Abstract
Objective:
To describe patient characteristics, treatment patterns, and health care costs among patients diagnosed with major headache disorders overall and by type (tension-type headache [TTH], migraine, cluster headache [CH], or >1 primary headache type), and secondarily to evaluate drug treatment patterns among triptan initiators with a major headache diagnosis.
Methods:
Using US claims data from January 2012 through December 2017, we identified adults with evidence of a major headache disorder: TTH, migraine, or CH; the first diagnosis date was deemed the index date. To evaluate triptan use specifically, patients who initiated triptans were identified; the first triptan claim date was deemed the index date. Patient characteristics, treatment patterns (concomitant treatments, adherence, number of fills), and annual health care costs data were obtained.
Results:
Of the 418,779 patients diagnosed with major headache disorders, the following 4 cohorts were created: TTH (8%), migraine (87%), CH (1%), and >1 primary headache type (4%). The majority used analgesic (54–73%) and psychotropic (57–81%) drugs, primarily opioids (36–53%). Headache-related costs accounted for one-fifth of all-cause costs. Of the 229,946 patients who initiated triptans, the following 7 study cohorts were analyzed: sumatriptan (68%), rizatriptan (21%), eletriptan (5%), zolmitriptan (3%), naratriptan (2%), frovatriptan (1%), and almotriptan (<1%). The major concomitant analgesic medication classes were opioids (41%) and nonsteroidal anti-inflammatory drugs (34%).
Conclusion:
The primary headache disorder treatment paradigm is complex, with significant variability. Predominant concomitant use of opioids and switching to opioids is of concern, necessitating solutions to minimize opioid use. Switching to non-oral/fast-acting or targeted preventive therapies should be considered.
Introduction
Approximately half of the world’s adult population has experienced a headache at least once within the last year; 1 more than 90% of patients who present to their primary care provider with symptoms are diagnosed with a primary headache disorder. 2 The global active prevalence of tension-type headache (TTH) is approximately 40% and migraine is 10%; cluster headache (CH) is rare, with a population prevalence of 0.1%; and together these account for the majority of primary headache disorder diagnoses. 2 Despite being recognized as a public health concern, there are few studies quantifying the prevalence and burden of all headache disorders; existing studies have focused on individual headache types. Understanding the treatment patterns and burden of the major headache types may assist in evidence-based treatment decisions as treatment paradigms tend to overlap across the different subtypes.
Headache disorders are estimated to cost US$31 billion in the United States each year. 3 As per a real-world assessment of the economic burden of migraine, compared with matched non-migraine patients, migraine patients were more likely to have work loss and longer periods of work loss, leading to significantly higher indirect costs. 4 While limited literature is available on the burden of TTH, indirect costs of non-migraine headaches (of which TTH is the major contributor) are also higher than that of migraine headaches. 5 Additionally, studies 6 –8 have demonstrated considerable absenteeism as a result of TTH, as high as three times more than in migraine. It is reported that CH patients have high health care costs associated with inpatient admissions and high indirect costs associated with absenteeism and short-term disability. 9 In a study that utilized US claims data, the CH-related total direct costs were US$3132 per patient per year. Additionally, CH-related inpatient hospitalizations (US$1604) and pharmacy (US$809) together (US$2413) contributed over 75% of the CH-related direct health care cost. 9
Previous research investigating pharmacologic treatment patterns from 2008 to 2009 in insured patients with migraine using a large claims database reported that 66% received acute migraine-specific medication, while only 20% received US Food and Drug Administration (FDA)-approved prophylactic migraine therapy. 10 The relatively low percentage of patients who received an FDA-approved migraine preventive therapy may be attributed to off-label preventive medication use for migraine. A secondary analysis of the pooled National Ambulatory Medical Care Survey from 2011 and 2012 demonstrated that of 12.9 million outpatient visits for pediatric patients with a migraine diagnosis, 66.7% received at least 1 migraine drug and, of these, off-label medications were prescribed 1.5 times more than FDA-approved medications for children (60.34% vs. 39.65%). 11 A systematic review of adults with episodic migraine reported that among off-label drug classes, angiotensin-inhibiting drugs (angiotensin-converting enzyme inhibitors and angiotensin receptor blockers) were more effective in reducing monthly migraine by ≥50% compared with antidepressants, off-label antiepileptics, and ergot alkaloids. 12 In a retrospective analysis using claims data from 2009 to 2014, the most commonly prescribed drug classes for CH patients included opiate agonists (41%), corticosteroids (34%), 5HT-1 agonists (32%), antidepressants (31%), nonsteroidal anti-inflammatory drugs (NSAIDs) (29%), anticonvulsants (28%), calcium antagonists (27%), and benzodiazepines (22%). 13 The literature on treatment patterns among patients diagnosed with TTH is scarce.
