Abstract
Background
Migraine is a prevalent disorder characterised by recurrent headache attacks preceded or accompanied by aura in a subgroup of patients. Migraine often occurs together with major depressive disorder (MDD). Alterations of adipokine levels have been reported both in migraine and in MDD. In this cross-sectional study, we aimed to assess the associations between serum leptin and adiponectin levels and migraine or migraine subtypes. Analyses were adjusted for a lifetime history of MDD in order to investigate the association between adipokines and migraine under consideration of depression status.
Methods
We included 3025 participants from the CoLaus/PsyCoLaus study. The impact of leptin and adiponectin levels on a diagnosis of migraine was analysed by binary regression analyses, adjusting for variables known to influence adipokine levels. Subgroup analyses were conducted based on the presence of aura.
Results
Crude leptin levels were significantly higher in subjects with migraine than controls (Mann–Whitney U = 515,102, p = 6 × 10−7). When performing adjusted analyses, leptin levels were found to be significantly higher in subjects with migraine (odds ratio = 1.22, p = 0.024) and migraine with aura (odds ratio = 1.34, p = 0.004).
Conclusion
High leptin levels might play a role in the pathogenesis of migraine and migraine with aura.
Introduction
Migraine is a disabling primary headache disorder of neurovascular origin that is three times more frequent in women (1), with a prevalence of 1 billion of people worldwide (2). Ranked as the third most prevalent disorder and seventh-highest specific cause of disability worldwide (2), migraine has a profound socio-economic and personal impact. Migraine without aura (MWOA) is the most common form of migraine and is characterised by the recurrence of unilateral, severe headache attacks lasting 4–72 hours with associated symptoms (3). In migraine with aura (MWA), migraine is preceded or accompanied by reversible focal neurological symptoms that last for fewer than 60 minutes (3). Recent findings suggest an association between migraine and obesity (4). This association could be mediated by adipokines (i.e. cytokines mainly secreted by adipose tissue), which participate in a number of processes that may play a role in migraine pathophysiology, such as inflammation and endothelial function (5). Among the adipokines, the 16-kDa peptide hormone leptin leads to the reduction of food intake and increase of energy consumption through the induction of anorexigenic factors. Importantly, leptin is able to act as a proinflammatory cytokine and modulate immune responses (6). Adiponectin is a 30-kDa protein that is involved in fatty acid oxidation and glucose synthesis (6,7) and has been suggested to exert both pro- and anti-inflammatory properties (6). While leptin levels are usually increased in obesity (8), adiponectin levels have been shown to be reduced (9). Studies that assessed the levels of these two adipokines in subjects with migraine have provided conflicting results (5,10–17). One reason for the contradictory findings could be the heterogeneity of migraine. Indeed, the two subtypes – MWOA and MWA – could be differentially associated with adipokines. However, previous studies that assessed the association between adipokines and migraine have only partially taken into account these subtypes.
Major depressive disorder (MDD), which has been frequently shown to be associated with both migraine and obesity, could also play a role in the association between migraine and obesity. Indeed, migraine patients have a 2-4-fold higher risk of MDD (18,19), and several reviews and meta-analyses found depression to be associated with obesity (20–22). MDD could either be a condition predisposing to both migraine and obesity or a mediator of the association between the two conditions (i.e. obesity may predispose to MDD, which predisposes to migraine, or the converse).
In the present study, we aimed to assess the associations between serum leptin and adiponectin levels and migraine or migraine subtypes. Analyses were adjusted for a lifetime history of MDD in order to investigate the association between adipokines and migraine under consideration of depression status.
