Abstract
Introduction
Galcanezumab is a humanized monoclonal antibody binding calcitonin gene-related peptide, used for migraine prevention.
Methods
A global, double-blind, 6-month study of patients with episodic migraine was undertaken with 915 intent-to-treat patients randomized to monthly galcanezumab 120 mg (n = 231) or 240 mg (n = 223) or placebo (n = 461) subcutaneous injections. Primary endpoint was overall mean change from baseline in monthly migraine headache days. Key secondary endpoints were ≥50%, ≥ 75%, and 100% response rates; monthly migraine headache days with acute migraine medication use; Patient Global Impression of Severity rating; the Role Function-Restrictive score of the Migraine-Specific Quality of Life Questionnaire.
Results
Mean monthly migraine headache days were reduced by 4.3 and 4.2 days by galcanezumab 120 and 240 mg, respectively, and 2.3 days by placebo. The group differences (95% CIs) versus placebo were 2.0 (−2.6, −1.5) and 1.9 (−2.4, −1.4), respectively. Both doses were superior to placebo for all key secondary endpoints. Injection site pain was the most common treatment-emergent adverse event, reported at similar rates in all treatment groups. Both galcanezumab doses had significantly more injection site reactions and injection site pruritus, and the 240 mg group had significantly more injection site erythema versus placebo.
Conclusions
Galcanezumab 120 or 240 mg given once monthly was efficacious, safe, and well tolerated.
Study identification
EVOLVE-2; NCT02614196; https://clinicaltrials.gov/ct2/show/NCT02614196.
Trial Registration
NCT02614196.
Introduction
Although the worldwide prevalence of migraine and its associated socioeconomic cost are well recognized (1–3), it is commonly inadequately treated (4,5). The American Migraine Prevalence and Prevention study revealed that 54% of patients with migraine needed bed rest or reported severe impairment, and 39% reported some impairment (6). Moreover, although prevention treatment was indicated in 39% of patients with migraine, only 13% received it (6). In addition, up to 68% of patients who use preventive medications stop doing so within 6 months because of insufficient benefit, dissatisfaction with the drug, or poor tolerability (7–9). There is a significant need for new treatment options with improved efficacy and tolerability.
Recent clinical investigations have pointed to the critical role of calcitonin gene-related peptide (CGRP), which is expressed throughout the nervous system and is prominent in the trigeminovascular system, in migraine pathophysiology (10–13). It is implicated in sensory neuromodulation, vasodilation, and mediation of neurogenic inflammation (12–14). Migraine attacks are accompanied by elevated blood levels of CGRP, and the infusion of CGRP induces headache in individuals with migraine (10,11,15–17). Selective antagonists to the CGRP receptor were efficacious in clinical trials for acute migraine (12). Galcanezumab is a humanized monoclonal antibody that binds CGRP and prevents its biological activity without blocking the CGRP receptor. Two Phase 2 clinical trials have shown that galcanezumab (120 and 300 mg) was safe and efficacious in migraine prevention (18,19). In the current manuscript, we hypothesized that at least one dose of galcanezumab was superior to placebo over a 6-month dosing regimen. Accordingly, we provide the results of a large, global, Phase 3 trial of galcanezumab that confirms its efficacy and safety in patients with episodic migraine.
Methods
Study design
EVOLVE-2 (NCT02614196) was a Phase 3, multi-center, placebo-controlled, double-blind, randomized clinical trial analyzing the efficacy and safety of two dosing regimens of galcanezumab in patients with episodic migraine. This study was conducted between January 2016 and March 2017 at 109 study sites in the United States, United Kingdom, Netherlands, Spain, Czech Republic, Germany, Argentina, Israel, Korea, Taiwan, and Mexico. Of these sites, 49 (45%) were in the US. The study was composed of four study periods (Figure 1). Study period I included medical examinations and washout of migraine preventive medications for ≥30 days (4 months for onabotulinumtoxin A). Study period II established the baseline number of migraine headache days (MHDs). Study period III was a 6-month double-blind treatment phase. During the post-baseline periods, patients were allowed to take acute migraine medications as needed, with the exception of medications containing opioids or barbiturates, which were limited to no more than three times per month. Study period IV was a 4-month post-treatment (washout) period and is not a topic of this manuscript. Key elements of the protocol are available at https://clinicaltrials.gov/ct2/show/NCT02614196.
Study protocol. aPatients randomized to the galcanezumab 120 mg dose received a loading dose of 240 mg at the first injection only (visit 3).
In all, 825 patients were planned to be randomized in a 2:1:1 ratio of placebo to galcanezumab 120 mg/month or 240 mg/month (target of 206 patients). It was estimated based on Monte Carlo simulations that this sample size would provide approximately 95% power that at least one dose level of galcanezumab would separate from placebo on the primary endpoint, at an overall one-sided 0.025 significance level, assuming a standardized treatment effect size of 0.33 and a 26% discontinuation rate.
