Abstract
Background:
Erenumab, a monoclonal antibody (mAb) developed to block the calcitonin gene-related peptide (CGRP) receptor, is approved for the prevention of migraine in adults. This retrospective observational study sought to describe early real-world use of erenumab.
Methods:
This study used the Practice Fusion ambulatory Electronic Health Record database, which represents approximately 6% of ambulatory care among primary care and specialist practices in the United States. Among migraine patients initiating erenumab, demographics, migraine type, comorbidities, and prior treatments were assessed during the 12-month baseline period. Treatment patterns including changes in acute and preventive medications, switches to other CGRP mAbs (fremanezumab and galcanezumab), and for erenumab, changes in dose and adherence were examined among patients with 6 months of follow-up.
Results:
Of 3,336 patients identified (85.9% female; mean age = 49.1 years), approximately 40% had documentation of chronic migraine. Common comorbidities included non-migraine headache, anxiety, depression, and hypertension. Most patients (76.3%) initiated on the 70 mg dose of erenumab. Among 1,638 patients included in the treatment pattern analysis, 53.1% used acute medications and 55.7% used other non-specific preventive migraine medications during follow-up, reductions of 9.8% and 10.2%, respectively, over the same period of time before index. Switching to fremanezumab and galcanezumab were observed in 12.2% and 13.8% of patients, respectively. The mean proportion of days covered by erenumab at 6 months was 79%. Dosage of erenumab increased (from 70 mg to 140 mg) in 13.0% and decreased (from 140 mg to 70 mg) in 24.9% of patients.
Conclusion:
This early real-world study showed high adherence among erenumab users. This combined with observed reductions in previously used acute and preventive medications are suggestive of erenumab’s benefit.
Introduction
Migraine is characterized by recurrent headache attacks with moderate to severe pain; it is the third most common disease in the world affecting up to 17% of the female and 6% of the male population. 1 –3 Disability due to migraine increases with the number of headache days experienced each month, with chronic migraine patients experiencing greater disability than episodic migraine patients. 4 The key goals of preventive migraine treatments are to reduce attack frequency and severity, to improve function, and reduce disability. 5
Guidelines for the management of people with migraine provide recommendations for both acute and preventive pharmacologic intervention. 5 –8 Expert consensus recommends that preventive pharmacologic treatment should be offered or considered depending on the number of headache days per month and level of headache-related disability. 6,8
Currently available oral migraine preventive medications are associated with low adherence rates due to lack of efficacy and/or poor tolerability. 9,10 A recent retrospective analysis of claims for 8,707 patients showed persistence of 25% after the first 6 months of treatment declining to 14% by 12 months, for the 14 most common medications used for migraine prevention. 11
Erenumab (erenumab-aooe in the United States [US]) was the first in a new class of monoclonal antibodies (mAbs) targeting the calcitonin gene-related peptide (CGRP) pathway and was approved in the US by the Food and Drug Administration in May 2018 for migraine prevention. 12,13 The efficacy and safety of erenumab is supported by a number of randomized clinical trials. 14 –22
A number of studies have reported on the real-world effectiveness for erenumab at 2, 3, and 6 months. 23 –28 However, there is little reported information on treatment patterns including adherence on erenumab. In addition, the real-world studies to date have all included relatively small sets of patients from a small number of headache centers. This is the largest study in the US that includes patients nation-wide from both specialty and primary care practices to describe characteristics and treatment patterns including adherence in patients initiating erenumab. The objective of this retrospective observational study was to provide descriptive information on real-world use of erenumab for patients seen in routine US clinical practice including demographic and clinical characteristics as well as treatment patterns including sequencing of treatments and treatment adherence.
Materials and methods
Study design and data source
We used de-identified data from the Practice Fusion electronic health record (EHR) database during the time period from May 2017 to September 2019. The Practice Fusion EHR database is sourced from clinical information entered through the Practice Fusion ambulatory EHR platform (Practice Fusion, San Francisco, CA, USA) and is currently in use at over 25,000 clinical sites, mostly single-provider or small group practices, in all 50 US states, representing approximately 6% of ambulatory care among primary care and specialist practices in the US. We consider this to be comparable to the US health care population in terms of age, gender, and geographic location. 29 Key data elements captured by the system include demographics, prescription data (National Drug Codes), diagnosis data (International Classification of Diseases, Ninth/Tenth Revision, Clinical Modification [ICD-9-CM/ICD-10-CM] codes), laboratory test results, physician characteristics, office visits, and vitals. Study parameters were drawn from information entered into the patient electronic medical chart as part of routine care and included provider specialty, patient demographics, migraine diagnosis type, comorbidities, and migraine therapy dose and timing. Data from these practices are made available for research in the form of a certified Health Insurance Portability and Accountability Act (HIPAA)–compliant de-identified research database. As a noninterventional, retrospective database study using data from a certified HIPAA–compliant de-identified research database, approval by an institutional review board was not required.
