Abstract
Symsagittifera roscoffensis is a small marine worm found in the intertidal zone of sandy beaches around the European shores of the Atlantic. S. roscoffensis is a member of the Acoelomorpha, a group of flatworms formerly classified with the Platyhelminthes, but now recognized as Xenacoelomorpha, a separate phylum of disputed affinity. We have used electron microscopy to examine the process of spermiogenesis (the final stage of spermatogenesis) in S. roscoffensis, by which spermatids form highly elongated spermatozoa. Their nuclei are long and thread-like, running most of the cell’s length, and during the process, a pair of flagella are fully incorporated into the cell body. Two previously undescribed interorganelle contact sites form at different stages of spermiogenesis. Strikingly, there is an extensive nucleus–plasma membrane contact site. Golgi-derived granules containing electron-dense filaments line up along the spermatid plasma membrane, undergo a conformational change, and donate material that forms a perinuclear layer that cements this contact site. We also show in spermatids at an earlier stage that the same granules are associated with microtubules, presumably for traffic along the elongating cell. We identify a second spermiogenesis-specific contact site where sheaths engulfing each internalizing flagellum contact the nuclear envelope.
Symsagittifera roscoffensis (von Graff 1891) (formerly Convoluta roscoffensis) is a small (5 mm long) acoel flatworm found intertidally on beaches in Europe. Adult individuals do not feed but rely on metabolites produced photosynthetically by a symbiotic alga, usually Platymonas convolutae, Tetraselmis convolutae, or Tetraselmis tetrathele (Douglas and Goodaw, 1982; Keeble and Gamble, 1907). The resulting green color is the source of their common name of mint-sauce worm. The acoel flatworms are members of the Acoelomorpha, a group once thought to be part of the Platyhelminthes (true flatworms) but shown using molecular data to be an independent group whose true affinities remain controversial (Ruiz-Trillo et al., 1999; Ruiz-Trillo et al., 2002; Telford et al., 2003; Wallberg et al., 2007; Achatz et al., 2010; Mwinyi et al., 2010; Achatz et al., 2013). It is widely accepted that the Acoelomorpha (which contains acoels such as Symsagittifera and a second clade—the Nemertodermatida) are most closely related to the Xenoturbellida (another group of marine worms once associated with the Platyhelminthes) in a phylum called the Xenacoelomorpha. The position of the Xenacoelomorpha relative to other animal phyla is also controversial. Some molecular phylogenies place them as the sister-group to all other bilaterian phyla. This early emergence makes sense of the simple body plan of these worms. Other molecular studies conclude, however, that Xenacoelomorpha are related to the Ambulacraria (echinoderms and hemichordates) and that their morphological simplicity is a result of evolution by loss of characters that were present in a more complex ancestor.
Study of spermatozoa in invertebrates and in the acoels in particular has proven useful for comparative phylogenetic analysis (Bedini and Papi, 1969; Hendelberg, 1969, 1977, 1983, 1986; Rieger et al., 1991; Raikova and Justine, 1994; Raikova et al., 1997; Justine et al., 1998; Littlewood et al., 1999; Hooge and Tyler, 2001; Raikova et al., 2001; Raikova, 2002; Petrov et al., 2004; Tekle et al., 2007; Achatz et al., 2010; Shafigullina and Zabotin, 2017). Like many acoels, S. roscoffensis produce long, thread-like (filiform), spermatozoa which develop according to a process of flagellar incorporation (Hendelberg, 1974; Henley, 1974; Hendelberg, 1983; see schematics in Figures 1 and 2). These sperm swim via undulations of a long tail containing two internalized, extended flagella and have undergone a polarity inversion, propelling the cell tail-first as compared to the head-first charge of archetypal primitive forms. The mid-section of the typical acoel sperm contains many mitochondria and large granules and is supported by an axial axoneme bundle of dense-cored, atypical microtubules forming a so-called keel. These characteristics are believed to reflect the peculiarities of acoel fertilization; the sperm often being introduced hypodermically into the body of the mate, penetrating between viscous cellular tissues as opposed to swimming freely in sea water as is the case with external fertilization. Although flagellar incorporation has been observed in numerous studies, a detailed mechanism of this process has not been proposed. As acoel sperm swim tail first, it has also been assumed by most that they do not have an acrosome, the large, granular vesicle that usually covers the crown of the nucleus in more familiar, primitive sperm that contact the egg head on and which facilitates fertilization with the egg. These adaptions have called into question interpretation of them as representing the form of the ancestral bilateran.

