Abstract
Background
Huanglianjiedu decoction (HLJDD) is a classical Traditional Chinese Medicine (TCM) prescription with thousand years of clinical use against various malignancies, including pancreatic adenocarcinoma (PAAD). However, its potential bioactive component and molecular mechanism remains unclear.
Aims
This study is to inspect the HLJDD mechanisms of action against PAAD via integrated computational and pharmacochemistry strategy,
Methods
A PAAD xenograft model was established by subcutaneous injecting Panc02 cells into C57BL/6 mice. Ultra-high performance liquid chromatography with tandem mass spectrometry (UHPLC-MS/MS) was engaged to determine constituents of HLJDD and assessed for pharmacokinetic scheme using the TCM Systems Pharmacology Platform (TCM-SP). Differentially expressed genes (DEGs) of PAAD was retrieved from the transcriptome dataset GSE43795, followed by recognizing overlapping targets the oncogenes and target genes of PAAD and HLJDD, respectively. Putative signaling pathways of HLJDD in treating PAAD were enriched using KEGG and GO analyses. The anti-PAAD effects of HLJDD was assessed
Results
HLJDD significantly suppressed the growth of transplanted PAAD tumors, constrained PAAD progression, and induced apoptosis and S-phase arrest. Seventy-five active components meeting the drug-likeness criteria and 278 target genes of HLJDD were identified. KEGG analysis indicated that the top three enriched pathways were cancer, AGE-RAGE signaling, and IL-17 signaling pathways. Disease enrichment analysis highlighted immune, pharmacological, and cancer-related diseases as the top three categories. A total of 47 potential target genes were identified. Immunoblotting revealed that HLJDD inhibited PI3K and MAPK-related signaling pathways, while immunohistochemical staining confirmed that HLJDD suppressed the expression of phosphorylated MAPK and ERK1/2.
Conclusion
HLJDD inhibited PAAD
Keywords
Introduction
Pancreatic adenocarcinoma (PAAD) is a fatal malignant tumor with high asymptomatic incidence, increasing prevalence and a 5-year survival rate of 11%.1,2 Although treatment strategies are continuously optimized, clinical outcomes for patients with PAAD remain unsatisfactory. Owing to the highly heterogenous nature of PAAD and the limited availability of precisely targeted treatment, together with the requirement of extensive research and development processes involving basic science research and multiple phases of clinical trials, the development of novel anti-PAAD western medicine have been a slow and challenging process. Nowadays, researchers have focused on the therapeutic effects of functional medicine with multiple targets and pathways. Traditional Chinese medicine (TCM) not only offers numerous natural remedies for cancer treatment without serious side effects, its thousand years of clinical application and its rich repository of bioactive components presents a promising alternative for novel pharmaceutical agent identification.
Huanglianjiedu Decoction (HLJDD) is a renowned classical TCM prescription with thousands of years of clinical applications against various malignancies, particularly for the management of gastrointestinal discomfort. With a combination of four herbal medicines, namely Huanglian (
Previous research has demonstrated the efficacy of HLJDD in inhibiting hepatocellular carcinoma (HCC) through the suppression of nascent protein synthesis via the inactivation of eukaryotic elongation factor-2 (eEF2). 3 This was attributed to the activation of AMP-activated protein kinase (AMPK) and subsequent activation of eEF2 kinase (eEF2 K), which inactivates eEF2 through phosphorylation. Additionally, HLJDD has shown promise in treating other types of cancer, such as oral squamous cell carcinoma (OSCC), by utilizing network pharmacology to identify active ingredients and core targets, followed by experimental validation, 4 highlighting the importance of pathways like MAPK/ERK and NF-κB in mediating its anti-cancer effects.
Moreover, single herbal extract and individual active compounds of HLJDD have been implicated in suppressing PAAD,5–13 suggesting that HLJDD possessed a high potential for treating PAAD. Palmatine suppresses the interaction between pancreatic cancer and stellate cells by inhibiting survivin and COL1A1, thereby disrupting the tumor microenvironment. Total flavonoid aglycones extract (TFAE) from Radix Scutellariae induces apoptosis and autophagy in pancreatic cancer cells by inhibiting the PI3 K/Akt/mTOR pathway, with autophagy functioning as a cytoprotective process against apoptosis. Wogonin (WOG) activates autophagy-related pathways and ROS generation while inhibiting the mTOR pathway, and suppresses oncogenic proteins such as Mcl-1 and CDK-9, demonstrating its potential for treating pancreatic cancer. Baicalein promotes apoptosis by releasing cytochrome c, activating caspases, and decreasing Mcl-1 expression. Additionally, baicalin inhibits cell proliferation and induces apoptosis via the JNK/Foxo1/BIM signaling pathway. These multifaceted actions of HLJDD's components underscore its comprehensive therapeutic potential against PAAD. However, studies have found that the precise and rigorous combination of herbs with multiple components, targets, and signaling pathways is more beneficial than single agents. 14 Hence, there is still a need to study the complicated connections between HLJDD and PAAD.
Recent investigations into TCM have highlighted significant challenges due to its intricate nature. TCM formulas, as aforementioned, composed of numerous chemical compounds, interact with various biological targets and pathways, complicating our understanding of their molecular mechanisms. 15 This complexity, combined with limited insights into their molecular actions, has impeded the development and wider acceptance of TCM. The emergence of bioinformatics and network pharmacology, which integrates systems biology, polypharmacology, and molecular network analysis, offers crucial information about the chemical compositions and ADME (absorption, distribution, metabolism, and excretion) properties, as well as the interplay between compounds, genes, targets, and diseases. 16 These approaches have considerably enhanced our grasp of the holistic and synergistic nature of TCM's molecular networks. 17 Our prior studies have successfully utilized these methods to delve into the intricacies of TCM. 18 This study leverages network pharmacology combined with ultra-high performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS) to analyze different cancer types, providing broader mechanistic insights. We identified several active components in HLJDD that meet drug-likeness criteria, offering a more comprehensive and detailed analysis than previous studies.
PI3 K and MAPK signaling pathways are with well-established roles in cancer biology. These pathways are critically involved in regulating key cellular processes such as growth, survival, proliferation, and apoptosis, which are often dysregulated in cancers, including PAAD. The PI3 K pathway is known for its role in promoting cell growth and survival through downstream effectors like AKT, while the MAPK pathway influences cell division and differentiation via cascades involving ERK and p38. By targeting these pathways, we aimed to elucidate the molecular mechanisms by which HLJDD exerts its therapeutic effects, ensuring a focused and relevant investigation grounded in current cancer research. Utilizing both
Materials and Methods
UHPLC-MS/MS Analyses
To prepare the samples for analysis, 100 μL of HLJDD was thoroughly mixed with 80% methanol (prechilled to 4 °C) by vortexing, followed by a 5-min incubation on ice. The samples were then centrifuged at 15,000 g for 20 min at 4 °C to collect the supernatant, which was subsequently diluted with LC-MS grade H2O to achieve a final concentration of 53% methanol. After a second centrifugation, the samples were analyzed using UHPLC-MS/MS analysis (Novogene Co., Ltd, Beijing, China). The analysis employed a 1.9μm Hypesil Gold column (100 mm × 2.1 mm) with a flow rate of 0.2 ml/min over a 17-min duration. Two eluents were utilized in different polarity modes: for positive and negative modes, eluent #1 consisted of 0.1% formic acid and 5 mM ammonium acetate (pH 9.0), while eluent #2 were methanol in both modes. The solvent gradient procedure was as follows: 2% methanol for 90 s, a linear increase to 85% methanol over 180 s, 85% to 100% methanol over 600 s, maintaining 100% methanol for 600 s, and finally returning to 2% methanol over 720 s. The mass spectrometer parameters, as previously indicated, 19 included an auxiliary gas temperature of 350 °C and S-lens RF level of 60 for both positive and negative polarity modes.
