Abstract
Honey has a long history of therapeutic properties for multiple diseases, including inflammation and oxidative stress. This review aimed to provide a better understanding and renewed interest in the potential role of honey in obesity control, obesity-related diseases treatment and weight management, with specific reference to its components and the effect of honey overall. There is compelling evidence that honey possesses the desired properties for this purpose, as seen in the in vitro, in silico, in vivo and clinical analyses discussed in this review. This review also highlights the components potentially responsible for the health benefits of honey. Honey and its components reduce blood sugar levels, improve insulin sensitivity and lipid metabolism by reducing triglycerides, and reduce total cholesterol and LDL levels while increasing HDL levels that prevent excessive weight gain and reduce the risk of obesity and its complications. Further controlled studies are necessary to validate the role of honey in the management of obesity, both as a preventive and as a therapeutic agent.
Introduction
The current global burden of obesity (defined as body mass index (BMI) ≥ 30.0 kg/m2) and overweight (BMI = 25.0-29.9 kg/m2) is estimated to have tripled since 1975, with more than 1.9 billion cases reported. 1 As the standard of living continues to rise worldwide, overweight and obesity have emerged as one of the most common and severe nutritional problems that confront many countries.
Obesity is a chronic disease that occurs in developed and developing countries and affects all age groups. 1 A study reported that from 1975 to 2015, the global rate for obesity elevated from 3.2% to 10.8% and 6.4% to 14.9% in men and women, respectively. 2 With this rapid increase in incidence rate, it is estimated that by 2025, the global population by over 18% (men) and 21% (women) will be overweight or obese. 2 Moreover, the World Health Organization (WHO) reported that in 2016, more than 340 cases of obese children and adolescents aged 5 to 19, and in 2019, 38 million children under five were obese and overweight. 3 Among South Asian countries, Malaysia has the highest overweight and obesity rate, with 64% and 65% of the male and female population, respectively, being either overweight or obese. 4 These figures represent a massive and increasing health and financial burden because the rate increase is directly correlated with the increased burden of obesity-related complications.
Recent findings show that excessive body weight and obesity are linked to an increased mortality rate, leading to numerous complications such as heart disease, cancer, dementia and common diabetes (see Figure 1 below).5‐7 These conditions are the imbalance of energy between calories consumed and calories expended. Changes in behavioural factors such as dietary choices and patterns in physical activities are often the result of environmental and societal changes associated with the lack of supportive government policies in sectors such as education, food processing and health. 8 However, obesity and overweight, as well as their related complications, are largely preventable. Changes in lifestyle such as limiting energy intake from sugars and total fat, increasing fruit and vegetable consumption, and engaging in regular physical activity 9 can prevent and ameliorate obesity and excessive body weight.

Complications due to obesity.
Bee honey, a natural product defined by convention as a collection of nectars processed by honeybees, has been used as food and for healing wounds and intestinal diseases, as recorded in ancient Chinese, Egyptian, Greek, Assyrian and Roman reports of traditional healings. For example, a rock painting from 6000 BC found in the caves of La Arana Shekter, Spain, depicts a prehistoric man gathering honey. 10 Traditional Ayurvedic experts also recommended honey as a remedy for indigestion, coughs, insomnia, and maintaining healthy gums and teeth.11,12 Honey is considered a health tonic in the Islamic medical system and the potential therapeutic value of honey is vividly illustrated in the holy Qur’an.13‐15
In modern times, honey is considered a nutritious supplement with medicinal properties recognised by society. According to the Food and Agriculture Organization of the United Nations (FAO), production of honey in Asia increased from 600 tons in 2007 to almost 1000 tons in 2017, which indicates a significant increase in the consumption of honey. 16
Honey exhibits multiple beneficial physiological effects; it is antibacterial, anticancer, anti-inflammatory, antioxidant, and improves cardiovascular health, oral health, the immune system, and gastric activity (see Figure 2 below). The use of honey on its own or with other nutritious foods for weight management became popular by the end of the 20th century. For example, a popular recipe for weight loss is a combination of honey and lemon or lime juice, or honey and cinnamon. 17 Honey is also used as a sugar substitute in beverages such as tea and fruit juice, desserts such as pancakes, or topping in sweetened food, to provide a nutritional supplement and improve its palatability. Although there is a dearth of validated evidence that these mixtures assist in weight loss, many anecdotal claims support the positive outcomes of consuming these regularly.

Despite the claims for honey's beneficial properties, the effects of this sweet product in weight management remain controversial because of its high sugar content. In this narrative review, we discuss the current understanding of honey and its effects in alleviating obesity, and the mechanism of the relevant signalling pathway that honey acts upon.
Composition of Honey
Honey is a natural blend of many constituents that include simple and complex sugars, water, organic acids, proteins, vitamins and minerals (see Figure 3 below). Honey produced by the honeybee (Apis mellifera) is 75–85% sugars. Meanwhile, honey from stingless bees such as Heterotrigina itama, Geniotrigona thoracica, Lepidotrigona terminata, Tetragonula laeviceps, and Trigona biroi only contain 16–36% sugars. Factors influencing honey composition include botanical sources, geographical origin, climate, and environmen. Further, honey contains minor constituents, many of which have antioxidant properties, including vitamins, minerals, organic acids, amino acids, phenolic acids, and flavonoids. The sweet aroma and taste of honey come from the existence of simple and complex sugars, acids, phenolics, amino acids, tannins, and volatile compounds. The gross composition of honey is summarised in Table 1 below, while the remaining part of this section discusses the desired effects of the components of honey in managing obesity and overweight.

In this section, the composition of different honeys will be discussed in terms of their effect on obesity and its related dysfunction and on weight reduction. The constituents used in each study discussed in this review do not reflect the actual amount found in honey. Instead, we highlight each unique constituent studied and prove its beneficial properties on the human body. We believe strongly that honey acts as a whole when it comes to its therapeutic effect, and discussing each of its components may give a better and clear understanding of the mechanism of action.