Schematic: Early Stages of Spermiogenesis. A: The products of meiosis remain closely associated by a cytophore. B and C: (Speculative) The basal bodies of the developing spermatocyte may have been separate or conjoined. They capture the nucleus; presumably via astral microtubules which wrap around the nuclear envelope (these giving rise to the manchette). D and E: The conjoined basal bodies and the internal elements of the flagella align with the elongating nucleus. The flagella begin to be assimilated into the cytoplasm of the spermatocyte, either by advancement of the cytoplasm over them, or by relative movements of the nucleus-associated axonemes and the cytoplasm. A multilaminate electron-dense complex and associated membrane contact site forms as the sheath develops, limiting a cytoplasmic canal, engulfing the flagella and linking them to the nucleus. E: The nucleus continues to elongate, as does the spermatocyte. The astral/manchette basal bodies remodel into the axial, atypical axoneme keel, elongating and pushing the basal bodies away from the nucleus.

Schematic: Later Stages of Spermiogenesis. F: dense granules associate with the medial keel axoneme (a circular arrangement of atypical microtubules) and with the manchette (nuclear associated microtubules). These two elements are probably contiguous with one another. G: The medial keel becomes two staves; the dense granules lose their association with it and line up with the degenerating sheath and associated plasma membrane. The dense granules realign their filamentous contents. H: The developing spermatozoan continues to elongate, the nuclear envelope becomes more elaborate as it merges with the dense granule membranes. Ultimately the filaments of the condensed granules will form a nuclear envelope/plasma membrane contact site.
In this article, we use transmission electron microscopy to examine spermiogenesis in S. roscoffensis. We identify previously overlooked aspects of the process, including the formation of unusual contact sites between membranes in the developing spermatozoon. Contact sites are specialized regions of organelle membranes that form close (usually less than 30 nm, though there are exceptions) associations with one another. These sites seem to constitute dynamic mechanical tethers, holding the two organelles in position with respect to one another while organelle remodelling (structural and/or biochemical) occurs. They may be the site of transfer of ions, complex metabolites or lipids, and act as scaffolds for the recruitment of molecular machines required to act at the site of membrane–membrane association (Prinz, 2014; Gatta and Levine, 2016). These contacts have been identified between nearly all organelles in most combinations. The ones we identify in this study occur during maturation of the complex nuclear envelope and the process of flagella incorporation and give clues to mechanistic elements of spermiogenesis. This is the first description of an elongated nuclear envelope–plasma membrane contact site.
We have also examined in detail the axial axoneme, composed of electron-dense atypical microtubules, which appears to facilitate traffic of dense core granules that donate material for nuclear envelope–plasma membrane contact site formation.
Materials and Methods
Specimens
Adult Symsagittifera roscoffensis were collected during low tide on the beach at Carantec (Brittany, France) in 2014. The animals were kept in plastic boxes with artificial seawater in incubators at 15°C with a 12-h light/12-h dark-light cycle.
Transmission Electron Microscopy
Adult specimens were fixed for two hours with cold (4°C) Karnovsky’s fixative (2% paraformaldehyde, 2.5% glutaraldehyde in 0.08 M cacodylate buffer). They were washed 3 times in phosphate buffer and osmicated with 1% osmium tetroxide in ddH2O for 1 hour. Samples were then washed 3 × 10 minutes in ddH2O and dehydrated with a series of ethanol dilutions: 30%, 50%, 70%, 90%, 3 × 100%, and 2 × propylene oxide (at least 20 minutes in each). They were infiltrated with 50:50 propylene oxide:araldite resin overnight and with several changes of 100% resin the next day. Blocks were cured at 60°C overnight. Sectioning was performed using a Leica Ultracut UCT microtome. Sections were counter-stained with Reynold’s lead citrate and were viewed on a JEOL 1010 TEM (JEOL USA, MA, USA).
Preparation to Enhance Contrast in Membranes
Specimens were fixed in Karnovsky’s EM fixative (as earlier) for 30 minutes (after Walton, 1979). They were washed 3 times with phosphate buffer. Samples were then incubated for 2 hours in 2% osmium tetroxide/1.5% ferricyanide and then washed 3 × 5 minutes in water. The samples were then placed in 1% thiocarbohydrazide solution for 10 minutes then washed 3 × 5 minutes in water. A second round of osmication was performed (2% osmium tetroxide (no ferricyanide) for 30 minutes) and the tissue washed again 3 × 5 minutes in water. The samples were then placed in aqueous 2% uranyl acetate overnight at 4 degrees.
The following day samples were placed in freshly made Walton’s lead aspartate (pH 5.5) at 60°C for 30 minutes and then washed 3 × 5 minutes in water. Samples were then dehydrated through ethanol (30%, 50%, 70%, 90%) to 100% × 3 then placed in acetone 2 × 20 minutes. Samples were infiltrated with Durcapan resin 25% plus acetone, then 50%, 75% (2 hours each) and 100% overnight. The following day the resin was exchanged, and the blocks hardened in an oven for 48 hours.
