Abstract
Objectives:
A significant improvement in pharmacological options is urgently required to meet the high standards of specificity and non-toxicity of modern medicines for cancer treatment. Honey is a natural product with an excellent nutritional profile and a wide range of anti-microbial, anti-oxidant, and probiotic activities that are largely used in traditional and complementary medicine. The present study aimed to evaluate the potential anti-neoplastic properties of a standardised, commercially available honey with approved dermatological and anti-inflammatory indications.
Methods:
The anti-proliferative, anti-metabolic, and anti-clonogenic activities of the honey were assessed in a panel of human and murine cell lines, including melanoma, lung, breast, colon and prostate cancer, and in spontaneously immortalised and human papilloma virus 16–transformed human keratinocyte lines. The in vitro effects of honey on intracellular reactive oxygen species generation, modulation of mitochondrial activity, cytoskeleton morphology, and anti-neoplastic activity in in vivo experimental tumours were also evaluated.
Results:
Experimental data indicated that Revamil© honey had a distinct anti-clonogenic activity, likely associated with a non-protidic, low-molecular-weight component, which was able to interact with a cell function shared by a panel of cell lines derived from tissues accounting for over 70% of the human tumour burden. This anti-clonogenic effect occurred along with a sharp induction of oxidative stress, acidification of intra-mitochondrial matrix, and severe derangement of cytoskeleton with cyto-chemical features of anoikis, and resulted in a strong suppression of cell mobility. In experimental tumours, a massive necrosis was induced.
Conclusion:
These preliminary results provide evidence that commercially available honey, registered with approved dermatological and complementary medicine indications, includes an unrecognised anti-neoplastic component potentially able to interfere with cellular mechanisms involved in the metastatic process.
Introduction
Honey is a popular natural product with an excellent nutritional profile, has numerous pharmacological activities with approved or proposed indications, and is widely used in traditional and complementary medicine. 1 Anti-oxidant, anti-inflammatory, immuno-modulating, wound-healing, bone repair, and cardiovascular potentiating effects of whole honey, or its purified components, have been demonstrated by several studies.1,2 Honey is commonly perceived as a defined nutrient with a well-characterised composition, although it is indeed a class of products whose composition in terms of nutrients and bioactive compounds is largely variable, and this also applies to uni-floral-derived honeys.3–5 Carbohydrates account for almost 99% of the total honey components and are mostly represented by fructose and glucose and a minor component by sucrose. Minor constituents are polyphenols, flavonoids, vitamins, free amino acids, minerals, peptides, and enzymes derived from the bee’s gastric apparatus.4,6 Polyphenols, generally the most abundant non-glycoside honey component, are a large family of molecules credited for most of the bioactive effects of honey, and are generally present at an average 0.05–0.08 M concentration.
A second class of potentially bioactive components are flavonoids, a plant-derived subclass of polyphenols, present, on average, at a 0.003–0.005 M concentration (Supplemental Table S1). Finally, the bee-derived glucose oxidase, through the generation of reactive oxygen species (ROS), is a key component contributing to the antimicrobial activity of honey.4,7 The bioactive properties of honey, or its purified components, are also relevant for cancer research and the number of scientific reports on their effects in cancer biology, both in vitro and in vivo, are steadily increasing.2,8,9 However, these data are rather heterogeneous and sometimes discordant or contradictory because of largely different experimental conditions, cell lines used, and variable composition of different honeys. In addition, very few reports are presently available about ability of honey to regulate the metastatic process, the ultimate, most relevant reason for cancer casualties. During metastasis, cancer cells migrate from the primary site of clonal growth, invade the local extracellular matrix (ECM), enter the blood/lymph stream, migrate through the matrix of a topographically distant and histologically different tissue, and establish a secondary neoplastic growth. This requires a coordinated and sequential activation and deactivation of a large number of specialised cell functions, including, but not limited to, adhesion, cytoskeleton remodelling, matrix digestion and deposition, and polarisation/depolarisation. The oral honey supplementation in animals, before injecting cancer cells, showed reduced number of lung metastases of colon adenocarcinoma in the rats 10 and mammary fibrosarcoma in mice, 11 but had almost no effect when administered after tumour establishment. Another study reported that intravenous administration of New Zealand honey could slightly reduce the mass of colon carcinoma experimental metastasis in the C67/BL6 mice. 12 Moreover, addition of crude honey inhibited the activity of matrix metalloproteinases (a class of enzymes involved in cell migration) in a glioblastoma multiforme cell line, although only when used at a very high, toxic concentration. 13 In the case of purified honey components, gallic acid, a polyphenol prototype, was found to reduce the migration of gastric cancer cells 14 and glioma cells 15 in wound healing and Boyden Chamber assays. Caffeic acid was found to reduce the invasiveness of PC3 prostate cancer cells by 50%, 16 and a recent study by Abel et al. 17 reported that a panel of crude commercial honeys from New Zealand and some of the purified polyphenolic components could partially reduce cell adhesion, migration, and invasion in the PC3 and DU145 prostate cancer cells. However, high concentrations of the polyphenols quercetin, gallic acid, kaempferol, chrysin, and caffeic acid were necessary to elicit only a partial effect, which was also reported for the artificially reconstituted mono- and disaccharide mixtures used as negative controls (CTRs). 17 The present study aimed to assess whether honey interferes with the metastatic process in cancer cells. To circumvent the problems of honey’s variable composition, experiments were performed with Revamil© honey, which is a commercially available, standardised medical honey with a constant analytical composition and retains edible and organoleptic properties. Based on this profile, Revamil© honey is a registered pharmacological product with several approved indications in complementary and traditional medicine, and represents a favourable starting point in the search for potential anti-neoplastic agents.
