Abstract
Introduction:
Migraine is a polygenic multifactorial disorder with a neuronal initiation of a cascade of neurochemical processes leading to incapacitating headaches. Headaches are generally unilateral, throbbing, 4–72 h in duration, and associated with nausea, vomiting, photophobia, and sonophobia. Chronic migraine (CM) is the presence of a headache at least 15 days per month for ≥3 months and has a high global impact on health and economy, and therapeutic guidelines are lacking.
Methods:
Using the Grading of Recommendations, Assessment, Development, and Evaluations system, we conducted a search in MEDLINE and Cochrane to investigate the current evidence and generate recommendations of clinical practice on the identification of risk factors and treatment of CM in adults.
Results:
We recommend avoiding overmedication of non-steroidal anti-inflammatory drugs (NSAIDs); ergotamine; caffeine; opioids; barbiturates; and initiating individualized prophylactic treatment with topiramate eptinezumab, galcanezumab, erenumab, fremanezumab, or botulinum toxin. We highlight the necessity of managing comorbidities initially. In the acute management, we recommend NSAIDs, triptans, lasmiditan, and gepants alone or with metoclopramide if nausea or vomiting. Non-pharmacological measures include neurostimulation.
Conclusions:
We have identified the risk factors and treatments available for the management of CM based on a grading system, which facilitates selection for individualized management.
Introduction
Headaches are a common occurrence in most people. Recurrent headaches are typically primary (i.e., nosological) entities on their own and are generally frequent, with tension-type headaches and migraines the main culprits. Migraine is a primary headache that has an episodic presentation and is characterized by pulsatile headache; unilateral moderate to intense pain; duration of 4–72 h; and is accompanied by photophobia, sonophobia, nausea and vomiting, and activity intolerance. It is occasionally preceded by an aura 5–60 min duration and sometimes prodrome and postdrome of variable symptomatology. 1 In Mexico, migraine occurs in 12.1% of women and 3.9% of men. Although migraine is second in frequency among primary headaches, disability associated with it significantly impacts family, social, and work life. 2 Unfortunately, therapeutic guidelines are lacking in the scientific literature.
Chronic migraine (CM) is a headache that occurs at least 15 days per month for ≥3 months and ≥8 days per month has migraine characteristics. Patients with CM are challenging to manage, as acute and prophylactic treatments are not always practical. Some of these treatments have not been investigated in controlled clinical trials. Therefore, many are used without sufficient scientific support or are based on acute treatment of patients with episodic migraine (EM). The justification for their use is often based on anecdotal cases or expert recommendations. In addition, epidemiological information in developing countries is scarce. Therefore, we should consider clinical features and comorbidity with psychiatric illnesses, being overweight or obese, and sleep disorders as risk factors for CM. The need to improve physician education and have guidelines in Mexico and other Latin American populations necessitates evidence-based proposals. As such, we (i.e., the Mexican Association of Headache and Migraine [AMCEMIG]) have developed guidelines for the prevention and management of CM. 3 –5
Importantly, we do not attempt to make mandatory recommendations but rather evidence-based suggestions that will help clinicians understand different therapeutic management options and select the most appropriate treatment. It is essential to consider the history of previous treatments that patients have received to reuse options that were not correctly used or avoid returning to what failed previously.
Methods
Working group
A working group formed by clinical neurologists with knowledge and interest in headaches and migraines, who belong to AMCEMIG, was convened. We questioned the treatment of CM, and aimed to better understand topics and limitations that a guide would address. This created a work schedule to be executed in five face-to-face sessions of 6–8 h each. Before each meeting, the topics and clinical questions were distributed among clinicians for their response and development in two panels. The working group members systematically formulated relevant answers to the questions posed by recommendations for developing Clinical Practice Guidelines (CCPG of the Grading of Recommendations, Assessment, Development, and Evaluation [GRADE] system). 6 Academic sessions were held to review the GRADE system and present our current rules and foundations to unify criteria and systematize the search, qualification, analysis, argumentation, and writing of specific statements and the derived synopses. 7
Working groups were formed by topic to search and evaluate scientific evidence and the elaboration and argumentation of recommendations.