Results
Our general observations of spermiogenesis in Symsagittifera roscoffensis are in line with those of others who have used electron microscopy to examine related species: Convoluta psammophila (Bedini and Papi, 1969^), Convoluta boyeri, and Convoluta philippinensis (Boyer and Smith, 1982; Bush, 1984); Paratomella rubra (Raikova et al., 1997); Convoluta saliensis (Raikova and Justine, 1999); Pseudaphanostema smithrii, Praeconvoluta tigrina, Convoluta pulchra, and Stromatricha hochbergi (Petrov et al., 2004). We observed multiple testes in various stages of development, each containing mature sperm and developing spermatocytes clustered around a cytophore, a residual domain of cytoplasm formed during meiosis that presumably serves as a communal source of biosynthesis (Figures 1A and 3A). Individual spermatocytes at various stages of development and mature sperm were found in the same TEM sections in various orientations.

Early Stages of Spermiogenesis. A: A primary spermatocyte containing an elongated, curved nucleus (black arrow). The cytoplasm is full of large granules. B: A close-up of a similar spermatocyte nucleus cut twice as it passes through the section. The manchette of single microtubules is clearly visible surrounding the nucleus. C: A montage image showing a later stage in spermiogenesis. Several spermatocytes (sp) are seen arranged around a common cytophore (c). The nuclei are elongated and the partially internalized flagella are intimately associated with them. D: A different orientation showing the cytophore containing large numbers of Golgi (G) and developing large granules and an elongating spermatocyte. At this stage the manchette of microtubules surrounding the nucleus (n) becomes associated with huge numbers of dense granules (dg) (see inset).
Changes in the Nucleus During Spermatogenesis
Early stage spermatocytes clustered around the common cytoplasmic cytophore contain an ovoid nucleus (Figure 3A), each surrounded by a complex manchette of single microtubules that enclose the nucleus in a cage-like structure (Figures 1C and 3B). The nuclear envelope appears highly fragmented and is perforated by large gaps. Each of these fragments is associated with a single manchette microtubule. Approximately 50% of the nuclear material appears to be exposed to the cytosol (see also Figure 4A and E).

Flagella Axoneme Association With the Nucleus. A: longitudinal section through a primary spermatocyte. Internalized flagellum to the left (FL1) showing the sheath of plasma membranes (Sh and white arrow). A single microtubule (Mi and black arrow) lies in the space between the flagellum and the NE. The noninternalized flagellum (FL2) to the right is associated with the nucleus via with a ring of microtubules of which two are observable in this section here (Mi black arrows). B: Transverse section of a different primary spermatocyte showing the same features identified above. C: A close-up from another specimen showing the dual membranes of the cytoplasmic canal/sheath (Sh) grey arrow of the internalized flagellum. The nucleus (N) is surrounded by a highly porous nuclear envelope. D and E: Non-internalized flagellum associated with the NE of the nucleus (N) by a ring of microtubules (black arrows). NE = nuclear envelope.
As spermiogenesis progresses, the nucleus dramatically elongates and the cytoplasmic content of the spermatocyte reorganizes; a complex rearrangement driven by internalization of the two flagella and elongation of an atypical microtubule structure called a keel (described in detail later).