Network Pharmacological Identification of Bioactive Components, Targets, Functional Enrichment and Potential Pathways
To identify the bioactive components and targets of HLJDD, we performed comprehensive network pharmacological analyses based on the results from UHPLC-MS/MS results. All phytochemicals from the four constituent herbs of HLJDD were retrieved from the TCM systems pharmacology database and analysis platform (TCMSP, http://tcmspw.com/). 20 The TCMSP database includes data on Chinese herbal medicines and the relationships among drugs, targets, and diseases, with properties such as oral bioavailability (OB), and drug likeness (DL).
The draggability of the bioactive compounds in HLJDD was assessed using criteria for OB and DL. 21 OB represents the fraction of a drug that reaches systemic circulation after oral administration, and DL estimates how drug-like a substance is. Compounds were considered bioactive if they had OB greater than or equal to 30% and DL greater than or equal to 0.18. 21 These parameters were screened using TCMSP database to identify compounds suitable for further analysis. 20
Protein targets of the identified compounds were retrieved from the TCMSP and further validated using the UniProtIDs Knowledgebase (UniProtKB, http://www.uniprot.org), which provides detailed protein sequence and fun ctional information. Functional enrichment analyses, including Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment and disease-related identification, were conducted using the Metascape (http://metascape.org) and Database for Annotation, Visualization and Integrated Discovery (DAVID, https://david.ncifcrf.gov/).
22
To obtain differentially expressed genes (DEGs) associated with PAAD, the GEO (https://www.ncbi.nlm.nih.gov/geo/) dataset GSE43795 was analyzed using the “DESeq” package of R (version 4.0.3), with the selection criteria of |Fold-Change | > 1 and
Preparation of HLJDD
The experimental drug HLJDD (Cat. #2140) was sourced from Nongbenfang of Pura Pharm International Ltd (H.K.), provided as a concentrated granule. The preparation process began by mixing all four crude herbs and boiling them in 1000 ml distilled water for 2 h to obtained herbal extract. After boiling, the solution was centrifugation at 10,000 rpm for 30 min. This extraction process was carried out twice, with the supernatants from both extractions combined and evaporated until dry. The dried granules of HLJDD were then dissolved in PBS (Gibco, USA) to achieve a final concentration of 20 mg/mL, followed by filtration through a 0.22 μm filter. The prepared solution was stored at −20 °C for future use.
Cell Lines and Culture Conditions
The mouse pancreatic cancer cell line Panc02 and the human pancreatic cancer cell line Capan-1 were obtained from the American Type Culture Collection (ATCC, USA) and Frederick National Laboratory for Cancer Research (USA), respectively. Both cell lines were cultured in DMEM (Gibco, USA) supplemented with 10% Fetal bovine serum (FBS, Invigentech, USA), 100 U/mL penicillin, and 100 μg/mL streptomycin (Gibco, USA). The cells were maintained in a humidified atmosphere with 5% CO2 at 37 °C.
Cells were seeded in 6-well plates at a density of 1 × 106 cells per well and allowed to adhere overnight, unless otherwise specified. The following day, cells were treated with various concentrations of HLJDD (0, 50, 100, 200 μg/mL) for 24 h. For the rescue experiments, cells were co-treated with either 740 Y-P (Y413958, Shanghai Aladdin Bio-Chem Technology, Shanghai China, a PI3 K activator) at 30 μM or PMA (HY-18739, MedChemExpress, USA, a PKC activator for induction of MAPK signaling) at 500 nM along with HLJDD treatment.
Cell Proliferation and Colony Formation Assays
The cell viability of Capan-1 and Panc02 cells following treatment with HLJDD was assessed using the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. Initially, Capan-1 and Panc02 cells (5000 cells in 100 μL of media per well of a 96-well plate) were treated with carious concentrations of HLJDD (0, 50, 100, 200, 400, 800, 1600, 3200 μg/mL) and incubated for 24, 48, and 72 h. After incubation, 10 μL of the MTT solution (5 mg/mL, Sigma-Aldrich, USA) was added to each well and further incubated at 37 °C for 4 h. Following this, 100 μL of dimethyl sulfoxide (DMSO, Sigma-Aldrich) was used to dissolve the formazan crystals. Absorbance was measured at 595 nm using a microplate reader (Molecular Devices, USA). The half-maximal inhibitory concentration (IC50) was calculated, and growth inhibition curves were plotted.
For the colony formation assay, Capan-1 and Panc02 cells were seeded at a density of 1 × 103 cells per well in a 12-well plate and treated with different concentrations of HLJDD (0, 50, 100, 200 μg/mL) for 7 days. The resulting colonies were fixed with 4% paraformaldehyde (PFA, Sigma-Aldrich) for 20 min and stained with crystal violet (Sigma-Aldrich) for 15 min at room temperature. After staining, the colonies were photographed using an XDS-500C microscope (Shanghai Cai Kang, China).
Apoptosis Assay and Cell Cycle Analysis
Capan-1 and Panc02 cells, seeded at a density of 1 × 106 cells per 6-well plate, were treated with HLJDD and incubated at 37 °C for 48 h. For the apoptosis assay, harvested cells were resuspended in 500 μL of 1 × binding buffer from the Annexin V-FITC/PI apoptosis detection kit (BD Bioscience, USA). The cells were then stained with Annexin V and propidium iodide (PI) for 20 min in the dark. For cell cycle analysis, harvested cells were fixed in ice-cold 70% ethanol overnight. After centrifugation, the cells were treated with PI staining buffer (BD Bioscience, USA) for 15 min in the dark. Both the apoptosis assay and cell cycle analysis were conducted using a CytoFLEXS flow cytometer (Beckman, USA).
Wound-Healing Migration and Transwell Invasion Assays
In the wound-healing migration assay, Capan-1 and Panc02 cells (2 × 106 cells per well) were seeded in 6-well plates and grown to near confluence. A scratch wound was created in the cell monolayer, and the cells were treated with HLJDD. Images of the wound were taken at 0, 24, and 48 h, and wound healing was quantified using Image J software.
For the invasion assay, Transwell chambers with an 8.0 μM pore size were used. The lower surface of the Transwell inserts was coated with Matrigel (1:2 dilution, Corning, USA). Cells (1 × 105 cells in 200 μL of serum-free medium) were placed in the upper chamber, while the lower chamber contained 800 μL of medium with 10% FBS. After incubating at 37 °C for 24 h with HLJDD, the cells that invaded through Matrigel and adhered to the lower surface of the membrane were fixed, stained, and counted under a light microscope to assess the level of invasion.
