Abstract
Background:
Relationships of adipokines (ADP) with migraine are not well-established. We examined the relationship of adiponectin and leptin with migraine by aura symptoms.
Methods:
In a baseline cross-sectional data of Brazilian Longitudinal Study of Adult Health (ELSA-Brasil), associations of ADP levels and migraine were assessed in a sample of 257 nondiabetic subjects, free from cardiovascular disease. Associations of ADP tertiles (dependent variable) and migraine status were tested using logistic regression models. Categories of migraine were created as follows: no headache (reference), migraine with aura (MA), and migraine without aura (MO) in all sample and by sex. Odds ratio (OR) with respective 95% confidence interval (CI) adjusted for age, sex, body mass index, and metabolic syndrome.
Results:
Among participants (46 years ± SD: 4.8), 47.5% were women and 36.2% had migraine (16.7% MA). Median values of leptin (ng/mL) increased gradually across subgroup: no headache: 9.5 (interquartile range (IQR): 5.5–15.7) versus MO: 17.0 (IQR: 10.9–34.2) versus MA: 20.9 (IQR: 11.7–29.3), overall p value <0.0001, but not for adiponectin levels. After full adjustment, the third of leptin was positively associated with MA (OR 2.89 (1.00–8.4)) and the second of adiponectin was associated with MO (OR 2.76; 95% CI: 1.09–6.96, p = 0.03). Positive associations with MA, second (OR 3.81; 95% CI: 1.07–13.59; p = 0.04) and third tertile of leptin (6.54; 95% CI: 1.74–24.57, p = 0.005), were also observed in women, but not in men.
Conclusions:
Positive associations between ADP and migraine, particularly between MA and leptin levels in women, raise the possibility of adipocytokines and play a role in migraine pathophysiology.
Introduction
It is estimated that more than a billion people suffer from migraine wordwide. 1 In female sex, headaches are most burdensome and migraine affects approximately 20.3 million of young and middle-aged women. 1 Considering the marked migraine-related disability particularly in working-age populations, deepening the knowledge about underlying factors is needed. The link between obesity and migraine has been previously reported showing controversial results. 2 –11 Most studies that reported a positive association between obesity and migraine were performed in women. 2 –7 Particularly in the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil) study, elevations in body adiposity were directly associated to increased risk for daily migraine, being >2.5 in the body mass index (BMI) category of 35.0–39.9 kg/m2, regardless of age, sex, educational level, race, and the use of migraine prophylaxis. 11
The association between obesity and migraine suggested that low-grade inflammation may be linked to these conditions. Further evidence supported that adipokines (ADP) produced by adipose tissue, such as adiponectin and/or leptin, could be mediating factors. 12,13 A recent systematic review has reinforced a role for leptin and adiponectin in the pathophysiology mainly of the chronic migraine. 14 Classically, leptin is increased in obesity, while adiponectin is decreased. 12,13 However, both have been associated with migraine episodes, particularly in those with aura in which inflammatory component is present. High leptin levels might trigger the cortex susceptibility to cortical spreading depression explaining aura symptoms. 13 Meanwhile, the role of adiponectin in inflammation is complex and dependent on its isoform. The low molecular weight (LMW) oligomer has been related to anti-inflammatory while the high molecular weight (HMW) oligomer to pro-inflammatory effects. Evidence suggests that the latter can participate in migraine pathophysiology. 13 Despite findings supporting relationships between migraine, particularly chronic migraine, and certain ADPs 14 –21 , some discrepancies persisted in more recent studies that have evaluated the relationship between leptin, adiponectin, and migraine according to aura symptoms in their analyses. 17,19,21 From those, only one found a positive relationship between migraine with aura (MA) and leptin levels. 19 The other two studies, including a case–cohort study that included the investigation between MA symptoms and HMW adiponectin, 17 found no associations between ADPs and MA. 17,21
As far as we know, no study has investigated the association of ADP and migraine according to aura symptoms stratified by sex in an admixture population in Latin America as the ELSA-Brasil. Of note, in the ELSA-Brasil, the 1-year prevalence of migraine is relatively high 22 representing a unique opportunity to explore whether the levels of ADPs are associated with the presence of aura. Thus, we aimed to evaluate the relationship between adiponectin and leptin with migraine according to aura symptoms in all sample and by sex.
