Abstract
Background
Short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) or with autonomic symptoms (SUNA) are grouped together within the trigeminal autonomic cephalalgias (TACs). However, the SUNCT and SUNA phenotype and management overlap with those of trigeminal neuralgia (TN). Additionally, a broad variety of cerebral pathologies are reportedly able to trigger either TN- or SUNCT-like pain, and emerging structural neuroimaging findings suggest the possible role of neurovascular conflict with the trigeminal nerve in SUNCT, further supporting aetiological and pathophysiological overlaps among SUNCT, SUNA and TN.
Case report
We present the first case of coexisting chronic SUNCT- and TN-like phenotypes caused by haemorrhagic infarct of the dorsolateral medulla.
Discussion
In light of our case, a perturbation of the dorsolateral medullary circuits may constitute an important pathophysiological component, supporting a unifying nosological hypothesis that considers SUNCT, SUNA and TN clinical variants of the same disorder.
Keywords
Introduction
Short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) or autonomic symptoms (SUNA) and trigeminal neuralgia (TN) are traditionally considered different disorders, thus grouped in separate sections of the International Classification of Headache Disorders (ICHD) (1). However, clinical and therapeutic similarities suggest that SUNCT, SUNA and TN may represent different variants of the same clinical entity (2). Additionally, a broad variety of cerebral pathologies are reportedly able to trigger either TN- or SUNCT-like pain (3,4). Emerging structural neuroimaging findings suggest the possible role of neurovascular conflict with the trigeminal nerve in SUNCT and SUNA, further supporting aetiological overlaps among SUNCT, SUNA and TN (5). We present a case of a patient who suffered a haemorrhagic infarct of the dorsolateral medulla causing a chronic headache condition characterised by coexistent SUNCT and TN attacks. This is the first case of SUNCT secondary to a brainstem stroke documented on magnetic resonance imaging (MRI) scanning. Furthermore, the exacerbation of headache attacks fulfilling the diagnostic criteria for SUNCT and TN, occurring ipsilaterally to the side of the medullary infarction, highlights the relevance of that anatomical structure in the pathophysiology of both disorders.
Case report
A 58-year-old male presented to our Headache Centre with a 16-year history of strictly right-sided headache, which began after a haemorrhagic infarct of the right dorsolateral aspect of the medulla, causing a clinical picture in keeping with a Wallenberg syndrome. At the time of the stroke the patient was diagnosed and managed within our Hospitals Trust, so we had access to the clinical and radiological information in the acute inpatient setting and for the subsequent outpatient follow-ups. A cerebral angiography at the time demonstrated a right vertebral artery dissection proximal to the origin of the posterior inferior cerebellar artery (PICA). The patient was started on warfarin for six months followed by aspirin and recovered satisfactorily from the immediate stroke-related neurological deficits. However, according to the patient and the neurologist who was looking after him at that time, approximately three weeks after the infarct, he began experiencing daily episodes of severe stabbing, sharp pain centred over the temple and the retro-orbital region with radiation to the cheek and the jaw. According to the patient’s headache diaries, the attacks lasted between five and 20 seconds and occurred on average 12–15 times daily, although occasionally he could experience up to 50–60 attacks daily. Approximately half of the headache attacks were accompanied by ipsilateral lacrimation, conjunctival injection, ptosis, eyelid oedema and rhinorrhoea. The remaining episodes had a similar phenotype but they were not associated with cranial autonomic symptoms. Both the headache attacks with and without autonomic symptomatology were witnessed in clinic by two of the authors (GL and MT). The pain could be triggered by touching the periorbital or temple area, cold wind blowing that side of the face, brushing teeth or bending over. The attacks occurred mainly during the daytime, seldom when he was asleep. An achy feeling along the face lasting for a few minutes often followed the severe headache attacks. There was no refractory period following triggered attacks. The patient did not have any familial history of headache and denied experiencing any headache prior to the infarct. His medical history was remarkable for hypertension and asthma. A recent MRI scan of the brain confirmed the presence of the mature haemorrhagic infarct of the right dorsolateral aspect of the medulla oblongata (Figure 1(a)) and also evidence of bilateral vascular loops at the root entry zone of both the sensory trigeminal roots without indication of compression, indentation or distortion of the nerves (Figure 1(b)). The pituitary gland appeared normal. A blink reflex study (Figure 2) showed normal early (R1) responses bilaterally, but absent ipsilateral and contralateral late (R2) responses to right supraorbital nerve stimulation, and indistinct, mildly delayed R2 responses to left supraorbital nerve stimulation consistent with a lesion in the spinal nucleus and/or tract of the trigeminal nerve in the medulla on the right with preservation of the main trigeminal sensory nucleus. At the time of the onset of the facial pain, a diagnosis of TN secondary to a medullary stroke was made. After the assessment in our centre, a diagnosis of chronic SUNCT and TN purely paroxysmal attributed to intracerebral haemorrhage was made. The patient failed to respond significantly to or tolerate trials of carbamazepine (800 mg/day), oxcarbazepine (1350 mg/day), gabapentin (900 mg/day), amitriptyline (40 mg/day, higher doses not tolerated), topiramate (not tolerated at 50 mg/day) and clonazepam (dose not known) at the time of the headache onset. We offered him trials of lamotrigine that was discontinued at the dose of 50 mg/day because of skin rash, duloxetine that was discontinued at the dose of 30 mg/day because of severe nausea and two sets of greater occipital and supraorbital nerve blocks that did not provide any benefit. The patient is currently on carbamazepine 600 mg/day and gabapentin 900 mg/day, which were only marginally effective in monotherapy, but they have been beneficial in combination by reducing the attack frequency and severity by 40%.
