Abstract
Background:
Erenumab, a fully human monoclonal antibody targeting the calcitonin gene-related peptide pathway, was developed specifically for preventive treatment of migraine.
Objective:
To compare the real-world effectiveness of erenumab and non-specific oral migraine preventive medication (OMPM) on acute medication usage and healthcare resource utilization (HCRU) among migraine patients.
Methods:
This retrospective US claims analysis included patients (≥18 years) diagnosed with migraine who initiated erenumab (May 01, 2018 and September 30, 2019) or OMPM (May 01, 2016 and October 31, 2017). Cohorts were matched 1:1 using the propensity score (PS) method with stratification. Acute medication usage, HCRU, and a composite endpoint of 1) outpatient visit with a migraine diagnosis and associated acute medication claim, 2) hospital admission with a primary migraine diagnosis, or 3) emergency room visit with a primary migraine diagnosis were assessed 6 months post-treatment initiation.
Results:
Following PS matching, both cohorts included 2,343 patients. At 6 months, erenumab was associated with significantly less acute medication usage versus OMPM, including number of types of acute medications used, number of claims per person, and proportion of patients using acute medication. HCRU and number of composite events were also significantly lower among erenumab users.
Conclusion:
Erenumab is more effective than OMPM at reducing acute medication usage and HCRU among migraine patients.
Trial registration:
N/A.
Introduction
Migraine is a common, complex, and long-term disabling neurological disease that imposes a significant burden on patients’ lives and on society. 1 –4 Migraine can either be categorized as episodic migraine (EM), defined as <15 headache days per month, or chronic migraine (CM), defined as ≥15 headaches days per month for more than 3 months, of which ≥8 days have features of migraine. 5 Guidelines recommend the use of both acute and preventive therapy for the pharmacological management of migraine, with their use depending on the severity and frequency of attacks, as well as patient characteristics and preferences. 6
The main goal of acute medication is to rapidly restore function, with minimal recurrence and avoidance of side effects. 7 However, in reality, older acute medications are often associated with limited efficacy, leading to an increased need for their use. 8 –13 This, in turn, can lead to the development of medication overuse headache, to chronification from EM to CM, to exacerbation or precipitation of comorbid medical and/or psychiatric conditions, and ultimately, to greater utilization of healthcare resources. 14 –18 Newer acute agents such as lasmiditan and gepants have become available in US since early 2020, which was beyond the study period of this retrospective study. In contrast, the goal of migraine preventive medication is to reduce the frequency, duration, and severity of attacks, the overall cost associated with migraine, and the use of acute medications, including migraine-specific medications. None of the currently available oral migraine preventive medications (OMPM, such as antihypertensives, anticonvulsants, or antidepressants), were developed specifically for migraine prevention, and many have limited to moderate efficacy, poor tolerability, or contraindications that may restrict their use. 19 –22 Adherence and persistence to OMPM are poor among patients with migraine, mainly due to lack of efficacy and intolerable side effects, leading many to rely on acute medication for pain relief. 21 –23
On May 17, 2018, the United States (US) Food and Drug Administration (FDA) approved erenumab (erenumab-aooe in the US; Aimovig®), a fully human monoclonal antibody (mAb) that targets the calcitonin gene-related peptide (CGRP) pathway. 24 Erenumab, along with other mAbs in its class, namely galcanezumab, fremanezumab, and eptinezumab, was specifically designed for the preventive treatment of migraine, and its clinical efficacy and safety have been established in several clinical trials. 25,26 We previously demonstrated that 6 months of erenumab treatment was associated with reduced acute medication usage and healthcare resource utilization (HCRU) in a real-world setting. 27 Furthermore, the magnitude of the reductions were generally higher in erenumab- than onabotulinumtoxinA-treated patients. 28 The aim of this study was to compare the effectiveness of erenumab and OMPM on acute medication usage (triptans, opioids, non-steroidal anti-inflammatory drugs [NSAIDs], and barbiturates) and HCRU among commercially insured patients with migraine in the US.
Methods
Data source
Data were extracted from the Optum’s de-identified Clinformatics® Data Mart (CDM), which is a database of administrative health claims from beneficiaries of commercial and Medicare Advantage health plans in the US. Data are derived from claims submitted by providers and pharmacies to obtain payment for healthcare services rendered, track plan membership for premium billing, and track participating physicians who have contracts with health plans to provide services. Such data provide a key source of information for a variety of research efforts, including research related to healthcare costs and resource utilization, as well as quality and effectiveness.
Study design
This was a retrospective, treatment effectiveness, non-interventional, observational study. The aim of the study was to evaluate the real-world impact of erenumab and OMPM on acute medication usage and HCRU (including emergency room [ER] visits/inpatient visits, office visits, neurologist or headache specialist visits, and other outpatient visits) among patients with migraine 6 months after treatment initiation.
This study was conducted in accordance with the Guidelines for Good Pharmacoepidemiology Practices (GPP) of the International Society for Pharmacoepidemiology (ISPE) 2016, the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, 29 and with the ethical principles laid down in the Declaration of Helsinki. This study was exempt from Institutional Review Board approval since only de-identified patient records were used.
Study population
Patients eligible for inclusion in the erenumab cohort were selected from the Optum’s de-identified CDM database based on having ≥1 migraine diagnosis (ICD10 codes is available in Supplementary Table S1) during the identification period between May 01, 2017 and September 30, 2019, ≥1 prescription for erenumab between May 01, 2018 and September 30, 2019, and ≥3 erenumab doses in the 6-month post-index period, including the index date (Supplementary Figure S1). The index date refers to the first erenumab or OMPM claim during the identification period. Eligibility criteria for inclusion in the OMPM cohort included ≥1 migraine diagnosis between May 01, 2015 and April 30, 2018, and ≥1 prescription (minimum of 28 days’ supply) for any OMPM during the identification period between May 01, 2016 and October 31, 2017. All patients in both cohorts were required to be aged ≥18 years as of the index date and have continuous medical/pharmacy coverage in the 12-month pre-index and 6-month post-index periods.
