Abstract
Treatment of medication-overuse headache (MOH) relies on detoxification, during which patients face rebound headache without alternative to painkiller. As oxygen has been proven effective for cluster and other headache subtypes, we sought to evaluate use of normobaric oxygen delivered by a high flow concentrator (HFC) in patients suffering MOH. For this purpose, twenty patients with MOH were included in this prospective monocentric open-labeled feasibility study. All patients received standard care with detoxification in addition to HFC delivering normobaric oxygen at 9 l/min, used to their discretion to treat rebound headache. Primary endpoint was acceptance of HFC and secondary endpoints evaluated its efficacy. Four patients were lost of follow-up after inclusion, one was excluded. HFC was accepted by 14/15 (93.3%). At M6 of follow-up, 15/15 (100%) reverted to episodic headache. In conclusion, normobaric oxygen delivered by HFC appears to be safe, feasible, and probably efficient to help patient with MOH who undergo withdrawal therapy. A larger double-blind, sham-controlled prospective study is needed.
Background
With an increasing prevalence of 1% to 3%, medication-overuse headache (MOH) is a frequent, costly, and highly disabling condition. 1 –3 The optimal management of MOH remains unknown but most experts agree that withdrawal of the overused medication, also called detoxification, should be the first line of the treatment. 4,5 Withdrawal therapy is often difficult for patients who face withdrawal symptoms such as rebound headache, nausea, vomiting, tachycardia, anxiety, sleep disturbances 4 and have no alternative to painkillers.
Oxygen therapy has been proven effective for the treatment of cluster headache attacks 6 but appears to be also efficient in attacks of other headache subtype, particularly in migraine attacks. 6 –8 The mechanism underlying the beneficial effect of oxygen on headache remains unknown but may involve the vasoconstrictive effect of hyperoxia (while hypoxia is a migraine trigger and induce arterial vasodilation) 8 and/or the anti-inflammatory effects of oxygen. 9
We hypothesized that the oxygen use to treat headache during the withdrawal therapy would be well accepted and tolerated and that, by giving patients an alternative to total abstinence of painkillers, might increase their compliance to painkillers reduction and, in fine, their chance of detoxification achievement.
High-flow oxygen usually requires repeated gas cylinder supplies and care plan which restricts its availability and affordability. High flow concentrator (HFC) delivering normobaric oxygen, a device which concentrates oxygen from ambient air, appears to be an affordable and easy access alternative to bring oxygen to patient’s home.
The aim of this pilot study was to evaluate for the first time the acceptance of oxygen delivered by a HFC, for the treatment of rebound headache in patient with MOH undergoing a withdrawal therapy. Additional data evaluating tolerability and efficacy of oxygen to achieve withdrawal and revert to episodic headache will also be collected.
Methods
This prospective monocentric (Lariboisiere Hospital, Paris) open-labeled feasibility study was approved by the local ethics committee (EUDRACT 2014-A01078-39) and registered at clinicaltrials.gov (NCT02302027).
Participants
Patients with a diagnosis of MOH (diagnosis according to International Classification of Headache Disorders (ICHD)-3β criteria 10 by trained headache specialists) considered for an outpatient care were invited to participate in this study if they met the following inclusion criteria: (1) age over 18 years, (2) underlying migraine or tension-type headache, and (3) no prophylactic treatment or treatment stable for at least 1 month. Non-inclusion criteria were (1) pregnancy, (2) previous withdrawal therapy, (3) analgesic use for another condition than headache, (4) contraindication to oxygen therapy, and (5) contraindication to ketoprofen or triptan use. Each patient signed a written informed consent.
Treatment
All patients received education on MOH and on the importance of withdrawal therapy. At D0, they were asked to stop all their usual acute medication but were prescribed “rescue” medication in the event of a non-tolerable headache but with the instruction to refrain their use as much as possible. For this purpose, investigator prescribed a unique box of ketoprofen or triptan. At M1 visit, restriction of this acute medication use was less strict, with a maximum of 8 days with intake within a month allowed. According to the standard care of MOH in our Department, introduction of prophylactic treatment was allowed if considered useful by the investigator.
In addition to this classical MOH treatment, oxygen delivery device (HFC: Platinum IRC9LXO2AWQ, 9 l/min) was supplied at patient’s home and patients were told to use it as much as they wanted at 9 l/min, for acute headache treatment, during the 6 months of this study.