In vivo Experiments
The research protocol was approved by the Ethics Committee of Animal Experiments, Cancer Research Center, (FUSCC-IACUC-S2022-0444). Twenty-four male C57BL/6 mice (4-6 weeks old, weighing 20-24 g) were obtained from Shanghai Jihui Experimental Animal Breeding Co., Ltd (Shanghai, China) and maintained in a specific pathogen-free environment. Panc02 cell suspensions (1 × 108 cells/mL), mixed with Matrigel at a 1:1 ratio, were subcutaneously injected into the right flank of each mouse (100 μL per injection). One-week post-injection, the mice were randomly divided into four groups: control (receiving 0.2 ml saline), low-dose HLJDD (50 mg/kg), medium-dose HLJDD (100 mg/kg), and high-dose HLJDD (200 mg/kg). The treatments were administered daily via oral gavage for 21 days. The HLJDD dosage was determined based on traditional Chinese medicine research methodologies. Tumor size and mouse body weight were monitored biweekly. On day 22, the mice were euthanized, and the tumors were dissected and fixed in 4% PFA (Biosharp, China) for 48 h for pathological examinations. Tumor volume was calculated using the formula: tumor volume = length × width × width /2.
Hematoxylin & Eosin (HE), Immunohistochemistry (IHC), and Immunofluorescence (IF) Staining
For Hematoxylin and Eosin (H&E) staining, tissue slides were processed using an H&E staining kit (Cat. #ab245880, Abcam, UK). For immunohistochemistry (IHC) and immunofluorescence (IF) staining, slides were first treated with 100 μL of hydrogen peroxide (H2O2) blocking solution for 10 min. Antigen retrieval was then performed by boiling the sections in a solution of 1 mmol Tris-EDTA (pH 9.0) for 15 min, followed by a 15-min warm incubation. After antigen retrieval, the sections were blocked and incubated overnight at 4 °C with primary antibodies. The primary antibodies used included anti-Ki-67 antibody (Cat. #ab15580, Abcam, UK), anti-p-ERK1/2 antibody (Cat. #4370, Cell Signaling Technology, CST, USA), and anti-p-p38 MAPK antibody (Thr180/Tyr182) (Cat. #4511S, CST). The following day, the slides were counterstained with DAPI (Sigma-Aldrich) or HPR-conjugated fluorescence antibody (Abcam) for 10 min at room temperature. The stained slides were then examined under an inverted fluorescence microscope (Nikon, Japan).
Immunoblotting
After incubating the cells with various concentrations of HLJDD for 24 h, cells were harvested, and total protein was extracted using RIPA buffer (Beyotime Biotechnology, China). Protein concentrations were determined using the Bradford protein assay kit (Beyotime Biotechnology, China).
The protein samples were subjected to immunoblotting via SDS-PAGE electrophoresis and transferred onto polyvinylidene difluoride (PVDF, Millipore) membranes. The membranes were then proved overnight with at 4 °C with primary antibodies against The membranes were then probed overnight at 4 °C with primary antibodies against GAPDH (14C10, Rabbit mAb, CST #2118S), mTOR (7C10, Rabbit mAb, CST #2983 T), p-mTOR (Ser2448, D9C2, XP® Rabbit mAb, CST #5536 T), pan-AKT1/2/3 (Affinity AF6261), p-AKT1/2/3 (Ser473, Affinity AF0016), PI3 K p85 alpha (Affinity AF6241), p-PI3 K p85 alpha (Tyr607, Affinity AF3241), p38 MAPK (D13E1, XP® Rabbit mAb, CST #8690 T), p-p38 MAPK (Thr180/Tyr182, D3F9, XP® Rabbit mAb, CST #4511 T), ERK1/2 (137F5, Rabbit mAb, CST #4695 T), and p-ERK1/2 (Thr202/Tyr204, D13.14.4E, XP® Rabbit mAb, CST #4370 T). Following primary antibody incubation, the membranes were incubated with HRP-linked antibodies (1:5000, CST) at room temperature for 2 h. Chemiluminescence reagent (Cat. #SQ201; Epizyme, China) was then applied to the membranes, and then protein bands were visualized using a chemiluminescent imaging machine (Tanon 6100, China). Band intensities were quantified using Image J software, with all protein levels normalized to GAPDH.
Data Analysis and Statistics
Experimental data were analyzed by GraphPad Prism 9.0. Continuous data were expressed as mean ± standard deviation (mean ± SD). Comparisons between two samples were performed using Student's
Results
HLJDD Suppressed PAAD Proliferation, Migration and Invasion in Vitro
To investigate the growth-inhibitory effects of HLJDD on pancreatic adenocarcinoma (PAAD) cell lines, Capan-1 and Panc02, several in vitro assays were conducted.
A proliferation assay was first employed to measure cell growth following HLJDD treatment. HLJDD demonstrated a time- and dose-dependent inhibition of proliferation in both Capan-1 and Panc02 cells (Figure 1A). The IC50 at 72 h were approximately 400 μg/mL for Capan-1 cells and 800 μg/mL for Panc02 cells. Based on these results, non-cytotoxic concentrations of HLJDD (0, 50, 100, and 200 μg/mL) were selected for subsequent experiments.
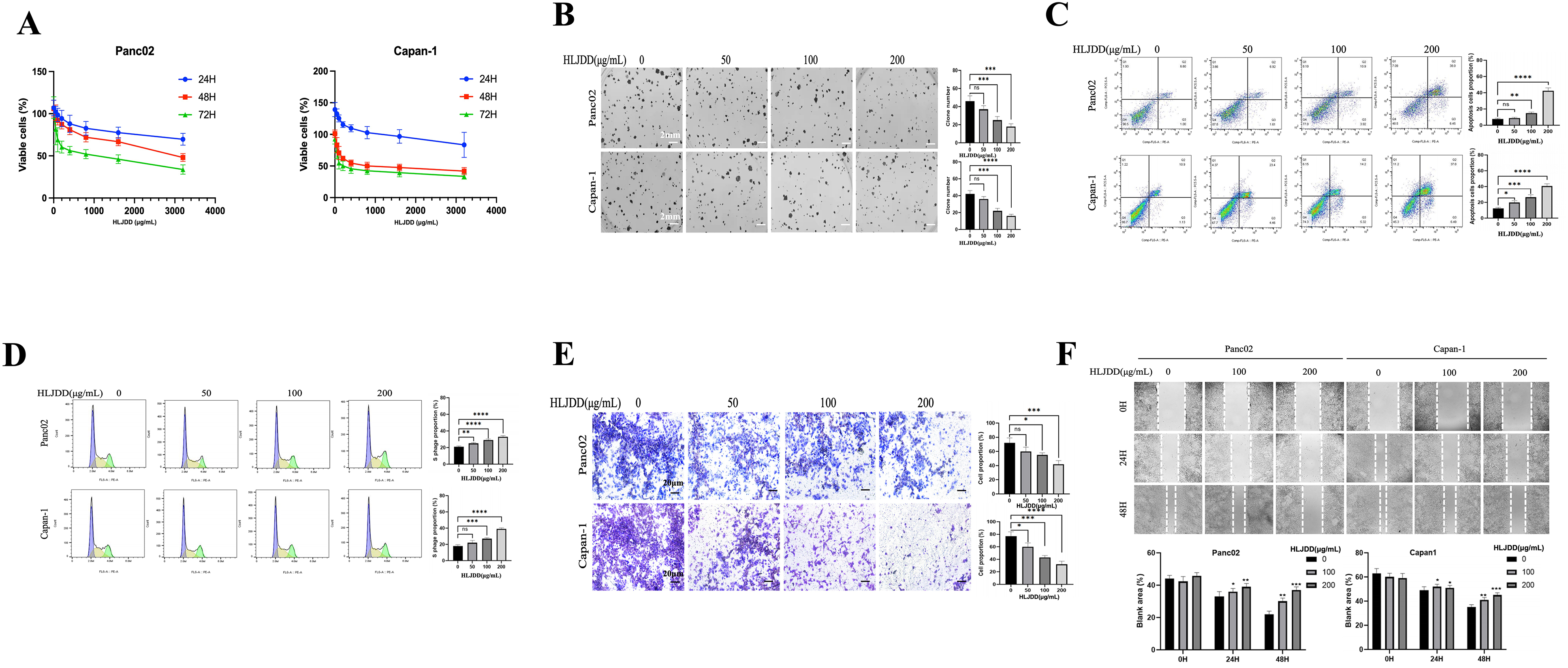
HLJDD suppressed PAAD cell proliferation
A colony formation assay was then performed to assess the long-term effects of HLJDD on the clonogenic potential of PAAD cells. Treatment with HLJDD at 100 and 200 μg/mL for 7 days significantly inhibited the colony formation in both Capan-1 and Panc02 cells (Figure 1B).