The objective of this study was to primarily describe patient characteristics, pharmacologic treatment patterns, annual all-cause and headache-related health care resource utilization (HCRU), and costs among patients diagnosed with major headache disorders overall and by type (TTH, migraine, CH, or >1 primary headache type) and secondarily to evaluate drug treatment patterns among triptan initiators with a major headache diagnosis.
Methods
Data source
Data on patient characteristics, treatment patterns, HCRU, and costs were obtained from the integrated health care claims data from the IBM MarketScan Commercial Claims and Encounters (commercial) and Medicare Supplemental (Medicare) databases. The commercial database consists of employer and health plan-sourced data containing medical and drug claims for over 50 million individuals annually. Enrollees are employees, Medicare-eligible retirees, and their dependents insured by employer-sponsored benefit plans. The Medicare Supplemental database contains the inpatient and outpatient medical and prescription claims of Medicare-eligible persons with supplemental insurance plans offered by their former employers. Since this study used deidentified patient records for analysis, it was exempt from institutional review board approval.
Study design and population
This study employed a retrospective cohort design using data from January 1, 2012, to December 31, 2017. For the analysis of major headache disorders overall, the target population was adults with evidence of a major headache disorder: TTH (
For the evaluation of triptan initiators specifically, the target population consisted of adults who initiated triptans during the identification period (January 1, 2013–December 31, 2016) with evidence of a major headache disorder diagnosis. The date of the first observed triptan claim during the identification period was defined as the index date. Patients were required to be continuously enrolled for the 12 months pre- and post-index date.
Patient demographic and clinical characteristics
Patient demographics such as age, gender, geographic region, health plan type, urban/rural residence, and index year were measured as of the index date. Clinical characteristics were measured for both study populations over the 12-month pre-index period and included the Charlson Comorbidity Index (CCI) score, which was computed for each patient based on the presence of ICD-9/10-CM diagnosis codes and represents overall comorbidity. In addition, specific comorbid conditions (i.e. angina, anxiety disorders, arthritis, osteoarthritis, rheumatoid arthritis, bipolar spectrum disorders, additional headache types, and so forth) and additional symptoms of interest (i.e. nausea, photophobia, phonophobia, and vomiting) were assessed using ICD-9/10-CM diagnosis codes.
Outcomes
HCRU and costs were computed by setting of care over the 12 months post-index; annual all-cause and headache-related (claims with a primary or secondary diagnosis of TTH, migraine, or CH) HCRU were presented. All costs were defined as the actual reimbursements paid by health plans plus any patient cost sharing in the form of deductibles, copayments, and coinsurance. All cost estimates were adjusted to the most recent year (2017) US$ by the medical component of the Consumer Price Index. Components of the total costs (medical and prescription) were computed.
The use of analgesic and psychotropic medications over the 12 months post-index was reported; the number of prescription claims was also computed. The proportions of patients on recognized treatments for headache, stratified by acute/abortive and preventive/prophylactic treatment, were captured over the 12 months post-index. For prophylactic medications, proportion of days covered (PDC) overall was reported (days with multiple drugs/drug classes [as applicable] on hand were counted once). For acute medications, the average number of claims by medication class was reported (a 90-day supply prescription counted as 3 claims). Recognized acute and prophylactic treatments were identified from a combination of published literature, 14,15 treatment guidelines, 16 –19 and clinical input.
Among triptan initiators, triptan use patterns were computed over a variable follow-up period: up to the end of enrollment or end of study period, whichever came first. The proportion of patients with only one triptan fill was reported. Patients who continued to refill the index triptan were considered persistent to their index triptan. In this way, persistency was defined as consecutive refills of the initially prescribed triptan, regardless of duration between refills. To better capture patients with a low frequency of attacks, patients were censored: if the end of the follow-up period occurred after 90 days following the last day of possession of the index triptan, that patient was classified as nonpersistent. 20 For nonpersistent patients, the number of index triptan fills before discontinuation was reported. Proportions of nonpersistent patients with evidence of a non-index triptan, opioid, or NSAID claim after the last index triptan claim were reported. Patients were required to have no evidence of use of the concerned class of medication over the 12 months prior to switch date to be classified as switchers.