Study procedure
After the day-0 (D0) inclusion visit, follow-up visits were scheduled at month 1 (M1), 3 (M3), and 6 (M6). Acceptance was evaluated at each follow-up visit by asking the patient if she/he regularly used of the device. Tolerability was assessed by asking the patients if she/he experienced any trouble with the device and with more specific questions about bulk, noise, and aesthetic tolerability. Occurrence of adverse event was considered at each visit. At D0 visit, the declarative number of days with headache, with migraine headache, and with painkillers use during the last month was recorded as well as the Hospital Anxiety and Depression Scale (HADS). During the whole follow-up, patients were asked to complete a headache diary containing days with oxygen use, rescue medications, and headache days. Patients were asked to fill in the Migraine Disability Assessment test (MIDAS) at D0, M3, and M6 as well as Patient Global Impression of Change (PGIC)—a scale between 1 and 7, from 1 = very much improved to 7 = very much worse—at each follow-up visit.
Outcome measures
The primary endpoint, acceptance, was the percentage of patents who regularly used the HFC during the follow-up, that is, answered at least two times “yes” at the question “Did you regularly used the HFC” that was asked at M1, M3, and M6. The principal secondary endpoint evaluating efficacy was the percentage at M6 of patients with no more chronic headache (less than 15 days of headache per month) and no longer in medication overuse. Other efficacy endpoints included evolution of headache days per month, of MIDAS, and PGIC score at M1, M3, and M6. A post hoc analysis with self-report data from the headache diary during the first month evaluated the percentage of attacks with response to oxygen (number of attacks with efficacy of HFC divided by total number of attacks with data on efficacy, including “unknown efficacy”) and the median individual efficacy rate (same calculation at the individual level).
Statistical analysis
Data were summarized as proportions for categorical variables, and median (min; max) for continuous variables. Percentages of patients who regularly used HFC were given with their 95% binomial confidence interval. We analyzed changes over time of headache days and MIDAS using linear-mixed models with two parameters (baseline and slope) and reported the estimated mean and standard error. PGIC score was dichotomized as 1–2 versus 3–7. All analyses were done with R.
Results
Twenty patients were included between January 2015 and November 2016. Because of protocol deviation (final diagnosis of cluster headache), one patient was secondary excluded. Of note, this 34 years old woman reported a good tolerability and efficacy of HFC at M1.
Fifteen out of 19 (78.9%) of patients completed at least one follow-up visit as 4 patients were lost of follow-up after inclusion. One patient did not attend the whole follow-up visits (patient number 7, vide infra).
Details of the 19 patients are shown in Table 1.
Characteristics of the 19 patients.

MIDAS: Migraine Disability Assessment test; HADS: Hospital Anxiety and Depression Scale; MD: missing data.
The underlying headache was migraine in all patients but one (tension-type headache, patient number 5). Median (min; max) age was 37 (19; 60) years, with 3 (16%) men. At D0, median declarative headache days per month was 25 (15–30), median MIDAS was 91 (13; 270), and median HADS 16 (5; 34). Fourteen (74%) were prescribed a new prophylactic treatment at inclusion (in addition to his previous stable propranolol treatment for patient number 7). There was no prophylactic treatment shift during the study but patient number 19 discontinued transiently her prophylactic treatment between M1 and M3.
HFC was accepted by 14 patients out of 15 (93.3% (68.0–99.8%)) who used it regularly. Patient 7 did not accept HFC as he did not use it regularly because he considered HFC inefficient. He had no tolerability concern but decided to discontinue the follow-up and agreed for a M6 phone call. No patient reported any adverse event or major trouble but four patients had significant complains about noise and bulk. Out of the 14 patients who regularly used HFC, 10 used it only to treat headache attacks while 4 (patient number 6, 8, 9, and 15) used it both for headache attacks treatment and outside attacks, mostly to try to prevent the occurrence of headache at work where they had no access to HFC. During the first month (which could be extracted from the diaries), the median (min; max) days with oxygen use was 10 (1; 30), the median number of oxygen intake of 14 (1; 47), the median duration for each intake was 30 min (12.1; 72), and the median total monthly use was 400 min (30; 1320).
All patients were prescribed ketoprofen as rescue treatment. During the first month, the median acute treatment intake was 2 (0; 6); three patients used other medication than ketoprofen (one used triptan, two used association of acetaminophen with codeine).
The analysis of our principal efficacy endpoint showed that at M6 of follow-up, 15/15 (100% (78.2–100%)) patients had no more chronic headache and were no longer in medication overuse.
Evolution of headache days per month and MIDAS at M1, M3, and M6 are shown in Figure 1.