Methods
Subjects and cohort
The data of the present paper stem from CoLaus/PsyCoLaus (23,24), a cohort study designed to study mental disorders and cardiovascular risk factors (CVRFs)/cardiovascular diseases in the general population. A total of 6733 individuals (CoLaus) aged between 35 and 75 years were randomly selected between 2003 and 2006 from the residents of the city of Lausanne (Switzerland) according to the civil register. In addition to anthropometric measures, DNA, serum and plasma samples were collected for the study of genetic variants and biomarkers associated with CVRFs (23). Subsequently, 67% of the participants of CoLaus in the age range of 35–66 years (n = 5535) agreed to take part in the psychiatric evaluation (PsyCoLaus), which resulted in a sample of 3719 individuals who underwent both the somatic/cardiovascular and psychiatric examination (24). A total of 92% of these individuals were Caucasians. The gender distribution of the PsyCoLaus sample (47% men) did not differ significantly from that of the general population in the same age range (24). Although the youngest 5-year band of the cohort was underrepresented and the oldest 5-year band overrepresented, participants of PsyCoLaus (mean age 50.9 years; SD 8.8 years) and individuals who refused to participate revealed comparable scores on the General Health Questionnaire (GHQ-12) (25), French translation (26), a self-rating instrument completed at the somatic examination.
Of the initial 3719 individuals, 656 were excluded because of missing adipokine levels, 30 because of missing migraine assessment, six because of missing lifetime MDD assessment, one because of missing blood glucose levels and one because of missing body mass index (BMI) information, leaving a total of 3025 subjects included in the present study. Excluded subjects were less frequently women (chi-square = 24.8, p < 0.001) and younger (Mann–Whitney U = 983,654, p = 0.012) than non-excluded ones.
The CoLaus and PsyCoLaus studies were approved by the Institutional Ethics Committee of the University of Lausanne. Informed written consent was obtained from all participants.
Assessment of clinical and biological data
The lifetime prevalence of migraine was assessed according to the criteria of the International Classification of Headache Disorders (ICHD-II) (3), using the validated French version of the Diagnostic Interview for Headache Syndromes (DIHS). Prevalence of MDD was assessed using the validated French translation (27) of the Diagnostic Interview for Genetic Studies (DIGS) (28). The French translation of the DIGS revealed excellent inter-rater reliability in terms of kappa and Yule’s Y coefficients for major mood and psychotic disorders, whereas the 6-week test–retest reliability was slightly lower (29). Interviews were conducted by trained psychologists or psychiatrists and reviewed by an experienced senior psychologist.
Body weight and height were assessed and a venous blood sample was obtained after an overnight fast (with a minimum fasting time equal to 8 hours). Samples were stored at −80℃ and sent on dry ice to the laboratory. Total serum leptin levels were measured by Pathway Diagnostics (Los Angeles, CA) using enzyme-linked immunosorbent assay (ELISA) (American Laboratory Products Company, Windham, NY; maximum inter-assay coefficient of variability (CV) = 12.8%; maximum intra-assay CV = 5.8%). Total serum adiponectin levels were measured using ELISA (R&D Systems, Inc., MN; maximum inter-assay CV = 8.3%; maximum intra-assay CV = 8.3%). Fasting plasma glucose levels were measured by the Centre Hospitalier Universitaire Vaudois (CHUV) laboratory using a glucose dehydrogenase assay (Roche Diagnostics, Switzerland; maximum inter-assay CV = 2.1%; maximum intra-assay CV = 1.0%).
Statistical analysis
The normal distribution of continuous variables was assessed using the Kolmogorov–Smirnov test. Differences in continuous or categorical variables between subjects with migraine and controls were assessed using Mann–Whitney U or Pearson chi-square tests, respectively. Correlations between adipokine levels and continuous variables were analysed using Spearman correlation. The association between adipokine levels and a diagnosis of migraine was assessed using binary logistic regression analysis. Three models of increasing complexity were constructed. In model 1, the association was adjusted for age and sex; model 2 included adjustments applied in model 1 and was additionally adjusted for BMI; model 3 was same as model 2 and also adjusted for fasting plasma glucose levels, lifetime MDD and regular intake of antidepressants. In all of these models, the dependent variable was the presence of migraine. In subsequent analysis based on migraine subtypes, the dependent variable was the presence of MWA or the presence of MWOA.