To evaluate the induction of apoptosis, Annexin V/PI staining was conducted on HLJDD-treated PAAD cells. The results indicated that HLJDD treatment induced significant early and late apoptosis in a dose-dependent manner in Capan-1 and Panc02 cells (Figure 1C). Additionally, PI staining of cellular DNA for the cell cycle analysis revealed that HLJDD induced an S- phase arrest in these cell lines (Figure 1D), indicating an interruption of cell cycle progression.
Finally, the effects of HLJDD on cell migration and invasion were examined. HLJDD treatment markedly suppressed the migratory (Figure 1E) and invasive (Figure 1F) capabilities of both Capan-1 and Panc02 cells, suggesting its potential to inhibit metastatic behavior. Together, these results demonstrate that HLJDD effectively suppressed several malignant behaviors of PAAD cells in vitro, including proliferation, colony formation, apoptosis induction, cell cycle arrest, and inhibition of migration and invasion.
HLJDD Suppressed PAAD Growth in Vivo
To validate the therapeutic effects of HLJDD on PAAD growth, a subcutaneous xenograft model was employed. To closely mimic a more authentic environment for tumor growth, we chose to use the Panc02 cell line in an immunocompetent C57BL/6 mouse model, rather than using the immunodeficient nude mouse model with Capan-1 cells. Throughout the treatment process, there were no significant differences in body weight between the treatment and control groups (Figure 2A). Oral administration of HLJDD significantly suppressed the growth of implanted PAAD in a dose-dependent manner (Figure 2B). At the endpoint of the experiment, tumors in the HLJDD-treated groups were notably smaller than those in the control group, further verifying the inhibitory effect of HLJDD on PAAD (Figure 2C). The corresponding tumor weights also demonstrated a significant reduction with HLJDD treatment (Figure 2D). These findings collectively demonstrated that HLJDD exerts a potent antitumor effect
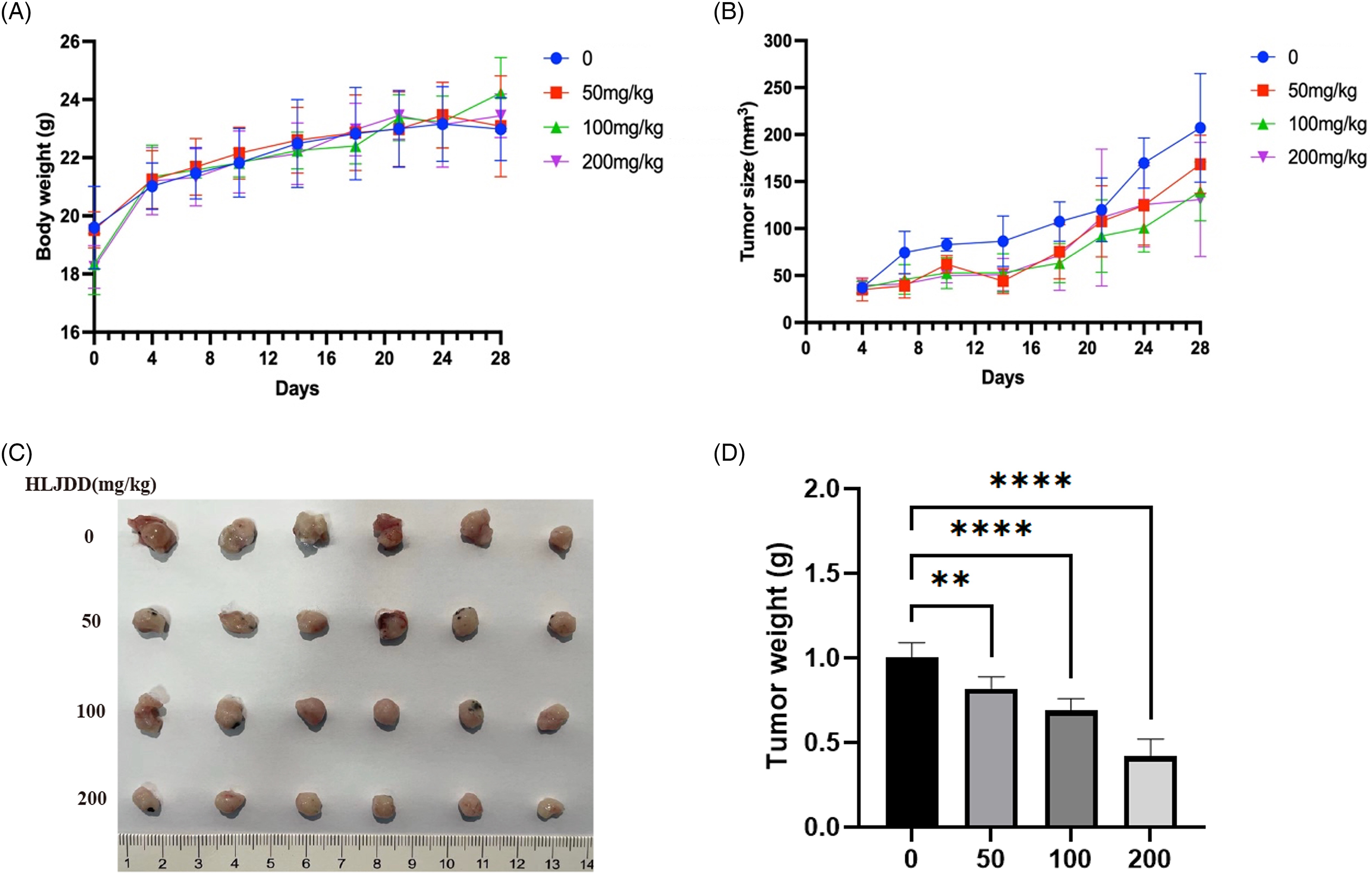
HLJDD inhibited PAAD growth
Active Compounds of HLJDD
The active compounds of HLJDD were identified using UHPLC-MS/MS in both negative and positive polarity modes (Figure 3A). Their respective ADME profile were retrieved from the TCMSP database. A total of 75 active ingredients meeting the inclusion criteria of OB ≥ 30% and DL ≥ 0.18 were selected (Supplementary Table 1). Among these, 14 active compounds were identified from RC, 30 from RS, 37 from CP, and 23 from FG. The details are shown in Table 1. The active compounds, along with 278 related target genes (Supplementary Table 2), were input into Cytoscape 3.7.2 to construct the “medicinal materials - active ingredients - target genes” network (Figure 3B).

