentin, pregabalin, carbamazepine, oxcarbazepine, levetiracetam, perampanel, lacosamide, carisbamate, clonazepam, tiagabine and vigabatrin), none of them have indication for migraine/headache (616).
Clinical practice guidelines for using ASMs for migraine treatment must consider the evidence regarding their efficacy, tolerability, and adverse events, including the risk of teratogenicity, which is particularly important considering that migraine is most prevalent in women of childbearing potential.
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for the Guideline on ASMs for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 4.2.1.
Results
Overall, we retrieved 1662 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening, we included seven systematic reviews and meta-analyses (617–623) as sources of RCTs. After analyzing full texts of these RCTs, we included 33 of them in the quantitative synthesis (596,624–655) (Figure 4.2.1).

Meta-analysis selection flow chart - anti-seizure medications.
Considering the most recently included systematic review/meta-analysis on the topic, the analysis of additional RCTs was performed for papers published since June 2020. Overall, we retrieved 1744 references from searching for RCTs and after removing duplicates we had three references to analyze (656–658). From the literature search updates performed in May and November 2023, we included 9 additional RCTs (656–664) (Figure 4.2.2).

RCT selection flow chart - anti-seizure medications. *trials published since June 2020 were screened.
Overall, 42 studies were considered for inclusion in the quantitative synthesis (meta-analyses) to develop the evidence-based guideline. However, 11 trials were excluded because they included medications that are not available on the market, the comparison was not pertinent, or they did not compare the ASM with another medication or placebo. Among the remaining 31 RCTs, 16 reported a comparison between active drugs and placebo and were presented in this section, while the 15 RCTs reporting head-to-head comparisons only are presented in Section 4.8.
Topiramate
Main evidence
We found four RCTs comparing topiramate to placebo in subjects with episodic migraine (625,628,630,664) that met the criteria to be included in the main evidence.
The pooled analysis of two RCTs (630,664) showed a significant improvement considering the outcome persisting monthly migraine days in patients treated with oral topiramate 100 mg and 200 mg daily as compared to placebo (Figure 4.2.3), while results did not show benefit of oral topiramate 50 mg daily over placebo for the same outcome. The quality of evidence for the outcome persisting monthly migraine days was considered low for the 50 mg daily dose and moderate for the 100 and 200 mg daily doses (Table 4.2.1).

Forest plot showing the comparison between oral topiramate (50, 100 or 200 mg) and placebo for the outcome persisting monthly migraine days in patients with episodic migraine.
GRADE evidence profile table of oral topiramate versus placebo in patients with episodic migraine

Only one trial.
Three RCTs reported the outcome change in monthly migraine days (625,628,630) (Figure 4.2.4). The pooled analysis of those studies showed benefits of oral topiramate 100 mg daily but not of oral topiramate 200 mg daily over placebo. No data for this outcome was available for the 50 mg daily dose. The quality of evidence for the outcome change in monthly migraine days was considered moderate for the 100 dose and low for the 200 mg dose (Table 4.2.1).

Forest plot showing the comparison between oral topiramate (100 or 200 mg) and placebo for the outcome change in monthly migraine days in patients with episodic migraine.
Two studies reported the outcome ≥50% responder rate (628,630). The pooled analysis of the two studies showed benefits of oral topiramate 100 and 200 mg daily dose over placebo (Figure 4.2.5). Only one of them addressed the 50 mg daily dose and showed benefits of the active treatment over placebo. The quality of evidence for the outcome ≥50% response rate was considered low for the 50 mg dose and moderate for the 100 and 200 mg dose (Table 4.2.1).

Forest plot showing the comparison between oral topiramate (50, 100 or 200 mg) and placebo for the outcome ≥50% response rate in patients with episodic migraine.
Additional evidence
We found three RCTs comparing topiramate to placebo in subjects with episodic migraine (627,629,638) that did not meet the criteria to be included in the main evidence. Overall, the risk of bias of included studies was very serious (Figures 4.2.6–4.2.8).
Forest plot showing the comparison between oral topiramate 200 mg and placebo for the outcome persisting monthly migraine days in patients with episodic migraine.

Forest plot showing the comparison between oral topiramate 50 mg and placebo for the outcome change in monthly migraine days in patients with episodic migraine.

Forest plot showing the comparison between oral topiramate (100 or 200 mg) and placebo for the outcome ≥50% response rate in patients with episodic migraine.
One study showed significant benefits of topiramate 50 mg daily over placebo considering the outcome change in monthly migraine days (629) (Figure 4.2.7), and another study showed significant benefits of topiramate 100 mg over placebo for the outcome ≥50% response rate (638) (Figure 4.2.8). The third study did not show significant benefits of topiramate 200 mg daily over placebo considering the outcomes persisting monthly migraine days (Figure 6) and ≥50% response rate (627) (Figure 4.2.8).
Evidence-based recommendation for PICO 4.2.1
1) In subjects with episodic migraine, we recommend oral topiramate 100 mg daily for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Strong (↑↑)
2) In subjects with episodic migraine, we recommend oral topiramate 200 mg daily for migraine
prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Strong (↑↑)
3) In subjects with episodic migraine, we suggest oral topiramate 50 mg daily for migraine prevention.
Strength of the recommendation: Weak (↑)
Quality of evidence: Low (⊕⊕⊖⊖)
Main evidence
We found one RCT comparing topiramate to placebo in subjects with chronic migraine (660) that met the criteria to be included in the main evidence. This study showed a significant improvement considering the outcome change in monthly migraine days in subjects treated with oral topiramate 200 mg daily as compared to placebo (Figure 4.2.9), while the results did not show benefit of oral topiramate 200 mg over placebo for the outcome 50% response (Figure 4.2.10). The quality of evidence for the outcomes change in monthly migraine days and ≥50% response was considered low (Table 4.2.2).

Forest plot showing the comparison between oral topiramate 200 mg and placebo for the outcome change in monthly migraine days in patients with chronic migraine.
Forest plot showing the comparison between oral topiramate 200 mg and placebo for the outcome ≥50% response rate in patients with chronic migraine.
GRADE evidence profile table of oral topiramate versus placebo in patients with chronic migraine

Only one trial; bSmall sample size (<50 patients per group).
Additional evidence
We found three RCTs comparing topiramate to placebo in subjects with chronic migraine (626,647,661) that did not meet the criteria to be included in the main evidence. Overall, the risk of bias of included studies was very serious (Figure 4.2.11–4.2.13) and for this reason the quality of evidence was considered very low.

Forest plot showing the comparison between oral topiramate (50 and 100 mg) and placebo for the outcome persisting monthly migraine days in patients with chronic migraine.

Forest plot showing the comparison between oral topiramate 100 mg and placebo for the outcome change in monthly migraine days in patients with chronic migraine.
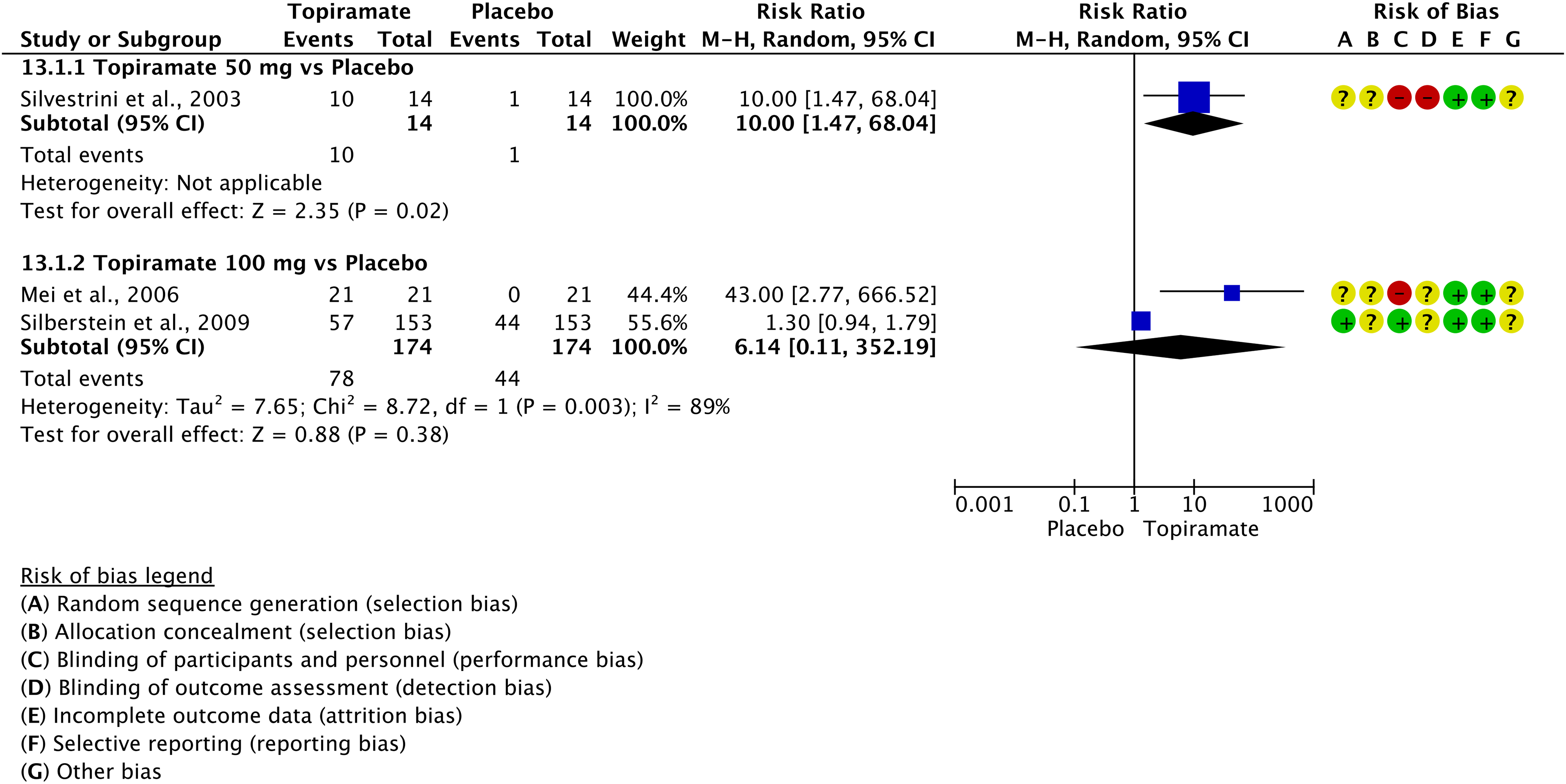
Forest plot showing the comparison between oral topiramate (50 and 100 mg) and placebo for the outcome ≥50% response rate in patients with chronic migraine.
Two RCTs showed significant benefits of oral topiramate 50 and 100 mg daily over placebo considering the outcome persisting monthly migraine days (626,661) (Figure 4.2.11), while the third RCT did not show significant benefit of 100 mg daily over placebo considering the outcome change in monthly migraine days (647) (Figure 4.2.12).
The three RCTs considered the outcome ≥50% response rate and one (626) showed significant benefit of oral topiramate 50 mg over placebo, while the pooled analysis of the other two RCTs (647,661) did not show significant improvement with oral topiramate 100 mg over placebo (Figure 4.2.13).
Evidence-based recommendation for PICO 4.2.2
1) In subjects with chronic migraine, we suggest oral topiramate 50 mg daily for migraine prevention.
Strength of recommendation: Weak (↑)
2) In subjects with chronic migraine, we suggest oral topiramate 100 mg daily for migraine prevention.
Strength of recommendation: Weak (↑)
3) In subjects with chronic migraine, we suggest oral topiramate 200 mg daily for migraine prevention.
Strength of recommendation: Weak (↑)
Quality of evidence: Very low (⊕⊖⊖⊖)
Quality of evidence: Very low (⊕⊖⊖⊖)
Quality of evidence: Low (⊕⊕⊖⊖)
Safety and tolerability of topiramate
Valproate
Main evidence
None.
Additional evidence
We found three RCTs comparing valproate to placebo in subjects with episodic migraine (632,633,639) that did not meet the criteria to be included in the main evidence. Overall, the risk of bias of included studies was very serious (Figures 4.2.14 and 4.2.15) and for this reason the quality of evidence was downgraded to very low.

Forest plot showing the comparison between oral valproate 1500 mg and placebo for the outcome change in monthly migraine days in patients with episodic migraine.
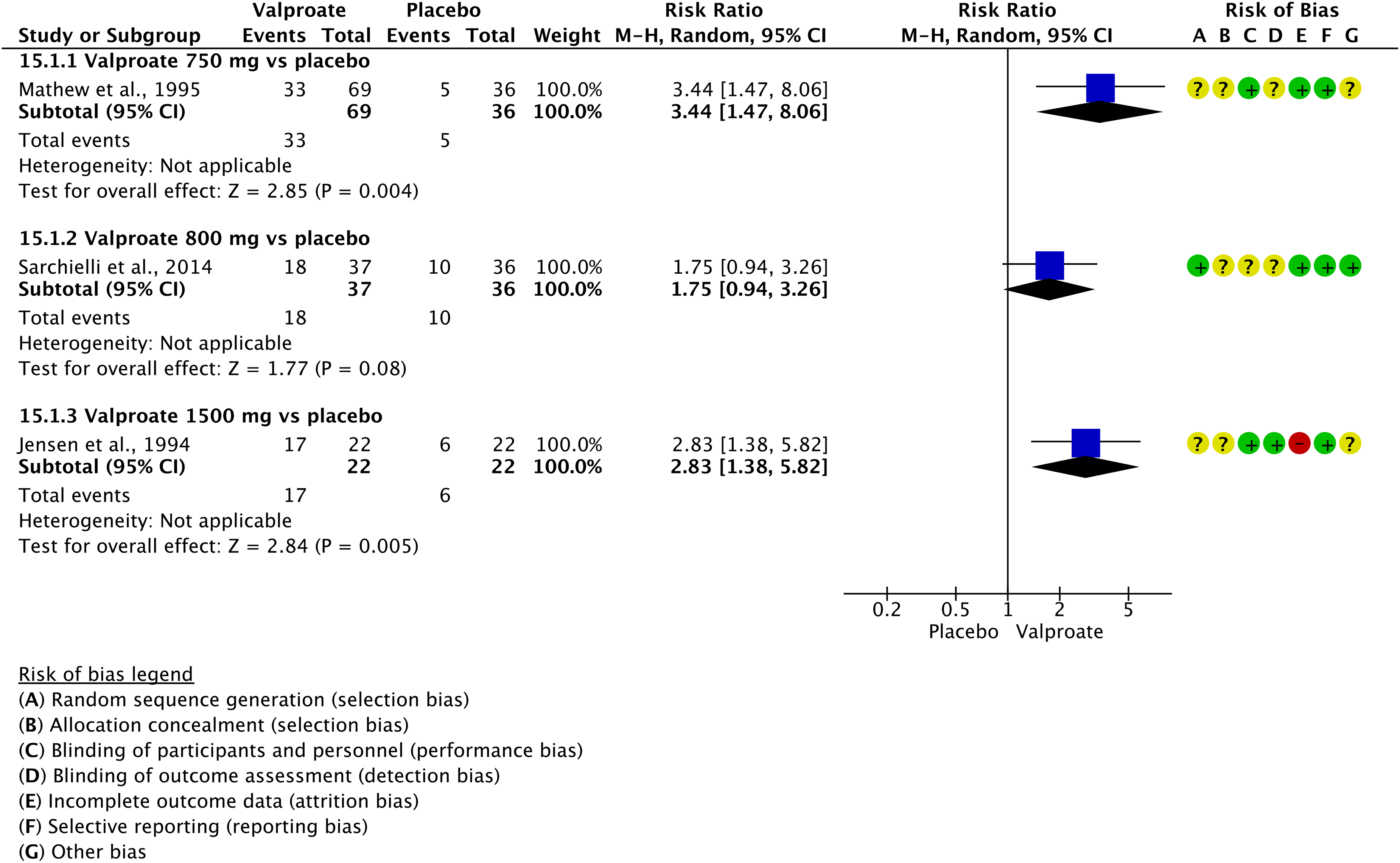
Forest plot showing the comparison between oral valproate (750, 800 or 1500 mg) and placebo for the outcome ≥50% response rate in patients with episodic migraine.
One RCT considered the outcome change in monthly migraine days and showed significant benefit of oral valproate 1500 mg daily over placebo (633) (Figure 4.2.14), even if with low numbers of participants (<50 per group).
Two RCTs showed significant benefits of valproate 750 mg and 1500 mg daily over placebo considering the outcome ≥50% response rate (632,639), while the third RCT (633) did not show significant benefit of valproate 800 mg daily over placebo considering the same outcome (Figure 4.2.15).
Evidence-based recommendation for PICO 4.2.3
1) In subjects with episodic migraine, we suggest oral valproate 750 mg daily for migraine prevention.
Strength of recommendation: weak (↑)
2) In subjects with episodic migraine, there is not enough evidence to establish the efficacy of oral valproate 800 mg daily for migraine prevention. Strength of recommendation: -.
3) In subjects with episodic migraine, we suggest oral valproate 1500 mg daily for migraine prevention.
Strength of recommendation: weak (↑)
Quality of evidence: very low (⊕⊖⊖⊖)
Quality of evidence: Very low (⊕⊖⊖⊖)
Quality of evidence: very low (⊕⊖⊖⊖)
Main evidence
We found two RCTs comparing oral valproate to placebo in subjects with migraine (no distinction between episodic and chronic) (636,651) that met the criteria to be included in the main evidence. The RCTs showed a significant improvement considering the outcome ≥50% response rate in subjects treated with oral valproate 500 mg and 1500 mg daily compared to placebo, while results did not show benefit of oral valproate 1000 mg over placebo for the same outcome (Figure 4.2.16). The quality of evidence for the outcome ≥50% response rate was considered very low (Table 4.2.3).

Forest plot showing the comparison between valproate (500, 1000 or 1500 mg) and placebo for the outcome ≥50% response rate in patients with migraine (no distinction between episodic and chronic).
GRADE evidence profile for oral valproate versus placebo in patients with migraine

only one trial; bSmall sample size (<50 per group); cconfidence interval crossing the threshold for clinical decision.
Additional evidence
We found two RCTs comparing oral valproate to placebo in subjects with migraine (no distinction between episodic and chronic) (631,653) that did not meet the criteria to be included in the main evidence. Overall, the risk of bias of included studies was very serious (Figures 4.2.17 and 4.2.18) and for this reason the quality of evidence was downgraded to very low. No recommendation could be issued given the low numbers of subjects (<50 per group) in each RCT.
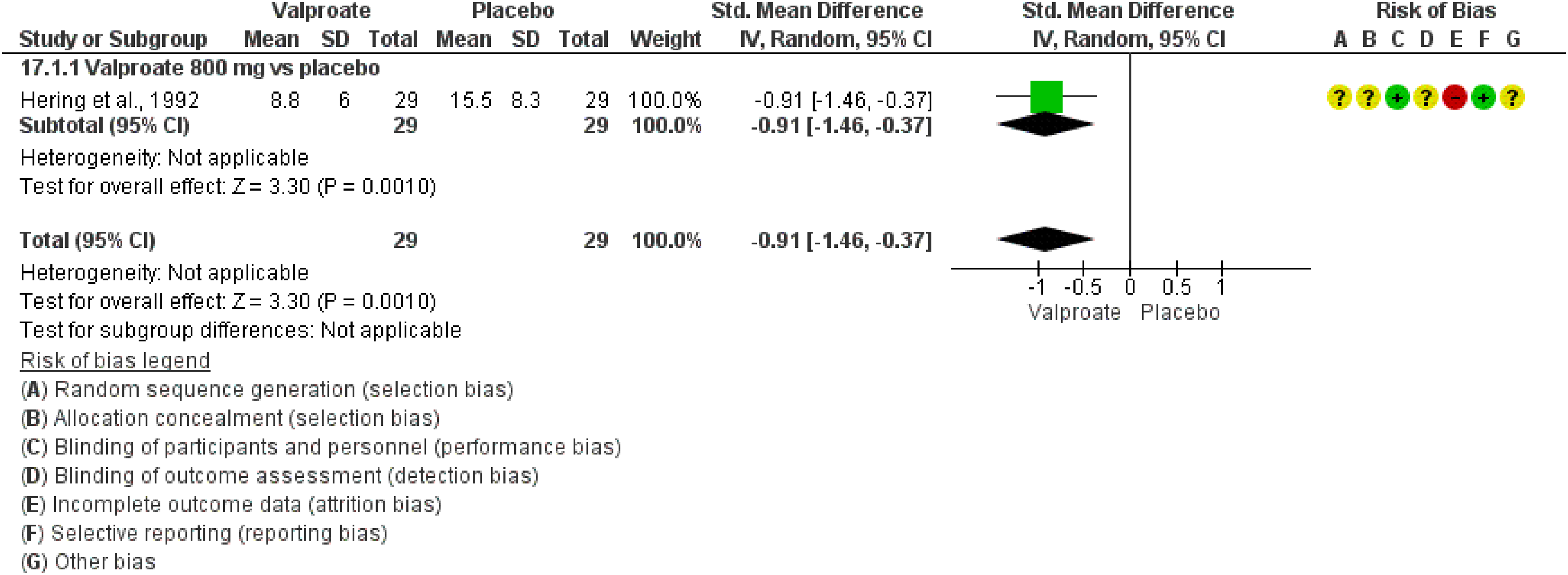
Forest plot showing the comparison between oral valproate 800 mg and placebo for the outcome persisting monthly migraine days in patients with migraine (no distinction between episodic and chronic).
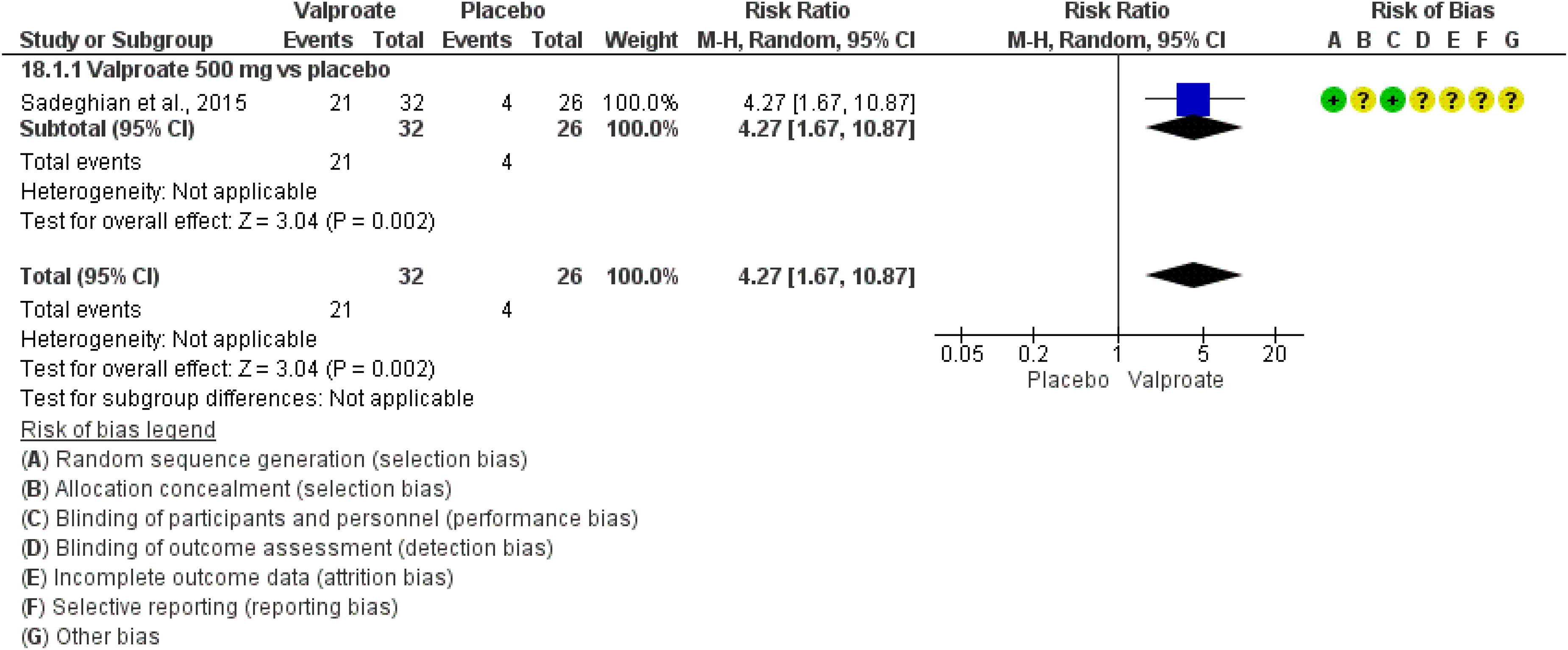
Forest plot showing the comparison between oral valproate 500 mg and placebo for the outcome ≥50% response rate in patients with migraine (no distinction between episodic and chronic).
One RCT considered the outcome persisting monthly migraine days (631) and showed significant benefit of oral valproate 800 mg daily over placebo (Figure 4.2.17).
One study considered the outcome ≥50% response rate (653) and showed significant benefit of valproate 500 mg daily over placebo (Figure 4.2.18).
Evidence-based recommendation for PICO 4.2.4 In subjects with migraine (no distinction between episodic and chronic), we suggest oral valproate (500–1500 mg) daily for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↑)
Safety and tolerability of valproate
Gabapentin
Main evidence
We found one RCT comparing gabapentin to placebo in subjects with migraine (635) that met the criteria to be included in the main evidence. The RCT considered only the outcome ≥50% response rate. The results did not show benefit of oral gabapentin 1200 to 3000 mg daily over placebo (Figure 4.2.19).