Methods
Study population and design
ELSA-Brasil is an ongoing prospective cohort of 15,105 civil servants aged 35–74 years performed in six state capital cities in Brazil. Baseline assessment occurred between 2008 and 2010. Further details of this cohort are described elsewhere. 23 –25 In brief, all active or retired civil servants of six academic institutions were eligible for the study. Exclusion criteria were current or recent pregnancy, intention to quit working at the institution, severe communication impairment, and residence outside of a study center’s metropolitan area. Participants were selected according to occupation classified as unskilled, technical/clerical, and faculty and professional staff permitting a gradient of socioeconomic position across the sample. 25 Initially, all eligible individuals were invited to an interview at the workplace for answering standard questionnaires, and further to visit the research center for clinical exams according to standard protocols. 23 –25
A random sample of 1000 individuals without diabetes and cardiovascular disease aged between 35 years and 54 years was drawn from 5061 participants of the ELSA-Brasil baseline in São Paulo Center to investigate associations of biomarkers of atherogenesis. 26 Of them, 872 participants had adiponectin and leptin levels determined. Fifty-two participants were excluded due to the use of lipid-lowering medication. From 820 participants, we ended up with 630 participants with definite diagnosis of migraine (n = 102) and no-migraine (n = 528). We further excluded four cases without information regarding aura symptoms, leaving 626 cases (migraine = 98 and no-migraine = 528). In order to eliminate some bias due to the possibility of increased ADP levels among individuals with high frequency of migraine as previously reported by other studies, 20,21 we excluded five participants who reported daily migraine. Of note, the frequency of migraine was classified into five groups (1 = once in a while; 2 = once or twice a month; 3 = once a week; 4 = more than once a week; 5 = daily). However, we did not exclude the group 4 (n = 22) because we might have both chronic and nonchronic cases in it.
From 621 participants, we excluded individuals with other headaches (n = 364) from the reference group to better evaluate the relationship between ADP, MA, and migraine without aura (MO). Thus, the final sample was composed of 257 participants (164 controls without headache, 50 with MO, and 43 with MA) with available data to be included in the present analyses (Figure 1).
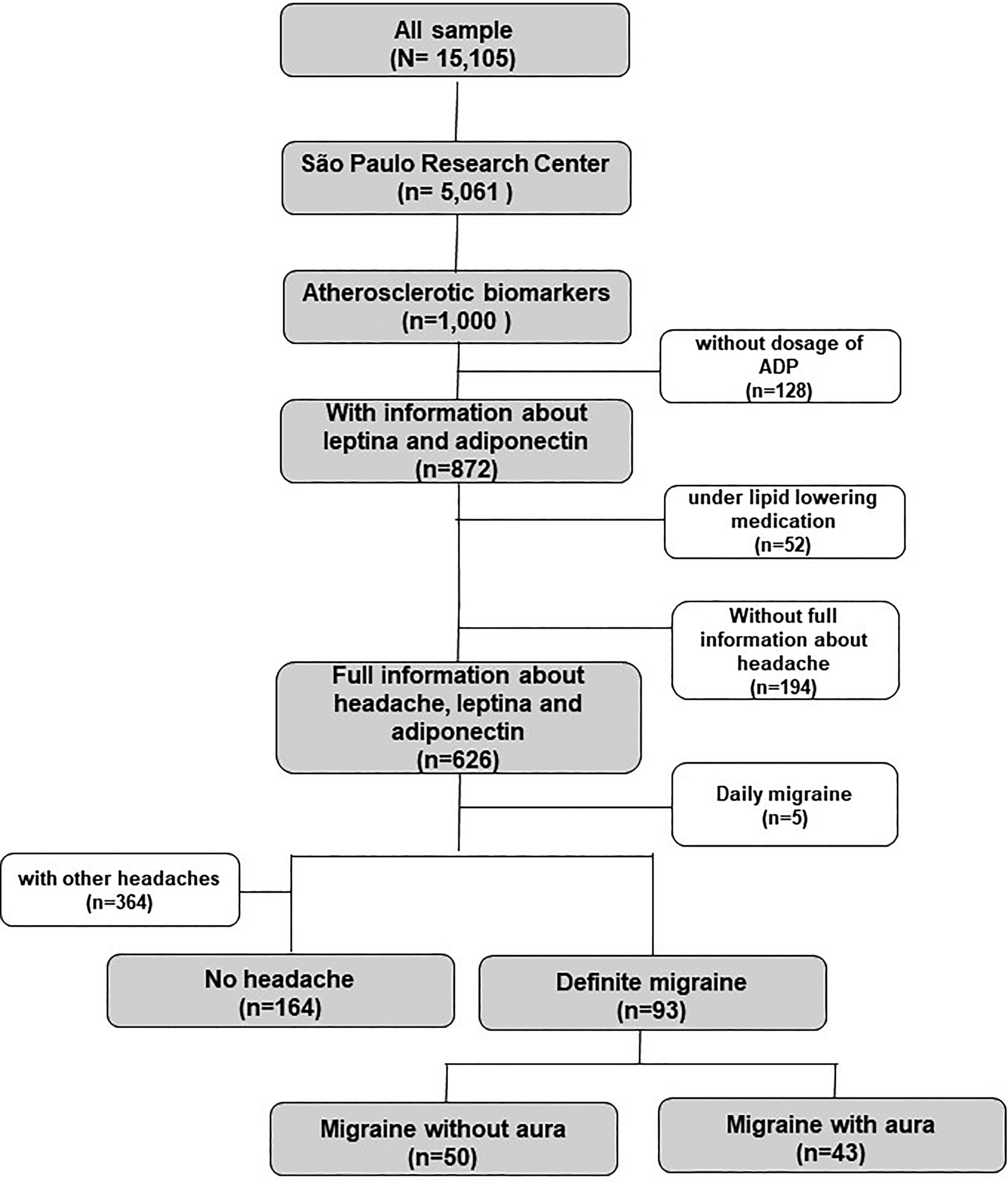
Casuistic from the ELSA-Brasil study. ELSA-Brasil: Brazilian Longitudinal Study of Adult Health.