GRADE system
We used the GRADE system to systematize the development of the document and evaluate the evidence to provide certainty about the knowledge and arguments that support each recommendation. 8 Using the GRADE system, we recognize that the available evidence is not always of optimal quality; therefore, we attempted to rate each evidence as high (A: systematic reviews, controlled clinical trials), moderate (B: observational studies, clinical trials with bias), low (C: case series, more consistent studies are required), or very low (D: anecdotal, unlikely for the evidence to be improved), to write specific recommendations. We also rated each recommendation uniformly and efficiently as either 1 (strong: benefits of action outweigh disadvantages, the recommendation is helpful, this is independent of the quality of evidence supporting it) or 2 (weak: benefits of action resemble disadvantages, this is independent of the quality of evidence of using the recommendation).
The topics addressed were factors that favor chronicity such as medication overuse and treatments of CM; abortive, preventive, non-pharmacological, monoclonal antibodies, onabotulinum toxin type A, peripheral nerve infiltrations, and CM; and pregnancy.
Subsequently, a search was conducted in Cochrane, MEDLINE and PubMed to assess risk factors, prevention, and treatment of CM in English and Spanish from January 1950 to May 2020. Specific keywords and MeSH terms related to the design of the study, treatment, and disease were as follows: migraine; chronic migraine; headache; chronic headache; treatment; therapy; trial; clinical trial; controlled trial; randomized clinical trial; guideline; meta-analysis; open label study; observational study; risk factors; modifying factors; chronification migraine factors; migraine AND headache; chronic migraine AND headache; chronic migraine AND chronic headache; headache AND chronic headache; migraine AND treatment; chronic migraine AND treatment; migraine AND therapy; chronic migraine AND therapy; (migraine AND headache) AND treatment; (chronic migraine AND headache) AND treatment; (chronic migraine AND chronic headache) AND treatment; (migraine AND headache) AND therapy; (chronic migraine AND headache) AND therapy; (chronic migraine AND chronic headache) AND therapy; (migraine AND headache OR chronic migraine AND headache OR chronic migraine AND chronic headache) AND (trial, clinical trial, controlled trial, randomized clinical trial, guideline, meta-analysis, open label study, observational study, risk factors, modifying factors, chronification migraine factors, migraine AND headache, chronic migraine AND headache); (migraine AND headache) AND treatment, (chronic migraine AND chronic headache) AND treatment, (migraine AND headache) AND therapy, (chronic migraine AND headache) AND therapy, (chronic migraine AND chronic headache)) AND therapy AND (trial, clinical trial, controlled trial, randomized clinical trial, guideline, meta-analysis, open label study, observational study, risk factors, modifying factors, chronification migraine factors, migraine AND headache, chronic migraine AND headache).
Selection of scientific articles
The type of article was chosen according to its content and the topic to be developed, privileging the analysis of primary literature (original observational and interventional articles), systematic reviews, and meta-analyses over secondary literature (narrative reviews, synthesis, memoirs, and consensuses); however, the reference list of secondary literature was analyzed to identify sources not identified in the initial searches.
The evidence and recommendations were weighed, rated, and classified into the four categories proposed in the GRADE system: high, moderate, low, and very low quality (Table 1). 9 These categories reflect a confidence gradient in the results offered by each study and how subsequent studies can modify the available results. With the GRADE system, the focus solely of evaluating the quality of evidence is not the methodological criticism of each study but rather the degree of confidence that a panel of experts has in the way that a study answers a PICO question(Patients, Intervention, Comparison, and Outcome) or a clinical problem statement. 10 An attempt was made to reach a consensus; the recommendations are classified as “points of good practice”, provided that the majority approved the statement in its complete and final form.