(A) UHPLC-MS/MS graph on positive and negative ion modes of HLJDD extract, and (B) the “medicinal materials-active ingredients-target gene” graph of HLJDD. Pos, positive ion mode; Neg, negative ion mode.
Components of the HLJDD Granule.

Target Gene Prediction and Pathway Analysis of HLJDD
The 278 target genes of HLJDD were analyzed using the Metascape website for KEGG pathway enrichment. The results indicated that the top three enriched pathways (Figure 4A) were the pathways in cancer, the AGE-RAGE signaling pathway, and the IL-17 signaling pathway. Notably, the pathways in cancer include crucial signaling mechanisms such as the PI3 K/AKT and MAPK signaling pathways, both of which play pivotal roles in cell growth and survival. For disease association analysis, the target genes were input into the DAVID database. The analysis revealed that the top three diseases categories associated with HLJDD were immune, pharmacogenomic, and cancer-related diseases (Figure 4B).

KEGG pathway enrichment and disease enrichment analysis of HLJDD. (A) The top 20 pathway enrichment analyses. (B) The top 20 related disease analyses. (C) The functional modules for protein interaction and function sets.
Furthermore, the target genes were entered into the STRING database for protein-protein interaction (PPI) network analysis. The results highlighted three internally distinct clusters interacting functional genes within the HLJDD targets (Figure 4C). The first cluster includes key oncogenes and signaling molecules such as AKT1, EGFR, and BCL2L1, indicating involvement in the PI3 K/AKT signaling pathway critical for cell growth and survival. The second cluster consists of genes related to neurotransmitter receptors and stress response, suggesting an impact on the tumor microenvironment. The third cluster features genes involved in immune response, inflammation, and cell cycle regulation, such as STAT1, MAPK1, and CDK4, highlighting potential modulation of immune and inflammatory pathways. These three clusters collectively suggest that HLJDD targets multiple pathways and processes involved in cancer progression, including cell cycle regulation, apoptosis, signaling pathways (PI3 K/AKT and MAPK), stress response, and immune modulation. The alignment between PPI clustering through network pharmacological analysis with the
Identification of Target Genes of HLJDD on PAAD
To identify the target genes of HLJDD in PAAD, the transcriptome dataset GSE43795 was screened from the GEO database This dataset includes RNA expression data analyzed using the Illumina HumanHT-12 v4 microarray from six PAAD tissues and five normal pancreatic tissues. By applying the criteria of | Log2FC | > 1 and
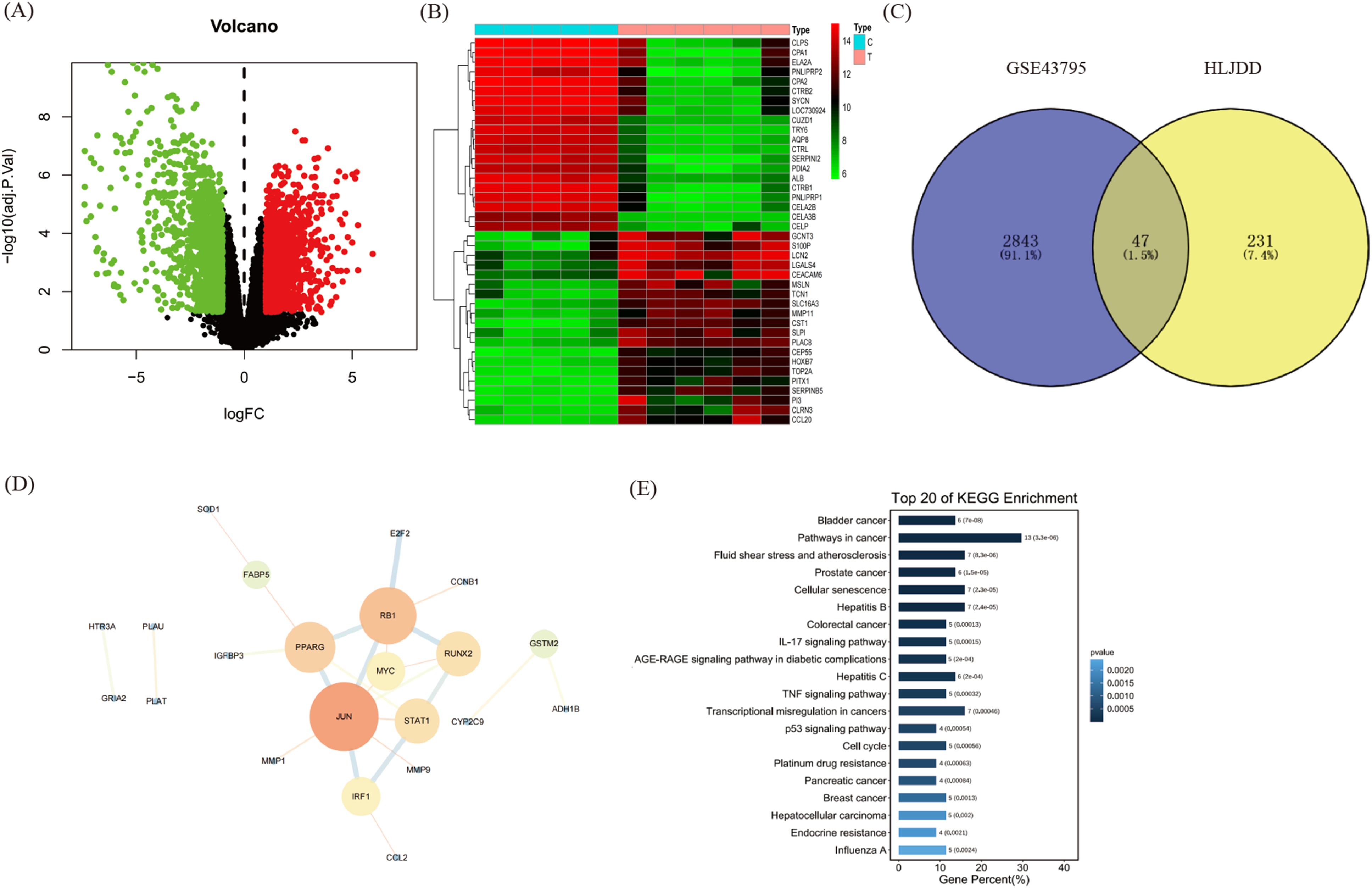
Targets identification of HLJDD in the treatment of pancreatic cancer. (A) The volcano plot of PAAD DEGs. (B) The heat map of PAAD DEGs. (C) The Venn diagram of core genes in HLJDD target genes and PAAD DEGs. (D) The PPI network diagram of the interaction between the 47 overlapping genes. (E) KEGG pathway analyses of the 47 overlapping genes.