The institutional ethics committee approved the study, and written consent was obtained from all individuals.
Analytical procedures
The blood samples were collected on the day the participant came to the research center to answer questionnaires and underwent several measurements such as anthropometrics, blood pressure (BP), electrocardiogram (ECG), pulse wave velocity, heart rate variability, and ultrasonography. Participants were advised to book another date if in the 24 h prior to the visit to the research center he/she presented any symptoms such as fever, any pain (including headaches), dizziness, and others. In the research center, the participants were asked again about the presence of symptoms. If the participant presented any symptom, a new date was booked. Therefore, no participants with headache underwent blood sample collection.
Venous blood samples were obtained 12 h after an overnight fast. Plasma glucose was determined by the hexokinase method, and HDL-cholesterol by homogeneous colorimetric, without precipitation (ADVIA Chemistry; Siemens, Deerfield, Illinois, USA). LDL-cholesterol was calculated using the Friedewald equation. When triglyceride concentration was greater than 400 mg/dL, the LDL-cholesterol level was directly measured. 27 Immunochemistry was used to measure high-sensitivity C-reactive protein (Dade Behring; Siemens, Marburg, Germany) and enzyme-linked immunoenzymatic assay for insulin (Siemens, Tarrytown, New York, USA), adiponectin, and leptin levels (Enzo Life Sciences, Farmingdale, New York, USA). Interleukin-6 (IL-6) were determined using the Bio-Plex® Pro Human Cytokine panel (Biorad, São Paulo, SP, Brazil). Intraassay and interassay coefficients of variation ranged from 1.8% to 7.2% and from 0.9% to 14.4%, respectively. Of note, our kit dosed the total level of adiponectin without differentiating its isoforms. 26 For adiponectin, the three outliers were concentrated in the reference group. For leptin, the few outliers (n = 2) were concentrated in the subgroups of MO and MA.
Migraine definition
All participants who answered “yes” to the question “In the last 12 months, did you have a headache?” during the ELSA-Brasil evaluation were invited to answer a detailed headache questionnaire based on the International Classification of Headache Disorders (ICHD-2nd) that was adapted, validated, and previously used in Brazil. 28,29 Briefly, it investigates pain frequency, duration, quality, location, intensity, triggering, and accompanying symptoms, such as nausea or vomiting. We classified individuals who answered “yes” to the question about headache and fulfilled all criteria for migraine as definite migraine. We classified individuals who answered “yes” to the question about headache and fulfilled all criteria for migraine but one as probable migraine.
All participants of the study with a migraine headache identified by the headache questionnaire in the research center of São Paulo (N = 1419) were selected for a telephone interview to investigate aura symptoms. The telephone interviews were conducted by the same medical doctor (ACG) with great expertise in headache classification, including aura symptoms, based on the ICHD-2nd. We classified individuals according to IHS (codes 1.1: MO or 1.2: MA). The reference group included participants with no headache during the last 12 months. Also, we considered in for present analyses only participants diagnosed as having definite migraine.
Finally, we collected information about prophylactic medications for migraine based on the American Academy of Neurology guideline as follows: level A: divalproex sodium, sodium valproate, topiramate, propranolol, metoprolol, and timolol and level B: amitriptyline, venlafaxine, atenolol, and nadolol. 30
Other study variables
Interview and examination were performed by trained personal with strict quality control. 25 Questionnaires addressed age (median and interquartile range (IQR)), years of education (up to incomplete high school, high school, or more than high school), self-defined race (White, Brown, Black, and others—Asian and Indigenous), smoking status (never, former, and current), and physical activity at leisure time (low, mild, or vigorous) using the International Physical Activity Questionnaire–Long Form. BP was taken using a validated oscillometric device Omron HEM 705CP INT (Omron Corporation, Kyoto Head Office, Shiokoji Horikawa, Shimogyo ku, Kyoto, Japan). Three measurements were taken at 1-min interval. The mean of the two latest BP measurements was considered as BP values. Hypertension was defined as use of medications to treat hypertension, a systolic BP ≥140 mmHg or a diastolic BP ≥90 mmHg. Insulin resistance was estimated using the homeostasis model assessment for insulin resistance (HOMA-IR) based on the formula: fasting glucose (mg/dL) × insulin (µIU/mL)/405 HOMA-IR values. In addition, dyslipidemia (cholesterol-LDL ≥130 mg/dL and/or lipid-lowering drugs) and metabolic syndrome (≥3 criteria: waist measurement >88 cm for women or >102 cm for men, HDL-cholesterol <50 mg/dL for women or <40 mg/dL for men, a systolic BP ≥130 mmHg or ≥85 mmHg, serum triglyceride levels ≥150 mg/dL, and fasting plasma glucose ≥110 mg/dL) diagnoses were defined according to NCEP ATP III criteria. 31
Statistical analysis
Descriptive data were provided according to migraine status (no headache, MO, and MA). Categorical variables were analyzed by χ 2 test and presented as absolute numbers and relative frequencies. Continuous variables were compared by Kruskal–Wallis (K-W) test and presented as median values with respective IQR. ADPs levels were compared in a 2 × 2 way in subgroups of study using Mann–Whitney U test, if significant difference was detected by K-W test (overall p < 0.05). The p values were adjusted for multiple comparisons using the Bonferroni method.
