Abstract
Background
Headache is the most frequent symptom of cerebral venous thrombosis (CVT) but there is limited information about the frequency and phenotype of headache, weeks to months after cerebral venous thrombosis (post-cerebral venous thrombosis headache, PCH).
Objective
To assess the frequency, characteristics and predictors of PCH.
Methods
In this cross-sectional study, the frequency and characteristics of PCH were assessed in cerebral venous thrombosis survivors. Patients were interviewed between six months and five years after the cerebral venous thrombosis diagnosis. Clinical and imaging characteristics at the time of cerebral venous thrombosis diagnosis, as well as history of headache prior to cerebral venous thrombosis were compared in subjects with (GroupPCH) and without PCH (Groupcontrol).
Results
Subjects (n = 100; 82% women) were assessed, on average, at 1.1 ± 1.6 years of follow-up. PCH was present in 59% of the patients, phenotypes of tension-type-like headache were present in 31/59 (52.6%) and of migraine-like headache in 16/59 (27.1%). History of primary headache prior to cerebral venous thrombosis was significantly more common (OR: 6.4; 95% CI: 1.7–36.3) in GroupPCH (33.9%) than in Groupcontrol (7.3%).
Conclusion
PCH was present in more than half of the patients. History of prior headache may be a risk factor for PCH. Prospective studies are required to confirm these findings and determine mechanisms, as well as interventions for prevention and treatment of PCH.
Keywords
Introduction
Cerebral Venous Thrombosis (CVT) is a rare cause of stroke, corresponding to about 1% of all cerebrovascular events (1). The interest for this condition increased after severe cases of CVT were reported after COVID-19 or after vaccination with ChAdOx1 and Ad26.COV2.S, during the SARS-CoV-2 pandemic (2). Before the pandemic, CVT had an overall good prognosis, with a mortality rate of around 8% (3–6). A modified Rankin Scale (mRS) score of 0 to 1 was typically observed in 85% to 90% of the patients at six months after diagnosis (5,7).
According to the International Classification of Headache Disorders (ICHD-3) (8), the headache attributed to CVT (6.6.1) can be of any type, must have a temporal relationship with the diagnosis of thrombosis or lead to the CVT diagnosis. In addition, the progress of pain must be correlated with improvement or worsening of the thrombosis. Headache is more frequent in CVT (up to 90% of the patients at the time of diagnosis) (4) than in ischemic stroke (9,10).
A prospective study reported headache at onset of ischemic stroke in 14.9% of 550 patients (10). A new type of headache was present in 56% of the patients, while headache with altered characteristics was reported by 36% (10). According to an international consensus, phases of recovery after stroke can be classified as hyperacute, acute, early subacute, late subacute, or chronic (11). At the chronic phase (>6 months) (11) after ischemic stroke, primary-like headache phenotypes (mainly tension-type-like headache) were reported by 50.6% of 89 subjects (12). The pain fulfilled criteria for tension-type headache (TTH, ICHD-3 code 2.) in 28.9% of the patients and for migraine (ICHD-3 code 1.) (8), in 46.7%. Overall, the characteristics of post-stroke headache differed from the characteristics of pain in the acute phase after stroke. Female sex and pre-ischemic stroke headache were independent predictors of post-stroke headache (12).
Weeks to months after CVT, headache was reported by 20% to 47% of the patients, being severe in up to 14% (6,13,14) (Table 1). Until now, headache at the chronic stage after CVT had not been assessed by structured interviews. Risk factors, characteristics of the pain and predictors of persistent post-CVT headache (PCH) remained to be clarified.
Summary of studies that followed patients with cerebral venous thrombosis for at least 12 months.

The aims of this study were to describe the frequency, characteristics and potential clinical/imaging predictors of PCH. In addition, we compared characteristics of PCH, headaches attributed to CVT (ICHD code 6.6.1) and headaches reported prior to CVT.
Methods
This was an observational, cross-sectional study, based on face-to-face interviews and assessment of medical records. Interviews were performed at the CVT Outpatient Clinic from the Neurology Department at Hospital das Clínicas/São Paulo University, the largest hospital in Latin America, from November 2016 to October 2019. After each interview, medical records were assessed to retrieve information about the admission at the time of CVT diagnosis (Figure 1).

Study design.
This study received ethical approval from our institutional Research Ethics Committee (number 56307316.70000.0068). All patients provided written informed consent.