Inclusion criteria
Enrolled patients had to be between the ages of 18 and 65 years (inclusive) with a diagnosis of migraine with or without aura (1.1 and 1.2), defined by International Headache Society (IHS) International Classification of Headache Disorders, 3rd edition, (ICHD-3) beta version (3). Patients were enrolled into the study by study staff, through referral or advertising. Included patients had migraine for at least 1 year prior to enrollment, migraine onset prior to age 50 years, 4–14 MHDs (including probable MHD), at least two migraine attacks during the baseline period, and an 80% compliance rate in using the electronic diary (ePRO). Finally, patients had to agree to use an acceptable method of birth control during the study and for at least 5 months afterwards. The study protocol was reviewed and approved by the appropriate institutional review board for each of the study sites, and the study was conducted according to Good Clinical Practice and the Declaration of Helsinki guidelines. Patients provided written informed consent before undergoing study procedures. Investigators at each study site evaluated and confirmed eligibility, obtained consent, and enrolled the patients.
Exclusion criteria
Patients were excluded if they had failed treatment with three or more migraine prevention drugs from different classes (level A or B evidence per American Academy of Neurology guidelines for episodic migraine prevention) (20) or if they were using opioids or barbiturates more than twice per month. Other conditions leading to exclusion were participation in another clinical trial within the past 30 days, prior exposure to galcanezumab or any another CGRP antibody, taking any therapeutic antibody in the past 12 months, known hypersensitivity to multiple drugs, or presence of any medical or psychiatric illness that would preclude study participation.
Randomization and blinding
Randomization to double-blind treatment was performed at visit 3. Patients were assigned in a double-blind manner by a computer-generated randomization sequence using an interactive web-response system (IWRS) to placebo or galcanezumab 120 or 240 mg (2:1:1 ratio) administered by subcutaneous injection once monthly for 6 months. Patients randomized to the 120-mg dose received a loading dose of 240 mg (two injections of 120 mg each) at the first dosing visit. To achieve balance among groups, randomization was stratified by country and migraine frequency (<8 vs. ≥8 MHDs/month) at baseline. All injections were administered by the investigator sites’ trained personnel. To preserve blinding, galcanezumab and matching placebo (i.e. excipients only) were supplied as 1-mL, single-dose, prefilled, disposable manual syringes with study-specific labels. The syringes were visibly indistinguishable from each other. Patients in all groups received two injections at each dosing visit to preserve blinding throughout the study.
Outcome measures
Efficacy analyses were performed in the intent-to-treat (ITT) population, where each randomized patient had received at least one dose of galcanezumab or placebo. The primary objective was to assess whether at least one of the studied dosing regimens of galcanezumab was superior to placebo in the prevention of migraine headache in patients with episodic migraine. The primary analysis evaluated the efficacy of each dosing regimen of galcanezumab compared with placebo on the overall mean change from baseline in the number of monthly MHDs during the 6-month treatment phase, based on the ePRO data. The key secondary outcome measures were the following:
The mean proportion of patients with reduction from baseline of ≥50%, ≥75%, and 100% in monthly MHDs during the 6-month double-blind treatment phase. Specifically, response rate was defined as the percentages of patients meeting predefined thresholds (i.e. ≥50%, ≥75%, and 100%) in the reduction from baseline in the number of MHDs for each month, and the overall percentages of patients meeting these thresholds averaged over months 1 through 6 were analyzed. The mean change from baseline in the Role Function-Restrictive (R-FR) domain score of the Migraine-Specific Quality of Life Questionnaire version 2.1 (MSQ v2.1), as an average of months 4–6. The overall mean change from baseline in the number of MHDs with acute migraine medication use during the 6-month double-blind treatment phase. The mean change from baseline in the Patient Global Impression of Severity (PGI-S) rating (average of months 4–6).
A secondary, but non-key, outcome measure that is included in the current manuscript is the Migraine Disability Assessment (MIDAS) total score at month 6.
Safety and tolerability
The safety population consisted of all randomized patients who received at least one dose of galcanezumab or placebo. Adverse events were coded by Medical Dictionary for Regulatory Activities (version 19.1). The safety parameters reported in the present manuscript are the treatment-emergent adverse events (TEAEs), serious adverse events (SAEs), deaths, discontinuations due to adverse events, discontinuation rates, vital signs, body weight, and immunogenicity. A comprehensive safety report is in preparation as a separate manuscript.
Assessments
The ePRO daily diary was used to record headache and other migraine symptoms. Based on the diary data, using an automated algorithm, the days were categorized as MHDs (including probable MHD). A probable migraine is defined as a headache with or without aura and lasting ≥ 30 minutes but missing one of the migraine features in the ICHD-3 beta criteria (3). The monthly number of MHDs with acute migraine medication use was obtained through the ePRO diary, and the PGI-S, MSQ, and MIDAS assessments were performed at the study site at every monthly visit for PGI-S and MSQ and on months 3 and 6 for MIDAS with the use of a slate device.