Magnetic resonance imaging sequencing of the brainstem showing a haemorrhagic infarct (black arrow) of the right dorsolateral medulla (a) and constructive interference in steady state (CISS) sequence showing bilateral vascular loops at the sensory trigeminal roots without indication of compression, indentation or distortion of the nerves (b). Blink reflex study. Right panel: stimulation of the left supraorbital nerve. Left panel: stimulation of the right supraorbital nerve. Upper four traces show responses recorded from the ipsilateral orbicularis oculi. Lower four traces show responses recorded from the contralateral orbicularis oculi.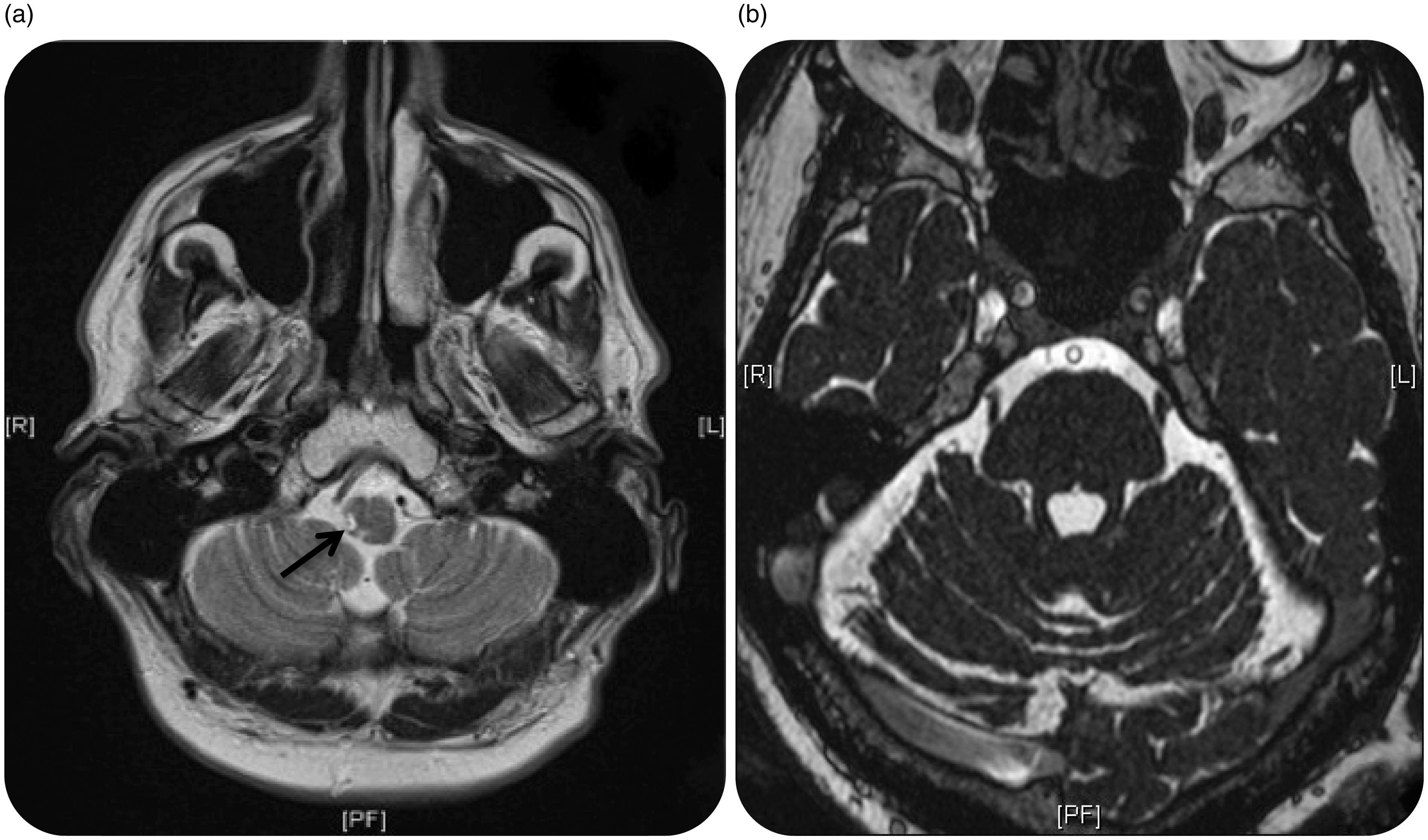

Discussion
Cases of SUNCT secondary to brainstem pathologies have been previously described (6). However, to date no cases of SUNCT secondary to an infarct of the brainstem, documented on neuroimaging, have been reported. Penart et al. (7) described a case of SUNCT syndrome secondary to a presumed brainstem infarction. However, the patient’s MRI scan revealed signs of cerebellar hemisphere infarction without brainstem involvement. Conversely, several cases of TN secondary to brainstem infarcts are described (4), including one case of TN secondary to a lateral medullary infarction (8). In our patient extensive neuroimaging studies conducted at the time of the onset of the acute neurological symptoms demonstrated a haemorrhagic infarct due to the PICA dissection, suggesting that vascular insults of the brainstem can exacerbate a SUNCT-like syndrome, as well as TN.
