Abstract
Background
High-quality research studies on therapeutic approaches for migraine management in pediatrics are scarce. Therefore, we conducted a systematic review of current clinical practice guidelines for the diagnosis and management of migraine in children and adolescents to identify areas of consensus and disagreement, as well as critical gaps, paving the way for future updates and improvements.
Methods
We searched PubMed, WOS, and SCOPUS for guidelines published in the English language in the last 10 years focused on the diagnosis and management of migraine in children and adolescents.
Results
Following a systematic search, eight eligible guidelines were identified. These guidelines were authored by the American Academy of Neurology (AAN), the European Headache Federation/European Academy of Neurology (EHF/EAN), the International Headache Society (IHS), National Institute for Health and Care Excellence (NICE), the Danish Headache Society, and the German Migraine and Headache Society and the German Society of Neurology. There was a broad consensus on considering the International Classification of Headache Disorders-3rd edition as the diagnostic reference. As regards management, paracetamol and ibuprofen were considered first line in the acute management of migraine, with sumatriptan and zolmitriptan nasal sprays rated as second line. Injectable medications for acute management were overlooked in most guidelines. On the other hand, targeted age groups, dosing, and preventive treatment choices showed areas of variability. Various non-pharmacological measures gained attention in all guidelines. Notably, CGRP antagonists and non-invasive neuromodulation techniques were identified as major gaps.
Conclusion
The current systematic review highlights areas of agreement and disagreement between existing guidelines on the management of childhood and adolescent migraine and emphasizes the current gaps and opportunities, thus offering a solid foundation for future studies and guidelines.
This is a visual representation of the abstract.
Introduction
Migraine is a common disabling primary headache disorder in the pediatric population, affecting approximately 10% of children and adolescents between 8–18 years of age (1,2). Frequent migraine episodes can have a considerable impact on the child's quality of life, and may lead to missed school days, disruption of parents’ work and family routines, impairment of social interactions and participation in extracurricular activities (3,4).
The diagnosis of migraine in the pediatric age group can be challenging, especially in younger children, as it relies primarily on a detailed history, often from a parent or an adult carer, and physical examination in the absence of objective tests. Differences in migraine between children and adults include shorter duration of attacks, frequent bilateral location, and gastrointestinal manifestations (5–7).
Management of migraine in children is even more complex and multifaceted. Evidence-based treatment for acute migraine attacks in children is still evolving, with the strongest evidence supporting paracetamol (acetaminophen), ibuprofen, sumatriptan nasal spray (NS) and zolmitriptan NS. Advancement in the understanding of migraine pathophysiology and the introduction of CGRP receptor antagonists in the treatment of adults with migraine are still to be assessed and approved for children (8). Evidence-based preventive treatment for children with migraine is also limited and equally challenging. The management of children with migraine is made even harder by other factors including the heterogeneity of physical and psychological comorbidities, psychosocial factors and triggers, that may underscore the need for individualized, evidence-based approaches and continuing research to optimize treatment (3,4).
The apparent complexity of migraine in children and the dearth of evidence in support of effective treatment make it necessary for national and international organizations to provide appropriate treatment guidelines. The guidelines should aim to synthesize the most robust scientific evidence, largely derived from randomized controlled trials, to formalize treatment algorithms, augment clinical decision-making, and provide clear guidance on the level of evidence supporting the utilization of different drugs in acute and preventive settings (5,9).
Over the past few years, several guidelines and practice recommendations have been published for migraine management in children and adolescents, encompassing the range of treatment aspects including lifestyle modification, triggers management, acute treatment, and preventive strategies (10–13).
Several challenges may limit the scope and value of any treatment guidelines for pediatric migraine. A major challenge is the limited evidence to support many of the preventive medications in children, making it difficult to establish universally accepted evidence-based guidelines on best and safest therapies to offer in this population (14).
Another important challenge is the high and consistent placebo response observed in pediatric migraine trials, which may obscure the true efficacy of active treatments and distort trial outcomes (15). In addition, there is a relatively little knowledge on the effectiveness of preventive treatments across different ages, sexes, levels of development, and patients’ profiles, including comorbidities and migraine subtypes (16).
Indeed, existing clinical practice guidelines for pediatric migraine had a noteworthy limitation. A major concern is the lack of standardization in the grading of evidence and the methodological approaches used for developing recommendations. Such inconsistencies may hinder clinicians’ decision-making and may lead to variability in management practices. Therefore, a systematic review of current guidelines is crucial to delineate areas of consensus, highlight discrepancies, and identify gaps that warrant further investigation.
This work aimed to conduct a systematic appraisal of the quality of recently published guidelines on the diagnosis and management of migraine in the pediatric population, and to compare their key recommendations to identify inconsistencies.
