Abstract
Background
Non-invasive vagus nerve stimulation (nVNS) is a proven treatment for cluster headache and migraine. Several possible mechanisms of action by which nVNS mitigates headache have been identified.
Methods
We conducted a narrative review of recent scientific and clinical research into nVNS for headache, including findings from mechanistic studies and their possible relationships to the clinical effects of nVNS.
Results
Findings from animal and human studies have provided possible mechanistic explanations for nVNS efficacy in headache involving four core areas: Autonomic nervous system functions; cortical spreading depression inhibition; neurotransmitter regulation; and nociceptive modulation. We discuss how overlap and interplay among these areas may underlie the utility of nVNS in the context of clinical evidence supporting its safety and efficacy as acute and preventive therapy for both cluster headache and migraine. Possible future nVNS applications are also discussed.
Conclusion
Significant progress over the past several years has yielded valuable mechanistic and clinical evidence that, combined with the excellent safety and tolerability profile of nVNS, suggests that it should be considered a first-line treatment for both acute and preventive treatment of cluster headache, an effective option for acute treatment of migraine, and a highly relevant, practical option for migraine prevention.
Introduction
The mechanisms of action for vagus nerve stimulation (VNS) have been investigated for several years. Potential peripheral and central mechanisms involved in VNS-induced anti-nociception were outlined by Yuan and Silberstein in their four-part comprehensive review published in 2016 and 2017 (1–4). At that time, evidence that non-invasive vagus nerve stimulation (nVNS) was effective in treating headache disorders came mainly from case series, open-label pilot studies, and some preliminary reports from small double-blind trials (3,4). Since then, nVNS has been established clinically as a safe and effective treatment option for migraine and cluster headache in addition to some of the other rarer trigeminal autonomic cephalalgias (TACs). Several possible mechanisms of action for VNS in headache treatment have been identified, some of which have been validated in human studies. This review provides an update on scientific and clinical research supporting the use of nVNS for treatment of headache disorders. We describe findings from mechanistic studies and their possible relationships to the clinical effects of nVNS therapy. We also explain and contextualize findings from clinical studies of nVNS for headache and describe ongoing efforts and future directions for mechanistic research and clinical indications for nVNS.
Mechanistic evidence