A step by step of the elaboration of the guidelines proposed by AMCEMIG.

All statements were reviewed by all authors and compiled into a single document that was then reviewed iteratively until a general agreement was reached. Once consensus was reached on the document’s final version, it was prepared in a single format, the latest version distributed via email for review and approval.
Results
Migraine chronification factors
Modifiable risk factors include overuse of analgesics for acute migraine, ineffective acute treatment, obesity, depression, and stressful life events. Low educational levels also increase the risk of CM.
Factors facilitating the presence of headache
The ICHD-III 2018 considers that any patient with CM may have a concurrent diagnosis of a MOH. Fifty percent of patients with CM are re-classified as having EM after stopping the overuse of migraine medications. Primary or secondary headache must be considered when the patient takes analgesics frequently, as it can become MOH. 7 MOH is a subtype of secondary headache, which occurs in patients with preexisting primary headache, and develops as a new type of headache or worsening of previous symptoms. The International Headache Society classifies this headache according to the subtype of medication overuse, either individually or in combination (e.g., paracetamol, ASA, or other NSAIDs), and diagnosis requires consuming the medication for at least 15 per month in the last 3 months. 1
Patients with MOH for opioids, combined analgesics, multiple drugs, or other medication classes for acute and symptomatic treatment of headache have consumed the medication for 10 or more days per month in the last 3 months. 2
MOH is complicated when it occurs with the following medical conditions: concurrent medical illness; relevant psychiatric comorbidity; a formal diagnosis of anxiety disorder, eating disorder, mood disorder, substance use disorder (including opiates, benzodiazepines, or barbiturates), relapse following previous detoxification or failure to stop medication with outpatient care, daily use of more than three doses of multiple symptomatic medications, or severe withdrawal symptoms (e.g., recurrent vomiting, migraine). In these patients, hospital treatment is more effective in to stopping the misuse. 16,17
Discontinuing medication overuse in CM patients and additional preventive measures to avoid headaches
In adults with CM, it is recommended NOT to use corticosteroids or celecoxib as a bridge therapy when ceasing the overuse and starting prophylaxis (strong recommendation, high quality of evidence, 1A). In an RCT, the use of prednisolone (75 mg) or celecoxib (400 mg) was not found to reduce headache days or the intake of overused medications as a bridge treatment when stopping overuse and initiating prophylactic treatment. 31
CM comorbidities to prevent and treat to improve quality of life
Migraine patients have consistently reported poor sleep both as a trigger and during attacks. Sleep is often thought to have a therapeutic role in ending the pain of an acute migraine attack. 31 Several studies have demonstrated that the high prevalence of sleep disorders is elevated in patients with CM. Moreover, 50% of CM patients have been found to meet the criteria for sleep disorders. Approximately 40% of patients with EM felt their sleep was adequate, compared to 34% of CM participants. 37 –41 Suzuki reports a total proportion of patients with OSAS of 12.3% and 16.6%, according to ICHD-II and ICHD-III beta, respectively. 36 Similarly, the review reports a prevalence of 12–18% of sleep apnea headaches in the middle-aged population. 34 Johnson et al. suggested that of 82 patients with chronic headache with migraine or tension-type headache or both, 63% had concomitant OSAS. 41 Provini et al. determined that OSAS is a common disorder in the general population with an estimated prevalence in the adult population of 2% in women and 4% in men. 42
The role of depression in the relationship between EM and its transformation to CM is unclear; however, a strong association has been demonstrated, and routine screening for depression should be considered patients with EM and moderate (OR = 1.77; 95% CI = 1.25–2.52), moderately severe (OR = 2.35; 95% CI = 1.53–3.62), and severe depression (OR = 2.53; 95% CI = 1.52–4.21) as they have a higher risk of developing CM. 63
The suspension of excessive and frequent use of caffeine improves the control of migraines. Therefore, based on prospective controlled clinical studies, we recommend that caffeine consumption be suspended for at least 2 weeks to 3 months in patients with CM to improve the efficacy (72% versus 40.3% with non-intervention) of acute migraine treatment. 71 –73
