Abstract
The aims of this study were to determine grayanotoxin (GTX-III) toxin level in mad honey using liquid chromatography-tandem mass spectrometry and examine the dynamic changes of certain biochemical parameters in blood serum of rats that consumed mad honey. For the experimental animal study, 20 Sprague-Dawley female rats were divided into 5 groups of 4 rats each, with one group being the control group (Group 1) and the others being the experimental groups (Groups 2-5). Groups 2, 3, 4, and 5 were, respectively, given mad honey extract at doses of 0.3, 0.6, 1.2, and 2.4 mg/g body weight/day via oral gavage for 8 days. According to results, the quantity of GTX-III found in the honey sample as 39.949 ± 0.020 μg GTX-III/g honey, and the biochemical analysis of the tested parameters (aspartate aminotransferase, alanine aminotransferase, lactate dehydrogenase, alkaline phosphatase, creatine kinase, and creatine kinase muscle and brain) showed a significant elevation with increasing concentration of honey. In conclusion, the use of increasing concentrations of Rhododendron honey was seen as a source of enzymatic symptoms.
Keywords
Cardiovascular disease includes all diseases and conditions of the heart and blood vessels. There are several types of risk factors of cardiac diseases such as hypertension, hypercholesterolemia, hyperhomocysteinemia, and diabetes mellitus. 1 Some new studies emphasize that unbalanced diets and specific food poisonings also cause this health problem. 2 Especially, mad honey intoxication is the most current poisoning, caused by the consumption of honey produced from the nectar of rhododendrons. Cardiac rhythm disturbances such as bradycardia or atrioventricular block, convulsions, vomiting, sweating, blurred vision, chills, and cyanosis can occur after consuming this honey due to grayanotoxins (GTXs). 3 –5 More than 25 grayanotoxin isoforms have been isolated from the Rhododendron family. Three members (GTX-I, GTX-II, and GTD-III) of the large grayanotoxin family can be evaluated as major toxins. While GTX-I and GTX-II are found in smaller amounts, the GTX-III isoform is the principal toxin in mad honey. 6
The main symptoms of GTXs are believed to be caused by continued sodium channel activation, cell depolarization, and hence stimulation of the vagal nervous system. 2 According to preclinical researches, the voltage-gated sodium channels of the neurons are a prominent target for grayanotoxins. In general, bradycardia occurs after the administration of GTXs and consumption of its source in foods. 7
The main drawback of the routine measurement of GTX levels is their molecular complexity. High-performance liquid chromatography with ultraviolet detector failed to find any grayanotoxins because the molecules do not consist of ultraviolet-active sites. A mass spectrometer detector is necessary for this method. Although GC-MS (gas chromatography-mass spectrometry) is a technique that has been used earlier for determining the GTXs, some potential doubts may be encountered (derivatization, long analysis time, high detection limits, etc) regarding the identity of the peaks due to the short pulse width and low power level of the transmitted signal. Liquid chromatography–tandem mass spectrometry (LC-MS/MS) has become a popular technique within clinical reference and referral and drug and toxicology laboratories worldwide, and it has started to penetrate into clinical and routine analysis laboratories for biological samples. 8,9 LC-MS/MS can combine high detection sensitivity and repeatability with high analyte specificity. It seems to offer all the advantages of GC-MS without the disadvantages of compromised detection sensitivity and long chromatography run-times. 8 Consequently, direct analysis from natural and biological samples could be carried out using this technique for the immediate determination of poisoning cases; although there have been some researches on the toxicity of mad honey, there is a lack of information on the preclinical studies performed on rats given mad honey.
From this perspective, this is the first comprehensive preclinical study on the effects of mad honey on the serum biochemical parameters of rats fed with different doses of honey extract.
Materials and Methods
Animal Ethics Committee Declaration
Experimental procedures were approved by the Animal Experimentation Ethics Committee of Karadeniz Technical University Faculty of Medicine (Trabzon, Turkey). All experiments were carried out according to local guidelines for the care and use of laboratory animals and the guidelines from Directive 2010/63/EU of the European Community Council for experimental animal care.