Sugars
Clinically, it is known that a high intake of sugars, particularly fructose, sucrose, and maltose, can cause obesity and overweight.33,34 However, some of these sugars have been reported to exhibit anti-obesity effects when taken at appropriate volumes and concentrations. Moreover, at the physiological level, sugars help the body function at an optimum level of homeostasis. Typically, raw honey is a concentrated aqueous solution composed of fructose (37.07-42.65%) and glucose (18.20-32.10%).16,22,28‐31,35
However, honey also contains small amounts of various other sugars such as disaccharides (0.36-16.57% of sucrose, maltose, turanose and leucrose), trisaccharides (panose, maltotriose), and oligosaccharides (isomaltotetraose, maltotetraose).22,35,36 Park et al 37 found that turanose, a sucrose isomer found naturally in honey, reduced adipogenesis of 3T3-L1 cells. Lipid accumulation increased significantly when the control cells were treated with 25 mM glucose. By contrast, they reported that turanose significantly reduced lipid accumulation at 50% and 75% of 25 mM (p < 0.001), and decreased the expression of sterol regulatory element-binding protein 1C (SREBP-1C), peroxisome proliferator-activated receptor γ (PPARγ) and fatty acid synthase (FAS) mRNA level, which are known to be adipogenic markers, in a dose-dependent manner (p < 0.005). 37
Another disaccharide called leucrose is found naturally in honey and pollen.22,36 Leucrose has been reported to improve blood glucose levels while reducing hepatic lipid accumulation. In a 12-week study, high-fat diet (HFD)-fed C57BL/6 mice supplemented with 25% and 50% leucrose solution showed downregulated lipogenesis-associated genes (PPARγ, SREBP-1C, and FAS) and upregulated the genes associated with β-oxidation (acyl CoA oxidase (ACOX1) and carnitine palmitoyltransferase 1A (CPT1A)) compared to HFD mice not supplemented with leucrose. 38 Trehalose, a disaccharide of glucose linked ‘head-to-head’ in an α-(1→1)-linkage, exhibited obesity-preventing properties by inducing white adipose tissue browning accompanied by suppression of white adipose hypertrophy. 39 A study reported that mice fed a normal diet supplemented with 0.3% w/v trehalose had lower blood glucose levels after 15 weeks when compared to controls. 39 An earlier study also demonstrated that trehalose (0.3% and 2.5% trehalose in HFD) mitigates insulin resistance by increasing serum adiponectin, and upregulates protein insulin receptor substrate-1 (IRS-1) and insulin receptor substrate-2 (IRS-2) in muscle after 14 weeks, thus suppressing adipocyte hypertrophy, which contributes to obesity and insulin resistance. 40 A recent study discovered stingless bee honey contains trehalulose as a major component (13 to 44 g per 100 g honey), almost four times the amount in honey from Apis species.41,42 Trehalulose is a disaccharide known to have a low insulinemic index and glycaemic index. This is due to unusual α-(1 → 1) glucose-fructose glycosidic linkages making the rate of release of monosaccharides into the bloodstream slower than in sucrose. 43
Water
After sugar, the second-largest constituent of honey is water.16,28‐30 The range of water content in honey varies between honeybee honey and stingless bee honey.44–47 Apart from that distinction, the water content of honey is also affected by other natural factors, such as the geographical and botanical origin of the soil, nectar and climatic conditions. Other than that, the season in which the honey was harvested, the intensity of nectar flux, the degree of maturation, manipulation by beekeepers during the period of harvest, and extraction, processing and storage conditions also play an important role in the amount of water content in honey.
According to the Harmonised Method of the International Honey Commission (IHC), the maximum moisture content allowed in honey is 20%. 48 However, honey from tropical countries seems to exhibit higher water content than the IHC standard, especially from stingless bee species. A study reported that four major stingless bee species in Malaysia showed higher water content (26.9 ± 1.78%) than that of the honeybee.31,45,49 Similarly, a study of stingless bee honey from Indonesia, Thailand, Brazil and Venezuela also showed high water content, ranging from 22% to 40%.49,50
Proteins
The protein content of honey varies according to bee species, from 0.22 to 2.93%.16,22,28‐30,32,51 This may be affected by vegetal sources and pollen content, fluid and nectar sources, and secretions from honeybee's salivary glands and pharynx. The contribution of that fraction to human protein intake is marginal, due to the meagre amount of it in honey. However, many types of protein that have been identified in honey have been shown to exert beneficial biological effects on human health.
Many essential and non-essential amino acids are found in honey and pollen proteins, such as arginine, glutamine, glycine, histidine, isoleucine, leucine and lysine, phenylalanine, and serine; but proline is the most abundant.26,52 A study reported that after 24 h, 10 mM of L-phenylalanine reduced food intake and plasma ghrelin in control and obese rats (p < 0.01). 53 A total of 100 mM L-phenylalanine stimulated glucagon-like peptide-1 (GLP1) and gastrointestinal (GI) hormone peptide YY (PYY) (p < 0.01) in ileal organoid cell culture from C57BL/6 mice via the activation of a calcium-sensing receptor (CaSR), a promiscuous amino-acid-sensing receptor expressed in the GI tract. 53
Glutamine, the most abundant circulating amino acid, was reported to have the potential for weight management. 54 In a four-week pilot study of obese women, a significant reduction in body weight and waist circumference was reported after glutamine supplementation (0.5 g/kg body weight) (p < 0.05). 55 Histidine can reverse HFD-increased body weight in rats and reduce the serum concentration of tumour necrosis factor-α (TNF-α), interleukin-6 (IL-6), C-reactive protein (CRP) and malondialdehyde (p < 0.05) after supplementation with 1.875 g/kg body weight for four weeks. 56 Other studies reported that histidine supplementation (4 g/kg body weight in two weeks) improved insulin resistance, BMI and fat mass in obese women with or without metabolic syndrome through the suppression of pro-inflammatory cytokine expression by the nuclear factor kappa-B (NF-KB) pathway in adipocyte cells. 57
A naturally-occurring amino acid that functions as a neurotransmitter called γ-aminobutyric acid (GABA) is also found in honey. 28 It was found that when the diet of HFD rats and mice was supplemented with GABA (15% in HFD for eight weeks; 2 mg in water for 12 weeks), body weight reduced significantly and insulin sensitivity improved, compared to HFD rats fed a diet not supplemented with GABA. 58
The nutritional and therapeutic roles of L-arginine are known in treating and preventing human metabolic disorders, including managing weight gain and reducing obesity. McKnight et al 59 reported that L-arginine reduced adiposity by stimulating mitochondrial biogenesis and brown adipose tissue development through enhanced synthesis of cell signalling molecules (polyamines, nitric oxide, carbon monoxide, cyclic guanosine monophosphate (cGMP) and cyclic adenosine monophosphate (cAMP)), as well as increasing the expression of genes that promote whole-body oxidation of energy substrates (glucose and fatty acids).
There is no fixed volume of protein and amino acid intake that will suit all age groups. Rather, people should adjust their intake according to their metabolic rate, physiological needs and health status.60,61 A sufficient intake of protein and both essential nutritional amino acids and non-essential amino acids should play a significant role in sustaining skeletal-muscle (and physical strength) protein function, mass and synthesis, and ameliorating ageing-associated sarcopenia while improving insulin sensitivity, as well as reducing white-fat accretion.61,62 It is possible that synergistic effects of the protein and amino acids may enhance their effects in many biological activities in the human body.
Enzymes
Enzymes in raw honey is derived from pollen, nectar, and bees.16,22,28–30 The number of enzymes present in honey depends on various factors, such as the bees’ diet, age, and physiological stage, as well as the state of the colony, the temperature of the hive, the nectar flow, and its sugar content.