The Chronic Migraine Epidemiology and Outcomes study by Lipton et al. identified several subgroups of individuals that may develop CM. Patients with comorbidities demonstrated the most severe clinical phenotype. Those with only pain syndromes or combinations of respiratory/psychiatric and respiratory/pain disorders had a moderately severe clinical phenotype, whereas those with a low prevalence of comorbidities or cardiovascular comorbidities alone had a milder clinical phenotype. Ongoing studies seek to understand whether any of these classes have a differential effect on migraine progression (from EM to CM) or remittance (from CM to EM). 74 Also, the Migraine in America Symptoms and Treatment study in 15,133 people with migraine showed cardiovascular, neurological, psychiatric, sleep, respiratory, dermatological, pain, and medical comorbidities. Participants with more comorbidities reported significantly more insomnia (OR = 3.79 [3.6, 4.0]), depression (OR = 3.18 [3.0, 3.3]), anxiety (OR = 3.18 [3.0 3.3]), gastric ulcers/gastrointestinal bleeding (OR = 3.11 [2.8, 3.5]), angina pectoris (OR = 2.64 [2.4, 3.0]), and epilepsy (OR = 2.33 [2.0, 2.8]). 75
Symptomatic treatment of CM
This treatment is focused on shortening the duration and severity of acute migraine episodes. With notable exceptions, symptomatic or abortive medicine does not reduce aspects such as the frequency, time, or severity of long-term migraine outbreaks; it should not be used for this purpose. Symptomatic treatments are evaluated in patients with acute headache attacks along the patient’s CM course. Many drugs have not been studied in CM and are only described as tools to manage migraine exacerbation in EM. Unfortunately, the response to symptomatic migraine management is not the same for patients with EM. The alternatives presented are summarized in Figure 1.

Algorithm of preventive pharmacological treatment of CM.
Most effective treatments (traditional and new) in CM for acute headaches associated with nausea, vomiting, and disability
In a meta-analysis that included a group who received step care within attacks (i.e., 800–1,000 mg ASA + 20 mg MTC) as initial treatment for all attacks found that patients who did not respond to treatment after 2 h required treatment with zolmitriptan (2.5 mg). A second group who received step care across attacks (i.e., 800–1,000 mg ASA + 10 mg MCT) demonstrated that patients who did not respond to medication in at least two of the first three attacks switched to zolmitriptan (2.5 mg) for the subsequent three episodes. In a third group (i.e., stratified care), patients with mild headaches were treated with ASA + MCT, while those with more severe headaches were treated with zolmitriptan. The latter two groups had significantly better outcomes than the first, measured by headache response and disability time; however, patients in the stratified group had the highest number of adverse events. 93
A recent study compared the combination of dexamethasone + metoclopramide with intravenous ketorolac, noting a similar response at the end, although it was faster with intravenous ketorolac. In this study, 86 patients were recruited (58.1% males), with a mean age of 37.6 ± 10.3 years. Thirty-five (40.7%) were in the ketorolac group, and 51 (59.3%) were in the dexamethasone + metoclopramide group. Success was defined as a reduction of 3 or more points in pain intensity. At 1 h, the reported pain intensity was 4.7 ± 2.0 in the ketorolac group and 6.2 ± 2.3 in the dexamethasone + metoclopramide group. At 2 h, the pain intensity was 3.4 ± 1.2 in the ketorolac group and 2.9 ± 1.3 in the dexamethasone + metoclopramide group. Thus, the pain reduction time was relatively shorter with ketorolac in acute cases, but the final response was similar between the two groups. 115
Reasons for hospitalizing a patient with CM and MOH and their management. Modified from Raggi et al. 2

Preventive treatment of CM in adults
Most effective treatments (traditional and new) in CM for preventive management
According to the International Headache Society guidelines, preventative treatment should be utilized; it is crucial to avoid overmedication and consider the comorbidities with CM. They emphasize that in trials of preventive treatment of CM, the choice is limited to only two agents that have shown superiority over placebo: TPM and onabotulinum toxin type A (BTA). 143 The alternatives presented are summarized in Figure 2.