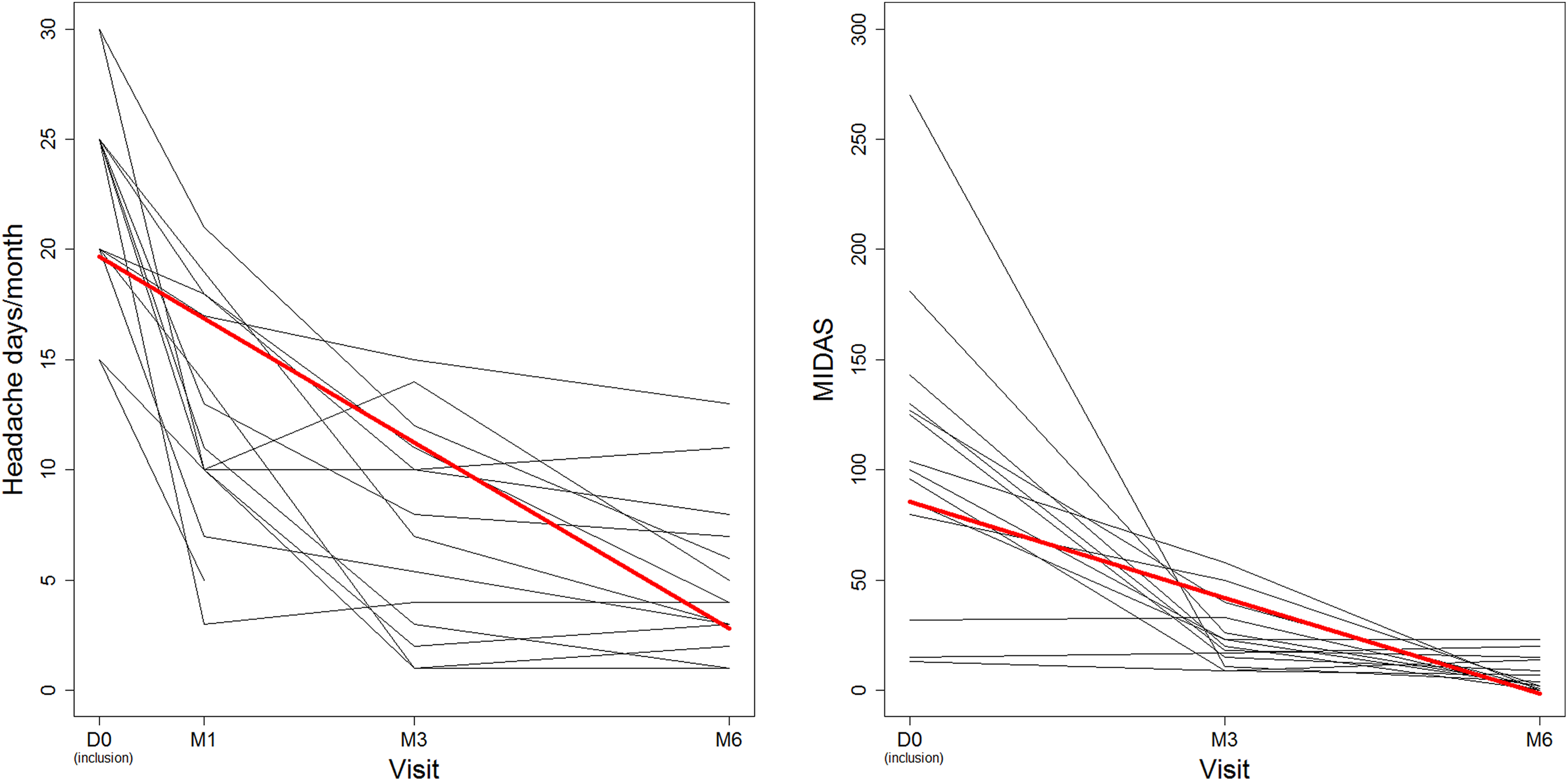
Spaghetti plot of evolution of headache days per month (right) and MIDAS (left). Red bold lines corresponds to the mean curve predicted by the linear-mixed model (N = 19 patients).
The mean (± standard error) initial headache days per month was estimated at 19.7 (±1.1) and decreased by −2.8 (±0.3) per month. The mean initial MIDAS was estimated at 85.4 (±9.6) and decreased by −14.5 (±2.6) per month. Having a score 1–2 at PGIC scale (patient considers being very much improved or much improved) during follow-up was 12/15 (80% (51.9–95.7%)) at M1, 11/14 (78.6% (49.2–95.3%)) at M3, and 14/14 (100% (76.8–100%)) at M6.