Statistical analyses
The changes from baseline to each scheduled postbaseline measurement for continuous efficacy measures were estimated for each treatment from mixed effect model repeat measurements (MMRM) analyses. This model included the fixed, categorical effects of treatment, pooled country, month, and treatment-by-month interaction, as well as the continuous, fixed covariates of baseline value and baseline-by-month interaction. The primary endpoint of overall mean change in monthly MHDs was estimated as the main effect of treatment (each galcanezumab dose group and placebo) during the 6-month treatment phase. This provided the average treatment effect across the 6-month double-blind treatment phase.
When the objective of the continuous secondary efficacy measures was to assess overall mean change during the 6-month treatment phase, the endpoint for comparing galcanezumab with placebo was the main effect of treatment from the MMRM analysis across months 1–6. Change from baseline of continuous variables with repeated measures included the change from baseline for MHD with acute medication use for months 1–6, as well as MSQ R-FR and PGI-S for months 4–6. The categorical longitudinal efficacy measures of ≥50%, ≥75%, and 100% reduction from baseline in the number of monthly MHD from months 1–6 were analyzed with a categorical, pseudo-likelihood-based repeated measures analysis. A superchain (multiple testing) procedure was used to ensure strong control of the familywise type I error rate for the primary and key secondary endpoints (Figure 2) (21). According to the specified procedure, testing of key secondary endpoints was to commence sequentially after testing of the primary endpoint. The superchain procedure provides appropriate adjustment for multiple testing across the two dose levels and all primary and key secondary endpoints. Categorical safety measures included both scheduled and unscheduled visits and were compared among treatment groups using Fisher’s exact test. For continuous safety measures, changes from baseline were assessed using an ANCOVA model. All statistical analyses were performed with the use of SAS Enterprise Guide 7.1 (SAS Institute, Cary, NC).
Superchain Multiple Testing Procedure. For the superchain multiple testing procedure, Dunnett’s test was used to test the primary null hypotheses. When at least one of the two primary null hypotheses was rejected, the Hochberg procedure was to be used to test the 50% and 75% response rate endpoints at the corresponding dose(s). Upon rejection of these null hypotheses, the Bonferroni-Holm procedure was to be used to test the change in the use of acute migraine treatment and the change in functioning for the MSQ RF-R, at the corresponding doses. After rejection of these null hypotheses, the hypotheses of 100% response rate and PGI-S were tested sequentially at the corresponding doses. When all null hypotheses for a single dose were rejected, available alpha was to be propagated to the other dose for continued testing of available nonrejected null hypotheses. In the diagram, propagation weights are denoted along edges connecting boxes, and families of hypotheses are shown in the boxes.
Results
Patient disposition and baseline characteristics
A total of 1696 patients entered study screening, and 922 were randomized (Figure 3). The most common reason for patients failing the screening phase was not having the required number of MHDs. The ITT population consisted of 915 patients who received at least one dose of galcanezumab or placebo, 785 patients (85.8%) completed the double-blind treatment phase, and 129 (14.1%) patients discontinued during study period III. The most common reason for discontinuation was the patient’s decision to withdraw, accounting for 64 (7%) patients. Adverse events (AEs) accounted for the discontinuation of 8 (1.7%) placebo patients, 5 (2.2%) galcanezumab 120-mg patients, and 9 (4.0%) galcanezumab 240-mg patients, and were not significantly different among the groups. In addition, 10 (2.2%) placebo patients, 7 (3.0%) galcanezumab 120-mg patients, and 0 (0%) galcanezumab 240-mg patients were lost to follow-up.
CONSORT diagram showing patient disposition throughout the trial.
Baseline demographics and disease characteristics.

Primary and *key secondary outcome measures. Other characteristics may be associated with non-key secondary measures and (except for MIDAS) fall outside the scope of this manuscript. They will be discussed in an upcoming publication.
North America includes Mexico and the United States.
MHD: migraine headache day; MIDAS: Migraine Disability Assessment; MSQ RF-R: Migraine-Specific Quality of Life Questionnaire Role Function-Restrictive; n: the intent-to-treat population for each dosing regimen; PGI-S: Patient Global Impression of Severity; SD: standard deviation.
Efficacy and measures of functioning
Primary objectives, key secondary objectives, and the MIDAS.