The infarct of the medulla caused the coexistence of two very similar chronic headache phenotypes: one fulfilling the criteria for SUNCT, in which the attacks are accompanied by pronounced cranial autonomic activation and another, fulfilling the criteria for TN, in which the attacks are featureless. Despite the fact that the laterality, the site of the pain, the pain character, the duration and frequency of occurrence of the attacks equally fulfilled the SUNCT and the TN diagnostic criteria (1), opting for a single diagnosis of SUNCT in our patient is not possible, according to the International Headache Society (IHS) diagnostic criteria, in view of the presence of featureless attacks. Similarly, a single diagnosis of TN with cranial autonomic features cannot be formulated according to the IHS criteria (1), despite case series of V1 (ophthalmic nerve territory) TN with attacks accompanied by mild and sporadic cranial autonomic features have been reported (9,10). The absence of a refractory period has also been thought to be a useful criterion in clinical practice to distinguish between SUNCT and TN (11). In our case the absence of a refractory period after triggered attacks may have suggested a diagnosis of SUNCT with some featureless attacks, rather than the coexistence of SUNCT and TN. However, neither the SUNCT nor the TN diagnostic criteria include the presence or absence of a refractory period (1). Moreover, a few cases of SUNCT with preserved refractory periods after triggered attacks have been described (12). This clinical overlap highlights the diagnostic complexity of distinguishing between these conditions, despite the supposedly distinctive clinical phenotype of SUNCT and TN for features such as cranial autonomic symptoms and the refractory period.
In terms of response to treatments, our patient obtained a partial benefit from a combination of carbamazepine and gabapentin. Interestingly, whereas carbamazepine is the drug of choice in TN (13), open-label evidence suggested only a partial effect in about one-third of SUNCT patients (14). Conversely, gabapentin has been shown to be effective in SUNCT (14), although its evidence in TN’s medical management is lacking. The two medications did not produce a noticeable improvement in monotherapy. However, in combination, they led to a moderate benefit with reduction in severity and frequency of both the SUNCT- and TN-like attacks, supporting the coexistence of the two conditions in the same individual.
The clinical and therapeutic similarities in SUNCT and TN suggest an involvement of similar pathophysiological mechanisms. The proposed pathophysiology for SUNCT resolves around the hyperexcitability of the trigemino-autonomic reflex possibly due to an abnormal modulation activity of the posterior hypothalamus via the trigemino-hypothalamic fibres, which seem to connect the posterior hypothalamus with the trigeminal nucleus caudalis (15). Central mechanisms may account for the pathogenesis of TN in patients with and without structural damage on the trigeminal sensory root. These mechanisms include a derangement in the physiological activity of the wide dynamic trigeminothalamic neurons (WDN) in the trigeminal nucleus caudalis (16). Furthermore, the occurrence of autonomic phenomena in some TN attacks may suggest the implication of similar structures involved in SUNCT syndrome, namely the trigeminal-autonomic reflex. Both SUNCT and TN pathophysiological theories highlight the key role of brainstem dysfunction at the level of the caudalis subdivision of the spinal trigeminal nucleus. In our case, both neuroimaging and neurophysiological findings suggest a lesion in close proximity to the trigeminal spinal nucleus, likely the pars caudalis, underpinning the strategic importance of medullary circuits in causing SUNCT- and TN-like phenotypes. In light of our case, a perturbation of dorsolateral medullary pathways may constitute an important shared pathophysiological mechanism supporting a unifying nosological hypothesis that considers SUNCT, SUNA and TN clinical variants of the same neurobiological disorder.
Clinical implications
Short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) syndrome can occur secondary to a brainstem vascular insult. The coexistence of SUNCT and trigeminal neuralgia ipsilaterally to the side of the infarction suggests the strategic importance of the dorsolateral medulla in the pathophysiology of both conditions. A perturbation of dorsolateral medullary pathways may constitute an important shared pathophysiological mechanism supporting the hypothesis that SUNCT and trigeminal neuralgia may be variants of the same disorder.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