Materials and methods
Medicinal honey
Revamil© (CE0344/2B; Bfactory Health Products BV, 3911TZ Rhenen, the Netherlands), a medicinal and nutraceutical honey with multiple approved indications for wound dressing and complementary medical treatments, was used throughout the present study. The whole commercial preparation was diluted in phosphate buffered saline (PBS), sterilised by microfiltration, and stored at −80°C in 0.5 mL aliquots until use. Aliquots were prepared starting from three 18-gram tubes, lot E5, expiring in November 2019. Unless otherwise stated, honey was used at a final 1% volume/volume (v/v) dilution in complete culture medium or PBS. Any unused remnants of the thawed aliquots were discarded. In any experiment, a reconstituted sugar mixture (RSM) containing fructose 39.0%/glucose 32.5%/sucrose 0.75%/PBS was used as a negative CTR based on the average sugar concentrations of the Mediterranean honey (Supplemental Table S1). 6
Cells
A total of 10 cell lines, outlined in Table 1, are used for this study.
Cell lines’ histological derivation and phenotypic appearance.

HPV16: human papilloma virus 16.
Cell line characteristics are fully described at www.cellosaurus.org.
Accession numbers, where available, are reported in column 5. NA: not available. Doctors S Soddu, O Segatto, P Giacomini, A Venuti, A Sacchi, G Zupi, all worked/are working at the Regina Elena National Cancer Institute. Professor S Campo is retired from Glasgow University, Glasgow, UK. Professor M Dürst is presently at the Friedrich Schiller University Jena, Jena, Germany.
Briefly, six of these cell lines were derived from human invasive cancers of different histological origins, including DU145 (prostate cancer), 18 H358 (bronco-alveolar carcinoma), 19 HT-29 (colon carcinoma), 20 M14 (amelanotic melanoma), 21 Siha (human papilloma virus 16 (HPV16)-positive invasive cervical squamous carcinoma), 22 and SK-BR-3 (breast carcinoma). 23 The non-tumourigenic HK-168 cell line was derived from the human skin keratinocytes in vitro transformed with whole HPV16 genome. 24 The HaCaT cells, derived from a solar skin lesion by Petra Boukamp et al. 25 were kindly donated by Prof Matthias Dürst. The C108 and B16 cell lines are of murine origin; the former is a sub-clone of the Lewis lung mouse carcinoma originally derived in this institute by Sacchi et al., 26 and the latter is derived from a poorly melanotic melanoma. 27 All the cell lines, except C108 and HK-168, were grown in high-glucose Dulbecco’s Modified Eagle’s medium (DMEM) supplemented with 10% foetal calf serum (FCS). The C108 cells required Roswell Park Memorial Institute medium (RPMI)-1640 medium plus 10% FCS for optimal growth, whereas the HK-168 cells, which retained the ability to terminally differentiate under high Ca2+ and FCS concentrations, were grown in keratinocyte serum-free medium (K-SFM), a chemically defined medium supplemented with human epidermal growth factor (EGF) and bovine pituitary extract (Thermo Fisher Scientific, Life Technologies, Monza, MI). Each cell line was sub-cultured twice a week at the appropriate split ratio according to the specific proliferative index. Antibiotics or antimycotics were not added to the culture media in any case. For the present study, all cell lines were retrieved from an in-house cell archive facility.
Cell proliferation and metabolic activity
The cells were seeded in 96-well microplates at a density allowing exponential growth rate for the subsequent 5-day incubation and, after overnight incubation, challenged either with media containing honey at 4%, 2%, 1%, and 0.5% (v/v), or 1% RSM as a negative CTR. 28 At indicated times, the number of cells for growth curve was evaluated by staining the cells with crystal violet (CV; Sigma-Aldrich, Merck Life Science 20149, Milano, Italy) and comparing the absorbance at 540 nm (A540) of the fixed dye to a standard reference curve. The metabolic activity of the cells was evaluated by MTT assay. The cells were seeded and treated as described above. After 24 and 48 h, they were challenged with 1.25 mg/mL MTT in a 100 μL final volume of fresh medium containing 0.1% FCS. After 2 h of incubation, the amount of reduced, insoluble dye (a proxy for the cell microsomal dehydrogenase activity) was evaluated by the A540 − A750 difference. 28 The metabolic activity of the cell is expressed as percentage ratio of treated cells to that of CTR cells (treated with RSM). All conditions were evaluated in eight parallel, independent replicates. The values are shown as mean ± standard deviation (SD).
Anti-clonogenic activity
The anti-clonogenic activity was evaluated using the current standard method. The cells were seeded at ‘clonal’ density (i.e. 0.5 cell/mm2) either in medium containing 1% honey or 1% RSM, and incubated without any further manipulation. After 9–14 days, according to the cell line–specific proliferative index, the cultures were decanted and stained with 0.2% CV/methanol for 5 min at room temperature. All conditions were tested in triplicates in a six-well culture plate. The anti-clonogenic activity of the treatments was evaluated using the colony-forming units (CFU) of treated versus CTR cultures. The specific anti-clonogenic activity of whole honey was assessed by limiting dilution clonogenic assay. Briefly, the cells seeded as described above were challenged with base 2 descending honey concentration. The minimal honey concentration, which could reduce 50% of the CFU of treated versus CTR culture was defined as colony-forming inhibitory dilution (CID50), and its inverse was taken as the specific inhibitory activity of undiluted whole honey. For a quantitative evaluation of clonogenicity, a part of the experiment was replicated using the INCUCYTE S3 Live Cell Analysis Imaging System (Incucyte, Sartorius Welwyn Garden City, Hertfordshire, UK). The analysis parameters for colony count were as follows: segmentation adjustment = 0.4; hole fill <5000 µm2; adjusted pixel = −2. The objects were scored as colonies once made of at least 24 cells, that is, with an area above 24,000 µm2 and an eccentricity of <0.750.
ROS assay
The cells were plated in 96-well plates at 3.0 × 104 per well at about 50% confluence density. After overnight incubation, the medium was decanted, monolayers were washed twice with warm PBS and challenged with fresh medium containing 1.0% honey. Ten minutes before each measurement, monolayers were washed twice with PBS and 100 µL of medium containing 10 mM 2′,7′-dichlorofluorescein diacetate (DCFH-DA; Sigma-Aldrich) was added. 29 At the indicated time periods, the fluorescence of DCFH-DA–loaded cultures was evaluated using a DTX Multimode Detector plate reader (Beckman Coulter SRL Cassina de’ Pecchi, Milano, Italy) with excitation/emission filters at 485/535 nm, respectively. This procedure measures the ROS quantity generated during the 10 min of DCFH-DA incubation. For the cumulative ROS evaluation, DCFH-DA was added at the beginning of the treatment and sequential readings were obtained thereafter. ROS-oxidised fluorescent DCF accumulates within the cells and the fluorescence intensity is proportional to the total ROS generated after DCFH-DA administration. All the experiments were repeated at least three times, and each data point represented the mean of eight parallel replicates. The width of the bars represents standard deviation (SD).