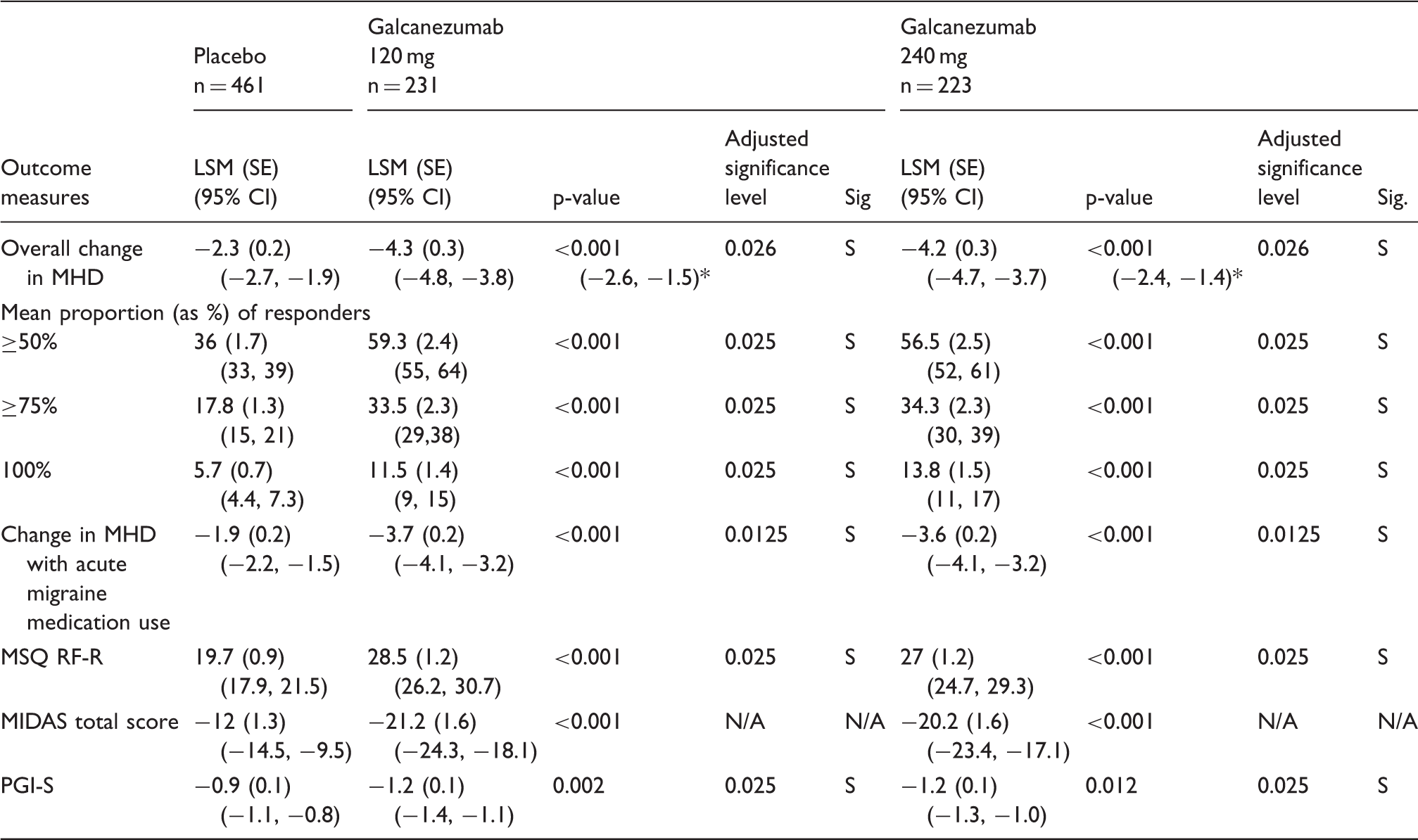
Data are presented as the change from baseline. P-values are the given versus placebo. The adjusted significance level represents the significance threshold adjusted for multiplicity. If p-value is less than or equal to the adjusted significance level, then the results are statistically significant after adjustment for multiplicity. MIDAS was not a key secondary outcome measure, and was not subjected to multiplicity adjustment.
Indicates the 95% CI for the difference from placebo for the primary endpoint (i.e. overall change in MHD).
CI: Confidence intervals; LSM: least squares mean; MHD: migraine headache day; MIDAS: Migraine Disability Assessment; MSQ RF-R: Migraine-Specific Quality of Life Questionnaire Role Function-Restrictive; n: the intent-to-treat population for each dosing regimen; N/A: not applicable; PGI-S: Patient Global Impression of Severity; PL: placebo; S: significant; SE: standard error, Sig: significance.