The vagus nerve modulates multiple systems within the brain and body. The most compelling mechanistic explanations for the efficacy of nVNS in headache disorders involve four core areas: Effects on autonomic nervous system functions; inhibition of cortical spreading depression (CSD); neurotransmitter regulation; and nociceptive modulation. Overlap and interplay among these areas contribute to the complex mechanisms underpinning the utility of nVNS in treating headache (Figure 1). Structures and pathways relevant to the pathophysiology of cluster headache and migraine are detailed in Figures 2 and 3, respectively (5,6).
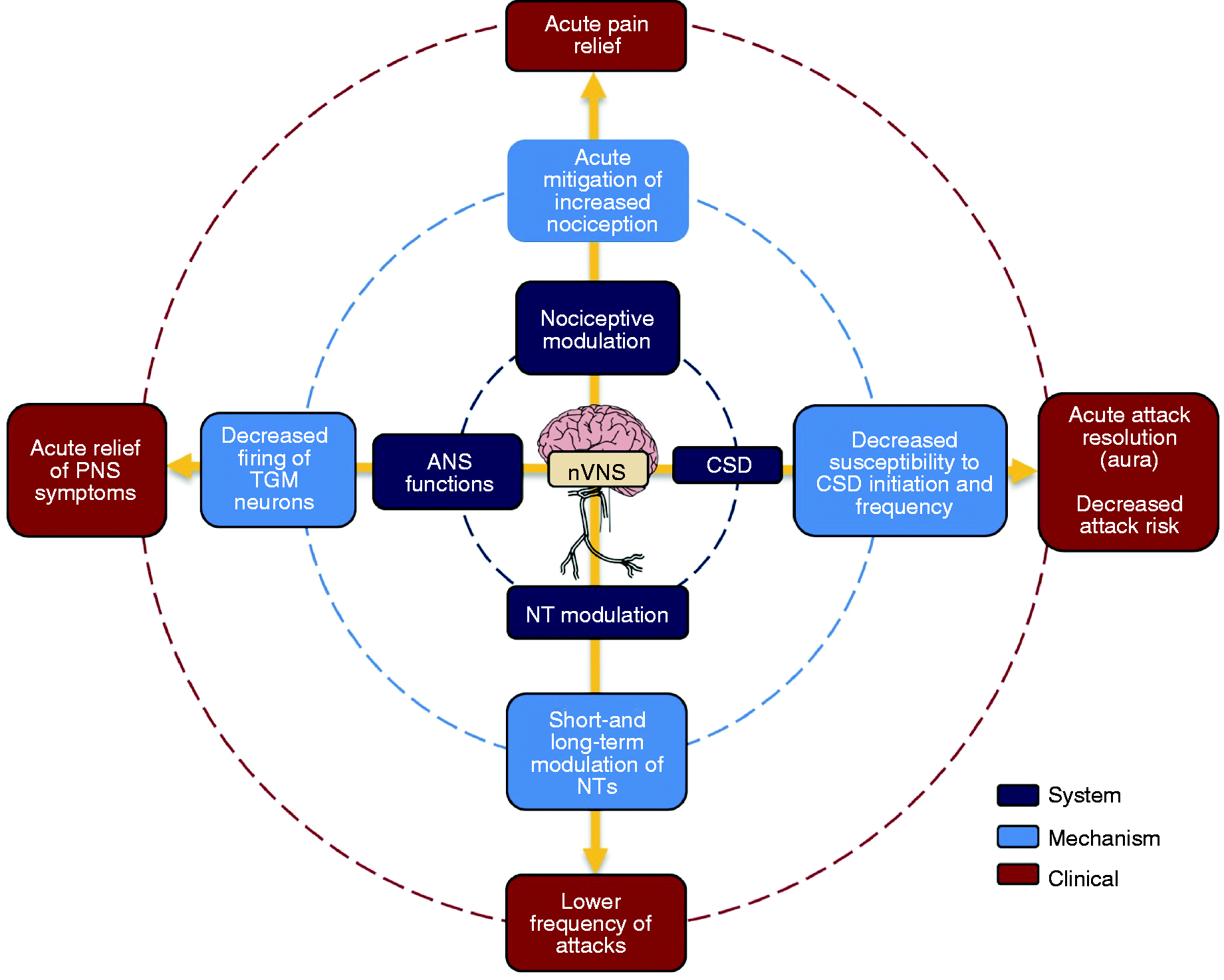
Involved systems, mechanisms of action, and clinical effects of nVNS in headache.

Pathophysiology of the TACs, with most of the data obtained from cluster headache (5). Several brain systems seem to be important in cluster headache, and neuromodulation of each of these systems can prevent cluster headache as shown. The system responsible for pain is the trigeminal system (red), including the TCC and TG. The cranial autonomic system (green) includes the superior SSN and SPG and connects to the trigeminal system via the trigeminal autonomic reflex. The HYP system (blue) connects to the autonomic and trigeminal systems. Recently, the vagus nerve and NTS system (orange) have been shown in neuromodulation studies to be involved in pain regulation, with connections to the trigeminal system, the autonomic system, and the HYP.

Structures involved in migraine pathophysiology (6).
Effects on autonomic nervous system functions
The trigeminal autonomic reflex is a pathway comprising a central brainstem connection between the trigeminal nerve and parasympathetic outflow from the facial cranial nerve via the superior salivatory nucleus (SSN) and the sphenopalatine ganglion (SPG) (7). The SSN innervates the submandibular and sublingual glands, and the SPG innervates the lacrimal glands and nasal mucosa. Postganglionic parasympathetic neurons contain nitric oxide synthase, vasoactive intestinal peptide, and pituitary adenylate cyclase–activating polypeptide (8–10). The trigeminal autonomic reflex plays a pivotal role in the pathophysiology of cluster headache (5,11) (Figure 2). Animal and human studies have demonstrated that direct and indirect connections between the trigeminal and vagus nervous systems in the brainstem are involved in head pain pathways, and that nVNS might inhibit firing of trigeminocervical neurons via such connections (12,13). In humans, nVNS has been shown to have a bilateral inhibitory effect on parasympathetic function (i.e. kinetic oscillation-evoked lacrimation [tearing]) within the trigeminal autonomic reflex (14).
Structural magnetic resonance imaging (MRI) study results are suggestive of the human trigeminal and vagus systems interconnecting anatomically. The existence of a trigeminovagal complex has been reported as a basis for the connections between the trigeminal and vagus systems (15). Recent functional MRI findings in humans suggest that nVNS may modulate the trigeminal autonomic reflex via a complex network that includes the hypothalamus, the spinal trigeminal nuclei, the left pontine nucleus, and the parahippocampal gyrus (16).