Methods
Systematic search
A systematic search of the literature was carried out to identify all published guidelines for the management of migraine in children and adolescents. The following electronic resources and databases were included in the search: Medline via PubMed, Scopus, and Web of Science. The following search terms were used:
Migraine OR “Status Migrainosus” OR “Sick Headache” OR headache Pediatric OR children OR adolescents OR adolescent OR youth OR youths OR teens OR teen OR teenagers OR teenager Guidelines OR consensus OR recommendations.
The whole search strategy for each database is provided in Online Supplementary Table S1.
Two authors (A.E., M.H.) screened all articles, independently, by title and abstract. The same authors downloaded and read all relevant articles in full text to determine eligibility for inclusion against the prespecified criteria. Conflicts were resolved between the two screeners through discussion. Unresolved conflicts were discussed by all authors.
Inclusion criteria
Articles with recommendations on treatment of migraine in children and adolescents.
Published Guidelines, practice recommendations, or consensus statements for treatment of migraine in children and adolescents.
Articles published in the English language within the last 10 years (between 2015 to September 2025).
Exclusion criteria
Articles that provided recommendations on treatment of migraine, exclusively, on adult patients.
Articles that provided recommendations on headache disorders other than migraine.
Articles that represented personal experience and opinion.
Abstracts of conference presentations and publications in languages other than English.
A critical reading of each article allowed the assessment of the consistency and reliability of the data. Cochrane “risk of bias” assessment was carried out (Online Supplementary Table S2). Although the Cochrane risk of bias tool is primarily designed for evaluating single clinical trials, we applied it to appraise the methodological transparency and potential biases in evidence selection and interpretation within the included guidelines. This approach was implemented to ensure consistency in evaluation, acknowledging that no standardized tool is currently available for assessing bias across clinical practice guidelines
The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) checklist was followed throughout the study.
Results
Search results and guidelines included in the analysis
The systematic review returned 201 articles out of 333, after removing duplicates from all databases; 14 full text articles were sought for retrieval after screening all articles by title and abstract. Finally, eight guidelines were included that represent the most up-to-date international recommendations addressing the management of migraine in pediatric populations (Table 1).
The American Academy of Neurology (AAN) and American Headache Society (AHS) Practice guidelines update summary: Acute treatment of migraine in children and adolescents- 2019 (17) The American Academy of Neurology (AAN) and American Headache Society (AHS) Practice guidelines update summary; Pharmacologic treatment for pediatric migraine prevention – 2019 (18) The Danish Headache Society's Guidelines, 3rd edition- 2020 (19) The European Headache Federation and European Academy of Neurology Guidelines −2021 (20) Guidelines of the German Migraine and Headache Society and the German Society of Neurology – 2022 (21) The International Headache Society (IHS) practice recommendations for acute pharmacological treatment of migraine - 2024 (10). The International Headache Society (IHS) practice recommendations for preventive pharmacological treatment of migraine −2024 (22). National Institute for Health and Care Excellence (NICE) guideline – 2025 (23).