The fragmented nuclear envelope of the spermatozoon becomes more elaborate, all gaps between fragments are closed and florid extensions of the membrane develop. These proliferations hold the now very long, filiform nucleus suspended through the centre of the sperm cell by connections to the two opposing faces of the plasma membrane (Figure 5A). This led us to wonder where the additional membrane came from and how the nucleus was able to form an attachment with the plasma membrane at these sites
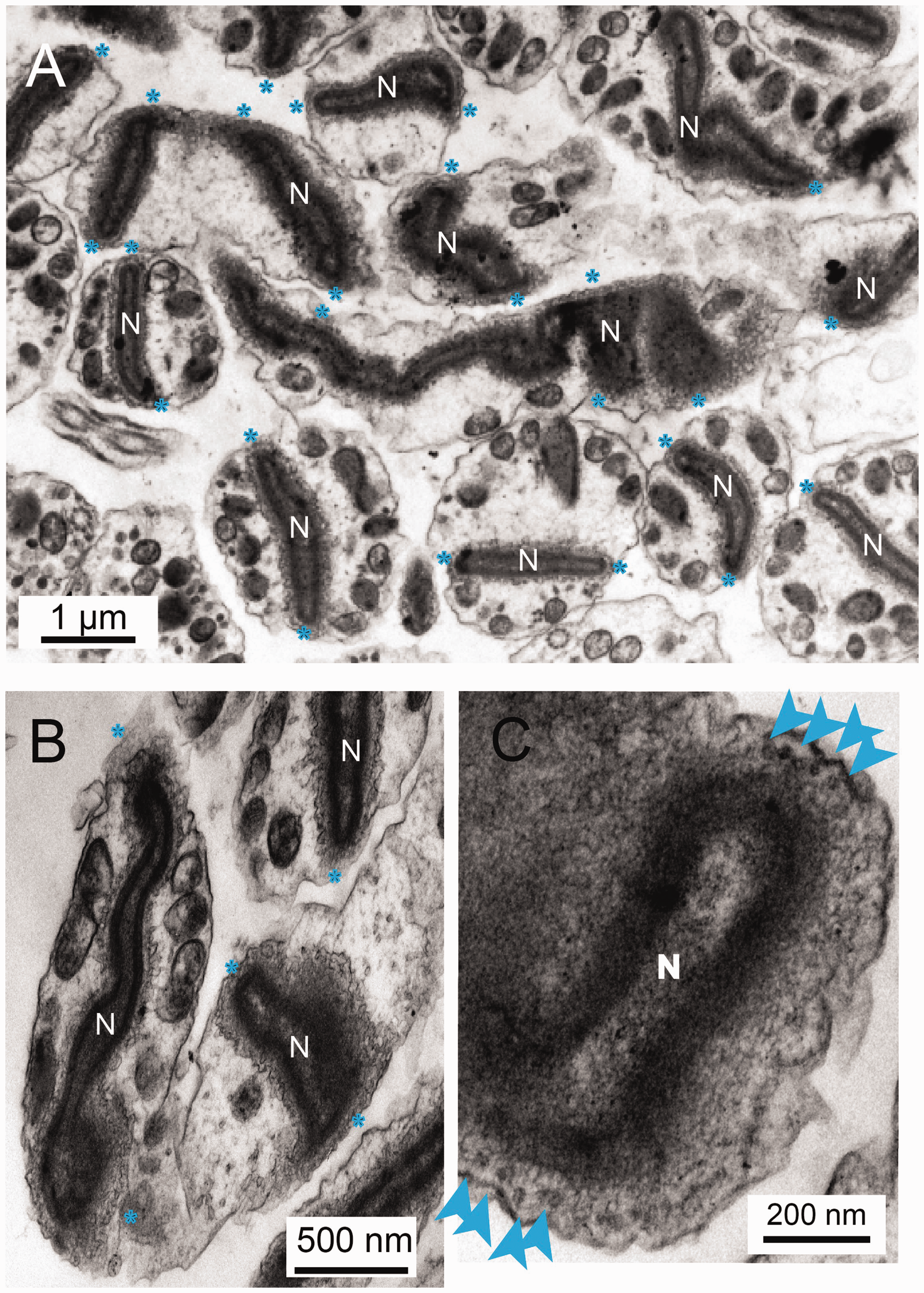
The Nuclear Envelope Retains Elements of the Fusion Events. A: In the mature spermatozoan the nucleus (N) appears suspended in the middle of the cytoplasm as a result of a number of contacts with the plasma membrane (blue stars (*)). B: More examples showing the complex elaborations of the membranes. C: Contact sites are characterized by (20 nm) rings of electron dense puncta (blue arrows).
Dense Granules Give Rise to a Nuclear Envelope–Plasma Membrane Contact Site
Formation and Redistribution of Dense Granules
Large numbers of small (50 nm) dense granules (DGs) were observed surrounding Golgi apparatuses in primary cytophores and primary spermatocytes (Supplemental Figure 1). These DGs have a characteristic structure, being flattened discs with a thickened circumferential rim. In the immediate vicinity of the Golgi we observed vesicles showing internal structures of varying degrees of condensation; some appearing as punctae, others as fibers and yet others as dense aggregates. We interpret these as immature DGs in the process of condensation, their contents, upon aggregation or polymerization, ultimately imposing structure on the mature discs.
At intermediate stages of spermiogenesis the DGs are arrayed in a spiral pattern around the medial keel axoneme (Figures 1F and 6A), to which they are attached by fibrous tethers. The medial keel axoneme may be continuous with the nuclear manchette microtubules at this stage (as indicated schematically in Figures 1 and 2). We do not know if DGs are transported down this structure or merely stored here at this stage of spermiogenesis. Later in spermiogenesis the medial keel reorganizes (Supplemental Figure 2C and F seen in transverse section, Supplemental Figure 2G in longitudinal section, Figure 6A and B), the DGs dissociate from them, and most are redistributed adjacent to the plasma membrane in the immediate vicinity of the lateral axonemes (Figure 6C and Table 1).

Movement of Dense Granules From the Medial to the Cortical Axonemes. A: In immature spermatids, the DGs are arranged spirally (see inset) around the circularly bundled, axial axoneme. B: Later in development the axoneme unfolds into two linear structures (a microtubular stave: see also inset). The DGs leave the axoneme and become associated with the cortical plasma membrane. C: In the vicinity of the cortical axoneme, the DGs (black arrows) become aligned to the plasma membrane, their electron-dense contents perpendicular to the membrane surface.
Arrangement and Distribution of Dense Granules on Membranes.

Dense Granules Form Contact Sites With the Plasma Membrane
Plasma membrane-associated DGs appear to be in a state of abeyant fusion (Figure 6C) with their internal filaments arranged perpendicular to the plasma membrane.