Cortical spreading depression inhibition
CSD is believed to be one of the major electrophysiological mechanisms underlying migraine aura (17,18). In rats, VNS inhibits susceptibility to CSD with a magnitude that is similar to that of migraine prophylactic drugs (e.g. topiramate, valproate, propranolol, amitriptyline) (17,19,20). In one rat study, bilateral CSD suppression developed within 30 minutes after unilateral VNS (20). Additional animal studies demonstrated that the effect of nVNS on CSD can last between 9 and 24 hours (21).
nVNS also affects CSD initiation and propagation. In rat models, nVNS led to a greater than two-fold increase in the electrical stimulation threshold necessary to evoke a CSD and was associated with decreases in both the frequency and the propagation speed of CSD (20). VNS inhibits CSD centrally as pharmacologic blockade of the nucleus tractus solitarius (the main relay for vagal afferents) by lidocaine or the glutamate receptor antagonist cyanquixaline prevented CSD suppression by nVNS (22). Suppression of CSD by nVNS was also inhibited by the depletion of norepinephrinergic or serotonergic neurotransmission using specific neurotoxins; such findings are suggestive of a role for both norepinephrine and serotonin in CSD inhibition by nVNS (22).
In a rat model of ischemic stroke, nVNS attenuated matrix metalloprotease-mediated damage to the blood-brain barrier, but it was unclear whether these effects resulted from CSD prevention and how they might be relevant to migraine (23,24). Studies investigating the capability of VNS to suppress the increased glutamate release associated with CSD would also be instructive. Once released from the depolarized tissue, glutamate diffuses into adjacent tissues and triggers the same depolarization cycle, thereby allowing contiguous propagation of CSD (20).
Neurotransmitter regulation
In a rat model of recurrent headache, repeated inflammatory dural stimulation by an inflammatory soup induced an increased response to glyceryl trinitrate associated with a chronic state of trigeminal hypersensitivity and significantly elevated levels of extracellular glutamate in the trigeminal nucleus caudalis (TNC) (25). This could be a possible mechanism for the transition of episodic to chronic headaches in humans. nVNS suppresses the increases in extracellular glutamate levels in the TNC that occur in response to glyceryl trinitrate infusion (26). This suggests that nVNS may relieve acute headache pain by inhibiting pain pathways that increase TNC activity (27). Recent rodent model study results have also suggested that inhibition of trigeminal nociception by nVNS may be mediated, in part, by activation of serotonergic receptors on inhibitory neurons in the spinal trigeminal nucleus, thus leading to release of γ-aminobutyric acid (GABA) and activation of GABAA receptors (28).
Together, glutamate and GABA comprise the major amino acid systems in the brain, and derangement of these systems has been implicated in migraine pathogenesis (29,30). Whereas glutamate promotes pain transmission, GABA inhibits glutamate-elicited neuronal activity in the trigeminocervical complex (30). Peripheral nociceptive hyperexcitability (e.g. increased release of calcitonin gene-related peptide and glutamate) together with microglial activation and loss of local inhibitory control from GABA likely contribute to the pain sensitization process. The ability of nVNS to inhibit glutamate release (26), suppress microglial activation (31), and enhance GABA release (28) provides a mechanistic rationale for how it can fundamentally restore the dysfunctional trigeminal system.
In rats, VNS induces release of norepinephrine and serotonin in the locus coeruleus and dorsal raphe nucleus, respectively (20). Both norepinephrine and serotonin have been linked to the pathophysiology of migraine (32,33).
Nociceptive modulation
Nociceptive activation of trigeminocervical neurons has been implicated in the mediation of migraine and cluster headache pain (34). In a rat model of episodic migraine, trigeminal nociceptive activation was triggered by injection of an inflammatory agent into the trapezius muscle, followed by exposure of the animals to pungent volatile compounds from California bay leaf oil extract. nVNS inhibited nocifensive responses (i.e. responses to pain or discomfort) to mechanical stimulation of sensitized trigeminal neurons and led to decreased expression of proteins associated with peripheral and central sensitization (35). In rat models of migraine-like (acute dural-intracranial) and cluster-like (trigeminal-autonomic) head pain, VNS suppresses acute nociceptive activation of trigeminocervical neurons (34) (Figure 4). VNS also inhibits tonic firing of trigeminocervical neurons (in addition to acting on evoked response), which may be relevant to prevention of migraine and cluster headache (34). Inhibition of the background activity level in these neurons may decrease the likelihood that they reach the activation threshold necessary to trigger attacks. In two rodent models of episodic migraine, nVNS was shown to be as effective as sumatriptan at inhibiting trigeminal nociception (28). Together, these findings suggest that modulation of central trigeminovascular neurons by VNS may contribute to its clinical benefits in the acute treatment of migraine and cluster headache (28,34).