There are three main enzymes in honey: invertase, glucose oxidase and diastase (amylase).14,28,63–65 Invertase hydrolyses sucrose into glucose and fructose, while glucose oxidase converts glucose into δ-gluconolactone.11,32,51,66 This δ-gluconolactone is later hydrolysed to gluconic acid and hydrogen peroxide (H2O2), which has been reported to exert antimicrobial activity.67,68 Interestingly, the antimicrobial activity of honey does not depend solely on H2O2. A study by Zainol et al 69 found that the antibacterial activity of non-peroxide honey did not give a significant result when compared to total honey (without H2O2 removal). In fact, the antimicrobial activity of non-peroxide honey was slightly reduced, indicating that H2O2 is still one of the major components of honey's antibacterial properties. 69 H2O2 was also reported to mimic insulin by activating many of the same physiological responses. 70 Meanwhile, diastase is an amylolytic enzyme that serves as an indicator of honey quality.30,51 It hydrolyses starch to produce β-amylases and dextrin that serve as precursors to the formation of maltose. 32
A report on the anti-diabetic activity of honey reported that among five multi-floral raw and processed honey samples, raw stingless bee (Trigona irridipennis) honey showed the highest α-amylase and α-glucosidase inhibitory activity compared to the raw honeybee (Apis cerana, A. mellifera, A. dorsata and A. florea) honey. 71 The study also reported that processed honey significantly decreased the inhibitory activity of both α-amylase and α-glucosidase. 71 Another study corroborated these findings, showing that heating and filtration reduced the final quantity of enzyme (73% drop in invertase level and 13% drop in phosphatase level). 72 These results suggest that honey could be a potential sugar replacement for diabetic patients who experience excessive weight gain, for reducing postprandial hyperglycaemia and obesity.
In a report on the association of amylase with metabolic syndrome, some obese and overweight people (and some diabetes patients) were found to have a higher likelihood of low serum amylase due to impaired insulin activity, insufficient insulin action and insulin resistance. 73 Further, a six-month weight-loss programme could significantly improve the reduction in serum pancreatic amylase and remain improved for a further ten months. Therefore, it is unclear whether α-amylase inhibitors alone would be beneficial for preventing overweight and obesity.
Interestingly, although honey was found to be an α-amylase and α-glucosidase inhibitor, anti-hyperglycaemic and anti-hyperlipidemic activity was also reported. Due to its varying composition, honey is believed to exert multiple pathways to ameliorate obesity and diabetes synergistically, or perhaps antagonistically.
Organic Acids
Honey tastes slightly acidic because it contains approximately 0.57–0.60% organic acids.16,22,28–30,32 Furthermore, the pH of honey from stingless bees (3.32-3.92) is slightly lower than that of the honeybee (3.73-3.95) due to the presence of gluconic acid from their various botanical sources.46,47,49,74 Organic acid content is useful for differentiating between honey according to their botanical and geographical origin; it also imparts honey's unique colour, flavour, and physicochemical properties. 74
Gluconic acid, glutamic acid, citric acid, propionic acid, and acetic acid are among the common organic acids found in honey.28,75–82 Of these, acetic acid and propionic acid showed potential for reducing obesity. Acetic acid (1.5% in water for six weeks) was found to protect type-2 diabetic rats and HFD mice against obesity and suppress body fat accumulation in both liver and muscle. 83 The suppression was due to the upregulation of gene peroxisome-proliferator-activated receptor α (PPARα) and fatty-acid-oxidation-related protein by α2 5’-AMP-activated protein kinase (AMPK) mediation in the liver. 83
On the other hand, propionic acid possesses ‘anti-obesity’ properties that ameliorate unhealthy levels of fasting glycaemia, body weight and insulin tolerance in animal models. It was found that just 10 mM of propionic acid and butyric acid inhibited lipolysis and de novo lipogenesis and increased insulin-stimulated glucose uptake in primary rat adipocytes, leading to healthier adipocytes and glucose uptake that could be beneficial in the context of preventing obesity and type-2 diabetes. 84
Vitamins
Honey has a small amount of vitamin content (<0.1%), which comes from pollen. 28 Vitamin B and C are found in honey, and due to honey's low pH value, any vitamins present tend to be preserved.14,28 However, vitamin C level can be unstable due to its chemical and enzymatic oxidation processes that could be accelerated by environmental factors such as light, heat and oxygen.30,85 In addition, the removal of pollen during commercial honey filtration may also cause a reduction in vitamin content. 28 Another process that causes a loss of vitamin content in honey is the oxidation of vitamin C by the hydrogen peroxide produced by glucose oxidase. 30 These factors may be the reason it is advisable to store honey in a closed container, at a low temperature and away from light.
B vitamins such as B1 (thiamine), B6 (pyridoxine) and B8 (biotin) have been reported to have positive effects on obese patients.11,14,79,82 These micronutrients are essential for dealing with increased load on metabolic pathways caused by high caloric intake in overweight people, particularly on glucose metabolism. A study review concluded that thiamine (in the form of thiamine diphosphate) plays a major role as a co-factor in glucose metabolism, especially for Krebs cycle key enzymes such as alpha-ketoglutaric acid dehydrogenase (KGDH) and pyruvate dehydrogenase (PDH). Maguire et al 86 assumed that by today's daily calorie intake standards, 0.5 mg thiamine is required to process 1000 kcal. Thus, it might be expected that an appropriate recommended daily allowance (RDA) would be 2.0 mg/day. Thiamine deficiencies may result in the accumulation of anaerobic metabolites, such as lactate, due to a mismatch between the caloric burden and the function of thiamine-dependent enzymes. 86
Pyridoxine, together with leucine, exhibited a long-term synergistic effect in reducing adipocyte lipid storage with increased muscle fat oxidation in obese subjects when supplemented with a nutraceutical containing 2.25 g leucine and 30 mg pyridoxine for 24 weeks, thus attenuating adiposity and reducing body weight in obese patients. 87 Another vitamin reported to attenuate obesity is biotin. Biotin plays an essential part in carbohydrate and lipid metabolism. Besides that, biotin also regulates the expression of genes essential for metabolism, and has stimulatory effects on genes, which favour glycaemic control. 88 In a study of a biotin and chromium picolinate combination (2 mg and 600 µg, respectively), the glycaemic control of overweight and obese patients with type-2 diabetes improved significantly after 90 days of supplementation. 88
Minerals
Honey contains various macro- and microelements such as calcium (3-31 mg per 100 g), sodium (1.6-17 mg per 100 g), phosphorus (2-15 mg per 100 g), magnesium (0.7-13 mg per 100 g), iron (0.03-4 mg per 100 g) and potassium, the last being found in the highest concentration in honey (40-350 mg per 100 g).51,89 These characteristics reflect the chemical components of the plant from which the nectar was harvested by the honeybees. According to various studies, honey from Spain was found to have the highest concentration of manganese, while honey from Hungary showed the lowest concentrations of chrome, cadmium, lead and copper. 30 A review by Santos-Buelga and Gonzalez-Paramas 28 concluded that the mineral content of honey could vary from 0.11% to 0.72%, depending on the types of soil in which the plant and nectar (the botanical sources of specific types of honey) were located.
As stated above, potassium is the most abundant element in honey, generally corresponding to one third (100-588 ppm) of the total mineral content found in honey. A systematic and meta-analytical review of potassium and obesity found that adequate daily intake of potassium can reduce the risk of obesity and metabolic syndrome. 90 High potassium intake was not associated with obesity. Rather, the patients with the highest potassium intake relative to sodium had the lowest risk of obesity.