Subjects
Consecutive patients followed at the CVT outpatient clinic were screened for eligibility criteria. All CVT survivors admitted at Hospital das Clínicas were evaluated in the CVT clinic.
The inclusion criteria were: outpatient subjects ≥18 years old; CVT diagnosed between six months and five years prior to enrollment, confirmed by MRI or venography (magnetic resonance venography – MRV, computed tomography venography, or conventional angiography); follow-up in the CVT Clinic; written informed consent to participate in the study. The exclusion criteria were: score of four or higher in the Modified Rankin Scale (15); inability to answer the questions from the interview (for instance, due to aphasia or cognitive impairment).
Patients’ characteristics
The following characteristics were assessed during a single face-to-face interview: gender, age, body mass index (BMI), medications, neurological impairment according to the National Institute of Health Stroke Scale (NIHSS) (16), functional independence according to the modified Rankin Scale (mRS) (15), cognitive status according to the Mini-Mental State Examination (MMSE) (17), quality of life according to the Stroke Impact Scale (SIS) 3.0 (18), symptoms of depression according to the Patient Health Questionnaire (PHQ9) (19,20), symptoms of anxiety according to the General Anxiety Disorder 7 (21,22). All scales were assessed on the day of the interview.
After the interview and physical examination, the following information was retrieved from medical records: age at the time of CVT; symptoms that led to CVT diagnosis; type and duration of treatment; neuroimaging results; CVT risk factors according to the institutional protocol (Supplementary Figure 1).
An experienced neuroradiologist, blinded to clinical information, evaluated head computed tomography (CT), computed tomography venography, magnetic resonance imaging (MRI) and magnetic resonance venography (MRV) performed at the time of CVT diagnosis to ascertain the presence of venous infarcts, intracranial hemorrhage and venous sinus(es) affected by thrombosis. In addition, recanalization was assessed in all patients who had performed computed tomography venography or MRV at least three months after CVT.
Assessment of headaches
Characteristics of the headaches prior to CVT, the headaches attributed to CVT, and PCH were assessed during the face-to-face interview.
A structured headache interview (Supplementary Table 1) (23) was used to evaluate characteristics of:
Prior history of headache unrelated to CVT – for instance, migraine (ICHD-3 code 1.) (8) or TTH (ICHD-3 code 2.) (8). Presence of headache attributed to CVT (6.6.1), defined according to ICHD-3 criteria (8) as any type of headache in the context of a sinus thrombosis with close temporal relation to that thrombosis, with headache improvement or worsening parallel with clinical or radiological improvement or worsening. Presence of PCH defined as at least three episodes of headache with the same characteristics within the three months prior to the structured interview. This time frame was chosen to limit memory bias.
The following characteristics were evaluated: frequency; duration; intensity; location; onset; associated symptoms; use of analgesic medications; possible triggers; number of days with pain per month; and absence from work due to pain. Initially, criteria from ICHD-2 (24) were used to classify the headaches. After the publication of ICHD-3 in 2018 (8), all headaches were reclassified according to the updated criteria. In subjects with PCH, additional tests were ordered according to the institutional protocol (Supplementary Table 2).
Outcomes
The primary outcome was the percentage of patients with PCH. The secondary outcomes were: characteristics of PCH; comparisons between characteristics of subjects who reported PCH (GroupPCH) and those who did not (GroupControl), to identify potential predictors of PCH; within-subject differences between types of headaches prior to CVT, headaches attributed to CVT, and PCH.
Headaches before CVT and PCH were classified according to ICHD-3 (8): for instance, migraine (ICHD-3 code 1.) (8), TTH (ICHD-3 code 2.) (8), secondary headaches or other types. Medication-overuse headache (ICHD-3 code 8.2) (8) was also assessed.
Sample size
We planned to include 100 consecutive patients followed in the clinic in order to assess the primary outcome in a representative sample of subjects with CVT, an uncommon type of cerebrovascular disease.
Statistical analysis
Means and standard deviations of normally distributed data are presented. Otherwise, medians and ranges are shown. Frequencies of categorical variables were calculated.
Characteristics of subjects in GroupPCH and in GroupControl were compared with chi-square tests, independent t-Student or Mann-Whitney tests, according to the nature and distribution of the data.
Wilcoxon tests were used to compare headache status (yes or no) prior to CVT and in the chronic phase after CVT, in all of the patients.