Characteristics of the included guidelines on migraine management in children and adolescents.
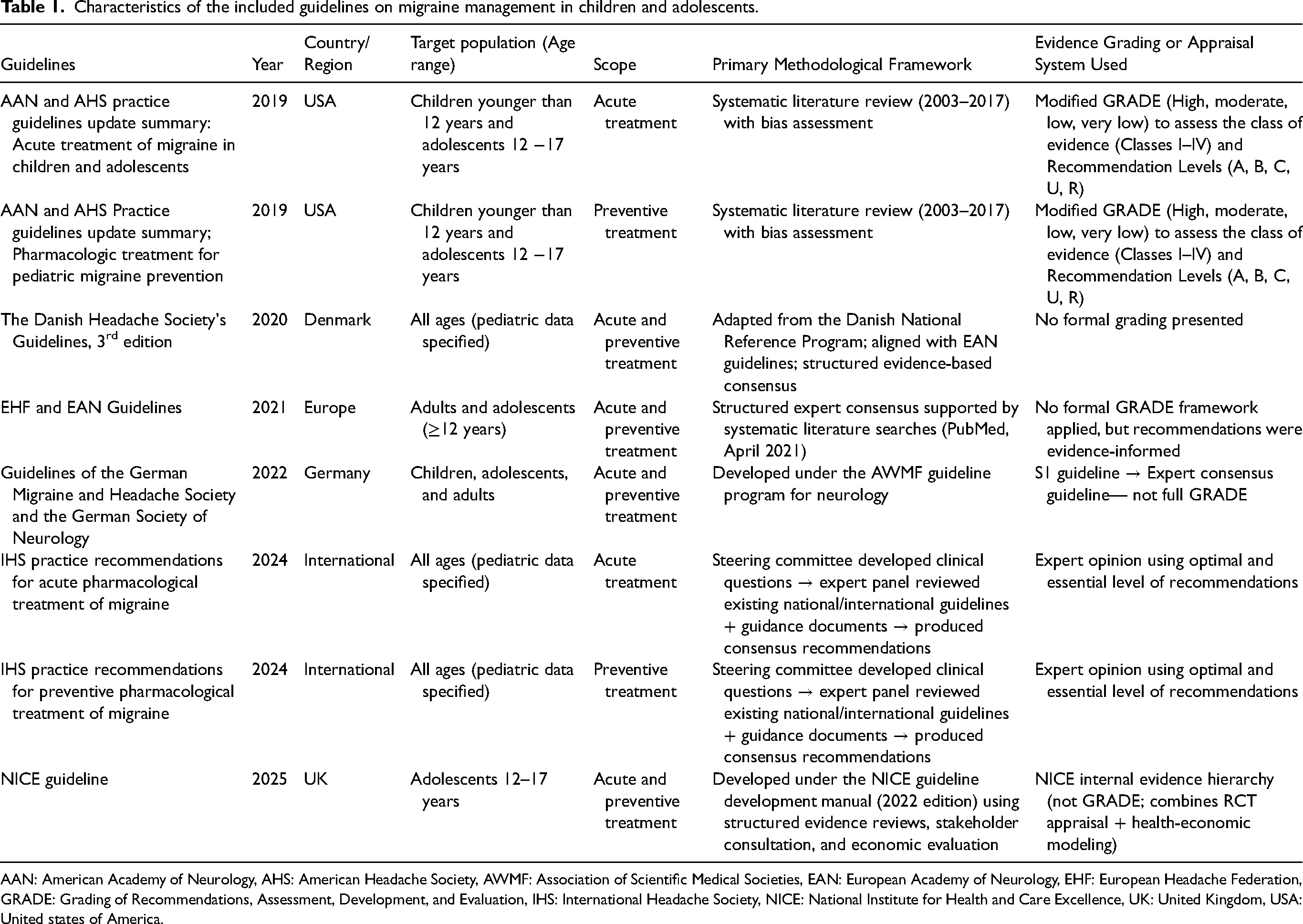
AAN: American Academy of Neurology, AHS: American Headache Society, AWMF: Association of Scientific Medical Societies, EAN: European Academy of Neurology, EHF: European Headache Federation, GRADE: Grading of Recommendations, Assessment, Development, and Evaluation, IHS: International Headache Society, NICE: National Institute for Health and Care Excellence, UK: United Kingdom, USA: United states of America.
Among the eight guidelines, six provided recommendations for the acute treatment and six for preventive treatment of migraine in children and adolescents reflecting both international consensus and regional variations. Figure 1 demonstrates a flow diagram for the search process.

The PRISMA flow diagram for the guidelines included.
Diagnostic considerations in pediatric migraine in the different guidelines
All the guidelines adopted the International Classification of Headache Disorders, 3rd edition (ICHD-3) (24), either in its original or modified form, for the diagnosis of migraine. The AAN guidelines (2019)
Acute management of migraine in the pediatric population
NSAIDs and analgesics
Regarding acute management, ibuprofen remains the most consistently endorsed medication across the included guidelines. The AAN guidelines (2019), the Danish guidelines (2020), the German guidelines (2022), and the IHS practice recommendations (2024) generally recommended a dose of 10 mg/kg, with the IHS practice recommendations (2024) further specifying a maximum daily dose of 30 mg/kg. The AAN guidelines (2019) suggested combining ibuprofen with triptans in cases where the response to triptans alone is inadequate, while the Danish guidelines (2020) emphasized combining it with domperidone. Notably, the Danish guidelines (2020) clarified that other NSAIDs have not been tested in randomized controlled trials involving children or adolescents, whereas the NICE guidelines (2025) highlighted that they can be administered in combination with triptans.
Paracetamol (acetaminophen) was recommended by the Danish guidelines (2020), the German guidelines (2022), the IHS practice recommendations (2024), and the NICE guidelines (2025), mostly at a dose of 15 mg/kg. The Danish guidelines (2020) advised its use in combination with domperidone, while the NICE guidelines recommended its use alongside triptans.
Antiemetics
Concerning antiemetics, the AAN guidelines (2019) did not recommend specific antiemetics in the treatment of migraine. Dopaminergic agents such as domperidone were endorsed by the Danish guidelines (2020), the EHF/EAN guidelines (2021), and the German guidelines (2022), particularly for adolescents older than 12 years and weighing more than 35 kg, at a dose of 10 mg. Domperidone was consistently favored over metoclopramide due to its lower risk of acute extrapyramidal side effects. This recommendation was explicitly highlighted in the Danish guidelines (2020) and the German guidelines (2022). However, metoclopramide was advised by IHS practice recommendations (2024) in cases with nausea or vomiting or in very disabling attacks.
Triptans
Among triptans, sumatriptan and zolmitriptan nasal spray formulations were consistently recommended across all guidelines for adolescents aged 12–17, possibly in combination with paracetamol, ibuprofen, or NSAIDs. Oral sumatriptan/naproxen combination was recommended by the AAN guidelines (2019) and the Danish guidelines (2020), whereas oral zolmitriptan was recommended by the Danish guidelines (2020) and the German guidelines (2022). Subcutaneous sumatriptan was endorsed by the German guidelines (2022) and IHS practice recommendations (2024) as 3rd line option for the treatment of severe, refractory cases.