Effect of two doses of iVNS on dural-evoked trigeminocervical neuronal responses ((a) and (b)) and SuS stimulation‒evoked trigeminocervical neuronal response (c) (34). Histograms of the time course changes in the average number of action potential spikes per sweep (mean ± SEM) of grouped data of intracranial dural-evoked multi-unlit trigeminocervical neuronal clusters that include inputs in the Aδ-fiber range (3–20 ms) and those whose latencies were extended and therefore also received inputs that include both Aδ- and C-fibers (“fast” neuronal responses (a)) and intracranial dural-evoked unitary discharges within the C-fiber latency range (“slow” neuronal responses (b)). Time course changes in the average number of action potential spikes per sweep of SuS stimulation-evoked longer-latency (>5 ms) multi-unit trigeminocervical neuron clusters (c). Ipsilateral iVNS × 2 inhibited SuS stimulation-evoked longer-latency neuronal responses in the TCC that only returned to baseline after 3 hours. Data have been normalized to represent the percentage change from baseline and are expressed as mean ± SEM.
Clinical results update
nVNS is Conformité Européen (CE) marked in the European Union for acute and/or prophylactic treatment of primary headache (migraine, cluster headache, and related TACs) and medication overuse headache (36). In the US, the Food and Drug Administration (FDA) cleared nVNS for acute treatment of migraine and episodic cluster headache as well as for the prevention of migraine and cluster headache (37).
Cluster headache
The use of nVNS as a first-line treatment for cluster headache is supported by randomized controlled trials and real-world evidence (38–43). Table 1 summarizes results for the primary endpoints of the randomized controlled trials, including therapeutic gain with nVNS for greater clinical context. The efficacy and safety of nVNS as acute treatment for episodic cluster headache were reported in two randomized, double-blind, sham-controlled clinical trials (ACT1, ACT2) (38,39) and a meta-analysis (44). Response rates (i.e. proportions of subjects who achieved a pain intensity score of 0 or 1 on a 5-point scale 15 minutes after treatment initiation with no rescue medication use) for the first treated attack (the ACT1 primary endpoint) were significantly higher with nVNS treatment than with sham treatment in ACT1 (38) and for the pooled ACT1/ACT2 populations (44). Also for patients with episodic cluster headache, the proportions of all treated attacks that achieved pain-free status at 15 minutes (the ACT2 primary endpoint) were significantly higher with nVNS than with sham in ACT2 (39) and for the ACT1 and pooled ACT1/ACT2 populations (44). The efficacy of nVNS for cluster headache prevention was explored in a randomized controlled clinical trial (PREVA) (40) that compared standard of care (SoC) treatment alone with SoC + nVNS. Compared with SoC alone, nVNS + SoC treatment was associated with a significantly greater reduction in the number of attacks per week and significantly higher (≥50%) response rates (Figure 5(a)), and nNVS + SoC treatment led to a significant decrease in abortive medication use (Figure 5(b)).
Summary of key randomized controlled clinical trials of nVNS for cluster headache and migraine.

aDefined as the proportion of subjects who achieved a pain intensity score of 0 or 1 on a 5-point scale at 15 minutes after treatment initiation with no rescue medication use.
bFrom chi-square test or Fisher exact test as appropriate.
cnVNS – control (sham or SoC).
dDefined as the proportion of all treated attacks that achieved pain-free status within 15 minutes after treatment initiation with no rescue medication use.
eFrom the generalized estimating equations model.
fFrom analysis of variance.
gDefined as the proportion of subjects who were pain-free for their first treated migraine attack of the double-blind period.
hFrom Student’s t-test (normal distribution) or Wilcoxon rank sum test (non-normal distribution) as appropriate.
iFrom linear regression adjusted for treatment group, center, presence/absence of aura, and number of migraine days in the run-in period.
jPost hoc analysis of the mITT population (i.e. subjects with ≥67% treatment adherence).
eCH: episodic cluster headache; ITT: intent to treat; mITT: modified intent to treat; nVNS: non-invasive vagus nerve stimulation; SoC: standard of care. Values in bold indicate statistical significance.

Results from PREVA (40). Percentage of subjects with ≥50% response to treatmenta for the ITT population (a); abortive medication use for the mITT population (b).