Reagents
GTX-III standard was supplied as Grayanotoxin III Hemi (ethyl acetate) from Sigma-Aldrich (St Louis, MO). Hypergrade methanol for LC-MS and glacial acetic acid were obtained from LiChrosolv (Merck KGaA, Darmstadt, Germany). High-quality ultrapure water was supplied by Human Zeneer Navi Power I Integrate (Human Corporation, Seoul, Korea). LC syringe filters (RC-membrane, 0.2 μm) were from Sartorius Minisart RC 15, Sartorius (Darmstadt, Germany).
Honey Sample
Mad honey produced in Düzce, Turkey, and was procured from a beekeeper in 2013. The samples were placed in a refrigerator in dark plastic containers until analysis using the LC-MS/MS instrument and used in rat experiments. The sample was tagged as monofloral Rhododendron (mad) honey by melissopalynological analysis. The melissopalynological characteristics were performed following the method described by Louveaux et al. 10 Acetolyzed pollen grains were mounted on glycerin jelly and sealed with paraffin. 11 For determining the frequency classes of pollen type, 500 pollen grains were counted and classified as dominant pollen (more than 45%).
Preparation of Honey for GTX-III Analysis
Thirty milliliters of methanolic extract was prepared with approximately 5 g of honey sample in a flask attached to a condenser at 60°C for 6 hours. After adjusting the final volume and concentration, the extract was evaporated until it dried using a rotary evaporator (IKA, Werke, Wilmington, NC) at 40°C. The residues were dissolved in 10 mL distilled water and transferred to a C18 SPE (solid-phase extraction) cartridge for the enrichment of terpenoids. The SPE cartridges were conditioned using 5 mL of methanol and washed with 3 mL of distilled water. The analytes (GTXs) were eluted from C18 SPE using 3 × 3 mL methanol. Finally, the organic solvents were concentrated by a rotary evaporator with reduced pressure at 40°C and GTX-III analysis was carried out by LC/MS-MS.
LC-MS/MS Analysis
A Thermo-Scientific LC coupled with a TSQ Quantum Access Max triple-quadrupole mass spectrometer (San Jose, CA) was used for GTX-III analysis. The analytical column was a Phenomenex C-18 (15 cm × 3 mm × 5 μm; (Torrance, CA). GTX-III chromatographic separation was accomplished under isocratic conditions using a mobile phase consisting 50:50 water/methanol solution containing 1% acetic acid at a flow rate of 0.3 mL/min in 8 minutes. The MS analyses were performed using SRM (selected reaction monitoring) in the negative ion mode after describing the real molecular weight of GTX-III by full scan in the range of 200 to 500 m/z. The instrument was tuned on MS/MS mode by optimizing the response of m/z 369 as the negative ion form of m/z 370 using 1 μg/mL.
GTX-III was passed into the mobile phase at a 10 μL/min flow rate with a flash syringe. Fragmentation pattern study was applied for m/z 279, m/z 297, m/z 315 mass ions as product masses of m/z 369 12,13 (Figure 1). 12,13

Molecular structure of GTX I-II-III, LC-MS/MS chromatogram of 1 μg/mL GTX-III and product masses of m/z 369 (m/z 279, m/z 297, and m/z 315).
Animal Experimental
Twenty Sprague-Dawley female rats were obtained from the University Animal Laboratory. For experimental design, rat body weights were recorded as 200 to 250 g, and they were divided into 5 groups of 4 animals each as follows: Group 1, normal control rats were fed with standard diet; and Groups 2, 3, 4, and 5 were, respectively, given Rhododendron (mad) honey water extract at doses of 0.3, 0.6, 1.2, and 2.4 mg/g body weight/day via oral gavage for 8 days. Animals were kept at 22 to 25°C in standard rat cages in 12-hour light/12-hour dark and were continuously fed with clean water and pellet feed during the experimental process. One hour after the last dose, the animals were maintained under light ether anesthesia and decapitated. Their blood samples were collected in plastic test tubes. Serum was separated by centrifugation for 10 minutes at 4000 rpm and kept refrigerated at −20°C until used for biochemical determination.
Biochemical Analysis
Analysis was done by Cobas Integra 800 biochemical autoanalyser (Roche Diagnostics, Basel, Switzerland) by using commercial kits supplied by Roche at the Karadeniz Technical University Medicinal Faculty Laboratory. Aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), alkaline phosphatase (ALP), creatine kinase (CK), creatine kinase muscle and brain (CK-MB), and Na+ (the positively charged sodium ion), K+ (the positively charged potassium ion), and Cl− (the negatively charged chlorine ion) were examined in the serum.