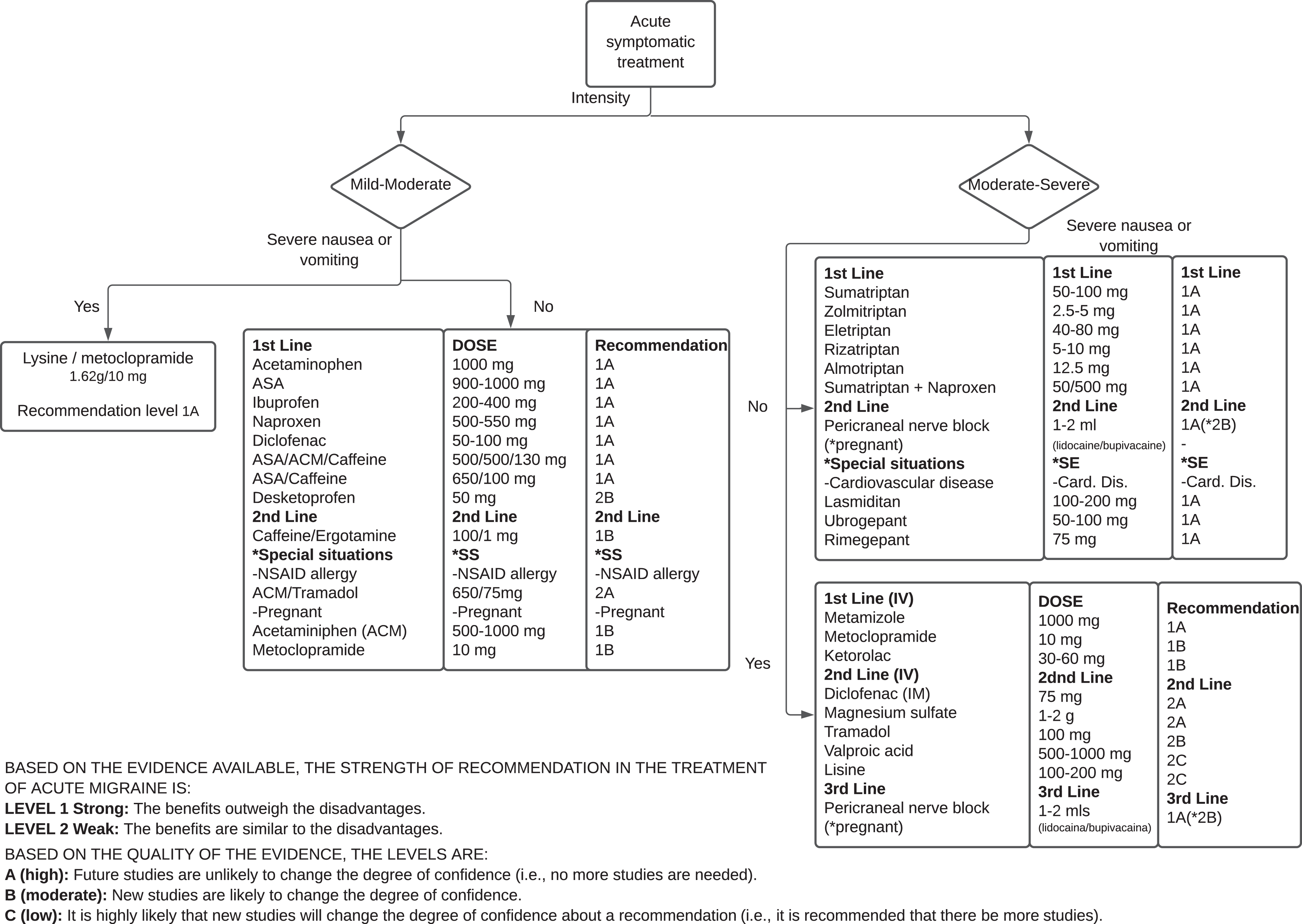
Based on the evidence available, the strength of recommendation in the treatment of acute migraine is: level 1 Strong: The benefits outweigh the disadvantages. level 2 Weak: The benefits are similar to the disadvantages. Based on the quality of the evidence, the levels are: A (high): Future studies are unlikely to change the degree of confidence (i.e., no more studies are needed). B (moderate): New studies are likely to change the degree of confidence. C (low): It is highly likely that new studies will change the degree of confidence about a recommendation (i.e., it is recommended that there be more studies).
