Abstract
Background:
Both propranolol and candesartan are prophylactic drugs for migraine, but with unknown mechanisms of action. The objectives of the present study were to investigate these drugs’ effects on arterial wall dynamics and the potential relation between their vascular and clinical effect.
Methods:
The study was based on data from a previously published randomized, placebo-controlled, triple-blinded, double crossover clinical trial comparing the prophylactic effects of candesartan and propranolol in 72 patients. Finapres noninvasive blood pressure curves were analyzed. On the descending limb of the pulse curve, a notch is produced by pulse wave reflection, and its relative height compared to the top of the curve (the notch ratio) was used as a marker of arterial wall stiffness.
Results:
Candesartan decreased the notch ratio from baseline (p = 0.005), reflecting more compliant arteries and vasodilation, whereas propranolol increased the notch ratio (p = 0.005), reflecting less compliant arteries and vasoconstriction. There was no difference in baseline notch ratio between clinical responders and nonresponders.
Conclusion:
The drugs are both efficient prophylactic medications, yet they have opposite effects on arterial wall dynamics. This suggests that drug effects other than those on arterial compliance must be responsible for their prophylactic effect in migraine.
Introduction
The pathophysiology of migraine is uncertain, and the current dominant hypothesis is that migraine mainly originates from the neural system. 1,2 The long-standing concept of migraine as primarily a vascular disorder in which headache is caused by dilation of blood vessels, and its throbbing is due to the arterial pulse, has been refuted by substantial evidence during the past two decades. 3,4 It is now clear that constriction of blood vessels is not a required mechanism of antimigraine therapies. 3 One hypothesis is that vessel wall dysfunction is involved in the initial stages of migraine attacks. A recent study, however, reveals that a genetic variant that reduces the expression of endothelin, which in turn leads to vasodilation, is strongly associated with migraine. 5 Another study indicates differences in cardiovascular regulation between migraine patients and healthy controls. 6 A review on arterial wall dynamics suggests peripheral vascular dysfunction in migraine patients, with evidence supporting greater stiffness or impaired compliance. 7
Several drug classes have documented beneficial prophylactic effects on the frequency and severity of migraine attacks. These include the antihypertensive drugs β-adrenergic receptor blockers (β-blockers) 8 and angiotensin II receptor blockers (ARBs). 9 These drugs target the cardiovascular system. Curiously, the two drug classes affect the arterial wall in opposite directions, as discussed later: nonselective β-blockers reduce, whereas ARBs increase arterial compliance and peripheral resistance. 10,11
In a randomized placebo-controlled crossover clinical trial of candesartan and propranolol, sequences of continuous blood pressure were measured at each follow-up visit, 12 providing pulse curves that could give information also on vessel wall properties. When the antegrade arterial systolic pressure wave reaches the arterioles, it is reflected and produces a small incision named the dicrotic notch, on the descending limb of the pulse pressure curve. Elastic arteries with high compliance will have a late, low location of their dicrotic notch, whereas stiff arteries will have an early, high notch. 13 The location of the dicrotic notch can be described as the notch ratio: the ratio between the notch height above the diastolic pressure and the pulse pressure (Figure 1). This allowed us to investigate the vessel wall dynamics in migraine patients, and the relationship between vessel wall responses and treatment effects of the nonselective β-blocker propranolol and the ARB candesartan.

Notch ratio: The height of the dicrotic notch (b) relative to the total height (a), that is, b/a.
The primary objective was to determine whether the drug-induced changes in notch ratio for each drug were different in clinical responders compared to that of nonresponders.
Due to a lack of earlier studies comparing baseline differences in notch ratio between migraine patients and healthy controls, we looked for differences in mean notch ratio values at baseline between migraine patients and a group of healthy controls as a secondary objective. We also wanted to determine whether clinical responders to each of the drugs had baseline notch ratios different from nonresponders.