PI3K and MAPK-Related Signaling Pathways may be Involved in the Therapeutic Effect of HLJDD on PAAD
The network pharmacological analysis indicated the potential involvement of cancer-related signaling pathways in the therapeutic effects of HLJDD on PAAD. To further explore these pathways, immunoblotting was employed, revealing that HLJDD effectively inhibited the PI3 K and MAPK-related signaling pathways. This inhibition was observed through reductions in the levels of phosphorylated proteins such as PI3 K, AKT, mTOR, p38 MAPK, and ERK1/2 (Figure 6). Furthermore, IHC staining was conducted to investigate the regulation of phosphorylated MAPK and ERK1/2 in tumor tissues. The results showed that HLJDD treatment at doses of 50, 100, and 200 mg/kg significantly suppressed the levels of phosphorylated p38 MAPK and ERK1/2 in vivo, compared to the vehicle control (Figure 7). The H&E staining demonstrated tumor architecture, and Ki67 staining indicated reduced proliferation with increasing HLJDD doses(Supplementary data). These findings confirm that HLJDD suppresses the PI3 K and MAPK signaling pathways both in vitro and in vivo, supporting its therapeutic potential against PAAD. The results confirmed that HLJDD indeed suppressed the levels of phosphorylated MAPK and ERK1/2
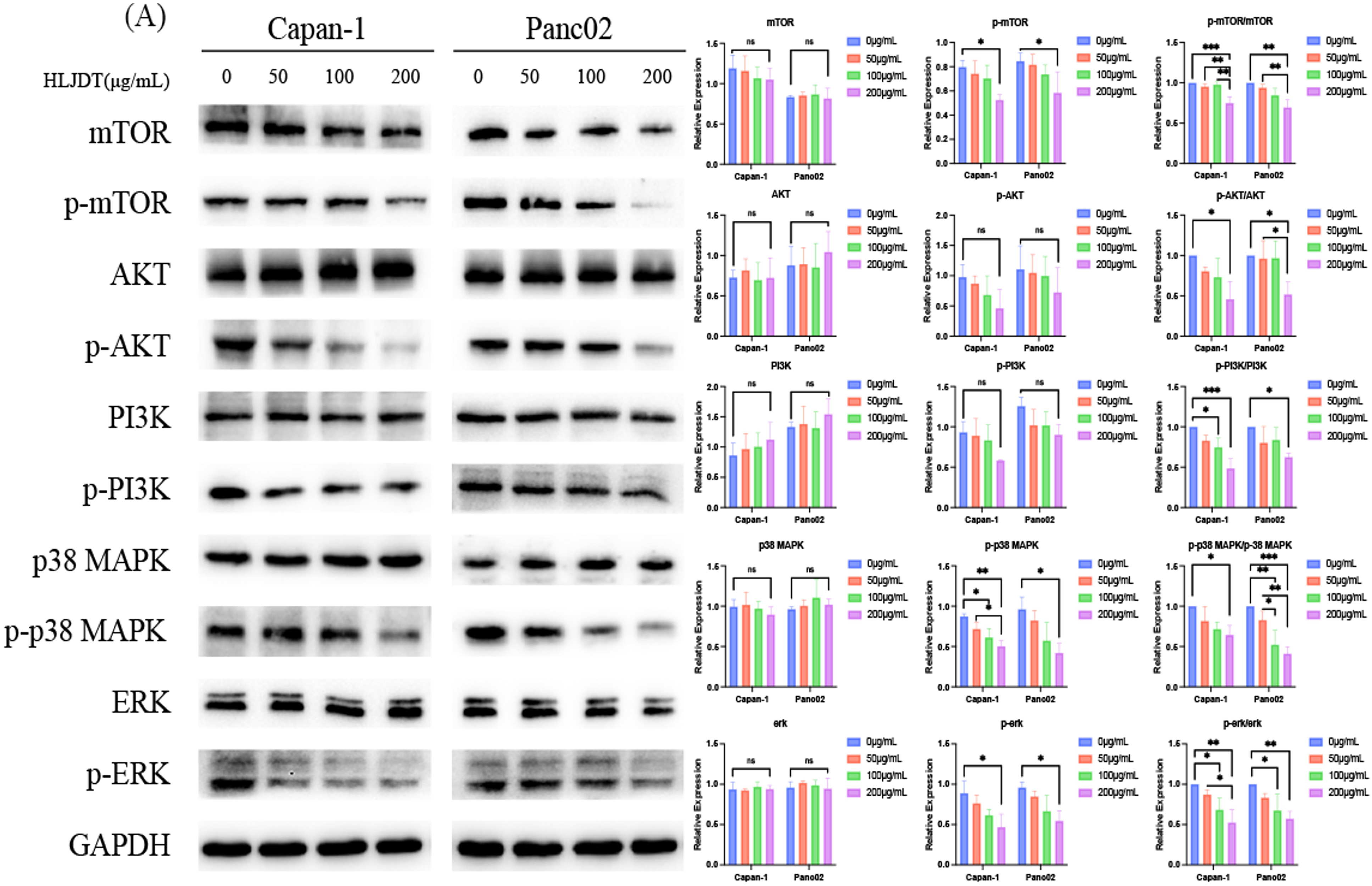
PI3K/AKT/mTOR and MAPK/ERK1/2 signaling pathways may be involved in the therapeutic effect of HLJDD on PAAD. Western blot analysis of protein levels of p-mTOR, mTOR, p-AKT, AKT, p-PI3K, PI3K, p38 MAPK, p-p38 MAPK, ERK1/2, p-ERK1/2 and GAPDH in Capan-1 and Panc02 cells. All data were expressed as mean ± SD (ns
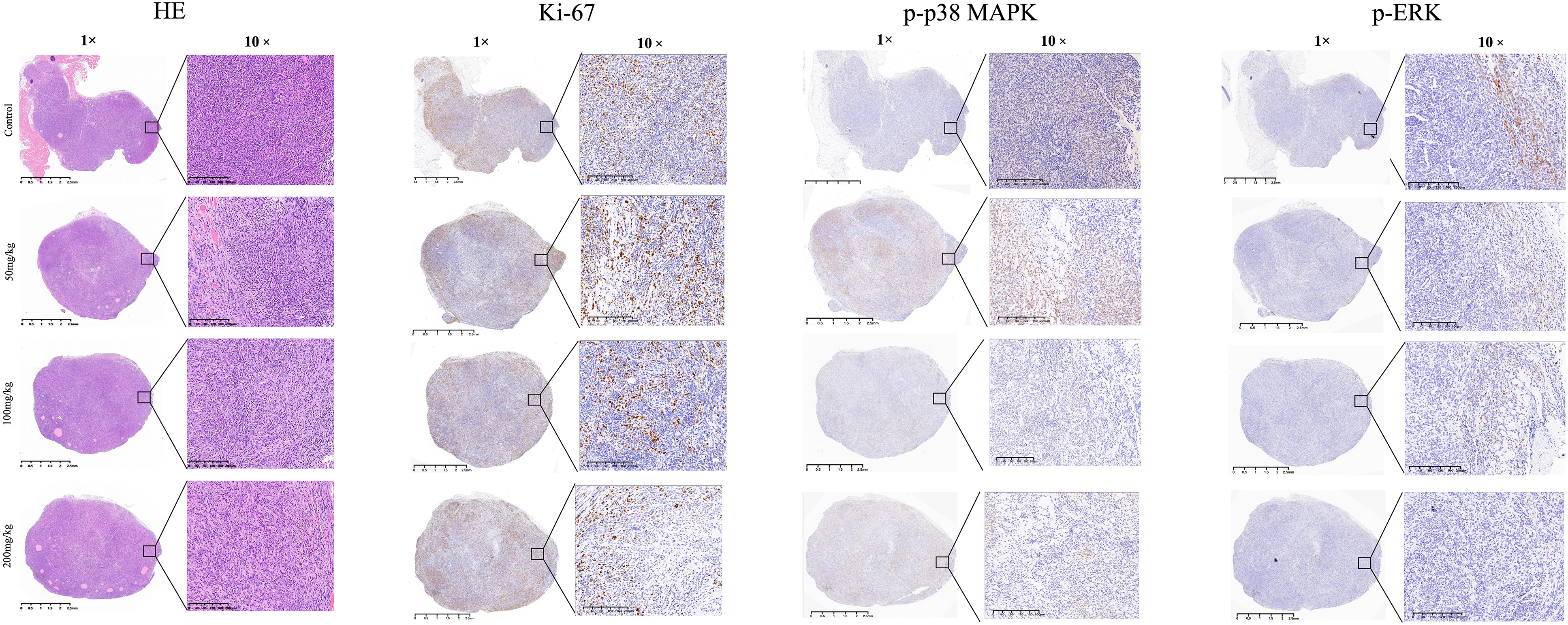
Histological and immunohistochemical analysis of tumor tissues from C57BL/6 mice with subcutaneous Panc02 xenografts treated with HLJDD. Hematoxylin and Eosin (HE) staining illustrates the tumor architecture at 1× and 10× magnification across control (vehicle), 50 mg/kg, 100 mg/kg, and 200 mg/kg HLJDD-treated groups. Ki-67 immunohistochemistry staining, shown at 1× and 10× magnification, indicates cell proliferation, with reduced staining intensity in HLJDD-treated groups, suggesting decreased proliferation. Phosphorylated p38 MAPK (p-p38 MAPK) and phosphorylated ERK (p-ERK) staining at 1× and 10× magnification show decreased staining intensity with increasing HLJDD doses, indicating inhibition of the MAPK and ERK signaling pathways. Scale bars represent 2.5 mm (1×) and 200 μm (10×).