Determination of intracellular pH
Intracellular pH was determined using the pH-sensitive fluorescent dye BCECF-AM (Thermo Fisher Scientific, Invitrogen, Monza, MI). 30 The cells were seeded in six-well plates (450 × 103 per well), incubated for 18–24 h and then incubated with 5 μM BCECF-AM for 20 min at 37°C. The cells were washed and incubated with different honey concentrations for 30 min at 37°C. At the end of the treatment, the cells from each of the conditions were detached with trypsin solution, divided into four aliquots, washed twice, and re-suspended in PBS or buffer containing 150 mM KCl and 5 μM nigericin (H+/K+ ionophore) at pH 6.0, 7.0, and 8.0, respectively. The fluorescence intensity (green/FL1 and red/FL3) was then recorded using BD Accuri C6Plus Cytofluorimeter (BD19 Biosciences, Becton, Dickinson and Company, Franklin Lakes, NJ, USA). The green/red fluorescence intensities from the sample aliquots at different pH were used to create a calibration curve to estimate the pH of the sample aliquot re-suspended in PBS.
Cell cycle analysis
Cell cycle analysis was performed by flow cytometry using propidium iodide (PI) staining. C108 cells were seeded in six-well plates (450 × 103 per well), grown overnight, and then differently treated for 3 and 6 h. The cells were detached using trypsin solution, washed with PBS, and fixed with 70% ethanol (added dropwise during vortex agitation). After overnight incubation at 4°C, the cells were sedimented, washed, and incubated for 30 min with 200 μL of PBS containing PI (30 μg/mL), Triton X-100 (0.1% v/v), and RNase (0.2 mg/mL) at room temperature. Fluorescence intensity of PI (FL2) was recorded using a cytofluorimeter (Accuri C6; Becton Dickinson) and cell cycle analysis was performed using a specific software for the cell cycle (Modfit LT; Verity Software House).The results were expressed as percentage of cells in each cell cycle phase according to PI intensity.
Cytoskeleton staining
C108 cells were seeded at 10,000 cells per well in eight wells, polystyrene chambers (Nunc), either in the presence of CTR or 1.0% honey. After 24 h, the media was gently aspirated and the cells were fixed with paraformaldehyde. The cells were then permeabilised with 0.5% Triton X-100 and stained with Phalloidin-iFluor 488 (Abcam Ltd, 330 Cambridge Science Park, Cambridge, UK) according to the manufacturer’s instructions, and the nuclei were counterstained with DAPI (4′,6-diamidino-2-phenylindole) dye (blue/violet). Cytochemical images were evaluated and viewed with an Olympus FV 10i-W confocal microscope (Olympus Italia Srl, 20080 Segrate, Milan, Italy).
Scratch wound-healing assay
Cell migration can be easily and precisely measured using an in vitro scratch wound-healing assay. This simple and inexpensive method combines the advantages of integrating cell adhesion, migration, and invasion in a single morphological assay that partially recapitulates the cell/ECM interaction occurring in vivo during wound healing and metastatic spread. 31 However, this cell/ECM interaction cannot be evaluated in the Boyden chamber due to the requirement of suspended cells as the starting material. To minimise the number of cells and reagents used, and thus scale up the number of replicates for a sound and quantitative evaluation, experiments were carried out in the INCUCYTE S3 Imaging System. The cells were seeded in a 96-well plate at confluence and incubated for 24 h. The confluent monolayers were then scratched with a 200-μL pipette tip, the medium was decanted and replaced with fresh medium, and cell mobility was monitored for up to 120 h under phase contrast, 4× magnification. At 12 h intervals, images were acquired and stored. The degree and velocity of the re-population of the defected area were taken as readout parameters for cell migration and tissue wound-healing capacity. 31
Animals
C57BL/6 mice were purchased from Allevamenti Plaisant S.r.l., Rome, Italy. All animal experiments were approved by the Ministry of Health, DGSAF, Rome (Approval number 212/2018-PR). The experiments were conducted at the Plaisant Facility, Castelromano, Rome, Italy (Authorisation number: 17/2015-UT), with strict compliance to the Italian National Animal Welfare laws (DL 116/92) and the European Communities Council Directives (86/609/EEC). The animals were maintained under standard conditions (12:12 light–dark cycle; temperature at 22°C; 50%−60% relative humidity) with unrestricted food and water access, and every effort was made to minimise the number of animals used and their sufferings. At the end of the experimental period, the surviving animals were sacrificed by carbon dioxide asphyxiation. A total of 44 six-week-old mice (22 males and 22 females) were used in this study. The experimental tumour was implanted in the right and left back flank by injecting 2 × 105 C108 cells re-suspended in a 100 µL volume. This cell multiplicity was shown in a preliminary experiment as the minimal inoculum that yielded a palpable experimental tumour (<2 mm in diameter) in 100% of the challenged animals within 7–8 days. For each experimental condition, a group of eight animals – four females and four males – was used. The animals were maintained under standard conditions and daily inspected for tumour appearance, scoring and sizing starting at 72 h post-inoculum. According to current recommendations, no anaesthesia was provided for this simple and low-pain-inducing procedure. A sentinel group of four untreated animals – two females and two males – was maintained parallel to the experimental groups. For the in vivo inhibition of tumour implantation, the time of tumour onset in mice inoculated with cells re-suspended in 1% RSM/PBS (group 1, CTR) was compared to that of mice inoculated with cells re-suspended in 1% honey/PBS (group 2). Regarding the anti-neoplastic activity of established tumours, starting from day 9 after the inoculation, experimental tumours were injected, intra-lesion, with 100 µL of either 50% RSM/PBS (group 3, vehicle CTR), 50% honey/PBS solution (group 4, honey tumour inhibition), or 25% honey/25% aloe vera extract/50% PBS (group 5, honey/anti-inflammatory tumour inhibition). At day 20, the animals were sacrificed, experimental tumours were collected and fixed in buffered 4% formaldehyde for 48 h, paraffin embedded, haematoxylin and eosin stained, and evaluated by an experienced pathologist. Image analysis of the histopathological slides was performed with the CS2 Aperio Digital Slide Scanner (Leica Biosystems). All the images were captured at 200× magnification, and the area of solid tumour and necrotic regions (green lines) was measured for each image.