Study population
We included adult patients (≥18 years) who had a diagnosis of migraine (ICD-9-CM/ICD-10-CM: 346.××/G43.××) or evidence of having migraine (1+ prescription record for an acute migraine-specific medication [triptans, ergotamines] listing 1 or more refills, or 2+ prescription records for an acute migraine-specific medication [triptans, ergotamines], without refills, within 365 days of one another) and had at least one prescription order for erenumab entered into the source EHR platform from May 2018 through March 2019 (the first 11 months that erenumab was available on the US market). The date of first erenumab prescription was defined as the index date. Additionally, patients were required to have an outpatient visit (in-person office visit, home visit, telemedicine, etc.) captured in the EHR system at least 1 year prior to their index date to establish a baseline period, which was considered to be the 12-month period prior to erenumab initiation and any time prior to erenumab initiation for diagnosis and comorbid conditions.
Outcome measures
Baseline characteristics included patient demographics, provider type, number of outpatient visits in the 12 months prior to the index date, migraine type (chronic; identified with ICD-9-CM codes 346.7x and ICD-10-CM codes G43.7x), initial dose of erenumab (70 mg or 140 mg), select comorbidities that are of interest in the migraine population (24 conditions), prior CGRP mAb use (fremanezumab and galcanezumab) and the mean number of days it took to switch to erenumab, use of acute medications to treat migraine (triptans, ergotamines, nonsteroidal anti-inflammatory drugs [NSAIDs], and opioids), and use of other non-specific preventive migraine medications (non-topiramate anticonvulsants, topiramate, antihypertensives, botulinum toxins, antidepressants, and other preventive agents [carisoprodol, cyproheptadine, guanfacine, memantine, methysergide, milnacipran, and tizanidine]). The total number of acute and preventive medications during baseline were also reported as categories (0, 1, 2, 3 or more different medications). Use of acute and other non-specific preventive migraine medications was based on either prescription orders written or medication use documented in the patient’s medical record.
Patients included in the treatment pattern analysis were required to have an outpatient visit or prescription captured in the EHR system at least 6 months after their index date. Acute and preventive medications used to treat migraine were captured in the 6-month period following index and were compared to use of these medications in the 6 months prior to the index. Drug switches from erenumab to other CGRP mAbs along with the number of days it took to switch, were captured during the follow-up period. Dose changes in erenumab (from 70 mg to 140 mg or 140 mg to 70 mg) were classified by the first observed change in erenumab dose prescribed during follow-up. The proportion of days covered (PDC) was used to assess adherence to erenumab at 3 months and at 6 months post-index. The PDC, often reported as a percentage truncated at 100%, was calculated as the days supply of medication dispensed to the patient (initial prescription and all subsequent refills combined) divided by the total number of days in the assessment period. 30 The PDC is commonly used in electronic database studies and has been endorsed by the National Quality Forum as a tool to measure health care quality. 31
Statistical analysis
Baseline demographic and clinical characteristics, treatments received, sequence of treatments, dose of erenumab, and adherence to erenumab at various time points were described by frequencies and percentages for categorical variables and means with standard deviations for continuous variables.
Results
Study population
Of 6,090 patients with a written prescription for erenumab, 3,336 patients met the study inclusion criteria; the majority of ineligible patients were excluded because they did not have at least one outpatient visit at least 365 days prior to erenumab initiation to establish a baseline period (Figure 1). Among the 3,336 eligible patients, 1,638 (49.1%) had at least 6 months of follow-up and were included in the 6-month treatment pattern analysis.

Patient inclusion criteria. *Follow-up defined by having an outpatient visit or a prescription 6+ months after index.