Real-world evidence provides further support for the effectiveness of nVNS as acute and preventive treatment for cluster headache. In a retrospective analysis of data from 30 patients with cluster headache, nVNS led to significant decreases from baseline in mean attack frequency (64%), duration (43%), and severity (23%) (45). Currently, nVNS is the only FDA-cleared device for any form of cluster headache and the only therapy that is FDA cleared/approved for both acute and preventive therapy in cluster headache. On the basis of this body of clinical evidence, nVNS was recently recommended by the National Institute for Health and Care Excellence as effective and cost-saving for the treatment of cluster headache (46).
Other trigeminal autonomic cephalalgias
The benefit of nVNS in cluster headache treatment led to its investigation in other TACs such as hemicrania continua (HC) and paroxysmal hemicrania (PH). Medical records from nine patients with HC and six with PH who had been treated with nVNS (treatment duration range, 3 months to 5 years) were retrospectively reviewed (47). Seven patients with HC (78%) reported reduced severity of continuous pain, and four patients with PH (67%) reported benefits of nVNS therapy, including attack freedom (n = 1) and reductions in attack frequency (n = 2), severity (n = 3), and/or duration (n = 1) (47). An open-label, prospective clinical audit included four patients with HC. After 3 months of nVNS therapy, two of these patients (50%) reported meaningful improvements, including 73% and 80% reductions in the number of headache exacerbations per month as well as 18% and 58% reductions in pain scores (48).
Migraine
The efficacy of nVNS for acute migraine treatment was assessed in an open-label 6-week pilot study (49). Among the 19 patients who had moderate or severe headache at baseline for their first attack, four (21%) were pain free and nine (47%) reported pain relief 2 hours after nVNS monotherapy. Similar results were seen when all attacks were pooled. These findings led to a large randomized, double-blind, sham-controlled clinical trial (PRESTO) (Table 1) (41). The PRESTO study provided Class I evidence to support the efficacy and safety of nVNS for acute treatment of migraine. nVNS was effective for aborting attacks as early as 30 minutes and up to 60 minutes after treatment and for relieving pain at 120 minutes. Rates of pain freedom 2 hours after treatment appear to be generally similar for nVNS and triptans (50); these pain-free rates for both nNVS and triptans compare favorably with those of ubrogepant (51) and somewhat unfavorably with those of lasmiditan (52). nVNS has the advantage of being very well tolerated and associated with limited adverse events, differentiating it from lasmiditan, which is a Schedule V controlled substance in the US with some potential for abuse, as indicated by the occurrence of certain adverse events that represent signs of abuse potential (e.g. euphoric mood, feeling abnormal, feeling drunk, abnormal dreams, hallucinations) during clinical trials (53,54). Unlike nVNS, lasmiditan is also associated with potentially serious warnings and precautions (e.g. driving impairment, central nervous system depression) (53).
An early pilot study (EVENT) (42) suggested that nVNS may also have benefit as a preventive treatment for chronic migraine (Table 1) with excellent tolerability and safety. These findings led to a larger randomized, double-blind, sham-controlled neuromodulation device study (PREMIUM) (43). Analysis of the primary endpoint in the PREMIUM study did not show a statistically significant benefit of nVNS over sham treatment (Table 1). A post hoc modified intent-to-treat (mITT) analysis of patients who were ≥67% adherent to treatment suggested that nVNS was associated with a significantly greater reduction (vs. sham) in monthly migraine days (difference, −0.74; p = 0.043), monthly headache days (difference, −0.86; p = 0.045), and acute medication days (difference, −0.80; p = 0.039). Therapeutic gains for the reduction in the number of migraine days were more pronounced for subjects with aura (nVNS, −2.83; sham, −1.41; p = 0.061) than for those without aura (nVNS, −2.22; sham, −1.71; p = 0.15).
The robust response seen in the sham group of the PREMIUM study, which was atypical in the context of neuromodulation device studies, raised the concern that the sham device may have stimulated the vagus nerve and provided an active therapeutic effect. This hypothesis was subsequently investigated in a randomized controlled study, which proved that the sham device used in the PREMIUM study significantly reduced autonomic output (i.e. lacrimation) after trigeminal stimulation as a result of active modulation of the trigeminal autonomic reflex. Thus, the stimulator generated a biological effect in the control group (55). Considering that the sham device used in PREMIUM provided vagal activation, the trial could be viewed as a study of two levels of vagal stimulation rather than one with an active treatment arm and a true placebo/sham arm. The number of migraine days decreased from baseline by 29% in the clinical dose nVNS group, which was slightly superior to the 22% decrease seen in the low-dose (sham) stimulation group. These benefits are similar in magnitude to the absolute benefit reported with some established preventive treatments (e.g. erenumab, fremanezumab, topiramate) (56). The efficacy rates from PREMIUM are similar to those reported from the trials of other neuromodulation devices that supported their FDA clearance for migraine prevention: Supraorbital transcutaneous stimulation (STS) (57) and single-pulse transcranial magnetic stimulation (sTMS) (58) (Figure 6). The PREMIUM study results are suggestive of nVNS being a valuable preventive treatment for migraine that provides meaningful benefits to patients.