Statistical analysis
The data for growth curves, metabolic activity, ROS generation, and pH modulation were obtained from at least three independent experiments. Each data point, for any single experiment, was the mean (±SD) of eight independent replicates. The data were analysed and plotted using Prism 6.0 software (GraphPad Software, San Diego, CA, USA). For research on animals to be approved by the Ministry of Health, DGSAF, the sample size was based on two-sided Wilcoxon–Mann–Whitney tests computed using the free software G*Power Version 3.1.9.2. In particular, the number of animals per treatment was based on means and variances from similar experiments and afforded detection of 20% differences between the experimental groups with a statistical power of 0.80, assuming a two-sided alpha of <0.05.
Results
Experimental anti-proliferative dose
To explore honey as a potential source of anti-neoplastic bioactive compounds and considering the variable and complex composition of honey of different origins, as a preliminary task, we had to set up an experimental working concentration. According to the literature, whole honey concentrations of up to 5% v/v have been commonly used in the short-term cell culture experiments.6,17,32,33 Based on these data, the maximal dose with minimal toxic effects was assessed using anti-proliferative and anti-metabolic dose–response curves generated in a panel of cancer cell lines derived from embryologically distinct organs and tissues that account for more than 70% of the cancer burden in Italy and South Europe. As shown in Supplemental Figure S1 and Figure S2, 1% whole honey concentration had almost no or negligible anti-proliferative effect, while 4% concentration invariably resulted in a complete inhibitory effect in both murine and human cell lines. Moreover, no anti-metabolic effect was observed, with 1% honey, both in C108 and B16 cells (Supplemental Figure S3). Taken together, these data indicate that 1% concentration of whole honey is devoid of unspecific anti-cellular or cytotoxic activity and can be conveniently used in in vitro tissue culture–based experiments.
Honey treatment suppresses cancer cell clonogenic activity and interferes with cell adhesion and polarisation
The anti-clonogenic activity was evaluated using the classical clonogenic efficiency assay. As shown in Figures 1 and 2, a complete suppression of clonogenic activity was consistently observed in all of the murine and human cell lines. For a quantitative evaluation, several reference experiments were replicated with the INCUCYTE S3 Live Cell Analysis Imaging System. As shown in Figure 1(c), proliferating clones were observed in CTR/untreated C108 cells within 9–12 h after seeding. The number of clones increased with a sigmoid profile reaching a plateau at 84–96 h after seeding, with a plating efficiency of about 60%. However, a complete suppression of clonal activity was observed in cells treated with 1% concentration. In the classical limiting dilution assay, the anti-clonogenic activity was evident even with 0.25% whole honey concentration, which reduced the colony number to 50%, along with a parallel reduction in the colony size (Figure 1(d)).

Honey anti-clonogenic activity on murine cell lines. Both in (a) C108 and (b) B16 cells, a 100% clonogenic inhibition activity was shown with the non-toxic, non-antiproliferative 1% v/v honey concentration (bottom rows). Top rows: Control cells (CTR) treated with 1% v/v RSM. (c) Live clonal efficiency analysis of CTR and honey-treated C108 cells. (d) Titration of honey colony-forming inhibitory activity. A 0.25% v/v dilution induced a 50% colony reduction on C-108 cells. (0.25% v/v honey = 1 CID50). A representative experiment in a set of three. Each point in (c) is the mean of four parallel replicas. Bars width represents SD.

Honey anti-clonogenic activity on human cell lines. Inhibition of clonogenic activity in M14; HT-29; Siha; H358, and DU145 human cells. CTR cells in top rows. 1% honey-treated cells in bottom rows. A complete inhibition of clonogenic activity was consistently shown in any treated cell line. HaCaT and HK-168 cells do not support growth at clonal density and were not used in this assay. Each plate corresponds to a representative experiment in a set of three.
Next, we defined the chronological window during which the cells were responsive to the anti-clonogenic effect. Time course experiments were performed with delayed treatments, that is, honey administration starting with increasing delay since plating. As shown in Figure 3, a distinct anti-clonogenic effect was observed with treatments starting up to 24 h post-seeding. Moreover, the anti-clonogenic activity was neither abolished nor partially reduced by 30 min thermal inactivation up to 100°C (Figure 3(b)).

Honey delayed treatment anti-clonogenic effect and thermal stability. (a) Honey treatment can be administered to HT-29 tissue cultures up to 24 h post plating with not even a minor reduction of anti-clonogenic activity. (b) Anticlonogenic activity of plain and heat-inactivated 1% honey dilutions. In each panel, a representative assay in a set of three. (RSM: reconstituted sugar mixture negative control; fructose 39.0%/glucose 32.5%/sucrose 0.75%/PBS).
Clonal suppression is associated with ROS generation and intracellular pH modulation
Oxidative balance and oxidative stress (OS) are major determinants of cell fate, and both pro-oxidant and anti-oxidant effects are reported for honey.34,35 In this study, we observed that honey treatment induced a sharp increase in the ROS generation in cancer cell lines (Figure 4(a)). The ROS generation was detected immediately after beginning the treatment with a peak at 1 h post treatment. This peak was pathologically meaningful as it was comparable, in height and sharpness, with the one induced by in vitro administration of 0.04 J/cm2 of ultraviolet (UV) B (a physiological daylight dose) to the human primary fibroblasts (Figure S4). One hour after the exposure, the rate of ROS generation decreased and returned to the initial level at 2–3 h post treatment. However, a second increase in ROS generation was observed starting from 4 h post treatment. This secondary increase was not followed by a compensatory activation of the anti-oxidant response as suggested by lack of the ‘two waves’ profile for UV treatment, moreover the cumulative ROS levels (Figure 4(b)) remained consistently above that of CTR cells, possibly indicating a lack of scavenging mechanisms. This elevated ROS generation was accompanied by a significant modulation of the endocellular pH modulation (Figure 5), which was clearly dose dependent and statistically significant in the case of 1% versus 5% treatments.

