Abstract
The antibiofilm and antimicrobial properties of tropical honey types including Malaysian stingless bee honey remain explicitly unexplored when compared with Apies honey. The antibiofilm and antimicrobial activities of the Malaysian Trigona honey were characterized with two stinging bee honey types (Centaurea hyalolepis and Citrus honeys) from Jordan. The antibiofilm and antimicrobial investigations were conducted on a set of seven microbial strains; five bacterial species of Pseudomonas aeruginosa ATCC 10145, Streptococcus pyogenes ATCC 19615, Staphylococcus aureus ATCC 25923, Escherichia coli ATCC 25922, Klebsiella pneumoniae ATCC 13883, and two fungal strains Candida albicans ATCC 10231 and Candida krusei ATCC 14243. The antimicrobial investigations revealed a broad spectrum activity for Trigona honey against Gram-positive, Gram-negative, and fungal strains over the two honey types. One-way ANOVA showed a significant difference (p < 0.001) in the zone of inhibition ranging from 9 to 25 mm and minimum inhibition activity (MIC) ranged from 9.4–29.6% (w/v) against the microbial strains. Moreover, the addition of honey to established biofilms has induced a degradation activity in the biofilm mass. Two-way ANOVA showed a significant biofilm degradation proportion (p < 0.001) ranging from 1.3% to 91.3% following treatment with Trigona honey and the other honey types in relevance to the concentration ranging from 10% to 50% (w/v). Moreover, the antibiofilm activity was highly consistent with MIC affecting bacterial growth inhibition. In conclusion, a robust antimicrobial and antibiofilm activity for Trigona stingless bee honey over the stinging bee Centaurea hyalolepis and Citrus honeys is noticed which endows the usage of Trigona honey in the antimicrobial industry.
Keywords
Introduction
Honey is a natural syrup with distinctive medical and nutritional characteristics. 1 Substantially, the honey-producing bees belong to the Apidae family including honey bees (Apis genus) and another type of bees known as stingless bees (Melipona and Trigona genera). Stingless bees are found in tropical and subtropical regions including Asia, North Australia, Africa, Central, and South America. 2 Commercial stingless bee honey also known as kelulut honey is produced by bees of Trigona species and is abundantly found in Malaysia. Kelulut honey is multi-floral honey that has been reported as medically and therapeutically useful syrup with potential antibacterial properties. 3 Stingless bee honey shares common characteristics with Apis bee honey with some variations. For instance, stingless bee honey shows a distinct taste is more aromatic, and has lower crystallization behavior. 4 Additionally, stingless bee honey contains several physiochemical characteristics that endorse an extraordinary antimicrobial activity against many different strains of bacteria.5,6 Trigona stingless bee honey is utilized locally in Malaysia for several purposes, such as an anti-aging agent, anticancer, treating respiratory illnesses, and relieving sore throat, cough, and cold. 7
The tropical forests contain an unexplored rich range of endemic flora in which bees compile pollens and nectar. Bees process the nectar and transform it into honey. The honey is classified into mono-floral or multi-floral honey in relevance to the source of nectar being used in bee feeding. A wide range of clinical applications has been described for the usage of honey as an alternative remedy for the treatment of microbial infections in skin ulcers, infected wounds, burns, and post-burns.8–11 Trigona honey is a multiflora honey produced by stingless bees reared on tropical flowers.