Additional real-world use of nVNS and additional randomized controlled trials will confirm the efficacy of nVNS in migraine prevention. A second large, randomized, double-blind, sham-controlled trial of nVNS for migraine prevention (PREMIUM II, NCT03716505) (59) is currently nearing completion. It addresses several limitations of the original PREMIUM study. In the original study, bilateral stimulations were used, but subsequent research suggested that unilateral stimulations may be more effective at suppressing CSD (60). In addition, the more robust response seen in ACT2 (39), which employed unilateral stimulations ipsilateral to the side of predominant pain, compared with the response in ACT1 (38), which employed right-side stimulations only, suggests that the former stimulation paradigm may be more effective than the latter for cluster headache pain. Thus, unilateral stimulations (ipsilateral to the side of predominant pain) are being used in PREMIUM II (59). Given the effect of nVNS on the autonomic nervous system, additional data on unilateral stimulation in cluster headache and migraine with autonomic features may yield results that lead to modification of the treatment paradigm. To increase adherence, the PREMIUM II study includes subjects with a higher overall headache burden than those in the original PREMIUM study, who may be more highly motivated to adhere to the three-times-per-day treatment regimen (43). The sham device used in PREMIUM produced some level of vagal activation (55). A modified inactive sham device has been adopted in the PREMIUM II study to address this issue (43). The challenges involved in conducting sham-controlled trials with neuromodulation devices are illustrated by the substantial monthly variation in treatment effects for the sham arm in the STS study, in which migraine days decreased by approximately 20% in month 1 and returned to near-baseline levels at month 3 (57).
Clinical safety and tolerability
No serious treatment-related adverse events were reported in any of the controlled clinical trials of nVNS for cluster headache (ACT1 (38), ACT2 (39), PREVA (40)) or migraine (PRESTO (41), EVENT (42), PREMIUM (43)). In total, 1091 subjects were randomly assigned to receive nVNS during these trials, and among them, 299 adverse device events (ADEs) were reported. The ADEs most commonly reported during the trials consisted of short-term discomfort at the site of device application (Table 2). These findings, which represent the largest dataset of any neuromodulation device (n = 1091), illustrate the benign safety and tolerability profile of nVNS.
Most commonly reported ADEs during clinical trials of nVNS (n = 1091).

Note: Data on file. electroCore, Inc.
ADE: adverse device event; nVNS: non-invasive vagus nerve stimulation.
Discussion
nVNS combines clinically meaningful benefits with good tolerability and safety. It can be used alone or in combination with pharmacologic treatments without the risk of drug interactions and for both mild and more complicated headache cases. Unlike many other available treatments, nVNS has been studied for up to 24 stimulations per day and may be used without restrictions on the number of treatments due to concerns about medication overuse headache (37,61). The safety profile of nVNS includes limited cardiac risk. Although nVNS has not been studied specifically in pregnant women and has limited data from use in adolescent patients, the lack of systemic off-target effects associated with nVNS suggests that it could be a possible alternative therapeutic option for such patients and for those who have not responded to/are unable to tolerate pharmacologic treatments (62,63). nVNS is controlled by the user; it stimulates the patient’s own body to treat itself without the need for pharmacologic intervention. In this way, nVNS may also contribute to improved quality of life for patients and provide cost savings over SoC (46).
By applying nVNS, the user activates afferent pathways that project to numerous brain regions to trigger modulation of descending pain pathways (16,28); top-down modulation strategies such as this have been hypothesized to progressively reduce allostatic load, which results in symptomatic improvement (64). Headache pathophysiology involves multiple mechanisms that differ depending on the type of headache and the individual patient. In cases when a particular mechanism(s) appears to be primarily responsible for a patient’s condition, the use of a specifically targeted agent could be ideal. When the headache etiology is multifactorial and/or is associated with comorbidities, a treatment that engages multiple pathways may be more effective. The effects of nVNS on autonomic nervous system functions, CSD inhibition, neurotransmitter regulation, and nociceptive modulation allow the activation of one or more of these mechanistic pathways in response to the headache. The diverse mechanistic pathways affected by nVNS, together with its exceptional safety and tolerability profile, allow its use as monotherapy or as a complement to existing medications.