Forest plot showing the comparison between oral gabapentin (1200–3000 mg) and placebo for the outcome ≥50% response rate in patients with episodic migraine.
The overall quality of evidence for the outcome was considered very low (Table 4.2.4).
GRADE evidence profile table of gabapentin versus placebo in patients with episodic migraine
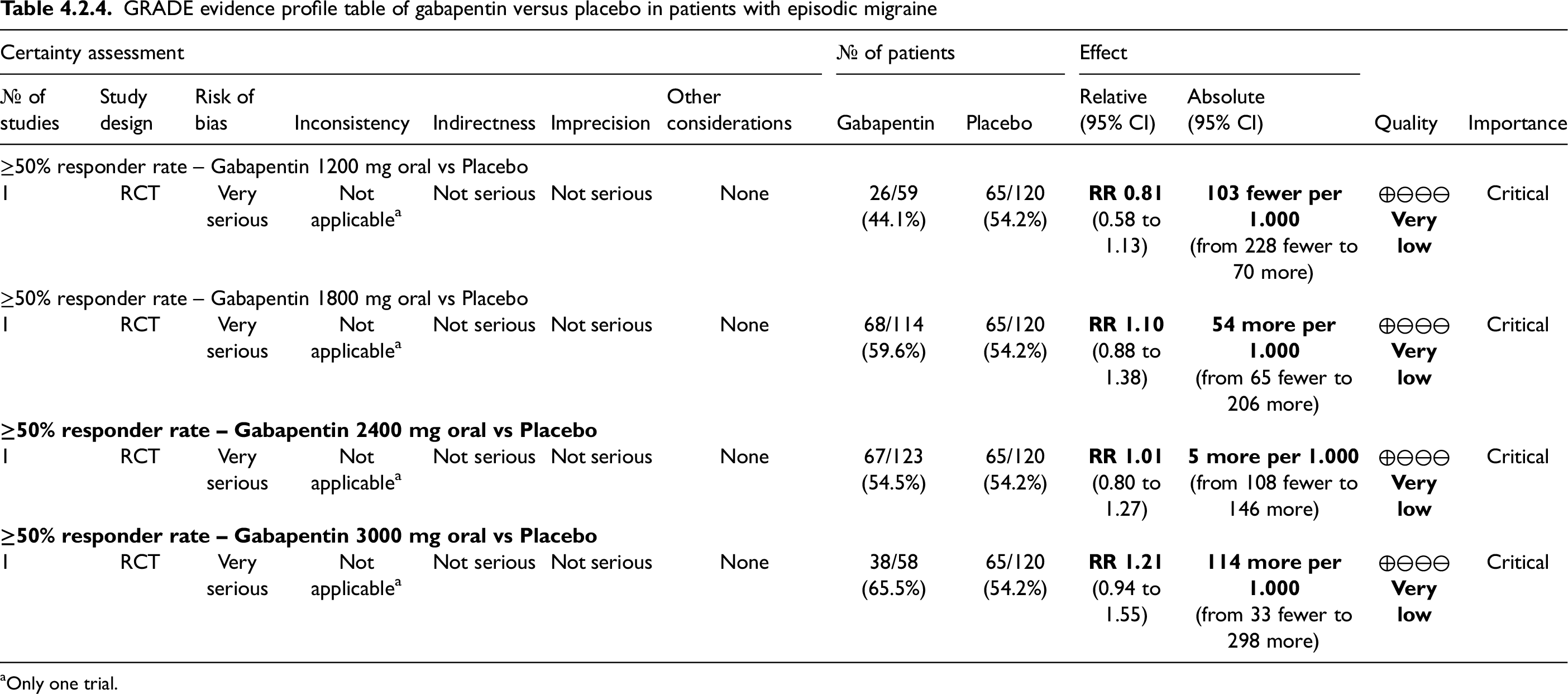
Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.2.5 In subjects with episodic migraine, we suggest against gabapentin for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↓)
Lamotrigine
Main evidence
We found two RCTs comparing lamotrigine to placebo in subjects with episodic migraine (638,641) that met the criteria to be included in the main evidence. One RCT (641) did not show any benefit of oral lamotrigine 200 mg daily over placebo for the outcome persisting monthly migraine days (Figure 4.2.20) whereas the other RCT showed significant improvement in the outcome change in monthly migraine days for oral lamotrigine 50 over placebo (638) (Figure 4.2.21). The overall quality of evidence for the outcome was considered very low for both persisting monthly migraine days and change in monthly migraine days (Table 4.2.5).
Forest plot showing the comparison between oral lamotrigine 200 mg and placebo for the outcome persisting monthly migraine days in patients with episodic migraine.

Forest plot showing the comparison between oral lamotrigine 50 mg and placebo for the outcome change in monthly migraine days in patients with episodic migraine.
GRADE evidence profile of oral lamotrigine versus placebo in patients with episodic migraine

Only one trial; bSmall sample size (<50 patients per group).
Additional evidence
None.
Evidence-based recommendation for PICO 4.2.6 In subjects with episodic migraine, we suggest oral lamotrigine 50 mg for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)
[NOTE: The suggestion in favor of lamotrigine was based upon the RCT that provided the most precise results. The RCT showing no benefit of lamotrigine over placebo had imprecise results. Further guidance on the use of lamotrigine for migraine prevention is provided in the Expert-Based Opinion Section.]
Levetiracetam
Main evidence
We found one RCT comparing oral levetiracetam to placebo in subjects with episodic migraine (640) that met the criteria to be included in the main evidence. The RCT considered only the outcome persisting monthly migraine days. The results showed significant benefit from oral levetiracetam 1000 mg daily over placebo (Figure 4.2.22).

Forest plot showing the comparison between oral levetiracetam 1000 mg and placebo for the outcome persisting monthly migraine days in patients with episodic migraine.
The overall quality of evidence for the outcome was considered very low (Table 4.2.6).
GRADE evidence profile of oral levetiracetam versus placebo in episodic migraine prevention

Relevant risk of bias detected in available RCTs; bonly one trial; cSmall sample size.
Additional evidence
None.
Evidence-based recommendation for PICO 4.2.7 In subjects with episodic migraine, we suggest oral levetiracetam 1000 mg for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)
[NOTE: The results from the included RCT are imprecise due to the low number of included subjects. Nevertheless, a recommendation was issued as the RCT was included in main evidence. Additional guidance is provided in the expert-based opinion section.]
Main evidence
None.
Additional evidence
We found one RCT comparing oral levetiracetam to placebo in subjects with migraine (no distinction between episodic and chronic) (653) that did not meet the criteria to be included in the main evidence. The risk of bias for this study was very serious (Figure 4.2.23) and for this reason the quality of evidence was downgraded to very low. This study considered the outcome ≥50% response rate and showed significant benefit of oral levetiracetam 500 mg daily over placebo (Figure 4.2.23). A recommendation could not be issued given the low numbers of participants (<50 per group).
Evidence-based recommendation for PICO 4.2.8 In subjects with migraine (no distinction between episodic and chronic), there is not enough evidence to recommend levetiracetam over placebo for migraine prevention.
Quality of evidence: -
Strength of the recommendation: -

Forest plot showing the comparison between oral levetiracetam 500 mg and placebo for the outcome ≥50% response rate in patients with migraine (no distinction between episodic and chronic).
Vigabatrin
Main evidence
None.
Additional evidence
We found one RCT comparing oral vigabatrin to placebo in subjects with migraine (no distinction between episodic and chronic) (650) that did not meet the criteria to be included in the main evidence. The risk of bias for this RCT was very serious (Figure 4.2.24) and for this reason the quality of evidence was downgraded to very low.
The RCT did not show benefits of oral vigabatrin 2000 mg daily over placebo considering the outcome persisting monthly migraine days (Figure 4.2.24). A recommendation could not be issued given the low numbers of subjects (<50 per group).
Evidence-based recommendation for PICO 4.2.9 In subjects with migraine (no distinction between episodic and chronic), there is not enough evidence to recommend vigabatrin over placebo for migraine prevention.
Quality of evidence: -
Strength of recommendation: -

Forest plot showing the comparison between oral vigabatrin 2000 mg and placebo for the outcome persisting monthly migraine days in patients with migraine (no distinction between episodic and chronic).
Oxcarbazepine
Main evidence
We found one RCT comparing oral oxcarbazepine to placebo in subjects with migraine (no distinction between episodic and chronic) (637) that met the criteria to be included in the main evidence. This study did not show benefits of oral oxcarbazepine 1200 mg daily as compared to placebo (Figure 4.2.25). The quality of evidence for the outcome persisting monthly migraine days was considered very low (Table 4.2.7).

Forest plot showing the comparison between oral oral oxcarbazepine 1200 mg and placebo for the outcome change in monthly migraine days in patients with migraine (no distinction between episodic and chronic).
GRADE evidence profile for oral oxcarbazepine versus placebo in patients with any migraine (no distinction between episodic and chronic)

Only one trial.
Accordingly, there was no difference in the outcome ≥50% response rate in subjects treated with oxcarbazepine 1200 mg daily as compared to placebo (Figure 4.2.26). The quality of evidence for the outcome ≥50% response rate was considered very low (Table 4.2.7).

Forest plot showing the comparison between oral oxcarbazepine 1200 mg and placebo for the outcome ≥50% response rate in patients with migraine (no distinction between episodic and chronic).
Additional evidence
None.
Evidence-based recommendation for PICO 4.2.10 In subjects with migraine, we suggest against oral oxcarbazepine for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↓)
Evidence-based guideline summary
Based on clinical trial results and the quality of evidence, the use of topiramate for episodic and chronic migraine prevention is recommended. There is limited evidence to support the use of valproate for episodic and chronic migraine and the use of lamotrigine and levetiracetam for episodic migraine. There is inadequate evidence to support the use of other anti-seizure medications (Figure 4.2.27).

Summary of evidence-based recommendations on anti-seizure medication for migraine prevention. *The weak recommendation in favor of oral valproate 1000 mg for patients with any migraine derives from the fact that the 500 mg and 1500 mg doses are superior to placebo and the results from the 1000 mg dose are imprecise. The quality of evidence is very low for all dosages.
Expert-based opinions
Optimization of topiramate use
Topic: Topiramate titration and dosing
Topic: Selection of candidates for migraine prevention with topiramate
However, topiramate should not be used during pregnancy or lactation, and caution should be exercised when prescribing it to subjects with a history of nephrolithiasis, narrow-angle glaucoma, or moderate/severe renal or hepatic impairment. For women of childbearing potential, it is essential to inform them about the potential teratogenic effects of topiramate, and the use of a highly effective method of contraception should be strongly recommended.
Topic: Selection of candidates for migraine prevention with topiramate
Optimization of valproate use
Topic: Valproate titration and dosing
Topic: Selection of candidates for migraine prevention with valproate
Topic: ASMs treatment duration
Topic: Lamotrigine in migraine prevention
Topic: Levetiracetam in migraine prevention
Topic: Switching among antiseizure medications
Introduction
Beta-blockers are available worldwide and used for many different conditions in medicine ranging from the discovery of beneficial effects for angina pectoris in the mid-1950s to the more recent use for migraine prevention (685). Nowadays, propranolol is listed among the most commonly used preventive drugs for migraine (686–688). In 2012, the American Academy of Neurology guideline recommended beta-blockers, specifically propranolol and metoprolol, as first line therapy for preventing migraine (689). Since then, several systematic reviews and meta-analysis supported the use of beta-blockers for migraine prevention (688), but, although they were employed in clinical practice, widely used in Europe, and recommended from international guidelines, the evidence for the efficacy of beta-blockers in migraine prevention still comes from old studies performed in the late 1970–80s (690–696). Of note, the mechanism of action of beta-blockers in migraine prevention is still not understood. It has been hypothesized that beta-blockers might penetrate the blood-brain barrier and interact with the catecholaminergic system and serotonin receptor in the brain, but the exact mechanism of action in migraine is still not understood (697,698). Beta-blockers are well-tolerated in general and considered safe in the absence of contraindications (asthma, congestive heart failure, cardiac dysrhythmia, depression), but adverse effects are reported in a minority of cases (691,699).
Section-specific methods
This section followed the general procedure to develop this guideline. However, in addition to those procedures, we also considered the outcomes of change in monthly migraine attacks and of persisting monthly migraine attacks. This choice was made to include even the oldest RCTs of beta-blockers. As the additional outcomes were not initially considered for the guideline, the resulting quality of evidence was considered by definition as very low.
Search strings for the Guideline on beta-blockers for systematic review/meta-analyses and for additional RCTs are reported in Online Appendix 4.3.1.
Results
Overall, we retrieved 2841 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included four systematic reviews and meta-analyses (686–688,700) that were considered as sources of randomized controlled trials (RCTs). After analyzing full texts of RCTs included in these reviews, we included 21 of them in qualitative or quantitative synthesis (628,649,691,696,697,699,701–715) (Figure 4.3.1).

Meta-analysis selection flow chart - beta blockers.
Overall, we retrieved 2386 references from searching for RCTs and after removing duplicates we had 2211 references to analyze. However, considering the most recent included meta-analysis on the topic (688), the analysis of additional RCTs was performed for papers published since 21 August 2018 (623 references). No additional RCT was included in meta-analyses (Figure 4.3.2). The literature update performed in May 2023 and November 2023 led to the inclusion of one further RCT (656) (Figure 4.3.2).

RCTs selection flow chart - beta blockers. *trials published since August 2018 were screened.
Overall, 22 RCTs were included in the literature synthesis to develop the evidence-based guideline. Among these studies, 12 reported a comparison between an active drug and placebo that could be included in quantitative analyses and generate recommendations, while the remaining 10 only reported head-to-head comparisons and were included in Section 4.8.
Results
Bisoprolol
Main evidence
None.
Additional evidence
We found one RCT comparing oral bisoprolol 5 mg and 10 mg for 12 weeks to placebo (710) in subjects with migraine (no distinction between episodic and chronic) that did not meet the criteria to be included in the main evidence. The study had a very serious risk of bias, and the quality of evidence was further downgraded to very low (Figures 4.3.3 and 4.3.4).

Forest plot showing the comparison between oral bisoprolol 5 or 10 mg and placebo for the outcome persisting monthly migraine days in patients with migraine (no distinction between episodic and chronic).
Forest plot showing the comparison between oral bisoprolol 5 or 10 mg and placebo for the outcome change in monthly migraine days in patients with migraine (no distinction between episodic and chronic).
The study showed benefits of oral bisoprolol 5 mg and 10 mg for 12 weeks over placebo considering the outcomes persisting monthly migraine days (Figure 4.3.3) and change in monthly migraine days (Figure 4.3.4).
Evidence-based recommendation for PICO 4.3.1 In subjects with any migraine (no distinction between episodic and chronic), we suggest oral bisoprolol 5 or 10 mg daily for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↑)
Metoprolol
Main evidence
None.
Additional evidence
We found two RCTs comparing metoprolol for 8 or 12 weeks to placebo (703,708) in subjects with any migraine (no distinction between episodic and chronic) that did not meet the criteria to be included in the main evidence. These studies showed high risk of bias and the evidence was downgraded to very low.
One study showed no differences between oral metoprolol 50 mg twice a day for eight weeks and placebo (708), while the other study showed benefits of oral metoprolol 200 mg for 12 weeks over placebo considering the outcomes persisting monthly migraine days (703) (Figure 4.3.5). Regarding the outcome change in monthly migraine days, one of the studies showed benefits of oral metoprolol 200 mg for 12 weeks over placebo (703) (Figure 4.3.6).
Evidence-based recommendation for PICO 4.3.2
1) In subjects with migraine (no distinction between episodic and chronic), we suggest against oral metoprolol 50 mg twice a day for migraine prevention.
Strength of recommendation: Weak (↓)
2) In subjects with any migraine (no distinction between episodic and chronic), we suggest oral metoprolol 200 mg daily for migraine prevention.
Strength of recommendation: Weak (↑)
Quality of evidence: Very low (⊕⊖⊖⊖)
Quality of evidence: Very low (⊕⊖⊖⊖)
Forest plot showing the comparison between oral metoprolol (50 mg BID or 200 mg) and placebo for the outcome persisting monthly migraine days in patients with migraine (no distinction between episodic and chronic).
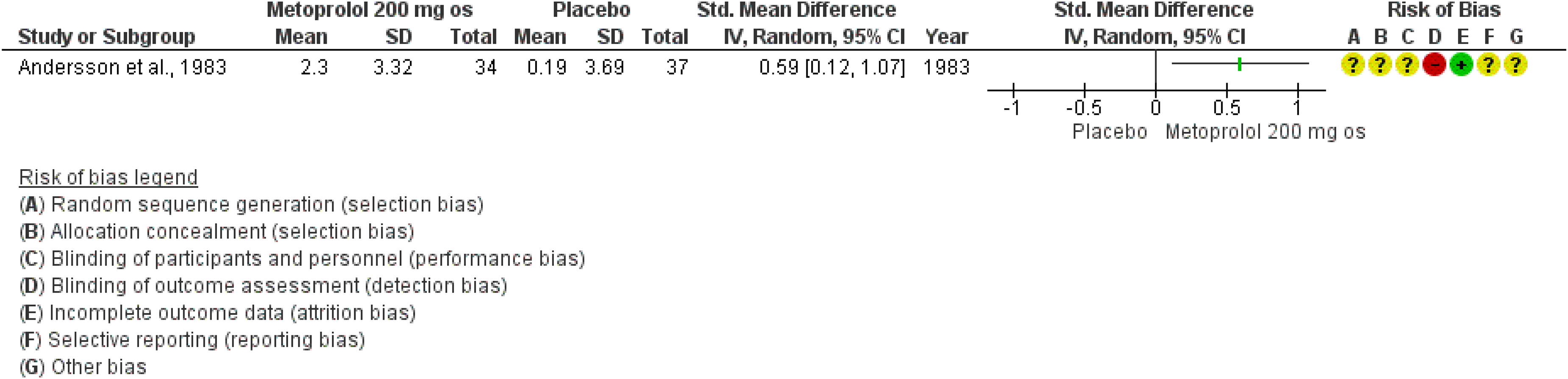
Forest plot showing the comparison between oral metoprolol 200 mg and placebo for the outcome change in monthly migraine days in patients with any migraine (no distinction between episodic and chronic).
Main evidence
We found one RCT comparing propranolol to placebo in subjects with episodic migraine (628) that met the criteria for main evidence. The risk of bias was considered serious (Figures 4.3.7 and 4.3.8). The RCT showed benefit of oral propranolol 160 mg daily over placebo considering the outcomes of change in monthly migraine days (Figure 4.3.7) and ≥50% responder rate (Figure 4.3.8). The quality of evidence was considered low (Table 4.3.1).

Forest plot showing the comparison between oral propranolol 160 mg daily and placebo for the outcome change in monthly migraine days in patients with episodic migraine.

Forest plot showing the comparison between oral propranolol 160 mg daily and placebo for the outcome ≥50% responder rate in patients with episodic migraine.
GRADE evidence profile for oral propranolol 160 mg versus placebo in patients with episodic migraine
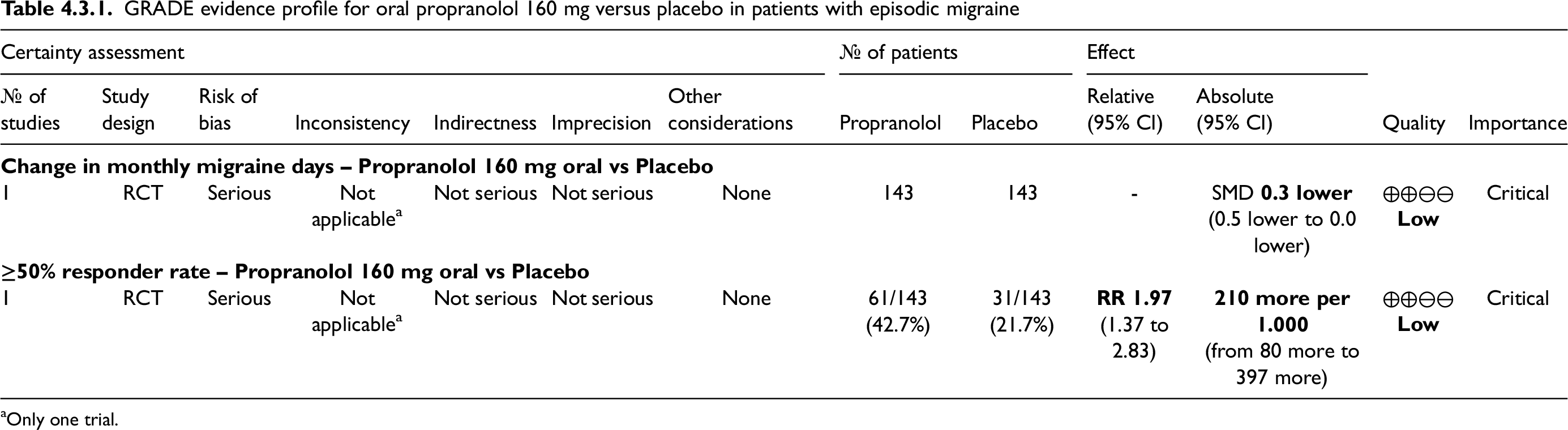
Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.3.3 In subjects with episodic migraine, we suggest oral propranolol 160 mg daily over placebo for migraine prevention.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of recommendation: Weak (↑)
Main evidence
We found one RCT comparing propranolol to placebo (709) that met the criteria for main evidence. The risk of bias was considered not serious (Figures 4.3.9 and 4.3.10). The RCT showed no benefits of oral propranolol 160 mg daily over placebo considering the outcomes of persisting monthly migraine days (Figure 4.3.9) and ≥50% responder rate (Figure 4.3.10). The quality of evidence was considered moderate (Table 4.3.2) due to the availability of only one RCT.
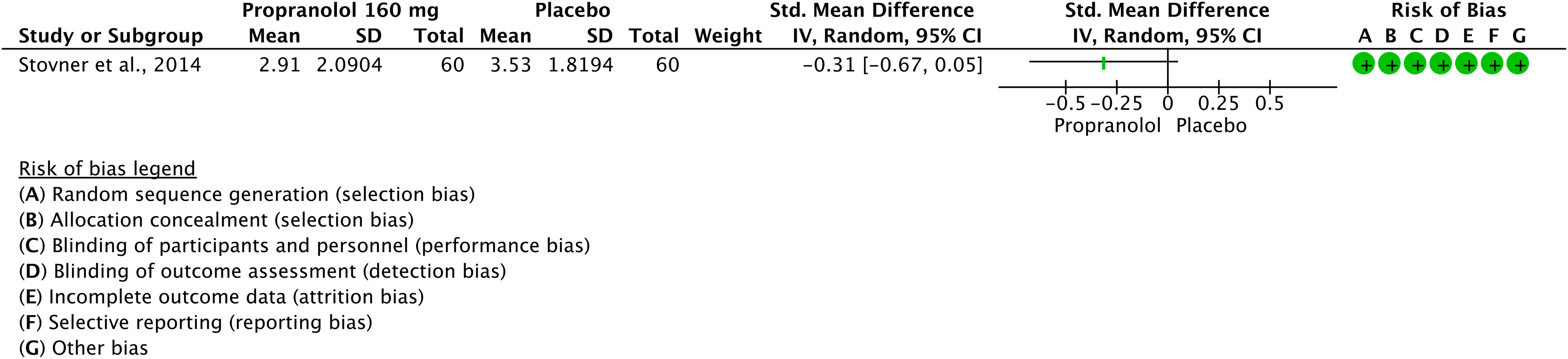
Forest plot showing the comparison between oral propranolol 160 mg daily and placebo for the outcome persisting monthly migraine days in patients with any migraine (no distinction between episodic and chronic).
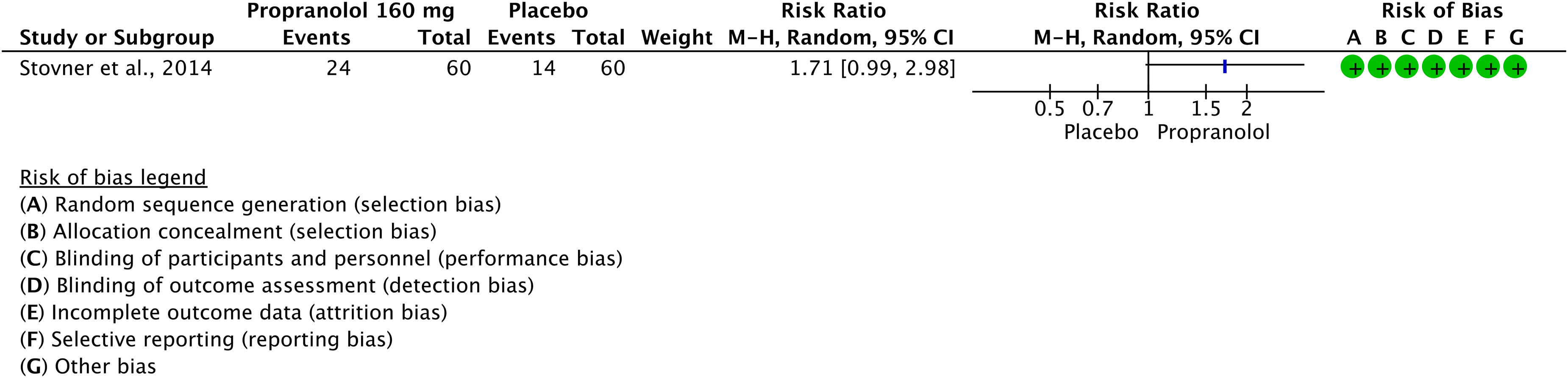
Forest plot showing the comparison between oral propranolol 160 mg daily and placebo for the outcome ≥50% responder rate in patients with any migraine (no distinction between episodic and chronic).
GRADE evidence profile for oral propranolol 160 mg versus placebo in patients with any migraine (no distinction between episodic and chronic)

Only one trial.
Additional evidence
We found eight RCTs comparing propranolol to placebo (691,696,697,707,712–715) that did not meet the criteria for main evidence. The overall risk of bias was considered serious (Figures 4.3.11 and 4.3.12). The RCTs showed benefits of oral propranolol 80 mg and 160 mg daily over placebo considering the outcome of ≥50% responder rate (Figure 4.3.11) and of oral propranolol 160 mg daily over placebo considering the outcome of persisting monthly migraine attacks (Figure 4.3.12). The quality of evidence was considered very low due to the assessment of an additional outcome – persisting monthly migraine attacks – that was not considered for the rest of the guideline.
Evidence-based recommendation for PICO 4.3.4 1) In subjects with migraine (no distinction between episodic and chronic), we suggest oral propranolol 160 mg daily over placebo for migraine prevention.
Quality of evidence: Very Low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↑)
2) In subjects with migraine (no distinction between episodic and chronic), we suggest against propranolol 80–120 mg daily over placebo for migraine prevention.
Quality of evidence: Very Low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↓)
[NOTE: The recommendation in favor of propranolol 160 mg daily was based upon the large number of positive RCTs available, even if the only RCT that could be included in the main evidence suggests against the benefit of propranolol. Evidence in favor of propranolol 160 mg daily refers to the slow-release formulation].