Among natural honey's main features is its capability to curb bacterial and fungal growth. The antimicrobial activity of honey is attributed to several factors among them, low pH, hydrogen peroxide, and bee defensin-1 secreted by the bee. 12 Several studies have revealed that the concentration of individual honey constituents is substantially very low to exert antimicrobial activity by themselves. Meanwhile, the therapeutic characteristic of honey is attributed to the synergistic effect of combining different ingredients of the phytochemical compounds.13–16
Nowadays, more highlight is pointed to the usage of honey as a medicine, and a relatively new era of alternative medicine called “Apitherapy” has emerged, focusing on medical applications of honey and other bee products such as propolis, bee pollen, royal jelly, and bee wax.17,18
The chemical investigations on the physicochemical properties of honey showed an absence of toxic metabolites in the constituents of freshly harvested honey. Moreover, biological lab screenings clarified a high margin of safety for the usage of honey on vital organs such as the liver, spleen, and kidneys. 19 To date, there are no reports either about bacterial strain resistance acquired against any type of honey or any toxicity or side effects of honey on humans. Also, the low cost of honey maintenance shared with the availability confers valuable advantages to using honey as an alternative antimicrobial therapy. 20
Biofilms can be explicitly described as communities of microorganisms that are attached to a surface. They encompass one single kind of microorganism or multiple microorganisms that are attached to a surface. 21 Biofilms are dominant in many environments and establish a variety of infections on the surfaces of medical devices and implants.22,23 Several research groups have studied the antibacterial and antibiofilm activities of honey against pathogenic microorganisms such as nosocomial bacteria as well as food spoilage bacteria.24,25
Medical devices serve as potential surfaces for biofilm formation, resulting in chronic and difficult-to-treat infections.26,27 For microbial biofilms, there is no generally agreed mechanism for resistance, for instance, some researchers argue that the higher resistance of antibiotics is attributed to the matrix formation and not to the bacterial cellular characteristics. 28 The Centers for Disease Control and Prevention (CDC) estimates that biofilm accounts for two-thirds of the bacterial infections that physicians encounter particularly in immunocompromised patients. 29
For instance, clinically relevant incidences include endocarditis, osteomyelitis, wound infection, otitis media, chronic pulmonary infection in cystic fibrosis patients, urinary tract infections, gingivitis, burn-related infections, and prostatitis are cases of human diseases caused by bacterial biofilm-associated infections.30–32
The majority of the research conducted on honey is focused mainly on Apis honey not taking into consideration the stingless bee honey due to the lack of knowledge and sources available for many scientists. For instance, the geographical abundance of stingless bees in tropical regions, whereas the Apis bees are predominantly distributed around the world.33,34 Malaysia is located geographically in the tropical region where stingless bees and their products are abundantly available.
Additionally, these honey types have been subjected to scientific research by different scientific groups to determine the valuable contents of the honey types and to determine the differences between Apis honey and stingless bee honey. 35
This study aims to insights into the versatility in medical activities of Trigona honey as an antibacterial and antifungal agent. We have explored the antimicrobial and biofilm degradation activities on seven microbial strains including Gram-positive, Gram-negative, and fungal strains. Moreover, to elucidate the therapeutic properties of stingless bee honey (Trigona, Malaysia) over other honey types we conducted a comparison between Trigona honey and two stinging bee honey types from another different geographical region (Jordan).
Material and Methods
Honey Samples and Preparations of Honey Solutions
Malaysian stingless Trigona honey and two stinging bees- Jordanian honey types (Centaurea hyalolepis, and Citrus honey) were used as reference honey types in the experiments of antimicrobial and antibiofilm activities. The three honey types were collected from March to May 2022.
Trigona honey is natural honey produced by stingless black bees reared in tropical regions in a virgin rainforest in Kelantan state on the East Coast of Peninsula, Malaysia. A commercial Trigona honey sample was purchased from Bee Haven Center, Kelantan, Malaysia.
Centaurea hyalolepis honey is a natural non-processed honey produced by Apis bees reared on wild Centaurea hyalolepis plants in the mountains of Jerash city, north of the Hashemite Kingdom of Jordan (HKJ) that are naturally grown in spring and early summer.
Citrus honey is a naturally produced honey by Apis bees reared in Citrus farms on the flowers of lime, lemons, and oranges in the region of Jerash city, north of HKJ.
Honey samples were stored in well-closed amber glass bottles, labeled accordingly, and placed in a dark place at 25 °C for further investigations. Honey samples have been freshly prepared for each experiment. For antibacterial activity studies, solutions of honey were prepared weight per volume (w/v) in saline 0.85%, then sterilized by passing through a non-sterile 0.7 μm glass fiber filter to remove larger particles. Honey solutions were prepared by starting with an original stock of 50% honey (w/v) in distilled water and serial dilutions were conducted to reach concentrations of 40%, 30%, 20%, and 10%. Honey preparations used in antibacterial activity assays were freshly prepared within 1 h of conducting experiments.36,37
Viable Bacterial Count
To start with a relevant microbial number in the antimicrobial and antibiofilm experiments; the Colony forming unit (CFU/ml) assay for seven reference bacterial strains was performed. Five bacterial strains of Pseudomonas aeruginosa ATCC 10145, Streptococcus pyogenes ATCC 19615, Staphylococcus aureus ATCC 25923, Escherichia coli ATCC 25922, and Klebsiella pneumoniae ATCC 13883, two fungal strains Candida albicans ATCC 10231 and Candida krusei ATCC 14243 were used in the microbial experiments. The procedure was conducted according to a previously published method. 20 Three morphologically identical colonies from each bacterial or fungal strain were aseptically picked up and grown aerobically in liquid broth in a shaker incubator (150 rpm) for 24 h. The bacterial strains were cultivated in 20 ml of Nutrient Broth (NB) (Sigma-Aldrich, Belgium) at 37 °C whereas fungal strains were cultivated in Sabouraud Dextrose Broth (SBD) at 25 °C. 38 For each microbial strain, five suspensions of bacteria or a fungus were prepared in 0.85% saline in relevance to Optical Density 600 nm (OD) absorbance of five McFarland standard concentrations of 0.5, 1, 2, 3, and 4 individually. To start with the same microbial burden for each strain each McFarland preparation was diluted 3 times in a ratio of 1:100 using 0.85% saline to have an accountable colony number. The adjusted bacterial suspensions were inoculated on 5 mm Nutrient Agar (NA) plates at 37 °C for 24 h and fungal strains were incubated on 5 mm Sabouraud Dextrose Agar (SDA) plates at 25 °C for 24 h. The CFU number was counted and calculated by the following formula: CFU/ml = (no. of colonies x dilution factor) / volume of the culture plate. Later on, the concentration of 1.5 × 108 CFU/ml of each strain was adopted for antimicrobial tests.