All the clinical studies of nVNS reviewed here allowed adjunctive use of preventive medications, acute treatments, or both, but no studies have been conducted to quantify the specific adjunctive benefits of nVNS when combined with individual pharmacologic products. Although there are no data to support recommendations for specific nVNS/medication combinations, its multiple mechanisms of action, lack of drug interactions, and ability to be used daily to decrease the frequency of attacks or acutely at any time during a headache attack enables its use as a complement to a variety of agents. For example, clinical experience suggests that patients who receive onabotulinumtoxinA or calcitonin gene-related peptide (CGRP) inhibitor therapy for migraine prevention benefit from acute treatment of attacks with nVNS, particularly near the end of the dosing period (3 months for onabotulinumtoxinA, 1 or 3 months for CGRP inhibitors). For patients who wish to use nVNS more broadly, it can be used as both preventive and acute treatment while still allowing for the addition of targeted preventive and/or acute treatments as necessary for persistent pain or breakthrough attacks.
In the absence of head-to-head studies, specific conclusions regarding the comparative efficacy of different treatments cannot be drawn. Nevertheless, several attributes of nVNS, together with the quality of evidence to support its efficacy, suggest that nVNS compares favorably with other non-invasive neuromodulation treatments for primary headache. nVNS is distinct in that it is the only device that has demonstrated efficacy in both acute and preventive treatment of cluster headache (34–36). It is the only FDA-cleared device for cluster headache and is the only therapy (device or pharmacologic) that is FDA cleared/approved for the prevention of both episodic and chronic cluster headache. For both acute and preventive treatment of migraine, results from multiple large randomized controlled trials of nVNS (37–39) are suggestive of efficacy similar to that of other neuromodulation devices (57,58,65–68). In a recent systematic review, nVNS was recognized as having a higher class/quality of evidence than other devices (62). nVNS also offers a unique combination of convenience, portability, and short stimulation time that is not available with other devices.
Future directions
Successful use of nVNS in treating primary headache has led to the study of its utility in other disorders. Clinical studies are planned or ongoing to investigate the use of nVNS as acute and preventive treatment of post-traumatic headache (69), in the treatment of headache associated with subarachnoid haemorrhage (70), and in supporting overall recovery from traumatic brain injury (71). Results from a recent study showed that nVNS reduces sympathetic activation in response to stress, which suggests possible clinical applications for post-traumatic stress disorder (72). A recent case report described attenuation of visual and somatosensory aura symptoms by nVNS in a patient with migraine (73). This finding, along with the mechanistic evidence of CSD suppression by nVNS, provides a compelling rationale for further comprehensive assessment of nVNS in treating migraine aura symptoms.
The interesting possibility of using nVNS during pregnancy and in adolescents with migraine needs to be explored further. A safety study suggested nVNS was safe, well tolerated, and effective for acute treatment of migraine in adolescent patients (74), but additional research is needed to confirm the study’s efficacy results. nVNS is being investigated for controlling the vestibular symptoms in patients with vestibular migraine (VM). In a retrospective record review that included 14 patients who had VM attacks during the study, vertigo severity decreased by a mean of 46.9% after nVNS use, with 13 patients (93%) reporting improvements (75).
One common challenge in the development of neuromodulatory treatments and their subsequent study is optimization of the stimulation parameters and protocols. In both clinical and mechanistic studies, the stimulation protocols used for nVNS have varied in the number of stimulations, the length of the interval (if any) between stimulations, and the use of unilateral or bilateral stimulations (38–43,49,60). Data from these studies suggest that a treatment paradigm including two or three unilateral stimulations is optimal for the treatment of migraine and cluster headache, respectively. Although there is subjectivity involved in determining an ideal stimulation protocol, additional variations in the nVNS treatment paradigm should continue to be considered. The lack of dose-limiting toxicity with nVNS offers a clinical benefit in this regard.
Conclusions
Scientific and clinical studies support the emergence of nVNS as a safe, well-tolerated, effective, and practical treatment for primary headache disorders. nVNS should be considered a) a first-line treatment for both the acute and preventive treatment of cluster headache, b) an effective option for acute treatment of migraine with therapeutic benefits similar to those of triptans, and c) a highly relevant, practical option for migraine preventive therapy.