Regarding other triptans, rizatriptan was recommended by the AAN guidelines (2019), the Danish guidelines (2020), the German guidelines (2022), and the IHS practice recommendations (2024), mostly at a dose of 5–10 mg. Almotriptan was also advised by the AAN guidelines (2019), the Danish guidelines (2020), and the German guidelines (2022) at a dose of 6.25–12.5 mg.
Other acute medications
Intravenous medications such as ketorolac, prochlorperazine, dihydroergotamine, valproate, and magnesium were reserved for severe refractory cases, as noted in the IHS practice recommendations (2024). Buccal prochlorperazine was explicitly endorsed by the NICE guidelines (2025). Notably, the German guidelines (2022) highlighted that ergotamine tartrate is approved for use from the age of 16 years (Table 2).
Comparison between the different guidelines regarding the suggested pharmacological options for the acute management of migraine in children and adolescents.

AAN: American Academy of Neurology, EAN: European Academy of Neurology, EHF: European Headache Federation, IHS: International Headache Society, IV: intravenous, NA: Not addressed in guideline, NICE: National Institute for Health and Care Excellence, NS: Nasal spray, NSAIDs: Non-steroidal anti-inflammatory drugs, ODT: Oral dispersible tablet, OT: Oral tablet, SC: Subcutaneous.
Preventive treatment of migraine in the pediatric population
Preventive pharmacotherapy remains a subject of ongoing debate, largely due to the scarcity of robust evidence in the pediatric population.
Topiramate
Topiramate was acknowledged by the AAN guidelines (2019) as having some supporting evidence, but potential side effects must be carefully discussed with patients and families. In contrast, the Danish guidelines (2020) and the EHF/EAN guidelines (2021) concluded that its efficacy has not been demonstrated in clinical trials, whereas the NICE guidelines (2025) supported its use off-label. The IHS practice recommendations (2024) endorsed its use as a second-line option.
Beta-blockers
A similar pattern is seen with propranolol; the AAN guidelines (2019) suggested its possible efficacy but advised caution and careful consideration of risks and benefits. Conversely, the Danish guidelines (2020), the EHF/EAN guidelines (2021), and IHS practice recommendations (2024) emphasized the limited supporting evidence of its use. The German guidelines (2022) are somewhat more permissive, suggesting doses of 0.25–0.5 mg/kg three to four times daily. The NICE guidelines (2025) advised against its use in adolescents with comorbid depression.
Amitriptyline
Amitriptyline was also considered in several guidelines. The AAN guidelines (2019) highlighted its benefit in combination with cognitive-behavioral therapy (CBT) while also underscoring the need to discuss its side effects. The Danish guidelines (2020) and EHF/EAN guidelines (2021) stated that its effectiveness was not proven in clinical trials. The IHS practice recommendations (2024) recommended its use as a second-line option.
Other preventive agents
Flunarizine was recommended by both the Danish guidelines (2020) and the IHS practice recommendations (2024), although the supporting evidence is still limited. Valproate has demonstrated some preventive effects in controlled trials, as stated by the Danish guidelines (2020), but significant side effects outweighed its benefits.
Frovatriptan and zolmitriptan were uniquely recommended by the NICE guidelines (2025) for adolescents with menstrual migraine, to be taken daily during the perimenstrual period (Table 3).
Comparison between the different guidelines regarding the prophylactic treatment of migraine in children and adolescents.

AAN: American Academy of Neurology, CBT: Cognitive behavioral therapy, EAN: European Academy of Neurology, EHF: European Headache Federation, IHS: International Headache Society, NA: Not addressed in guideline, NICE: National Institute for Health and Care Excellence.
Lifestyle modifications in the pediatric population with migraine
Lifestyle modifications represent a cornerstone of migraine management and were therefore emphasized across most of the guidelines. Counseling and family education were consistently recommended by the AAN guidelines (2019), the Danish guidelines (2020), and the German guidelines (2022). The identification and avoidance of migraine triggers were highlighted by the AAN guidelines (2019), the Danish guidelines (2020), and the German guidelines (2022) as being crucial in migraine management.
Adequate fluid intake, regular meals, following a well-regulated sleep schedule, avoidance of excessive TV/screen time, and identification of possible comorbidities were specifically endorsed by the Danish guidelines (2020). Physical activity was recommended by the Danish guidelines (2020) and the German guidelines (2022). Stress management was highlighted by the Danish guidelines (2020) and the German guidelines (2022). Rest and sleep (during the attack) was specifically recommended by the German guidelines (2022). The AAN guidelines (2019) and the German guidelines (2022) further underscore the importance of avoiding medication overuse. The AAN guidelines (2019) uniquely noted the significance of maintaining a headache diary and promoting migraine-healthy habits. Finally, the need to screen for mood and anxiety disorders was emphasized in both the AAN guidelines (2019) and the Danish guidelines (2020) (Table 4).