Antimicrobial Activity of the Honey Types
The standard protocols of the Kirby-Bauer Disk Diffusion Susceptibility Test were applied with modifications.39,40 The antimicrobial activity of the three types was tested against the same set of microbial strains mentioned in the viable bacterial count experiment.
Bacterial strains were grown at 37°C for 24 h in Muller Hinton Broth (MHB). Muller Hinton Agar (MHA) was used for conducting the agar diffusion test. Fungal strains were cultivated in SDB at 25°C for 24 h, whereas, the agar diffusion test was conducted on SDA. The microbial concentration was adjusted to a concentration of 108 CFU/ml followed by a dilution to 106 CFU/ml using 0.85% normal saline. 20 A sterile swab was used for streaking bacteria on the MHA, whereas, fungal strains were inoculated on SDA. Pores of 8 mm were bunched on the agar to allow proper filling of 100 µl of the 100% (w/v) honey samples. As a control for the test, a 5 µg disk of reference drug ciprofloxacin was used as a positive control for the antimicrobial activity of bacterial plates whereas a 25 µg disk of fluconazole was used as an antifungal reference drug and eventually water was used as a negative control. Bacterial plates were incubated at 37 °C, and the growth of the microbial cells was evaluated after 22 ± 2 h by measuring the zone of inhibition around the wells using a caliper. Whereas, fungal strains were cultured at 25°C for 22 ± 2 h and the diameter of the inhibition zone was measured. The test was performed in triplicate and the average value for each microbial strain was considered.
Determination of minimum Inhibitory Concentration of Honey Against the Microbial Strains
The Minimum Inhibitory Concentration (MIC) was conducted using the broth microdilution method with modifications reported by.41,42
Overnight culture of the three bacterial strains was cultivated at 37°C in NB whereas the two fungal strains were cultivated in SDB at 25 °C for 24 h. The microbial inoculum was adjusted using a spectrophotometer at OD600 to a concentration of around 1.5 × 108 CFU in 0.85% saline. The adjusted bacterial suspensions were dispensed in 1 in 40 in 4X NB whereas fungal strains were diluted in the same manner in SDB to achieve a final concentration of 1X of broth in the 96 well microtiter plates. Five concentrations of the three honey types of 10%, 20%, 30%, 40%, and 50% (w/v) were prepared using sterile water. Using a flat bottomed 96 well microplates 150 µl of the relevant honey preparation was pipetted into wells. 50 µl of the 4x nutrient broth and the bacterial suspension was pipetted in the 96 well plates whereas 50 µl of 4x SDB and fungal preparation was pipetted in the relevant microbial row to achieve a final concentration of 5 × 105 CFU/ml for the microbial strains with a final volume of 200 µl.