Calcium is another micronutrient found in honey. It regulates many cellular processes such as bone formation, cell proliferation and differentiation, and has been reported to prevent or treat obesity.91,92 The 2011 Institute of Medicine Dietary Reference Intake committee recommended dietary allowances for calcium at 1300 mg/day for children aged 9 to 18 years, and 1000–1200 mg/day (varying by age) for healthy adults. 93 Many studies have suggested the possible anti-obesity mechanism of calcium supplementation such as regulation of adipogenesis in 3T3-L1 adipocytes (10 mM), fat metabolism, absorption and excretion (0.6% w/w) in HFD rats and mice, adipocyte (precursor) proliferation, and apoptosis, thermogenesis, and gut microbiota.91,94
Phosphorus and magnesium intake have been inversely correlated with weight management. The modernisation and globalisation of food industries have been correlated with increased consumption of products containing negligible amounts of phosphorus and magnesium. Studies have reported that low consumption of phosphorus (<2.5 g/day) is positively associated with increased body weight.95,96 Another study reported that phosphorus supplementation (375 mg in every main meal, for 12 weeks) and dietary magnesium (10 mg per 1000 kcal/day, for eight months) halted weight gain and BMI growth while significantly decreasing waist circumference in healthy and obese participants, thus suggesting that phosphorus supplementation and magnesium have the potential to prevent obesity.95,97
Antioxidant Properties
The antioxidant property of honey is associated with phenolic compounds, a chemically heterogeneous group divided into two different classes, according to their basic chemical structure: phenolic acids and flavonoids. These two groups of phenolic compounds are the main functional components of honey. They contribute significantly to the total antioxidant activity of honey and have accompanying beneficial effects on human health98–100. Like other honey components, the phenolic compounds mostly depend on their floral source; in fact, these compounds can be used as a tool for classification and authentication, especially in the case of mono-floral honey varieties. Additionally, studies reported that phenolic compounds, especially flavonoids, are found in greater proportions in stingless bee honey than in honeybee honey.32,101–104
However, the antioxidant property of honey may change in response to several factors such as time, storage, and commercial heating processes. Before it is sold, honey usually undergoes a heating process to maintain uniform colour, taste and viscosity in every batch. Studies have shown that after heating at 100 ⁰C for 24 h, honey's antioxidant content and radical scavenging activities against peroxide and 2,2-diphenyl-1-picrylhydrazyl (DPPH) radicals increased significantly. Its angiotensin I-converting enzyme (ACE) increased and hyaluronidase activities decreased gradually after extended heating.102,105
Phenolic acids
Phenolic acid compounds in honey, such as vanillic acid, syringic acid, gallic acid, ferulic acid, chlorogenic acid, and ellagic acid, were shown to exert an anti-obesity effect. In HFD rats, vanillic acid (0.5% w/w in HFD for 15 weeks) contributed to a reduction in body weight gain, improved glucose tolerance, and insulin resistance by activating brown adipose tissue thermogenesis and promoting inguinal white adipose tissue browning. 106 Further, 10 mg/kg/day of vanillic acid significantly decreased the body weight of obese mice during four weeks of administration. Adipogenic markers (PPARγ, C/EBPα) were also reduced while AMPKα, the key factor in energy metabolism, was increased in the white adipose tissue and liver cells of vanillic acid-treated obese mice. 106 In 3T3-L1 adipocytes, vanillic acid (10 µM) markedly decreased the expressions of adipogenic markers (PPARγ and C/EBPα), which was associated with AMPK activation. 107
Syringic acid (0.05% w/w for 16 weeks) was found to ameliorate insulin resistance and reduce body weight, hepatic lipid content, serum levels of leptin, TNFα, interferon γ (IFNγ), IL-6 and monocyte chemoattractant protein-1 (MCP-1). 108 Meanwhile, it also increased the circulation of adiponectin in HFD mice. In an in vitro study, 500, 750, and 1000 µmol/mL of syringic acid concentration reduced lipid accumulation, adipogenesis and reactive oxygen species (ROS) while increasing adipolysis in 3T3-L1 adipocytes. 109
Gallic acid, another type of phenolic compound, is a naturally occurring polyphenol and has been investigated extensively because of its effectiveness in attenuating obesity and ameliorating obesity-mediated metabolic complications. The consumption of gallic acid of 10 mg/kg body weight (BW) for nine weeks; 50 and 100 mg/kg BW for ten weeks for HFD rats and mice significantly decreased body weight and liver weight. 110 Serum triglyceride, phospholipid, cholesterol, and insulin were also significantly decreased after administering gallic acid. It was believed that gallic acid plays its role by activating AMPK and by regulating mitochondrial function via the activation of PPARγ coactivator 1α.