Comparison between the different guidelines regarding the recommended lifestyle modifications in children and adolescents with migraine.

AAN: American Academy of Neurology, EAN: European Academy of Neurology, EHF: European Headache Federation, TV: Television.
Non-pharmacological management of migraine in the pediatric population
Non-pharmacological interventions also feature prominently in the guideline recommendations. CBT was universally endorsed by most of the guidelines as a practical approach to migraine management in children and adolescents. Relaxation training and biofeedback were also advocated in the Danish guidelines (2020) and the German guidelines (2022). Notably, acupuncture was uniquely advised by the NICE guidelines (2025) in patients >12 years, whereas local cooling was recommended by the German guidelines (2022) (Table 5).
Comparison between the different guidelines regarding the non-pharmacological management of migraine in children and adolescents.
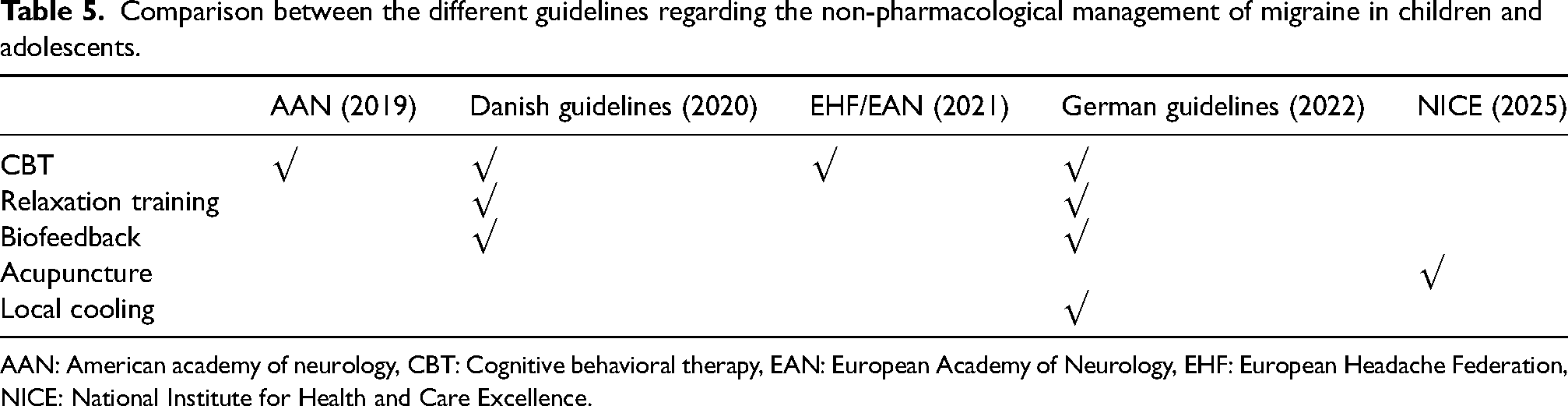
AAN: American academy of neurology, CBT: Cognitive behavioral therapy, EAN: European Academy of Neurology, EHF: European Headache Federation, NICE: National Institute for Health and Care Excellence.
Discussion
This systematic review indicates a broad international consensus on the principles of pediatric/adolescent migraine management, with minor variations in the guidelines regarding pharmacological and non-pharmacological recommendations. These variations are likely based on the level of available evidence, differences in regulatory approvals, and the clinical practice across the different regions (23).
There is a broad consensus across clinical guidelines that ibuprofen and paracetamol should be considered as the first-line treatments for acute migraine in children and adolescents. This is likely because of their proven safety, availability in most parts of the world at low cost, and their use in clinical practice for many years (24–27). There is some evidence from several randomized controlled trials (RCTs) suggesting the efficacy of ibuprofen for pediatric migraine (26,28), while paracetamol, typically seen as safe, has fewer well-designed, controlled trials within this population (26,28,29).
The AAN makes a recommendation for ibuprofen (Level B); however, there was no formal recommendation for acetaminophen, illustrating a conservative, evidence-based approach that follows only pediatric RCTs. In contrast, other guidelines invoke broader clinical experience or extrapolated adult data to provide a rationale for acetaminophen as a first-line agent.
In practice, acetaminophen is a reasonable option, especially in patients with a contraindication or intolerance to NSAIDs. In pediatric populations, further trials would be useful to support its use as a treatment option.
Antiemetics are often used to treat nausea and vomiting, commonly associated with various pediatric conditions, such as acute gastroenteritis (30,31). However, there was insufficient evidence from clinical trials on the use of antiemetics in pediatric migraine patients. (29,32)
The recommendations regarding dopaminergic antiemetic medications supplementing the first-line treatments in the Danish guidelines (2020) were apparently more selective, as the drug was recommended primarily for adolescents over 12 years old and weighing >35 kg to control nausea and vomiting symptoms and improve oral drug absorption. This recommendation was based on safety considerations, such as the potential extrapyramidal side effects (33), which led to the preference for domperidone over metoclopramide, and on pharmacokinetic considerations related to age and body weight (34,35).