Plates were incubated at 37 °C for bacterial strains and 25 °C for fungal strains and the growth was evaluated after 22 ± 2 h by measuring the OD absorbance at a wavelength of 570 nm measured using an Elisa plate reader (Tecan Infinite 200 PRO, Austria). 43
Biofilm Formation and Degradation for Microbial Strains
Biofilm formation assay was performed using the crystal violet method in 96-well microtiter plates. The biofilm formation experiment was conducted based on a previously published method.15,44 The biofilm formation experiment was conducted on the seven microbial strains mentioned in the viable bacterial count experiment. The bacterial strains were grown separately in a TSB medium at 37 °C for 24 h with constant shaking at 150 rpm whereas fungal strains were cultivated in SDB at 25 °C overnight. The CFU of the microbial strains was adjusted to 1 × 105 cells; bacterial strain concentration was adjusted using TSB and the fungal strains were adjusted using DSB. Both broth media were supplemented with 1% (w/v) glucose to enhance biofilm formation. For each type of bacteria, 200 μL of the suspension was pipetted in each well in 96-well microtiter plates, TSB supplemented with 1% glucose without microbial strains was used as a negative control for biofilm formation study, and then the plates were incubated at 37 °C for 24 h. After incubation, the microbial cells were discarded by inverting the plates upside down and washing them with distilled water to remove any excess unattached cells and allowed to be air-dried. After that, 200 μL of 0.1% crystal violet (CV) solution was added to each well of the microtiter plate and incubated at room temperature for 15 min. Following the incubation, the plates were rinsed with phosphate-buffered saline (PBS) (Invitrogen, UK) three times to remove the unabsorbed stain, and then the plates were tapped gently on a towel paper. Finally, 200 μL of 95% ethanol was added to each well to solubilize the stained biofilms and the biofilm formation was estimated by measuring the absorbance of the CV at 570 nm using an Elisa plate reader (Tecan Infinite 200 PRO, Austria).
To determine the biofilm degradation activity for the three honey types against the established biofilms of the seven microbial strains, the bacterial suspensions above the established biofilms have aspired to remove the planktonic bacteria, discarded from each well, and displaced by 200 µl of TSB for bacterial strains and SDB for fungal strains supplemented with 1% (w/v) glucose-containing in each plate kind of honey (Trigona, Hyalolepis, and Citrus) at different concentrations ranging from 10%—50% (w/v). The plates were incubated at 37°C for a further two hours to study the biofilm degradation activity of different honey types. Wells containing microbial suspensions with a honey concentration of 100% (w/v), TSB, or SDB supplemented with 1% glucose were used as a positive control for biofilm degradation activity. Whereas, wells containing only microbial suspensions without any honey kind, TSB or SDB supplemented with 1% glucose were used as a negative control. Following the incubation, planktonic cells were removed carefully without scratching the bottom and walls of the wells and the wells were washed with distilled water. The plates were inverted upside down to be air-dried for 15 min at room temperature before being stained with 200 µl of 0.1% (w/v) CV solution for 15 min at room temperature. Later on, the biofilms were washed three times with PBS to remove the unabsorbed stain. Eventually, 200 μL of 95% ethanol was added to each well to solubilize the stained biofilms and biofilm degradation was estimated by measuring the absorbance of the CV at 570 nm using an Elisa plate reader (Tecan Infinite 200 PRO, Austria). The experiment was performed in triplicates, and the average value was considered.
The classification of the biofilm formation for each bacterial strain was measured by the following formulas45,46: Non-adherent (NA = OD ≤ ODC), Weak adherent (WA = ODC < OD ≤ (2×ODC)), Moderate adherent (MA = (2×ODC) < OD ≤ (4×ODC)) and strong adherent (SA = (4×ODC) < OD).
Biofilm degradation%= [(OD growth control - OD sample) / OD growth control] x100
Statistical Analysis
All samples were repeated three times separately and the mean value was expressed. CFU of the bacterial strains, MICs, and zone of inhibition for the three honey types were analyzed using one-way ANOVA analysis. Biofilm formation of different bacterial strains and biofilm degradation activity of different honey types in relevance to the honey concentrations were analyzed by two-way ANOVA followed by Turkey's post hoc analysis using the Statistical Package for the Social Sciences (SPSS) program, version 20 (IBM, Chicago, IL, USA). Values with P < 0.05 are regarded as statistically significant.
Results
Viable Bacterial Count for P. aeruginosa and S. pyogenes
The CFU-counting assay was performed for seven microbial strains to start with a relevant microbial number in the anti-microbial and anti-biofilm experiments. The CFU number for each microbial strain was significantly different (p < 0.001) than the number of other strains when we used only the McFarland standard in the preparation of bacterial suspension as the lowest log CFU 6.12 (1.43 × 106) was demonstrated in C. albicans whereas the highest log CFU 8.79 (6.29 × 108) was in E. coli when the reference 0.5 McFarland standard used as a reference for suspension preparation (Supplement 1). There was a significant variation between the anticipated microbial count and the actual viable microbial count in different strains when we compared the seven microbial strains at McFarland standards 0.5, 1, 2, 3, and 4 (p < 0.001) (Supplement 1). Therefore a viable CFU of 1.5 × 108 was prepared for the reference strains and the relevant absorption for each strain was adopted for further investigations.