High triptan use was defined as dispensing of triptan pills, sprays, or injections at a level that suggested the patient was treating more than 24 headaches during any 90-day rolling period. For this calculation, it was assumed that a quantity of two pills or injections (regardless of strength or specific triptan), two sprays of nasal sumatriptan, one subcutaneous injection of zolmitriptan, or one spray of nasal zolmitriptan was used for each headache. 10
Statistical analysis
The study was descriptive in nature. All demographic and clinical variables measured during the baseline period and outcomes measured during the follow-up period were analyzed descriptively through the tabular display of means, standard deviations (SDs), medians, and percentiles for continuous measures and frequency distributions for categorical variables. All data management and analyses were conducted using SAS version 9.4.
Results
For the analysis of all major headache disorders, a total of 418,779 patients were included; the following 4 cohorts were created: TTH (8%;
Baseline demographics and clinical characteristics of patients diagnosed with major headache disorders.

CCI: Charlson Comorbidity Index; CDHP: consumer-driven health plan; CH: cluster headache; EPO: exclusive provider organization; FFS: fee-for-service; GERD: gastroesophageal reflux disease; HDHP: high-deductible health plan; HMO: health maintenance organization; POS: point of service; PPO: preferred provider organization; SD: standard deviation; TTH: tension-type headache.
a Specific primary/secondary headaches and comorbidities <5% have not been reported.
For the evaluation of triptan initiators, of the 1,114,406 patients with at least 1 claim for a triptan, 229,946 (20.6%) were included in the final sample; patients were mainly excluded due to noncontinuous enrollment in the health plan during the 12-month pre- and post-index periods. Of the final sample, the majority were sumatriptan initiators (68.0%), followed by rizatriptan (20.7%), eletriptan (5.1%), zolmitriptan (3.3%), naratriptan (1.7%), frovatriptan (0.9%), and almotriptan (0.4%) (Table 2). The mean (SD) age of the triptan initiators was 40.0 (12.9) years, ranging from 39.5 (13.0) years (sumatriptan) to 45.1 (12.7) years (almotriptan). The majority of patients were 35–54 years old, female (81.4%), and from the South (44.4%) and resided in urban areas (85.8%). The mean (SD) CCI was 0.3 (0.8) for the overall study population and remained similar across study cohorts. Approximately, three-fourths of the study population had a CCI score of 0. The most prevalent comorbidities overall were sinusitis (23.9%), hypertension (16.8%), circulation problems (25.3%), and anxiety disorders (16.7%). The proportions of patients with specific comorbidities and symptoms did not differ significantly across study groups.
Baseline demographics and clinical characteristics of triptan initiators.

CCI: Charlson Comorbidity Index; CDHP: consumer-driven health plan; CH: cluster headache; EPO: exclusive provider organization; FFS: fee-for-service; GERD: gastroesophageal reflux disease; HDHP: high-deductible health plan; HMO: health maintenance organization; POS: point of service; PPO: preferred provider organization; SD: standard deviation; TTH: tension-type headache.
a Specific primary/secondary headaches and comorbidities <5% have not been reported.
HCRU and costs
Among patients diagnosed with major headache disorders, one-tenth (7.7–9.8%) of the patients were hospitalized over the 12-month post-index period, and one-third had emergency department (ED) visits (26.3–36.3%). Almost all patients had a physician office visit over the post-index period (97.9–99.7%), with mean (SD) number of visits over the 12-month period ranging from 8.4 (7.1) (CH) to 12.3 (9.1) (>1 headache type). Physician office visits were further evaluated by specialty type, and 20.7–54.2% of patients had evidence of a neurologist visit (mean [SD] number of visits = 0.6 [1.8] to 2.1 [3.1]), 58.1–65.8% of patients visited a primary care provider (mean [SD] number of visits = 2.7 [4.9] to 3.8 [6.0]), and 15.3–38.1% of patients visited an obstetrician/gynecologist (mean [SD] number of visits = 0.3 [1.2] to 0.9 [1.9]) over the 1 year. Patients diagnosed with >1 headache type incurred the highest mean (SD) total annual all-cause costs (US$17,853 [US$32,073]), followed by migraine (US$15,320 [US$31,802]), CH (US$15,037 [US$40,018]), and TTH (US$12,825 [US$27,829]). The medical costs were primarily driven by outpatient/ancillary visits (other than physician office visits). Headache-related costs accounted for approximately one-fifth of all-cause costs.