Domperidone was also endorsed as a potential adjunct antiemetic by the EHF/EAN (2021) and German (2022) guidelines for adolescents (>12 years old) with prominent gastrointestinal symptoms, reinforcing its potential role in this population.
Notably, the IHS was the only guideline to recommend adding metoclopramide for pediatric/adolescent migraine patients with severe nausea, vomiting, or very disabling attacks.
Taken together, while both agents may be considered in clinical practice for symptomatic relief, domperidone appears more consistently supported across guidelines, whereas metoclopramide should be reserved for select cases due to potential safety concerns.
When considering further options for management of acute migraine attacks, there was a strong consensus regarding triptans, especially sumatriptan and zolmitriptan nasal sprays in adolescents aged 12 to 17 years; these were most often recommended in conjunction with NSAIDs when either treatment alone was insufficient, based on the available evidence suggesting potential additive efficacy (36,37). Rizatriptan and almotriptan were more carefully considered. The AAN (2019), German (2022), and IHS (2025) guidelines allow for their use in adolescent patients, with weight-based dosing recommendations for rizatriptan provided explicitly by the IHS. (Rizatriptan 5 mg if <40 kg or 10 mg if ≥40 kg). It is worth noting that rizatriptan is the only triptan approved by the Food and Drug Administration (FDA) to be used in pediatric patients aged six to 11 years (38). The guidelines show a shift in triptan usage toward acceptance in pediatric/adolescent migraine treatment, with efficacy and safety data emerging. The inconsistent acceptance may be related to the relative lack of strong, large randomized controlled trials assessing efficacy/safety in this age group for rizatriptan and almotriptan versus the longer-established agents sumatriptan and zolmitriptan (39–51).
Based on the available evidence, zolmitriptan and sumatriptan are the most supported triptans for adolescent migraine, whereas newer agents such as rizatriptan and almotriptan are also considered appropriate in selected patients until additional pediatric trials confirm their efficacy and safety in this age group.
For children and adolescents, third-line treatments for acute migraine show some variance between guidelines and are not typically recommended in cases that are not severe or refractory in nature. The German guidelines (2022) have recommended subcutaneous sumatriptan and ergots. In contrast, the IHS guidelines suggest these two options and also mention additional therapies such as IV ketorolac, prochlorperazine, dihydroergotamine, valproate, and magnesium. The NICE guidelines uniquely recommend buccal prochlorperazine. Unresolved differences owing to variations in regional approvals and the limited available evidence guiding treatment in children and adolescents highlight the need for further research to develop robust, evidence-based guidelines.
Clinical trials of preventive treatments for pediatric migraine have shown high responses to placebo. A network meta-analysis conducted in 2020, comparing head-to-head and placebo-controlled trials, found no significant long-term effects for migraine prophylaxis relative to placebo (52). Another double-blind randomized controlled trial conducted in 2017 found that 61% of participants receiving a placebo experienced a 50% or greater reduction in headache days, compared to 52% for amitriptyline and 55% for topiramate. This difference was not statistically significant (53). Based on moderate-quality evidence (53–59). The AAN guidelines recommended that clinicians may consider short-term treatment trials of the preventive medications, topiramate, propranolol, and amitriptyline through shared decision-making for patients who could benefit (Level B).
On the other hand, most of the other guidelines described the supporting evidence for the use of preventive medications as limited. Interestingly, the NICE guidelines, recommended that a course of up to 10 sessions of acupuncture over five to eight weeks can be considered in patients older than 12 who did not benefit from, did not tolerate or had contraindications to propranolol, topiramate, and amitriptyline. These differences suggest that, in the absence of high-quality, pediatric-focused clinical trials, treatment selection in clinical practice is guided by local expertise and safety considerations.
Based on the current evidence, short-term trials of topiramate, amitriptyline, or propranolol may be considered for patients with frequent or disabling attacks. Given the high placebo responses observed in pediatric trials, careful monitoring of efficacy and side effects are recommended.
Although drugs targeting calcitonin gene-related peptide (CGRP) showed efficacy and have received regulatory approval for use in adults with migraine, their testing in the pediatric population is more recent with a study on erenumab showing comparable safety and pharmacokinetics that could help guide future recommendations (60–62).
The European Medicines Agency (EMA) has not yet approved any of the anti-CGPRs for migraine prevention in children or adolescents. On the other hand, the FDA, in 2025, has broadened the indication for fremanezumab for episodic migraine prevention based on the phase 3 SPACE clinical trial (63). The SPACE trial was a large, multicenter, double-blind randomized controlled trial with a low risk of bias, providing Level 1 evidence according to the Oxford Centre for Evidence-Based Medicine (OCEBM), Level B evidence under the AAN classification, and high-quality evidence according to Grading of Recommendations, Assessment, Development, and Evaluations (GRADE). Given the paucity of preventive options for pediatric migraine and the fact that current guidelines predate the publication of the SPACE trial, there is a strong rationale for updating recommendations to incorporate fremanezumab as a standard preventive therapy (63).