Patients were excluded from the erenumab cohort if they used other non-erenumab anti-CGRP therapies during the 12-month pre-index period or 6-month post-index period. Patients were excluded from the OMPM cohort if they used any anti-CGRP therapy in the 12-month pre-index period or in the follow-up period until September 30, 2019. Patients were also excluded from the OMPM cohort if they used onabotulinumtoxinA in the 12-month pre-index period or in the 6-month post-index period.
Different identification periods were used for the selection of eligible patients in the erenumab and OMPM cohorts due to the FDA approval and subsequent availability of CGRP pathway targeted therapies since May 2018 in the US. The development of CGRP pathway targeted therapies initiated a new era in the acute and preventive treatment of primary headache disorders. 30,31 Thus, matching patients in the erenumab and OMPM cohorts to ensure similarity of baseline disease severity becomes challenging after May 2018. To ensure the closest alignment as possible to clinical practice following the introduction of CGRP pathway targeted therapies, a period of 2 years prior to May 2018 was chosen to identify OMPM patients.
Data from the 12-month pre-index period for both the erenumab and OMPM cohorts were used to determine most baseline characteristics (e.g. preventive medication use and HCRU). Data from the 6-month pre-index period were used to determine baseline acute medication use.
Given that non-migraine specific acute and preventive medications are approved for other conditions, a claim associated with a migraine diagnosis was required. Use of non-migraine specific acute medication (NSAIDs, opioids, and barbiturates) required a migraine diagnosis on or before 7 days of the medication claim. Use of non-migraine specific preventive medication required a migraine diagnosis on or before 14 days of the medication claim, with a supply of ≥28 days. Patients identified for the OMPM cohort were allowed to have switched between classes of OMPM, which included anticonvulsants, antidepressants, angiotensin II receptor blockers, angiotensin-converting enzyme inhibitors, beta blockers, calcium channel blockers, and ergots (methysergide maleate; not commonly used for preventive treatment).
Outcomes
To evaluate the effectiveness of erenumab and OMPM on acute medication usage among patients with migraine in the US, the number of types of acute medication used at a specific class level (triptans, opioids, NSAIDs, and barbiturates) or at generic drug level, the number of claims per person for acute medication, and the proportion of patients using acute medication in the 6-month post-index period were assessed.
To evaluate the effectiveness of erenumab and OMPM on HCRU, the number of all-cause and migraine-specific ER/inpatient visits, office visits, neurologist or headache specialist visits, and other outpatient visits per person 6 months post-treatment initiation of erenumab or OMPM were assessed. The proportion of patients with all-cause and migraine-specific visits to healthcare providers 6 months post-treatment initiation of erenumab or OMPM was also evaluated.
An exploratory objective was to explore the composite endpoint of 1) outpatient visits with a diagnosis of migraine and an associated acute medication claim, 2) hospital admissions with a primary diagnosis for migraine, or 3) ER visits with a primary diagnosis for migraine. Any events that occurred ≤3 days apart were counted only once.
Statistical analysis
Data analyses were performed using SAS® studio 3.8 (Copyright © 2018, SAS Institute Inc., Cary, NC). The erenumab and OMPM cohorts were matched 1:1 using the propensity score (PS) method with stratification. Stratification was performed based on gender, diagnosis of CM without aura, onabotulinumtoxinA use in the 12-month pre-index period, number of preventive drugs used during the 12-month pre-index period, index prescriber specialty, migraine-specific inpatient and ER visits in the 12-month pre-index period, and the number of migraine-specific office visits, including the number of visits to a neurologist or headache specialist, and other outpatient visits in the 12-month pre-index period. Greedy nearest neighbor matching with caliper = 0.05 was used. The variables for PS matching included age, gender, Charlson Comorbidity Index, insurance type, region, selected comorbidities (insomnia, depression, cardiovascular disease, irritable bowel syndrome, and fibromyalgia), CM without aura, acute/preventive drug use, and HCRU. Bivariate analyses of baseline characteristics on pre- and post-PS matching were performed, with the standardized mean difference reported to assess the heterogeneity of the two cohorts.
PS-matched data were used to assess the comparative effectiveness of erenumab and OMPM; logistic regression with covariate adjustment was used for dichotomous variables, and a negative binomial model with covariate adjustment was performed for count variables, with odds ratios (OR) and rate ratios (RR) and 95% confidence intervals (CI) calculated. A proportional odds model with covariate adjustment was performed to assess the odds of having a higher number of different types of acute medication for both cohorts. Sensitivity analysis was performed using the inverse probability of treatment weighting (IPTW) model with 1:3 randomized PS-matched data for erenumab and OMPM, respectively (Supplementary Materials).
Results
A total of 4679 erenumab patients met the entry criteria and were identified between May 01, 2017 and September 30, 2019; and 75,861 OMPM patients met the entry criteria and were identified between May 01, 2015 and April 30, 2018 from the Optum’s de-identified CDM database (Supplementary Figure S2). Following stratified PS matching, the number of patients included in the analyses was 2343 for each cohort.
Demographic and baseline clinical characteristics for the pre-matched and post-matched cohorts are shown in Table 1. In the post-matched data, 88.4% were female, 39.9% had a diagnosis of CM, and the mean age was 49 years. Most patients (70.8%) were prescribed erenumab or OMPM by a neurologist or headache specialist, while 19.7% received their prescription from a general practitioner. Greater than 60% of all patients in both cohorts used some form of acute medication to treat their migraine, with the most common being triptans and opioids. The baseline data were well matched with SMD below 0.1 for all covariates included on the PS model.
Demographics and baseline clinical characteristics of study patients.