DGs associated with the plasma membrane have a narrow neck region when viewed from the side and a dense circular connection to the plasma membrane. Between 10 and 18 filaments, around 3 nm in diameter are packed hexagonally, emanating from a point close to the plasma membrane then running parallel before coming together again (Figure 7). This structure appears to be cross-linked by fine lateral filaments. The DG membrane, initially circular in outline ( Supplemental Figure 3A and B), becomes progressively more undulating as spermatid maturation progresses (Figure 7A and F) and the 3 nm internal filaments crowd together in the center. Later still, the filaments condense further, becoming shorter and the membrane expands to become flower-shaped (Figure 7E and F and Supplemental Figure 3C and D ).

Schematic: Demonstrating the Condensation of the Dense Granules into the Multicomponent Nucleus–Plasma Membrane Contact Site. A: A dense granule aligned with the plasma membrane observed perpendicular to the plasma membrane showing the internal filaments (blue). B: A granule at a similar stage of fusion sectioned transversely showing arrangement of an additional layer of internal filaments (purple) inside the outer layer (blue). Radiating filaments which reach out to the membrane are also clearly seen. C: A slightly later stage: the outer membrane is crenelated and internal filaments shorten and aggregate. D: A granule cut transversely at an early stage of fusion showing the internal filaments equidistantly spaced. Very thin horizontal connections can be seen interconnecting the filaments. Presumably these retain the structure’s geometry. E: Later the filaments coalesce and are bundled asymmetrically in the granule. F: A late-stage granule cut transversely. The internal filaments condense to dense puncta. In this example the radiating fine filaments are quite prominent. The schematic at the bottom of the figure is a graphic representation of where these sections have been taken.
Assimilation of Dense Granules Into the Nuclear Envelope and Formation of a Plasma Membrane–Nucleus Contact Site
As the elongating nucleus passes by the DGs it appears that it establishes a contact site with them, possibly fusing and incorporating their membrane into that of the nuclear envelope ruffles. The DG membrane is slightly more osmophilic (appearing more electron dense) than that of other regions of the nuclear envelope, and in regions where fusion seems to be actively occurring, remnants of DGs are visible ( Supplemental Figure 1E and F ). In many sections, we could identify intermediates in the process of fusion and we also observe rings of electron-dense particles we interpret as the remnants of the DG dense fibers ( Supplemental Figure 4A and insets). This process was easier to observe in samples processed to enhance membrane staining ( Supplemental Figure 5 ). This entire process is summarized schematically in Figure 8.

Proposed Model of Nuclear Migration Leading to Incorporation of Dense Granules Into the Elaborating Nuclear Envelope (N = nucleus, A = axoneme, M = mitochondrion, dg = dense granule, pm = plasma membrane).
Less frequently, there were larger, more complex, focal accumulations of material within nucleus–plasma membrane contacts. These varied in number between specimens, so we conclude they represent a transitory state in spermiogenesis ( Supplemental Figure 4B and F ).
Microtubule Rearrangements Associated With Spermiogenesis
We did not see many examples of centrioles in the cytoplasm of early stage spermatocytes, but it has been suggested from related species that they migrate from near the plasma membrane towards the nucleus (Raikova and Justine, 1999).
As the spermatocyte extends and the nucleus elongates. The flagella are simultaneously incorporated into the cytosol of the spermatocyte progressing in a disto-proximal mode (Figures 1D and E and 3C).
The resultant spermatozoon has a long, thread-like (filiform) nucleus which, by moving distally as the spermatid matures, but not passing between the centrioles of the developing tail, comes to lie in front of the incorporated binary flagella (Figure 2).
Sections through the elongating spermatocyte at different stages reveal that both flagellar axonemes come to be associated with the elongating nucleus. Flagellar incorporation is asymmetrical. At early stages in spermiogenesis one axoneme is predominantly free in the cytosol while the other is entirely surrounded by a cytoplasmic canal or sheath, a ∼20 nm luminal void within the cytoplasm generated as the flagellum is drawn into the cytoplasm (Figure 4A). The internalized flagellar sheath appears to be held close to the nucleus by association with a single microtubule (Figure 4A to C) which runs between it and the nuclear envelope. The cytoplasmic free axoneme is also associated with the nucleus but in this case by a ring of approximately 12 satellite single microtubules which surround it (Figure 4D and E).
As spermiogenesis proceeds, the remaining external projections of both flagella are drawn into flagellar sheaths. These internalized extracellular domains progressively reduce in volume to form narrow canals that no-longer encircle the entire flagellum. The result is that ultimately both the flagella axonemes are effectively free in the cytosol and flagellar incorporation is complete. The flagella migrate past the nucleus and are fully incorporated as lateral axonemes, 9 + 0 rings of microtubule doublets on opposite sides of the sperm (Figure 1A). They are no-longer associated with the nucleus, which has moved beyond them. The nucleus elongates, becoming flattened and condensed (Figures 2 and 3D).