Materials and methods
The present study describes pulse curve analysis (not reported before) from a previously published randomized, placebo-controlled, triple-blind, double crossover study, where candesartan was compared with propranolol as prophylactic medication for migraine with aura. 12 Trial registration: EUDRACT (2008-002312-7), ClinicalTrials.gov (NCT00884663), 2016/1004/REK Mid-Norway. The treatment study protocol has been published in detail elsewhere, 12 but briefly describes a 4-week baseline period followed by three 12-week periods, one for each of the treatment arms: candesartan, propranolol, or placebo. There was a 4-week washout period between each treatment period.
In addition to the 72 patients included in the main study, 103 age- and sex-matched healthy controls were recruited by posters at universities and large companies in the area, as well as by intranet advertisements at the same sites. Each healthy subject was matched to one patient, with age not differing more than 5 years. All investigations were performed in the same time period with patients and controls in random order. The investigator was also blinded to the subjects’ status (patient/healthy control). Seventeen patients were recruited from the neurological outpatient clinic of St. Olavs University Hospital (Trondheim, Norway), and 55 patients were included among those who contacted the study team voluntarily after advertisement on national TV, in the newspapers, and on the Internet.
Inclusion criteria for patients in the original study were as follows: age between 18 and 65 years; signed informed consent; migraine with or without aura, or chronic migraine; two or more migraine attacks per month over the last 3 months before inclusion, two or more migraine attacks during the 4-week baseline period; debut of migraine more than 1 year prior to inclusion, before age of 50. The controls and the migraineurs were similar in age and sex (Table 1).
Baseline characteristics of patients (n = 61) and controls (n = 80).

SD: standard deviation.
Clinical assessment
The primary effect variable in the original study of Stovner et al. 12 was number of days per 4 weeks with moderate or severe headache, either lasting for over 4 h or treated with the patient’s usual acute medication, according to recommendations from guidelines for prophylactic migraine studies. 14 A number of secondary measures were also made, of which only the number of drug responders will be discussed in this article. Drug responders were participants with a reduction in migraine days by 50% or more, compared with baseline.
Pulse wave recording
At the end of each treatment period, continuous finger blood pressure was measured after 10 min of supine rest, during 5 min spontaneous breathing, during 5 min paced breathing, first at 6/min, then at 15/min (Finapres® Nova, Finapres Medical Systems, The Netherlands). Finapres (acronym for Finger Arterial Pressure) is a noninvasive method of measuring blood pressure. The system uses a finger cuff mounted with an infrared sensor and an inflatable bladder. A fast-acting servo system responds to the changing intra-arterial pressure by inflating the bladder when the intra-arterial pressure rises and deflating when the pressure lowers. Thus, the arteries are clamped at a fixed diameter; the transmural pressure is zero and the intra-arterial pressure always equalizes the cuff pressure.
Finapres continuous blood pressure recordings were obtained at each outpatient visit; four times for each patient (at baseline and during the last 1–2 weeks of each of the three treatment periods), and once for each of the controls. This sums up to 288 recordings for the patients and 103 recordings for the controls, 391 recordings in total. In addition to the Finapres measurements, brachial cuff blood pressures were also obtained during each outpatient visit.
The pulse curves were analyzed in LabChart 8 Pro (ADInstruments, Sydney, Australia). An automated integrated blood pressure module was used to determine maximum points of the curves (peak systolic pressure), minimum points (end diastolic pressure), and the dicrotic notch. LabChart detects the dicrotic notch of the pulse curve by evaluating the adjacent rise after the notch (detection threshold 0.5% of the total height). The notch ratio was calculated as the ratio between the height of the notch above the diastolic pressure (b) and the pulse pressure (a) (Figures 1 and 2). In some cases, the automated integrated blood pressure module in LabChart was unable to detect the notch, which was then determined manually from 10 to 15 contiguous pulse cycles in the start, the middle, and the end of each of the three 5-min breathing sequences, and the mean ratio was calculated. A split peak on the blood pressure curve could indicate poor quality of measurement, making the reading less accurate. Furthermore, when the dicrotic notch had a very high position, it could be less distinct, or sometimes invisible. All these curves, along with atypically looking curves, were rejected (Figure 3).