Forest plot showing the comparison between oral propranolol and placebo for the outcome ≥50% responder rate in patients with any migraine (no distinction between episodic and chronic).

Forest plot showing the comparison between oral propranolol and placebo for the outcome persisting monthly migraine attacks in patients with any migraine (no distinction between episodic and chronic).
Safety and tolerability of beta-blockers
Evidence-based guideline summary
There is low-quality evidence to suggest oral bisoprolol 5–10 mg, oral metoprolol 200 mg, and oral propranolol 160 mg for migraine prevention (Figure 4.3.7).

Summary of evidence-based recommendations on beta-blockers for migraine prevention.
Expert based opinions
Topic: Beta-blockers other than bisoprolol, metoprolol, and propranolol
Optimization of beta-blockers
Topic: Bisoprolol titration and dosing
Topic: Metoprolol titration and dosing
Topic: Propranolol titration and dosing
Introduction
Calcium channel blockers and drugs acting on the renin-angiotensin system (RAS), such as angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs), have been used for migraine prevention.
Flunarizine is one of many preventive treatment options for migraine (720–722). Flunarizine is a nonselective calcium antagonist, which has also been shown to block voltage-gated sodium channels. Action at these two sites may reduce neuronal excitability and normalize cortical hyperexcitability in migraine. Furthermore, flunarizine acts as a D2 dopamine antagonist and has in this regard been shown to possess (at high doses) comparable antipsychotic efficacy as haloperidol (723). It has also been shown that flunarizine can reduce the number and duration of cortical spreading depolarization (CSD) waves (724) and can alleviate CSD-induced mitochondrial injury (725). Finally, flunarizine increases leptin levels, and this activity has been suggested to reflect leptin resistance, which may potentially explain the treatment-related weight gain (565). Flunarizine is largely regarded as effective, inexpensive, and easy to use with its once-daily dosing (721).
ACE inhibitors and angiotensin receptor blockers (ARBs) modulate RAS, which contributes to control of maintenance of extracellular volume homeostasis and blood pressure (726). Renin is a plasmatic enzyme produced by kidneys. It converts angiotensinogen to angiotensin I, which is subsequently converted to angiotensin II by ACE. Angiotensin II can bind to specific receptors (AT), the most important being AT1 and AT2 (727). AT1 receptors are implicated in mediating vasoconstriction, sympathetic nervous system activation and cardiovascular remodeling, while AT2 activation results in vasodilation, antiproliferative effects and apoptosis (728,729). RAS involvement in migraine is not fully understood. Centrally, RAS may influence endorphin metabolism altering nociception, cerebrovascular blood flow, neurohormone levels, neurotransmitter levels, mast cells degranulation, proinflammatory cytokines and NO production, oxidative stress, vasodilation and neurogenic inflammation (730,731). Moreover, RAS modulates cerebrovascular blood flow, and inhibition and blockade of RAS may provide protection against cortical oligemia that occurs during a migraine attack. There may also be a genetic association between RAS and migraine. In fact, the ACE-D allele is significantly more frequent in subjects with migraine compared to controls (732,733), and the ACE-DD genotype positively correlated with a higher frequency of migraine attacks (734). ACE inhibitors (captopril, enalapril, lisinopril) are suggested to be effective for the prevention of migraine because they block the ACE enzyme preventing the conversion of angiotensin I to angiotensin II. ARBs represent another option for pharmacological prevention of migraine (735), because they compete directly with angiotensin II at its receptor (736). Candesartan and telmisartan are selective and competitive ARB at the AT1. In this way, they inhibit the sympathetic tone by reducing angiotensin II-induced vasopressin and adrenal catecholamine release and decreasing expression of nitric oxide-synthase (737). Furthermore, ARBs can partially agonize peroxisome proliferator-activated receptor gamma which exerts an anti-inflammatory action in the brain, by reducing levels of tumor necrosis factor and inducible nitric oxide (730).
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for the guideline on antihypertensives for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 4.4.1.
Results
Overall, we retrieved 1498 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included eight systematic reviews and meta-analyses that were considered as sources of randomized controlled trials (RCTs). After analyzing full texts of these RCTs, we included four RCTs in the quantitative synthesis (709,736,738,739) (Figure 4.4.1).
Meta-analysis selection flow chart - calcium channel blockers and blood pressure-lowering medications.
Overall, we retrieved 1525 references from searching for RCTs and after removing duplicates we had 1438 references to analyze. However, considering the most recent included systematic review and meta-analysis on the topic, the analysis of additional RCTs was performed for papers published since January 2020 (217 references). We finally included no additional RCTs (Figure 4.4.2). No additional RCTs were included after the literature update performed in May and in November 2023 (Figure 4.4.2).

RCT selection flow chart - calcium channel blockers and blood pressure-lowering medications.
Overall, four studies were included in the literature review and quantitative synthesis (meta-analyses) to develop the evidence-based guideline (709,736,738,739). All of them reported a comparison between an active drug and placebo and were presented in this section. One RCT also included a head-to-head comparison (709) which was reported in Section 4.8.
Candesartan
Main evidence
We found one RCT comparing oral candesartan to placebo (709) in subjects with migraine that met the criteria to be included in main evidence. The risk of bias was considered not serious (Figures 4.4.3 and 4.4.4). The study did not show a significant benefit of oral candesartan 16 mg compared to placebo considering the outcome persisting monthly migraine days while it showed significant benefit for the outcome ≥50% responder rate (Figures 4.4.3 and 4.4.4). The quality of evidence for both outcomes was considered low (Table 4.4.1).
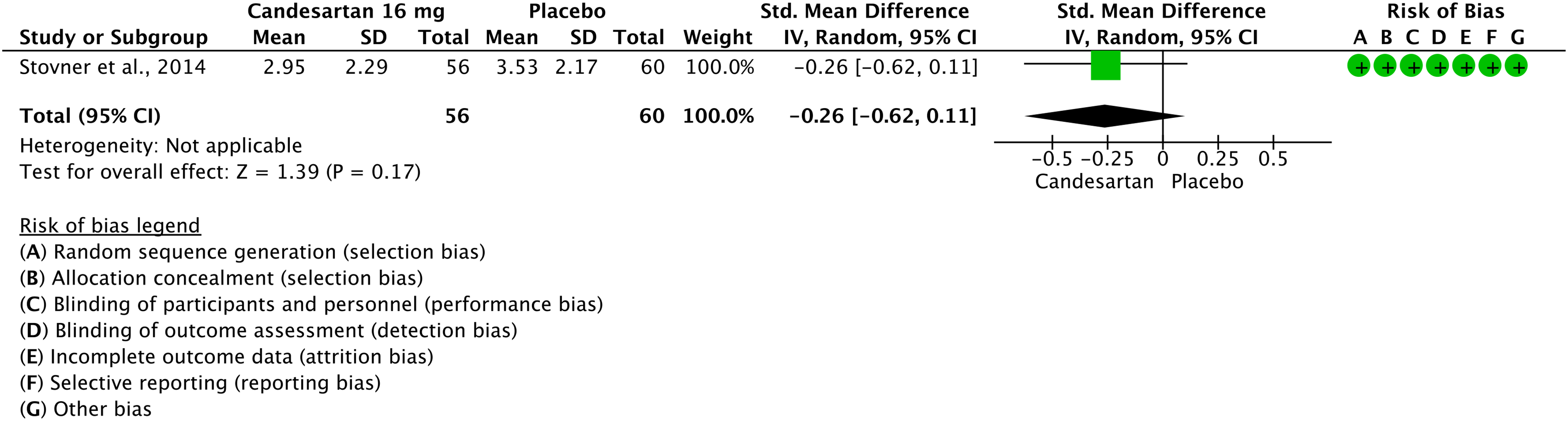
Forest plot showing the comparison between oral candesartan 16 mg and placebo for the outcome change in monthly migraine days in patients with migraine (no distinction between episodic and chronic).

Forest plot showing the comparison between oral candesartan 16 mg and placebo for the outcome ≥50% response rate in patients with migraine (no distinction between episodic and chronic).
GRADE evidence profile table for oral candesartan 16 mg versus placebo in patients with migraine (no distinction between episodic and chronic)

Only one trial; bInconsistency of findings in the same study.
Additional evidence
None.
Evidence-based recommendation for PICO 4.4.1 In subjects with migraine (no distinction between episodic and chronic), we suggest oral candesartan 16 mg for migraine prevention. Quality of evidence: Low (⊕⊕⊖⊖).
Strength of the recommendation: Weak (↑)
Main evidence
We found one RCT comparing oral candesartan to placebo (738) in subjects with episodic migraine that met the criteria to be included in main evidence. The study showed a significant benefit of oral candesartan 16 mg compared to placebo considering the outcome persisting monthly migraine days (Figure 4.4.5). The quality of evidence for the selected outcomes was considered moderate (Table 4.4.2).
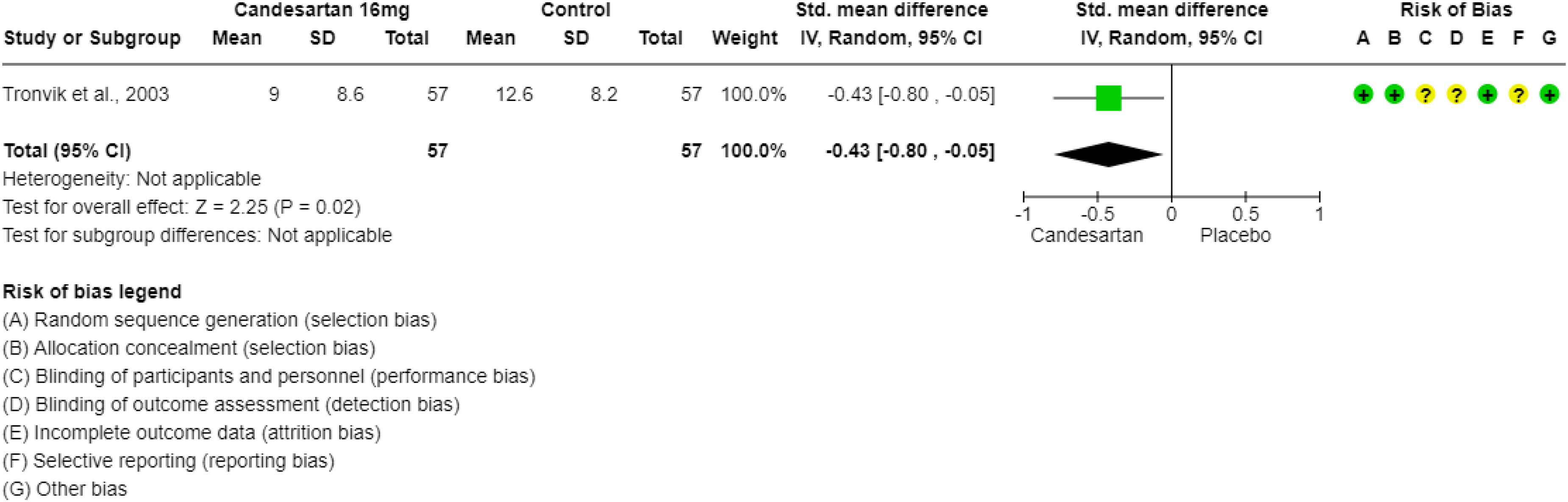
Forest plot showing the comparison between oral candesartan 16 mg os and placebo for the outcome persisting monthly migraine days in patients with episodic migraine.
GRADE evidence profile table for candesartan 16 mg os versus placebo in episodic migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.4.2 In subjects with episodic migraine, we suggest oral candesartan 16 mg for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Weak (↑)
Lisinopril
Main evidence
We found one RCT comparing oral lisinopril to placebo (739) in subjects with migraine that met pre-defined criteria to be included in main evidence (Figure 4.4.6).

Forest plot showing the comparison between oral lisinopril 20 mg and placebo for the outcome persisting monthly migraine days in patients with episodic migraine.
Its analysis showed a significant benefit of oral lisinopril 20 mg compared to placebo for the outcome persisting monthly migraine days (Figure 4.4.6). No other outcome was reported. The quality of evidence was considered low (Table 4.4.3).
GRADE evidence profile table for oral lisinopril 20 mg versus placebo in patients with episodic migraine
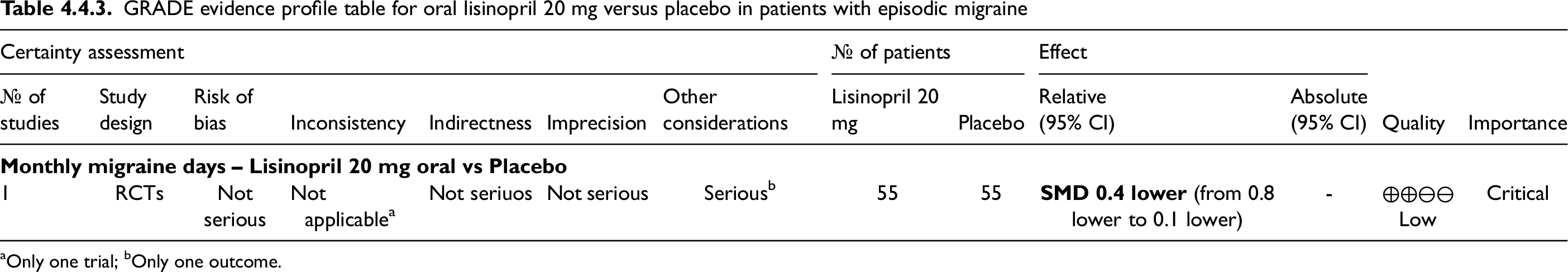
Only one trial; bOnly one outcome.
Additional evidence
None.
Evidence-based recommendation for PICO 4.4.3 In subjects with episodic migraine, we suggest oral lisinopril 20 mg for migraine prevention.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Weak (↑)
Telmisartan
Main evidence
We found one RCT comparing oral telmisartan to placebo (736) in subjects with migraine that met the criteria to be included in main evidence (Figures 4.4.7 and 4.4.8). Its analysis did not show a significant benefit of oral telmisartan 80 mg compared to placebo for the outcomes change in monthly migraine days and for the outcome ≥50% response rate (Figure 4.4.7 and 4.4.8). The quality of evidence for the selected outcomes varies from moderate to low and the overall quality of evidence across both outcomes was rated as low (Table 4.4.4).

Forest plot showing the comparison between oral telmisartan 80 mg and placebo for the outcome change in monthly migraine days in patients with episodic migraine.
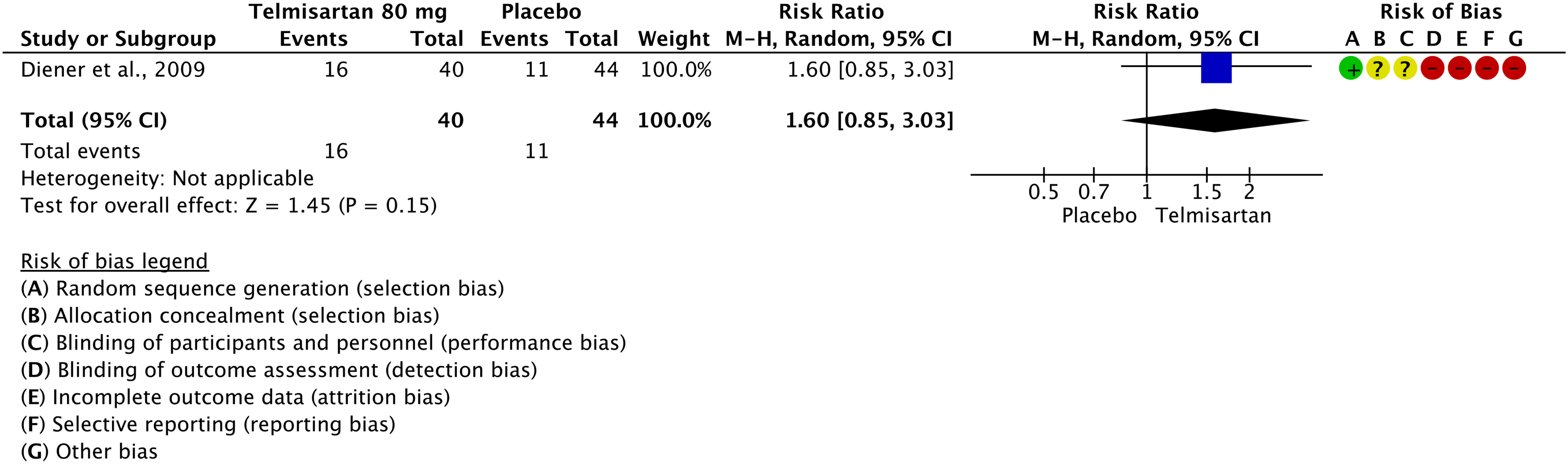
Forest plot showing the comparison between oral telmisartan 80 mg and placebo for the outcome ≥50% response rate in patients with episodic migraine.
GRADE evidence profile table for oral telmisartan 80 mg versus placebo in episodic migraine

Only one trial; bLow numbers of patients (<50 per group).
Additional evidence
None.
Evidence-based recommendation for PICO 4.4.4 In subjects with episodic migraine, we suggest against oral telmisartan 80 mg for migraine prevention.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Weak (↓)
Safety and tolerability
Calcium-channel blockers
ACE inhibitors
ARBs
Evidence-based guideline summary
We found only studies on ARBs and ACE inhibitors and not studies on calcium-channel blockers to be included for deriving evidence-based recommendations for this guideline. Evidence was available for candesartan, lisinopril and telmisartan.
The available evidence is limited to migraine (no distinction between episodic and chronic) and episodic migraine. No studies were included for chronic migraine.
There is evidence to recommend the use of candesartan for episodic migraine prevention while other agents’ studies do not allow to establish any recommendation (Figure 4.4.9).

Summary of evidence-based recommendations on antihypertensives for migraine prevention.
Expert based opinions
Calcium channel blockers
Topic: Calcium-channel blockers in migraine prevention
Main efficacy data for flunarizine versus placebo; data are taken from a systematic review (725)

RCTs assessing the efficacy and safety of flunarizine for migraine prevention could not be included in our guidelines as either main or additional evidence as they did not measure the outcomes that were required to be included in the present guideline. Nevertheless, given the large amount of available data, positive results against placebo, and clinical experience, we would like to provide guidance on the use of flunarizine for migraine prevention.
According to evidence from RCTs and clinical experience, there is no need to titrate flunarizine. The 10 mg oral dose, taken once daily, is the one with the best evidence for efficacy (721). However, a head-to-head RCT did not show any difference in efficacy between the 5 mg and the 10 mg dose of flunarizine (704). The 5 mg dose of flunarizine was associated with a lower proportion of adverse events compared with the 10 mg dose, although not significantly (704). Therefore, it might be reasonable to employ a 5 mg dose of flunarizine in subjects with high expected probability of adverse events, such as those with low weight or depressive symptoms, or in those reporting adverse effects with the 10 mg dose. It might also be reasonable to take the drug in the evening due to the possible drug-related sedation.
Optimization of candesartan use
Topic: Candesartan titration and dosing
Topic: Selection of candidates for migraine prevention with candesartan
Introduction
The clinical observation that in some people onabotulinumtoxinA injected for cosmetic purposes appeared to alleviate headache led to the hypothesis that the drug may be a useful preventive treatment for migraine (756). The mechanism of action of onabotulinumtoxinA in migraine subjects is still not fully understood (757). However, sensory effects of onabotulinumtoxinA in migraine are supported by many findings. Indeed, onabotulinumtoxinA is thought to achieve its therapeutic effect through blocking activation of unmyelinated meningeal nociceptors by cortical spreading depression (758). Moreover, onabotulinumtoxinA inhibits the release of CGRP, substance P and other neuropeptides that are deeply involved in migraine pathophysiology (759). More recently, onabotulinumtoxinA was also found to alter inflammatory gene expression and immune cells in chronic headache subjects (760). Since 2010, onabotulinumtoxinA has been approved in many countries for the prevention of chronic migraine based on two large RCTs (761,762). The recommended reconstituted dose is 155 to 195 units, administered intramuscularly as 0.1 mL (five units) injections to between 31 and 39 sites in the head and neck.
In 2018, a consensus statement from the European Headache Federation recommended onabotulinumtoxinA as an effective and well-tolerated treatment of CM (763). Indeed, in the same year, Cochrane analyses reported that onabotulinumtoxinA may reduce the number of migraine days per month by two days compared with placebo treatment. Conversely, it was uncertain whether this treatment is effective for people with episodic migraine (764). Since Cochrane analyses, RCTs and real-world evidence have expanded the evidence and knowledge for this treatment.
Section-specific methods
This section followed the general procedure to develop this guideline. In general, for the guideline, we excluded dose-finding dosages that were not marketed for each of the considered drugs. In the case of botulinum toxin, given the peculiarities and the possibility to customize dosages we included all doses adopted by the selected studies.
Search strings for the guideline on botulinum toxin for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 4.5.1.
Results
Overall, we retrieved 1108 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included 1087 references.
Finally, we included one systematic review and meta-analysis as source of randomized controlled trials (RCTs) (764). After analyzing full texts of these RCTs, we included 13 of them in the quantitative synthesis (663,761,762,765–774) (Figure 4.5.1).

Meta-analysis selection flow chart - botulinum toxin.
Overall, we retrieved 1336 references from searching for RCTs and after removing duplicates we had 1185 references to analyze. However, considering the most recent included systematic review/meta-analysis on the topic, the analysis of additional RCTs was performed for papers published since 31 August 2020 (275 references). From the literature search updates performed in May 2023 and November 2023, we retrieved 211 references and examined 205 of them after removing duplicates. We finally included no additional RCTs (Figure 4.5.2).

RCTs selection flow-chart. *trials published since 31 August 2020 were screened.
Overall, 13 studies were included in the quantitative synthesis (meta-analyses) to develop the evidence-based guideline. Among these, 11 reported a comparison between an active drug and placebo and are presented in this section, while the remaining two reported head-to-head comparisons and were included in Section 4.8. Four studies practiced the PREEMPT injection protocol; different protocols are reported in Table 4.5.1.
Protocol of onabotulinumtoxinA injections in the included studies. *PREEMPT: 31 fixed sites of injection (Corrugator 10 U, Procerus 5 U, Frontalis 20 U, Temporalis 40 U, Occipitalis 30 U, Cervical/paraspinal 20 U, Trapezius 30 U) + 8 sites of injection with follow the pain approach (40 U)

OnabotulinumtoxinA
Main evidence
None.
Additional evidence
We found three RCTs comparing onabotulinumtoxinA to placebo (761,769,773) in subjects with episodic migraine that did not meet the pre-defined criteria to be included in main evidence. Risk of bias of included studies was serious and the quality of evidence was considered very low (Figures 4.5.3 and 4.5.4).