Down the centre of the spermatozoon is a so-called medial keel, a ring of 10 microtubule-like rods. These do not show the typical doublet ring of microtubules of conventional axonemes or appear as single microtubules. They are larger than the latter, each measuring 26.7 ± 3.2 nm compared to 21.2 ± 2.9 nm for conventional microtubules (n = 100, p ≪ .01). These rods have no internal cavity appearing in the very clearest, transverse sections to be spirally-arranged bundles of protein units rather than rings (data not shown). We refer to these as dense core atypical microtubules. They are separated one from the other at a distance of 43.5 ± 4.6 nm (n = 60) (when either a circular ring of 10 or in two rows of 5). In some cases a few thin linear connections can be identified between them (data not shown) suggesting they are held together by proteinaceous tethers. It is likely that this structure represents an elongated centriolar-derivative.
Flagellar Incorporation: Another Nuclear Envelope–Plasma Membrane Contact Site
At the point of entry of the internalized flagella into the developing spermatocyte, a multilaminate, electron-dense complex (MEC) forms between the flagellar sheath and the nucleus (Figure 9A). This structure maintains an intimate association of the flagella with the nucleus as nuclear elongation and flagellar incorporation proceeds (Figure 9B to E, schematic Figure 1D and E). The juncture of each internalized flagellum and the nucleus is marked by an electron-dense membrane contact site (Figure 9D) where the nuclear envelope appears to be fixed to the MEC and associated internalized plasma membrane.
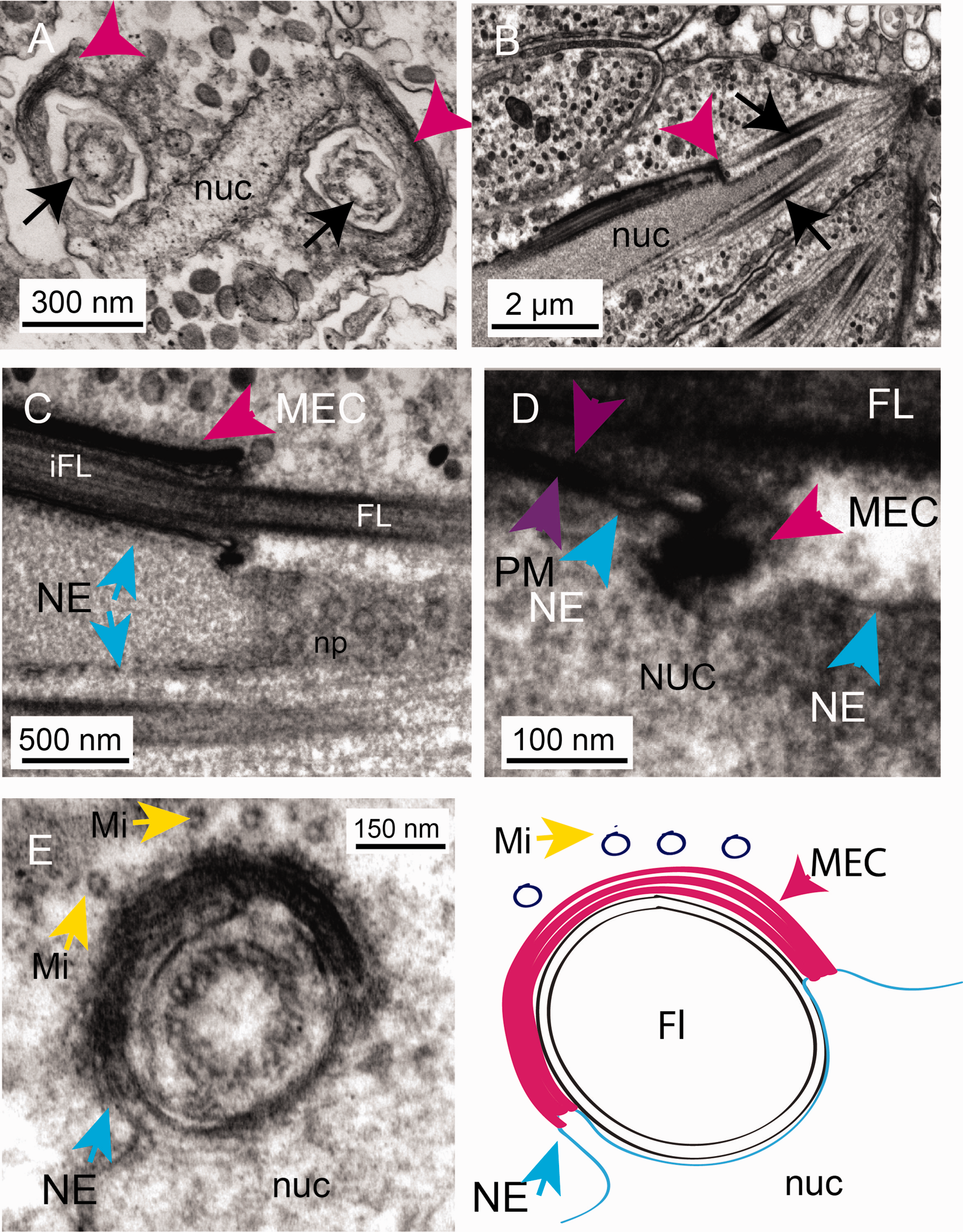
A Multilaminate, Electron-Dense Complex (MEC; a Nucleus–Plasma Membrane Contact Site). A: A transverse section of the tip of the developing spermatocyte showing the flattened nucleus (nuc) and development of a multilaminate, electron-dense complex (MEC; pink arrowheads) that forms as the flagella (black arrows) are drawn into the developing flagellar sheath. B: A transverse section showing the internalising flagella (black arrows) and the association of the MEC (pink arrowhead) with the nuclear envelope (blue arrows). C: The MEC is intimately associated with the nuclear envelope (note the nuclear pores (np) seen in this glancing section across the nucleus). D: A close up of this region showing the contact site between the sheath (composed of invaginated plasma membrane [purple arrowheads] and the nuclear envelope [blue arrowheads]). E: Note the drawing in of the nuclear envelope (NE and blue arrow) into the MEC. There are microtubules associated with the MEC complex at this stage (Mi yellow arrows). E: Schematic to show the arrangement of membranes at the nuclear envelope/sheath contact site mediated by the MEC. nuc = nucleus; NE = nuclear envelope; Mi = microtubule; FL = flagellum; iFL = internalized flagellum; MEC = multilamellar electron-dense complex.
An MEC-like object has also been observed in electron micrographs of spermatocytes of Symsagittifera bifoveolata and Symsagittifera schulzei (Raikova et al., 2001). This is presumably a temporary structure as it is not seen later in spermatogenesis.
Discussion