Ferulic acid and chlorogenic acid, phenolic acids abundant in fruits, vegetables and leaves, are also found in honey. A study found that 0.5% w/w of ferulic acid suppressed increases in body weight, body fat accumulation and blood glucose, and suppressed hepatic lipid accumulation and inflammatory cytokines (IL-6 and TNF-α) (p < 0.05) in HFD rats after six weeks of supplementation. 111 Similarly, a 15-week study on HFD rats demonstrated that 10 mg/kg ferulic acid significantly reduced body weight gain, adipocyte size and fat accumulation. 112 Serum lipid profiles (amylase and lipase activities), blood glucose, and insulin hormone were also reduced after ferulic acid supplementation. 112 Chlorogenic acid (150 mg daily for six weeks) was found to reduce weight gain, lipid levels in plasma and altered mRNA expression of lipogenesis- and lipolysis-related genes in adipose tissue. 113 The same study also demonstrated that chlorogenic acid supplementation significantly inhibited gut microbiota dysbiosis, including the growth of Desulfovibrionaceae, Ruminococcaceae, Lachnospiraceae, and Erysipelotrichaceae; and increased the growth of Bacteroidaceae and Lactobacillaceae after supplementation of 150 mg/kg daily for six weeks in HFD mice. 113
Ellagic acid is another promising dietary phenolic acid compound that could modulate the development of obesity and associated metabolic consequences. To date, much evidence has emerged that ellagic acid may decrease symptoms of chronic metabolic diseases, including dyslipidemia, insulin resistance, type-2 diabetes, and non-alcoholic fatty liver disease. 114 Ellagic acid inhibits adipogenesis and reduces lipid accumulation in 3T3-L1 cells by significantly reducing early adipogenic markers (Kruppel-like factor 4, Kruppel-like factor 5, early growth response protein 2, PPARγ and CCAAT/enhancer-binding protein α (C/EBPα)) via cell cycle arrest. 115
Flavonoids
Flavonoids are a diverse group of phytonutrients, a class of polyphenolic secondary metabolites synthesised by the polypropanoid pathway. Quercetin, myricetin, galangin, kaempferol, chrysin, apigenin, and hesperetin are some of the flavonoids known to exhibit anti-obesity properties.32,101–104 Kassim et al 116 reported that honey contains approximately 223 μg of myricetin, 69 μg of chrysin, 66 µg of quercetin and 16 μg of kaempferol per 100 g. Animal studies found that quercetin, myricetin and galangin protected HFD mice and rats from adipose tissue accumulation and body weight gain.117–119 These also suppress lipogenesis and the gene expression of adipogenesis key factors, C/EBPα, by downregulating the gene levels of acetyl-CoA carboxylase (ACC), FAS and sterol regulatory element-binding protein 1c (SREBP-1c). An in vitro study on primary adipocytes reported that quercetin induced lipolysis by increasing hormone-sensitive lipase (HSL) and cAMP levels. 120 Quercetin (925 mg/kg) also inhibited adipogenesis by decreasing PPARγ, C/EBPα and C/EBPβ expression. 117 The level of adipokines (angiopoietin-like 4, adipsin, PAI-1) and glycolysis-associated enzymes (ENO2, PFKP, PFKFB4) associated with obesity and adipose tissue dysfunction were also significantly reduced when treated with quercetin. 120
Kaempferol (30 µM) also showed lipid-accumulation inhibition in 3T3-L1 adipocytes and mediated the regulation of early adipogenic factors, resulting in the attenuation of late adipogenic factors (C/EBPα and PPARγ). 121 Similarly, 50 µM of chrysin induced 3T3-L1 adipocytes browning via the upregulated expression of brown fat-specific genes (PPARα, PPARγ, PPARδ, p-AMPK, p-acyl-CoA carboxylase, HSL, PPARγ coactivator-1α, carnitine palmitoyltransferase 1, perilipin, acyl-coenzyme A oxidase 1 and uncoupling protein 1) as well as enhanced lipid metabolism (p < 0.05). 122 Chrysin (100 mg/kg) also exerted an anti-obesity effect by inhibiting pancreatic lipase, reducing the calorie intake and sucrose preference of HFD rats, thus significantly decreasing their body weight, BMI, adiposity index and calorie intake. At the same time, it significantly increased faecal cholesterol (p < 0.05). 123
Apigenin at 50 µM reduced the expression of PPARγ by the activation of AMPK, thus leading to decreased expression of adipogenic genes (p < 0.05) and subsequent suppression of adipogenesis in 3T3-L1 adipocytes. 124 In another study, hesperetin inhibited pre-adipocyte differentiation and suppressed lipid accumulation by arresting adipocyte maturation in human mesenchymal stem cells (hMSCs), inducing adipogenesis. 125
Evidence that Honey has the Potential to Control Obesity, Obesity-Related Dysfunction and Weight Gain
This section discusses the evidence for honey's use in weight management in in vitro, in silico, in vivo and clinical studies, and is summarised in Table 2. Examples of honey in combination with other food products to reduce weight gain will also be discussed. Although honey's excess-weight-reduction effect has been associated with its anti-obesity properties, caution must be taken not to label honey as an anti-obesity medication, as not enough study has been done to prove honey an effective anti-obesity medication.
in Vitro, in Vivo, and Clinical Trials Carried out with Honey in Relation to Obesity and Weight Gain Management.
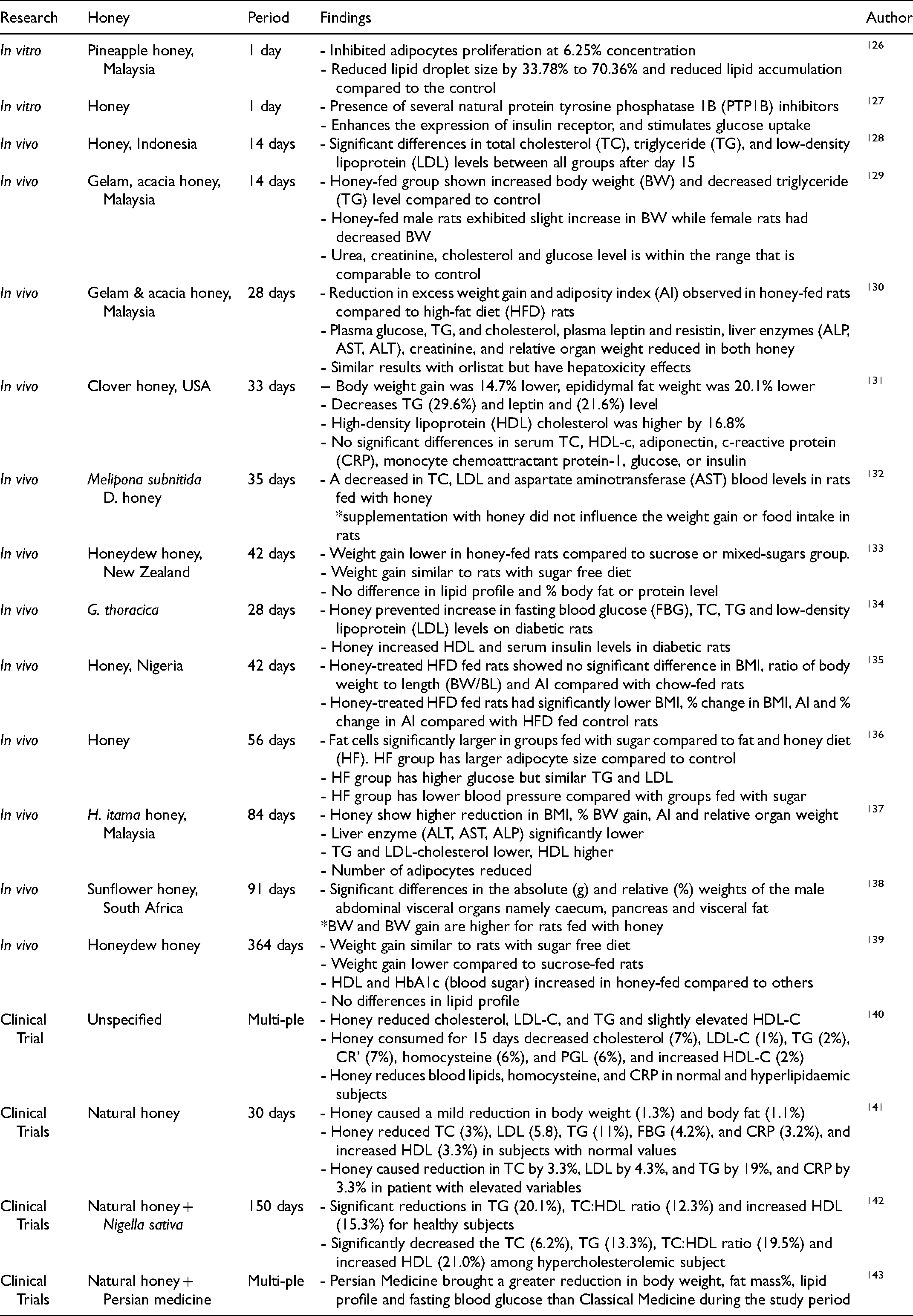
Effect of Honey Observed in in Vitro Model
For the in vitro study – to assess the application of honey in reducing excess weight reduction – a different approach must be taken since the effect on weight reduction cannot be assessed at the cellular level. This could be achieved by evaluating the levels of biomarkers or cellular products related to weight gain, such as lipid metabolites.