In order to assess whether sex or a lifetime history of MDD exerted an impact on the association between adipokine levels and migraine, models were first tested for potential interactions between adipokine levels, sex and lifetime diagnosis of MDD with respect to migraine and migraine subtypes. Specifically, the following interactions terms were included in the model: ‘sex × leptin levels’, ‘sex × adiponectin levels’, ‘MDD × leptin levels’ and ‘MDD × adiponectin levels’. In the final model, interaction terms that did not reach the level of statistical significance were removed.
A p-value < 0.05 was considered significant. Statistical analyses were performed using SPSS v.21 (IBM, Armonk, NY).
Results
Demographic and clinical characteristics of the sample
Demographic and clinical characteristics of the sample (n = 3025).

Continuous variables are expressed as mean ± SD; p-values are calculated using the Mann–Whitney U test or Pearson chi-square test, comparing subjects with migraine or migraine subtypes versus controls. Significant differences are indicated in bold.
BMI: body mass index; MDD: major depressive disorder; MWA: migraine with aura; MWOA: migraine without aura.
Adipokine levels and the prevalence of migraine and MWA
Leptin levels were higher in women (mean = 16.2 ng/mL, SD = 11.4) than men (mean = 8.2 ng/mL, SD = 7.2; p < 0.001) and were positively correlated with age (rho = 0.14) and BMI (rho = 0.38; p < 0.001 for both variables). Similarly, adiponectin levels were higher in women (mean = 12.3 µg/mL, SD = 9.4) than men (mean = 7.2 µg/mL, SD = 5.7; p < 0.001) and correlated positively with age (rho = 0.13) and inversely with BMI (rho = −0.29; p < 0.001 for both variables). Before adjustments, leptin levels were significantly higher in subjects with migraine than controls, regardless of the migraine subtype (Table 1). In contrast, there was no association between adiponectin levels and migraine or its subtypes.
In order to test the impact of sex or comorbidity with MDD on the association between adipokine levels and migraine, a binary logistic regression model with interaction terms for sex and lifetime history of MDD was applied in a first step. We found no significant interactions between sex or lifetime MDD and adipokine levels regarding the risk for migraine, MWA or MWOA (Supplementary Table 1). Therefore, all interaction terms were removed from the model.
Associations between adipokine levels and a diagnosis of migraine and migraine subtypes according to logistic regression models.
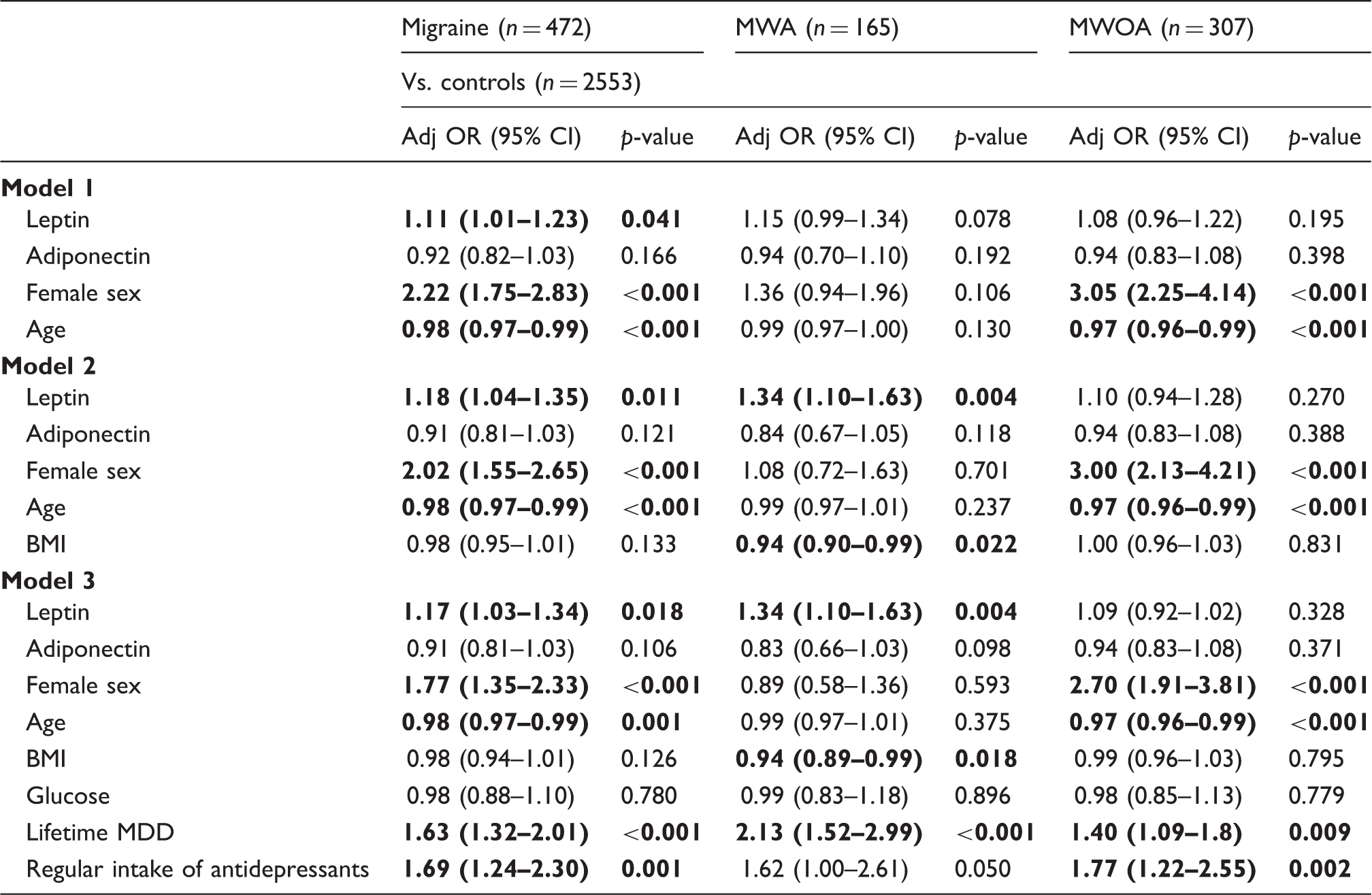