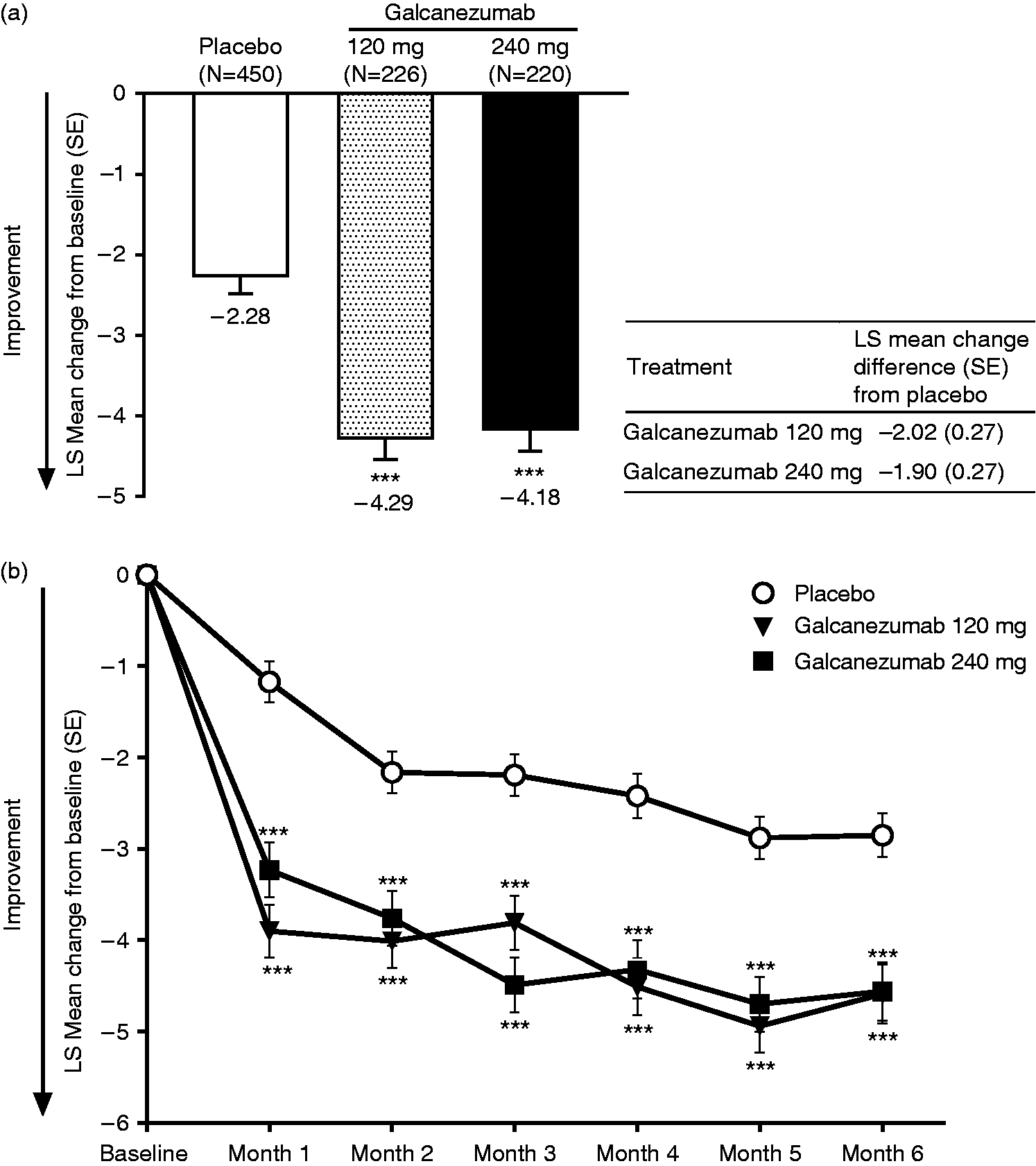
(a) The overall LS mean change from months 1 through 6 in monthly migraine headache days. The inset table shows the LS mean change from placebo for both doses of galcanezumab; and (b) the LS mean change at each of months 1 through 6 in monthly migraine headache days.
Significantly (p < .001) greater mean proportions of patients in the galcanezumab 120 and 240 mg groups experienced ≥ 50% reduction in MHDs compared to the placebo group (59% and 57% vs. 36%, p < .001; Figure 5). Additionally, significantly greater mean proportions of patients in the galcanezumab 120 and 240 mg groups experienced ≥ 75% and 100% reduction in monthly MHDs compared to the placebo group (Figure 5). Galcanezumab 120 and 240 mg resulted in significant (p < .001) reductions in MHDs with acute migraine medication use compared to the placebo group (Figure 6).
The mean percent of patients with ≥ 50%, ≥ 75%, and 100% reductions in monthly migraine headache days. The LS mean change in migraine headache days with the use of acute migraine medications.

Treatment with both dosing regimens of galcanezumab was associated with reduced functional impairment due to migraine as measured in the MSQ RF-R. Both galcanezumab 120 and 240 mg significantly (p < .001) improved the LS means of the MSQ RF-R score averaged over months 4−6 versus placebo (Figure 7). Both galcanezumab groups showed improvement in patients’ global impression of severity of their disease as assessed by PGI-S rating (Figure 8). The LS mean change in PGI-S ratings for the galcanezumab 120 and 240 mg groups were significantly (p = .002 and p = .012, respectively) greater than in the placebo group. In addition, both treatment groups of galcanezumab significantly (p < .001) improved total MIDAS scores at month 6, which was the prespecified time point of interest, compared to the placebo group (Figure 9).