Statistical Analysis
All data are expressed as mean ± standard deviation. The statistical analysis was performed using the Statistical Package for Social Sciences software (SPSS for Windows, Chicago, IL). The association of the amount of honey with AST, ALT, LDH, ALP, CK, CK-MB, Na+, K+, Cl− were analyzed by using paired t test. A P value of less than .05 was considered statistically significant.
Results
Analytical Findings
Rhododendron “mad” honey was analyzed by using LC-MS/MS. The molecular weight of GTX-III is 370 g/mol, appearing at m/z 369 in negative ion mode. GTX-III concentration was found to be 39.949 ± 0.020 μg GTX-III/g honey from Düzce city locations in the West Black Sea region of Turkey (Table 1).
Parameters of Using Mad Honey Properties.

aValue is the mean ± standard deviation of triplicate measurements.
bGTX-III values are expressed as μg of GTX-III per g honey.
Laboratory Findings
In this study, the rats that were used in experiments were decapitated by the end of the eighth day. Blood serum analysis of AST, ALT, LDH, ALP, CK, CK-MB, Na+, K+, and Cl− were measured, and the values are provided in Table 2. None of the electrolytes showed significant differences in the mean when the values of the experimental groups are compared with the values of the control group (P > .05). However, the levels of enzyme values in serum increased linearly in relation to honey dose concentration. The statistical test used was within the scope of this study, and the test results are shown in Tables 3 and 4.
Biochemical Results of Rat Blood Markers.
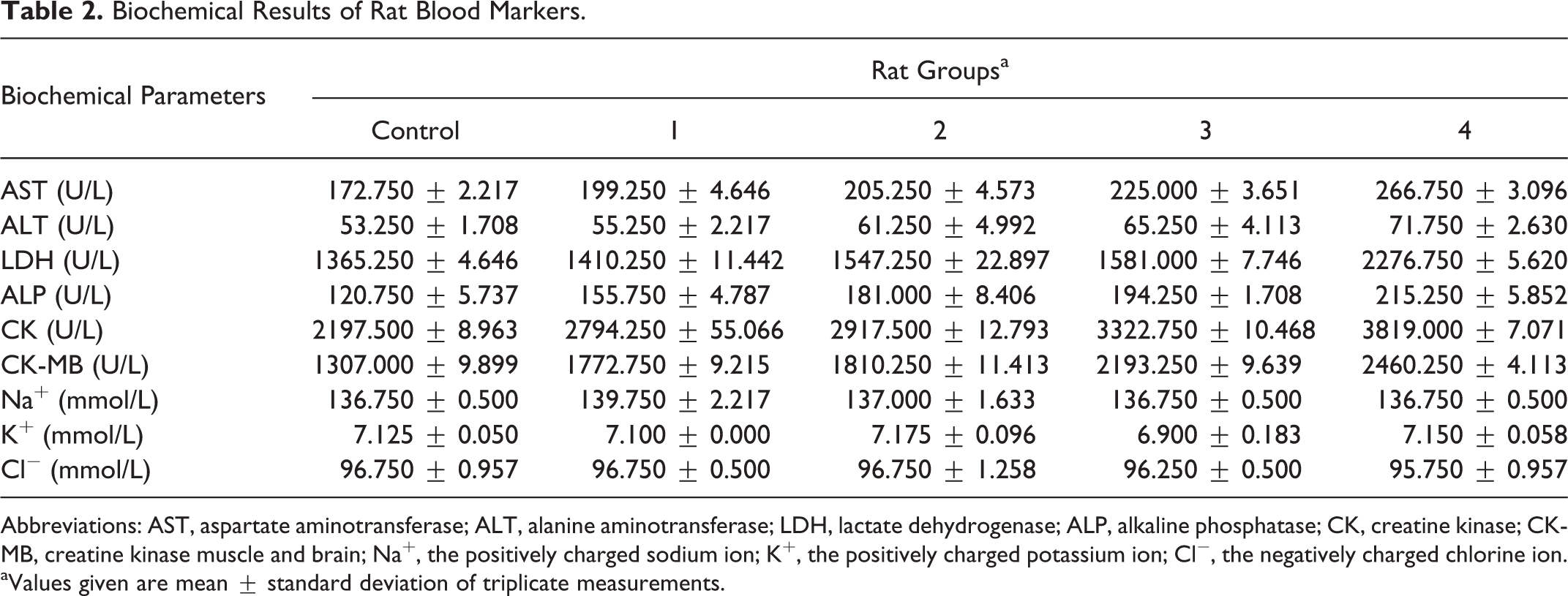
Abbreviations: AST, aspartate aminotransferase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; ALP, alkaline phosphatase; CK, creatine kinase; CK-MB, creatine kinase muscle and brain; Na+, the positively charged sodium ion; K+, the positively charged potassium ion; Cl−, the negatively charged chlorine ion.