Logistic regression models were built to evaluate the relationship between ADP tertiles (dependent variable, first tertile: reference) and migraine status according to the following subgroups: no-migraine (reference), MA, and MO. Odds ratio (OR) with respective 95% confidence interval (95%CI) was calculated as crude (model 1), age and sex-adjusted (model 2), further adjusted by BMI (model 3) and metabolic syndrome (model 4) and the full model that included the adjustments for age, sex, BMI, and metabolic syndrome (model 5). We also performed stratified analyses by sex. Further additional adjustments for other potential confounding variables such as inflammatory biomarkers and medication for migraine prophylaxis were also tested in multivariate models.
For all analyses, p values <0.05 were also considered significant. Statistical analyses were performed with the statistical software SPSS version 25.0.
Results
Table 1 shows baseline characteristics according to migraine status. From 257 participants (mean age: 46 years ± SD: 4.8, 47.5% women), 36.2% (93/257) were migraineurs. From the total, 16.7% (43/257) had MA. Among migraineurs, the proportion of participants with and without aura was 46.2% (n = 43/257) and 53.8% (n = 50/257), respectively. Of note, participants with MA were 3 years younger than those with no headache (45 years vs. 48 years, p < 0.0001). Also, they presented lower frequency of hypertension, metabolic syndrome, alcohol consumption, lower levels of waist circumference, fasting glucose, triglycerides, as well as higher levels of HDL-cholesterol than no-headache subgroup (p < 0.05).
Demographical, clinical, and laboratorial characteristics of 257 participants from the ELSA-Brasil, according to migraine status.a

ELSA-Brasil: Brazilian Longitudinal Study of Adult Health; IQR: interquartile range; HOMA-IR: homeostasis model assessment for insulin resistance; Hs-CRP: high-sensitivity C-reactive protein; IL-6: interleukin-6; BMI: body mass index; BP: blood pressure.
a Continuous variables are demonstrated in median with IQR as well as categorical variables are described in absolute numbers and relative frequencies. Missing values are at most 6.2% in the variable “physical activity.” Hypertension was defined by systolic BP ≥140 mmHg, diastolic BP ≥90 mmHg, or current treatment. Dyslipidemia was defined as LDL-cholesterol ≥130 mg/dL. Metabolic syndrome (NCEP ATP III) was defined by the presence of ≥ three of the following criteria: waist measurement > 88 cm for women or > 102 cm for men, HDL-cholesterol < 50 mg/dl for women or < 40 mg/dl for men, a systolic blood pressure ≥ 130 m Hg or ≥ 85 mm Hg, serum triglyceride levels ≥ 150 mg/dl, and fasting plasma glucose ≥ 110 mg/dl. High hazardous alcohol consumption per week: ≥140 g for women and ≥210 g for men.
b p Values were from χ 2 test for categorical variables and from Kruskal–Wallis test for continuous variables: p value <0 0.05.
c p Values were from χ 2 test for categorical variables and from Kruskal–Wallis test for continuous variables: p value < 0.0001.
As stated in the Methods section, in the present analysis, we excluded the few cases (n = 5) of daily migraine to better evaluate the relationship between ADPs and migraine according to aura symptoms. From 93 cases classified as definite migraine (MO and MA) the following frequencies were reported: 16.1% = once in a while; 46.2% = once or twice a month; 14% = once a week; 23.7% = more than once a week. No statistical difference regarding frequency was noticed between MO and MA (p value =0.55). Of all episodes, 2.2% (2/93) were mild, 71% (66/93) as moderate, and 26.9% (25/93) as severe with no statistical difference between both migraine subtypes (p value = 0.41).
The frequency of use of prophylactic medication was of 2.0% in MO and 11.6% in MA (p value =0.06). Use of prophylactic medication was low even considering that the sample has more access to health services than the general population in Brazil.
Regarding biomarkers, HOMA-IR, Hs-CRP, and IL-6 levels were not significantly different across the subgroups (no headache, MO, and MA). However, higher median values of leptin (ng/mL) were observed among migraineurs, particularly in those with aura symptoms: no headache: 9.5 (IQR: 5.5–15.7) versus MO: 17.0 (IQR: 10.9–34.2) versus MA: 20.9 (IQR: 11.7–29.3), overall p value <0.0001 (Figure 2). Both MA and MO subgroups presented significatively higher levels of leptin than no-headache subgroup (both p values <0.0001). No significant differences were detected for adiponectin levels according to migraine status (p = 0.13). In stratified analyses by sex, leptin levels (ng/mL) were progressively higher across migraine status in women (no headache: median: 14.5 (IQR: 7.8–23.9) vs. MO: median 18.3 (IQR: 12.8–34.3 ng/mL) vs. MA: median 26.4 (IQR: 16.5–40.8), p = 0.02)), but not in men. One more time, leptin levels were significantly higher in MO and MA compared to the no-headache subgroup (p = 0.02 and p < 0.0001, respectively). After the adjustment for multiple comparisons, all p values <0.0001 (leptin levels by MA and MO compared to no headache) remained significant, except for a borderline significance for the difference in leptin levels between MO and no-headache subgroup in women (p = 0.053).