In model 1, adipokine levels are adjusted for age and sex; in model 2, adipokine levels are adjusted for the variables included in model 1 plus BMI; in model 3, adipokine levels are adjusted for the variables included in model 2 plus fasting glucose levels, lifetime history of MDD and regular intake of antidepressants. Significant results are reported in bold.
ORs with 95% CIs are calculated in relation to an increase of 1 unit of SD in leptin or adiponectin levels.
Adj: adjusted; BMI: body mass index; CI: confidence interval; MDD: major depressive disorder; MWA: migraine with aura; MWOA: migraine without aura; OR: odds ratio.
Among the covariates included in the models, female sex was associated with migraine and the MWOA subtype. Moreover, increasing age was associated with a reduced prevalence of migraine and the MWOA subtype, and increasing BMI was associated with a reduced prevalence of the MWA subtype. Finally, the lifetime histories of MDD and migraine and its subtypes were strongly associated.
Discussion
In the current study, we evaluated the association between adipokine levels and a diagnosis of migraine in a large population sample including 3025 participants from the CoLaus (23) and PsyCoLaus (24) studies. The major finding was the strong association between high leptin levels and migraine after adjustment for a comprehensive series of variables known to influence leptin levels, including demographic characteristics, BMI and fasting glucose levels. Although a lifetime history of MDD was strongly associated with migraine and its two subtypes, the association between migraine and leptin levels was not explained by depression status.
Previous studies that explored the relationship between leptin levels and migraine yielded inconsistent results. Given our evidence of differential associations between leptin levels and migraine subtypes, the conflicting results of previous research could be due to variance of subtypes across studies. One study found lower leptin levels in 61 patients with episodic migraine compared with 64 healthy subjects before adjusting for fat mass (11), whereas another study documented higher crude but not adjusted leptin levels in 44 non-obese female migraine patients (13 of whom had symptoms of aura) compared with 40 female controls (12). Another study assessing migraine subtypes reported negative results in a sample of 72 patients with migraine, 59 with a diagnosis of probable migraine and 850 controls (14). However, only 23 patients with migraine and 12 with probable migraine had a history of aura, which consisted of having experienced a visual aura in the past, rather than having met the MWA criteria according to ICHD-II.