aValues given are mean ± standard deviation of triplicate measurements.
Paired Samples t Test Correlation Coefficients.
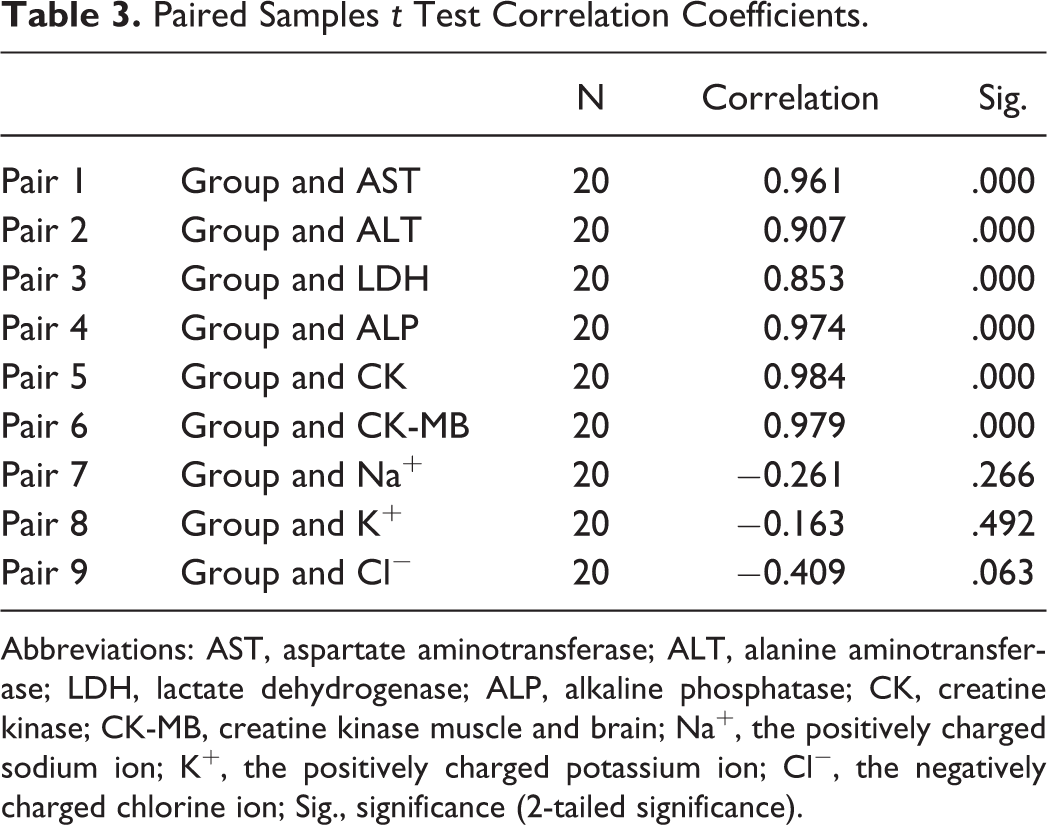
Abbreviations: AST, aspartate aminotransferase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; ALP, alkaline phosphatase; CK, creatine kinase; CK-MB, creatine kinase muscle and brain; Na+, the positively charged sodium ion; K+, the positively charged potassium ion; Cl−, the negatively charged chlorine ion; Sig., significance (2-tailed significance).
Analysis of Paired Samples t Test.

Abbreviations: SD, standard deviation; SEM, standard error of the mean; CI, confidence interval; df, degrees of freedom; Sig., significance (2-tailed significance); AST, aspartate aminotransferase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; ALP, alkaline phosphatase; CK, creatine kinase; CK-MB, creatine kinase muscle and brain; Na+, the positively charged sodium ion; K+, the positively charged potassium ion; Cl−, the negatively charged chlorine ion.