ROS induction by honey treatment. C108 cells were seeded in a 96-well plate and challenged with 1% honey or RSM. (a) At indicated time since honey challenging, DCFH-DA was added and the amount of endocellular ROS was evaluated by fluorimetry (excitation/emission at 485/535 nm) and expressed as relative fluorescent units (RFU). An ROS burst is evident at 1 h post challenging. The rate of ROS generation returns to control values at 2 h. Subsequently, ROS level starts to steadily increase with no restriction. (b) DCFH-DA is loaded on cells before the beginning of treatment and sequential measures were taken thereafter. The RFU level thus gives an account of the cumulative amount of ROS produced since the beginning of treatment.
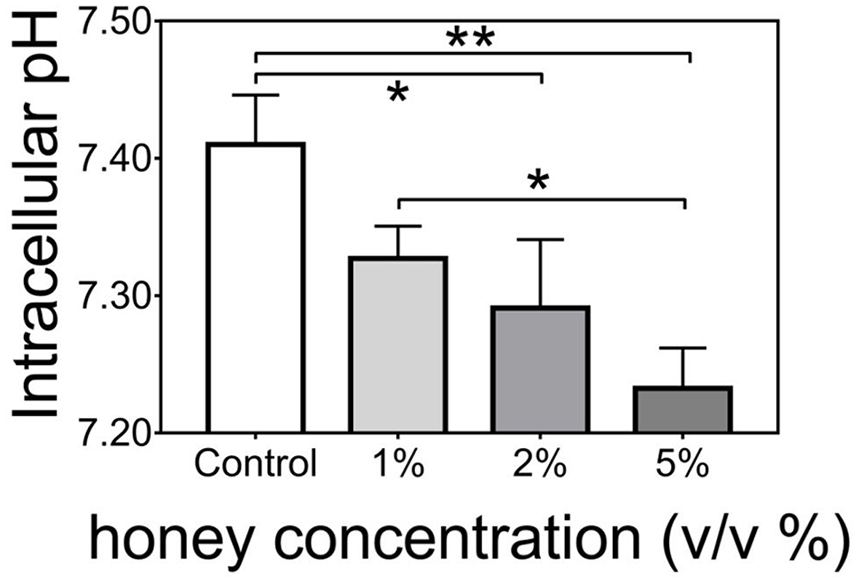
Endocellular pH modulation. Endocellular pH modulation by honey treatment. Histograms report the pH value of control C-108 cells (treated with 1% RSM) compared to cells exposed to different honey concentrations for 30′ at 37°C. A representative experiment in a set of three is reported. Each value is the mean of three parallel replicas and bars indicate standard deviation (SD). The data were analysed by analysis of variance (ANOVA) one-way, Tukey’s multiple comparisons test using GraphPad 6.0 software, significant differences are marked with asterisks (*p < 0.05; **p < 0.01).
Honey treatment induces late cell cycle arrest, cytoskeleton derangement, and inhibition of cell migration
Oxidative status and pH balance play a major role in the regulation of many fundamental cell functions. Thus, we aimed to check their association with cell cycle regulation and cell migration. As shown in Figure 6, an accumulation in G2/M phase was evident in the C108 cells 3 h post-treatment, although a rather high dose of 3% was necessary for the significant modulation, and this arrest persisted till 6 h post-treatment, suggesting the induction of a cell cycle arrest, or delay, at the G2/M checkpoint.

Honey treatment induces a late cell cycle arrest. (a) Differences in cell cycle distribution of C108 control cells or treated for 3 h (left) and 6 h (right) with honey 3% (v/v). (b) As compared to honey treatment, C108 cells were analysed after 3 and 6 h incubation in the presence of selected drugs affecting the cell cycle, such as doxorubicin (1 μM), paclitaxel (0.1 μM), and camptothecin (3 μM). A representative experiment in a set of three is reported.
The effects of paclitaxel and doxorubicin (arrest the cell cycle at the G2/M checkpoint) and camptothecin (arrest cell cycle at the S1 checkpoint) in C108 cell line are shown in Figure 6(b).
The honey treatment led to a significantly lower velocity of cell migration in the treated versus CTR cells during the 5-day end-point wound-healing assay and resulted in the coverage of a sharply smaller growth surface (Figure 7)

Inhibition of cell migration (wound-healing assay). Honey effect on wound healing assay. (a–f) CTR C-108 cell monolayers at times 0, 24, 48, 72, 96, and 108 h since scratching, respectively. (g–k) 1% honey-treated C108 monolayers at corresponding experimental times. After 108 h, the wound is almost completely repaired by control cells, whereas only one-half of scratched surface was roughly covered by treated cells. The orange colour is arbitrarily attributed by INCUCYTE live cell Imagine System to mark, in processed images, confluent counted areas.
The cell adhesion process and subsequent morphological and functional polarisation require fully efficient cytoskeleton activity. Considering that experiments with delay in starting honey suggested that honey treatment interferes with the cytoskeleton function and integrity, the cytoskeleton morphological organisation was analysed by phalloidin staining and fluorescence visualisation under UV illumination. The results obtained in a representative experiment, shown in Figure 8, indicated that, in the treated cells, the inhibition of clonogenic activity was accompanied by cytoskeleton structural changes typical of anoikis, a specialised form of programmed cell death operated by cells failing to complete the process of adhesion to the ECM and subsequent polarisation.

Cytoskeleton derangement by honey treatment. (a) CTR C108. Cytoskeleton is finely arranged around the nucleus, which presents a slightly uneven colour uptake. (b and c) C108 cell after a 24 h 1% v/v honey treatment. The cytoskeleton appears poorly stained; the fibrils are not connected with the nucleus, which, in turn, is faintly stained and fragmented.