Treatment patterns
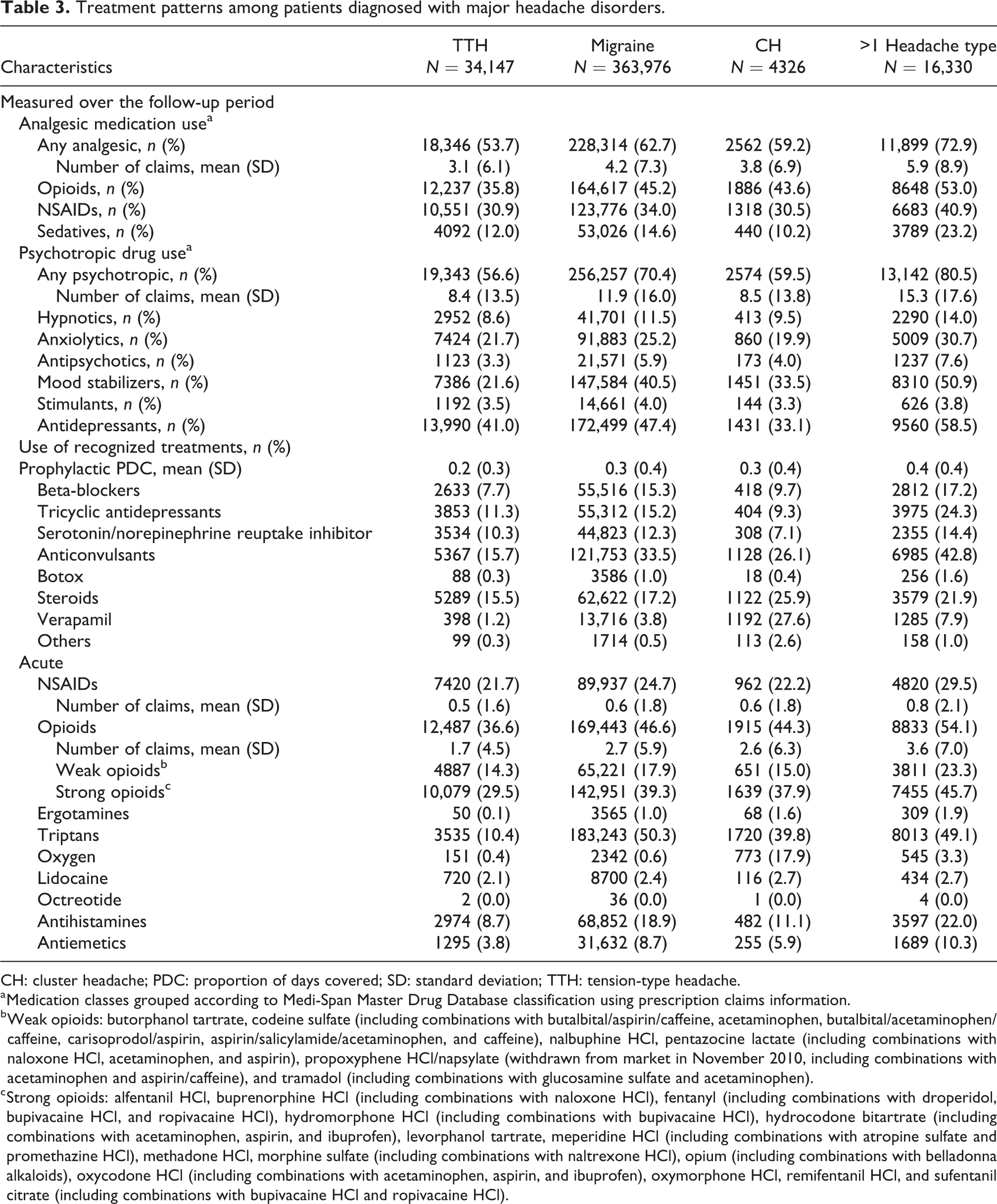
Among patients diagnosed with major headache disorders, the majority had evidence of analgesic (53.7–72.9%) (proportions vary across primary headache disorder diagnoses [Table 3]) and psychotropic (56.6–80.5%) drug use. The predominant analgesic medication classes used were opioids (35.8–53.0%) and NSAIDs (30.5–40.9%). The mean (SD) number of analgesic prescription claims over 1 year ranged from 3.1 (6.1) for the TTH cohort to 5.9 (8.9) for patients diagnosed with >1 headache type. The mean (SD) number of opioid prescription claims ranged from 1.7 (4.4) for the TTH cohort to 3.5 (6.7) for patients diagnosed with >1 headache type. The predominant psychotropic medication classes were antidepressants (33.1–58.5%) and mood stabilizers (21.6–50.9%). Patients diagnosed with >1 headache type had the highest mean (SD) number of psychotropic prescription claims over the year (15.3 [17.6]), followed by migraine (11.9 [16.0]), CH (8.5 [13.8]), and TTH (8.4 [13.5]) cohorts. Antidepressant and mood stabilizer use was highest among patients diagnosed with >1 headache type (mean [SD] number of prescription claims = 6.8 [9.4] and 5.1 [8.5], respectively), followed by the migraine cohort (mean [SD] number of prescription claims = 5.3 [8.4] and 3.9 [7.5], respectively). Adherence to recognized prophylactic treatments was low; the mean (SD) PDC ranged from 0.2 (0.3) for the TTH cohort, to 0.4 (0.4) for patients diagnosed with >1 headache type.