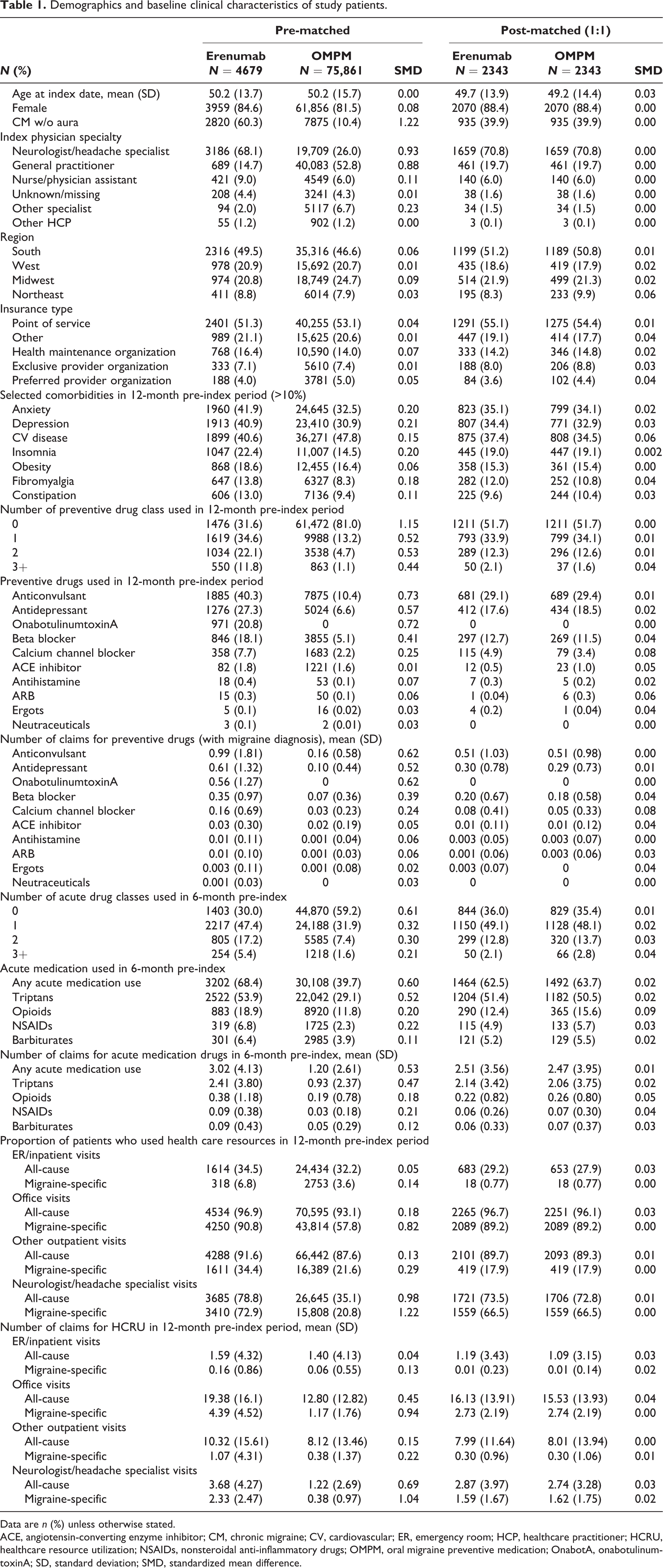
Data are n (%) unless otherwise stated.
ACE, angiotensin-converting enzyme inhibitor; CM, chronic migraine; CV, cardiovascular; ER, emergency room; HCP, healthcare practitioner; HCRU, healthcare resource utilization; NSAIDs, nonsteroidal anti-inflammatory drugs; OMPM, oral migraine preventive medication; OnabotA, onabotulinumtoxinA; SD, standard deviation; SMD, standardized mean difference.
Acute medication usage in the 6-month post-index period
At 6-month post-index period, the adjusted number of types of acute medications used (i.e. 0, 1, 2, 3+) was significantly lower in the erenumab cohort compared with the OMPM cohort (OR 0.70 [95% CI: 0.63, 0.79]; p < 0.0001) (Table 2).
Number of type of acute medications used in the 6-month post-index period.
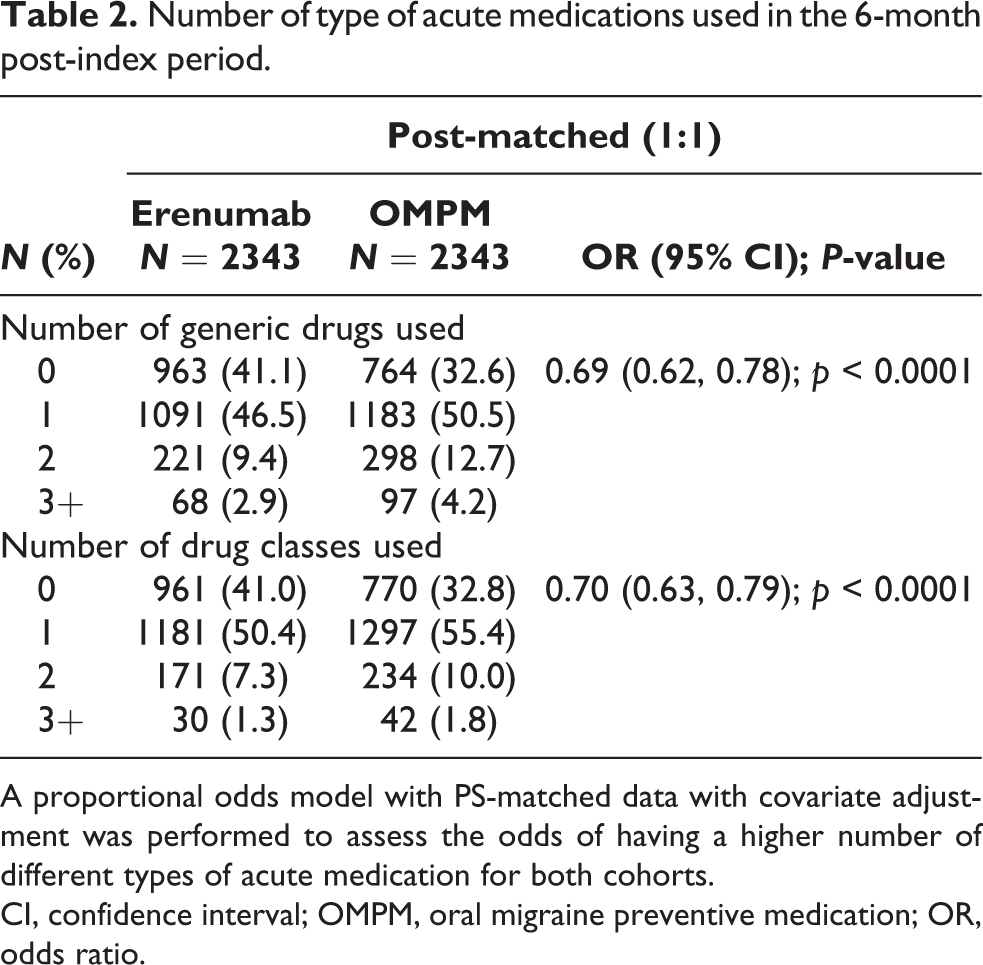
A proportional odds model with PS-matched data with covariate adjustment was performed to assess the odds of having a higher number of different types of acute medication for both cohorts.