Effect of honey on in vivo establishment and growth of experimental tumours
The effect of honey on experimental tumour implantation was evaluated by comparing the time for onset of experimental tumour by C108 cells re-suspended in 1% RSM (group 1, tumour implantation CTR) versus cells re-suspended in 1% honey (group 2, tumour implantation inhibition). The effect of honey on the established neoplastic lesions was evaluated with three additional experimental treatments with daily intra-lesional injection. The mice in group 3 (negative CTR, vehicle tumour treatment) were injected with 100 µL of PBS; mice in group 4 (honey cancer growth inhibition) were injected with a 50% honey/PBS solution; and mice in group 5 (combined honey/anti-inflammatory cancer growth inhibition) were injected with 25% honey/25% aloe vera extract/PBS. Group 6 consisted of four untreated animals (no tumour control).
In all the animals of groups 1–5, irrespective of the sex, treatment and conditions, the growth of experimental tumours became evident around day 7/8, while those in group 6 remained unaffected (Supplemental Figure S5). Group 1 (tumour control), group 2 (inhibition of tumour implantation), and groups 3, 4, and 5 showed no differences at the end-point in terms of time and prevalence of the tumour onset, appearance and mass growth; no difference was also noted between male and female animals. Interestingly, in treated animals from groups 4 and 5, manual inspection of tumours suggested considerably softer masses than those in control subjects (group 3). At the histological examination (Figure 9), treated tumours (Figure 9(e) and (f)) revealed large areas of tumour necrosis that, in a number of cases, involved more than 50% of the total tumour mass. Conversely, in the negative control tumours treated with intra-lesional PBS inoculation (Figure 9(c) and (d)), only limited signs of necrosis were observed, with an extent comparable to that in untreated tumours (Figure 9(a) and (b)).

Histological presentation of honey-treated experimental tumours. (a) Group 1, control tumour established inoculating C108 cells in PBS (mouse ID 31462); (b) group 2, tumour established inoculating C108 cells resuspended in PBS + 1% NP (mouse ID 31455); (c and d) group 3, tumours injected with 100 µL PBS per day (mice ID 33006 and ID 31471); (e) group 4, tumours injected with 100 µL 50% honey/PBS per day (mouse ID 33003); (f) group 5, tumour injected with 100 µL honey 25%/Aloe vera 25% per day (mouse ID 33008). Large colliquated areas are evident in lesions treated with honey (animals in groups 4 and 5).
Discussion
Cancer is a class of diseases with divergent, highly specific biomolecular fingerprints. To meet the high standards of specificity and non-toxicity that a therapy is required by modern medicine, a significant expansion of pharmacological options is urgently needed. Honey is largely consumed as food and extensively used for its healing effects. 36 However, highly variable composition of honey is partly the reason for the largely dissimilar, and sometimes conflicting, results that have contributed to delay its utilisation as a potential source of pharmacological products. Revamil© is a standardised, medicinal honey with a constant analytical composition approved for several dermatological and wound-dressing conditions, and thus represents a favourable starting point in the search for newer anti-neoplastic agents. To benefit from the previous experiences, which are largely obtained by experiments with whole honey preparations, and to address the synergistic effects potentially arising among its many components, we decided to work with the whole Revamil© formulation. Based on the literature review, the 1% v/v honey concentration, corresponding to weight/volume concentration of 14.1 mg/mL, emerged as a convenient non-toxic concentration for cell-based experiments.6,17,32,37,38 However, these data were derived from studies dealing with different honey preparations used under varying experimental conditions, mostly involving only one or a few cell lines. Therefore, we decided to select the concentration to be used in our work based on experimental confirmatory tests. Cell lines have greatly contributed to fundamental discoveries in any field of experimental biology, although each of them represents a highly singular biological system,39,40 and as a consequence, the results derived from a limited number of cell lines are generally of scant significance. In order to mitigate, at least in part, such an unavoidable drawback, a panel of 10 human and murine cell lines were used for performing the confirmatory tests. These cell lines are derived from histologically different organs and tissues representing the histological origin of about 70% of the total cancer incidence in Italy and South Europe. As reported in supplemental materials, the preliminary tests confirmed 1% whole honey dilution to be robustly and consistently devoid of coarse anti-proliferative and anti-metabolic effects in each cell line and were therefore used in subsequent experiments. However, this concentration completely suppressed the clonogenic activity across the complete panel of cell lines, indicating the ability of honey to interfere with cellular mechanisms of general relevance that are widely conserved across largely divergent cell lines derived from both the neuro-ectodermal and -endodermal compartments. Such an effect was observed even with a 0.25% concentration corresponding to whole honey specific activity of 400 CIDs per millilitre. In addition, data from experiments with delayed administration suggested that honey interferes with mechanisms crucial to the initial phases of colony establishment, such as cell adhesion and/or functional polarisation. Cell adhesion and polarisation also occur during cell proliferation under standard tissue culture conditions, where no or minimal inhibitory effect is observed. As a possible explanation, we hypothesise that the mechanism(s) of anti-clonal effect of honey are mostly effective in the biochemical environment of cells growing in low-density/clonal culture conditions.
Some of the pharmacological properties of honey are indeed associated with peptides and enzymes derived from the bee’s gastric apparatus. 7 However, the present anti-clonogenic activity is neither abolished nor partially reduced by a high-temperature treatment or tryptic digestion (De Marco et al., unpublished data). This stability is unlikely to be associated with peptides and enzymes, while it is strongly consistent with the profile of non-peptide, low molecular weight compounds. In honey, these are represented by polyphenols, flavonoids, vitamins, pigments, minerals, and free amino acids.5,36,38 Polyphenols are anti-oxidants and modulatory molecules, and as isolated compounds, they have been associated with many health-promoting effects in several conditions, including cancer.41,42 As a component, they are present in honey with a global roughly 8 mM concentration. 6 Flavonoids are plant’s secondary metabolic product present in honey with a total 0.5 mM concentration and reported to have a variety of immune-modulatory, anti-inflammatory, and antimicrobial functions in mammals. 6 We have not identified the exact component of honey that is responsible for inhibition of clonogenicity. However, assuming that the molecule responsible for the observed effect has the same molecular weight as catechin (the prototype of flavonoids), and that it is present at a concentration equal to median concentration of flavonoids (about 1.0 µM), and that undiluted honey has a concentration of about 400 CID50, this putative compound would be present in honey at a concentration in the range of 1–10 nM. This figure is consistent with the hypothesis that the reported effects depend on a mechanism triggered by a receptor–ligand interaction.