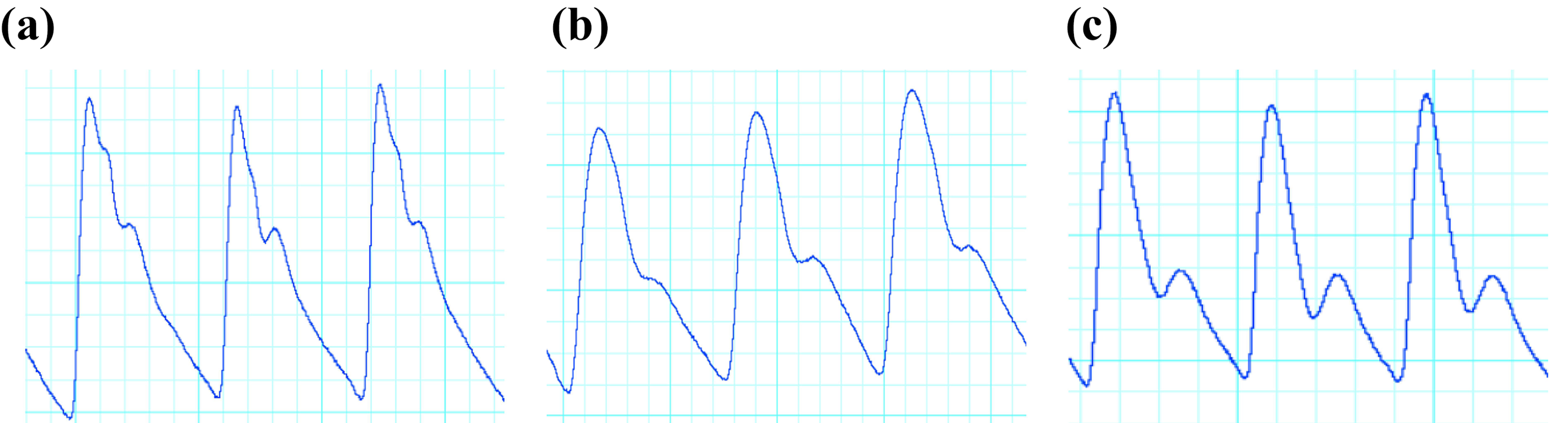
Vasoconstriction (a), normal (b), and vasodilation (c).

Flow of patients and pulse curve recordings. *Unavailable owing to dropout from Stovner et al. 12 **Excluded owing to poor quality. ***Seven patients lacked all recordings, three patients had only one recording, and one patient had only propranolol and placebo recordings but lacked baseline. Exclusion of these 11 patients leads to removal of a total of nine recordings.
Statistical analyses
Continuous variables are described with mean and standard deviation for normally distributed variables or with median and range for variables with skewed distributions. Categorical data are presented as counts and percentages.
An independent t-test was used to compare the means of baseline notch ratios between patients and healthy controls. Paired samples t-tests were used to determine whether the mean difference in notch ratio between baseline and on-treatment was different from zero for all the treatment groups: candesartan, propranolol, and placebo. Independent t-tests were used to compare baseline notch ratios between responders and nonresponders on all treatments. Delta notch ratios (ΔNR), which we defined as the difference between treatment values and baseline values, were not normally distributed. The nonparametric Mann–Whitney–Wilcoxon test was used to analyze the possible differences between responders and nonresponders on all treatments. To test a possible carry-over period effect, we fitted a logistic regression statistical model with a time × treatment interaction term. All tests were two-sided, and the values of p < 0.05 were considered statistically significant. The analyses were considered exploratory. Thus, no correction for multiple testing was performed. Missing data were excluded pairwise for each analysis.
Results
Of the 391 potential recordings (serial from patients, baseline only for controls), 58 were unavailable owing to technical failure or dropout, and 24 were rejected owing to problems with locating the dicrotic notch. The remaining 309 recordings were analyzed in a blinded and randomized order (Figure 3). Only patients with baseline recordings and at least one additional recording were included in the final main analyses, which comprised a total of 220 recordings from 61 patients. Their age ranged from 18 to 60, and 51 (84%) were females (Table 1). Two patients did not complete the main study of Stovner et al. 12 according to the study protocol, and therefore did not have a defined responder/nonresponder status. These were left out from the analyses concerning responder status in the present study, but their baseline data were used.