CI, confidence interval; OMPM, oral migraine preventive medication; OR, odds ratio.
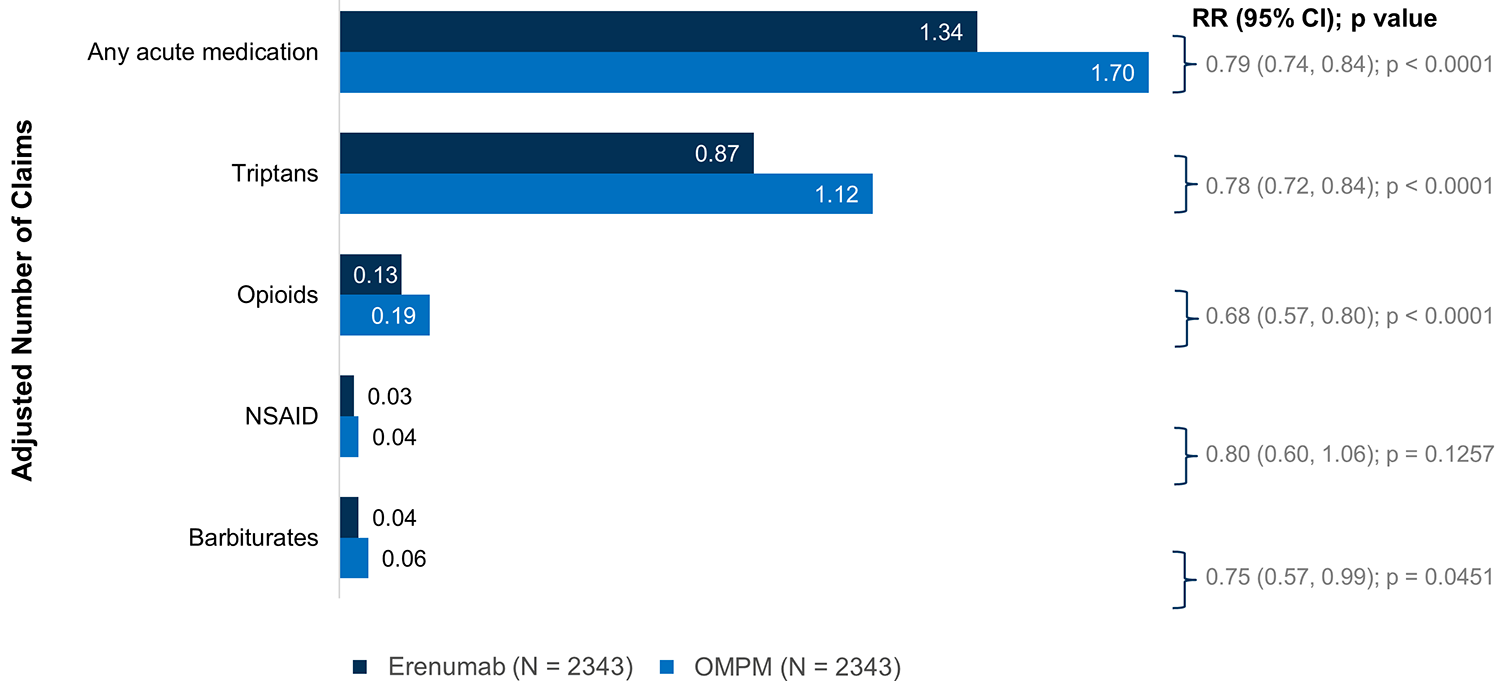
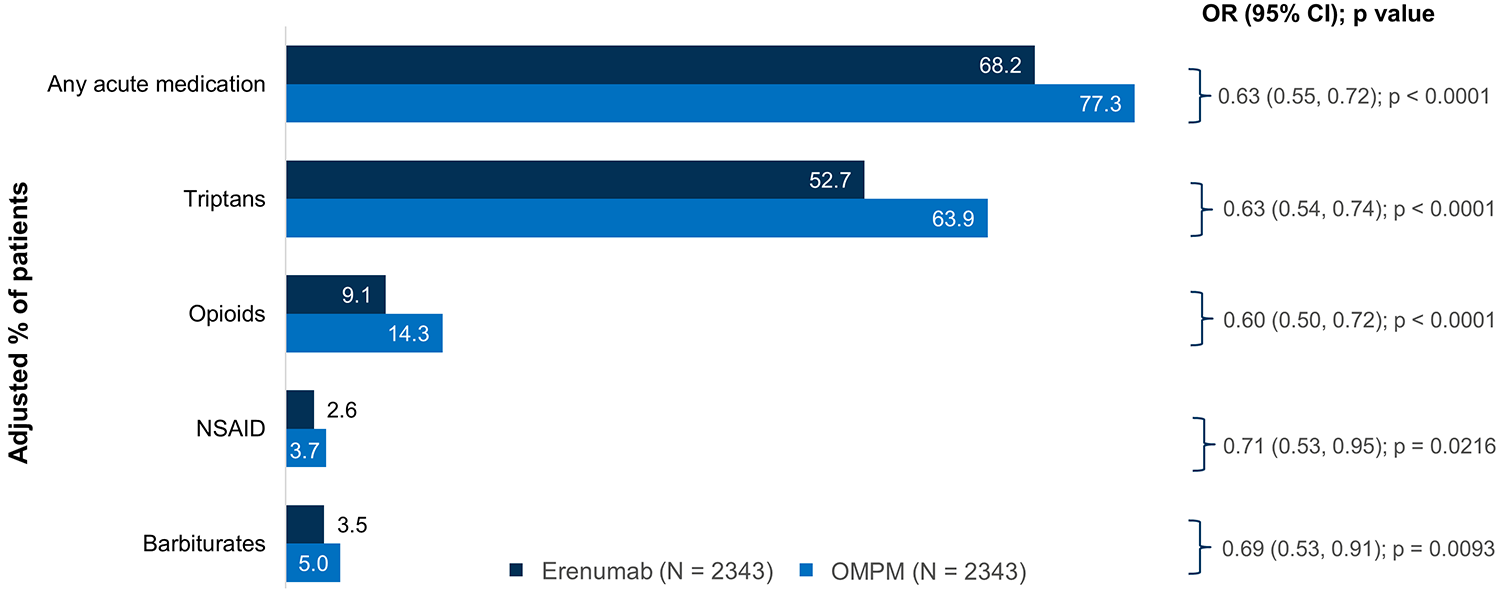
The adjusted average number of claims per person for any acute medication was also significantly lower in the erenumab cohort in the 6-month post-index period (1.34 vs 1.70 in the OMPM cohort; RR 0.79 [95% CI: 0.74, 0.84]; p < 0.0001) (Figure 1), as was the adjusted proportion of patients who used any acute medication (68.2% vs 77.3%; OR 0.63 [95% CI: 0.55, 0.72]; p < 0.0001) (Figure 2). The adjusted average number of claims per person for triptans in the 6-month post-index period was lower in the erenumab cohort (0.87 vs 1.12 in the OMPM cohort; RR 0.78 [95% CI: 0.72, 0.84]; p < 0.0001) (Figure 1). Similarly, the adjusted proportion of patients who used triptans was lower in the erenumab cohort (52.7% vs 63.9% in the OMPM cohort; OR 0.63 [95% CI: 0.54, 0.74]; p < 0.0001) (Figure 2).
Number of claims per person for acute medication use in the 6-month post-index period. Use of non-migraine specific acute medications (NSAIDs, opioids, and barbiturates) required a migraine diagnosis on or before 7 days of the medication claim. Negative binomial model with PS-matched data with covariate adjustment (age, gender, CCI, insurance type, region, selected comorbidities (CM, insomnia, depression, CV, IBS, fibromyalgia), number of acute/preventative drug use by class (numerical), number of acute drugs (barbiturates, triptans, NSAIDS, opioids, antiemetics), all-cause inpatient or ER visit (binary), number of migraine-specific inpatient/ER visit, office visits, and others visits, and number of office visits to neurologists. CCI, Charlson Comorbidity Index; CI, confidence interval; CM, chronic migraine; CV, cardiovascular; ER, emergency room; IBS, irritable bowel syndrome; NSAID, nonsteroidal anti-inflammatory drug; OMPM, oral migraine preventive medication; PS, propensity score; RR, rate ratio.