To date, published pediatric/adolescent migraine guidelines do not recommend the use of anti-CGRPs. However, a special interest group of the American Headache Society did offer expert opinion-based recommendations for the use of anti-CGRP monoclonal antibodies in post-pubertal adolescents with frequent migraine and moderate to severe migraine-related disability (64,65). The guidelines and recommendations generally agree that pharmacologic treatment should be reserved for children and adolescents who do not improve with non-pharmacological measures and need rapid resolution of the most disabling migraine attacks. This approach is considered appropriate in clinical practice and aligns with current evidence.
Therefore, it may be reasonable to offer CGRP-targeted treatments only for selected adolescent patients experiencing frequent attacks and significant migraine-related disability, broader use should be deferred until additional high-quality pediatric studies confirm efficacy and long-term safety.
Emerging evidence suggested that peripheral nerve blocks, sphenopalatine ganglion (SPG) block, and onabotulinumtoxinA injections are promising, relatively safe adjuncts or alternatives in children/adolescents with migraine, particularly when conventional pharmacologic treatments are ineffective or poorly tolerated. Whereas these interventions are not yet considered first-line treatments in pediatrics, their expanding use highlights the growing need for robust RCTs to establish optimal protocols, evaluate long-term safety, and compare their effectiveness against standard preventive therapies (66–69).
Non-pharmacologic interventions were emphasized across all the pediatric/adolescent migraine guidelines utilized in our review. Counseling, family education, and trigger identification have been frequently offered as recommendations and represent reasonable strategies in pediatric migraine management.
Physical activity is recommended by both the Danish and German guidelines among the non-pharmacological strategies to reduce migraine frequency despite its limited evidence (70–72). On the other hand, the German guidelines suggest rest and sleep during acute attacks to relieve symptoms. These recommendations highlight that children/adolescents with migraine should maintain a balance by staying active most of the time while resting only during migraine attacks to relieve their symptoms.
All reviewed guidelines endorse at least one psychological intervention, such as CBT, mindfulness, relaxation training, or biofeedback. Moreover, some recommendations, such as local cooling, are only mentioned in the German guidelines (2022) and may be based on regional experiences rather than global consensus. Overall, non-pharmacologic therapies target similar goals, but differences in recommendations suggest that these therapies need to be tailored based on individual patient conditions.
It must be mentioned that although nutraceuticals such as magnesium, riboflavin, and coenzyme Q10 are gaining attention as potential therapeutic options in the pediatric age group (73,74), none of the reviewed guidelines formally endorsed their use. This warrants systematic evaluation to determine their possible inclusion in future pediatric migraine guidelines.
It is also worth noting that, although the first FDA cleared and EU approved non-invasive neuromodulation devices for adolescent migraine became available in 2019, these were not included in the reviewed guidelines, which primarily focused on pharmacologic treatments. Rigorous sham-controlled trials in adults have demonstrated favorable safety and efficacy profiles for these non-invasive neuromodulation devices, with evidence comparable in quality to RCTs of pharmacologic treatments (75,76). In contrast, pediatric-specific data remain preliminary and are largely limited to studies focused on safety. This highlights the need for well-designed RCTs in children and adolescents before strong, evidence-based recommendations can be made.
Finally, we suggest that future international guideline development initiatives pay explicit attention to regional regulatory and economic differences with the goal of ensuring that recommendations are evidence-based, applicable worldwide, and with implementation pathways suited to a range of healthcare systems.
Strengths and limitations
A significant strength of this review is that it is the first study to systematically compare different published guidelines for the treatment of pediatric/adolescent migraine and thus provides important insights into the areas of consensus and variation among recommendations.
On the other hand, a considerable limitation of the included guidelines is the substantial variability in their methodological approaches. Whereas some are developed through structured systematic reviews using predefined evidence-grading criteria, others primarily relied on expert consensus without standardized appraisal methods. Such methodological heterogenicity hampers direct comparison between guidelines and can lead to discrepancies in clinical recommendations, which, in turn, may limit the harmonized implementation of evidence-based practices in pediatric migraine management.
Additionally, the guidelines were published at different times, hence some of the differences presented may be due to evolving evidence rather than scientific disagreement. Furthermore, the target populations of available guidelines varied extensively. While some were designed specifically for pediatric and adolescent patients, others span a broader age range. This heterogeneity makes direct comparison among guidelines more complex because differing age groups inevitably influence treatment recommendations, types and degrees of evidence, and consequently the applicability of findings. Other limitations include differences in the regional healthcare systems, availability of drugs, and regulatory approvals (e.g., FDA versus EMA).