The notch ratio was normally distributed, both in controls and patients, and the notch ratio at baseline was not different comparing patients with controls (p = 0.68).
Comparing the patients’ notch ratio during treatment with baseline, the notch ratio decreased on candesartan (p = 0.005), increased on propranolol (p = 0.005), and was unchanged on placebo (p = 0.48) (Table 2 and Figure 4). There was no periodic effect during the crossover trial (all p > 0.50).
Baseline and delta values of mean notch ratio.

ΔNR: delta notch ratio; n: number of recordings; m: number of missing recordings.
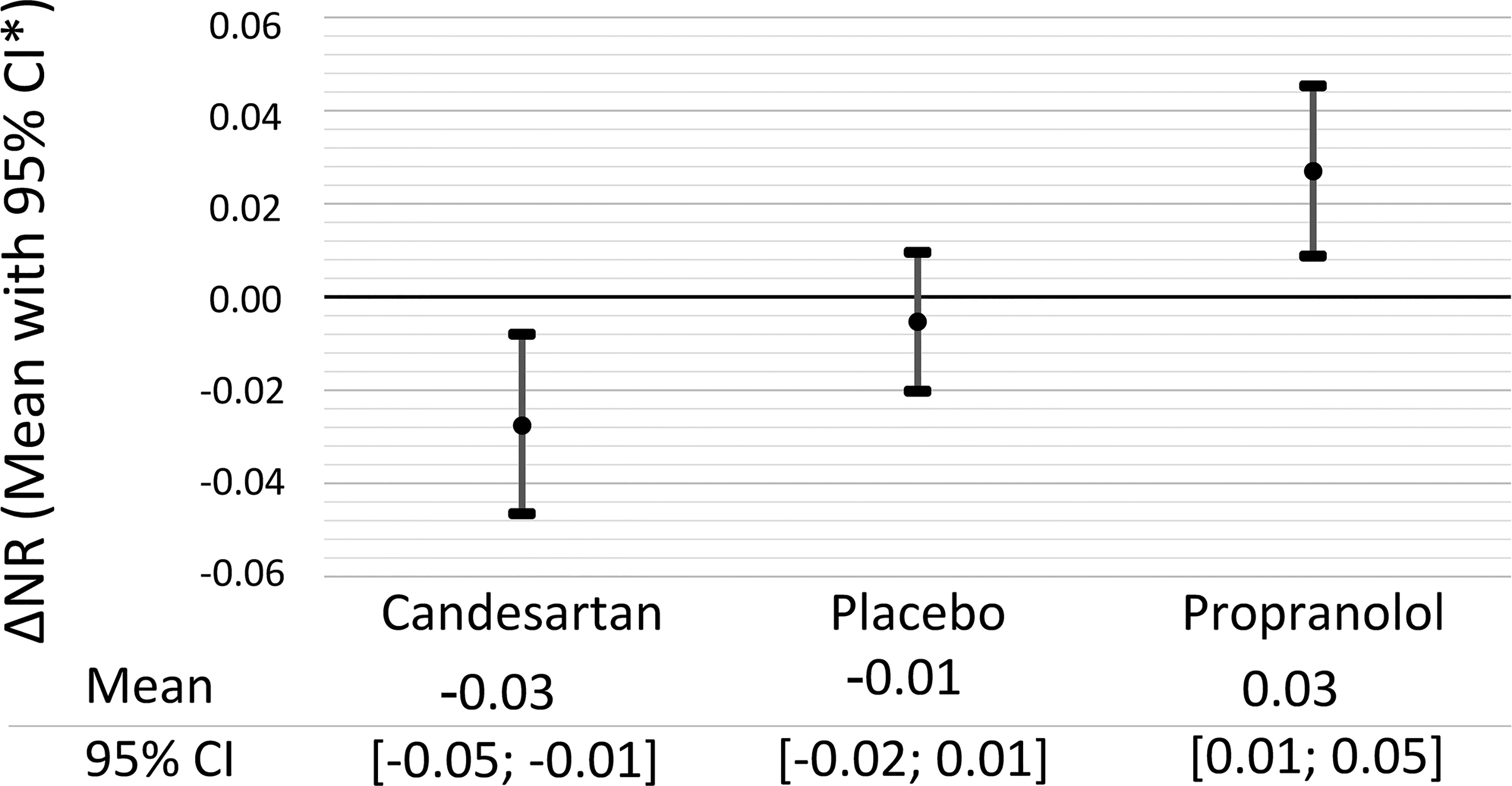
Change in notch ratio from baseline to treatment. *95% confidence interval.