Distribution of serum adiponectin (a) and leptin levels (b) at the baseline of ELSA-Brasil, according to migraine status in all sample (N = 257).
Overall, adiponectin levels were not significantly different across migraine status for both sexes (Figures 3 and 4).

Distribution of serum adiponectin (a) and leptin levels (b) at the baseline of ELSA-Brasil, according to migraine status in 122 women.

Distribution of serum adiponectin (a) and leptin levels (b) at the baseline of ELSA-Brasil, according to migraine status in 135 men.
After multivariate adjustment, the second tertile of adiponectin was positively associated to MO (OR 2.76; 95% CI: 1.09–6.96, p = 0.03) and third tertile of leptin was associated with MA (OR 3.38; 95% CI: 1.10–10.37, p = 0.03) (Table 2). Additional adjustments for other inflammatory biomarkers, including adiponectin and leptin, and migraine prophylaxis did not modify the direction of our main findings. In secondary analyses, we did not find any significant association between migraine status with obesity, overweight, or even metabolic syndrome (data not shown).
OR (95% CI) for the relationship between adiponectin and leptin tertiles according to migraine status in all sample (N = 257).

OR: odds ratios; CI: confidence interval; BMI: body mass index.
a First tertiles of adiponectin (0.80–7.05 mcg/mL) and leptin (0.11–7.94 mcg/mL) were considered as reference. Multivariate adjusted by age, sex, BMI, and metabolic syndrome.
In stratified analysis by sex, positive associations between leptin tertiles and MA were detected only for the female sex: second tertile: OR 3.81; 95% CI: 1.07–13.59; p = 0.04 and third tertile: OR 6.54; 95% CI: 1.74–24.57, p = 0.005 (Table 3); but no differences were found among men (Table 4). Overall, no significant associations between migraine and adiponectin were found for both sexes (Tables 3 and 4).
OR (95% CI) for the relationship between adiponectin and leptin tertiles according to migraine status in women (n = 122).
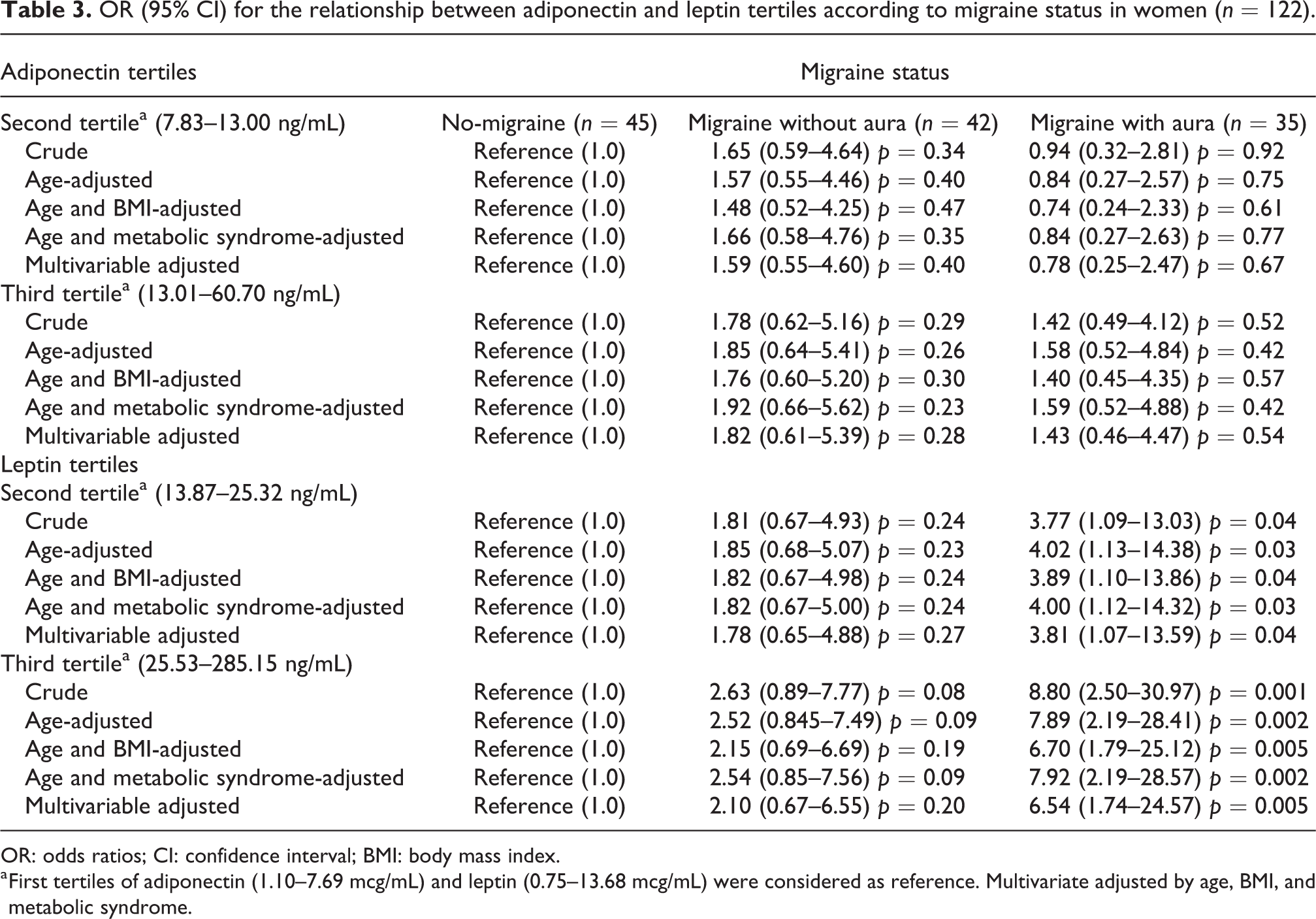
OR: odds ratios; CI: confidence interval; BMI: body mass index.