Interestingly, in our study, an association between leptin levels and migraine was specifically seen for subjects with MWA. This finding is well in line with preclinical study results showing that hyperleptinaemia has the potential to increase cortex susceptibility to cortical spreading depression (CSD), a mechanism that is suggested to induce auras during migraine attacks. CSD is characterised by a slowly propagating neuronal and glial depolarisation wave followed by hyperpolarisation (30). Chronic hyperleptinaemia has recently been found to increase CSD in a rat model after intraperitoneal leptin administration (31), and Zucker fatty rats (a model of obesity with intrinsic hyperleptinaemia) display a significantly higher frequency of KCl-induced CSD than Zucker lean rats (31).
Mechanistically, it cannot be excluded that higher levels of leptin in patients with MWA may also be a feedback response to prolonged stress and hypothalamic–pituitary–adrenal (HPA) axis activation. Interestingly, in food-deprived mice, leptin administration was shown to prevent the induction of corticotropin-releasing hormone (CRH) production in the paraventricular nucleus and to lower corticosterone levels (32). Moreover, in vivo photostimulation of orexin neurons in the lateral hypothalamic area was shown to induce a stress response through regulation of CRH secretion. Leptin administration inhibited orexin neuron activation through leptin receptor b-expressing inhibitory neurons, in this way suppressing the HPA axis (33).
Our finding of a lack of association between migraine and adiponectin levels is in line with results of a meta-analysis (13) and one recent study (16), and contrasts with one study that revealed higher adiponectin levels in 68 migraine patients compared with 65 controls, independently of psychiatric comorbidities, migraine impact and allodynia (15). Another study documented an association only in men older than 50 years of age, but not in women with migraine (14).
Our results need to be interpreted in the light of several limitations. First, the participation rate was relatively low, which was probably due to the comprehensive physical and psychiatric assessments. Second, the cross-sectional nature of the data reported herein did not allow us to determine the temporal sequence between adipokine level increases and the onset of migraine. Third, total adiponectin but not high- and low-molecular-weight oligomers were measured and assessed for association with migraine. These oligomers could be differentially associated with migraine (10). Fourth, all subjects except one had a diagnosis of episodic migraine. Therefore, we were not able to specifically evaluate whether chronic migraine was associated with higher adiponectin levels, as suggested by a previous study (10). In conclusion, the present study suggests that high leptin levels are associated with the risk of migraine and MWA. Prospective studies that can provide clues as to the temporal sequence between leptin level increases and the onset of migraine are needed in order to confirm our findings and to further elucidate the role of leptin in migraine pathogenesis. Future studies should also subtype into high- and low-molecular-weight oligomers in order to unravel the potential differential effects of these specific oligomers on the risk of migraine.
Article highlights
Subjects with migraine showed higher leptin levels than controls. Higher leptin levels were specifically associated with migraine with aura. The association between leptin levels and migraine was observed independently of major depressive disorder.
Footnotes
Acknowledgements
The authors would like to express their gratitude to the investigators who recruited the CoLaus sample, particularly to Prof. Peter Vollenweider and Prof. Gérard Waeber.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: a grant from the Swedish Society for Medical Research (SSMF) to JM and a grant from the Swedish Research Foundation to HBS. The CoLaus/PsyCoLaus study was and is supported by research grants from GlaxoSmithKline, the Faculty of Biology and Medicine of Lausanne and the Swiss National Science Foundation (grants 3200B0-105993, 3200B0-118308, 33CSCO-122661, 33CS30-139468 and 33CS30-148401). The present paper is supported in part by NIH and the Intramural Research Program/NIMH (MHØØ2932).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