Of all patients with both valid blood pressure recordings and complete diary data, 42% (22/53) improved clinically (fewer days with migraine) on candesartan, 40% (22/55) on propranolol, and 24% (13/55) on placebo. Table 2 shows the baseline values and changes in notch ratio for drug responders and nonresponders on each drug and placebo. The notch ratio was similar at baseline between responders and nonresponders, both for candesartan (p = 0.79) and propranolol (p = 0.36). However, nonresponders to candesartan had a greater reduction in notch ratio than responders (p = 0.028) (Table 2).
Patients had a slightly higher brachial systolic blood pressure and mean arterial blood pressure (136 mmHg and 104 mmHg) than controls (130 mmHg and 98 mmHg) (p = 0.004 and p = 0.002, respectively). There was no difference in baseline systolic or mean blood pressure between responders and nonresponders to candesartan (p = 0.15 and p = 0.39) or propranolol treatment (p = 0.75 and p = 0.76).
Discussion
The present study demonstrates that the two drugs tested have opposite effects on the notch ratio, a proxy for arterial compliance, and yet both are efficient as prophylactic medication. This suggests that there must be drug effects other than those upon peripheral arterial compliance that are responsible for their prophylactic effect in migraine patients. Also, there was no difference in baseline notch ratio values between patients and controls, indicating that vessel wall dynamics measured with the methods of the present study give little information on the predisposition to migraine.
A previous study supports our findings that candesartan lowers the notch ratio, reflecting more compliant arteries and vasodilation during treatment with candesartan.
10
We are not aware of any human study investigating the effect of propranolol on the notch ratio. However, no effect was found in a rabbit study,
15
and furthermore, in humans, pulsed Doppler velocimetry revealed no effect of propranolol upon arterial compliance.
16
Interestingly, however, initial studies on the use of nonselective β-blockers in patients with peripheral artery diseases revealed a series of complications such as cold extremities, absent peripheral pulses, and in some cases cyanosis.
11
This suggests that propranolol has an opposite effect on arterial compliance to that of candesartan, favoring vasoconstriction (Figure 2). There are several studies that suggest that a nonselective β-blocker can cause or worsen Raynaud
We examined the arterial compliance as assessed by the height ratio of the dicrotic notch in several settings, and the results were consistent: no difference between migraineurs and controls at baseline, and no baseline difference between drug responders and nonresponders. We are not aware of previous such investigations in migraine patients.
The numbers of clinical responders and nonresponders to prophylactic migraine treatment differ somewhat from the original study of Stovner et al., 12 because in the present study only patients with both valid Finapres blood pressure recordings and complete diary data were analyzed.
During treatment with candesartan, the notch ratio decreased in nonresponders, reflecting increased vascular compliance, but not in responders (p = 0.028). It is difficult to interpret the physiological meaning of this. The finding may be spurious, or may indicate that migraineurs with clinical effect of candesartan are less responsive to the vascular effects of the drug. On propranolol, no such difference was seen.