The rescue experiment results reveal that HLJDD exerts inhibitory effects on the PI3 K/AKT/mTOR and MAPK signaling pathways in both Capan-1 and Panc02 cell lines. Treatment with HLJDD at 200 μg/mL significantly reduced the levels of phosphorylated PI3 K, AKT, and mTOR, while the total levels of these proteins remained unchanged, indicating a specific effect on phosphorylation. The addition of 740 Y-P, a PI3 K activator, partially reversed this inhibition, suggesting that HLJDD's impact on the PI3 K/AKT/mTOR pathway can be mitigated by pathway activation (Figure 8A). Similarly, HLJDD decreased the levels of phosphorylated p38-MAPK and ERK1/2 without altering the total protein levels. This inhibition was partially reversed by the addition of PMA, a PKC activator, indicating that HLJDD also targets the MAPK pathway (Figure 8B). GAPDH served as a loading control, confirming equal protein loading across samples. These findings collectively demonstrate that HLJDD inhibits critical oncogenic pathways, and the partial rescue by specific activators underscores its targeted action on the PI3 K/AKT/mTOR and MAPK signaling pathways, which are essential for cancer cell proliferation and survival.
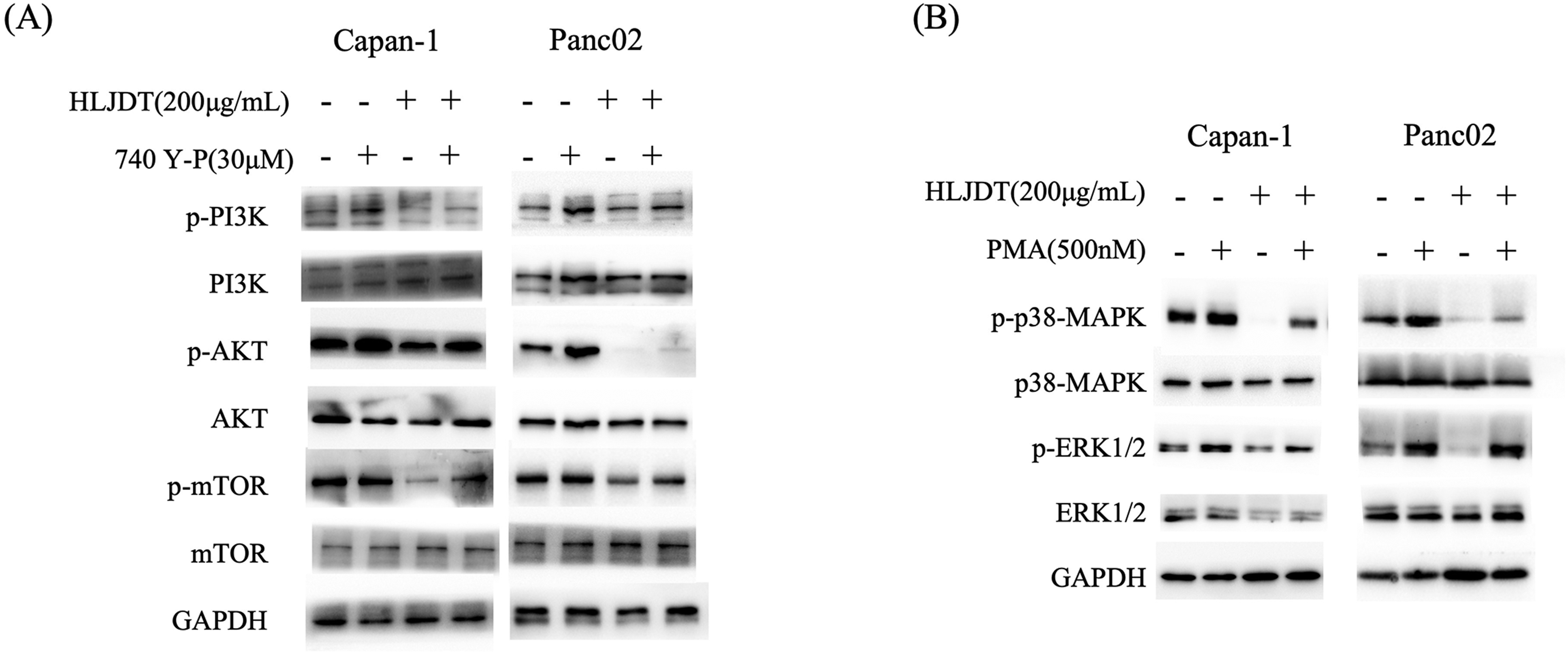
Rescue experiment demonstrating the effects of HLJDD on the PI3K/AKT/mTOR and MAPK signaling pathways in Capan-1 and Panc02 cells. (A) Immunoblot analysis of PI3K, p-PI3K, AKT, p-AKT, mTOR, and p-mTOR in Capan-1 and Panc02 cells treated with HLJDD (200 μg/mL) in the presence or absence of the PI3K activator 740 Y-P (30 μM). HLJDD treatment reduces the levels of phosphorylated PI3K, AKT, and mTOR, indicating inhibition of the PI3K/AKT/mTOR pathway. The addition of 740 Y-P partially reverses this inhibition, restoring phosphorylation levels. (B) Immunoblot analysis of p38 MAPK, p-p38 MAPK, ERK1/2, and p-ERK1/2 in Capan-1 and Panc02 cells treated with HLJDD (200 μg/mL) in the presence or absence of the PKC activator PMA (500 nM). HLJDD treatment reduces the levels of phosphorylated p38 MAPK and ERK1/2, indicating inhibition of the MAPK pathway. The addition of PMA partially reverses this inhibition, restoring phosphorylation levels. GAPDH serves as a loading control in all experiments.