Forest plot showing the comparison between intramuscular onabotulinumtoxinA and placebo for the outcome change in monthly migraine days in patients with episodic migraine. The study by Aurora et al., 2007 included the two populations of “placebo non responders” and “placebo responders”.
Forest plot showing the comparison between intramuscular onabotulinumtoxinA and placebo for the outcome ≥50% response rate in patients with episodic migraine.
One of the RCTs compared onabotulinumtoxinA 100 UI with placebo (769). This study did not show significant benefit from onabotulinumtoxinA over placebo in the change in monthly migraine days (Figure 4.5.3) or in ≥50% response rate (Figure 4.5.4).
One of the RCTs compared onabotulinumtoxinA 150 UI and 255 UI with placebo (773). Overall, this study did not show any benefit from onabotulinumtoxinA treatment in the change in monthly migraine days (Figure 4.5.3) but showed a significant increase in ≥50% response rate in patients treated with onabotulinumtoxinA (Figure 4.5.4).
One additional RCT did not show any benefit from onabotulinumtoxinA treatment (761) in the change of monthly migraine days associated with onabotulinumtoxinA use (Figure 4.5.3). This study did not address ≥50% response rate. In this study, all patients were initially treated with placebo and classified as “placebo responders” and “placebo non responders”. Afterwards, all patients were randomized to receive onabotulinumtoxinA (from 110 to 260 UI) or placebo and results were provided separately.
Evidence-based recommendation for PICO 4.5.1 In subjects with episodic migraine, we suggest against onabotulinumtoxinA (100 to 255 UI) for migraine prevention.
Level of evidence: Very low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↓)
Main evidence
We found three RCTs comparing onabotulinumtoxinA to placebo (761,762,772) in subjects with chronic migraine that met the criteria to be included in main evidence.
Two RCTs (761,762) considered the outcome change in monthly headache days. Their pooled analysis showed a significant mean reduction in monthly headache days in subjects who were treated with onabotulinumtoxinA (Figure 4.5.5). The quality of evidence for the outcome was considered high (Table 4.5.2).

Forest plot showing the comparison between intramuscular onabotulinumtoxinA and placebo for the outcome change in monthly headache days in patients with chronic migraine.
GRADE evidence profile table for intramuscular onabotulinumtoxinA versus placebo in patients with chronic migraine

Although only one study was available, it reported a patient-level pooled analysis of two RCTs.
Only one study (772), which provided a patient-level pooled analysis of two RCTs, considered the outcome ≥50% response rate. This study showed a significantly increased probability of ≥50% response rate in subjects treated with onabotulinumtoxinA as compared to those treated with placebo (Figure 4.5.6). The quality of evidence for the outcome ≥50% response rate was considered high (Table 4.5.2).

Forest plot showing the comparison between intramuscular onabotulinumtoxinA and placebo for the outcome ≥50% response rate in patients with chronic migraine.
Additional evidence
We found four additional RCTs comparing onabotulinumtoxinA to placebo (767,770,771,774) in subjects with chronic migraine that did not meet all the pre-defined criteria to be included in main evidence. Overall risk of bias was considered not serious, and the quality of evidence was considered low (Figures 4.5.7 and 4.5.8).

Forest plot showing the comparison between intramuscular onabotulinumtoxinA and placebo for the outcome change in monthly headache days in patients with chronic migraine.

Forest plot showing the comparison between intramuscular onabotulinumtoxinA and placebo for the outcome ≥50% response rate in patients with chronic migraine.
Two RCTs compared onabotulinumtoxinA 100 UI with placebo (771,774). Overall, those studies did not show a significant reduction in monthly migraine days in subjects treated with onabotulinumtoxinA as compared to those treated with placebo (Figure 4.5.7). One additional RCT (767) compared onabotulinumtoxinA 155–195 UI with placebo. This RCT showed a marginally non-significant reduction in monthly migraine days in subjects treated with onabotulinumtoxinA as compared to those treated with placebo (Figure 4.5.7). However, this study had a small sample size which could have been inadequate to detect significant differences.
One additional RCT (770) reported for onabotulinumtoxinA 100 UI the outcome ≥50% response rate and results did not show a significant increased probability of ≥50% response in subjects treated with onabotulinumtoxinA 100 UI as compared to those treated with placebo (Figure 4.5.8).
Evidence-based recommendation for PICO 4.5.2 In subjects with chronic migraine, we recommend intramuscular onabotulinumtoxinA (155–195 UI quarterly administered according to the PREEMPT injection paradigm) for migraine prevention.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of recommendation: Strong (↑↑)
Safety and tolerability of onabotulinumtoxinA
AbobotulinumtoxinA
Main evidence
None.
Additional evidence
We found one RCT comparing abobotulinumtoxinA with placebo (768) that did not meet all the pre-defined criteria to be included in the main evidence. The study reported an overall serious risk of bias, and the quality of evidence was downgraded to very low (Figure 4.5.9).
Evidence-based recommendation for PICO 4.5.3 In subjects with episodic migraine, we suggest against intramuscular abobotulinumtoxinA (120 UI or 240 UI) for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of recommendation: Weak (↓)
Forest plot showing the comparison between intramuscular abobotulinumtoxinA and placebo for the outcome change in monthly migraine days in patients with episodic migraine.
Overall, this study did not show any benefit from abobotulinumtoxinA 120 or 240 UI in the outcome persisting monthly migraine days (Figure 4.5.9). Data showed a non-significant trend toward benefit from abobotulinumtoxinA 120 UI and 240 UI when considering ≥50% response rate (Figure 4.5.10). The pooled analysis of the two different doses indicated a significant benefit from abobotulinumtoxinA considering this same outcome (768).

Forest plot showing the comparison between intramuscular abobotulinumtoxinA and placebo for the outcome ≥50% response rate in patients with episodic migraine.
Evidence-based guideline summary
We found studies on onabotulinumtoxinA and abobotulinumtoxinA to be included for deriving evidence-based recommendations for this guideline. No studies on other toxins met eligibility criteria. For onabotulinumtoxinA, evidence covered episodic migraine and chronic migraine. No studies were included for any migraine. For abobotulinumtoxinA, evidence covered episodic migraine. No studies were included for any migraine or chronic migraine.
There is high quality of evidence to recommend the use of onabotulinumtoxinA for chronic migraine prevention while there is low quality of evidence indicating lack of benefits from onabotulinumtoxinA and abobotulinumtoxinA for episodic migraine prevention. (Figure 4.5.11).

Summary of evidence-based recommendations on botulinum toxin for migraine prevention. *155-195 UI for patients with chronic migraine.
Expert-based opinion
Topic: OnabotulinumtoxinA titration
Topic: OnabotulinumtoxinA dosing
Topic: First evaluation of effectiveness of onabotulinumtoxinA
Topic: Administration scheme other than PREEMPT
Introduction
Gepants are oral preventives for the treatment of migraine that act as competitive antagonists of the CGRP receptor (787). Gepants are highly selective for the canonical CGRP receptor (CLR/RAMP1), with poor affinity for other members of the CGRP receptor-family (788). This is due to their binding site between the CLR and the RAMP1 components of the canonical CGRP receptor (789). Their great affinity for this receptor is confirmed by an inhibition constant (Ki) in the nanomolar range (790). Multiple preclinical studies demonstrate the action of gepants on the trigeminovascular system. Firstly, they reduce CGRP-mediated meningeal vasodilation (791). Furthermore, gepants suppress activation of trigeminal Aδ and, partially, of C-fibers (792). As gepants cross the blood brain barrier (BBB) to a miniscule degree (523), some authors speculate that they may provide central effects (793).
The first generation of gepants, telcagepant and olcegepant, were developed in the early 2000s as acute treatments for migraine, but their development was interrupted due to liver toxicity (526). However, after the anti-CGRP monoclonal antibodies demonstrated efficacy for prevention, gepant development was resumed. The second generation of gepants were chemically modified from the first and did not show hepatotoxicity. Among these, atogepant was specifically developed for migraine prevention, whilst rimegepant was first explored for acute and subsequently for preventive treatment (794). Indeed, the favorable safety profile of the gepants, including the intriguing possibility that gepants may not induce MOH, led to their use also as preventive treatment (794). Nowadays, atogepant is marketed at doses of 10 mg, 30 mg and 60 mg and rimegepant at the dose of 75 mg.
Section-specific methods
This section followed the general procedure to develop this guideline. However, there was only one literature update performed in November 2023, without that performed in May 2023 (see also Section 3.8).
Search strings for the guideline on gepants (preventive treatment) for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 4.6.1.
Results
Overall, we retrieved 139 references from searching for systematic reviews and meta-analyses. After duplicate removal and selection phases, we included two systematic reviews and meta-analyses (795,796) as sources of randomized controlled trials (RCTs) (Figure 4.6.1).

Meta-analysis selection flow chart - gepants for migraine prevention.
After analyzing full texts of these RCTs, we included two of them in the quantitative synthesis (797,798) (Figure 4.6.2).

Flow chart of article selection - gepants for migraine prevention. *search was updated by the Authors directly in November 2023.
Overall, we retrieved 299 references from searching for RCTs. After removing duplicates, 245 references remained for analysis; three RCTs were finally included (543,797,798). From the literature search update performed in November 2023, we retrieved 136 references and 121 were examined after removing duplicates. Three further studies were included from this search (799–801) (Figure 4.6.2).
Overall, five studies were included to develop the evidence-based guideline as they reported a comparison between an active drug and placebo and are presented in this Section. One RCT reported a head-to-head comparison and was included in Section 4.8.
Atogepant
Main evidence
We found three RCTs (797–799) comparing oral atogepant to placebo in subjects with episodic migraine that met the criteria to be included in main evidence.
The pooled analysis showed benefits of oral atogepant 10, 30 and 60 mg daily over placebo considering the outcomes of change in monthly migraine days (Figure 4.6.3) and ≥50% responder rate (Figure 4.6.4) (797,798). The quality of evidence for both outcomes was considered high (Table 4.6.1).

Forest plot showing the comparison between oral atogepant and placebo for the outcome change in monthly migraine days in episodic migraine.
Forest plot showing the comparison between oral atogepant and placebo for the outcome ≥50% response rate in patients with episodic migraine.
GRADE evidence profile table of oral atogepant versus placebo in episodic migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.6.1 In subjects with episodic migraine, we recommend oral atogepant 60 mg daily for migraine prevention. Quality of evidence: High (⊕⊕⊕⊕).
Strength of the recommendation: Strong (↑↑)
[NOTE: In Europe and in the USA atogepant is approved only in the 60 mg dose (802); therefore, the recommendation applies to commercial doses only].
Main evidence
We found one RCT comparing oral atogepant to placebo (800) in subjects with chronic migraine that met the criteria to be included in main evidence.
The pooled analysis showed benefits of oral atogepant 30 twice a day and 60 mg daily over placebo considering the outcomes of change in monthly migraine days (Figure 4.6.5) and of ≥50% responder rate (Figure 4.6.6) (800). The quality of evidence was considered high (Table 4.6.2).
Evidence-based recommendation for PICO 4.6.2 In subjects with chronic migraine, we recommend oral atogepant 60 mg daily for migraine prevention. Quality of evidence: High (⊕⊕⊕⊕).
Strength of the recommendation: Strong (↑↑)
[NOTE: In Europe and in the USA atogepant is approved only in the 60 mg dose (802); therefore, the recommendation applies to commercial doses only].

Forest plot showing the comparison between oral atogepant and placebo for the outcome change in monthly migraine days in patients with chronic migraine.
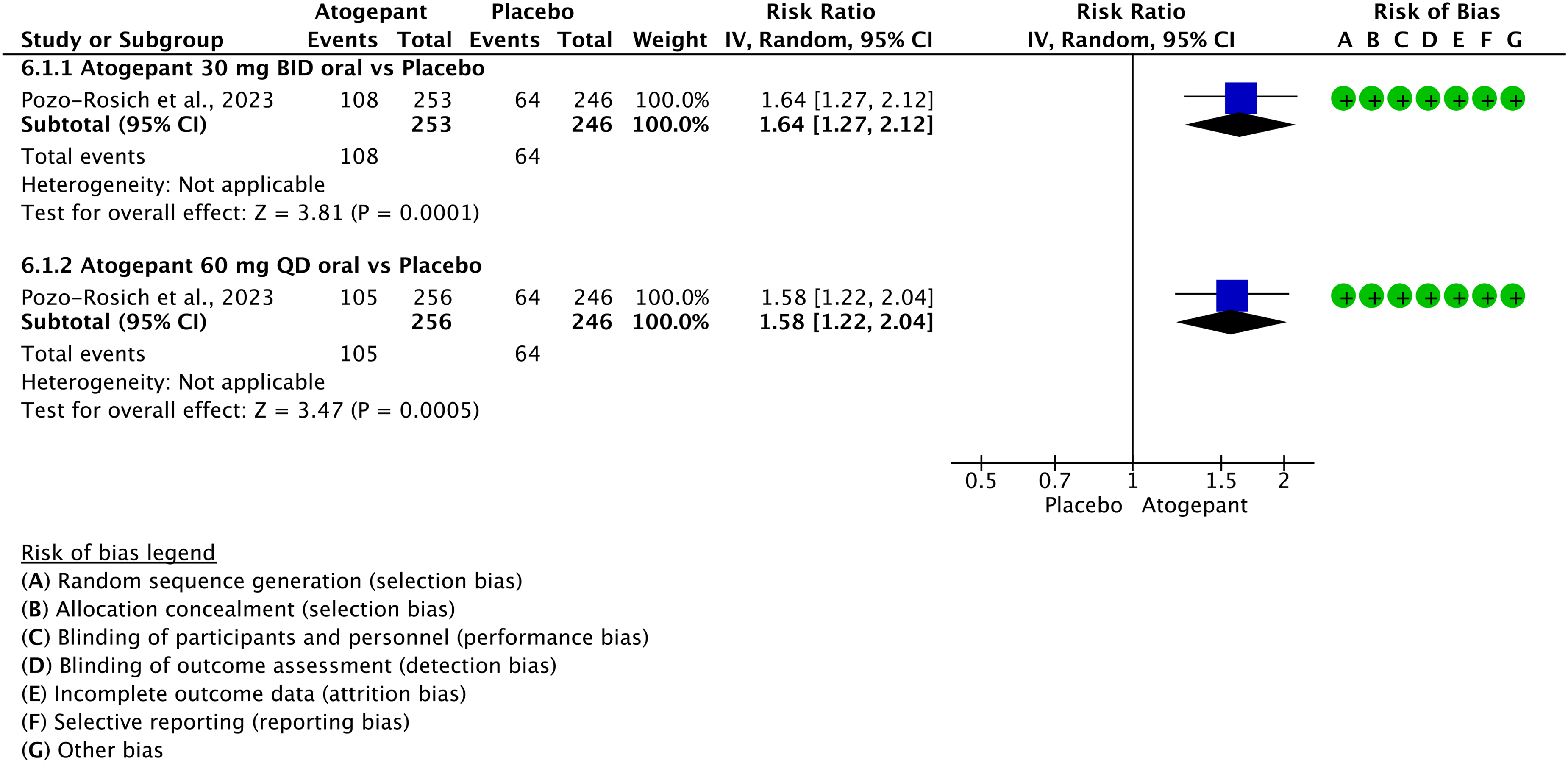
Forest plot showing the comparison between oral atogepant and placebo for the outcome ≥50% responder rate in patients with chronic migraine.
GRADE evidence profile table of oral atogepant versus placebo in chronic migraine

Only one trial; bUpgraded because of a very low risk of bias.
Safety and tolerability of atogepant
Rimegepant
Main evidence
We found one RCT comparing oral rimegepant to placebo (543) in subjects with migraine (between four and 18 monthly migraine days at baseline) that met the pre-defined criteria to be included in main evidence.
The RCT showed benefits of oral rimegepant 75 mg every other day over placebo considering the outcomes change in monthly migraine days (Figure 4.6.7) and ≥50% response rate (Figure 4.6.8) (543). The quality of evidence for both outcomes was considered moderate (Table 4.6.3) due to the availability of only one RCT.

Forest plot showing the comparison between oral rimegepant 75 mg and placebo for the outcome change in monthly migraine days in any migraine (no distinction between episodic and chronic).

Forest plot showing the comparison between oral rimegepant 75 mg and placebo for the outcome ≥50% response rate in patients with any migraine (no distinction between episodic and chronic).
GRADE evidence profile table for oral rimegepant versus placebo in migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.6.3 In subjects with migraine (between four and 18 monthly migraine days), we recommend oral rimegepant 75 mg every other day for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Strong (↑↑)
[NOTE: Current approvals of rimegepant only include subjects with episodic migraine (552), which represent most subjects treated in the available RCT.]
Safety and tolerability of rimegepant
Evidence-based guideline summary
We found five studies meeting eligibility criteria to be included as main evidence in this guideline on atogepant and rimegepant in migraine prevention. This study class is still under investigation in clinical trials and new results are expected that may change the present guideline. There is evidence to recommend the use of atogepant and rimegepant for episodic migraine prevention (Figure 4.6.9). No studies are currently available specifically for chronic migraine.

Summary of evidence-based recommendations on gepants for migraine prevention. *patients with 4- 18 monthly migraine days. **Only 30 mg twice a day or 60 mg daily.
Expert-based opinion
Optimization of atogepant use
Topic: Which patients should currently receive limited or no treatment with gepants?
Patients in whom gepants should be used with care

4.7 Monoclonal antibodies targeting the CGRP pathway
Introduction
Four monoclonal antibodies against the CGRP pathway (anti-CGRP mAbs) are currently available on several markets (805). One (erenumab) is directed against the CGRP receptor (R) (anti-CGRP/R mAb) and three (eptinezumab, galcanezumab and fremanezumab) are against the ligand (L) (anti-CGRP/L mAbs). RCTs and real-world studies demonstrated similar efficacy and tolerability of all these new drugs in chronic and episodic migraine, regardless of the presence of medication overuse and various previous treatment failures (804–806). Furthermore, anti-CGRP mAbs demonstrated a very favorable AE profile, with reported AEs slightly superior to the placebo group (807). The introduction of the anti-CGRP mAbs, which are disease-specific and mechanism-based treatments for the prevention of migraine, substantially improved patients’ management even if raising costs. Providing evidence-based clinical guidance on the use of the CGRP mAbs is of paramount importance to optimize the use of highly effective although currently costly treatments.
Section-specific methods
This section followed the general procedure to develop this guideline.
Search strings for the guideline on monoclonal antibodies targeting the CGRP pathway for systematic review/meta-analysis and for additional RCTs are reported in Online Appendix 4.7.1.
Results
Overall, we retrieved 1308 references from searching for systematic reviews and meta-analyses. After duplicate removal and screening stages, we included 11 systematic reviews and meta-analyses (618,808–817) that were considered as sources of randomized controlled trials (RCTs). From the 11 systematic reviews and meta-analyses 19 RCTs were included in the quantitative synthesis (818–836) (Figure 4.7.1).

Meta-analysis selection flow chart - monoclonal antibodies targeting the CGRP pathway.
More recent papers, published since October 2020 and not reported in the reviews and meta-analyses, were searched. We retrieved 2762 references from which, after removing duplicates, five RCTs were selected and analyzed (837–841). From the literature search update performed in May and November 2023, three further RCTs were included (842–844) (Figure 4.7.2).

RCTs selection flow chart - monoclonal antibodies targeting the CGRP pathway. *trials published since October 2020 were screened.
Overall, 26 studies were included in the quantitative synthesis to develop the evidence-based guideline (meta-analyses) and are presented in this Section. One RCT reported a head-to-head comparison and was presented in Section 4.8.
Eptinezumab
Main evidence
We found one RCT comparing eptinezumab to placebo in subjects with episodic migraine (818) that met the criteria to be included in the main evidence.
The RCT showed a significant improvement in subjects treated with intravenous (IV) eptinezumab 100 and 300 mg quarterly over placebo considering the outcomes of change in monthly migraine days (Figure 4.7.3) and ≥50% response rate (Figure 4.7.4). The quality of evidence for both outcomes was considered moderate (Table 4.7.1).

Forest plot showing the comparison between intravenous eptinezumab (100 or 300 mg quarterly) and placebo for the outcome change in monthly migraine days in patients with episodic migraine at 12 weeks.
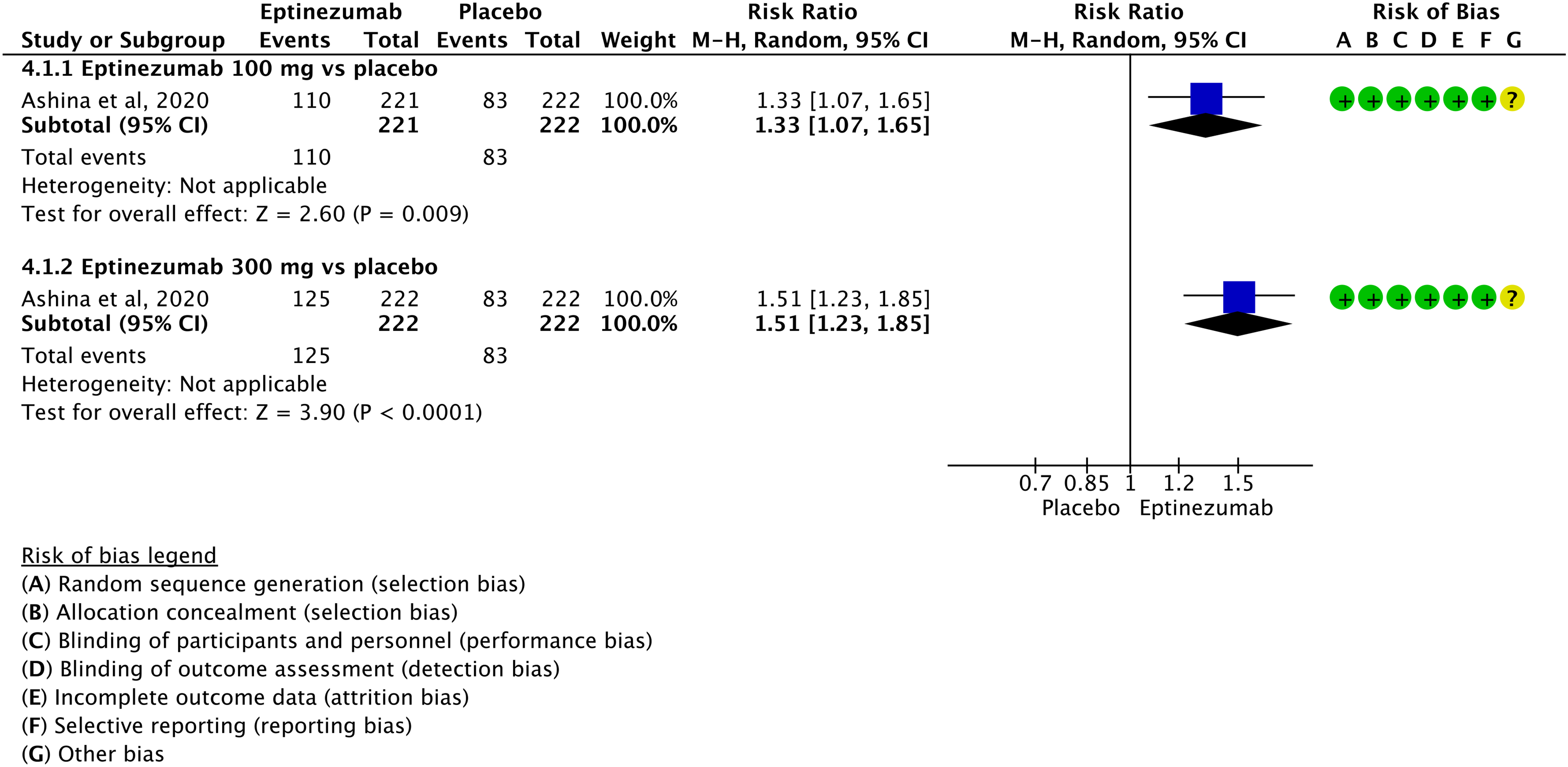
Forest plot showing the comparison between intravenous eptinezumab (100 and 300 mg quarterly) and placebo for the outcome ≥50% response rate in patients with episodic migraine at 12 weeks.
GRADE evidence profile table for intravenous eptinezumab versus placebo in episodic migraine

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.7.1 In subjects with episodic migraine, we recommend IV eptinezumab 100 or 300 mg quarterly for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Strong (↑↑)
Main evidence
We found two RCTs comparing eptinezumab to placebo in subjects with chronic migraine (823,827) that met the criteria to be included in the main evidence.
The pooled analysis showed a significant improvement in subjects treated with IV eptinezumab 100 and 300 mg quarterly over placebo considering the outcomes of change in monthly migraine days (Figure 4.7.5) and ≥50% responder rate (Figure 4.7.6). The quality of evidence for both outcomes was considered high (Table 4.7.2).

Forest plot showing the comparison between intravenous eptinezumab (100 or 300 mg quarterly) and placebo for the outcome change in monthly migraine days in patients with chronic migraine at 12 weeks.

Forest plot showing the comparison between intravenous eptinezumab (100 or 300 mg quarterly) and placebo for the outcome ≥50% response rate in patients with chronic migraine at 12 weeks.
GRADE evidence profile table for intravenous eptinezumab versus placebo in chronic migraine

Additional evidence
None.
Evidence-based recommendation for PICO 4.7.2 In subjects with chronic migraine, we recommend intravenous eptinezumab 100 or 300 mg quarterly for migraine prevention.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Main evidence
We found one RCT comparing eptinezumab to placebo in subjects with migraine (no distinction between episodic and chronic) (843) that met the criteria to be included in the main evidence. This study only included subjects with previous failure to at least 2–4 classes of migraine preventatives.
The RCT showed a significant improvement in patients treated with IV eptinezumab 100 and 300 mg quarterly over placebo considering the outcomes of change in monthly migraine days (Figure 4.7.7) and ≥50% responder rate (Figure 4.7.8). The quality of evidence for both outcomes was considered moderate (Table 4.7.3).