Treatment patterns among patients diagnosed with major headache disorders.
CH: cluster headache; PDC: proportion of days covered; SD: standard deviation; TTH: tension-type headache.
a Medication classes grouped according to Medi-Span Master Drug Database classification using prescription claims information.
b Weak opioids: butorphanol tartrate, codeine sulfate (including combinations with butalbital/aspirin/caffeine, acetaminophen, butalbital/acetaminophen/caffeine, carisoprodol/aspirin, aspirin/salicylamide/acetaminophen, and caffeine), nalbuphine HCl, pentazocine lactate (including combinations with naloxone HCl, acetaminophen, and aspirin), propoxyphene HCl/napsylate (withdrawn from market in November 2010, including combinations with acetaminophen and aspirin/caffeine), and tramadol (including combinations with glucosamine sulfate and acetaminophen).
c Strong opioids: alfentanil HCl, buprenorphine HCl (including combinations with naloxone HCl), fentanyl (including combinations with droperidol, bupivacaine HCl, and ropivacaine HCl), hydromorphone HCl (including combinations with bupivacaine HCl), hydrocodone bitartrate (including combinations with acetaminophen, aspirin, and ibuprofen), levorphanol tartrate, meperidine HCl (including combinations with atropine sulfate and promethazine HCl), methadone HCl, morphine sulfate (including combinations with naltrexone HCl), opium (including combinations with belladonna alkaloids), oxycodone HCl (including combinations with acetaminophen, aspirin, and ibuprofen), oxymorphone HCl, remifentanil HCl, and sufentanil citrate (including combinations with bupivacaine HCl and ropivacaine HCl).
Triptans were the second predominant acute headache-related treatment used (10.4–50.3%) after opioids (36.6–54.1%) among all study cohorts, except patients diagnosed with TTH. The TTH cohort had evidence of predominant opioid use (36.6%) followed by NSAID use (21.7%). The migraine cohort had the highest proportion of patients on triptans (50.3%), with a mean (SD) number of prescriptions = 2.5 (4.6) over the 12-month post-index period. Patients diagnosed with >1 headache type had the highest proportion with opioid use (54.1%), followed by migraine (46.6%), CH (44.3%), and TTH (36.6%) cohorts. Higher proportions of patients used strong opioids (29.5–45.7%) across all study cohorts than weak opioids (14.3–23.3%). Oxygen therapy was predominant among patients diagnosed with CH (17.9%) compared with the other study cohorts (0.4–3.3%). Sumatriptan was the most commonly used triptan followed by rizatriptan; a higher proportion of triptan users among patients diagnosed with CH used sumatriptan (81.1%) when specific triptan use was compared across study cohorts. Non-oral sumatriptan use was more common among patients diagnosed with CH when compared across all study cohorts (Figure 1).