The frequencies of phenotypes of headache prior to CVT and the frequencies of phenotypes of PCH according to ICDH-3 criteria, considering all of the patients, were compared with chi-square tests. P-values <0.05 were considered statistically significant.
In addition, in GroupPCH, two within-subject comparisons were performed with Wicoxon tests: in the subgroup of patients who presented headache attributed to CVT, between the type of headache attributed to CVT (6.6.1) and the type of PCH (same/different headache); in the subgroup of patients with history of headache prior to CVT, between the type of headache prior to CVT, and PCH (same/different headache). Bonferroni’s corrections for multiple comparisons (25) were performed and p-values <0.025 were considered statistically significant.
Wilcoxon tests were also used to compare, in the subgroup of subjects with history of headache prior to CVT and headache attributed to CVT, the types of these headaches (same/different).
Multivariate binomial regression was planned with PCH as the dependent variable. Characteristics of the patients prior to CVT would be included as independent variables in the model, according to results of between-group comparisons (p < 0.05, GroupPCH versus GroupControl) for each variable. Statistical Analysis was performed with JASP® 0.10.2 and IBM® SPSS® Statistics 24.
Results
Subjects
Figure 2 shows the flow of subjects through the study. From 1st November 2016 to 31st October 2019, 100 patients were included (Table 2). The mean age was 38.3 ± 13.7 years and 82% of the patients were women. Subjects were assessed, on average, at 1.1 ± 1.6 years after CVT. The minimum interval between the diagnosis of CVT and the assessment was six months and the maximum was five years, with a median of one year. Interviews were performed less than three years after the diagnosis of CVT in 72% of the subjects. At the time of inclusion in the study, the median score in the NIHSS and in the mRS was 0 (ranges: NIHSS, 0–6; mRS, 0–3). The most frequent risk factors for CVT were use of oral contraceptives in women (69.5%), followed by the prothrombin gene mutation G20210A (11%) (Supplementary Table 3). In 18 subjects (18%), no risk factors were identified. Headache prior to CVT was reported by 23% of the patients. Most (91.3%) of the headaches prior to CVT were primary: migraine (47.8%; ICHD-3 code, 1.) and tension-type headache (TTH, 43.5%; ICHD-3 code, 2.) (Table 3). Characteristics of the pain prior to CVT and associated symptoms are shown in Tables 4 and 5.

Flow of the subjects through the study.
Characteristics of all subjects and in the subgroups without (Groupcontrol) and with (GroupPCH) recurrent headache within the three months prior to the interview.

achi-square. bMann Whitney test. cStudent-t test.
GAD: Generalized Anxious Disorder; mRS: modified Rankin Scale; NIHSS: National Institute of Health Stroke Scale; PHQ: Patient Health Questionnaire; SIS: Stroke Impact Scale.
**In some cases, CVT was identified in our hospital, after the patients had been discharged from other hospitals without diagnosis and sought medical assistance in our Emergency Department.
Comparisons between frequencies of different types headache prior to CVT, and of types of headaches at the chronic phase after cerebral venous thrombosis (PCH)a.

aTypes of headaches were classified according to ICDH-3 based on characteristics reported by the patients. The terms “migraine” and “tension-type headache” refer to headaches prior to Cerebral Venous Thrombosis. The terms “migraine-like” and “tension-type like” refer to types of PCH with phenotypes of these primary headaches.
bchi-square.
Pain characteristics and associated symptoms.