Antimicrobial Activity for the Honey Types
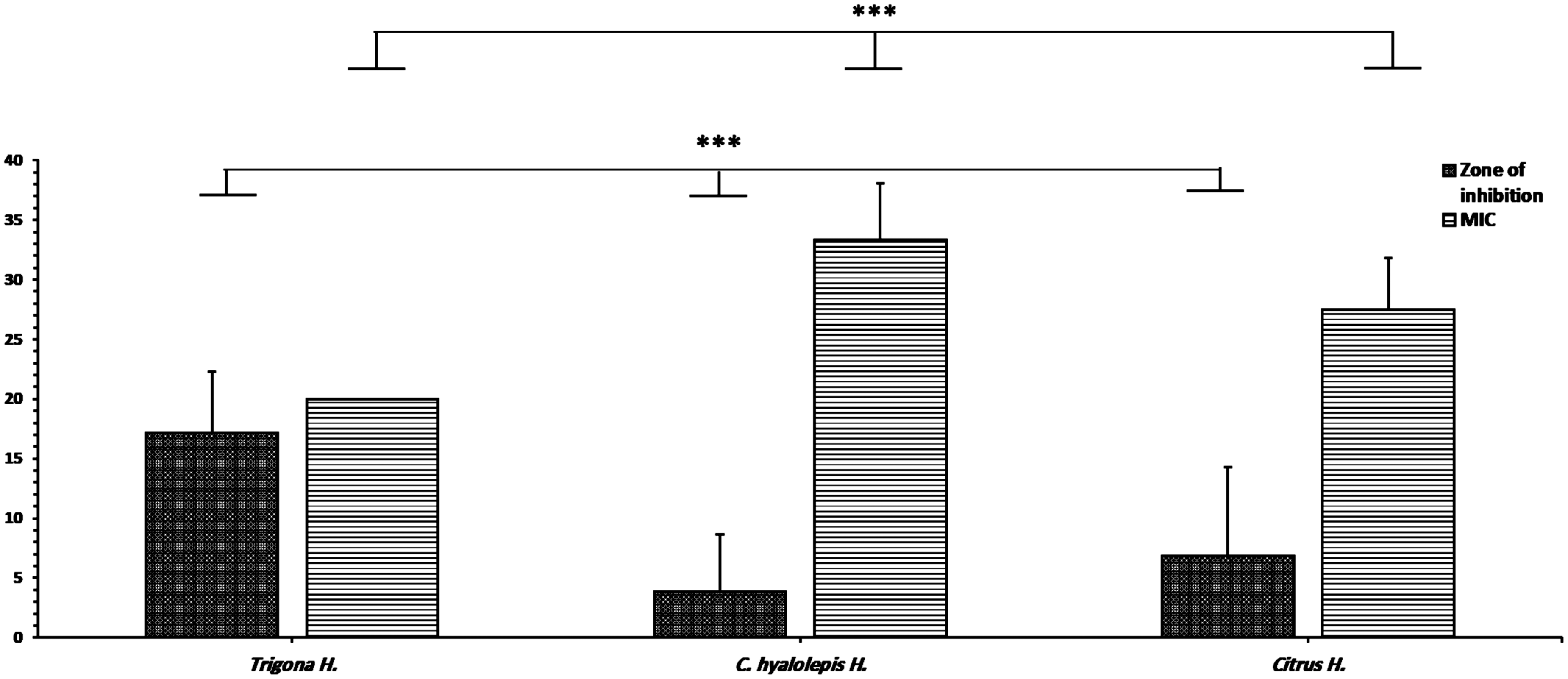
Honeys were screened for antimicrobial activity against a set of bacterial and fungal strains using a well diffusion assay and MICs. The three honey types showed antimicrobial activity with a significant variation (p < 0.001) in both zone of inhibition and MIC activities. Trigona honey exerted the highest anti-microbial activity against different microbial strains over the other two honey types. The mean zone of inhibition for Trigona honey was 17.14 ± 5.1 mm, whereas, C. hyalolepis was 3.8 ± 4.8 mm and Citrus 6.8 ± 7.5 mm (Figure 1, Table 1). E. coli showed the highest susceptibility to Trigona honey with a zone of inhibition of 25 mm whereas C. krusei showed the least susceptibility at 13 mm (Table 1). The MIC activity of the three honey types is summarized in Table 1. Trigona honey showed the lowest MIC mean value of 20% followed by Citrus honey at 27.5% and eventually, C. hyalolepis with MICs of 33.3%.