A post hoc analysis evaluating the efficacy of each oxygen intake during the first month (244 oxygen use: 102 considered efficacious, 33 inefficacious, 17 unknown, and 92 missing data) showed a percentage of attacks with response to oxygen of 67.1% and a median individual efficacy rate of 46.2% (20.9–71.4%).
Discussion
This first feasibility study confirms that oxygen delivered by HFC is well accepted and tolerated by patients undergoing a withdrawal therapy, with no significant adverse event. Even if four patients had substantial complains about noise and bulk related to HFC, this did not refrain their use, suggesting a good benefit/risk ratio. Noise, related to the compressor engine, remains a problem for patients who often face phonophoby, even if most of patients dealt with it by using a long tube while HFC was placed in distant room. Bulk was a limitation mostly for patients with small home and for travelers who could not bring HFC (dimension of Platinum 9 are 67 × 46 × 36 cm and weight 24 kg). Manufacturers should consider these limitations when working on the next generation device. Of note, tolerability of HFC and regular oxygen use decreased over time (data not shown). This probably relies on rarefaction of rebound headache remotely from withdrawal and on the occurrence of scarcer but more severe migraine attack, which appear to be less responsive to oxygen therapy, as reported by patients and concordant with previous data from Singhal et al. who suggested a better response to oxygen for moderate headache. 8 To our opinion, this suggests that oxygen therapy should be considered for a shorter period of time than 6 months, when rebound headache are more prevalent.
This study also suggests probable promising efficacy results; even if it was not designed for efficacy, the study reveals that all patients who completed the study reverted to episodic course of their primary headache with no more medication overuse. The evolution of headache days, the disability scale as well as the patient global impression of change confirms a good evolution of all patients. Of course, these results should be taken with caution as, in this small open-labeled study, improvement might rely on oxygen but also on withdrawal by itself, on prophylactic treatment, and/or on placebo effect. The good evolution of the patient who did not use HFC (patient 7) further supports this fact and the need for further larger double-blinded sham-controlled studies. The potential confounding effect of the introduction of prophylactic treatment should be considered when larger double-blinded studies will be build: As some teams start detoxifications without introduction of prophylactic treatment, 5 this should not raise any ethical problem.
Because our prespecified efficacy endpoint, achievement of withdrawal and reversion to episodic headache, is prone to bias, one could argue that we should have focused the efficacy of oxygen to treat each attack. The results of our post hoc analysis suggest that oxygen is efficient to treat two attacks out of three but not for all patients. However, the results should be taken with caution because of the post hoc nature of the analysis and because of the high number of missing data. These results underline the interest to evaluate this endpoint in future RCT as well as the interest of electronic diaries, as the paper ones are more permissive regarding unanswered questions responsible for missing data.
Larger studies will have to take into account our limitations: We did not include a run-in period: this would be of great interest when planning next studies, to improve accuracy of headache days before treatment. Another limitation may rely on the four patients lost of follow-up, which may weaken the results. We are aware of this but our drop-out rate, 21% (6–45%) (4/19) is close to others, as Carlsen et al. reported a 18% drop-out rate in their withdrawal therapy study. 5 Furthermore, if we consider the worst scenario—that all patients lost of follow-up did not accept the treatment and did not improve—the acceptance would be 14/19 (74% (49–91%)) and the percentage of patient reverting to episodic would be 15/19 (79% (54–94%)), which remains satisfying. Of note, in their study, Carlsen et al. reached a maximum of 70% of patients who reverted to episodic, without wakening their results as we did with the lost of follow-up.
In conclusion, oxygen delivered by HFC appears to be safe, feasible, and possibly efficient to help patient who have to undergo withdrawal therapy. A larger double-blind, sham-controlled prospective study is needed.
Clinical implications
– Oxygen delivered by a HFC for the treatment of headache is safe and well tolerated. – Oxygen may help patients with MOH to face the difficult detoxification period.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jerome Mawet received travel, accommodation, and meeting expenses from SOS oxygen, Air Liquide, AMGEN, and Homeperf or not funded by industry and received punctual payments for consultancy from Air Liquide and Novartis. Dominique Valade received received travel, accommodation, and meeting expenses from Adep Assitance and a punctual payment for consultancy from Air Liquide. Caroline Roos received travel, accommodation, and meeting expenses from Adep Assistance, Linde, TEVA, and SOS Oxygene or not funded by industry and received punctual payments for consultancy from Biogen, Teva, and Novartis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Association pour le Developpement du Centre d’Urgences des Cephalees (ADCUC) and Association pour la Recherche au Centre d’Urgence des Cephalees (ARCUC), two non-profit research associations that received fundings from Air Liquide and Homeperf.