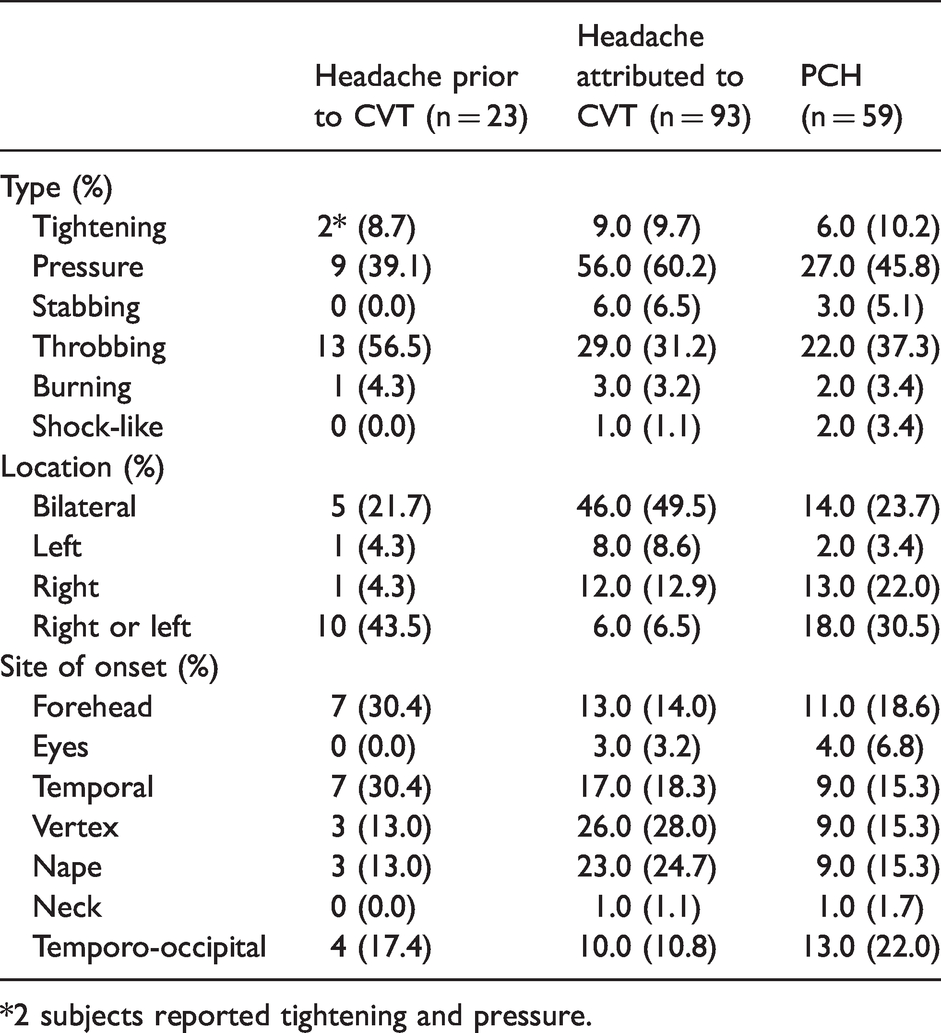
*2 subjects reported tightening and pressure.
Symptoms associated with headache prior to cerebral venous thrombosis (CVT), headache attributed to CVT and post-CVT headache (PCH).

The interval between the first symptom and CVT diagnosis was 21 ± 4 days (mean ± standard deviation) (Table 2). Presentation was acute (within 0h–48h) in 21% (p = 0.193), subacute in 60% (within 6–30 days) (p = 0.354), and chronic (>30 days) in 19% (p = 0.068).
Headache attributed to CVT (6.6.1) was reported by 93% of the subjects and was the only symptom in 17%. Other symptoms and signs were: seizures (39%); hemiparesis (20%); papilledema (19%); decrease in level of consciousness (14%); visual loss (12%); diplopia (11.8%); aphasia (11%); tinnitus (7%), behavioral abnormalities (7%); dizziness (6%); and coma (4%).
Tables 4 and 5 show the phenotypes of headaches attributed to CVT (ICHD-3 code 6.6.1). Some subjects (11/93, 11.8%) had more than one headache phenotype. The mean duration of the headache attributed to CVT (ICHD-3 code 6.6.1) was 122 ± 102 hours. In 17/93 (18.2%) subjects, headache was described as pressure-type, bilateral, and associated with nausea, vomiting, or, less frequently, with phono or photophobia. In 11/93 (11.8%) subjects, headaches had characteristics of migraine but red flags such as seizures, papilledema, visual loss, hemiparesis or cognitive symptoms were present (26). In one subject with a migraine-like, new-onset headache, no other symptoms or signs were present. In 20/23 (87%) subjects with history of headache prior to CVT, the headache attributed to CVT (ICHD-3 code 6.6.1) represented a change in the headache pattern, while 3/23 (13%) did not have headache attributed to CVT. In these three patients, CVT was suspected because of other neurological symptoms. Red flags were present in all of the patients who presented headache attributed to CVT.
The diagnosis of CVT was confirmed by computed tomography venography in 89% of the subjects. MRI and MRV were performed in 74% of the subjects during hospital admission. Supplementary Table 4 shows the number of thrombosed sinuses, sites of thromboses, frequencies of venous infarction and hemorrhagic lesions as well as the percentage of sinus recanalization.