Proportion of patients who used acute medication in the 6-month post-index period. Use of non-migraine specific acute medications (NSAIDs, opioids, and barbiturates) required a migraine diagnosis on or before 7 days of the medication claim. Logistic regression model with PS-matched data with covariate adjustment (age, gender, CCI, insurance type, region, selected comorbidities (CM, insomnia, depression, CV, IBS, fibromyalgia), number of acute/preventative drug use by class (numerical), acute drug use by class (categorical), number of acute drugs (barbiturates, triptans, NSAIDS, opioids, antiemetics), all-cause inpatient or ER visit (binary), number of migraine-specific inpatient/ER visit, office visits, and others visitis, and number of office visits to neurologists. CCI, Charlson Comorbidity Index; CI, confidence interval; CM, chronic migraine; CV, cardiovascular; ER, emergency room; IBS, irritable bowel syndrome; NSAID, nonsteroidal anti-inflammatory drug; OMPM, oral migraine preventive medication; OR, odds ratio; PS, propensity score.
The adjusted average number of claims per person for opioids in the 6-month post-index period was lower in the erenumab cohort (0.13 vs 0.19 in the OMPM cohort; RR 0.68 [95% CI: 0.57, 0.80]; p < 0.0001) (Figure 1), as was the proportion of patients who used opioids (9.1% vs 14.3%; OR 0.60 [95% CI: 0.50, 0.72]; p < 0.0001) (Figure 2). The adjusted average number of claims per person for barbiturates in the 6-month post-index period was also lower in the erenumab cohort (0.04 vs 0.06 in the OMPM cohort; RR 0.75 [95% CI: 0.57, 0.99]; p = 0.0451) (Figure 1). Similarly, the proportion of patients who used barbiturates in the 6-month post-index period was lower in the erenumab cohort (3.5% vs 5.0% in the OMPM cohort; OR 0.69 [95% CI: 0.53, 0.91]; p = 0.0093) (Figure 2).
The number of claims per person for NSAIDs was not significantly different between the two cohorts, however, the proportion of patients who used NSAIDs in the 6-month post-index period was significantly lower in the erenumab cohort (2.6% vs 3.7% in the OMPM cohort; OR 0.71 [95% CI: 0.53, 0.95]; p = 0.0216 (Figure 2). The results for ergot use in the 6-month post-index period are not reported due to insufficient data.
Sensitivity analysis of the IPTW model using the 1:3 randomized PS-matched data demonstrated similar findings for the number of types of acute medications used (Supplementary Table S2), number of claims per person for acute medication (Supplementary Figure S3), and for the proportion of patients who used acute medication (Supplementary Figure S4) in the 6-month post-index period.