Discussion
Chinese herbal formulas, comprising specific herbal ingredients with precise therapeutic effects, represent the essence of Traditional Chinese Medicine (TCM). Among them, Huanglian Jiedu decoction (HLJDD) stands out as a renowned TCM formula for its “heat-clearing and detoxicating” properties. This formula is meticulously designed to enhance efficacy while minimizing potential side effects, following the principles of TCM's “sovereign, minister, assistant, and courier” theory. In this formula,
Analyzing the network pharmacology of TCM in diverse diseases offers a systematic way to understand its therapeutic mechanisms. In our study, we employed network pharmacology analysis as the initial step to identify potential biologically active compounds and target genes associated with HLJDD. A total of 75 potentially bioactive compounds of HLJDD, meeting the criteria of “OB ≥ 30%” (indicating satisfactory pharmacokinetic absorption) and “DL ≥ 0.18” (suggesting favorable distribution, metabolism, and excretion properties), were extracted from the TCMSP database. Subsequently, 278 target genes related to these compounds were considered as plausible targets of HLJDD. Further KEGG pathway enrichment revealed that the top three pathways enriched by HLJDD were cancer, the AGE-RAGE, and the IL-17 signaling pathway. In terms of disease enrichment analysis, the top three enriched categories were immune-related diseases, pharmacological effects, and cancer. These findings align with previous studies indicating the anti-cancer effects of HLJDD. Previous network pharmacological analyses have demonstrated HLJDD's ability to inhibit cancer growth and progression by modulating the p53 signaling pathway, and studies have highlighted its potential anti-cancer effects in several cancers, 23 including hepatocellular carcinoma, 24 lung cancer, 25 myeloma, 26 and oral cancer. 4 In light of these considerations, we proceeded to investigate whether HLJDD exhibits anti-cancer effects in the context of PAAD, along with exploring the associated target genes and signaling pathways.
The transcriptome dataset GSE43795, comprising RNA expression data from 6 PAAD tumor tissues and 5 normal pancreatic tissues, was utilized for the identification of DEGs in PAAD. Applying the criteria of “| Log2FC | > 1” and “
To substantiate the anticancer effects of HLJDD in the context of PAAD, investigations were conducted
Numerous studies have been dedicated to the identification and characterization of the pharmacodynamically active constituents present in HLJDD. Alkaloids, such as those found in
Recent pharmacological studies have investigated HLJDD's antitumor activities and its bioactive components. Our previous research highlighted the suppressive effect of berberine hydrochloride on PAAD progression in both
To further investigate the impact of HLJDD treatment on the regulation of the PI3 K and MAPK-related signaling pathways, we conducted immunoblotting and immunohistochemical staining analyses. Our results indicated that HLJDD treatment led to the inhibition of the PI3 K/AKT/mTOR pathway by suppressing the protein- phosphorylation levels of PI3 K, AKT, and mTOR
The PI3K/AKT/mTOR signaling pathway is a well-established pathway involved in numerous cellular processes, including cellular growth, motility, migration, and metabolism. 37 In the context of PAAD, this pathway serves as a major downstream effector of KRAS, regulating kye characteristics of the disease. Several studies found that the aberrant activation of the PI3 K/AKT/mTOR pathway leads to enhanced cell proliferation and growth while suppressing cancer cell apoptosis. Consequently, there is growing interest in the development of inhibitors targeting this signaling pathway as potential therapeutic strategy for PAAD. To date, various small molecule inhibitors have been thoroughly assessed in preclinical and clinical settings. 38
In the realm of tumorigenesis and tumor progression, the MAPK/ERK1/2 signaling pathway hold significance and has been extensively studied. Recent clinical trials have found the potential of MAPK/ERK1/2 pathway inhibitors as effective anticancer agents. 39 The findings of our current study suggest that HLJDD may possess the capability to act as a dual inhibitor of both the PI3 K and MAPK-related signaling pathways. Subsequent investigations should explore the synergistic effects of combining HLJDD with chemoradiotherapy, immunotherapy, and targeted therapy. It is important to acknowledge certain limitations in our study. We were unable to conduct specific assays to elucidate the molecular mechanisms through which HLJDD inhibits the PI3 K and MAPK-related pathways. Furthermore, verification of targeted mRNA expressions and protein levels, as well as the evaluation of tissue-derived target mRNA and protein expression, requires further research.
Conclusion
In conclusion, HLJDD exhibited significant inhibitory effects on the growth and progression of pancreatic adenocarcinoma (PAAD) both
Supplemental Material
sj-docx-1-chp-10.1177_2515690X241291381 - Supplemental material for “Huanglianjiedu Decoction” Against Pancreatic Adenocarcinoma Proliferation of by Downregulating the PI3K/AKT/mTOR and MAPK/ERK1/2 Signaling Pathways
Supplemental material, sj-docx-1-chp-10.1177_2515690X241291381 for “Huanglianjiedu Decoction” Against Pancreatic Adenocarcinoma Proliferation of by Downregulating the PI3K/AKT/mTOR and MAPK/ERK1/2 Signaling Pathways by Shu Dong, MD, Panling Xu, MD, Peiwen Yang, MD, Juying Jiao, MD, Chien-shan Cheng, MD, PhD, Lianyu Chen, MD and PhD in Journal of Evidence-Based Integrative Medicine
Supplemental Material
sj-docx-2-chp-10.1177_2515690X241291381 - Supplemental material for “Huanglianjiedu Decoction” Against Pancreatic Adenocarcinoma Proliferation of by Downregulating the PI3K/AKT/mTOR and MAPK/ERK1/2 Signaling Pathways
Supplemental material, sj-docx-2-chp-10.1177_2515690X241291381 for “Huanglianjiedu Decoction” Against Pancreatic Adenocarcinoma Proliferation of by Downregulating the PI3K/AKT/mTOR and MAPK/ERK1/2 Signaling Pathways by Shu Dong, MD, Panling Xu, MD, Peiwen Yang, MD, Juying Jiao, MD, Chien-shan Cheng, MD, PhD, Lianyu Chen, MD and PhD in Journal of Evidence-Based Integrative Medicine
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank XuRan Biotech Co.Ltd (Shanghai,China) for providing RNA sequencing help; and Dr Zhen Chen from the Department of Integrative Oncology, Fudan University Shanghai Cancer Center for providing project consulting help.
Authors’ Contribution
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was partly supported by the National Natural Science Foundation of China [grant numbers 82204850,81973629]; the Shanghai Technology Development Funds [grant number 22YF1425800, 21ZR1414100].
Ethics Approval
This study protocol had been reviewed by the animal and ethics committee of Fudan University Shanghai Cancer Center (FUSCC-IACUC-S2022-0444/ 050432-4-2108*/1612167-18)
Data Availability
Data will be made available on request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