The LS mean change in the Migraine-Specific Quality of Life Questionnaire Role Function-Restrictive domain. The LS mean change in the Patient Global Impression-Severity rating. The LS mean change in the Migraine Disability Assessment score.


Safety and tolerability
Overview of adverse events.

P-values vs. placebo. No p-values were computed for “Deaths,” since all values were 0.
AEs: adverse events; n: the safety population for each dosing regimen; TEAEs: treatment-emergent adverse events.
Treatment-emergent adverse events ≥2% Galcanezumab total.

P-values vs. placebo.
n: the safety population for each dosing regimen; TEAE: treatment-emergent adverse event.
Immunogenicity
At baseline, prior to randomization, anti-drug antibodies (ADAs) were detected in 8.4%, 8.1%, and 11.2% of patients in the placebo, galcanezumab 120 mg, and galcanezumab 240 mg groups, respectively. There were 19 (8.6%), 11 (5.1%), and 2 (0.5%) patients in the galcanezumab 120 mg, galcanezumab 240 mg, and placebo groups, respectively, with treatment-emergent ADA. The difference between each galcanezumab treatment group and placebo was statistically significant (p < .001). Among all treatment-emergent ADA+patients, 29 of them had neutralizing ADAs present. The ADA titers were generally low. Titers ranged from not detectable to a maximum of 1:1280, which occurred in four patients. Patients who were ADA+ at any time and who also reported a TEAE had titers ranging from 1:10 to 1:640. Treatment-emergent ADAs or neutralizing ADAs had no impact on either safety or efficacy.
Discussion
Treatment with the currently available migraine prevention drugs is associated with a relatively poor tolerability and low rates of patient satisfaction (8,9). Consequently, the prevention of migraine remains an important unmet clinical need. The results of this large Phase 3 randomized clinical trial demonstrate that galcanezumab is well-tolerated and efficacious in preventing migraine headache.
Both dose regimens of galcanezumab met the primary and all key secondary endpoints. Galcanezumab significantly reduced the monthly MHDs relative to placebo within the first month of the trial and throughout the 6-month treatment period, representing an annualized gain of approximately 7 weeks of migraine-free days. In addition, a majority of galcanezumab-treated patients had response rates of ≥50%, a well-established benchmark indicating a clinically meaningful result (22). Similarly, ≥75% and 100% response rates were also statistically significantly greater in both galcanezumab treatment groups relative to placebo. Treatment with both dose regimens of galcanezumab was also associated with a significant decrease in the days of use of acute migraine medications. This is an important consideration because their overuse may lead to the development of medication overuse headache (3,10). Galcanezumab was also found to be superior to placebo in reducing the migraine-related impairment in functioning in the MSQ R-FR and the MIDAS rating scales. Both dose regimens of galcanezumab significantly improved the patients’ global impression of severity of disease (PGI-S) relative to placebo. Taken together, the results of this global clinical trial indicate that treatment with galcanezumab (a) demonstrates a clinically meaningful level of efficacy in preventing migraine headaches in patients with episodic migraine and (b) replicates the findings of the identically designed North American study (EVOLVE-1) (23).
Injection site pain, the most common TEAE, was reported by approximately 9% of patients, regardless of treatment. TEAEs significantly more commonly reported by the galcanezumab-treated, versus placebo-treated, patients, were all injection site related: Injection site reaction, injection site pruritus, injection site swelling, and injection site erythema; most were mild to moderate, and none were serious. Only four patients receiving galcanezumab discontinued the study due to injection site reaction. Discontinuations due to TEAEs were low, as was the rate of SAEs, and they did not differ among the treatment groups. Together, these results show that galcanezumab is well tolerated, with a small number of patients experiencing relatively minor injection-site related adverse effects.
Galcanezumab presents a promising investigational drug for migraine prevention. By the end of the 6-month treatment period, more patients receiving placebo than those receiving galcanezumab discontinued from the study for any reason. Similarly, the number of placebo-treated patients who discontinued the study due to lack of efficacy was three times as high as the corresponding number of the galcanezumab-treated patients. These results are especially encouraging when one considers that as many as two-thirds of patients using currently available preventive therapies discontinue their use because of poor tolerability or lack of efficacy over a 6-month period (7–9). Notably, this study also included patients who had tried and failed several preventive treatments from up to two different classes.
Limitations of the study include certain restrictions in the inclusion criteria that may limit the generalizability of the results. Patients with serious medical or psychiatric conditions, high body mass index, substantial opioid use, and high risk for major cardiovascular events were excluded. Although the study was of a relatively large size and long duration, it may not be sufficient to detect possible rare adverse events or long-term risks.
Conclusions
Galcanezumab treatment was associated with a statistically significant and clinically meaningful reduction in monthly MHDs at doses of 120 and 240 mg given subcutaneously once monthly. Moreover, patients treated with either dose of galcanezumab reported a reduction in the migraine-related impairment of function and a significant reduction in migraine-related disability, while also reducing the use of acute migraine medications. Galacanezumab treatment was safe and well tolerated. No clinically meaningful differences in either efficacy or safety were found between the two galcanezumab dose regimens. The results of this investigation confirm findings from several previously published migraine prevention studies of galcanezumab.