Forest plot showing the comparison between intravenous eptinezumab (100 or 300 mg quarterly) and placebo for the outcome change in monthly migraine days in patients with any migraine (no distinction between episodic and chronic) at 24 weeks.

Forest plot showing the comparison between intravenous eptinezumab (100 or 300 mg quarterly) and placebo for the outcome ≥50% response rate in patients with any migraine (no distinction between episodic and chronic) at 24 weeks.
GRADE evidence profile table for intravenous eptinezumab versus placebo in patients with migraine (no distinction between episodic and chronic)

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.7.3 In subjects with migraine (no distinction between episodic and chronic), we recommend intravenous eptinezumab 100 or 300 mg quarterly for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Strong (↑↑)
Erenumab
Main evidence
We found six RCTs comparing erenumab to placebo in subjects with episodic migraine (822,826,831,835,840,841) that met the criteria to be included in the main evidence.
The pooled analysis showed a significant improvement in subjects treated with subcutaneous (SC) erenumab 70 and 140 mg monthly over placebo considering the outcomes of change in monthly migraine days (Figure 4.7.9) and ≥50% response rate (Figure 4.7.10). The quality of evidence for both outcomes was considered high (Table 4.7.4). Analyses were pooled for 12-week and 24-week outcomes.

Forest plot showing the comparison between subcutaneous erenumab (70 or 140 mg every 4 weeks) and placebo for the outcome change in monthly migraine days in patients with episodic migraine at 12 or 24 weeks.

Forest plot showing the comparison between subcutaneous erenumab (70 or 140 mg every four weeks) and placebo for the outcome ≥50% response rate in patients with episodic migraine at 12 or 24 weeks.
GRADE evidence profile table for subcutaneous erenumab versus placebo in patients with episodic migraine

Additional evidence
None.
Evidence-based recommendation for PICO 4.7.4 In subjects with episodic migraine, we recommend subcutaneous erenumab 70 or 140 mg monthly for migraine prevention.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Main evidence
We found two RCTs comparing erenumab to placebo in subjects with chronic migraine (836,840) that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of SC erenumab 70 and 140 mg monthly over placebo considering the outcomes of change in monthly headache days (Figure 4.7.11) and ≥50% response rate (Figure 4.7.12). The quality of evidence for the outcome of monthly headache days was considered high for erenumab 70 mg and moderate for erenumab 140 mg; the quality of evidence for ≥50% response rate was considered moderate for both 70 and 140 mg doses (Table 4.7.5). Analyses were pooled for 12-week and 24-week outcomes.

Forest plot showing the comparison between subcutaneous erenumab (70 or 140 mg every 4 weeks) and placebo for the outcome change in monthly migraine days in patients with chronic migraine at 12 or 24 weeks.

Forest plot showing the comparison between subcutaneous erenumab (70 or 140 mg every 4 weeks) and placebo for the outcome ≥50% response rate in patients with chronic migraine at 12 weeks.
GRADE evidence profile table for subcutaneous erenumab versus placebo in patients with chronic migraine
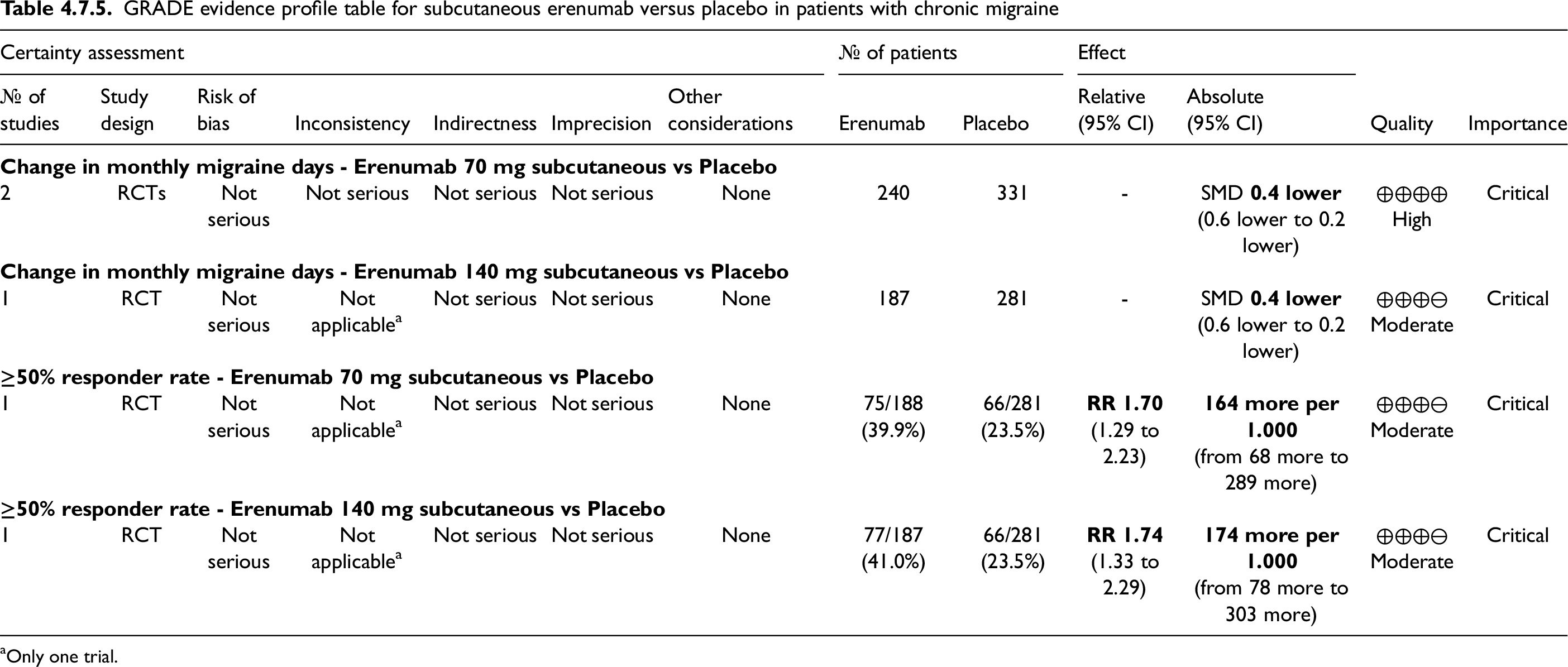
Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.7.5 In subjects with chronic migraine, we recommend subcutaneous erenumab 70 or 140 mg monthly for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Strong (↑↑)
Fremanezumab
Main evidence
We found four RCTs comparing fremanezumab to placebo in subjects with episodic migraine (819,824,825,838) that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of subcutaneous fremanezumab 225 mg monthly and 675 mg quarterly over placebo considering the outcomes of change in monthly migraine days (Figure 4.7.13) and ≥50% response rate (Figure 4.7.14). The quality of evidence for both outcomes was considered high (Table 4.7.6).

Forest plot showing the comparison between subcutaneous fremanezumab (225 mg monthly or 675 mg quarterly) and placebo for the outcome change in monthly migraine days in patients with episodic migraine at 12 weeks.

Forest plot showing the comparison between subcutaneous fremanezumab (225 mg monthly or 675 mg quarterly) and placebo for the outcome ≥50% response rate in patients with episodic migraine at 12 weeks.
GRADE evidence profile table for fremanezumab versus placebo in episodic migraine

Additional evidence
None.
Evidence-based recommendation for PICO 4.7.6 In subjects with episodic migraine, we recommend subcutaneous fremanezumab 225 mg monthly or 675 mg quarterly for migraine prevention.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Main evidence
We found four RCTs comparing fremanezumab to placebo in subjects with chronic migraine (820,825,832,839) that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of subcutaneous fremanezumab 225 mg monthly (with 675 mg loading dose) and 675 mg quarterly mg over placebo considering the outcomes change in monthly headache days (Figure 4.7.15) and ≥50% response rate (Figure 4.7.16). The quality of evidence for both outcomes was high for fremanezumab 675 mg quarterly. For both outcomes related to fremanezumab 225 mg monthly the quality of evidence was downgraded to moderate since in RCTs the intervention also included a loading dose of 675 mg that is not approved for clinical use (Table 4.7.7). However, it should be noted that the loading dose was deemed not necessary by regulatory agencies as not influencing the efficacy of the drug or its onset of effect.

Forest plot showing the comparison between subcutaneous fremanezumab (225 mg monthly plus 675 mg loading dose or 675 mg quarterly) and placebo for the outcome change in monthly migraine days in patients with chronic migraine at 12 weeks.

Forest plot showing the comparison between subcutaneous fremanezumab (225 mg monthly plus 675 mg loading dose or 675 mg quarterly) and placebo for the outcome ≥50% response rate in patients with chronic migraine at 12 weeks.
GRADE evidence profile table for subcutaneous fremanezumab versus placebo in patients with chronic migraine

RTC used a 675 mg loading dose not approved for clinical practice.
Additional evidence
None.
Evidence-based recommendations for PICO 4.7.7
1) In subjects with chronic migraine, we recommend subcutaneous fremanezumab 225 mg monthly for migraine prevention.
Strength of the recommendation: Strong (↑↑)
2) In subjects with chronic migraine, we recommend subcutaneous fremanezumab 675 mg quarterly for migraine prevention.
Strength of the recommendation: Strong (↑↑)
Quality of evidence: Moderate (⊕⊕⊕⊖)
Quality of evidence: High (⊕⊕⊕⊕)
Galcanezumab
Main evidence
We found five RCTs comparing galcanezumab to placebo in subjects with episodic migraine (828,830,833,834,844) that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of SC galcanezumab 120 mg monthly (with 240 mg loading dose at the first administration) over placebo considering the outcomes of change in monthly headache days (Figure 4.7.17) and ≥50% responder rate (Figure 4.7.18). The quality of evidence for both outcomes was considered high (Table 4.7.8). Analyses were pooled for three-month and six-month outcomes.

Forest plot showing the comparison between subcutaneous galcanezumab (120 mg monthly plus 240 mg loading dose) and placebo for the outcome change in monthly headache/migraine days in patients with episodic migraine at 3 or 6 months.

Forest plot showing the comparison between galcanezumab (120 mg monthly plus 240 mg loading dose) and placebo for the outcome ≥50% responder rate in patients with episodic migraine at 3 or 6 months.
GRADE evidence profile table for subcutaneous galcanezumab versus placebo in patients with episodic migraine
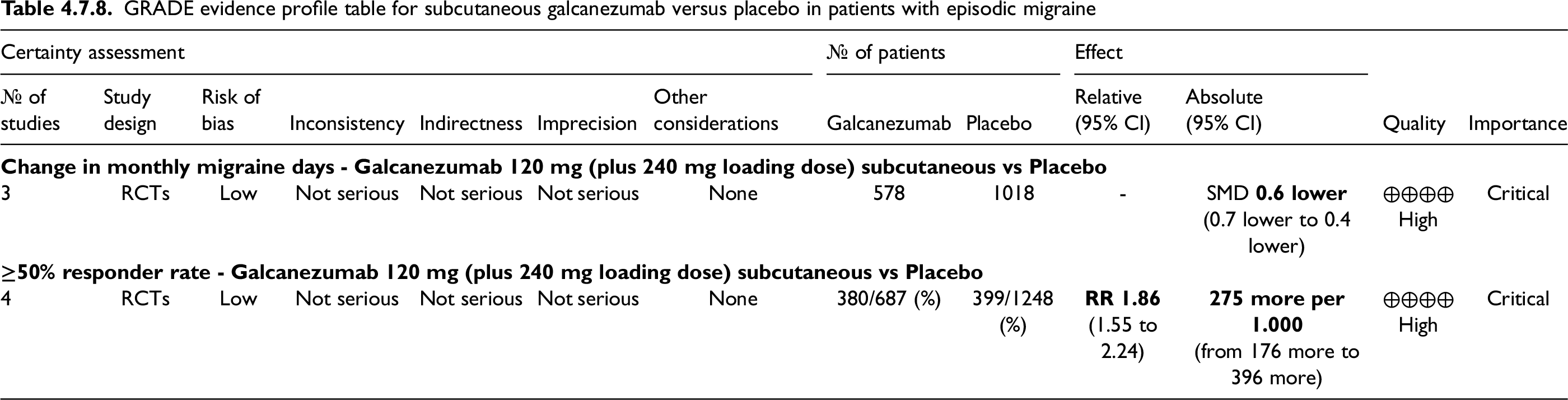
Additional evidence
None.
Evidence-based recommendation for PICO 4.7.8 In subjects with episodic migraine, we recommend subcutaneous galcanezumab 120 mg monthly (with 240 mg loading dose at the first administration) for migraine prevention.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Main evidence
We found two RCTs comparing galcanezumab to placebo in subjects with chronic migraine (821,828) that met the criteria to be included in the main evidence.
The pooled analysis showed benefits of subcutaneous galcanezumab 120 mg monthly (with 240 mg loading dose at the first administration) over placebo considering the outcomes change in monthly headache days (Figure 4.7.19) and ≥50% response rate (Figure 4.7.20). The quality of evidence for both outcomes was considered high (Table 4.7.9).
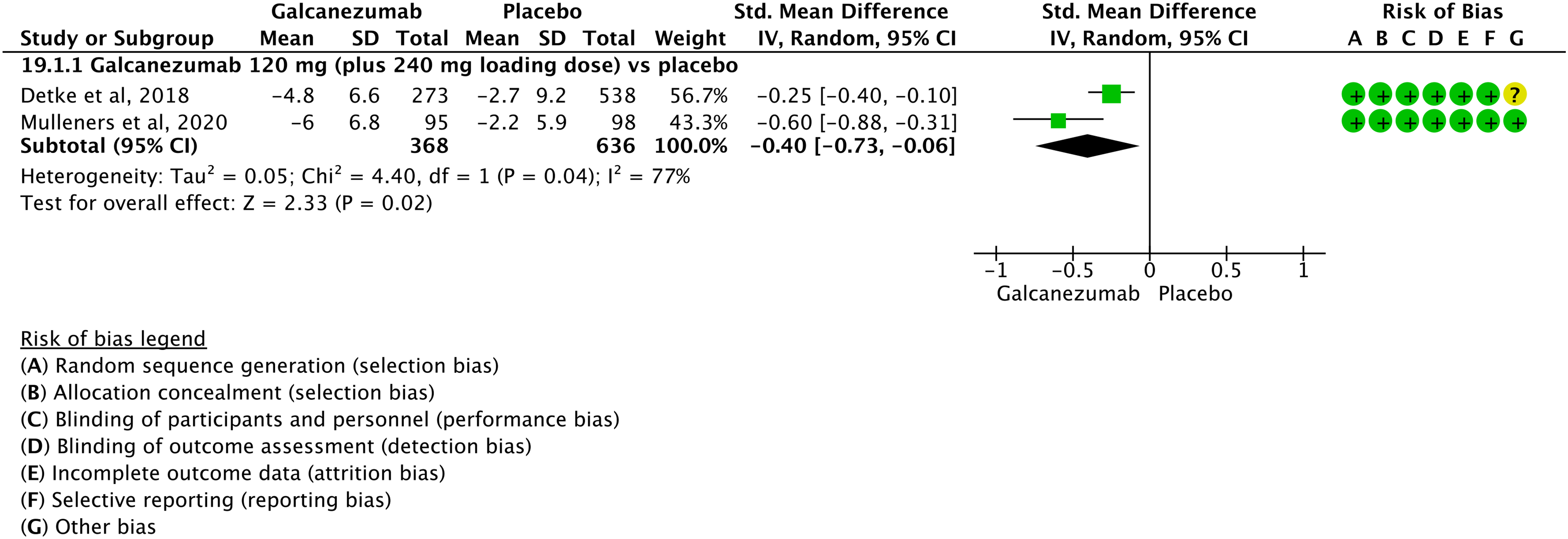
Forest plot showing the comparison between subcutaneous galcanezumab (120 mg monthly plus 240 mg loading dose) and placebo for the outcome change in monthly headache/migraine days in patients with chronic migraine at 3 months.

Forest plot showing the comparison between subcutaneous galcanezumab (120 mg monthly plus 240 mg loading dose) and placebo for the outcome ≥50% response rate in patients with chronic migraine at 3 months.
GRADE evidence profile table for subcutaneous galcanezumab versus placebo in patients with chronic migraine

Additional evidence
None.
Evidence-based recommendation for PICO 4.7.9 In subjects with chronic migraine, we recommend subcutaneous galcanezumab 120 mg monthly (with 240 mg loading dose at the first administration) for migraine prevention.
Quality of evidence: High (⊕⊕⊕⊕)
Strength of the recommendation: Strong (↑↑)
Safety and tolerability of anti-CGRP monoclonal antibodies
Evidence-based guideline summary
We found 25 studies that met eligibility criteria to be included in the main evidence on CGRP mAbs in migraine prevention. There is evidence to recommend the use of CGRP mAbs for episodic and chronic migraine prevention (Figure 4.7.21).

Summary of evidence-based recommendations on CGRP mAbs for migraine prevention. *240 mg loading dose.
Expert based opinion
Topic: Eptinezumab dosing and titration
Topic: Erenumab dosing and titration
Topic: Fremanezumab administration
Topic: Intravenous versus subcutaneous administration
Topic: Switch in real life studies
Topic: First evaluation of effectiveness
Topic: In which patients should monoclonal antibodies targeting the CGRP pathway be used with caution?
Conditions in whom monoclonal antibodies targeting the CGRP pathway should be used with caution

Introduction
A systematic examination of head-to-head comparisons of migraine preventive drugs might guide the decision for the choice of those drugs and for their combination. When choosing a single drug for migraine prevention, it would be reasonable to select a drug that is more effective than others. On the other hand, when choosing a combination of migraine preventive drugs, it is reasonable to combine medications that have a better tolerability profile. Head-to-head comparisons of preventive drugs are also important to simplify migraine treatment by establishing the best sequence of preventive drugs to try in each patient.
In the present section, we reported all head-to-head comparisons between different migraine preventive drugs for which RCTs are available. Both efficacy and safety information were discussed.
Section-specific methods
All RCTs included in this section were retrieved from the search strings of the other sections dedicated to RCTs of the respective migraine preventive drugs. We considered only PICOs including at least one drug as intervention or comparator that has an independent comparison against placebo.
At first, we collected all the head-to-head RCTs retrieved from the literature search. Duplicates were removed as an initial step. All the remaining RCTs were grouped by prioritizing injectables (CGRP-mAbs, onabotulinumtoxinA), followed by oral drugs. All RCTs were considered only once, even if eligible for more than one comparison. Comparisons were structured to be read in either direction, without the concept of a main intervention and its comparator. The terms “intervention” and “comparator” were replaced by “Drug 1” and “Drug 2” in the PICO structure (Table 4.8.1).
PICO structure of randomized controlled trials included in the present chapter

RCTs were included in the main evidence if they met all the following criteria:
A hypothesis of non-inferiority or superiority of one drug over the other was clearly stated The study sample size was justified.
All trials not meeting those criteria were excluded from the main evidence and considered for additional evidence.
Figure 4.8.1 reports the results of the literature search. Table 4.8.1 reports the PICO question structure of the present section.

Flow chart of study selection.
It is important to note that PICO questions referring to the present section were structured after the literature search, as there were no expected results prior to the search.
Main evidence
We found one RCT comparing erenumab to topiramate in subjects with any migraine that met the criteria to be included in the main evidence (836). The risk of bias was low (Figures 4.8.2 and 4.8.3).

Forest plot showing the comparison between subcutaneous erenumab (70 mg or 140 mg monthly) and oral topiramate (up to 100 mg daily) for the outcome change in monthly headache days in patients with any migraine (no distinction between episodic and chronic).

Forest plot showing the comparison between subcutaneous erenumab (70 mg or 140 mg monthly) and oral topiramate (up to 100 mg daily) for the outcome ≥50% response rate in patients with any migraine (no distinction between episodic and chronic).
The RCT showed benefits of SC erenumab 70 mg or 140 mg quarterly over oral topiramate up to 100 mg daily considering the endpoints change in monthly headache days (Figure 4.8.2) and ≥50% response rate (Figure 4.8.3). The quality of evidence for both outcomes was considered low (Table 4.8.2) as the RCT was designed to address tolerability and not efficacy as primary endpoint.
GRADE evidence profile table for subcutaneous erenumab versus oral topiramate in any migraine (no distinction between episodic and chronic)

Only one trial; bPrimary outcome of the trial was tolerability and not efficacy.
GRADE evidence profile table for oral topiramate 100mg or 200mg daily vs oral propranolol 160mg daily in patients with episodic migraine

Only one trial.
GRADE evidence profile table for oral topiramate 100 mg daily vs oral propranolol 160mg daily in patients with chronic migraine

Only one trial.
GRADE evidence profile table for oral topiramate 50mg daily vs oral propranolol 80mg daily in patients with any migraine (no distinction between episodic and chronic)

Only one trial; blow numbers of patients (<50 per group).
GRADE evidence profile table for oral topiramate 100 mg daily vs oral amitriptyline up to 100 mg daily in patients with any migraine (no distinction between episodic and chronic)

Only one trial.
GRADE evidence profile table for oral topiramate 50mg daily vs oral lamotrigine 50mg daily in patients with any migraine (no distinction between episodic and chronic)

Only one trial.
GRADE evidence profile table for oral propranolol 160 mg daily vs oral candesartan 16 mg daily in patients with any migraine (no distinction between episodic and chronic)
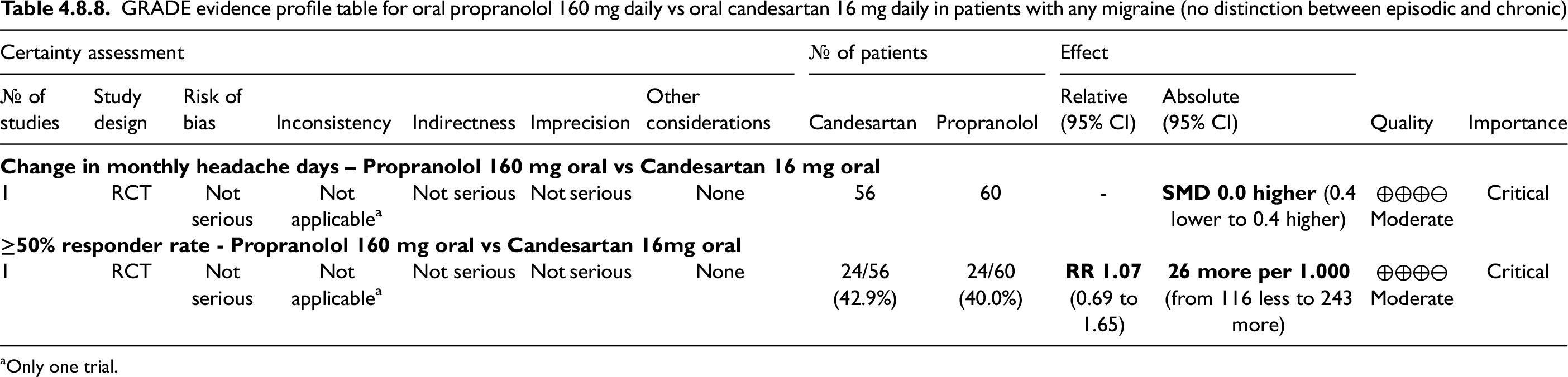
Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.1 In subjects with any migraine (no distinction between episodic and chronic), we recommend subcutaneous erenumab 70 mg or 140 mg monthly over oral topiramate up to 100 mg daily for migraine prevention.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: Strong (↑↑)
Safety and tolerability of erenumab compared with topiramate
In the only head-to-head RCT (HER-MES) of a monoclonal antibody acting on the CGRP pathway, erenumab was more effective than topiramate for migraine prevention (837). The primary endpoint of HER-MES was tolerability, which was met as erenumab showed a far higher tolerability than topiramate. For those reasons, monoclonal antibodies acting on the CGRP pathway could be preferable to oral migraine preventive treatments.
Main evidence
None.
Additional evidence
We found two RCTs comparing onabotulinumtoxinA with topiramate in subjects with chronic migraine (662,663). Overall, the risk of bias of included studies was high (Figure 4.8.3).
The meta-analysis of RCTs did not show any benefit of onabotulinumtoxinA (100–200 units) over topiramate up to 100 mg daily for ≥50% response (Figure 4.8.4). The quality of evidence for the outcome was considered very low as the RCTs had a serious risk for indirectness and imprecision. Indirectness was due to the lower dose of onabotulinumtoxinA used in the RCTs – 100–200 IUs – compared with clinical practice – 155–195 IU. Imprecision was due to the low number of participants in each RCT. No recommendation was issued due to the low numbers of participants (<50 in each group).
Evidence-based recommendation for PICO 4.8.2 None.
Quality of evidence: -
Strength of the recommendation: -

Forest plot showing the comparison between intramuscular onabotulinumtoxinA (100–200 units) and oral topiramate (up to 100 mg daily) for the outcome ≥50% response rate in patients with chronic migraine.
Safety and tolerability of onabotulinumtoxinA over topiramate
According to the available evidence, the efficacy profile of onabotulinumtoxinA is comparable to that of topiramate. However, the two drugs might have a different tolerability profile. Topiramate is an anticonvulsant medication that crosses the blood-brain barrier, while onabotulinumtoxinA is applied locally and has no systemic effects. Therefore, it is expected that onabotulinumtoxinA has a better tolerability profile than topiramate. Surprisingly, the RCTs included in this guideline did not find any difference in adverse events between onabotulinumtoxinA and topiramate.
Main evidence
None.
Additional evidence
We found one RCT comparing onabotulinumtoxinA (100 UI) with valproate (250 mg bid) in subjects with any migraine (766). The risk of bias was high (Figure 4.8.5).