Baseline characteristics
Of 3,336 patients meeting study inclusion criteria, 85.9% were female and the mean age was 49.1 (standard deviation [SD]: 13.0) years, with 63.8% between the ages of 40 and 64 (Table 1). Geographically, the patient distribution was similar to that of the overall US population with lower representation of the Midwest region and slightly more representation in the Northeast, South, and West regions. Chronic migraine was in the diagnosis history (any time prior to the index date) for 39.7% of patients. Most patients (81.1%) had 3 or more outpatient visits recorded in the EHR during their 12-month baseline period with a mean of 7.5 (SD: 9.4) outpatient visits per patient.
Demographic and clinical characteristics of patients.
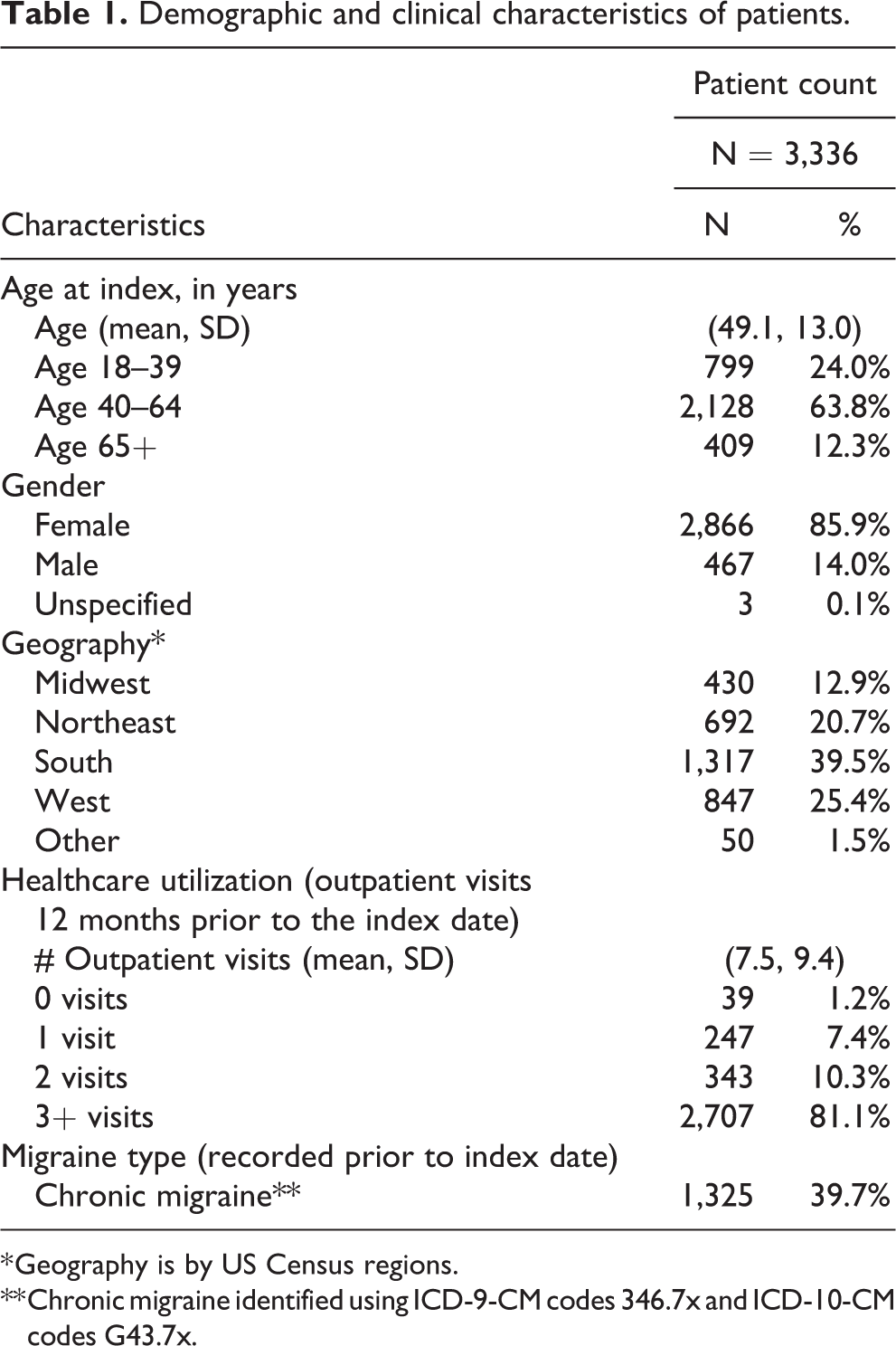
* Geography is by US Census regions.