Article highlights
The most compelling mechanistic explanations for the efficacy of nVNS in headache disorders involve four core areas: Effects on autonomic nervous system functions; inhibition of cortical spreading depression; neurotransmitter regulation; and nociceptive modulation. Evidence from recent clinical trials and real-world use supports the efficacy of nVNS as acute and preventive treatment for both cluster headache and migraine. Safety and tolerability from clinical trials of nVNS, which represent the largest dataset for any neuromodulation device (n = 1091), illustrate the benign safety and tolerability profile of nVNS. Clinically, nVNS offers advantages in being usable at any stage in the course of treatment, alone or with other treatments without risk of drug interactions, and for up to 24 stimulations per day without restrictions. nVNS provides a unique level of control to the patient as it offers a safe, flexible, and effective non-drug option.
Footnotes
Acknowledgments
Writing assistance and editorial support were provided by Elizabeth SO Barton, MS, of MedLogix Communications, LLC, in cooperation with the authors.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SS has received honoraria from Abide Therapeutics; Alder BioPharmaceuticals (now Lundbeck); Allergan, Inc.; Amgen; Autonomic Technologies; Biohaven Pharmaceuticals; Cefaly; Curelator, Inc.; Dr. Reddy’s Laboratories; Egalet Corporation; GlaxoSmithKline, LLC; electroCore, Inc.; eNeura, Inc.; Impel Pharmaceuticals; Lilly USA LLC; Medscape LLC; the National Institute of Neurological Disorders and Stroke; Satsuma Pharmaceuticals; Supernus Pharmaceuticals, Inc.; Teva Pharmaceuticals; Theranica; and Trigemina, Inc.
HY has received consultant fees from Supernus Pharmaceuticals, Inc.
UN reports no disclosures.
JA has received consultancy fees from Alder BioPharmaceuticals, Allergan, Biohaven Pharmaceuticals, Eli Lilly and Company, Impel, Lundbeck, Revance, Satsuma Pharmaceuticals, Teva Pharmaceutical Industries, and Zosano Pharma; speaker fees from Alder BioPharmaceuticals, Allergan, Amgen, Biohaven Pharmaceuticals, Eli Lilly and Company, Lundbeck, and Teva Pharmaceutical Industries; and support for clinical trials from Allergan, Biohaven Pharmaceuticals, Eli Lilly and Company, Satsuma Pharmaceuticals, and Zosano Pharma. She is also a section editor for Current Pain and Headache Reports.
ALdM reports no disclosures.
PGM has served as a consultant for Allergan, Amgen, Avanir, Biohaven, BioMobie, Cowen, electroCore, Inc., Ipsen, Lilly, Novartis, Promius, Revance, Satsuma, Stealth BioTherapeutics, Supernus, Takeda, Theranica, Teva, and Upsher-Smith. He has no ownership interests and does not own any pharmaceutical company stocks. He served as an uncompensated consultant (not in his capacity as a faculty member of the Harvard Medical School/Brigham & Women’s Hospital).
EL is an employee of electroCore, Inc., and receives stock ownership.
CT has received consultancy fees from Allergan S.p.A., electroCore, LLC, Eli Lilly and Company, and Novartis AG and research grants from the European Commission and the Italian Ministry of Health. She is also a principal investigator or collaborator for RCTs sponsored by Alder BioPharmaceuticals Inc., Eli Lilly and Company, and Teva Pharmaceutical Industries Ltd.
H-CD has received honoraria for participation in clinical trials and for contributions to advisory boards and oral presentations sponsored by 3M Medica, Addex Therapeutics, Alder BioPharmaceuticals, Allergan, Almirall, Amgen, AstraZeneca, Autonomic Technologies, Bayer, Berlin-Chemie, Boehringer Ingelheim, Bristol-Myers Squibb, Chordate Medical, Coherex Medical, CoLucid Pharmaceuticals, Data Sciences International, electroCore, Inc., Eli Lilly, GlaxoSmithKline, Grünenthal, Janssen-Cilag, Johnson & Johnson, Labrys Biologics, Roche Pharma, Medtronic, Menarini, Minster Pharmaceuticals, MSD, Novartis, Pfizer, Pierre Fabre, Sanofi, Schaper and Brümmer, St. Jude Medical, Vital, and Weber & Weber. He has also received research funding from Allergan, Almirall, AstraZeneca, Bayer, electroCore, Inc., GlaxoSmithKline, Janssen-Cilag, MSD, and Pfizer. He has received additional research support from the European Union, the German Ministry of Education and Research, and the German Research Foundation. He has no ownership interests and does not own any pharmaceutical company stocks.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was funded by electroCore, Inc.