Unfortunately, at present there is limited information on cellular-level processing regarding the effect of honey on influencing or reducing obesity, with only a few prominent studies. One of these was conducted to observe the effect of Malaysia's pineapple honey on 3T3-L1 adipocytes and lipid accumulation. 126 When cells were treated directly with pineapple honey at 25, 50% and 100% (v/v) concentrations, a significant reduction was observed in the size of the lipid droplets produced by the cells, ranging from 33% to 70.36%. Also, the accumulation of lipid droplets was significantly reduced compared to the untreated control in a dose-dependent manner. However, the researchers noted that higher concentrations of honey (>25% concentration) were detrimental to adipocyte viability; smaller lipid droplet sizes and lower lipid accumulation were observed. It may be that the honey disrupted the normal cellular process; however, further investigation is required. 126
Another study focused on the impact of different floral varieties of Tuscany honey (honeydew, chestnut, wildflowers and acacia) on the insulin signalling pathway. 127 The honey was extracted with 80% v/v ethanol, and the insoluble residue was removed centrifuged. The study reported that all honey extracts at a 10 µL/mL concentration inhibited the enzyme PTP1B, one of the main negative regulators of insulin receptor signalling. Using liquid chromatography–mass spectrometry (LC-MS) for physicochemical analysis, the study identified different compounds such as chrysin, naringenin, kurarinone, acacetin, pinocembrin, apigenin and caffeic acid, in all four types of honey; wildflower honey produced the highest values. These flavonoid compounds are recognised as PTP1B inhibitors, suggesting that such molecules could contribute to the PTP1B inhibitory control of the honey extracts used in that study. The honey extracts (0.02% extract concentration) also enhanced the expression of insulin receptors and stimulated glucose uptake in HepG2 cells after 96 h of incubation. 127 This study proved that a low concentration of honey inhibits PTP1B enzymes and upregulated insulin receptor expression, thereby increasing insulin sensitivity.
These two studies show that lipids play an important role in obesity and weight management research, as they serve as the body's storage repository for excess energy. Excessive amounts of lipids in the body could lead to various obesity-related metabolic disorders. At present, the mechanism employed by honey to reduce lipid accumulation and size is still unknown. Nevertheless, this presents an opportunity to highlight honey's potential and warrants more research at the cellular level.
Effect of Honey Shown in in Silico Model
In silico processes are starting to be widely used in studies to predict drug-body interactions; the in silico molecular docking simulation model is rapidly emerging as a means to understand and predict possible modes of action and interaction between a ligand and a target biomolecule. The advancement of molecular technology has identified new drug targets for new pathways in the pathophysiology of obesity. Fat mass and obesity-associated protein (FTO) is one of the significant genes implicated in obesity and its complication of metabolic diseases, and is currently a potential target for an anti-obesity drug. 144 FTO gene expression is associated with the regulation of energy balance and food intake; it increases food intake and the desire for high-calorie foods, which increases the risk of obesity.144,145
To date, there have been limited in silico studies on using honey to address obesity. However, a study on the antioxidant content of plants showed a high binding affinity with FTO protein structure, suggesting a possible anti-obesity mechanism. Mathew et al 146 found that selected flavonoids (luteolin, quercetin, capsaicin, abscisic acid, ajoene, and diosgenin) may be effective drugs against FTO protein, and could be used therapeutically for the prevention of obesity. When a docking simulation procedure was performed with the FTO protein structure (PDB ID: 3LFM), the flavonoids – especially luteolin – showed a high affinity with the docking score at −25.632 kcal/mol, followed by abscisic acid, capsaicin, ajoene, quercetin and diosgenin.
From the discussion in the previous section, honey contains various bioactive compounds, including phenolic acids and flavonoids, that positively affect obesity and weight management. Although there are limited obesity-related in silico studies using honey as a target ligand, it is safe to assume that the bioactive compounds found in honey have a high affinity for FTO protein. Nevertheless, further studies using in vitro and in vivo model validation are necessary to better understand the underlying pathways.
Effect of Honey Observed in in Vivo Model
The in vivo study evidence for honey's potential in weight management is presented in Table 2. Earlier studies focused on comparing its effect on honey-fed rats and sugar-fed rats. Long-term studies by Chepulis, 133 and Chepulis and Starkey, 139 found that despite the similarity in food intake, the overall weight gain percentage was significantly lower in rats fed with honey (10% honeydew honey from New Zealand) than in the rats fed with sucrose (8% solution), or mixed sugars (8% solution) after 52 weeks of supplementation. The studies also reported a significant reduction in HbA1c levels, while HDL-cholesterol was significantly increased in rats fed with honey compared with rats fed sucrose.133,139
An eight-week study by Romero-Silva et al 136 reported no significant difference in overall weight gain, body-fat mass, blood glucose, triglyceride and LDL between sugar-fed and honey-fed rats. However, blood pressure and fat-cell size were significantly lower in honey-fed than in sugar-fed rats.
A study by Nemoseck et al 131 found that after 33 days, clover honey-fed rats (300 g/kg daily) had lower weight gain and food intake percentages, at 14.7% and 13.3%, respectively. In addition, epididymis fat weight was 20.1% lower, leptin and triglycerides serum levels were lower by 21.6% and 29.6%, respectively, and non-HDL cholesterol increased by 16.8% (p < 0.05).
Furthermore, a study by Ajibola et al 138 reported no significant differences in body weight between honey-fed rats (sunflower honey from South Africa), HFD-fed rats and control after 13 weeks; however, honey-fed (10 mL/kg of 50% solution) rats showed increased pancreas and caecum weights in male rats. The increased weight of the organs could elevate the digestive functions by the intestinal microflora in the caecum and enzymatic activities of the pancreas. The study also reported that in contrast with HFD-fed rats, honey-fed rats showed enhanced growth of intestinal villi, and no pathology in the rodents’ abdominal viscera, suggesting a potential nutritional benefit of honey as a substitute for refined sugars. 138 Similarly, a short-duration study by Nurmasitoh and Pramaningtyas 128 reported that 0.8 mL honey supplementation per day could reduce the triglyceride, total cholesterol, and LDL serum levels in HFD rats after 15 days (p < 0.05).
More recent studies have been conducted to observe the positive effect of honey in HFD rats. An eight-week study on the effect of honey (1 g/kg BW) on HFD rats found no significant difference in body weight, BMI or adiposity compared with control rats. 135 When compared with rats fed with HFD, rats fed with HFD supplemented with honey had significantly reduced BMI (p < 0.01), body weight and adiposity percentages. 135 Mohd Rafie et al 137 showed that HFD rats fed a supplement with 750 mg/kg of body weight of stingless bee honey (Heterotrigona itama) showed significantly higher reductions in percentages of body-weight gain, BMI, adiposity index, and relative organ weight (ROW). The rats lost weight (−2.30 ± 2.9%) compared to their initial body weight, and their HDL levels were higher than those of control obese rats. 137 This suggests that honey can improve indicators associated with obesity reduction.