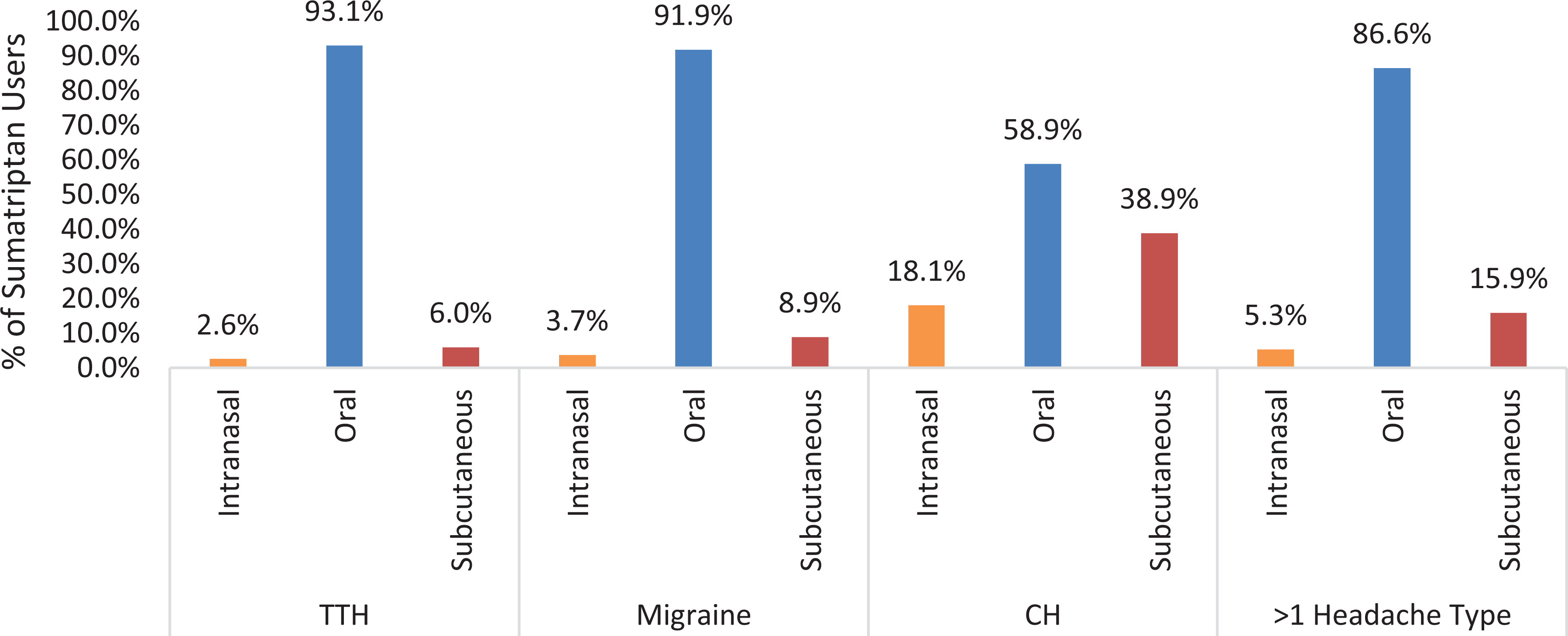
Sumatriptan routes of administration by headache type. CH: cluster headache; TTH: tension-type headache.
Among triptan initiators, the majority had evidence of concomitant analgesic (58.4%) and psychotropic (64.8%) drug use (Table 4). The predominant analgesic medication classes used were opioids (40.6%) and NSAIDs (34.1%). The mean (SD) number of analgesic prescription claims over the 12-month post-index period was 3.0 (5.7). Higher proportions of naratriptan initiators had evidence of concomitant analgesic drug use (including a higher mean number of analgesic prescription claims) compared with the other triptan initiators, whereas the proportion of sumatriptan initiators with concurrent analgesic, psychotropic, and prophylactic drug use was the lowest among all triptan initiators. The majority of the triptan initiators had evidence of concomitant prophylactic headache-related treatment (56.5%), and the mean (SD) PDC for prophylactic treatments was 0.3 (0.3).
Treatment patterns among triptan initiators.

CH: cluster headache; NSAID: nonsteroidal anti-inflammatory drug; PDC: proportion of days covered; SD: standard deviation; TTH: tension-type headache.
a Medication classes grouped according to Medi-Span Master Drug Database classification using prescription claims information.
b Weak opioids: butorphanol tartrate, codeine sulfate (including combinations with butalbital/aspirin/caffeine, acetaminophen, butalbital/acetaminophen/caffeine, carisoprodol/aspirin, aspirin/salicylamide/acetaminophen, and caffeine), nalbuphine HCl, pentazocine lactate (including combinations with naloxone HCl, acetaminophen, and aspirin), propoxyphene HCl/napsylate (withdrawn from market in November 2010, including combinations with acetaminophen and aspirin/caffeine), and tramadol (including combinations with glucosamine sulfate and acetaminophen).
c Strong opioids: alfentanil HCl, buprenorphine HCl (including combinations with naloxone HCl), fentanyl (including combinations with droperidol, bupivacaine HCl, and ropivacaine HCl), hydromorphone HCl (including combinations with bupivacaine HCl), hydrocodone bitartrate (including combinations with acetaminophen, aspirin, and ibuprofen), levorphanol tartrate, meperidine HCl (including combinations with atropine sulfate and promethazine HCl), methadone HCl, morphine sulfate (including combinations with naltrexone HCl), opium (including combinations with belladonna alkaloids), oxycodone HCl (including combinations with acetaminophen, aspirin, and ibuprofen), oxymorphone HCl, remifentanil HCl, and sufentanil citrate (including combinations with bupivacaine HCl and ropivacaine HCl).