Comparison of the antimicrobial activity between the three honey types. Zone of inhibition was measured in mm whereas MIC was calculated in percentage. Asterisks indicate a significant difference p < 0.001in the zone of inhibition and mean MIC activities between the three honey groups after conducting a one-way ANOVA analysis.
Zone of Inhibition and MICs for the Three Honey Types Against Reference Strains.

H = honey, mm = zone of inhibition in mm; the zone of inhibition was expressed as mean ± standard deviation, MICs = minimum inhibition concentration, ND = not detected, NA = not applicable. Significant difference p < 0.001 in the zone of inhibition and mean MIC activities between the three honey types after conducting a one-way ANOVA analysis.
Biofilm Formation and Degradation for the Microbial Strains
Bacterial growth analysis of the microbial strains showed a high growth rate and higher turbidity in the biofilm formation assay compared to the negative control wells. All microbial strains have developed biofilms, the OD570 of the biofilms formed by seven strains were four times higher than the OD570 of the negative control, so according to the interpretation guidelines, biofilms formed by microbial strains are classified as strong adherent biofilms (data not shown).
The addition of the three honey types individually for two hours to the established microbial biofilms has amended the biofilm formation and confronted biofilm degradation compared to the negative control of the biofilm degradation experiment (Figure 2). Two-way ANOVA analysis has revealed a significant biofilm degradation activity (p < 0.001) for the three honey types in relevance to the different honey concentrations used in the biofilm degradation experiment. The statistical analysis of the biofilm degradation activity was conducted using different honey concentrations in each honey type and in comparison to the negative control of biofilm degradation wells without honey treatment (Supplement 2). Trigona honey showed the highest biofilm degradation activity against all reference strains with a mean biofilm degradation percentage of 90.3% against bacterial strains and 81.3% for the fungal strains (Figure 2 A). Both honey types of C. hyalolepis and Citrus showed a lower antibiofilm activity than Trigona honey as the mean biofilm degradation activity for Citrus honey was 65.47% for the bacterial strain and 52.3% for the fungal strains (Figure 2 B, C). Meanwhile, the least antibiofilm activity was noticed for C. hyalolepis honey as the highest antibiofilm activity for bacterial strains was 56.47% whereas the highest antibiofilm activity for fungal strains was 47.30% (Figure 2B).
Biofilm degradation percentage after the treatment with different honey types and concentrations against a set of five bacterial strains and two fungal strains. Degradation activity A. Trigona stingless bee honey B. Centaurea hyalolepis stinging bee honey C. Citrus sting honey. Positive control: biofilm of each microbial strain treated with 100% (w/v) honey. Negative control: honey untreated established biofilm. Biofilm degradation data were analysed using Two-way ANOVA for the degradation activity of each honey type, concentration, and microbial strain in comparison with the negative control followed by the Tukey post hoc test. All honey-treated biofilms were compared with the negative control of the untreated biofilms. *** P < 0.001.
The highest biofilm degradation activity was noticed in the concentration of 50% (w/v) honey in the different honey types. The concentration of 20% (w/v) of Trigona honey was the lowest concentration capable of conferring a significant biofilm degradation activity in the three honey strains against the different bacterial and fungal strains (Figure 2). A sequential significant biofilm degradation activity, and there was decomposition in the biofilm mass in relevance to the honey concentration used in the treatment.
Discussion
In the current study, Trigona honey demonstrated a substantial deleterious effect against the reference microbial strains of bacteria and fungi over the other two honey kinds; C. hyalolepis and Citrus honeys. The antimicrobial activity was not exerted on a specific microbial strain on the contrary a broad spectrum activity was noticed against all microbial strains when samples were treated with Trigona honey. Among the seven reference strains, Gram-negative bacteria were the highest vulnerable strains to different honey types. On the contrary, fungal strains explicitly had the lowest antimicrobial susceptibility toward the three honey types. Whereas, Gram-positive strains showed a similar biofilm degradation activity compared with Gram-negative strains when treated with Trigona honey whereas the degradation activity was significantly lower when biofilms treated with C. hyalolepis or Citrus honey types.