Clinical implications
Galcanezumab is safe and efficacious in preventing migraine. Galcanezumab reduces migraine-related disability and impairment in functioning. The study completion rates of the galcanezumab-treated groups were higher than that of placebo.
Footnotes
Acknowledgments
Most importantly, the authors thank the patients who participated in the study, and the following investigators and their respective institutions: William D Koltun, MD, Medical Center for Clinical Research; Marigene D Salazar Sharma, MD, Albuquerque Clinical Trials; Michael Ament, MD, MBA, IMMUNOe International Research Centers; Itay D Melamed, MD, IMMUNOe International Research Centers; Eugene Andruczyk, DO, Clinical Research of Philadelphia; Lesley M Arnold, MD, University of Cincinnati College of Medicine; Thomas V Ballard, MD, Mountain View Clinical Research, Inc; William D Byars, MD, Mountain View Clinical Research, Inc; Mushtque Chachar, MD, Novex Clinical Research; Mohammad Munir, MD, Novex Clinical Research; Herman J Downey, MD, Westside Center for Clinical Research; Michael J Downing, MD, FutureSearch Trials; Alexander P Feoktistov, MD, PhD, Diamond Headache Clinic; Chester L Fisher, Jr, MD, MPH, Health Research of Hampton Roads Inc; Kristi K George, MD, Josephson Wallack Munshower Neurology; Ira D Glick, MD, Pacific Research Partners, LLC; Dan C Henry, MD, Foothill Family Clinic; Jose C Rafecas, MD, Neurology & Neuroscience Associates, Inc.; Cynthia L Huffman, MD, Meridien Research; Lissette Jimenez, MD, SomniCare Sleep Institute; Nandita M Joshi, MD, Clinical Neuroscience Solutions Inc; Shawn K Hassler, MD, Optimus Medical Group; Timothy M Koehler, DO, Heartland Research Associates; Rise A Futterer, MD, MPH, Dean Foundation for Health Research and Education; Amelito B Malapira, MD, Sentara Neurology Specialists; Mia Moon, MD, Catalina Research Institute, LLC; Gilbert J Martinez, MD, Catalina Research Institute, LLC; Braden T Nago, MD, POLYCLINIC; Margarita Nunez, MD, High Point Clinical Trials Center; Ashok K Patel, MD, Bio Behavioral Health; Larry D Reed, MD, PhD, Healthcare Research Network – Hazelwood; Alan J Reichman, MD, Clinical Trial Network; Barry J Riskin, MD, Christie Clinic, LLC; Aamer Habib, MD, Christie Clinic, LLC; Christian E Schenk, MD, Cortex, PSC; Jose M Itzcovich-Schuster, MD, Schuster Medical Research Institute; Cynthia B Strout, MD, Coastal Carolina Research Center, Inc.; Duncan Sze-Tu, MD, Regional Clinical Research; Louise A Taber, MD, Arizona Research Center; Louise M Thurman, MD, MPH, IPS Research Company; Joshua A Tobin, MD, Xenoscience; Nicholas G Vatakis, MD, SPRI Clinical Trials, LLC.; Robert A Riesenberg, MD, Atlanta Center of Medical Research; Arifulla Khan, MD, Northwest Clinical Research Center; Steven B Graff-Radford, DDS, Cedars Sinai Medical Center; Alan C Newman, DDS, Cedars Sinai Medical Center; Jeffrey B Rosen, MD, Clinical Research of South Florida; Jeffrey White, MD, Urgent Care Specialists, LLC; Narinder Saini, MD, Urgent Care Specialists, LLC; Steve H Choi, MD, Urgent Care Specialists, LLC; JienSup Kim, MD, Axiom Research; Abraham J Nagy, MD, Nevada Headache Institute; Kelly N Taylor, MD, Sensible Healthcare; Thomas G Ledbetter, MD, ClinPoint Trials, LLC; Katie A Julien, MD, Jordan River Family Medicine; Harvey D Schwartz, MD, Sunrise Clinical Research; Fayyaz Ahmed, MD, Hull Royal Infirmary; Brendan Davies, MD, Royal Stoke University Hospital; Peter Goadsby, MD, PhD, Kings College Hospital; Alok Tyagi, MD, Queen Elizabeth University Hospital; Nicholas Silver, MD, PhD, Walton Centre for Neurology and Neurosurgery; Emile Couturier, MD, Boerhaave Medisch Centrum; Wim Mulleners, MD, Canisius-Wilhelmina Ziekenhuis; Michel Ferrari, MD, PhD, Leids Universitair Medisch Centrum; Veronique Triebels, MD, Zuyderland Medisch Centrum; Peter van den Berg, MD, PhD, Isala Klinieken; Guus