Post-cerebral venous thrombosis headache (PCH)
More than half (59%) of the subjects reported PCH.
Table 3 shows the types of PCH. The most frequent type of PCH was tension-type-like headache (31/59; 52.6%), followed by migraine-like headache (16/59; 27.1%). Only one subject (1.7%) had a secondary headache attributed to a dural fistula (ICDH-3 code 6.3.3) (8) at the chronic phase after CVT. Pulsatile tinnitus, aural fullness, progressive worsening of headache and lack of response to analgesic drugs were red flags in this case (26).
Characteristics of PCH and associated symptoms are shown in Tables 4 and 5. The intensity of PCH was moderate in 28 (47.5%) of the subjects and severe in 14 (23.7%). The number of days with pain per month (average ± standard deviation) was 13.6 ± 9.9 days and the number of days of absence from work, 2.3 ± 5.1 per month. Table 6 shows metrics of headache burden according to the type of PCH.
Burden from headache after cerebral venous thrombosis.

aVisual Analog Scale ranges from zero to 10.
Comparisons between characteristics of the subjects in GroupControl and GroupPCH
Table 2 and Supplementary Tables 3, 4 and 5 show characteristics of the subjects in GroupControl and GroupPCH. There were no statistically significant differences in characteristics between GroupPCH and GroupControl, except for prior history of headache, significantly more frequent in GroupPCH (33.9%) than in GroupControl (7.3%) (Table 2; OR: 6.4; 95% CI: 1.7–36.3).
Figure 3 shows the progress of headache according to history of headache prior to CVT. After CVT, the headache status (yes/no) significantly changed (p < 0.001; n = 100). Most subjects (20/23; 87%) with history of headache prior to CVT reported PCH, while almost half (37/77; 48%) of the subjects without history of headache prior to CVT also reported PCH (Table 2, Figure 3).

Headache prior to cerebral venous thrombosis in subjects with (GroupPCH) or without (GroupControl) headache at the chronic phase after cerebral venous thrombosis.
Only one subject was not treated with anticoagulation at the time of CVT diagnosis. Only three subjects in GroupPCH, and three subjects in GroupControl, were treated with surgical or endovascular procedures in the acute phase after the diagnosis of CVT: dural fistula embolization (1), ventriculoperitoneal shunt + optic nerve sheath decompression (1), empyema drainage (1), ventriculoperitoneal shunt (1), optic nerve sheath decompression (1), intracerebral hemorrhage drainage (1).
At the time of the interview, subjects in GroupPCH presented a higher burden of depressive and anxious symptoms, as well as lower scores in the SIS memory domain, than subjects in GroupControl (Table 2 and Supplementary Table 5). Medication-overuse headache (ICDH-3 code 8.2) (8) was more frequent (33.9%) in GroupPCH than in GroupControl (0%).
Multivariate analysis was not performed because just one variable (history of prior headache) was significantly more common in GroupPCH than in GroupControl. We opted not to include PHQ9, GAD7 and SISmemory in the multivariate regression, because scores in these scales were measured at the time when PCH was assessed. Changes in these scales may have occurred after the onset of persistent headache and in association with frequent episodes of pain. Therefore, their scores should not be used as “predictors” of headaches after CVT.
Within-subject comparisons between characteristics of headache prior to CVT, headache attributed to CVT, and PCH
Figure 4 shows the progress of different types of headache prior to CVT, and to PCH.

History of prior headache and type of headache in subjects with (GroupPCH) or without (GroupControl) headache at the chronic phase after cerebral venous thrombosis.
In GroupPCH, 56/59 (94.9%) subjects had headache attributed to CVT (ICHD-3 code 6.6.1). In these 56 subjects, there was a statistically significant difference (p<0.001) between the type (same/different) of headache attributed to CVT (ICHD-3 code 6.6.1) and the type of PCH. Only 1/56 (1.8%) patient reported PCH with the same characteristics of the headache attributed to CVT. Neuroimaging revealed partial recanalization of the superior sagittal and lateral sinuses in this patient.
In GroupPCH (n = 59), in the subgroup of patients with history of headache prior to CVT (n = 20), the type of prior headache (same/different) was not significantly different from the type of PCH (p = 0.9).