Spermatogenesis in the acoel Symsagittifera roscoffensis appears to occur in a manner broadly similar to that seen in related species (Figures 1 and 2) (Bedini and Papi, 1969; Hendelberg, 1969; Hendelberg, 1977; Boyer and Smith, 1982; Bush, 1984; Raikova and Justine, 1994; Raikova et al., 1997; Justine et al., 1998; Littlewood et al., 1999). Nuclear elongation and spermatid elongation and internalization of the flagella (to form lateral axonemes) occur in tandem. We describe many aspects of this process in detail, focusing on the relationship between the nucleus and adjacent membranes: the plasma membrane, the sheaths of internalized flagella and the origin and fate of DGs containing filamentous material.
Dense Granules and Formation of a Nucleus–Plasma Membrane Contact Site
Structurally complex, DGs arise in the Golgi and accumulate on the medial keel axoneme of the spermatids and on the manchette microtubules of the elongating nucleus. DGs of various morphologies have been observed in other, related acoels (Hendelberg, 1974, 1977; Raikova et al., 1997; Petrov et al., 2004). As spermiogenesis proceeds, the medial keel axoneme reorganizes and the circle of 10 breaks into two lines or semi-circles of 5. The DGs relocate to a narrow domain of the plasma membrane in the immediate vicinity of internalized lateral (flagellar) axonemes. At this site they form a series of intimate membrane contact sites in which the DG membrane is directly opposed to the plasma membrane. The DGs do not form direct physical interactions with the lateral microtubules themselves, suggesting that the membrane in the vicinity of the lateral axonemes is in some way different from that covering the rest of the spermatozoon. At this site they undergo a dramatic structural reorganization; their internal electron dense filaments (initially held in a geometric pattern by filamentous cross fibers) align perpendicular to the plasma membrane before condensing into an electron-dense core with spider’s-web like projections reaching out to the vesicular membrane.
The structural changes we see are reminiscent of the conformational changes observed in filaments during the acrosomal reaction (acrosome exocytosis) observed in other phyla during sperm maturation and fertilization. Acrosomal fusion usually occurs between the closely apposed outer acrosomal membrane (OAM) and the plasma membrane. This results in the controlled release of the soluble protease components of the acrosome while an insoluble pseudo-crystalline matrix, recently shown to be an amyloid (Hardy et al., 1991; Olson and Winfrey, 1994; Kim et al., 2001; Kim and Gerton, 2003; Guyonnet et al., 2014) is retained. Such amyloids (self-assembled complexes of beta-sheets) are resistant to the proteolysis characteristic of the activated acrosome environment, and potentially allow a regulated release of contents (Hardy et al., 1991; Kim et al., 2001; Buffone et al., 2008; Guyonnet et al., 2014). Other authors noting the presence of granules in acoel spermatozoa have suggested that they might be components of an acrosome (Hendelberg, 1969, 1977, 1986), although it is generally accepted that, with the exception of putative vesicles at the very tip of the sperm of some Nemertodermatida which have monoflagellate sperm with a large head, acrosomes are missing from acoel sperm. Our results reveal the possibility that in some acoels the acrosome has become modified into an extended nucleus plasma-membrane contact site. It would be interesting to ascertain if any acrosome-like biochemical properties were retained.