Statement: Prophylactic pharmacological management in CM can be with TPM, calcium antagonists, and antidepressants (amitriptyline is widely used and effective, although it does not have enough RCTs to support its use, especially in anxiety and moderate depression). Melatonin can be an alternative, especially in patients with insomnia.
Non-pharmacological treatment of adults with CM
Most effective treatments (traditional and new) in CM for preventive management
Riederer et al. 169 designed a study with 67 patients with at least two migraine attacks per month with a follow-up of 3 months. They found a significant response of 38.5% compared to 12% of patients who received a placebo. Russo et al. 170 confirmed these results in an open study with 24 migraine patients. Finally, Vikelis et al. 171 conducted a study of patients with EM and CM intolerant to TPM. A small but statistically significant decrease in the number of days with headache and the need for acute medication by 3 months was found.
In an open study, Di Fiore et al. 172 assessed 23 patients with CM who received active neurostimulation; 35% reported a good safety profile and satisfaction. In 75% of participants, a reduction greater than 50% was achieved. D’Ostillo et al. 173 conducted a multicenter survey of 2,313 patients with migraines and reported that 53.3% of patients were satisfied with the device with a non-response rate of 18.6%. Importantly, adverse events occurred in 4.3% of the patients, most of which were paresthesia, agitation, or changes in the sleep pattern, and only 2% suspended use of the device.
Neurofeedback
Monoclonal antibodies
Molecularly designed monoclonal CGRP treatments for the preventive management of CM
Silberstein et al. conducted a controlled clinical study with 1,130 participants divided into three groups (the first receiving 675 mg at the beginning and placebo at weeks 4 and 8, the second receiving 675 mg at the beginning and 225 mg at weeks 4 and 8, and the third receiving placebo at the beginning and weeks 4 and 8, subcutaneously). There was no statistical difference between the two groups receiving the active drug; however, the percentage of patients with at least a 50% reduction in the average number of days with headache per month was 38% in the quarterly fremanezumab group, 41% in the monthly fremanezumab group, and 18% in the placebo group. Fremanezumab showed comparable safety to placebo, although application site reactions were common. 208
BTA in the treatment of CM
Application of BTA and anesthetics alone or in combination with other preventive disease management
The painful trigger points found in CM are postulated to result from the excessive release of acetylcholine in the neuromuscular junction and result in tension bands. The peripheral nerves infiltrated in the CM are the major and minor occipital nerves. The local anesthetic drugs used for peripheral nerve blocks are lidocaine, bupivacaine with or without methylprednisolone, dexamethasone, and or triamcinolone reversible inhibition voltage-dependent calcium channels and at low doses selectively on sensitive fibers. 226 Another mechanism proposed in migraine is the nociceptive inhibition of some fibers of the trigeminal-cervical complex. By infiltration in peripheral nerves, inhibition of all systems is achieved. Although the duration of the anesthetic effect is typically in hours, the migraine’s impact could last months, which could be explained by breaking the circle of the pain in CM. The frequency of application varied from 1 to 8 weeks; the marker of success was numbness in the distribution of the blocked nerve, evaluation of the improvement of the headache the day of the application, or improvement in the following days. 227 The effect’s duration is variable; it can be hours to months, most commonly weeks. The vasovagal complications reported are dizziness and pain at the injection site and contraindications include infection, skull defects, allergy to anesthetics, and use of anticoagulants. 228 It is a procedure that, in most cases, is performed in the doctor’s office and is inexpensive.