** Chronic migraine identified using ICD-9-CM codes 346.7x and ICD-10-CM codes G43.7x.
Over half of patients were first prescribed erenumab by neurologists or pain specialists (63.2%) with most of the remaining patients being prescribed erenumab by primary care physicians (33.0%) (Table 2). Most patients (76.3%) initiated on the 70 mg dose of erenumab. A small number of patients switched to erenumab from another CGRP mAb prior to the erenumab index date: 19 (2.1%) switched from fremanezumab after a mean of 54.1 days and 7 (0.8%) switched from galcanezumab after a mean of 30.1 days.
Initial erenumab prescription summary of patients.
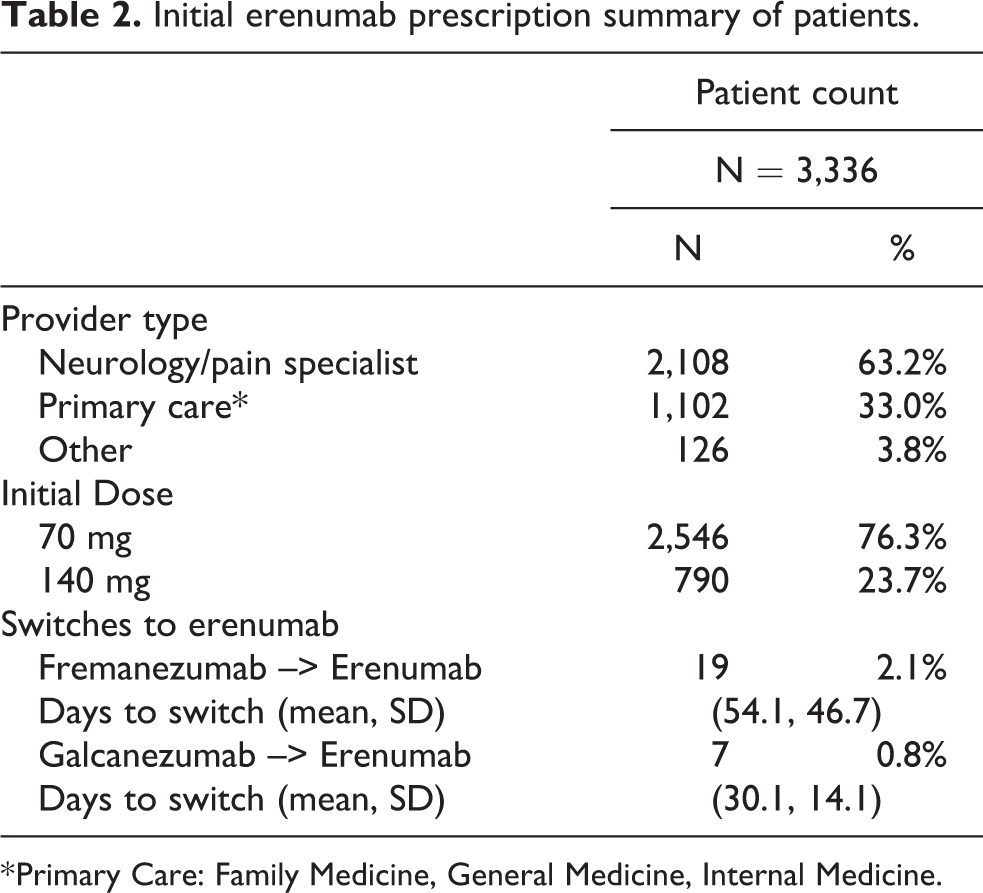
* Primary Care: Family Medicine, General Medicine, Internal Medicine.
Of 24 comorbid conditions selected for review and recorded prior to erenumab initiation, the five most frequent were other non-migraine headache (31.4%), anxiety (24.9%), depression (21.0%), hypertension (17.1%), and osteoarthritis (11.8%) (Table 3).
Comorbidities* of patients.

* Recorded at any time prior to index.