All of the subjects with history of headaches prior to CVT (n = 23) presented headache attributed to CVT (ICHD-3 code 6.6.1). In these subjects, there was a statistically significant difference between the types of headaches attributed to CVT (same/different) and the types of prior headaches (p<0.001).
Individual classification of headaches is shown in Supplementary Table 6.
Discussion
The frequency of PCH (59%) was higher than rates reported in other studies (47% in VENOPORT (6); 43% according to the 6-item Headache Impact Test in functionally independent patients (14); 20% of headache more than once a week in a Finnish cohort (13); 14% of severe headache in ISCVT (5)). This discrepancy is likely due to different ascertainment criteria because structured headache interviews were not performed in prior studies. Differences in study design and characteristics of the patients may also have contributed to differences in rates between the present study and previous reports.
The most common type of PCH was tension-type-like headache (52.5%, 31/59), followed by migraine (27.1%, 16/59). Except for the report from one patient, PCH was described as different from the headache attributed to CVT (ICDH-3 6.6.1) and often fulfilled criteria for primary headaches in the outpatient CVT survivors included in this study. History of headache prior to CVT was more than four times more frequent in subjects with PCH than in those without. Other patients’ characteristics, clinical features at the time of CVT diagnosis, imaging features (site, extent or thrombosis recanalization), CVT risk factors and time since the diagnosis of CVT were not significantly different between the two groups of patients.
On the other hand, both history of headache prior to CVT and female sex were significantly more frequent in subjects with headache, compared to headache-free patients after ischemic stroke (12). This difference may be related to a ceiling effect in our series that was predominantly composed of women (82%) compared to the series of ischemic stroke (39.3%).
These findings suggest two main categories of PCH. In patients with history of headache prior to CVT, PCH may consist of reactivation of a previous primary headache, possibly with similar underlying mechanisms. On the other hand, in subjects with no history of prior headache, PCH seems to represents a new category of headache. PCH might be a Persistent Headache attributed to a past Cerebral Venous Thrombosis (similar to ICHD-3 6.1.1.2 persistent headache attributed to past ischemic stroke – cerebral infarction). The criteria for this condition may include: a new-onset headache different from the headache attributed to CVT (ICHD-3 code 6.6.1); the pain may mimic any primary headache, in patients without history of primary headache disorder; secondary headaches associated with complications of CVT (chronic intracranial hypertension, dural fistulae) must be excluded. However, PCH may also represent a “de novo” primary headache, triggered by a relevant clinical event. In order to test these hypotheses, comparisons between rates of primary headaches in groups with or without history of CVT in prospective or case-control studies are required. New criteria for PCH should be defined in order to standardize headache classification and to facilitate multicenter collaborations (27).
CVT survivors with PCH had significantly higher rates of medication-overuse headache (ICHD-3 code 8.2), as well as more symptoms of anxiety or depression than those without PCH. It is not possible to establish a causal relationship between PCH and medication-overuse headache or psychiatric conditions in our series, given the design of our study. The associations between pain, decline in cognitive functions and symptoms of depression or anxiety are well recognized in the literature (28–30). These results highlight the importance of screening CVT survivors for psychiatric symptoms and chronic use of painkillers. Also, future work is required to assess if treatment with prophylactic drugs early after CVT can decrease the burden of PCH.
Previous guidelines (31–34) provided a list of red and orange flags to select patients in need of screening for potential secondary causes, leading to the acronym SNNOOP10 (26). Red flags were present in all of the patients who presented headache attributed to CVT and were included in this study. A change in headache pattern, one of the red flags, was present in 87% of the subjects who had history of headache prior to CVT and had headache attributed to CVT. Patients who did not present headache attributed to CVT had other neurological symptoms or signs.
The main strength of this study is the detailed assessment of headache in a series of patients with CVT, a rare type of cerebrovascular disease. Clinical evaluation, including mood symptoms and medication-overuse headache as well as radiological assessment were thoroughly performed. Headache data were collected blindly to information about CVT location, extension or treatment. Sites of CVT were assessed blindly to clinical features.
The main limitations of this study are the retrospective design and the lack of a control group without CVT, matched by sex and age. Recall bias may have influenced the characterization of the headache that led to CVT diagnosis. Still, we consider recall bias unlikely because the mean interval between CVT and the interview was not long (average, 1.1 years). Other limitations are: return to work or social/economic burden attributed to PCH were not evaluated; not all patients with PCH underwent extensive investigation.