Migraine and pregnancy
Options for acute and preventive management of CM in pregnancy
In general, drugs to be used in pregnancy are divided into categories by FDA recommendation. Category A is safe (e.g., vitamins and thyroid hormone at substitute doses); Category B demonstrates no evidence of risk in humans or animals, but there are not enough controlled studies for pregnant women; Category C states that the drug has shown risk in animals, but there are no studies in women; and Category D demonstrates that the drug has been shown to cause harm, although the potential benefits may outweigh the risks. A category X would be when the drug is contraindicated in pregnancy. 233,234
For prophylactic management, the use of magnesium, riboflavin, memantine, cyproheptadine, propranolol, and gabapentin are minimally recommended and offer moderate evidence for pregnancy (2B). 246,247
Statement: In pregnancy, discontinuation of drugs of abuse is mandatory. Administration of paracetamol is indicated in exacerbation (although other considerations may be helpful if there is resistance, including opioids). In prevention, TPM, valproate, and candesartan should be avoided. Memantine, riboflavin, propranolol, and gabapentin could be considered if preventive treatment is required. BTA and nerve block could be other alternatives.
Conclusion
The treatment of CM is a challenge and must be individualized, as it considerably affects quality of life. Fortunately, therapeutic options have increased, and we now have molecularly designed drugs to manage symptoms. Comorbid conditions and pregnancy create additional difficulties in care. The development of clinical guidelines can facilitate the best therapeutic approach in the affected population. Importantly, the validity of the current guidelines is likely to be transitory. We believe that 5 years of use is recommended, and we hope to have new possibilities and evidence for managing this problem in the future.
Article highlights
There are risk factors for migraine chronification that should be avoided, especially medication abuse, sleep disturbances (especially OSAS), obesity, asthma, and mood disorders (e.g., depression and anxiety).
Symptomatic management demonstrates an excellent response to NSAIDs and triptans and their combination. Metoclopramide can be used if the migraine is associated with vomiting.
New options for acute management are lasmiditan and gepants.
In prevention, TPM and BTA have good evidence; however, monoclonal anti-CGRP ligands or receptor antibodies are a novel molecularly designed safe alternative with robust evidence.
There are non-pharmacological devices with adequate evidence for the acute and preventive management of CM.
In pregnancy, discontinuation of drugs of abuse is mandatory. For exacerbation, paracetamol can be safely used. Nerve blocks and botulinum toxin are alternatives that need further assessment, but appear safe.
Footnotes
Acknowledgments
We thank María Luisa Díaz García for her support and guidance in the classification and systematization of the created guidelines.
Declaration of conflicting interests
The author(s) declare that there is no conflict of interest. The authors have no personal, financial, or institutional interest in any of the drugs, materials, or devices described in this publication.
Ethical considerations
These guidelines required no written approval from an Ethics Committee, as there was no clinical intervention or direct participation of subjects for this research. Controlled trials for Chronic Migraine should be performed in accordance with the Declaration of Helsinki II.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The meetings and workshops held by the authors when creating these guidelines were financially supported by Allergan, Armstrong Laboratorios de Mexico; Eli Lilly; and Novartis. However, the authors received no financial compensation for the research, authorship, and publication of this article.
Any opinions, findings, and conclusions, or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the pharmaceutical companies stated above.