Use of both acute and non-specific preventive medications for migraine were common among study patients in the 12-month baseline period. Acute medications (triptans, ergotamines, NSAIDs, opioids), were noted among 2,367 patients (71.0%) and non-specific preventive medications (non-topiramate anticonvulsants, topiramate, antihypertensives, botulinum toxins, antidepressants, other) were noted among 2,427 patients (72.8%) (Table 4). Nine hundred ninety-eight patients (29.9%) had one prescription for a non-specific preventive medication, 742 (22.2%) had two, and 687 (20.6%) had three or more prescriptions for non-specific preventive migraine medications in the baseline period. Triptans (53.2%) were the most common acute medication and anticonvulsants (including topiramate; 56.5%) were the most common preventive medication.
Acute and other non-specific preventive migraine medications in the 12 months prior to first erenumab prescription.

Follow-up characteristics
In the analysis restricted to the 1,638 patients with at least 6 months of follow-up, use of acute and other non-specific preventive medications for migraine in the follow-up period was less frequent compared to the baseline period. Prescriptions for acute medications in the 6-month follow-up period were noted among 869 patients (53.1%), a reduction of 9.8% compared to that in the 6-month baseline period (Table 5). Prescriptions for other non-specific preventive migraine medications in the 6-month follow-up period were noted among 913 patients (55.7%), a reduction of 10.2% compared to that in the 6-month baseline period. Prescription of 3 or more non-specific preventive migraine medications was noted among 157 patients (9.6%), a reduction of 24.5% compared to that in the 6-month baseline period. Use of all acute and preventive medications for migraine decreased from the 6-month baseline period, with triptans (37.4%) remaining the most common acute medication and anticonvulsants (including topiramate; 35.3%) remaining the most common non-specific preventive migraine medication.
Acute and other non-specific preventive migraine medications in the 6 months prior to and 6 months following first erenumab prescription among patients with 6 months of follow-up.

Treatment patterns
Among patients with 6 months of follow-up, mean PDC was 86% at 3 months and 79% at 6 months (Table 6). The majority of patients (1,103, 67.3%) initiated erenumab on the lower 70 mg dose while 535 (32.7%) initiated erenumab on the higher 140 mg dose. Among patients initiating on the 70 mg dose, 143 (13.0%) increased their dose to 140 mg after a mean of 95 (SD: 62) days, and among patients initiating on the 140 mg dose, 133 (24.9%) decreased their dose to 70 mg after a mean of 196 (SD: 78) days. Switching from erenumab to one of the other two CGRP mAbs during the follow-up period was similar with 200 patients (12.2%) switching to fremanezumab after a mean of 138.0 (SD: 92.9) days and 226 patients (13.8%) switching to galcanezumab after a mean of 202.4 (SD: 92.0) days.
Erenumab treatment pattern in patients with 6 months of follow-up.

Discussion
In this real-world study, we identified more than 3,000 patients prescribed erenumab and meeting our study inclusion criteria. Patients were predominantly female with an average age of almost 50 years, and over a third had a diagnosis of chronic migraine. The most common comorbidities identified were other non-migraine headache, anxiety, depression, and hypertension. Prior to initiating erenumab, patients were prescribed a number of acute and preventive medications. Just under half of the patient population had 6-month follow-up data for the treatment pattern analysis. For these patients, reductions in acute and preventive medications, along with high adherence, suggest that erenumab may have benefits.