a First tertiles of adiponectin (1.10–7.69 mcg/mL) and leptin (0.75–13.68 mcg/mL) were considered as reference. Multivariate adjusted by age, BMI, and metabolic syndrome.
OR (95% CI) for the relationship between adiponectin and leptin tertiles according to migraine status in men (n = 135).
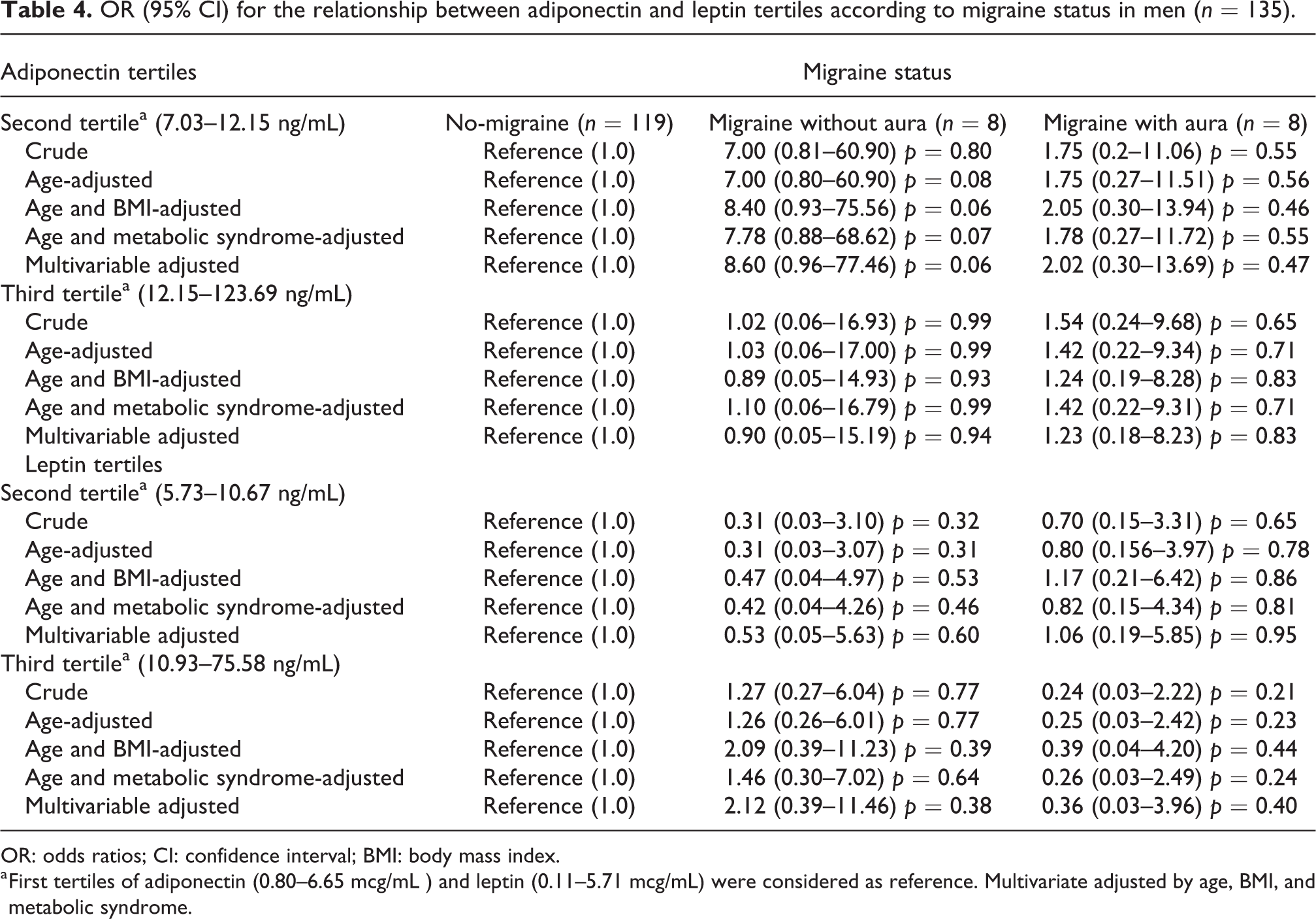
OR: odds ratios; CI: confidence interval; BMI: body mass index.
a First tertiles of adiponectin (0.80–6.65 mcg/mL ) and leptin (0.11–5.71 mcg/mL) were considered as reference. Multivariate adjusted by age, BMI, and metabolic syndrome.