Forest plot showing the comparison between intramuscular onabotulinumtoxinA (100 units) and oral valproate (250 mg bid) for the outcome change in monthly migraine days in patients with any migraine (no distinction between episodic and chronic).
The RCT did not show any benefit of onabotulinumtoxinA over valproate for the outcome of change in monthly migraine days (Figure 4.8.5). The quality of evidence for the outcome was considered very low as the RCT had a serious risk for indirectness and imprecision. Indirectness was due to the lower dose of onabotulinumtoxinA compared with clinical practice, i.e., 100 IU instead of 155–195 IU. Imprecision was due to the low number of participants in the RCT. It should also be noted that 100 IU of onabotulinumtoxinA is a dosage that did not prove superior to placebo (see Section 4.5). No recommendation was issued due to the low numbers of participants (<50 in each group).
Evidence-based recommendation for PICO 4.8.3 None. Quality of evidence: - Strength of the recommendation: -
Safety and tolerability of onabotulinumtoxinA over valproate
According to the available evidence, the efficacy profile of onabotulinumtoxinA is comparable to that of valproate. However, onabotulinumtoxinA was better tolerated compared with valproate, with less adverse events and less dropouts due to adverse events. Although this result suggests that onabotulinumtoxinA might be more tolerable than valproate with similar efficacy, and thus generally preferable to valproate, we should also note that the evidence comes from a single RCT on few patients and with a lower dose of onabotulinumtoxinA compared with current clinical practice. Additionally, the RCT was performed in subjects with either episodic or chronic migraine, while the current indication of onabotulinumtoxinA is chronic migraine only.
Main evidence
None.
Additional evidence
We found one RCT comparing onabotulinumtoxinA (250 UI) to amitriptyline (25–50 mg daily) in subjects with chronic migraine (590). The RCT did not meet the quality criteria (Figure 4.8.6) as it lacked a clear superiority or non-inferiority hypothesis and a sample size estimate.

Forest plot showing the comparison between intramuscular onabotulinumtoxinA (250 U quarterly) and oral amitriptyline (25–50 mg daily) for the outcome ≥50% response rate in patients with chronic migraine.
The RCT did not show differences between onabotulinumtoxinA and amitriptyline considering the outcome of ≥50% responder rate (Figure 4.8.6). The quality of evidence was considered very low due to the availability of only one RCT and the high risk of bias. Imprecision was also a concern as onabotulinumtoxinA was used in the RCT at higher doses than those used in clinical practice (250 U vs 155–195 U). No recommendation was issued due to the low numbers of participants (<50 in each group).
Evidence-based recommendation for PICO 4.8.4 None. Quality of evidence: - Strength of recommendation: -
Safety and tolerability of onabotulinumtoxinA over amitriptyline
According to the available evidence, the efficacy profile of onabotulinumtoxinA is comparable to that of amitriptyline. We should note that the dosage of onabotulinumtoxinA used in the trial (250 IU) was higher than the maximum dosage used in clinical practice (195 IU).
Main evidence
None.
Additional evidence
We found one RCT comparing topiramate (50 mg daily) to valproate (400 mg daily) in subjects with episodic migraine (654) that did not meet the criteria to be included in the main evidence due to the absence of a pre-specified sample size calculation. The risk of bias of the RCT was high (Figures 4.8.7 and 4.8.8).

Forest plot showing the comparison between oral topiramate 50 mg daily and oral valproate 400 mg daily for the outcome persisting monthly headache days in patients with episodic migraine.

Forest plot showing the comparison between oral topiramate 50 mg daily and oral valproate 400 mg daily for the outcome ≥50% response rate in patients with episodic migraine.
The RCT showed no benefit of topiramate over valproate considering the outcomes of monthly headache days (Figure 4.8.7) and ≥50% responder rate (Figure 4.8.8). The quality of evidence for both outcomes was considered very low due to the low number of subjects and high risk of bias. Additionally, both drugs were used at lower doses than those tested against placebo. No recommendation was issued due to the low numbers of participants (<50 in each group).
Evidence-based recommendation for PICO 4.8.5 None. Quality of evidence: - Strength of the recommendation: -
Safety and tolerability of topiramate compared with valproate
In the head-to-head RCT of topiramate versus valproate for episodic migraine, drug dosages and number of participants were low. Therefore, tolerability considerations are as difficult to draw as those on efficacy. In the included RCT, topiramate and valproate users reported comparable proportions of adverse events (64.3% vs 78.6%, respectively), even if they were different in quality; weight loss occurred in the topiramate group and weight gain in the valproate group.
Main evidence
None.
Additional evidence
We found one RCT comparing topiramate (75 mg daily) to valproate (750 mg daily) in subjects with chronic migraine (644) that did not meet the criteria to be included in the main evidence due to the absence of a pre-specified sample size calculation. The risk of bias of the RCT was high (Figures 4.8.9 and 4.8.10).
Forest plot showing the comparison between oral topiramate 75 mg daily and oral valproate 750 mg daily for the outcome persisting monthly headache days in patients with chronic migraine.

Forest plot showing the comparison between oral topiramate 75 mg daily and oral valproate 750 mg daily for the outcome ≥50% response rate in patients with chronic migraine.
The RCT did not show any difference between topiramate and valproate considering the endpoints persisting monthly headache days (Figure 4.8.9) and ≥50% responder rate (Figure 4.8.10). The quality of evidence for both outcomes was considered very low due to the low number of subjects and high risk of bias. No recommendation was issued due to the low numbers of participants (<50 in each group).
Evidence-based recommendation for PICO 4.8.6 None. Quality of evidence: - Strength of the recommendation: -
Safety and tolerability of topiramate compared with valproate
In the head-to-head RCT of topiramate over valproate for chronic migraine, safety and tolerability were not specifically assessed. The number of dropouts was similar in both the topiramate and valproate groups (three subjects each); however, this is not enough evidence to conclude on a similar tolerability profile between the two drugs. Besides, the two drugs have different adverse event profiles, which should be considered in clinical practice for treatment choice.
Main evidence
None.
Additional evidence
We found one RCT comparing topiramate (50 mg daily) to valproate (400 mg daily) in subjects with any migraine (no distinction between episodic and chronic) (652) that did not meet the criteria to be included in the main evidence due to the absence of a pre-specified sample size calculation. The risk of bias of the RCT was high (Figure 4.8.11).
Forest plot showing the comparison between oral topiramate 50 mg daily and oral valproate 400 mg daily for the outcome change in monthly headache days in patients with any migraine (no distinction between episodic and chronic).
The RCT showed a slight benefit of topiramate over valproate considering the endpoint change in monthly headache days (Figure 4.8.11). The quality of evidence for the outcome was considered very low due to the low number of subjects and high risk of bias. A further concern is that topiramate and valproate were both used at lower doses than those tested in placebo-controlled trials. No recommendation was issued due to the low numbers of participants (<50 in each group).
Evidence-based recommendation for PICO 4.8.7 None. Quality of evidence: - Strength of the recommendation: -
Safety and tolerability of topiramate compared with valproate
In the head-to-head RCT of topiramate over valproate for migraine (no distinction between episodic and chronic), safety and tolerability were comparable between the two drugs with a different profile of adverse events. Notably, no dropouts were recorded in any of the two groups. It is likely that valproate and topiramate had a low proportion of adverse events because of the low doses used in the RCT.
Main evidence
We found one RCT comparing topiramate (100 mg or 200 mg daily) to propranolol (160 mg daily) in subjects with episodic migraine (628) that met the criteria to be included in the main evidence. The risk of bias of the RCT was low (Figures 4.8.12 and 4.8.13).

Forest plot showing the comparison between oral topiramate 100 mg or 200 mg daily and oral propranolol 160 mg daily for the outcome change in monthly headache days in patients with episodic migraine.

Forest plot showing the comparison between oral topiramate 100 mg or 200 mg daily and oral propranolol 160 mg daily for the outcome ≥50% response rate in patients with episodic migraine.
The RCT showed no difference between topiramate and propranolol considering the endpoints change in monthly headache days (Figure 4.8.12) and ≥50% responder (Figure 4.8.13). The quality of evidence for both outcomes was considered moderate (Table 4.8.3) due to the availability of only one RCT. Nevertheless, the RCT included a placebo group together with active groups, which increases the reliability of findings.
GRADE evidence profile table for oral metoprolol 200 mg daily versus oral flunarizine 10 mg daily in patients with any migraine (no distinction between episodic and chronic)

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.8 In subjects with episodic migraine, we suggest either oral topiramate 100 mg or 200 mg daily or oral propranolol 160 mg daily as equivalent options for migraine prevention.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: =
Safety and tolerability of topiramate compared with valproate
In the head-to-head RCT of topiramate over propranolol for chronic migraine, adverse events leading to treatment limitation or discontinuation were more frequent in the topiramate 200 mg group (44%) compared with the topiramate 100 mg group (28%) and the propranolol 160 mg group (20%). Therefore, based on safety data, it could be reasonable to treat subjects with a 100 mg daily dose of topiramate rather than the 200 mg daily dose, which could not give any efficacy advantage while increasing the probability of tolerability issues.
Main evidence
We found one RCT comparing oral topiramate (100 mg daily) with oral propranolol (160 mg daily) in subjects with chronic migraine (656) that met the criteria to be included in the main evidence. The risk of bias of the RCT was low (Figure 4.8.14).
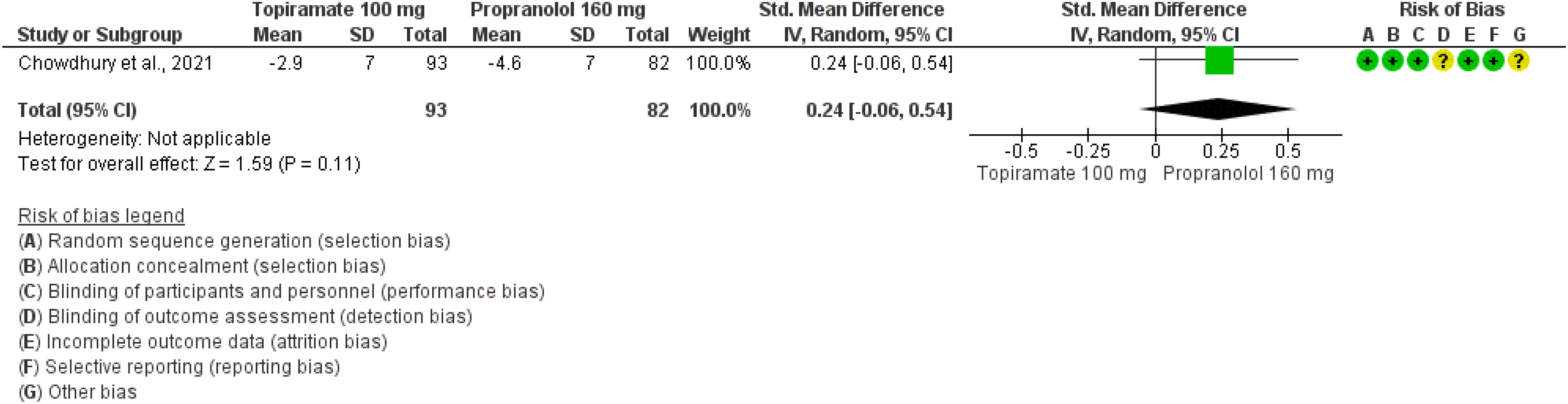
Forest plot showing the comparison between oral topiramate 100 mg daily and oral propranolol 160 mg daily for the outcome change in monthly headache days in patients with chronic migraine.
The RCT showed no benefit of topiramate over propranolol considering the outcome change in monthly headache days (Figure 4.8.14). The quality of evidence for the outcome was considered moderate (Table 4.8.4) due to the availability of only one RCT.
GRADE evidence profile table for subcutaneous galcanezumab (120 mg monthly with 240 mg loading dose) vs oral rimegepant dispersible tablets (75 mg every other day) in patients with any migraine (no distinction between episodic and chronic)

Only one trial.
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.9 In subjects with chronic migraine, based on efficacy data, we suggest oral topiramate 100 mg daily and oral propranolol 160 mg daily as equivalent options for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of the recommendation: Weak (=)
Safety and tolerability of topiramate compared with propranolol
In the head-to-head RCT of topiramate over propranolol for chronic migraine, the safety profile was comparable between the two drugs, even if the kind of adverse events was different between the two drugs. Literature suggests that the choice between topiramate and propranolol be done according to safety more than to efficacy data.
Main evidence
We found one RCT comparing topiramate (50 mg daily) with propranolol (80 mg daily) in subjects with migraine (no distinction between episodic and chronic) (649) that met the criteria to be included in the main evidence. The risk of bias of the RCT was considered moderate (Figure 4.8.15) due to low numbers.

Forest plot showing the comparison between oral topiramate 50 mg daily and oral propranolol 80 mg daily for the outcome change in monthly headache days in patients with any migraine (no distinction between episodic and chronic).
The RCT showed a benefit of topiramate over propranolol considering the outcome change in monthly headache days (Figure 4.8.15). The quality of evidence for the outcome was considered moderate (Table 4.8.5) due to the availability of only one RCT.
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.10 In subjects with any migraine (no distinction between episodic and chronic), we suggest oral topiramate 50 mg daily over oral propranolol 80 mg daily for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)
Safety and tolerability of topiramate compared with propranolol
In the head-to-head RCT of topiramate versus propranolol for migraine (no distinction between episodic and chronic), the safety profile was comparable between the two drugs. It should be noted that both drugs were used in lower doses if compared with those of placebo controlled RCTs.
Main evidence
We found one RCT comparing topiramate (50 mg daily) with amitriptyline (recommended dose 25–100 mg daily) in subjects with migraine (596) that met the criteria to be included in the main evidence. The risk of bias of the RCT was low (Figure 4.8.16).

Forest plot showing the comparison between oral topiramate 100 mg daily and oral amitriptyline 25–100 mg daily for the outcome ≥50% response rate in patients with any migraine (no distinction between episodic and chronic).
The RCT showed that topiramate up to 100 mg daily was at least as effective as amitriptyline 100 mg daily considering the outcome of ≥50% responder rate (Figure 4.8.16). The quality of evidence for the outcome was considered moderate (Table 4.8.6) due to the availability of only one RCT.
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.11 In subjects with any migraine (no distinction between episodic and chronic), based on efficacy data, we suggest oral topiramate (100–200 mg daily) and oral amitriptyline (25–150 mg daily) as equivalent options for migraine prevention.
Quality of evidence: Low (⊕⊕⊖⊖)
Strength of the recommendation: =
Main evidence
None.
Additional evidence
We found one RCT comparing topiramate (up to 200 mg daily) with amitriptyline (up to 150 mg daily) in subjects with episodic migraine (589) that did not meet the criteria to be included in the main evidence as it did not pre-specify a sample size calculation. The risk of bias of the RCT was serious (Figure 4.8.17).

Forest plot showing the comparison between oral topiramate 200 mg daily and oral amitriptyline 150 mg daily for the outcome monthly headache days in patients with episodic migraine.
The RCT showed that topiramate was at least as effective as amitriptyline considering the outcome of monthly headache days (Figure 4.8.17). The quality of evidence for the outcome was considered very low due to the availability of only one RCT.
Evidence-based recommendation for PICO 4.8.12 In subjects with episodic migraine, we suggest oral topiramate (200 mg daily) and oral amitriptyline (150 mg daily) as equivalent options for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =
Safety and tolerability of topiramate compared with amitriptyline
In the head-to-head RCTs of topiramate versus amitriptyline for migraine (no distinction between episodic and chronic), the safety profile was comparable between the two drugs.
Main evidence
None.
Additional evidence
We found one RCT comparing topiramate (100 mg daily) with flunarizine (5 mg daily) and a combination of the two drugs in subjects with migraine (634) that did not meet the criteria to be included in the main evidence due to the lack of sample size calculation. The risk of bias of the RCT was high (Figure 4.8.18). For the present analysis, we considered the topiramate and flunarizine arm and discarded the arm of combination treatment.

Forest plot showing the comparison between oral topiramate 100 mg and oral flunarizine 5 mg daily for the outcome 50% response rate in patients with any migraine (no distinction between episodic and chronic).
The RCT did not show any difference between topiramate up to 100 mg daily and flunarizine 5 mg daily considering the outcome of ≥50% response rate (Figure 4.8.18). No recommendation was issued due to the low numbers of participants (<50 in each group).
Evidence-based recommendation for PICO 4.8.13 None. Quality of evidence: - Strength of the recommendation: -
Safety and tolerability of topiramate compared with flunarizine
In the head-to-head RCTs of topiramate over flunarizine for migraine, adverse events leading to treatment limitation or discontinuation were comparable between the two groups, in the absence of dropouts. However, the high risk of bias of the included RCT did not allow to draw conclusions on potential safety differences.
Main evidence
We found one RCT comparing topiramate (50 mg daily) with lamotrigine (50 mg daily) in subjects with migraine (638) that met the criteria to be included in the main evidence. The risk of bias of the RCT was high (Figures 4.8.19 and 4.8.20).

Forest plot showing the comparison between oral topiramate 50 mg and oral lamotrigine 50 mg daily for the outcome change in monthly headache days in patients with any migraine (no distinction between episodic and chronic).

Forest plot showing the comparison between oral topiramate 50 mg and oral lamotrigine 50 mg daily for the outcome ≥50% response rate in patients with any migraine (no distinction between episodic and chronic).
The RCT did not show any difference between topiramate 50 mg daily and lamotrigine 50 mg daily considering the outcomes of change in monthly headache days (Figure 4.8.19) and ≥50% responder rate (Figure 4.8.20). The quality of evidence for both outcomes was considered low (Table 4.8.7) due to the availability of only one RCT and the high risk of bias. Nevertheless, the RCT included a placebo group together with active groups, which increases the reliability of findings.
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.14 In subjects with any migraine (no distinction between episodic and chronic), we suggest oral topiramate 50 mg daily and oral lamotrigine 50 mg daily as equivalent options for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: =
Safety and tolerability of topiramate vs lamotrigine
Although numerically higher (15% vs 10%), the rate of adverse events in patients treated with topiramate 50 mg did not significantly differ from that of patients treated with lamotrigine 50 mg daily. Notably, the two doses of both drugs were lower than those used in comparable RCTs and lower than those commonly used in clinical practice.
Main evidence
None.
Additional evidence
We found one RCT comparing topiramate 100 mg daily to zonisamide 200 mg daily in subjects with episodic migraine (624). This RCT did not meet the quality criteria for main evidence and therefore the quality of evidence was considered low.
The study did not show differences between topiramate up to 100 mg daily and zonisamide up to 200 mg daily considering the outcomes of persisting monthly headache days (Figure 4.8.21) and ≥50% responder rate (Figure 4.8.22). The quality of evidence for both outcomes was considered very low due to the availability of only one RCT and the high risk of bias. No recommendation was issued due to the low numbers of participants (<50 per group).
Evidence-based recommendation for PICO 4.8.15 None. Quality of evidence: - Strength of recommendation: -
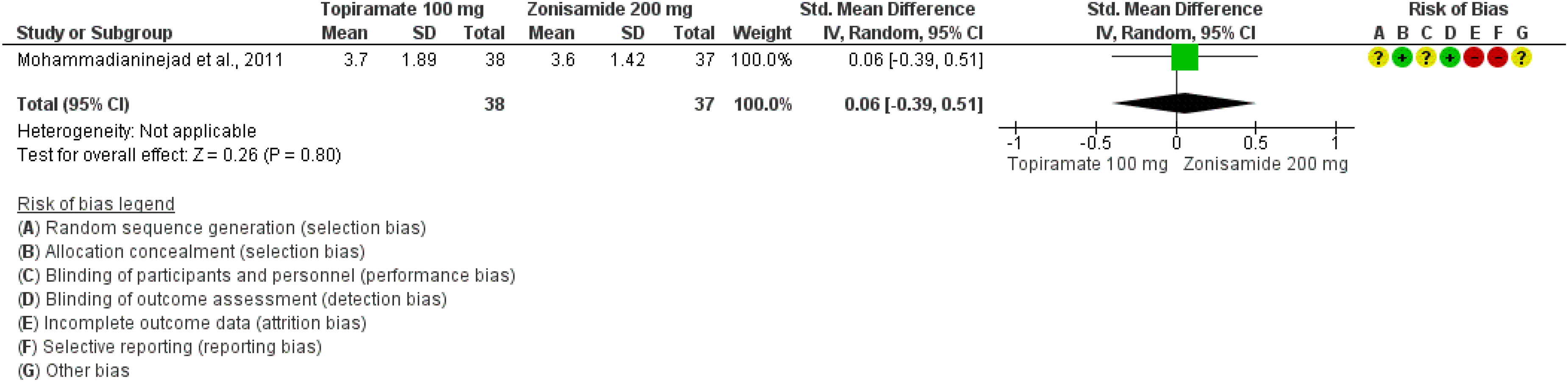
Forest plot showing the comparison between oral topiramate 100 mg and oral zonisamide 200 mg daily for the outcome persisting monthly headache days in patients with episodic migraine.

Forest plot showing the comparison between oral topiramate 100 mg and oral zonisamide 200 mg daily for the outcome ≥50% response rate in patients with episodic migraine.
Safety and tolerability of topiramate compared with zonisamide
The RCT comparing topiramate with zonisamide did not find differences in the safety of the two drugs. It should be noted that the evidence base for topiramate is much larger than that of zonisamide, which does not have a placebo-controlled trial for migraine prevention.
Main evidence
None.
Additional evidence
We found two RCTs comparing valproate to cinnarizine in subjects with episodic migraine (648,655). Those RCTs did not meet the pre-defined quality criteria (Figures 4.8.23 and 4.8.24) and for this reason the quality of evidence was considered very low.

Forest plot showing the comparison between oral valproate (400 or 600 mg daily) and oral cinnarizine (50 or 75 mg daily) for the outcome persisting monthly headache days in patients with episodic migraine.

Forest plot showing the comparison between oral valproate (600 mg daily) and oral cinnarizine (75 mg daily) for the outcome ≥50% response rate in patients with episodic migraine.
The studies did not show differences between valproate (400 mg or 600 mg daily) and cinnarizine (50 mg or 75 mg daily) considering the outcome of persisting monthly headache days (Figure 4.8.23), nor between valproate 600 mg daily and cinnarizine 75 mg daily considering the outcome and ≥50% responder rate (Figure 4.8.24).
Evidence-based recommendation for PICO 4.8.16 In subjects with episodic migraine, we suggest oral valproate (400 or 600 mg daily) and oral cinnarizine (50 or 75 mg daily) as equivalent options for migraine prevention. Quality of evidence: very low (⊕⊖⊖⊖). Strength of recommendation: = .
Safety and tolerability of valproate compared with cinnarizine
The tolerability of valproate and cinnarizine was comparable in the RCT. However, cinnarizine does not have a RCT with a comparison against placebo. Therefore, the evidence base for use in clinical practice is higher for valproate than for cinnarizine.
Main evidence
None.
Additional evidence
We found one RCT comparing valproate to amitriptyline in subjects with episodic migraine (600). The RCT did not meet the pre-defined quality criteria (Figure 4.8.25) and for this reason the quality of evidence was considered very low.

Forest plot showing the comparison between valproate (1000 mg daily) and amitriptyline (100 mg daily) for the outcome persisting monthly headache days in patients with episodic migraine.
The study did not show differences between valproate 1000 mg daily and amitriptyline 100 mg daily considering the outcome of persisting monthly headache days (Figure 4.8.25). The quality of evidence for monthly headache days was considered very low due to the availability of only one RCT and the high risk of bias. No recommendation was issued due to the low numbers of participants (<50 in each group).
Evidence-based recommendation for PICO 4.8.17 None. Quality of evidence: - Strength of recommendation: -
Safety and tolerability of valproate compared with amitriptyline
In the RCT comparing valproate with amitriptyline, there was no difference in safety between the two drugs. However, the low number of patients reduces the reliability of safety considerations.
Main evidence
None.
Additional evidence
We found one RCT comparing amitriptyline (75 mg daily) to venlafaxine (150 mg daily) in subjects with migraine (588). The RCT did not meet the quality criteria for main evidence (Figure 4.8.26) and for this reason the quality of evidence was considered low.