In relation to other studies, the profile of patients in our study is similar to that seen in other early reports of erenumab users, that is a predominantly female population aged in their mid to late 40 s with recognized migraine comorbidities including anxiety and depression. 23 –25,27,28,32 Compared to a US center-based chart review and early administrative claims-based analysis the proportion of patients with a recorded diagnosis of chronic migraine was substantially lower, a finding that is not surprising given that our data comes from single providers or small group practices. 32,33 It is important to note that patients in real-world studies examining erenumab, including ours, have tended to be a bit older with a larger proportion of females than migraine patients included in both the American Migraine Prevalence and Prevention (AMPP) and Chronic Migraine Epidemiology and Outcomes (CaMEO) studies of migraine in the US population. 34,35 This is most likely due to the use of a new class of preventive medication being provided to patients with a longer history of migraine or more severe migraine that may be more refractory to previously available options. For the same reason it is not surprising that patients in the current study were also more likely to have chronic migraine, 39.7% compared to 6.6% in the AMPP and 8.8% in the CaMEO studies. 4
Adherence to erenumab in our study was high with reported measures of PDC of 86% at 3 months and 79% at 6 months. These measures of adherence are generally comparable to that seen in other studies. A study of erenumab adherence using US pharmacy claims data reported a mean PDC of 72% at 3 months. 36 Additionally, a study in Italy reported that among patients with 6 months of follow-up, 85.4% received 6 doses of erenumab, while a US study reported that 60.5% of patients received all 6 doses during a 6-month follow-up period. 25,27 Patients, for the most part, appear to be adherent to the recommended monthly dosing regimen. In the present study, much of the lack of adherence to erenumab was due to patients who discontinued erenumab by switching to other CGRP mAbs as they became available on the US market. As a result, the in-class adherence would be higher suggesting that providers and patients are willing to try multiple products in seeking what works best; however, we lacked information on reasons for discontinuation which would have provided additional insight on both efficacy and tolerability of erenumab. A common finding in early reports is the use of the lower 70 mg dose for the majority of patients with some evidence of dose titration. 24,25,27
Finally, our finding of reductions in acute and preventive medications after initiating erenumab is also congruent with a large US study using pharmacy claims data. 36,37 Reductions in medication usage after erenumab initiation could be viewed as a proxy for migraine improvement and suggests a benefit of erenumab, which is consistent with findings from clinical trials and real-world studies. 14 –22,24 –28
The strengths of our study include its relatively large sample size, 6-month follow-up availability, and use of data sourced from a large EHR system used in all 50 US states. The database also includes patients covered by public and private insurance as well as patients who have no insurance. Minimum inclusion and exclusion criteria were applied to keep study results representative of as large a portion of the migraine population as possible. Our study also has important limitations. We relied on data collected as part of routine clinical care through a single EHR network that is made up primarily of small, independent practices, and as such results may not be generalizable to larger care settings. There may be misclassification of medication use as medications prescribed outside of these practices may not be recorded. For instance, our analysis showed that 27.2% of migraine patients had used 0 preventive medications and 29.9% had used 1 preventive medication in the year prior to the first erenumab prescription despite the fact that erenumab is generally prescribed to migraine patients who have failed multiple migraine preventive treatments. Moreover, medications that are prescribed may not necessarily be dispensed and then actually used by patients. We were also unable to include data on patient-reported outcomes such as monthly migraine days or migraine severity because these were not consistently available before and after first erenumab use. In addition, it is unclear how the design and use of patient access programs (ie, financial assistance sponsored by pharmaceutical companies to cover the entire prescription cost or some of the patient’s out-of-pocket costs) may have impacted use and adherence to erenumab and switching between CGRP mAbs.
Despite the insights provided by this and other real-world studies to date, there remain important unanswered questions. Results reporting changes in patient-reported outcomes from larger patient populations is an obvious gap in current evidence. In addition, insights from studies with longer follow-up and additional information on changes in healthcare resource utilization and costs is also needed.
Conclusion
In this early look at US clinical practice, the majority of patients prescribed erenumab were women, who had received several acute and preventive medications for migraine prior to the initiation of erenumab, with more than a third having chronic migraine. Most patients were adherent to recommended monthly dosing. This, combined with reductions in the use of acute and preventive medications, may be suggestive of an erenumab benefit; however, more data on patient-reported outcomes and healthcare resource utilization, with longer follow-up, is needed.
Clinical implications
Erenumab use in US clinical practice appears to be focused on migraine patients that have had inadequate response to previous migraine management options
Generally, high adherence to erenumab and reductions in previously used acute and preventive medications may be suggestive of an erenumab benefit
More research is required to better understand real-world effectiveness especially impacts on patient-reported outcomes
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A. Bogdanov, L. Fischer, and L. Kallenbach are employees of Veradigm Health, under contract with Amgen Inc. for the conduct of the study. V. Chia, R. Urman, and S. Rasmussen are employees and shareholders of Amgen, Inc. M. Bensink is a previous employee and current shareholder of Amgen Inc. now working as an independent consultant and receiving consulting fees from Amgen, Inc. C. Szekely was employed by Amgen, Inc. at the time this work was completed and is a shareholder of Amgen, Inc.
Ethics or institutional review board approval
This retrospective observational study used de-identified data from the Practice Fusion EHR database. Patient consent was not required as data were collected and entered into the EHR platform as part of usual patient care. The resulting EHR data is made available for research in the form of a certified Health Insurance Portability and Accountability Act (HIPAA)–compliant de-identified research database. As a noninterventional, retrospective database study using data from a certified HIPAA–compliant de-identified research database, approval by an institutional review board was not necessary.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Amgen, Inc.