The antimicrobial activity of honey is attributed to its physicochemical properties, as honey is a hygroscopic syrup that causes dehydration of the bacterium by decreasing the moisture of the environment around it. Additionally, the high sugar content and low pH level are unfavorable conditions for bacterial growth. 47 In addition to the physicochemical properties of the honey, researchers clarified that the antibacterial properties of honey are governed by the content of polyphenolic compounds, the presence of hydrogen peroxide, and antioxidant properties. 48 Kwakman et al reported a robust antimicrobial effect of Revamil source honey which is a peroxide-producing honey over Manuka honey a non-peroxide-producing honey against a set of pathogenic bacteria of Bacillus subtilis methicillin-resistant Staphylococcus aureus (MRSA), E. coli and P. aeruginosa. 49 Furthermore, the antibacterial activity of honey is highly dependent on the sources of the nectar, and geographical factors like temperature, type of soil, flower origin, and humidity.50,51 The phytochemical analysis conducted by Seder et al revealed a higher concentration of polyphenolic compounds in Trigona honey 42 X than the other Stinging bee honey types. Additionally, low pH value and low moisture content are distinctive characteristics of Trigona honey than other honey types which can donate an augmented antimicrobial activity. 37
In the current study, several concentrations of Trigona honey ranging from 50% to 10% (w/v) were challenged against a wide range of Gram-positive, Gram-negative, and fungal strains. Nonetheless, Trigona honey showed the lowest MIC value among the three honey types as the concentration of 20% was sufficient to cause an inhibitory effect on the whole microbial strains. More precisely, Trigona honey was the only honey that inhibited the growth of S. aureus, K. pneumoniae, and C. krusei \whereas, C. hyalolepis and Citrus honey didn’t show antimicrobial activity against the mentioned strains. These results are congruent with Zainol et al who reported an MIC value of 20% (w/v) for Trigona honey against Staphylococcus aureus, Bacillus cereus, Escherichia coli, and Pseudomonas aeruginosa.. 35 Nweze reported MIC values between 12.5% to 25% (w/v) and 6.3% to 25% (w/v), against isolates of multidrug-resistant E. coli and P. aeruginosa when he used Trigona honey and Melipona honey, respectively. 52 On the other hand, the other types of Apis honey showed a weaker antibacterial activity when compared with Trigona honey. Bhushanam reported that the MIC of Apis honey against a set of pathogenic isolates was 25% (w/v) 53 likewise, E. Brown, showed a lower MIC for stingless bee honey 16% (w/v) against four common pathological strains S. aureus, E. coli, S. pyogenes, and Haemophilus influenza compared with the MIC of Apis honey which was 32% (w/v) against same strains. 54 Additionally, several scientists reported antifungal activity for Trigona honey against different fungal species such as C. albicans, C. glabrata, C. krusei, and Saccharomyces crevasses and MICs ranged from 10% to 25% (w/v).55,56 Fausto et al have reported recently an exploratory study about the antibiofilm activity of 35 different types of honey collected from different provinces in Ecuador, they showed that stingless-bee honey (Melipona) is one of the best five honey types exerted a robust antibiofilm activity against a set of Staphylococcus aureus, Klebsiella pneumoniae, Candida albicans, and Candida tropicalis. They reported a biofilm degradation activity of 15% (v/v) against the microbial set with a variation in the degradation proportion attributed to cellular changes in size and shape. 57 These findings suggested that the variations in the antimicrobial activity of the kinds of honey could be attributed to the bee species and type of multiflora that the bees were feeding on in the home country, as well as, other factors like the handling procedure, the storage duration between the sampling and conducting the experiments which can exaggerate or render the activity. 58
In this study, the concentration of 20% (w/v) Trigona honey successfully degraded the biofilms of the whole microbial strains of bacteria and fungi, whereas, the biofilm degradation activity was sequential with the increase in honey concentration. Trigona honey affects the cell wall consistency through massive cell destruction and lysis in Gram-positive and Gram-negative bacteria,. 46 Consistently, a study was conducted using honey that has the same geographical origin as Malaysia, Manuka honey at a concentration of 20% (w/v) affected the structure and viability of both Gram-positive and negative strains by distorting the cells.59,60 Meanwhile, another study conducted on the antimicrobial activity of Manuka honey showed that a concentration of 10% (w/v) honey affected the structure of S. aureus. 61 Studies by62,63 showed that stingless bee honey and sider honey have disrupted the cell wall and inhibited cell division of P. aeruginosa, S. pyogenes, and S. aureus. Additionally, Nishio et al reported an MIC inhibition of 0.62–10% for a stingless bee honey called Scaptotrigona bipunctata Lepeletier honey. 62