Earlier that year, Abdul Aziz et al 134 reported that 1 g/kg BW of stingless bee honey (from Geniotrigona thoracica) increased the body weight of control rats (p < 0.05). However, when stingless bee honey was administered to diabetic rats, the increase in body weight was reduced. Fasting blood glucose, total cholesterol (TC), triglyceride (TG) and low-density lipoprotein (LDL) levels were also reduced. HDL and insulin in diabetic rats increased after supplementation with stingless bee honey (p < 0.05). Prior to animal testing, the study revealed the presence of L-phenylalanine and other chemical compounds in the honey used. The study also suggested that there is a possibility that the presence of phenylalanine might be responsible for the anti-diabetic effects of the honey. 134 Another study using stingless bee honey (from Melipona subnitida D.) on dyslipidemic rats for 35 days found that the honey lowered food consumption, increased glucose tolerance and superoxide dismutase (SOD) activity, and decreased total cholesterol, LDL and AST blood levels, compared to untreated dyslipidemic rats. 132
A 14-day study by Samat et al 129 using Malaysia's gelam and acacia honey (2 g/kg body weight each) found the increase in body weight in rats in all honey-supplemented groups similar to that of the control group. However, in contrast to the rats in HFD groups (sucrose-only diet), acacia honey significantly reduced total calories and total food intake, while the gelam honey significantly reduced the triglyceride serum level compared to the control group. The study demonstrated that gelam honey, multi-flora honey, exhibits lower adiposity index and weight gain percentage, reduced triglyceride, glucose and cholesterol compared to the acacia (mono-flora) honey. 129 Furthermore, the study found that total flavonoid content (TFC) and total phenolic content (TPC) were higher in gelam honey than in acacia honey, which might be due to the different multi-floral and mono-floral sources. Based on histological analysis, the study reported that HFD with honey is safer, with liver histological evaluation similar to that of non-obese rats. 129
All these studies agreed that honey-supplemented rats experienced similar or increased body weight compared to control rats. This result is expected since honey comprises mostly simple sugars, and the body stores excess sugar, which translates into a slight body weight increase. However, the overall percentage weight gain was significantly lower in honey-supplemented rats compared to sugar-supplemented rats.
Effect of Honey Shown in Human Clinical Trial
Although there have been quite a number of studies that have focused on the beneficial effects of honey on obesity in vitro and in vivo, there have been only a limited number of clinical trials and human studies performed to study the effects of honey on obesity. These clinical studies vary widely, determining the effect of honey alone on body weight, or combinations of honey with other nutrition, and ranging in duration from three weeks to six months.
In a study on obese and overweight patients, honey reduced 3% of TC, 4.2% fasting blood glucose, 5.8% LDL-cholesterol, 11% triglyceride, 3.2% CRP, and increased HDL-cholesterol by 3.3%; a subject with elevated variables had TC reduced by 3.3%, LDL-cholesterol by 4.3%, triacylglycerols by 19%, and CRP by 3.3%. 141 Honey also reduced cholesterol, LDL-cholesterol, TG, CRP and blood plasma glucose in healthy, diabetic and hypertriglyceridemic subjects, compared to sugar solution (dextrose) and artificial honey. 141 A study from Farakla et al 147 showed that honey did not result in significant changes in BMI or parameters (glucose, TC, TG, HDL-cholesterol, insulin, CRP, HbA1C, Vitamin A and E, interleukins, TNF-α and MCP-1) when tested on healthy obese adolescence girls (15 g of honey daily for six months). However, the study notes that although some results were not statistically significant, they might be clinically important or attain statistical significance if honey was consumed for a longer period of time. 147
Few studies have reported the effect of honey in combination with other nutrition. For example, supplementation with a mixture of natural honey and Nigella sativa seeds (50 mg/kg body weight) significantly reduced triglycerides serum level by 20.1% and TC:HDL-cholesterol by 12.3%, and increased HDL-cholesterol by 15.3% for healthy subjects after five months consumption. 142 Moreover, in a hypercholesterolemic subject, the mixture significantly decreases 6.2% of TC, 13.3% of triglycerides and 19.5% of TC:HDL-cholesterol, increasing about 21% HDL-cholesterol. 142 Another example: honey supplemented with a mixture of Persian herbs such as Nigella sativa, Trachyspermum ammi, Zataria multiflora and Mastic gum, in the ratio 1:1:1:1 to 12 units of pure and natural honey, resulted in a significant reduction in body weight, lipid profile, fat mass percentage, and fasting blood glucose in overweight women (BMI: 27-29.9 kg/m2) from 18 to 60 years old, compared to a standard diet with orlistat. 143
However, similarly to the in vivo model study, none of the studies above provided any data on the chemical components of the honey used, and some studies combining honey with other products make it harder to pinpoint the cause of the effect. Also, differences in type of flower source, geographical area, climate, conditioning by processing techniques and storage, and the different honeybee and stingless bee species involved in honey production cause great variation in the composition, taste, smell and colour of different types of honey. This makes the physicochemical analysis of honey relevant, and essential to better understanding the effect exerted by the honey in our study.
Honey in Weight Gain Control: Possible Mechanism and Challenges
Possible Mechanism for Controlling Obesity, Weight Gain and Related Complications
The favourable effects of honey on obesity and its complications can be attributed to the sugars, which make up the majority of the honey components, together with the proteins, organic acids, vitamins and minerals, and phenolic compounds it contains in water solution. A conceptual summary of the proposed mechanism by which honey may reduce obesity is presented in Figure 4.

Possible mechanism for how honey ameliorates obesity and weight gain.
Figure 4 suggests that honey modulates obesity by reducing the expression of PPARγ, FAS, SREBP-1C and CEBPα protein, while increasing the expression of insulin receptors (IRS-1 and IRS-2) and adiponectin protein. These lead to a reduction in adipogenesis activity, thus reducing lipid accumulation in adipocyte cells. The serum levels of TNF-α, IL-6 and CRP are also reduced, leading to a reduction in NF-KB, thus improving insulin sensitivity. NF-KB is a significant factor for insulin resistance in adipocyte cells. In liver cells, honey decreases the expression of PPARγ and CEPBα while increasing the expression of PPARα and AMPK. If the honey is consumed regularly for an extended period of time, all these changes could lead to improved glucose homeostasis, insulin-stimulated glucose uptake, and adipolysis activity. The presence of unique sugars such as leucrose, turanose and trehalulose, an antioxidant such as chrysin, apigenin and gallic acid, and also the essential and non-essential amino acids in honey could work together or individually to suppress adipogenesis and decrease lipid accumulation.38,110,123,124,148,149
The changes made at the cellular level led to reductions in serum levels of triglyceride, total cholesterol, LDL, CRP, blood glucose, leptin and HbA1C in the blood vessel. The phenolic acids found in honey can increase glucose tolerance by improving glucose homeostasis and lowering cholesterol levels by reducing lipid accumulations. These reductions are accompanied by increases in HDL, GLP1 and PYY. GLP1 is a hormone that stimulates insulin production and inhibits glucagon release, thus reducing blood glucose levels. PYY is a peptide known to affect appetite.