Discussion
The general findings of our analyses revealed direct associations of leptin and adiponectin with migraine add knowledge to the complex relationship of adipocyte-produced cytokines in pathophysiology of migraine. We provide subsidies that may enhance understanding of the increased frequency of migraine associated to ADPs and the differences between sexes regardless of obesity. Interestingly, the highest levels of leptin were associated with MA while adiponectin levels with MO. In women, the association of leptin levels with MA was independent of body adiposity while in men, no association between migraine and adipocytokine levels was confirmed.
Leptin and migraine
Overall, leptin levels varied between migraineurs and controls across the studies that evaluated this intricate relationship involving aura symptoms. 17,19,21 Particularly in our study, leptin levels were higher in migraine than controls, mainly in middle-aged women with MA. Similar results regarding the association between leptin levels and migraine were reported in a population-based prospective CoLaus/PsyCoLaus cohort, designed to study mental disorders and cardiovascular diseases in individuals aged from 35 years to 75 years (mean age 48.8 years). 19 In 3025 participants, the impact of leptin levels was evaluated by lifetime migraine prevalence considering the presence of aura. Although MA prevalence in the CoLaus/PsyCoLaus cohort was lower (35%) than in the ELSA-Brasil study (46.2%), higher leptin levels were also observed in MA (14.6 ng/mL, p = 0.013) and MO (14.8 ng/mL, p < 0.001) compared to controls ((no-migraine), 12.2 ng/mL). Also, in the adjusted regression models, it was reported leptin levels positively associated with all-migraine (OR 1.17; 95% CI 1.03–1.34) and MA (OR 1.34; 95% CI 1.10–1.63). 19 Both studies depicted similar findings in the same age bracket as our cohort. In both studies, leptin levels were higher in women than men. However, no regression analyses stratified by sex were performed in the CoLaus/PsyCoLaus cohort. 19
The role of ADPs and migraine was also investigated in the Atherosclerosis Risk in Communities (ARIC) study, a nested case–control with 981 nondiabetic older migraine and no-migraine control participants (mean age 52.8 years). 17 Leptin levels measured in both sexes at baseline (visit 1) were not associated with migraine, even considering aura symptoms. 17 Although both cohorts evaluated nondiabetic individuals, differences on the population characteristics between ELSA-Brasil and ARIC might explain the divergent results regarding the association between MA and leptin, being the main one related to age, since ELSA-Brasil participants were 6 years younger than ARIC cohort. Other differences were related to the time of evaluation of migraine (6 years later than the ADPs dosage in the ARIC study), and the frequency of migraine (23% vs. 46.2% of MA among definite migraineurs in the ARIC and the ELSA-Brasil study, respectively).
Other small cross-sectional study of Domínguez et al. among 111 migraineurs (mean age 39.7 years, 93% female) and 24 healthy controls (mean age 35.9 years, 90% female) evaluated the relationship of leptin, and adiponectin in association with migraine considering aura symptoms. Leptin levels (15.2 ng/mL vs. 3.1 ng/mL; p < 0.001) were higher in all migraineurs compared to the control group. In the regression analyses, leptin levels were positively associated with all-migraine after adjustments for age, sex, BMI, and several other biomarkers. Although the frequency of aura among migraineurs was similar to our study (44.2% vs. 46.2%), no statistical differences were observed between migraineurs according to aura in univariate analysis in the Domínguez study. 21 Participants included in the Domínguez study were younger than those from ELSA-Brasil study. Also, the small size of the sample and the lack of matching between cases and controls may corroborate to the nonsignificant findings between leptin and migraine. 21
Adiponectin and migraine
In the ARIC study, adiponectin levels were associated with all-migraine among male. It was observed an increased OR of migraine for the entire sample (88% for each SD increase in total adiponectin) and for men (OR 1.86; 95% CI: 1.15–3.01) but not for women. Total and HMW adiponectin was also evaluated in the ARIC, and similar finding regarding the positive association between HMW adiponectin (pro-inflammatory oligomer) and all-migraine was reported. 17 However, due to the small number of male participants with aura (n = 6), reliable interpretation of adiponectin levels stratified by sex according to aura was not possible, as it was performed in our study.
In the study of Domínguez et al., also adiponectin levels were higher in migraineurs than in controls (72.3 mg/mL vs. 37.7 mg/mL; p < 0.001). However, no significant finding was detected in univariate analysis considering the association between MA and crude adiponectin levels (p = 0.49). 21 One of the factors associated to the lack of significance 21 was due to unmatched patients and controls by age or sex, and the presence of adiposity, 32 as we did in our analyses.