HCRU in the 6-month post-index period
By 6 months, the adjusted average number of migraine-specific office visits per person was significantly lower in the erenumab cohort (1.34 vs 1.62 in the OMPM cohort; RR 0.83 [95% CI: 0.78, 0.87]; p < 0.0001); however, the average number of all-cause office visits per person, although numerically lower in the erenumab cohort, was not statistically different between cohorts (Figure 3). Nearly all patients in both cohorts had an all-cause office visit in the 6-month post-index period, but the proportion of patients with migraine-specific office visits was lower in the erenumab cohort (77.2% vs 79.8% in the OMPM cohort; OR 0.86 [95% CI: 0.75, 0.98]; p = 0.0249) (Figure 4).

Number of visits to healthcare providers in the 6-month post-index period. *1:1 PS-matched model did not converge (results from sensitivity analyses are shown). Negative binomial model with PS-matched data with covariate adjustment (age, gender, CCI, insurance type, region, selected comorbidities (CM, insomnia, depression, CV, IBS, fibromyalgia), number of acute/preventative drug use by class (numerical), number of acute drugs (barbiturates, triptans, NSAIDS, opioids, antiemetics), all-cause inpatient or ER visit (binary), number of migraine-specific inpatient/ER visit, office visits, and others visits, and number of office visits to neurologists. CCI, Charlson Comorbidity Index; CI, confidence interval; CM, chronic migraine; CV, cardiovascular; ER, emergency room; IBS, irritable bowel syndrome; NSAID, nonsteroidal anti-inflammatory drug; OMPM, oral migraine preventive medication; PS, propensity score; RR, rate ratio.
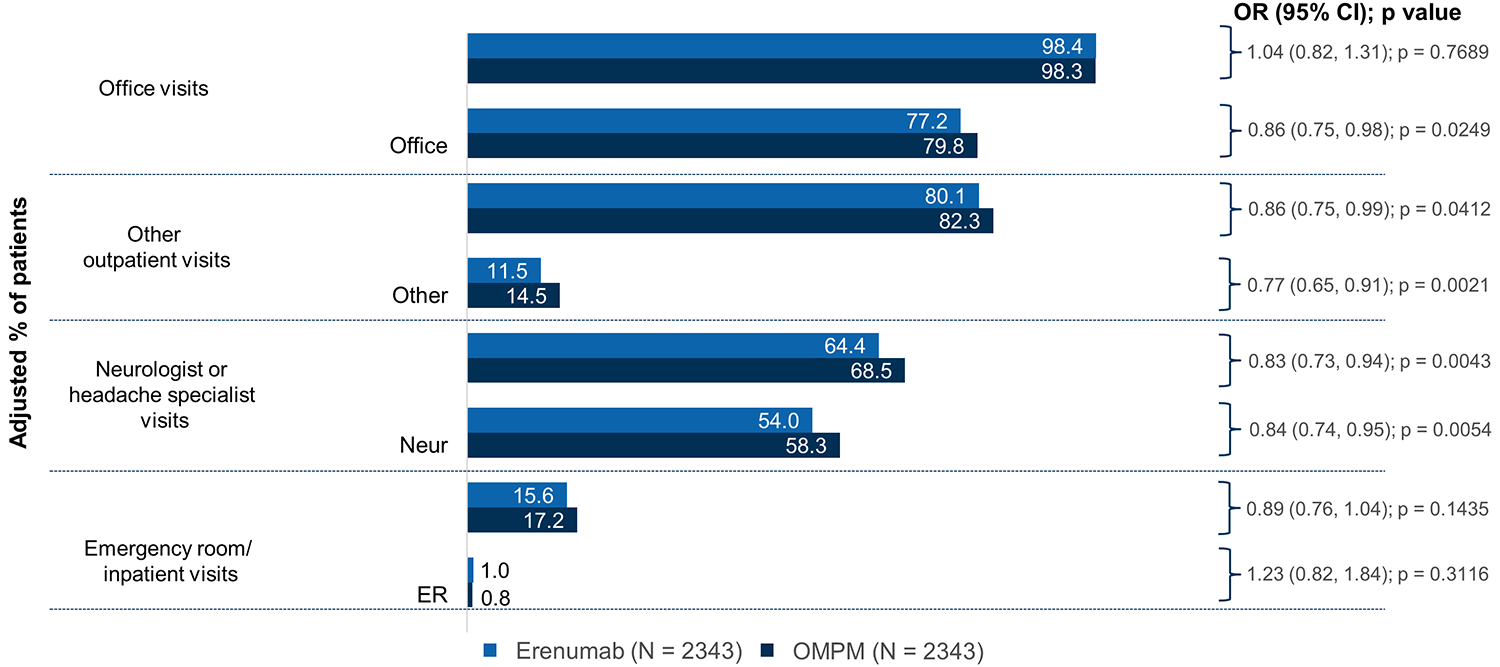
Proportion of patients with visits to healthcare providers in the 6-month post-index period. *1:1 PS-matched model did not converge (results from sensitivity analyses are shown). Logistic regression model with PS-matched data with covariate adjustment (age, gender, CCI, insurance type, region, selected comorbidities (CM, insomnia, depression, CV, IBS, fibromyalgia), number of acute/preventative drug use by class (numerical), acute drug use by class (categorical), number of acute drugs (barbiturates, triptans, NSAIDS, opioids, antiemetics), all-cause inpatient or ER visit (binary), number of migraine-specific inpatient/ER visit, office visits, and others visits, and number of office visits to neurologists. CCI, Charlson Comorbidity Index; CI, confidence interval; CM, chronic migraine; CV, cardiovascular; ER, emergency room; IBS, irritable bowel syndrome; NSAID, nonsteroidal anti-inflammatory drug; OMPM, oral migraine preventive medication; OR, odds ratio; PS, propensity score.