Index triptan use patterns were computed over a variable follow-up period. The mean (SD) follow-up time for the study population was 29.9 (13.2) months and relatively similar across all study cohorts. Overall, 42.4% of the study population had only one claim for a triptan (including the index claim) over follow-up; the proportion of patients with only one triptan fill was highest among sumatriptan initiators (44.2%) and lowest among almotriptan initiators (29.9%). Overall, 84.0% of triptan initiators were nonpersistent to index triptan therapy and had on average 2.7 (±3.2) index triptan fills before discontinuation. The proportion of patients nonpersistent to index triptan therapy was similar across all triptan initiators. Of the nonpersistent patients, 45.4% switched to NSAIDs, opioids, or a non-index triptan and had on average 1.9 (±2.1) index triptan fills before switching. Of the switchers, 64.9% switched to opioids, 62.1% switched to NSAIDs, and 6.6% switched to a non-index triptan. These results were similar across study cohorts. Additionally, 9.3% of triptan initiators had evidence of high triptan use; the proportion of patients with high triptan use was the highest among almotriptan initiators (13.5%) and lowest among sumatriptan initiators (8.4%). Higher proportions (27.2–36.4%) of patients who initiated non-oral triptans had evidence of oral triptans over follow-up, whereas among patients initiating oral triptans, only 2.8% and 1.7% had evidence of a subcutaneous and intranasal triptan over follow-up. Differential discontinuation of treatment by index route of administration may affect this conclusion.
Discussion
This retrospective observational cohort study was conducted using a linked medical and pharmacy claims database to evaluate patient characteristics and treatment patterns among patients diagnosed with major headache disorders overall and by headache type, and treatment patterns among triptan users diagnosed with TTH, migraine, or CH. The study demonstrated that the majority of patients diagnosed with major headache disorders had evidence of analgesic and psychotropic drug use, primarily opioids, NSAIDs, and antidepressants. Adherence to recognized prophylactic treatments was low. Triptans were the predominant acute headache treatment after opioids for all cohorts except TTH patients. Patients diagnosed with CH had evidence of triptan use and were more likely to use non-oral triptans. Headache-related resource utilization accounted for approximately 20% of total health care costs incurred by these patients.
Opioids were the most predominant concomitant acute headache-related treatment used, followed by NSAIDs. Higher proportions of patients used strong opioids overall than weak opioids; this was consistent across all study cohorts. Among triptan initiators, switching to opioid or NSAID therapy was prevalent. The use of oral triptans was more favorable in comparison to non-oral triptan therapy (>90% of the population had a migraine diagnosis). Approximately one-tenth of patients had evidence of high triptan use.
Consistent with previous research, 13,14 opioids were the predominant concomitant acute headache-related treatment used across all study cohorts. Opioid treatment for acute headache is highly controversial, as there are a number of adverse events (AEs) associated with acute and chronic opioid treatment, 21 including euphoria, sedation, sleep disturbance, respiratory depression, cough suppression, pupillary constriction, truncal rigidity, and nausea and vomiting. Additionally, AEs with opioids may be amplified when use is daily, which may include significant gastrointestinal dysfunction, dependence, and addiction. Overuse of opioids may be associated with worsening of migraine, increasing the risk of chronic migraine. 22 High rates of resource utilization (inpatient and ED admissions) have been reported among patients diagnosed with migraine or CH, with evidence of concomitant opioid medication use. 13,23 Moreover, opioid use for migraine resulted in more severe headache-related disability, symptomology, and comorbidities (primarily psychiatric and cardiovascular) and greater HCRU. 24
Solutions to minimize opioid use among patients diagnosed with major headache disorders are needed. Successful management of the disorder depends on patient–provider collaboration. Detailed records or patient diaries with information on headache days, severity, medication use, response and adverse effects, headache triggers, and so on, and frequent review of this information by the provider and patient can help develop better preventive strategies tailored to the individual patient. 25 Oral cycling and high switching rate may also infer patient dissatisfaction. 26 Patients should be switched to non-oral/fast-acting treatments to optimize management of acute migraine episodes first rather than opioids. Additional real-world data on opioid use and dosage in the treatment of major headache disorders and their effectiveness are warranted.