Although the presence of aura has been attributed to a more accentuated inflammation in migraine pathophysiology, 33 we did not find association between adiponectin and MA. In fact, we observed a positive association between the second tertile of adiponectin and MO for all sample in our analysis. We speculate that the lack of association of adiponectin with migraine according to aura in sex-stratified analyses could be attributed to sociodemographic characteristics of our sample as age. In our study, MO participants were 2 years older than those with MA. As previously reported in the literature, adiponectin levels differ in men and women and increase with age, especially in men. 32 Also, we cannot rule out the lack of power to detect some association between migraine and adiponectin due to few cases of men in our study.
Adipocytokines have been more commonly linked to metabolism of glucose and lipids because their inflammatory effects deteriorate insulin signaling. In addition, ADPs have also been involved in migraine physiopathology, particularly when this condition is preceded by aura. 13 In contrast to other ADP (e.g. leptin), adiponectin levels are usually decreased in obesity 13 ; however, evidence from studies in migraineurs has shown that both, leptin and adiponectin, can be directly associated with the occurrence of migraine. 17,19,21 Previous study has suggested that high leptin levels might be involved in the mechanism of aura symptoms. 19,33 On the other hand, adiponectin by its HMW oligomer form, which has an inflammatory role, can be implicated with migraine pathophysiology, as well. 17 We reinforce that high leptin levels were independently associated to the presence of MA, particularly in women, regardless of obesity and metabolic syndrome. Previously, ELSA-Brasil investigators have reported higher leptin levels in female sex, 26 which is in agreement with previous literature. 19
Strengths
We emphasize here that the strength of our study was to examine ADP levels according to the presence of aura in stratified analysis by sex compared to no-headache subgroup. Such strategy allowed us to identify that the highest leptin levels were verified in women with MA, reinforcing the hypothesis that inflammation may play a role for aura symptoms at least in female sex, since leptin levels are greater in reproductive age women than men. 13 In addition, adiponectin levels that had been previously associated with migraine were included among the adjustments of the leptin regression model with persistence of the association.
Moreover, evidence demonstrated by ELSA-Brasil on the association of leptin with MA aura in middle-aged women reinforces the possible inflammatory-mediating role, regardless the presence of obesity or metabolic syndrome. 17,19,21 Also, the availability of data about medication for migraine prophylaxis was important to improve interpretation of our results.
Limitations
Limitations of our study are related to its cross-sectional design, which does not allow inferences about causal relationships. Moreover, few cases of daily migraine (n = 5) in our sample impeded deepening association analyses of ADP in these cases. Also, we could not use the classification of chronic migraine (more than 15 days of pain per month) according to the ICHD-2nd edition. Thus, we cannot rule out some bias related to the association between ADP levels and chronic migraine because we might have included few cases of chronic migraine in the subgroup who reported a frequency of migraine more than once a week (n = 22).
The subsample included in this analysis derived from an ancillary study in the ELSA-Brasil research center of São Paulo, which evaluated atherosclerosis biomarkers from a random sample of 1000 participants selected from these 5061 subjects of our research center, was less than 55 years of age and presented no cardiovascular disease or diabetes at baseline of study. In addition, participants under lipid-lowering medication were also excluded. As in our subsample MA participants are younger than participants with MO and the use of lipid-lowering mediation is higher in older people, these filters may have affected the selection of participants explaining the higher frequency of MA (46.2%, n = 43/93) in the current study. Of note, previous studies that evaluated ADP levels and migraine reported frequencies of MA ranging from 23% to 44.2%. 17,19,21
As the main focus of the study was on cardiovascular diseases, participants were eligible if they were in the age strata between 35 years and 74 years. Therefore, we have no information about migraine headaches in adults with less than 35 years of age. Also, the paucity of men may also limit analyses for this gender. Finally, adiponectin mensuration involved total level in our study. As previously reported, 13 adiponectin can have a dual role in inflammation process depending on its isoform. The LMW oligomer is associated to anti-inflammatory pattern while the HMW oligomer to pro-inflammatory effects. Moreover, HMW adiponectin can play a role in migraine pathophysiology, 13 particularly chronic migraine. 20,21 Unfortunately, conclusions about the biological functions of adiponectin isoforms in the migraine headache according to aura symptoms were not possible to be done in our study.
Conclusions
Positive associations between ADP and migraine, particularly between MA and leptin levels in women, raise the possibility of adipocytokines and play a role in migraine pathophysiology.
Footnotes
Acknowledgements
The authors thank the research team of the ELSA-Brasil study for their contributions.
Clinical implications
ELSA-Brasil brings relevant information in the complex relationship between adiponectin, leptin, and MA symptoms. In this population of middle-aged individuals, MA was associated with the highest levels of leptin, while MO was associated with intermediary levels of adiponectin. Our findings yield information about the inflammatory role of ADP as possible predictor among migraineurs with aura, particularly in women.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics or institutional review board approval
Ethics committee of each ELSA-Brasil centers approved this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The ELSA-Brasil baseline study was supported by the Brazilian Ministry of Health (Science and Technology Department) and the Brazilian Ministry of Science and Technology (Financiadora de Estudos e Projetos and CNPq National Research Council) (grants 01 06 0010.00 RS, 01 06 0212.00 BA, 01 06 0300.00 ES, 01 06 0278.00 MG, 01 06 0115.00 SP, 01 06 0071.00 RJ).