According to several researchers, the antibacterial and biofilm degradation activities of stingless bee honey could be attributed to major biological characteristics of honey as the low acidity, non-hydrogen peroxide effect, and the presence of natural phytochemical components.64–68 For instance, a study conducted on Malaysian Meliponini honey; a non-hydrogen peroxide honey, showed antibacterial activity against S. aureus. 65 The combining of these factors is beneficial in controlling bacterial colonization and additionally disruption of biofilms. The acidity of Trigona honey, which is within the range of pH 3.2 to 4.5, creates an unfavorable environment for bacterial growth, whereas, their optimum growth pH is about pH 7.2 to 7.4. 48 The high osmotic effect of honey due to the high content of sugar also plays a role in reducing biofilm mass. 48 likewise, Abbas et al, reported a mean biofilm degradation activity of 92.3% for stingless bee honey against P. aeruginosa, S. aureus, P. mirabilis, and Klebsiella spp. compared to 75.6% for endemic Egyptian honey. 69 Additionally, Kafaween et al demonstrated antibiofilm activity for Trigona honey at a concentration of 20% (w/v) against S. pneumoniae. 70 Sindi et al reported MICs of 6.7–28% (w/v) for Manuka honey against a set of Gram-positive and negative bacteria. They reported a reduction in the biofilm formation attributed to an inhibition in the quorum sensing pathway. In previous work for our group, a comprehensive analysis of microarray in P. aeruginosa biofilms after the treatment with Trigona honey has revealed a reduction of a key biofilm regulator c-di-GMP production. 38
In this study, the conditions for biofilm formation were optimized to achieve a representative biofilm mass, the findings were similar to several other research groups’ results.71,72 The modifications in the initial bacterial inoculum, the type of culture media, and the incubation temperature have affected drastically the biofilm mass in the current experiment. Concomitantly, several studies point out that environmental signals are an essential driving force for microbial development and environmental cues play a vital role in biofilm development.73,74 Kafaween et al showed a strong biofilm formation for both P. aeruginosa and S. pyogenes in similar conditions to our experiment. 75 Several factors affect the biofilm mass like presence and absence of nutrients, temperature, surface type, and quorum sensing. 73
Conclusion
Malaysian Trigona stingless bee honey has shown antibiofilm activity and hampered biofilm formation in both bacterial and fungal strains in a dose-dependent manner. Moreover, there was a robust antimicrobial activity of Trigona honey against the pathogenic microorganisms which endow for an implementation of Trigona honey usage as an additive in the medical devices industry as part of development.
Supplemental Material
sj-docx-1-chp-10.1177_2515690X241271978 - Supplemental material for Characterization of Antibiofilm and Antimicrobial Effects of Trigona Stingless Bee Honey Compared to Stinging Bee Centaurea hyalolepis and Citrus Honeys
Supplemental material, sj-docx-1-chp-10.1177_2515690X241271978 for Characterization of Antibiofilm and Antimicrobial Effects of Trigona Stingless Bee Honey Compared to Stinging Bee Centaurea hyalolepis and Citrus Honeys by Walid Salem Aburayyan, Nesrin Seder, O’la Al-fawares, AbdulFattah Fararjeh, Ibrahim S. Majali and Yousef Al-Hajaya in Journal of Evidence-Based Integrative Medicine
Supplemental Material
sj-docx-2-chp-10.1177_2515690X241271978 - Supplemental material for Characterization of Antibiofilm and Antimicrobial Effects of Trigona Stingless Bee Honey Compared to Stinging Bee Centaurea hyalolepis and Citrus Honeys
Supplemental material, sj-docx-2-chp-10.1177_2515690X241271978 for Characterization of Antibiofilm and Antimicrobial Effects of Trigona Stingless Bee Honey Compared to Stinging Bee Centaurea hyalolepis and Citrus Honeys by Walid Salem Aburayyan, Nesrin Seder, O’la Al-fawares, AbdulFattah Fararjeh, Ibrahim S. Majali and Yousef Al-Hajaya in Journal of Evidence-Based Integrative Medicine
Footnotes
Acknowledgements
The authors would like to thank the Dr Helmi Abu Baker University of Terengganu for his their support, cooperation and assistance throughout the period of study.
Author Contributions
Walid AbuRayyan, and Nesrin Seder designed the study. O’la Al-fawares, AbdulFattah Fararjeh, Nesrin Seder collected the data. Ibrahim S Majal, Yousef Al Hajaya Nesrin performed data analysis and interpretation. All authors drafted the manuscript and approved the final version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