Forest plot showing the comparison between oral amitriptyline 75 mg daily and oral venlafaxine 150 mg daily for the outcome persisting monthly headache days in patients with migraine (no distinction between episodic and chronic).
The study did not show differences between amitriptyline and venlafaxine considering the outcome of persisting monthly headache days (Figure 4.8.26). The quality of evidence was considered low due to the availability of only one RCT.
An additional RCT compared amitriptyline at a daily oral dosage of 25 mg and venlafaxine at a daily oral dose of 37.5 mg (875). The RCT could not be included in quantitative analyses as it did not contain the outcomes considered for quantitative analyses. The RCT showed no difference in efficacy between amitriptyline and venlafaxine in decreasing the number of monthly headache attacks. Conversely, amitriptyline showed a worse tolerability profile, as the adverse drug reactions in the amitriptyline group exceeded those of the venlafaxine group.
Evidence-based recommendation for PICO 4.8.18 In subjects with any migraine (no distinction between episodic and chronic) we suggest oral amitriptyline 75 mg daily and oral venlafaxine 150 mg daily as equivalent options for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of recommendation: Weak (=)
Safety and tolerability of amitriptyline compared with venlafaxine
Safety and tolerability of the two drugs were comparable in the only available head-to-head RCT. The evidence base for placebo-controlled RCTs is higher for amitriptyline than for venlafaxine. However, venlafaxine has a better antidepressant profile than amitriptyline. Therefore, in patients with depressive comorbidities venlafaxine could be a suitable option.
Main evidence
None.
Additional evidence
We found one RCT comparing amitriptyline (50 mg daily) to citalopram (20 mg daily) in subjects with migraine (591). The RCT did not meet the pre-defined quality criteria (Figure 4.8.27) and for this reason the quality of evidence was considered low.
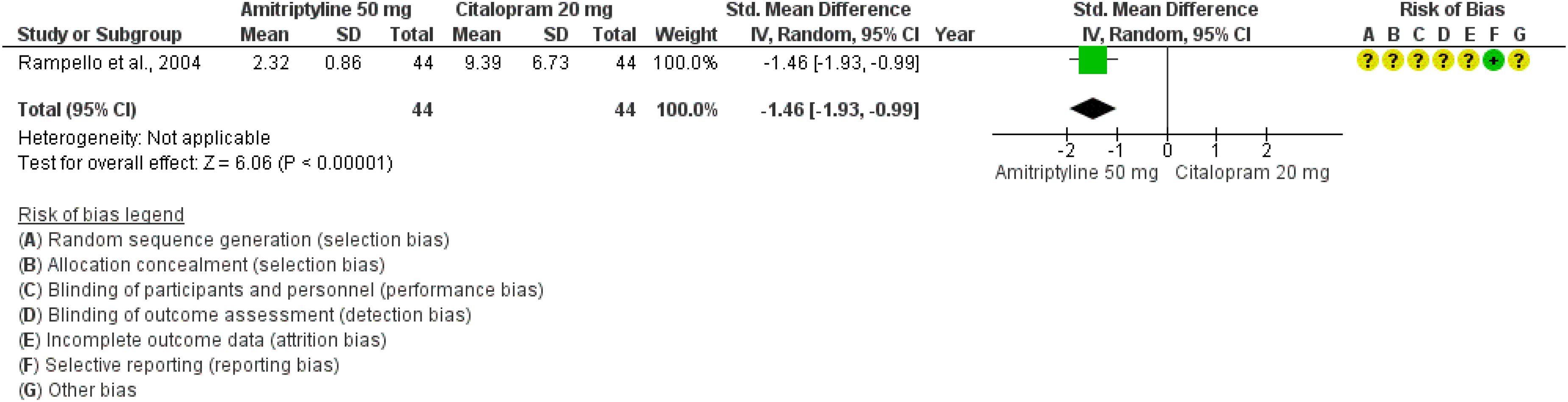
Forest plot showing the comparison between oral amitriptyline 50 mg daily and oral citalopram 20 mg daily for the outcome persisting monthly headache days in patients with any migraine (no distinction between episodic and chronic).
The study did showed the superiority of amytriptiline over citalopram considering the outcome of persisting monthly headache days (Figure 4.8.27). The quality of evidence was considered low due to the availability of only one RCT. It should also be noted that citalopram was not tested against placebo for migraine prevention. No recommendation was issued due to low numbers of participants (<50 per group).
Evidence-based recommendation for PICO 4.8.19 None. Quality of evidence: - Strength of recommendation: -
Main evidence
None.
Additional evidence
We found one RCT comparing venlafaxine (37.5 mg daily) to nortriptyline (25 mg daily) plus propranolol (40 mg daily) in subjects with migraine (601). The RCT did not meet the pre-defined quality criteria (Figure 4.8.28) and for this reason the quality of evidence was considered low.

Forest plot showing the comparison between oral venlafaxine 37.5 mg daily and oral nortriptyline 25 mg daily plus propranolol 40 mg daily for the outcome persisting monthly headache days in patients with any migraine (no distinction between episodic and chronic).
The study did not show differences between venlafaxine and nortriptyline plus propranolol considering the outcome of persisting monthly headache days (Figure 4.8.28). The quality of evidence was considered very low due to the availability of only one RCT and to the high risk of bias. No recommendation was issued due to the low number of participants (<50 per group).
Evidence-based recommendation for PICO 4.8.20 None. Quality of evidence: - Strength of recommendation: -
Main evidence
We found one RCT comparing propranolol (160 mg daily) to candesartan (16 mg daily) for migraine prevention (709). The RCT met the quality criteria for main evidence (Figure 4.8.30) and had a low risk of bias.
The study did not show differences between propranolol and candesartan considering the outcomes of persisting monthly headache days (Figure 4.8.29) and ≥50% responder rate (Figure 4.8.30). The quality of evidence was considered moderate (Table 4.8.8) due to the availability of only one RCT.

Forest plot showing the comparison between oral propranolol 160 mg daily and oral candesartan 16 mg daily for the outcome persisting monthly headache days in patients with any migraine (no distinction between episodic and chronic).
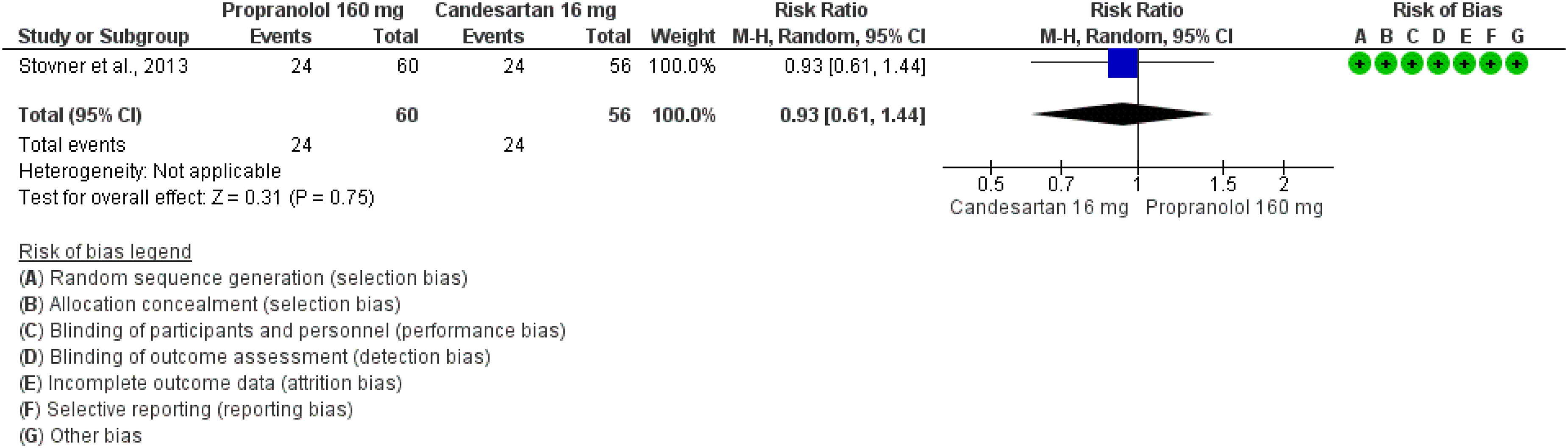
Forest plot showing the comparison between oral propranolol 160 mg daily and oral candesartan 16 mg daily for the outcome ≥50% response rate in patients with any migraine (no distinction between episodic and chronic).
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.21 In subjects with any migraine (no distinction between episodic and chronic), we suggest oral propranolol (160 mg daily) and candesartan (16 mg daily) as equivalent options for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of recommendation: Weak (=)
Main evidence
None.
Additional evidence
We found one RCT comparing propranolol (80 mg daily) to nortriptyline (40 mg daily) for migraine prevention (705). The RCT did not meet the pre-defined quality criteria (Figure 4.8.32) due to the lack of a pre-specified sample size calculation and had a high risk of bias.
The study did not show differences between propranolol and nortriptyline considering the outcome ≥50% responder rate (Figure 4.8.31). The quality of evidence was considered very low due to the availability of only one RCT and the high risk of bias. It should be mentioned that nortriptyline does not have any placebo-controlled RCT. No recommendation was issued due to the low numbers of participants (<50 per group).
Evidence-based recommendation for PICO 4.8.22 None. Quality of evidence: - Strength of recommendation: -

Forest plot showing the comparison between propranolol 80 mg daily and nortriptyline 40 mg daily for the outcome ≥50% response rate in patients with any migraine (no distinction between episodic and chronic).
Main evidence
We found one RCT comparing metoprolol (200 mg daily) with flunarizine (10 mg daily) in subjects with migraine (876). The risk of bias was unclear (Figure 4.8.32).

Forest plot showing the comparison between oral metoprolol 200 mg daily and oral flunarizine 10 mg daily for the outcome change in monthly migraine days in patients with any migraine (no distinction between episodic and chronic).
The RCT showed superiority of flunarizine over metoprolol for the outcome change in monthly migraine days (Figure 4.8.32). The quality of evidence for the outcome was considered low as there was only one available RCT with low numbers and an unclear risk of bias.
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.23 In subjects with any migraine (no distinction between episodic and chronic), we suggest oral flunarizine (10 mg daily) over oral metoprolol (200 mg daily) for migraine prevention. Quality of evidence: Low (⊕⊕⊖⊖). Strength of the recommendation: Weak (↑)
Main evidence
None.
Additional evidence
We found one RCT comparing oral metoprolol (80 mg daily) to oral acetylsalicylic acid (300 mg daily) for migraine prevention (699). The RCT did not meet the quality criteria for main evidence due to the lack of a sample size calculation and had a high risk of bias.
The study showed superiority of metoprolol over acetylsalicylic acid for the outcomes of persisting monthly headache days (Figure 4.8.33) and change in monthly headache days (Figure 4.8.34). The quality of evidence was considered very low due to the availability of only one RCT and the high risk of bias.
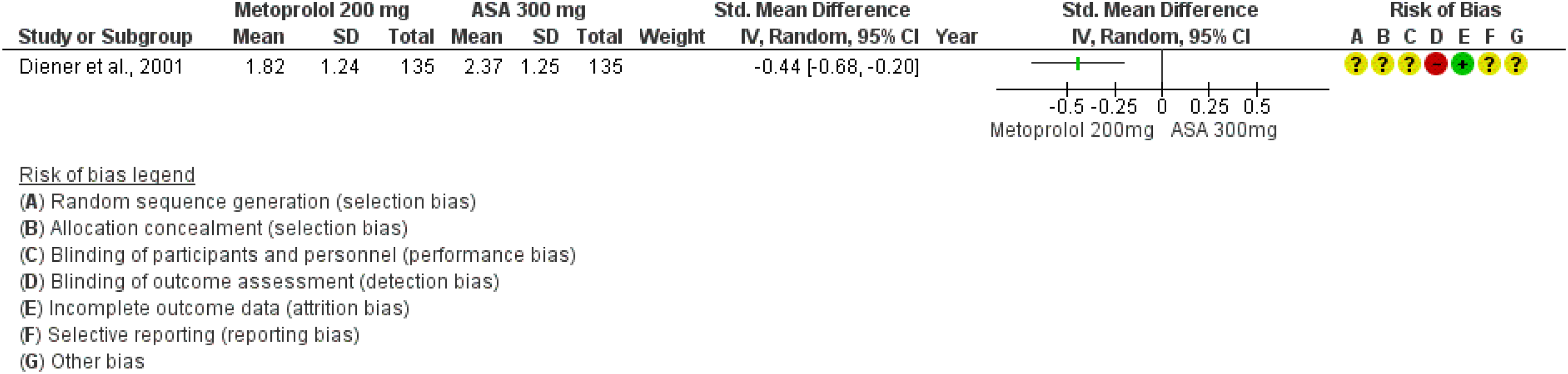
Forest plot showing the comparison between oral metoprolol 200 mg daily and oral acetylsalicylic acid 300 mg daily for the outcome persisting monthly headache days in patients with any migraine (no distinction between episodic and chronic).

Forest plot showing the comparison between oral metoprolol 200 mg daily and oral acetylsalicylic acid 300 mg daily for the outcome change in monthly headache days in patients with any migraine (no distinction between episodic and chronic).
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.24 In subjects with any migraine (no distinction between episodic and chronic), we suggest oral metoprolol (200 mg daily) over oral acetylsalicylic acid (300 mg daily) for migraine prevention.
Quality of evidence: Very low (⊕⊖⊖⊖)
Strength of the recommendation: Weak (↑)
Main evidence
None.
Additional evidence
We found one RCT comparing oral propranolol (60 mg three times per day - TID) to nifedipine (30 mg three times per day - TID) for migraine prevention (701). The RCT did not meet the criteria for main evidence (Figure 4.8.35) due to the lack of a pre-specified sample size calculation and had a high risk of bias.

Forest plot showing the comparison between oral propranolol 60 mg three times per day and oral nifedipine 30 mg three times per day for the outcome persisting monthly headache days in patients with any migraine (no distinction between episodic and chronic).
The study showed not significant difference between the two drugs for the outcome of persisting monthly headache days (Figure 4.8.35). The quality of evidence was considered very low due to the availability of only one RCT and the high risk of bias. No recommendation was issued due to the low numbers of participants (<50 per group).
Evidence-based recommendation for PICO 4.8.25 None. Quality of evidence: - Strength of recommendation: -
Main evidence
We found one RCT comparing subcutaneous galcanezumab 120 mg monthly (with 240 mg loading dose) with oral rimegepant 75 mg (dispersible tablets) every other day (842) that met the criteria for main evidence. The risk of bias was considered low. The RCT showed no difference between galcanezumab and rimegepant considering the outcome of change in monthly migraine days (Figure 4.8.36) and ≥50% response rate (Figure 4.8.37). The quality of evidence was considered moderate (Table 4.8.25) due to the availability of only one RCT.

Forest plot showing the comparison between subcutaneous galcanezumab (120 mg monthly with 240 mg loading dose) and oral rimegepant dispersible tablet (75 mg every other day) for the outcome change in monthly migraine days in patients with any migraine (no distinction between episodic and chronic).

Forest plot showing the comparison between subcutaneous galcanezumab (120 mg monthly with 240 mg loading dose) and oral Rimegepant dispersible tablet (75 mg every other day) for the outcome ≥50% response rate in patients with episodic migraine.
Additional evidence
None.
Evidence-based recommendation for PICO 4.8.26 In subjects with episodic migraine, we recommend subcutaneous galcanezumab (120 mg monthly with 240 mg loading dose) and oral rimegepant dispersible tablets (75 mg every other day) as equivalent options for migraine prevention.
Quality of evidence: Moderate (⊕⊕⊕⊖)
Strength of recommendation: Strong (=)
Safety and tolerability of galcanezumab compared with rimegepant
Both galcanezumab and rimegepant act on the CGRP pathway, the former as a ligand and the latter as a receptor antagonist. The safety profile of both drugs is excellent due to their specific mechanism of action. Both drugs should be used with caution in patients with previous vascular events, Raynaud's phenomenon, severe constipation, and bowel disease (see Sections 4.6 and 4.7). Given the equivalent efficacy of the two drugs, patients’ preference and attitude toward the route of administration (oral or subcutaneous) is important. The oral route might be preferable for patients who do not manage injections, while the subcutaneous route could be preferable in those with poor compliance to oral medications.
Summary of evidence for head-to-head comparison trials of preventive treatments
Figure 4.8.38 shows the summary of evidence for comparisons reported in the present section.

Summary of evidence for head-to-head comparisons of preventive treatments.
Expert-based opinion
Topic: Other head-to-head comparisons between preventive treatments
Topic: Which is the best sequence of oral preventive treatments for migraine?
Topic: When choosing an oral preventive treatment for migraine, should clinicians prioritize efficacy or safety?
Topic: When combining more than one oral preventive treatment for migraine, should dose of individual drugs be reduced?
Topic: When combining more than one oral preventive treatment for migraine, which safety elements should be considered?
Supplemental Material
sj-docx-1-cep-10.1177_03331024241305381 - Supplemental material for Evidence-based guidelines for the pharmacological treatment of migraine
Supplemental material, sj-docx-1-cep-10.1177_03331024241305381 for Evidence-based guidelines for the pharmacological treatment of migraine by Raffaele Ornello, Valeria Caponnetto, Fayyaz Ahmed, Haidar M. Al-Khazali, Anna Ambrosini, Sait Ashina, Carlo Baraldi, Alessia Bellotti, Filippo Brighina, Paolo Calabresi, Francesco Casillo, Sabina Cevoli, Shuli Cheng, Chia-Chun Chiang, Alberto Chiarugi, Rune Hackert Christensen, Min Kyung Chu, Gianluca Coppola, Ilenia Corbelli, Santiago Crema, Roberto De Icco, Marina de Tommaso, Cherubino Di Lorenzo, Vincenzo Di Stefano, Hans-Christoph Diener, Esme Ekizog. lu, Adriana Fallacara, Valentina Favoni, Kimberly N. Garces, Pierangelo Geppetti, Maria Teresa Goicochea, Antonio Granato, Franco Granella, Simona Guerzoni, Woo-Seok Ha, Amr Hassan, Koichi Hirata, Jan Hoffmann, Eva-Maria Hussler, Mona Hussein, Luigi Francesco Iannone, Bronwyn Jenkins, Alejandro Labastida-Ramirez, Anna Laporta, Morris Levin, Antonino Lupica, Edoardo Mampreso, Daniele Martinelli, Teshamae S. Monteith, Ilaria Orologio, Aynur Ozge, Li-Ling Hope Pan, Lavindren Luke Panneerchelvam, Mario F. P. Peres, Marcio Nattan Portes Souza, Patricia Pozo-Rosich, Maria Pia Prudenzano, Silvia Quattrocchi, Innocenzo Rainero, Volodymyr Romanenko, Marina Romozzi, Antonio Russo, Grazia Sances, Paola Sarchielli, Todd J. Schwedt, Marcello Silvestro, Diego Belandrino Swerts, Cristina Tassorelli, Alessandro Tessitore, Mansoureh Togha, Gloria Vaghi, Shuu-Jiun Wang, Messoud Ashina, and Simona Sacco in Cephalalgia
Footnotes
Acknowledgements