The adjusted average number of all-cause visits per person to a neurologist or headache specialist in the 6-month post-index period was lower in the erenumab cohort (1.21 vs 1.36 in the OMPM cohort; RR 0.89 [95% CI: 0.82, 0.96]; p = 0.0024), as was the adjusted number of migraine-specific visits per person to a neurologist or headache specialist (0.70 vs 0.91 in the OMPM cohort; RR 0.77 [95% CI: 0.72, 0.82]; p < 0.0001) (Figure 3). The proportion of patients with all-cause visits to a neurologist or headache specialist in the 6-month post-index period was also lower in the erenumab cohort (64.4% vs 68.5% in the OMPM cohort; OR 0.83 [95% CI: 0.73, 0.94]; p = 0.0043), as was the proportion of patients with migraine-specific visits to a neurologist or headache specialist (54.0% vs 58.3% in the OMPM cohort; OR 0.84 [95% CI: 0.74, 0.95]; p = 0.0054) (Figure 4).
The adjusted average number of all-cause other outpatient visits per person in the 6-month post-index period was lower in the erenumab cohort (3.15 vs 3.80 in the OMPM cohort; RR 0.83 [95% CI: 0.76, 0.91]; p < 0.0001), as was the number of migraine-specific other outpatients visits (0.18 vs 0.23 in the OMPM cohort; RR 0.78 [95% CI: 0.67, 0.91]; p = 0.0022) (Figure 3). Similarly, the proportion of patients with all-cause other outpatient visits was lower in the erenumab cohort (80.1% vs 82.3% in the OMPM cohort; OR 0.86 [95% CI: 0.75, 0.99]; p = 0.0412), as was the proportion of patients with migraine-specific other outpatient visits (11.5% vs 14.5%; OR 0.77 [95% CI: 0.65, 0.91]; p = 0.0021) (Figure 4).
The adjusted average number of all-cause ER/inpatient visits in the 6-month post-index period was lower in the erenumab cohort (0.34 vs 0.43 in the OMPM cohort; RR 0.78 [95% CI: 0.64, 0.95]; p = 0.0153); however, the average number of migraine-specific ER/inpatient visits was similar for both cohorts (Figure 3). The proportion of patients with all-cause and migraine-specific ER/inpatient visits was also similar for both cohorts (Figure 4).
Sensitivity analysis of the IPTW model using the 1:3 randomized PS-matched data demonstrated similar findings for the number of all-cause and migraine-specific visits to healthcare providers (Supplementary Figure S5) and for the proportion of patients with all-cause and migraine-specific visits to healthcare providers (Supplementary Figure S6) in the 6-month post-index period.
Composite endpoint
As noted, the exploratory composite endpoint consisted of 1) outpatient visits with a diagnosis of migraine and an associated acute medication claim, 2) hospital admissions with a primary diagnosis for migraine, or 3) ER visits with a primary diagnosis for migraine. Patients in the erenumab cohort had a significantly lower number of composite events compared with the OMPM cohort (0.40 vs 0.57; RR 0.71 [95% CI: 0.65, 0.77; p < 0.0001) (Figure 5(A)). A lower proportion of patients with any of the three composite events was also observed in the erenumab cohort compared with the OMPM cohort (30.5% vs 40.7%; OR 0.64 [95% CI: 0.56, 0.73; p < 0.0001) (Figure 5(B)).

Number of composite events per person (A) and proportion of patients with a composite endpoint event (B) in the 6-month post-index period. (A and B) Composite endpoint of 1) outpatient visit with a diagnosis of migraine and associated acute medication claim, 2) hospital admission with a primary diagnosis for migraine, or 3) emergency room visit with a primary diagnosis for migraine. Any events occurring ≤3 days apart were counted only once. Logistic regression for binary endpoint and negative binomial model for count data were used with PS-matched data with covariate adjustment (age, gender, CCI, insurance type, region, selected comorbidities (CM, insomnia, depression, CV, IBS, fibromyalgia), number of acute/preventative drug use by class (numerical), number of acute drugs (barbiturates, triptans, NSAIDS, opioids, antiemetics), all-cause inpatient or ER visit (binary), number of migraine-specific inpatient/ER visit, office visits, and others visits, and number of office visits to neurologists. CCI, Charlson Comorbidity Index; CI, confidence interval; CM, chronic migraine; CV, cardiovascular; ER, emergency room; IBS, irritable bowel syndrome; NSAID, nonsteroidal anti-inflammatory drug; OMPM, oral migraine preventive medication; OR, odds ratio; PS, propensity score; RR, rate ratio.
Sensitivity analysis of the IPTW model using the 1:3 randomized PS-matched data demonstrated similar findings for the number of composite events per person (Supplementary Figure S7A) and for the proportion of patients with a composite event (Supplementary Figure S7B) in the 6-month post-index period.
Discussion
This real-world effectiveness analysis suggests that over a period of 6 months, erenumab-treated patients had significantly decreased use of acute medication versus those treated with OMPM (RR 0.79; 95% CI: 0.74, 0.84; p < 0.0001). Erenumab-treated patients were also less likely to require different classes of acute medications after being treated with erenumab (RR 0.70 [95% CI: 0.63, 0.79]; p < 0.0001). All of the acute medication classes evaluated showed lower use in the erenumab group than the OMPM group. Importantly, this reduction was present for both opioid and barbiturate acute medication usage, however, it is important to note the limitations of the small number of patients treated with opioids and barbiturates. This reduced use can have profound medical, psychiatric, economic, and social implications, given the opioid crisis and the need to reduce chronic and repeated use of scheduled medications.
Furthermore, compared with the initiation of OMPM, the initiation of erenumab was associated with a lower HCRU relating to migraine-specific office visits (RR 0.83; 95% CI: 0.78, 0.87; p < 0.0001) and to a lower number of migraine-specific visits per person to a neurologist or headache specialist (RR 0.77 [95% CI: 0.72, 0.82]; p < 0.0001). In fact, this reduction of at least 23% in HCRU is considered a meaningful change especially given the widespread prevalence of migraine and the associated high rates of resource utilization.