Contacts Between the Nucleus and Other Organelles
We have shown that during their incorporation, the flagella migrate into the cystosol and initially come to lie parallel to the nucleus before eventually losing their surrounding membranes and becoming fully incorporated as lateral axonemes. We have identified structures that might provide a mechanism for this. The free flagella represent an axoneme surrounded by a thin layer of cytoplasm and a flagella-associated plasma membrane. If formation of the flagellar sheath involved them being drawn into the cytoplasm rather than merely fusing laterally with the spermatocyte, one might imagine a mechanism by which the plasma membrane becomes associated with the flagellum at some form of collar as it is pulled in, otherwise the surface of the spermatocyte would flow over the flagellar axoneme and there would be no cytoplasmic canal formed. We suggest that the MEC we observe surrounding the plasma membrane of the internalising flagellum is this structure (Figure 9). It is well placed both to initiate and maintain the canal/sheath as it is drawn in. This structure is also associated with a novel membrane contact site at the junction between the base of this cytoplasmic canal and the nuclear envelope, physically linking the flagella to the nucleus. This would allow the processes of nuclear elongation and that of flagellar incorporation to proceed in unison.
A combination of both lateral fusion of flagella and subsequent formation of cytoplasmic canals (sheaths) has been suggested to give rise to the complex set of membranes observed during spermiogenesis around the internalized flagella in the closely related acoel P. rubra (Raikova et al., 1997). Although there are significant differences between S. roscoffensis and P. rubra in terms of the final structure of the spermatozoon; these authors also identified the intimate association of the internalized flagella with the nucleus during spermiogenesis (this persists in the final spermatozoon in the latter case). The authors identify a gap in the nuclear envelope between the nucleus and the internalized flagella, and, as we see in S. roscoffensis, they identify microtubules in close proximity to these junctures. They do not identify an MEC or a contact site between the flagella and the nucleus, but it is possible that this is short-lived and could have been overlooked. This underlines how certain elements of the process of spermiogenesis are common to acoels, though others may be lost or modified.
Conclusions
We have examined spermiogenesis in the acoel Symsagittifera roscoffensis. We identify the MEC as an extended membrane contact site that maintains a close linkage between the nuclear envelope and the plasma membrane-derived flagellar sheath as the developing spermatozoan undergoes structural reorganization. We follow rearrangements of atypical dense-cored microtubules which form the axial keel characteristic of convolute, aberrant sperm. We describe how thousands of Golgi-derived DGs initially associate with the axial keel but then migrate and by means of membrane contacts between themselves, a subdomain of the plasma membrane and the nuclear envelope, act to rivet the nucleus to the plasma membrane. We believe these to be the first descriptions of extended nucleus–plasma membrane contact sites.
Summary
During spermiogenesis in the acoel flatworm Symsagittifera roscoffensis, two previously unidentified contact sites contribute to the structure of the mature spermatozoon.
Supplemental Material
sj-jpg-1-ctc-10.1177_2515256420926354 - Supplemental material for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis
Supplemental material, sj-jpg-1-ctc-10.1177_2515256420926354 for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis by Matthew J. Hayes, Anne-C. Zakrzewski, Timothy P. Levine and Maximilian J. Telford in Contact
Supplemental Material
sj-jpg-2-ctc-10.1177_2515256420926354 - Supplemental material for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis
Supplemental material, sj-jpg-2-ctc-10.1177_2515256420926354 for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis by Matthew J. Hayes, Anne-C. Zakrzewski, Timothy P. Levine and Maximilian J. Telford in Contact
Supplemental Material
sj-jpg-3-ctc-10.1177_2515256420926354 - Supplemental material for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis
Supplemental material, sj-jpg-3-ctc-10.1177_2515256420926354 for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis by Matthew J. Hayes, Anne-C. Zakrzewski, Timothy P. Levine and Maximilian J. Telford in Contact
Supplemental Material
sj-jpg-4-ctc-10.1177_2515256420926354 - Supplemental material for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis
Supplemental material, sj-jpg-4-ctc-10.1177_2515256420926354 for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis by Matthew J. Hayes, Anne-C. Zakrzewski, Timothy P. Levine and Maximilian J. Telford in Contact
Supplemental Material
sj-jpg-5-ctc-10.1177_2515256420926354 - Supplemental material for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis
Supplemental material, sj-jpg-5-ctc-10.1177_2515256420926354 for Nucleus–Plasma Membrane Contact Sites Are Formed During Spermiogenesis in the Acoel Symsagittifera roscoffensis by Matthew J. Hayes, Anne-C. Zakrzewski, Timothy P. Levine and Maximilian J. Telford in Contact
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