Earlier this year, the FDA approved three targeted preventive therapies for migraine in calcitonin gene-related peptide monoclonal antibody drugs, ushering in a new era of migraine treatment. Studies using more recent data that evaluate the effect of this new class of therapies on opioid use in this population are required. As recently as January 2020, The Institute for Clinical and Economic Review released an evidence report assessing the comparative clinical effectiveness and economic value of lasmiditan (Reyvow™, Eli Lilly), rimegepant (Biohaven), and ubrogepant (Ubrelvy™, Allergan). 27 The report concludes that triptans will likely remain first-line therapy for patients with acute migraine, who can tolerate triptans, as the evidence suggests that lasmiditan, rimegepant, and ubrogepant all offer either a comparable or inferior net health benefit to triptans. They appear to be less effective overall than triptans and are expected to have a comparatively higher price tag.
Limitations
Claims data are not inherently designed for research and therefore do not capture clinical severity measures and patient characteristics that might influence physician prescribing behavior. Additional insight from physician notes on observed high opioid use and other treatment patterns were not available. While claims data allow for the determination of the medications prescribed to patients, they do not lend insight into how the medications were used, nor provide data on reasons for discontinuation/nonpersistence. This is a principal limitation, especially when studying medications taken as needed, rather than in a prescribed regimen. Information on over-the-counter medication use was not available. Further, a prescription filled does not always translate to medication use. Adherence/persistence results obtained using claims data should be evaluated with the above limitations in mind. Additionally, prescription data lack information on the primary diagnosis associated with the prescription claim. Therefore, some of the acute and prophylactic treatments discussed above could have been prescribed for comorbid conditions prevalent in the study population, other than the primary headache disorders. Limitations related to the accuracy of coding in the claims data may also affect study results. Prevalence of secondary headache types and suicidal ideation and attempt, as well as symptoms of interest (photophobia and phonophobia), were underestimated due to lack of coding. The database only provides information on medications received in an outpatient setting; administration of headache-related treatments in an inpatient setting could not be captured. However, such events, if any, should have been rare. Additionally, the coding system used to capture diagnoses in claims data transitioned from the 9th to the 10th Revision on October 1, 2015, which could impact the identification of the study population across calendar years and the prevalence of diseases and symptoms. The study analyzed data up to December 2017, following which there has been significant media coverage and awareness regarding opioid overuse; the use of opioids within this patient population may have changed since then. Finally, the results are only generalizable to a population insured through employer-sponsored plans and may not be applicable to other populations such as Medicaid.
Conclusion
In conclusion, this study supports existing perspectives on primary headache disorders as complex, with significant variability in treatment patterns. Patients diagnosed with >1 primary headache type have higher comorbidity and economic burden.
Patients diagnosed with CH have evidence of triptan use and are more likely to use non-oral triptans, probably due to the relatively quicker onset and higher severity of a headache episode among CH patients compared with patients diagnosed with other major headache disorders. However, 60% of CH patients with evidence of sumatriptan used oral sumatriptan at least once, highlighting the need for therapy tailored to the headache type and severity.
Predominant concomitant use of any opioids across all primary headache types (35.8–53.0%)—and specifically strong opioids—and high rates of switching to opioids are of concern and may be indicative of poor treatment optimization.
Further studies are needed to understand the role of natural disease progression, including the burden of other comorbidities, while evaluating physician prescribing behaviors for various headache-related treatments. In-depth analysis of the use of opioids for headache treatment is also recommended.
Footnotes
Acknowledgments
The authors thank Xcenda colleague, Cynthiya Ruban, MS, PhD, for her assistance with manuscript writing.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author Kudrow is the Director of the California Medical Clinic for Headache & Neurological Research Institute. Author Munjal is an employee of Promius Pharma LLC. Author Bensimon was an employee of Promius Pharma LLC throughout the course of the analysis and manuscript development. Authors Lokhandwala and Coutinho are employees of Xcenda, a consultancy that assisted in the completion of this study. Author Yue was an employee of Xcenda throughout the course of the analysis and manuscript development. Author Silberstein is the director of the Jefferson Headache Center.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Promius Pharma LLC.