Since both of the studied drugs have prophylactic effects on migraine attacks, but opposite effects on arterial wall dynamics, alternative organ targets for drug effects should be sought. Both drugs are fat-soluble and cross the blood–brain barrier, 24,25 but their molecular mechanisms of action in the nervous system are not fully understood. Beside its cardiovascular use, propranolol is used for treatment of anxiety symptoms 24 and essential tremor. There is hence a possibility that the prophylactic effect on migraine is mediated through effects on the central nervous system. β-blockers may influence cortical excitability via noradrenaline and serotonin repressing systems and affect cortical spreading depression. 26 Beta 1 receptor blockade may inhibit the release of noradrenaline, and β-blockers may also reduce serotonergic activity by blocking 5-HT receptors. An inhibitory effect of propranolol on retinal spreading depression has been shown, 27 and a rat study has demonstrated that propranolol blocks cortical spreading depression. 28
As to the ARBs, they may definitely influence migraine by mechanisms other than effects on blood pressure and the vasculature through the peripheral renin–angiotensin system. Notably, there is a renin–angiotensin system within the brain, working separately from that in the periphery, and known to be involved in pain regulation. 29
The associations between migraine and endothelin have recently been reviewed. 30 Migraine is associated with genes that influence endothelin activity. 5 There are increased concentrations of endothelin-1 both during the pre-ictal aura phase and the ictal headache phase, and both β-blockers 31 and ARBs 32 decrease production and release of endothelin-1. Thus, endothelin-1 may be a common candidate mediator for the preventive effect of the two drug classes in migraine, despite their opposite effects on arteries.
There is a possibility that the prophylactic effect on migraine is due to lowering blood pressure. Patients had slightly higher systolic and mean arterial blood pressure than controls. However, the difference was not substantial (systolic blood pressure 136 mmHg and 130 mmHg, mean arterial blood pressure 104 mmHg and 98 mmHg in patients and controls, respectively), and there were no differences in blood pressure between responders and nonresponders for either drug. Therefore, we could not find a relation between the blood pressure-reducing effects of the drugs and the prophylactic effect in migraine patients. However, studies suggest that different types of antihypertensive drugs may give a prophylactic effect in migraine patients. 33
Strengths and limitations
Major strengths of the study are the randomized double-blind placebo-controlled crossover design, and the application of a vascular physiological method on the clinical neurological puzzle that the two drugs studied have opposing effects on arterial wall dynamics, yet despite this, both reduce the number of migraine attacks.
A limitation of the present study is that the measurements were performed when neither prodromal nor migraine symptoms were present. Possibly, the drug-induced changes in arterial compliance relevant for the drug response might only be detectable during migraine attacks, when for instance endothelin levels are increased. 30
The Finapres measurements give information on peripheral arterial wall dynamics only, and we cannot exclude the possibility that measures of intracerebral arterial compliance could yield different results.
Our findings suggest that arterial compliance is equal in patients and controls, however, we cannot conclude with certainty that arterial wall dynamics do not play a role in migraine pathophysiology. This may be more important when addressing acute medication rather than prophylaxis.
In cases with an early and high location of the dicrotic notch, the notch was difficult to detect reliably, and this sometimes led to rejection (7.2%), leading to a slightly higher proportion of curves with late, low dicrotic notch locations.
Several methods can be used to assess arterial compliance. Forearm flow-mediated dilation, carotid-femoral flow velocity, and augmentation index from arterial pulse curves have all been studied in migraine patients compared to healthy controls, with divergent results. 7 In the present study, the only method available was the dicrotic notch ratio.
The patients’ age affects arterial pulse curves. However, the control group and the migraine patients were matched for age and sex, and the drug study had a crossover design. The study was negative, with no significant group differences, no prespecified hypothesis on age effects, and a limited number of patients. For these reasons we refrained from subgroup analyses.
Conclusion
Since candesartan and propranolol have opposite effects upon vascular compliance, but both drugs reduce headache frequency, this study suggests that vessel wall dynamics do not play a major role in the mechanism of action for prophylactic treatment of migraine.
Clinical implications
The clinical prophylactic effects of candesartan and propranolol in migraine patients seem to be unrelated to the drugs’ vasoactive effects.
Candesartan causes peripheral vasodilation and propranolol causes peripheral vasoconstriction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. The study of Stovner et al. 12 received an unconditional grant of NOK 500.000, and study drugs were provided by AstraZeneca. The study design, data collection, analysis, and interpretation were done without involvement from the company. The present study has not received external funding.