| SISC-IHS Working Group | ||
|---|---|---|
| 1. Summary of recommendations | Raffaele Ornello, Simona Sacco | Department of Biotechnological and Applied Clinical Sciences, University of L'Aquila, L'Aquila, Italy |
| 2. Guideline methods | Valeria Caponnetto | University of L'Aquila, Department of Life, Health and Environmental Sciences, l’Aquila, Italy |
| Raffaele Ornello, Simona Sacco | Department of Biotechnological and Applied Clinical Sciences, University of L'Aquila, L'Aquila, Italy | |
| Eva-Maria Huessler | Institute for Medical Informatics, Biometry and Epidemiology, University Hospital Essen | |
| Messoud Ashina | Danish Headache Center, Department of Neurology, Rigshospitalet - Glostrup, Faculty of Health and Medical Sciences, University of Copenhagen, 2600, Glostrup, Denmark; Department of Clinical Medicine, Faculty of Health and Medical Sciences, University of Copenhagen, Copenhagen, Denmark | |
| 3.1. Non-steroidal anti-inflammatory drugs (NSAIDs) and cycloxygenase 2 (COX2) inhibitors | Maria Pia Prudenzano, Adriana Fallacara | Headache Center, Amaducci Neurological Clinic, Polyclinic Hospital-University Consortium Bari, Italy |
| Anna Laporta | Department of Basic Medical Science, Neuroscience and Sense Organs, University of Bari Aldo Moro, Bari, Italy | |
| Santiago Crema, Maria Teresa Goicochea | Headache Clinic, Neurology Department, Fleni, Buenos Aires, Argentina | |
| Marina de Tommaso | DiBrain Department, Neurophysiopathology Unit, Bari Aldo Moro University, Bari, Italy | |
| 3.2. Triptans | Antonio Russo, Marcello Silvestro, Ilaria Orologio, Alessandro Tessitore | Headache Centre of Department of Advanced Medical and Surgical Sciences University of Campania “Luigi Vanvitelli” Naples, Italy |
| Kimberly Garces, Teshamae S. Monteith | Department of Neurology-Headache Division, University of Miami, Miller School of Medicine, Miami, FL, USA | |
| 3.3. Paracetamol (acetaminophen) | Gianluca Coppola, Francesco Casillo | Department of Medico-Surgical Sciences and Biotechnologies, Sapienza University of Rome Polo Pontino ICOT, Latina (Italy), Rome, Italy |
| Esme Ekizoglu | Istanbul University, Istanbul Faculty of Medicine, Department of Neurology, Istanbul, Turkey | |
| Aynur Özge | Mersin University School of Medicine, Department of Neurology, Mersin, Turkey | |
| 3.4. Combination analgesics | Ilenia Corbelli, Paola Sarchielli | Neurological Clinic, Santa Maria Misericordiae Hospital, Department of Medicine, University of Perugia, Perugia, Italy |
| Alessia Bellotti | Neurology, Department of Medicine and Surgery, University of Perugia, Perugia, Italy | |
| Marcio Nattan Portes Souza | Hospital das Clínicas da Universidade de São Paulo, São Paulo, Brazil | |
| Volodymyr Romanenko | Ukrainian Medical Academy, Kyiv, Ukraine | |
| 3.5. Antiemetics | Marina Romozzi | Dipartimento di Neuroscienze, Università Cattolica del Sacro Cuore, Rome, Italy |
| Francesco Casillo, Gianluca Coppola | Department of Medico-Surgical Sciences and Biotechnologies, Sapienza University of Rome Polo Pontino ICOT, Latina (Italy), Rome, Italy | |
| Haidar M. Al-Khazali | Department of Neurology, Danish Headache Center, Copenhagen University Hospital - Rigshospitalet, Copenhagen, Denmark | |
| Mansoureh Togha | Headache Department, Iranian Center of Neurological Research, Neuroscience Institute, Tehran University of Medical Sciences and Headache Department, Neurology Ward, Sina Hospital, Medical School, Tehran University of Medical Sciences, Tehran, Iran | |
| Paolo Calabresi | Dipartimento di Neuroscienze, Organi di Senso e Torace, Fondazione Policlinico Universitario Agostino Gemelli IRCCS, and Dipartimento di Neuroscienze, Università Cattolica del Sacro Cuore, Rome, Italy | |
| 3.6. Opioids | Daniele Martinelli | IRCCS Mondino Foundation - Headache science and Neurorehabilitation Center, Pavia, Italy |
| Koichi Hirata | Dokkyo Medical University, Mibu, Japan | |
| Franco Granella | Unit of Neurosciences, Department of Medicine and Surgery, University of Parma, Parma, Italy | |
| 3.7. Ditans | Marina Romozzi | Dipartimento di Neuroscienze, Università Cattolica del Sacro Cuore, Rome, Italy |
| Li-Ling Hope Pan | Brain Research Center, National Yang Ming Chiao Tung University, Hsinchu, Taiwan | |
| Shuu-Jiun Wang | Department of Neurology, Taipei Veterans General Hospital, and College of Medicine, National Yang Ming Chiao Tung University, Taipei, Taiwan | |
| Paolo Calabresi | Dipartimento di Neuroscienze, Organi di Senso e Torace, Fondazione Policlinico Universitario Agostino Gemelli IRCCS, and Dipartimento di Neuroscienze, Università Cattolica del Sacro Cuore, Rome, Italy | |
| 3.8. Gepants for acute treatment | Simona Guerzoni, Carlo Baraldi | Digital and Predictive Medicine, Pharmacology and Clinical Metabolic Toxicology -Headache Center and Drug Abuse - Laboratory of Clinical Pharmacology and Pharmacogenomics, AOU of Modena, Modena, Italy |
| Diego B. Swerts | Department of Neurology Irmandade Santa Casa de Misericordia de São Paulo, Department of Neurology, Hospital Israelita Albert Einstein, São Paulo, Brazil | |
| Mario F. P. Peres | University of São Paulo, São Paulo, Brazil | |
| 3.9. Head-to-head comparisons – acute treatments | Valeria Caponnetto | University of L'Aquila, Department of Life, Health and Environmental Sciences, l’Aquila, Italy |
| Patricia Pozo-Rosich | Headache Clinic, Neurology Department, Vall d'Hebron Hospital, and Headache and Neurological Pain Research Group, VHIR, Department of Medicine, Universitat Autònoma de Barcelona, Barcelona, Spain | |
| Cherubino Di Lorenzo | Department of Medico-Surgical Sciences and Biotechnologies “Sapienza” University of Rome Polo Pontino, Rome, Italy | |
| Raffaele Ornello, Simona Sacco | Department of Biotechnological and Applied Clinical Sciences, University of L'Aquila, L'Aquila, Italy | |
| 4.1. Antidepressants | Gloria Vaghi, Roberto De Icco | Department of Brain and Behavioral Sciences, University of Pavia and Headache Science & Neurorehabilitation Center, IRCCS Mondino Foundation, Pavia, Italy |
| Alejandro Labastida-Ramírez | Headache Group, Wolfson Sensory Pain and Regeneration Centre, King's College London, London, UK | |
| Jan Hoffmann | Wolfson Sensory, Pain and Regeneration Centre, Institute of Psychiatry, Psychology & Neuroscience, King's College London, London, UK | |
| Grazia Sances | Headache Science & Neurorehabilitation Center, IRCCS Mondino Foundation, Pavia, Italy | |
| 4.2. Anti-seizure medications | Luigi Francesco Iannone | Section of Clinical Pharmacology and Oncology, Department of Health Sciences, University of Florence, Florence, Italy |
| Todd J. Schwedt | Mayo Clinic, Department of Neurology, Phoenix, AZ, USA | |
| Chia-Chun Chiang | Department of Neurology, Mayo Clinic, Rochester, MN, USA | |
| Alberto Chiarugi | Section of Clinical Pharmacology and Oncology, Department of Health Sciences, University of Florence, Florence, Italy | |
| 4.3. Beta-blockers | Vincenzo Di Stefano, Antonino Lupica, Filippo Brighina | Department of Biomedicine, Neuroscience, and Advanced Diagnostic (BIND), University of Palermo, Palermo, Italy |
| Amr Hassan | Kasr Al Ainy Hospitals, Faculty of Medicine,Cairo University, Cairo, Egypt | |
| Mona Hussein Tawfik | Beni-Suef University, Beni-Suef, Egypt | |
| 4.4 Calcium-channel blockers and blood-pressure lowering agents | Marina Romozzi | Dipartimento di Neuroscienze, Università Cattolica del Sacro Cuore, Rome, Italy |
| Francesco Casillo, Gianluca Coppola | Department of Medico-Surgical Sciences and Biotechnologies, Sapienza University of Rome Polo Pontino ICOT, Latina (Italy), Rome, Italy | |
| Paolo Calabresi | Dipartimento di Neuroscienze, Organi di Senso e Torace, Fondazione Policlinico Universitario Agostino Gemelli IRCCS, and Dipartimento di Neuroscienze, Università Cattolica del Sacro Cuore, Rome, Italy | |
| 4.5 Botulinum toxin | Silvia Quattrocchi, Valentina Favoni, Sabina Cevoli | IRCCS Istituto Delle Scienze Neurologiche di Bologna, Programma Cefalee e Algie Facciali, Bologna, Italy |
| Lavindren Luke Panneerchelvam, Fayyaz Ahmed | Hull University Teaching Hospitals NHS Trust, Hull, UK | |
| 4.6 Gepants for migraine prevention | Simona Guerzoni, Carlo Baraldi | Digital and Predictive Medicine, Pharmacology and Clinical Metabolic Toxicology -Headache Center and Drug Abuse - Laboratory of Clinical Pharmacology and Pharmacogenomics, AOU of Modena, Modena, Italy |
| Sait Ashina | Department of Neurology and Department of Anesthesia, Critical Care and Pain Medicine, Harvard Medical School, Beth Israel Deaconess Medical Center, Boston, MA, USA and Department of Clinical Medicine, Faculty of Health Sciences, University of Copenhagen, Copenhagen, Denmark | |
| Rune Häckert Christensen | Department of Neurology, Danish Headache Center, Copenhagen University Hospital - Rigshospitalet, Copenhagen, Denmark; Department of Clinical Medicine, Faculty of Health and Medical Sciences, University of Copenhagen, Copenhagen, Denmark; Harvard Medical School, Boston, MA, USA; Department of Anesthesia, Critical Care and Pain Medicine, Beth Israel Deaconess Medical Center, Boston, MA, USA | |
| 4.7 Monoclonal antibodies targeting the CGRP pathway | Luigi Francesco Iannone | Section of Clinical Pharmacology and Oncology, Department of Health Sciences, University of Florence, Florence, Italy |
| Shuli Cheng | Department of Neurology, Alfred Health, Melbourne, Australia | |
| Bronwyn Jenkins | Department of Neurology, Royal North Shore Hospital, Sydney, Australia | |
| Pierangelo Geppetti | Section of Clinical Pharmacology and Oncology, Department of Health Sciences, University of Florence and Headache Department of Molecular Pathobiology and Pain Research Center, College of Dentistry, New York University, NY, USA | |
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Raffaele Ornello: Consulting fees, Allergan-AbbVie, Eli Lilly. Honoraria: Eli Lilly, Novartis, Pfizer, Teva. Support for attending meetings, Allergan-AbbVie, Eli Lilly, Lundbeck, Novartis, Pfizer, Teva. Participating, Allergan-AbbVie, Eli Lilly. Leadership, Jr. Editorial Board member J Headache Pain. Receipt of equipment, Novartis, Eli Lilly. Other financial or non-financial, Eli Lilly, Novartis, Teva.
Valeria Caponnetto: Honoraria, Teva. Support for attending meetings, Teva. Honoraria, AbbVie. Honoraria for lecture.
Fayyaz Ahmed: Honorarium to be on the ad board and for lecturing from Abbvie, Pfizer, Dr Reddy's laboratory, TEVA, Eli Lilly, Lundbeck, Electrocore, Eneura, Novartis. Quality improvement grant from Pfizer. Treasurer of the International Headache Society, chair of the advisory board of headache section of the Association of British Neurologists, treasurer of the Anglo-Dutch Migraine Association, honorary advisor of the British Association for the Study of Headache.
Haidar M. Al-Khazali: Personal fees from Pfizer and Lundbeck, outside of the submitted work.
Anna Ambrosini: Speaker honoraria by Novartis, Teva, Eli-Lilly, Pfizer. Congress attendance supported by Teva. Secretary of the Italian Headache Society.
Sait Ashina: Sait Ashina: Consulting: Allergan/AbbVie, Eli Lilly, Impel NeuroPharma, Linpharma, Lundbeck, Pfizer, Satsuma, Teva, Theranica. Honoraria, Lectures presentations: AbbVie, Eli Lilly, Teva, Pfizer, Lundbeck. Leadership: Associate Editor for Cephalalgia, BMC Neurology, Frontiers in Neurology, Headache and Pain Research, Neurology Reviews, and Trustee of the International Headache Society Board.
Carlo Baraldi: Honoraria, AbbVie. Support for meeting attendance, AbbVie, Lilly, Pfizer, Lundbeck.
Filippo Brighina: Honoraria for lectures, presentations from: Pfizer, Lilly, TEVA, Alnylam, AbbVie, Lundbeck, Alexion. Support for attending meetings, TEVA, Alnylam, Roche, Sanofi, Biogen, AbbVie, Alexion, Lundbeck, Lilly.
Paolo Calabresi: Speaker honoraria from: AbbVie, Bayer Schering, Bial, Biogen-Dompè, Biogen-Idec, Eisai, Genzyme, Lundbeck, Lusofarmaco, Merck- Serono, Novartis, Prexton, Teva, UCB Pharma, Zambon. PC received support to attend national and international conferences from: AbbVie, Bayer Schering, Bial, Biogen-Dompè, Biogen-Idec, Eisai, Genzyme, Lundbeck, Lusofarmaco, Merck-Serono, Novartis, Prexton, Teva, UCB Pharma, Zambon. Participation on the Advisory Board for Lilly. Participation in Data Safety, Roche, Therapy for Parkinson's Disease. PC is Past President of the Società Italiana per lo Studio delle Cefalee (SISC).
Sabina Cevoli: Honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events for Angelini, Teva, Novartis, AbbVie, Lundbeck, Pfizer. Support for attending meetings and/or travel by AbbVie, Lilly. Participation on the Advisory Board for Lilly. Advisor of ANIRCEF. PI for trial of Lilly, Novartis, Teva, Lundbeck.
Shuli Cheng: Headache Australia website medical reviewer. AbbVie to attend 2022 Australia New Zealand Neurologist Association ASM. Co-secretary of the Australian New Zealand Headache Society- current.
Chia-Chun Chiang: Research support from the American Heart Association with funds paid to her institution. (Unrelated to this manuscript). Consulting, Satsuma, eNeura. Honoraria, HMP Global for migraine education lectures. Leadership, American Migraine Foundation Editorial Board.
Alberto Chiarugi: Filed patents on novel uses of anti-CGRP biologics and gepants.
Rune Häckert Christensen: iHEAD support from IHS. Travel support from the Augustinus Foundation.
Min Kyung Chu: Honoraria, Allergan Korea, Handok-Teva, Eli Lilly and Company, and Yuyu Pharmaceutical Company.
Gianluca Coppola: Consulting fees, Pfizer, AbbVie. Honoraria, Pfizer, Lilly. Meeting support, TEVA, Lilly, Pfizer. Safety boards, Pfizer, AbbVie. He has received funding for clinical trials from Pfizer, AbbVie, Novartis, and Teva. Leadership: Trustee and co-chair of the membership committee for the International Headache Society, member of the board at large of the European Headache Federation, Member of the study center of the Italian Society for the Study of Headaches, member of the Special Interest Group on Brainstem of the International Federation of Clinical Neurophysiology. Associate Editor for Cephalalgia, Cephalalgia Reports, The Journal of Headache and Pain, BMC Neurology (Pain section), Frontiers in Neurology (Neurotechnology section), and Frontiers in Human Neuroscience (Brain Imaging and Stimulation section).
Santiago Crema: Funding for travel, accommodation and congress registration provided by Pfizer.
Roberto De Icco: Honoraria from Eli-Lilly; TEVA; Lundbeck; Pfizer; AbbVie. Funding support for meeting attendance, TEVA; Lundbeck. Advisory boards, Pfizer; AbbVie. Junior Editor for Cephalalgia.
Marina de Tommaso: Honoraria, presentations at national conferences supported by TEVA, Lundbeck, Pfizer, Lilly. Support from TEVA for EHF last meeting in Barcelona. Leadership, President-Elect Italian Society for Headache Study (SISC).
Cherubino Di Lorenzo: Consulting, Pfizer. Honoraria, Pfizer, Eli Lilly.
Vincenzo Di Stefano: Support for travels for attending meetings from Alexion, Alnylam, Argenx. Vincenzo Di Stefano is member of Italian Society of Neurology (SIN), Italian Society for the study of headache (SISC), Italian Society of Peripheral Nervous System (ASNP), Italian Society of Clinical Neurophysiology (SINC). Vincenzo Di Stefano received compensation for speaking from Alexion, and Alnylam; he is Sub-Investigator in clinical trials for Alexion, Alnylam, Argenx, Dianthus, and Sanofi.
Hans-Christoph Diener: Grants from German Research Council. Honoraria, Lundbeck, Novartis, Teva. Leadership, Clinical Trials Committee of the IHS NorHead Norway.
Esme Ekizoglu: Received honoraria for the preparation of educational materials from Allergan-AbbVie.
Valentina Favoni: Consulting fees from AbbVie, Eli-Lilly and Teva. Honoraria from Eli-Lilly, Lundbeck, Pfizer and Teva. Support for attending meetings from AbbVie, Lundbeck and Teva.
Pierangelo Geppetti: European Research Council - Advanced grant; European research Council - Proof of Concept grant. Royalties for books of the series Handbook of Experimental Pharmacology, Springer. Compensations from Abbott; AbbVie; Allergan; Eli Lilly; Lundbeck; Pfizer; TEVA; Novartis. Recipient of 3 Patents owned by FloNext Srl. Founder and shareholder of the Academic Spinoff (University of Florence) FloNext Srl. Editor in Chief, Confinia Cephalalgica; Editor in Chief, Advancements in Health Research.
Maria Teresa Goicochea: Independent Medical Education Grant from Pfizer. Speaker or advisory board for: Teva, AbbVie, Pfizer. Support for attending meetings AbbVie, Pfizer.
Antonio Granato: Lectures supported by Lilly, TEVA, Novartis, AbbVie. Educational meeting supported by Lilly, TEVA, AbbVie.
Franco Granella: Honoraria from Lundbeck and Pfizer. Leadership, President of the Italian Society for the Study of Headaches (SISC).
Simona Guerzoni: Consulting fees, AbbVie, Lilly, Angelini, Pfizer, Lundbeck. Honoraria, AbbVie, Lilly, Angelini, Pfizer, Lundbeck. Meeting attendance, AbbVie, Lilly, Pfizer, Lundbeck. Advisory boards, AbbVie, Lilly, Angelini, Pfizer, Lundbeck,
Woo-Seok Ha: Lecture on online education program for nursing patients with epilepsy in request of Korean Nurse association (2024-01-17).
Amr Hassan: Consulting fees, Novartis, Sanofi Genzyme, Biologix, Merck, Hikma Pharma, Janssen, Inspire Pharma, Future Pharma, Elixir pharma. Honoraria for lectures/ presentations: Novartis, Allergan, Merck, Biologix, Janssen, Roche, Sanofi Genzyme, Bayer, Hikma Pharma, Al Andalus, Chemipharm, Lundbeck, Inspire Pharma, Future Pharma and Habib Scientific Office, and Everpharma. Support for attending meetings, congresses, symposia and/or travel: Novartis, Allergan, Merck, Biologix, Roche, Sanofi Genzyme, Bayer, Hikma Pharma, Chemipharm, and Al Andalus and Clavita pharm. Advisory boards, Clavita Pharm Advisory board: Novartis, Sanofi Genzyme, Biologix, Merck, Hikma Pharma, Janssen, Inspire Pharma, Future Pharma, Elixir pharma. Leadership, Member of Education committee of IHS Member of Membership committee of IHS Member of regional societies committee of IHS Vice President of MENAA headache society Secretary General of headache chapter of Egyptian society of Neurology.
Koichi Hirata: Consulting, Otsuka Pharmaceutical Co., Ltd Honoraria, Amgen Astellas BioPharma K.K., Daiichi Sankyo Company, Limited, Eisai Co., Ltd, Eli Lilly Japan K.K., MSD Co., Ltd, Otsuka Pharmaceutical Co., Ltd, Pfizer Japan Inc. Support for meeting attendance, Amgen Astellas BioPharma K.K., Daiichi Sankyo Company, Limited, Eisai Co., Ltd, Eli Lilly Japan K.K., MSD Co., Ltd, Otsuka Pharmaceutical Co., Ltd, Pfizer Japan Inc. Advisory boards, Amgen Astellas BioPharma K.K., Eli Lilly Japan K.K., MSD Co., Ltd, Otsuka Pharmaceutical Co., Ltd Leadership, Japan Patient Advocacy Coalition.
Jan Hoffmann: At the time of drafting and submitting this manuscript he was employed as an academic researcher and clinician at King's College London, UK. Since May 2024 he is a full-time employee of H. Lundbeck A/S. Since his employment with H. Lundbeck A/S he has approved the manuscript but made no changes to its content. In the 36 months preceding his employment at H. Lundbeck A/S, he held research grants from the following entities (unrelated to this manuscript): Bristol Myers Squibb, Migraine Trust, International Headache Society, National Institute for Health and Care Research (NIHR), Medical Research Council (MRC). Consulting and/or advisory boards for AbbVie, Cannovex, Chordate Medical, Eli Lilly, H. Lundbeck A/S, Sanofi, Teva. Lectures/speaking/educational events: Chordate Medical, MD-Horizonte, H. Lundbeck A/S, Pfizer, Teva. Manuscript writing/associate editor work: NEJM Journal Watch, Oxford University Press, Quintessence Publishing, Sage Publishing, Springer Healthcare. Data Monitoring Committee: Chordate Medical. Advisory Board: AbbVie, Chordate Medical, Eli Lilly, H. Lundbeck A/S, Teva. Past Member of the Board of Trustees of the International Headache Society. Past Council Member and Treasurer of the British Association for the Study of Headache. Past Associate Editor for Cephalalgia, Cephalalgia Reports, Journal of Oral & Facial Pain and Headache. Associate Editor for Journal of Headache and Pain, Frontiers in Pain Research. Stock options for Chordate Medical.
Luigi Francesco Iannone: IHS junior grant. Consulting fees, Eli-Lilly, AbbVie. Honoraria, Eli-Lilly, AbbVie, Pfizer, TEVA, ORGANON. Meeting attendance support, TEVA, Lundbeck, Eli-Lilly.
Bronwyn Jenkins : Honoraria, Allergan/AbbVie, Eli Lilly, GPCE, HealthEd, Lundbeck, Pfizer, Teva. Leadership, Immediate Past President of the Australian and New Zealand Headache Society.
Alejandro Labastida-Ramírez: Grants, ALR was supported by a fellowship from the Migraine Trust.
Morris Levin: Royalties, Oxford University Press $600. Advisory boards $6000. Expert testimony, $5000. Board of Directors Headache Cooperative of the Pacific Unpaid.
Edoardo Mampreso: Honoraria, AbbVie, Elli-Lilly, TEVA, Pfizer, Lundbeck. Meeting attendance support, AbbVie, Elli-Lilly, TEVA, Pfizer, Lundbeck.
Daniele Martinelli: LSC LifeSciences Consultants. Honoraria, AbbVie, Lundbeck.
Teshamae S. Monteith: Grants from AbbVie, Amgen Site PI: Rehaler, AbbVie, Ipsen, Eli Lilly. Consulting fees, Pfizer, AbbVie, Merz. Honoraria, Medscape, American Headache Society, American Academy of Neurology. Novartis, Massachusetts Medical Society. Advisory boards, Teva, Pfizer, AbbVie, eNeura, LinPharma. Leadership: IHS Board of Trustee, Florida Society of Neurology, American Headache Society, American Academy of Neurology, American Neurological Association, Editorial Boards: Cephalalgia, Neurology, Brain and Life Magazine, American Migraine Foundation, Continuum Audio.
Aynur Özge: Consulting fees, AbbVie, Drogsan, ABdi İbrahim. Honoraria, AbbVie, Drogsan, ABdi İbrahim. President of Global Migraine and Pain Society, President of Mersin Alzheimer Society, Board member of IHS.
Mario F. P. Peres: Consulting fees, AbbVie, Pfizer, Lundbeck, Eurofarma, Libbs, Teva, Lilly. Honoraria, AbbVie, Pfizer, Lundbeck, Eurofarma, Libbs, Teva, Lilly. Patents, BR 10 2020 020706–7 US 11,826,177 B2. Advisory boards, AbbVie, Pfizer, Lundbeck, Eurofarma, Teva. Leadership, IHS, ABRACES.
Marcio Nattan Portes Souza: Honoraria, Libbs, TEVA, Pfizer, Allergan/AbbVie, Lundbeck. Funding for traveling by TEVA. Advisory boards, Pfizer, TEVA, Libbs.
Patricia Pozo-Rosich: Her research group has received research grants from AbbVie, Novartis and Teva; as well as, Instituto Salud Carlos III, EraNet Neuron, European Regional Development Fund (001-P-001682) under the framework of the FEDER Operative Programme for Catalunya 2014–2020 - RIS3CAT; has received funding for clinical trials from AbbVie, Amgen, Biohaven, Eli Lilly, Novartis, Teva. Received, in the last 36 months, honoraria as a consultant for: AbbVie, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer and Teva. Received, in the last three years, honoraria as a speaker for: AbbVie, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva. Norwegian Clinical Trial driven by the investigator. She is the Honorary Secretary of the International Headache Society. She is on the editorial board of Revista de Neurologia. She is an associate editor for Cephalalgia, Headache, Neurologia, The Journal of Headache and Pain and Frontiers of Neurology. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society.
Maria Pia Prudenzano: Consulting fees, Allergan, Pfizer, Lilly, Lundbeck, Novartis, Teva. Honoraria, AbbVie, Allergan, Eli-Lilly, Idorsia, Italfarmaco, Lundbeck, Pfizer, Teva, Valeas. Meeting attendance support, Idorsia, Lilly, Lundbeck, Teva. Advisory boards, Allergan, Lilly, Lundbeck, Pfizer, Teva. Leadership, Italian Society for the Study of Headache (SISC), Italian Society of Neurology,
Innocenzo Rainero: Honoraria, Pfizer, Eli Lilly Italy, Teva. Support for meeting attendance, AbbVie. Advisory board, Pfizer, Volodymyr Romanenko: Ukrainian Headache Research Society, President.
Antonio Russo: Honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events (in the past 36 months) from Ely-Lilly, AbbVie, Teva Pharmaceuticals, Pfizer. Support for attending meetings and/or travel (past 36 months) from Ely-Lilly, AbbVie, Teva Pharmaceuticals, Pfizer Lundbeck. Participation on a Data Safety Monitoring Board or Advisory Board (past 36 months) from Ely-Lilly, AbbVie, Teva Pharmaceuticals, Pfizer. Specialty Chief Editor for Headache and Neurogenic Pain Frontiers in Neurology. Member of executive board of the Italian Headache Society (S.I.S.C.).
Grazia Sances: Honoraria for presentations, speakers bureaus or educational events from Novartis, Eli Lilly, Teva, Lundbeck, Pfizer. Participation on Advisory Board for Novartis, Eli Lilly, Teva, Lundbeck, Pfizer. Support for attending meetings and/or travel by Novartis, Eli Lilly, Teva, Lundbeck. Member of executive board of the Italian Headache Society (S.I.S.C.).
Paola Sarchielli: Presentation and moderations for Eli-Lilly and Lundbeck. Annual congress of SISC and for Florence Symposium on CGRP (2022, 2023). Leadership, Didactic Leader for High School of Headache of SISC Italian society.
Todd J. Schwedt: In the prior 24 months, TJS has received personal compensation for consulting with AbbVie, Amgen, Eli Lilly, Linpharma, Lundbeck, Satsuma, Scilex, and Theranica, royalties from UpToDate, and has held stock options in Aural Analytics and Nocira. Research grants have been received from American Heart Association, Henry Jackson Foundation, National Headache Foundation, National Institutes of Health, Patient Centered Outcomes Research Institute, Pfizer, Spark Neuro, and United States Department of Defense.
Marcello Silvestro: Speaker honoraria from Novartis, Pfizer, Allergan, Teva and Lilly.
Alessandro Tessitore: I have received honoraria for lectures and speakers bureaus from AbbVie, Everpharma, Bial, Lusofarmaco, Zambon. Expert testimony, I have received support from Bial, AbbVie, Zambon. I serve as associate Editor for the European Journal of Neurology.
Mansoureh Togha: I received support from the European Headache Federation for accommodation and travel tickets to attend EHC 2022 as a scientific committee member. I am the president of the Iranian Headache Association.
Gloria Vaghi: Honoraria for educational event from Lundbeck.
Shuu-Jiun Wang: For this manuscript received research grants from the National Science and Technology Council of Taiwan, Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, Taipei Veterans General Hospital, Taiwan Headache Society and Taiwan branches of Eli Lilly, Novartis and Orient Europharma. Received honoraria as a moderator from AbbVie, Biogen, Eli-Lilly, Hava Biopharma, and Pfizer . Received consulting fees from AbbVie, Eli-Lilly Taiwan, Percept Co., and Pfizer Taiwan, and has been the PI in trials sponsored by Eli-Lilly, Lundbeck, and Novartis.
Messoud Ashina: For this manuscript, Institutional grants from Lundbeck Foundation, Novo Nordisk Foundation, Lundbeck, and Novartis. In the past 24 months, MA has received personal compensation for consulting with AbbVie, AstraZeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals. Associate Editor of The Journal of Headache and Pain, Associate Editor of Brain.
Cristina Tassorelli: Institutional fees for conducting clinical trials for AbbVie, Eli Lilly, Ipsen, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals. Consulting fees from AbbVie, Dompé, Eli Lilly, Ipsen, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals. Associate Editor Cephalalgia. Past-President of IHS.
Simona Sacco: For this manuscript, Novartis, Uriach. Royalties, Abbott, Allergan-AbbVie, AstraZeneca, Boheringer, Eli Lilly, Lundbeck, Novartis, NovoNordisk, Pfizer, Teva. Consulting fees, Abbott, Allergan-AbbVie, AstraZeneca, Boheringer, Eli Lilly, Lundbeck, MedScape, Novartis, NovoNordisk, Pfizer, Teva. Expert testimony, Abbott, Allergan-AbbVie, AstraZeneca, Boheringer, Eli Lilly, Lundbeck, Novartis, NovoNordisk, Pfizer, Teva, Bayer, Medtronic, Starmed, Bristol-Myers-Squibb, Daiichi-Sankyo. Patents, Allergan-AbbVie, AstraZeneca, Eli Lilly, Lundbeck, Novartis, Pfizer, Teva. Leadership, President-elect European Stroke Organization, Editor-in-Chief Cephalalgia and Cephalalgia Reports, assistant editor for Stroke.
Alessia Bellotti, Francesco Casillo, Ilenia Corbelli, Adriana Fallacara, Kimberly N. Garces, Eva-Maria Huessler, Mona Hussein, Anna Laporta, Antonino Lupica, Ilaria Orologio, Li-Ling Hope Pan, Lavindren Luke Panneerchelvam, Silvia Quattrocchi, Marina Romozzi, Diego Belandrino Swerts. No conflict of interest to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