Patients in the erenumab cohort had a significantly lower number of composite endpoints, that is, 1) outpatient visits with a diagnosis of migraine and an associated acute medication claim, 2) hospital admissions with a primary diagnosis for migraine, or 3) ER visits with a primary diagnosis for migraine (0.40 vs 0.57; RR 0.71 [95% CI: 0.65, 0.77; p < 0.0001), which may be associated with a greater reduction in the frequency of migraine attacks or severity of attacks than in those treatment with OMPM, and which in turn could lead to an overall reduction in the burden of the disease in patients with migraine. A significant reduction of 29% in the number of the composite endpoint events shows the overall benefit of erenumab in the real world. A composite endpoint such as the one reported in this study may potentially be used as a proxy to evaluate migraine attacks, although further validation is needed.
The use of claims data is subject to several limitations. As Optum’s CDM is a US database, the results gained from claims analysis apply only to the insured population in the US, which may not be generalizable to the overall population, or to the international population. US claims data are dependent on professional International Classification of Diseases (ICD) coding, not the International Classification of Headache Disorders, 3rd Edition (ICHD-3) diagnosis, and therefore, some diagnoses may be missed, different professional types may have different coding patterns, and not all coding may be accurate. Further limitations of prescription claims data are that over-the-counter (OTC) medications used to manage acute migraine attacks are missed, and a prescription does not always guarantee patient adherence. Patients receiving erenumab from the free drug program are also not captured in the Optum’s de-identified CDM database, thus, it is impossible to ascertain whether the first erenumab claim is truly indicative of the first time erenumab is used by a given patient. If used previously, the actual effectiveness might be larger than observed as baseline data may have been affected. In spite of these limitations, claims data are a valuable resource for exploratory analyses of a variety of health services research questions.
Given the widespread prevalence of migraine and the associated high rates of resource utilization, concerted efforts to reduce HCRU would help reduce burden upon the healthcare system. The personal, economic, and societal burden of migraine can be eased by optimizing acute care therapy and earlier initiation of effective preventive therapy. These findings suggest that erenumab, as a migraine preventive therapy, may reduce the holistic burden of the disease as well as overall cost in the real world.
Clinical implications
Treatment with erenumab is associated with significantly less acute medication usage compared with oral migraine preventive medication, including the number of types of acute medications used, the number of claims per person for acute medications, and the proportion of patients using acute medications.
More reduction in triptan, opioid, and barbiturate use in the erenumab group compared with conventional, non-specific oral migraine prevention should have favorable medical, psychiatric, economic, and social implications.
Healthcare resource utilization is meaningfully reduced in patients treated with erenumab compared with oral migraine preventive medication.
Erenumab treatment may be associated with less frequent migraine attacks compared with oral migraine preventive medication, as assessed using a composite endpoint (outpatient visits with a diagnosis of migraine and an associated acute medication claim; hospital admissions with a primary diagnosis for migraine; or ER visits with a primary diagnosis for migraine) as a proxy to evaluate migraine attacks.
These findings suggest that erenumab, as a migraine preventive therapy, may reduce the holistic burden of the disease as well as overall cost in the real world.
Supplemental material
Supplemental Material, sj-docx-1-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-docx-1-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-docx-2-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-docx-2-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-tif-1-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-tif-1-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-tif-2-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-tif-2-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-tif-3-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-tif-3-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-tif-4-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-tif-4-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-tif-5-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-tif-5-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-tif-6-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-tif-6-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-tif-7-rep-10.1177_25158163211040061 - Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study
Supplemental Material, sj-tif-7-rep-10.1177_25158163211040061 for Comparative effectiveness of erenumab versus oral preventive medications among migraine patients: A US claims database study by Stewart J Tepper, Juanzhi Fang, Lujia Zhou, Pamela Vo, Ahmad Abdrabboh, Mrudula Glassberg and Matias Ferraris in Cephalalgia Reports
Footnotes
Acknowledgements
Medical writing support was provided by Bronwyn Boyes and funded by Novartis.
Availability of data and materials
Raw data were generated by Optum, Inc. and are subject to their data security and privacy protocols. Derived data supporting the findings of this study are available from the corresponding author [JF] on reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.J.T. reports: Grants for research (no personal compensation): Allergan, Amgen, ElectroCore, Eli Lilly, Lundbeck, Neurolief, Novartis, Satsuma, Zosano. Consultant and/or Advisory Boards (honoraria): Aeon, Align Strategies, Allergan/AbbVie, Alphasights, Amgen, Aperture Venture Partners, Aralez Pharmaceuticals Canada, Axsome Therapeutics, Becker Pharmaceutical Consulting, BioDelivery Sciences International, Biohaven, ClearView Healthcare Partners, CoolTech, CRG, Currax, Decision Resources, DeepBench, DRG, Eli Lilly, Equinox, ExpertConnect, GLG, Guidepoint Global, Healthcare Consultancy Group, Health Science Communications, HMP Communications, Impel, InteractiveForums, Krog and Partners, Lundbeck, M3 Global Research, Magellan Rx Management, Medicxi, Navigant Consulting, Neurolief, Nordic BioTech, Novartis, Pulmatrix, Reckner Healthcare, Relevale, SAI MedPartners, Satsuma, Slingshot Insights, Spherix Global Insights, Sudler and Hennessey, Synapse Medical Communications, System Analytic, Teva, Theranica, Thought Leader Select, Trinity Partners, XOC, Zosano. Salary: Dartmouth-Hitchcock Medical Center, American Headache Society, Thomas Jefferson University. CME honoraria: American Academy of Neurology, American Headache Society, Cleveland Clinic Foundation, Diamond Headache Clinic, Elsevier, Forefront Collaborative, Hamilton General Hospital, Ontario, Canada, Headache Cooperative of New England, Henry Ford Hospital, Detroit, Inova, Medical Learning Institute Peerview, Medical Education Speakers Network, Miller Medical Communications, North American Center for CME, Physicians’ Education Resource, Rockpointe, ScientiaCME, WebMD/Medscape; J.F., P.V., A.A., M.F., and M.G. are employees and shareholders of Novartis; L.Z. is employed by KMK Consulting Inc., at the time of this study, which received funding from Novartis to conduct this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Novartis Pharma AG. Erenumab is co-developed by Amgen and Novartis.
Novartis employees contributed to study design, analysis of the data, and the decision to publish the results. KMK Consultants conducted analysis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