Discussion
The mad honey sample was critically important, as it has previously been reported that mad honey from the Black Sea region of Turkey causes cardiac complication such as GTX poisoning by blocking sodium channels in the cell membrane. 7
The cardiovascular system is responsible for transporting nutrients (eg, oxygen) and removing gaseous waste from the body. Providing the muscle contraction by electrical impulse on this system occurs with diffusion of cellular ions (sodium, potassium, calcium, and chloride).
The functional disorders can be seen by overdose consumption of Rhododendron honey on cardiac side effects such as bradycardia and severe hypotension. Modification of Na+ and K+-ATPase channel is also the most common cause of these disorders. The most important result of Na+/K+ modification, also called “cardiotoxicity,” can be measured by blood serum biomarker levels such as AST, ALT, LDH, CK, CK-MB, troponin T, troponin I, ANP, and BNP.
Up to now, many mad honey intoxication cases have been reported, especially cardiac effects of human and experimental animals. Definitely, grayanotoxin forms also act directly on the serum biomarker parameters generally related to cardiac enzymes. Our results of preclinical efficacy of GTXs supported previous literature, and the electrolytes levels analyzed remained unchanged.
The range of hepatocyte enzyme profiles showed an AST of 172.750 to 266.750 U/L (R 2 = 0.907, P < .05) and an ALT of 53.250 to 71.750 U/L (R 2 = 0.961, P < .05) (Tables 2-4). Cetin et al 14 reported AST and ALT levels as 1175 U/L and 479 U/L, respectively. Their finding levels are upper to approximately 40 to 100 times when compared with the normal serum levels. These higher enzyme levels have thus supported to our finding. 14 ALP has a hydrolase group and is responsible for removing phosphate groups from many types of molecules. 15 Thus, the enzyme may have triggered the release of phosphate from bone tissue with increasing muscle contraction. 16 The significant increasing was seen in the ALP activities of the experimental groups with honey dose-dependent (120.750-215.250 U/L; R 2 = 0.974, P < .05) when compared with the control group. Our expected and observed results correlated well.
In our study, in light of some scientific evidences, a simultaneous increase was seen on total CK, MB isoenzyme by consumption of honey extract in rats (Table 2). A case study of mad honey and its effects on CK and CK-MB was reported by Yaylaci et al. 17 According to these findings, after the consumption of honey, values of cardiac enzymes (total CK and CK-MB) were more than twice the normal cellular levels. This change is based on the following assumptions: increasing sodium channel permeability and activating the vagus nerve. 17,18
Another specific heart muscle cells enzyme is LDH. In our study, this enzyme range level was determined to be 1365.250 to 2276.750 U/L (R 2 = 0.853, P < .05; Tables 2-4). The reason for this difference can be explained by a possible heart attack and the condition of hypoxia. When a heart attack (myocardial infarction) has occurred, the abnormal elevation of this enzyme in the blood stream can occasionally be the marker. High level of LDH can be also seen in some case of hypoxia too. 19
Conclusion
In all analyses except blood electrolyte levels, statistical differences of increasing proportions were seen. Although there have been only a definite mechanism on cardiac toxicity by augmenting sodium channel permeability, some different mechanisms can be suggested in cellular effects of GTX due to unchanged electrolytes. Thus, different mechanisms of GTXs on cellular systems can bring out further in vitro analysis of cardiotoxicity in future studies. This study could be evaluated as a wide perspective but there were some limitations. The most important limitation of the present study is its small sample size of rats and the number of groups. Furthermore, the research results cannot be generalized to all mad honey samples. To make the results more accurate and reliable, the number of honey samples should be increased, and different localities should be studied, because the GTX contents of mad honeys can vary depending on factors such as seasonal climatic variations, season production, and the maturity of honey.
Footnotes
Author Contributions
All authors designed the study and directed its implementation, including quality assurance and control. They helped supervise the field activities and designed the study’s analytic strategy, and also helped conduct the literature review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Experimental procedures were approved by the Animal Experimentation Ethics Committee of Karadeniz Technical University Faculty of Medicine (Trabzon, Turkey) with 2012/46 approval number.