Interestingly, phenylalanine found in honey can increase the level of PYY, thus reducing appetite. This makes honey very suitable for obese or overweight people; it can control their high body weight and high caloric intake, and at the same time improve insulin sensitivity and glucose homeostasis. Honey is known for reducing high levels of blood cholesterol and blood glucose, which simultaneously reduces cardiovascular disease and diabetes risk. Persistent hypercholesterolemia and hyperglycaemia can lead to various health conditions (see Figure 5 below).11,12,14,32 Drugs may target and reduce microvascular complications, while honey ameliorates macrovascular complications. Moreover, based on this review, honey may work synergistically with clinically useful drugs without antagonising their actions.

Possible targets of honey to ameliorate complications due to obesity.
Based on the suggested pathways in Figures 4 and 5, modulating the obesity factors is just one of the various pathways through which honey can reduce obesity complications. This means that honey may exert multiple effects, such as lipid-accumulation-lowering ability, or improving-glucose-homeostasis ability, as well as other pathways for ameliorating obesity complications. Therefore, the various bioactive components of honey, such as proteins, vitamins, minerals, organic acids and phenolic compounds, work synergistically.
Challenges and Limitations of Research Using Honey
The composition of honey is as diverse as its floral sources and species of bee (honeybee or stingless bee). 28 Thus, ‘standardising’ honey is challenging, as its composition varies according to geographical origin, seasonal factors, the floral surroundings of the beehive, and storage conditions, all of which play an essential role in influencing particular types of honey.
Unfortunately, most of the studies discussed above do not include an analysis of the honey used, such as its physicochemical data and antioxidant content. Understandably, this might not be their main objective; however, data from the honey analysis can assist our understanding of the mechanisms behind honey's effects. If the concentrations of honey components such as sugars, vitamins, proteins and polyphenols were identified, it would reflect a better understanding of their results. Also, honey quality changes over time, at least in terms of its antioxidant capabilities, which depend on storage method and duration.30,150–154 We suggest involving honey analysis in any honey-related studies, including physicochemical and antioxidant analysis, for clearer evaluation of the quality of honey used; the selection of which data to focus on should be open to the research community for discussion.
Another important reason for compositional analysis of honey is to distinguish between natural (pure) and adulterated honey. Due to the effect on the market of the scarcity of pure honey and the profit motive, some irresponsible traders produce and sell adulterated honey, which is far cheaper and easier to produce than pure honey. Unfortunately, adulterated honey exhibits none of the beneficial therapeutic effects of natural honey, and long-term consumption can negatively affect the body.18,155–157 The issue of the existence of adulterated honey should be solved promptly to protect consumer rights.
Besides identifying the composition of natural honey, recent studies have discovered what is potentially a new way to identify fake and adulterated honey, by using the fluorescence excitation-emission matrices. A study from Dramicanin et al 158 reported that there are significant differences between the fluorescence of natural versus adulterated honey. The majority of the fluorescence comes from aromatic amino acids, phenolic compounds, and fluorescent Maillard reaction products. These exist in five spectral regions of emission and excitation wavelength, work with 100% accuracy, and are based on linear discriminant analysis (LDA). 158 Such accuracy proposes that fluorescence excitation-emission spectroscopy could be a promising method for detecting honey adulteration.
Although this review discusses the positive effects of honey and its components towards alleviating obesity and its potential mechanism of action, some studies reported the opposite, especially in clinical analysis. An eight-week study from Sadeghi et al 159 concluded that daily intake of 50 g honey might have an adverse effect on LDL and adiponectin levels in type 2 diabetic patients. The serum adiponectin level in the honey group reduced significantly compared to that of the control group, while honey did not reduce LDL-C to the same extent as in the control group, suggesting an apparent adverse effect of honey on LDL-C. 159 A randomised cross-over study by Al-Tamimi et al 160 showed that honey does not adversely affect serum lipid levels in adult men and women. The four-week study showed that there was no effect on serum insulin, TC, LDL and HDL levels for the honey and sucrose groups. 160 A systematic review and meta-analysis study by Gholami et al 161 concluded that honey did not affect lipid profile (TG, TC, LDL, HDL, and LDL: HDL ratio), contrary to the previous meta-analysis study results by Tul-Noor et al, 162 which showed a beneficial effect of honey on lipid profile (TG, LDL and HDL).
The effect of honey on lipids mechanisms can be influenced by many factors, including gender, population, honey, and geographical condition. Therefore, further studies must be conducted with a larger sample, different populations and various types of honey to elucidate the effect of honey intake on lipid profile.
Conclusions
In this paper, we presented two different approaches to assessing the effects of honey on obesity, its complications and weight gain, ie (a) by looking into specific individual components and (b) by considering honey as a whole. However, honey is comprised of thousands of compounds, some of which are still being identified by researchers. Plus, the task of isolating adequate amounts of any specific compound from honey in order to exert the desired effect could prove difficult. Hence, the study of using honey as a whole on obesity is viewed as a more practical approach. The idea that the components of honey may work synergistically is becoming more prominent and relevant; and it is also the view of the author that honey should be studied (and applied) as a whole in future studies.
Though the complete mechanisms are yet to be widely agreed on, various investigations have shown that honey ameliorates obesity through its intervention with complicated cell-signalling pathways. The various components of honey have specific functions in controlling obesity and overweight, despite their minuscule amounts. Honey encourages the adipolysis process and improves glucose homeostasis and insulin sensitivity in adipocyte cells, leading to lower glucose, triglycerides, cholesterol and LDL in the blood system. However, more research is needed to unravel the possible mechanisms of honey action, and detailed, long-term clinical trials are required. As shown in this review, only a handful of clinical trials have tried to study the effects of honey on obesity. This presents an opportunity for the scientific community to conduct more studies as obesity rates continue to grow rapidly. A deeper understanding of the mechanisms of honey's effect on obesity is essential to promoting the consumption of honey not only as a natural sweetener, but as an alternative means of managing obesity, its complications and weight.
Footnotes
Acknowledgments
We would like to thank Mohd Naim Fadhli Mohd Radzi for the work on compiling the original draft for this review. We also appreciate Jonel P. Saludes (JPS) and Doralyn S. Dalisay (DSD) for their valuable advice throughout this review. JPS and DSD acknowledge the support of Philippines Department of Science and Technology (DOST) Balik Scientist Program. The Balik (Filipino word for Returning) Scientist Program (BSP) seeks highly trained Filipino scientists, technologists, experts, and professionals residing abroad to return to the Philippines and transfer their expertise to the local community for the acceleration of scientific, agro-industrial and economic development of the country.
Author Contributions
All authors contributed in the conceptualization, preparation, writing, review, and editing of the manuscript. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
No ethical approval was required or obtained for the purpose of this review research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the For Women in Science (FWIS): Loreal-UNESCO Fellowship, (grant number 53349).