It is important to highlight that the design of clinical trials in pediatric populations with migraine remains a critical limitation that warrants careful consideration. Most existing pediatric trials have primarily focused on efficacy outcomes, with relatively insufficient emphasis on tolerability and safety. Indeed, accounting for these factors is crucial to yield more robust, meaningful and clinically relevant evidence. Also, implementation of the recently published IHS Guidelines for Controlled Trials of Preventive Treatment of Migraine in Children and Adolescents in future studies may further help standardize methodology and strengthen the evidence base for pharmacological management in this population (73).
Another important limitation lies in diagnostic uncertainty, particularly among younger children where migraine symptoms may be atypical or insufficiently characterized. Overcoming these challenges requires the adoption of refined diagnostic criteria that account for the developmental stage and pubertal status. This may enhance the reliability of future research and support the generation of high-quality, evidence-based, pharmacological treatment in pediatric population.
Conclusions and future directions
This review provides an overview of the content of current guidelines and recommendations on the pharmacological and non-pharmacological management of migraine in the pediatric population. It identifies some areas of agreement but mostly points to the need for methodologically robust RCTs that properly assess the efficacy of pharmacological and non-pharmacological interventions for migraine treatments in children and adolescents.
Availability of robust comparative data across available migraine treatments would not only strengthen clinical recommendations and allow superior, uniform decision-making but also ensure better patient outcomes and the possibility to address early in life a disease that may persist for decades and become more severe over time. Such data are particularly important considering heterogeneity among responses to therapies across populations, unequal access to drugs, and the economic burden of new or less available therapies. Through integration of these considerations, comparative evidence can help make sure that guidelines are both clinically effective and implementable in different healthcare settings. An update of current guidelines is warranted to incorporate fremanezumab as a recommended option for migraine prevention in children and adolescents.
While this review highlighted fremanezumab as the first CGRP-targeted preventive therapy supported by robust evidence, emerging evidence for other CGRP antagonists and monoclonal antibodies is expected soon. Future guideline updates should therefore consider the broader class of CGRP-targeted therapies alongside other promising interventions such as nutraceuticals, onabotulinumtoxinA, neuromodulation devices, and peripheral nerve blocks based on the evolving evidence. Dedicated systematic reviews assessing these modalities in the pediatric population are warranted to support their potential inclusion in forthcoming guideline revisions.
Beyond identifying current gaps, overcoming the paucity of evidence in pediatric migraine requires coordinated, multi-level strategies. Greater investment in pediatric headache research is crucial, as funding for this area remains disproportionately low relative to its disease burden (74). Regulatory initiatives, such as the Pediatric Research Equity Act, the Best Pharmaceuticals for Children Act in the United States (US), and any other non-US related mandates, should be more rigorously enforced to ensure adequate pediatric trials representation. Furthermore, evidence from clinical registries and electronic medical records can complement traditional RCTs when large-scale pediatric trials are not feasible. Integrating such data into future guideline may accelerate the advancement of high-quality evidence -based effective pharmacological treatment of migraine in children and adolescents.
Article highlights
There is an urgent need for methodologically robust RCTs that properly assess the efficacy of pharmacological and non-pharmacological treatment for migraine in children and adolescents. Upcoming guidelines need to address differences in therapeutic responses, unequal access to medications, and financial challenges. Fremanezumab should be acknowledged in the future guidelines as a preventive treatment option for pediatric migraine
Supplemental Material
sj-docx-1-cep-10.1177_03331024251413283 - Supplemental material for Management of migraine in children and adolescents: A systematic review of guidelines and practice recommendations
Supplemental material, sj-docx-1-cep-10.1177_03331024251413283 for Management of migraine in children and adolescents: A systematic review of guidelines and practice recommendations by Amr Hassan, Alaa Elmazny, Anas Elgenidi, Ahmed Dahshan, Ishaq Abu-Arafeh, Rehab Magdy, Cristina Tassorelli, Mario Fernando Prieto Peres, Francesca Puledda, Aynur Ozge and Mona Hussein in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024251413283 - Supplemental material for Management of migraine in children and adolescents: A systematic review of guidelines and practice recommendations
Supplemental material, sj-docx-2-cep-10.1177_03331024251413283 for Management of migraine in children and adolescents: A systematic review of guidelines and practice recommendations by Amr Hassan, Alaa Elmazny, Anas Elgenidi, Ahmed Dahshan, Ishaq Abu-Arafeh, Rehab Magdy, Cristina Tassorelli, Mario Fernando Prieto Peres, Francesca Puledda, Aynur Ozge and Mona Hussein in Cephalalgia
Footnotes
Acknowledgments
Authors report that the content has not been published or submitted for publication elsewhere. The authors used [Qwen 2.5 max and ChatGPT plus] in order to check writing mistakes and polish the manuscript language. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Author contributions
A.H., A.E., A.E., A.D., M.H. and R.M. participated in collecting scientific material, writing, and helped to draft manuscript. I.A., C.T., M.F.P.P., F.P. and A.O. revised the final manuscript. All authors read and approved the final manuscript.
Data availability statement
Authors report that the datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