Schoonman, MD, PhD, TweeSteden Ziekenhuis; Angel Luis Guerrero Peral, MD, Hospital Universitario de Valladolid; Jose Miguel Lainez Andres, MD, PhD, Hospital Clínico Universitario de Valencia; Agustin Oterino Duran, MD, Hospital Universitario Marques De Valdecilla; Patricia Pozo Rosich, MD, PhD, Hospital Universitari Vall d’Hebron; Vicente Medrano Martinez, MD, Hospital General De Elda; Joan Prat Rojo, MD, Hospital Universitari de Bellvitge; Michal Bajacek, MD, PhD, Neurologicka ordinace; David Dolezil, MD, PhD, MBA, DADO MEDICAL, s.r.o.; Fabienne Kosova, MD, Clintrial, s.r.o.; Yuliya Rizova, MD, Clintrial, s.r.o.; Hynek Lachmann, MD, Axon Clinical, s.r.o.; Irena Novotna, MD, Fakultni Nemocnice U svate Anny; Klaudia Vodickova-Borzova, MD, Brain-Soultherapy s.r.o; Andreas Böger, MD, DRK-Kliniken Nordhessen; Matthias Böhringer, MD, NeuroZentrum Bielefeld; Stefan Braune, MD, Neurozentrum Prien; Tobias Freilinger, MD, Klinikum der Eberhard-Karls-Universität Tübingen; Charly Gaul, MD, Migräne- und Kopfschmerzklinik GmbH & Co. KG; Heike Israel-Willner, MD, MBA, Charité Universitätsmedizin Berlin; Torsten Kraya, MD, Martin-Luther-University Halle-Wittenberg; Arne May, MD, PhD, Universitätsklinikum Hamburg-Eppendorf; Jörg Peltz, MD, PhD, Neuropsychiatrisches Facharztzentrum; Kasja Solbach, MD, Universitätsklinikum Essen; Dagny Holle-Lee, MD, Universitätsklinikum Essen; Peter Storch, MD, Klinikum der Friedrich-Schiller-Universität Jena; Philipp Stude, MD, Praxis Dr. Philipp Stude; Conrado J Estol, MD, PhD, Stat Research; Bibiana B Saravia, MD, Mautalen Salud e Investigacion-Centro de Osteopatías Médicas; Gustavo Fischbein, MD, Instituto de Investigaciones Metabolicas; Osvaldo CJ Bruera, MD, Instituto de Investigaciones Metabolicas; Fernanda M Paez, MD, Aprillus Asistencia e Investigacion - Servicio de neurologia; Yaron River, MD, Hillel Yaffe Medical Center; David Yarnitsky, MD, Rambam Medical Center; Yelena Feingold, MD, Chaim Sheba Medical Center; Gabriel Vainstein, MD, Maccabi Healthcare Services, Kfar Saba; Soo-Jin Cho, MD, PhD, Hallym University Dongtan Sacred Heart Hospital; Chin-Sang Chung, MD, PhD, Samsung Medical Center; Byung Kun Kim, MD, PhD, Eulji General Hospital; Man Ho Kim, MD, PhD, Seoul National University Hospital; Kyungmi Oh, MD, Korea University Guro Hospital; Heui Soo Moon, MD, PhD, Kangbuk Samsung Hospital; Min Kyung Chu, MD, PhD, Hallym University of Medicine, Kangnam Sacred Heart Hospital; Kao-Chang Lin, MD, Chi-Mei Medical Center – Yung Kang; Shiang-Ru Lu, MD, Kaohsiung Medical University Chung-Ho Memorial Hospital; Po-Jen Wang, MD, Sin-Lau Hospital; Shuu-Jiun Wang, MD, Taipei Veterans General Hospital; Tzu-Hsien Lai, MD, Far Eastern Memorial Hospital; Freddy G Castro Farfan, MD, Grupo Médico Camino SC; Jose A Gien Lopez, MD, Medical Care and Research, S.A. de C.V.; Elmer G Lopez Meza, MD, Neurociencias Estudios Clinicos; Jose H Nicolini Sanchez, MD, PhD, Grupo Medico Carracci; Sarug Reyes Morales, MD, Instituto Biomedico de Investigacion AC. The authors also thank Sarah Becker-Marrero of Syneos Health for help in editing and proofreading the manuscript. The authors also thank the study team members, Qi Zhang and Holly Detke, of Eli Lilly and Company, for their contributions to this study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VS, BAM, and JYY are full time employees and minor stock holders of Eli Lilly and Company. MM serves on the advisory board for Allergan, Autonomic Technologies Inc, St Jude Medical, and Medtronic, and has received payment for the development of educational presentations from Allergan, Autonomic Technologies Inc, Medtronic, and electroCore. BKK received lecture fees from Allergan, Sandos, YuYu pharm, and SK pharm. MHO is a full-time employee of Syneos Health.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was sponsored by Eli Lilly and Company.