After confirming that the observed anti-clonogenic activity was indeed shared across a wide range of cancer cell lines, to simplify and expedite the set-up of in vivo experiments with the C57BL/6 mice strain, we focussed the subsequent characterisation in the fast growing, highly clonogenic C108 cell line. ROS generation, oxidative balance, and OS are critical factors in cell biology and play a major, double-edged function in cancer development and regulation. 43 Honey is a well-known source of free radical scavengers with sharp anti-oxidant activities 44 and is generally perceived as an anti-oxidant agent. However, oxidative balance modulates several fundamental functions of the cell. Therefore, any perturbation of this fine balance, regardless of whether oxidative or reductive, can have severe and unpredictable effects on cell homoeostasis. In this study, we observed a sharp elevation in the rate of ROS generation with a peak at 1 h post treatment. This elevation was pathologically relevant and not an artefact due to cell manipulation as its extension and height was comparable to those induced in the human primary fibroblasts exposed to UV-B irradiation, and appeared to severely damage the cell balance. Moreover, the ROS level increased at 4 h post treatment and proceeded unrestricted for the subsequent 20 h with no evidence of the ‘two waves’ metabolic response typically observed following UV treatment. The lack of such a modulated response was further observed with the cumulative ROS levels in treated cells that consistently remained above that of untreated CTR cells, possibly representing the driving mechanism for the observed clonal inhibition.
The mitochondrion is the main source of intracellular ROS generation, and mitochondrial outer membrane permeabilisation is a crucial step for induction of all forms of cell death. It is reported that tumour tissues undergo an acidic shift in the extracellular environment coupled with alkali shift in intracellular compartments. 45 Considering that the mitochondrion emerged as a central player in regulating cellular pH in the last decade, we tested whether the anti-clonogenic ROS-generating condition was associated with perturbation of mitochondrial homoeostasis; interestingly, a decrease in the mitochondrial pH was observed in treated cells than in untreated cells. This result, although obtained at 3% concentration of honey, a dose associated with a considerable anti-proliferative effect, is consistent with the data reported about purified quercetin used at equivalent concentrations. 43 This flavonoid component of honey leads to mitochondrial outer membrane alteration in different cell lines, thus supporting the hypothesis that members of the flavonoid fraction may be involved in ROS generation and mitochondrial acidification.
Deregulated cell proliferation and cell clearing are basic features of cancer and represent two main targets in cancer therapy. Many honey preparations have demonstrated induction or modulation of apoptosis and programmed cell death.46 In Wistar rats, application of honey plus the anti-inflammatory Aloe vera extract decreased the expression of the anti-apoptotic protein Bcl-2, while increasing the expression of pro-apoptotic protein Bax and stimulated the activation of caspase 9. Moreover, the cells treated with honey undergo a cytoskeleton derangement and nuclear chromatin alterations typical of anoikis, a specialised form of cell death occurring in cells failing to establish connection with the ECM. The acquisition of resistance to anoikis is an elective trait of metastatic cells, allowing them to survive the hostile blood/lymphatic environment to reach the distant permissive tissues. 47 To the best of our knowledge, this is the first study to describe the induction of anoikis in cancer cells by treatment with honey or its derivatives.2,9,38,48 Assuming the efficiency of colony formation as an in vitro indicator of resistance to anoikis, it is interesting to note that treatment with honey induced morphological and functional restoration of anoikis even in the fast-growing, highly aggressive C108 cell line, which, under standard conditions and without any need for adhesion-promoting treatments such as plastic coating with fibronectin or collagen, has a clonal efficiency index as high as 60% of plated cells. The in vitro ‘wound-healing’ test is a well-established method to probe cell detachment, protrusion dynamics, and cell adhesion functions in the integrated context of cell migration. 31 The results of wound-healing experiments showed that the treatment with Revamil could sharply suppress the movement of cells at the margin of the lesion, that is, those cells needing to detach, migrate, and repopulate the damaged area. However, cells endowed in the confluent areas, and therefore not involved in the repair process remained unaffected. This double-edged result is consistent with the apparent contraddiction of sharp clonal inhibition coupled with the lack of suppression on growth curves seeded at sub-culture density. This contraddiction can be explained by the sharply different growth and microenvironmental conditions respectively occurring at high and low cell density. There is a growing interest in honey as a potential source of anti-neoplastic agents, although very little is known about its anti-metastatic potentials at the cellular level. A recent analysis by Abel et al., 32 which indicated that three different commercial honeys, from New Zealand, and some of their purified polyphenolic components, could partially reduce the cell adhesion in PC3 and DU145 human prostate cancer cells on collagen-I (but not on fibronectin-I)-coated plastics, and also reduce the matrigel invasion by PC3 cells but not DU145 cells. However, all these effects were of limited extent, considerably variable between the three different formulations, and could be elicited only by very high honey concentrations (up to 5%) invariably associated with severe toxic effects. Conversely, here we report about a complete clonal inhibition, sharply evident in all the cell lines of both murine and human origin, all characterised by a robust plastic adhesion under standard conditions without the need for collagen-I or fibronectin-I coating treatments. Based on the in vitro effects in diverse cancer cell lines, we investigated the in vivo effects of honey in experimental tumours. As previously reported, the treatment had no effect on the in vivo tumour implantation and growth. We anticipate that this discrepancy between the in vitro and in vivo effects can be due to the rapid removal of the components injected with the tumour cells by the subcutaneous vasculature, and supply of pro-survival and pro-clonogenic factors to the tumour microenvironment that can overcome the inhibitory effects of short-term exposure to honey during the inoculum preparation. In the mice bearing C108 tumours, no clear differences were observed between the groups in terms of time of onset and tumour size. However, in both the honey-treated groups, a marked reduction in tumour consistency at palpation was associated with the presence of multiple areas of tumour necrosis. The presence of these necrotic chambers did not prevent or reduce tumour growth or improve the quality of life in the animals. Nonetheless, we interpreted these results supposing that the intra-tumoural injection, due to the compactness of the tumoural mass, allows the active honey components to persist within the tumour microenvironment for the time necessary to elicit a distinct cytotoxic effect. Moreover, this cytotoxicity was restricted to the inner part of the tumour mass where the poor blood flow and reduced metabolic exchanges extend the persistence of honey and maximise its anti-neoplastic effect.