We do not routinely interrupt anticoagulation to perform lumbar puncture, or order lumbar punctures in every single patient with PCH in clinical practice. We chose to order additional tests, including lumbar puncture and computed tomography venography or MRV when there was a suspicion of secondary headache (lack of criteria for a primary headache, lack of response to treatment, signs of intracranial hypertension or other red flags).
Conclusion
PCH occurred in more than a half of CVT survivors. Phenotypes pf PCH were similar to phenotypes of primary headaches. History of headache prior to CVT was significantly more frequent in patients with PCH than in those without. Patients with PCH often reported pain with moderate or severe intensity and presented medication-overuse headache (ICDH-3 code 8.2), as well as symptoms of anxiety or depression.
These results pave the way for future multicenter studies about diagnostic criteria, underlying mechanisms, prevention and treatment of PCH.
Clinical Implications
Headache was present in 59% of the patients at the chronic phase of cerebral venous thrombosis (CVT). Post-CVT headache (PCH) was different from the headache that led to CVT diagnosis and often fulfilled criteria for primary headaches. The only predictor of PCH was history of headache prior to CVT. PCH was associated with symptoms of depression, anxiety and medication-overuse headache.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221113825 - Supplemental material for Headache at the chronic stage of cerebral venous thrombosis
Supplemental material, sj-pdf-1-cep-10.1177_03331024221113825 for Headache at the chronic stage of cerebral venous thrombosis by Alexandre Souza Bossoni, Mario Fernando Prieto Peres, Claudia da Costa Leite, Ida Fortini and Adriana Bastos Conforto in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024221113825 - Supplemental material for Headache at the chronic stage of cerebral venous thrombosis
Supplemental material, sj-pdf-2-cep-10.1177_03331024221113825 for Headache at the chronic stage of cerebral venous thrombosis by Alexandre Souza Bossoni, Mario Fernando Prieto Peres, Claudia da Costa Leite, Ida Fortini and Adriana Bastos Conforto in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024221113825 - Supplemental material for Headache at the chronic stage of cerebral venous thrombosis
Supplemental material, sj-pdf-3-cep-10.1177_03331024221113825 for Headache at the chronic stage of cerebral venous thrombosis by Alexandre Souza Bossoni, Mario Fernando Prieto Peres, Claudia da Costa Leite, Ida Fortini and Adriana Bastos Conforto in Cephalalgia
Supplemental Material
sj-pdf-4-cep-10.1177_03331024221113825 - Supplemental material for Headache at the chronic stage of cerebral venous thrombosis
Supplemental material, sj-pdf-4-cep-10.1177_03331024221113825 for Headache at the chronic stage of cerebral venous thrombosis by Alexandre Souza Bossoni, Mario Fernando Prieto Peres, Claudia da Costa Leite, Ida Fortini and Adriana Bastos Conforto in Cephalalgia
Supplemental Material
sj-pdf-5-cep-10.1177_03331024221113825 - Supplemental material for Headache at the chronic stage of cerebral venous thrombosis
Supplemental material, sj-pdf-5-cep-10.1177_03331024221113825 for Headache at the chronic stage of cerebral venous thrombosis by Alexandre Souza Bossoni, Mario Fernando Prieto Peres, Claudia da Costa Leite, Ida Fortini and Adriana Bastos Conforto in Cephalalgia
Supplemental Material
sj-pdf-6-cep-10.1177_03331024221113825 - Supplemental material for Headache at the chronic stage of cerebral venous thrombosis
Supplemental material, sj-pdf-6-cep-10.1177_03331024221113825 for Headache at the chronic stage of cerebral venous thrombosis by Alexandre Souza Bossoni, Mario Fernando Prieto Peres, Claudia da Costa Leite, Ida Fortini and Adriana Bastos Conforto in Cephalalgia
Supplemental Material
sj-jpg-7-cep-10.1177_03331024221113825 - Supplemental material for Headache at the chronic stage of cerebral venous thrombosis
Supplemental material, sj-jpg-7-cep-10.1177_03331024221113825 for Headache at the chronic stage of cerebral venous thrombosis by Alexandre Souza Bossoni, Mario Fernando Prieto Peres, Claudia da Costa Leite, Ida Fortini and Adriana Bastos Conforto in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ABC received a scholarship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq/305568/2016-7).
Ethic approval and patient consent
This study received ethical approval from our institutional Research Ethics Committee (number 56307316.70000.0068). All patients provided written informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