Limitations
As for many already published reports, this study was conducted with the whole honey preparation. Thus, the reported effect could be due to a synergistic interaction of several components that are simultaneously present in honey. Second, the molecular mechanisms involved in the reported effects have not yet been identified. Therefore, further studies are needed to unravel the complex problems of in situ drug delivery to replicate the reported results in vivo.
Conclusion
The present study demonstrated that Revamil© honey can induce a sharp suppression of cell adhesion, cell migration, and clonogenic activity. These effects are absolute, consistent, and robust across a panel of 10 cancer cell lines derived from histologically divergent organs and tissues, which account for approximately 70% of the human cancer burden. Such clonal inhibition is linked to cell functions specific to the early stage of cell adhesion and functional polarisation and appears attributable to a non-protidic, thermostable, low-molecular-weight compound. This clonal suppression is accompanied, in the fast growing, highly clonogenic and tumourigenic C108 cells, by the increased production of ROS, mitochondrial acidification, and cytochemical aspects of anoikis; and thus, appears to potentially suppress the metastatic spread, the ultimate cause of death in cancer patients. Further molecular and cellular studies are underway in our laboratories to characterise the molecular mechanisms underlying these stimulating results, highlighting honey as a precious source of the yet-unrecognised, powerful biological anti-neoplastic agents.
Supplemental Material
Figure_S1_2020-04-30 – Supplemental material for A potentially anti-metastatic agent in an approved medicinal honey formulation
Supplemental material, Figure_S1_2020-04-30 for A potentially anti-metastatic agent in an approved medicinal honey formulation by Federico De Marco, MD PhD, Susanna Falcucci, Umberto Brocco MSci, Pierluigi Melissi, Andrea Venturi, Fabio Altieri, Paolo Visca and Roberto Becelli in SAGE Open Medicine
Supplemental Material
Figure_S2_2020-05-01 – Supplemental material for A potentially anti-metastatic agent in an approved medicinal honey formulation
Supplemental material, Figure_S2_2020-05-01 for A potentially anti-metastatic agent in an approved medicinal honey formulation by Federico De Marco, MD PhD, Susanna Falcucci, Umberto Brocco MSci, Pierluigi Melissi, Andrea Venturi, Fabio Altieri, Paolo Visca and Roberto Becelli in SAGE Open Medicine
Supplemental Material
Figure_S3_2020-05-08 – Supplemental material for A potentially anti-metastatic agent in an approved medicinal honey formulation
Supplemental material, Figure_S3_2020-05-08 for A potentially anti-metastatic agent in an approved medicinal honey formulation by Federico De Marco, MD PhD, Susanna Falcucci, Umberto Brocco MSci, Pierluigi Melissi, Andrea Venturi, Fabio Altieri, Paolo Visca and Roberto Becelli in SAGE Open Medicine
Supplemental Material
Figure_S4_2020-05-07 – Supplemental material for A potentially anti-metastatic agent in an approved medicinal honey formulation
Supplemental material, Figure_S4_2020-05-07 for A potentially anti-metastatic agent in an approved medicinal honey formulation by Federico De Marco, MD PhD, Susanna Falcucci, Umberto Brocco MSci, Pierluigi Melissi, Andrea Venturi, Fabio Altieri, Paolo Visca and Roberto Becelli in SAGE Open Medicine
Supplemental Material
Figure_S5_2020-05-11 – Supplemental material for A potentially anti-metastatic agent in an approved medicinal honey formulation
Supplemental material, Figure_S5_2020-05-11 for A potentially anti-metastatic agent in an approved medicinal honey formulation by Federico De Marco, MD PhD, Susanna Falcucci, Umberto Brocco MSci, Pierluigi Melissi, Andrea Venturi, Fabio Altieri, Paolo Visca and Roberto Becelli in SAGE Open Medicine
Supplemental Material
Supplemental_materials_titles_and_descriptions_2020-09-15 – Supplemental material for A potentially anti-metastatic agent in an approved medicinal honey formulation
Supplemental material, Supplemental_materials_titles_and_descriptions_2020-09-15 for A potentially anti-metastatic agent in an approved medicinal honey formulation by Federico De Marco, MD PhD, Susanna Falcucci, Umberto Brocco MSci, Pierluigi Melissi, Andrea Venturi, Fabio Altieri, Paolo Visca and Roberto Becelli in SAGE Open Medicine
Supplemental Material
Table_S1_Mean_Honey_composition_2020-05-14 – Supplemental material for A potentially anti-metastatic agent in an approved medicinal honey formulation
Supplemental material, Table_S1_Mean_Honey_composition_2020-05-14 for A potentially anti-metastatic agent in an approved medicinal honey formulation by Federico De Marco, MD PhD, Susanna Falcucci, Umberto Brocco MSci, Pierluigi Melissi, Andrea Venturi, Fabio Altieri, Paolo Visca and Roberto Becelli in SAGE Open Medicine
Footnotes
Acknowledgements
We thank Dr Graziana Palmieri, Plaisant S.r.l., for her valuable help in preparing and submitting the request for animal experimentation; and Dr Enzo Gallo, Regina Elena National Cancer Institute, for his technical assistance in histological image analysis. We thank doctors S Soddu, O Segatto, P Giacomini, A Venuti, A Sacchi, and G Zupi; and Professors S Campo and Matthias Durst for providing the cell lines.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Ministry of Health, DGSAF, Rome (approval number 212/2018-PR).
Informed consent
Informed consent was not sought for the present study because no human being has been involved in it.
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
