Abstract
The wide-ranging expression of glutamate receptors in peripheral tissues suggests an unexpectedly wider role(s) of
Keywords
L-Glutamate is an excitatory chemical transmitter that plays an essential role in neuronal plasticity, behavior, learning, and memory in the central nervous system (Foster and Fagg 1984; Mayer and Westbrook 1987). To use
Vesicular glutamate transporters (VGLUTs) play an essential role in glutamate signal output through vesicular storage of
In this study we investigated the expression and localization of VGLUT1 and VGLUT2 in non-neuronal cells of rat peripheral tissues using specific antibodies with special reference to pancreas, stomach, intestine, and testis. Because the GI contains more than 18 kinds of endocrine cells (Solcia et al. 1987), we expected that some of these cells would exhibit a glutamatergic phenotype, as in the case of islets of Langerhans. The testis expresses glutamate receptors (Storto et al. 2001), indicating that some cells are glutamatergic in nature. Here we show that VGLUT1 and VGLUT2 are present in pancreatic polypeptide-containing secretory granules of F-cells in the islets of Langerhans and that VGLUT2 is present in a subset of pancreatic polypeptide-containing stomach cells, L-cells containing glucagon-like immunoreactive peptide and PYY in ileum mucosa, and acrosomes of testicular spermatids. These results strongly suggest that
Materials and Methods
Animals
Male Wistar rats at 7–8 postnatal weeks were used throughout the study. All animal use procedures and care were in fully accordance with guidelines for the animal care at Okayama University.
Antibodies
DNA fragments encoding the C-terminal cytosolic regions of VGLUT1 (198 bp) (Ni et al. 1994) and VGLUT2 (270 bp) (Aihara et al. 2000), which correspond to G509-Y560 and G500–S582, respectively, were amplified by PCR and then cloned into the EcoRI site of expression vector pGEX3X for VGLUT1 and pGEX4T-2 for VGLUT2 (Amersham Pharmacia Biotech; Piscataway, NJ) to form GST fusion plasmids. The expression plasmids were transformed to
Expression of VGLUT1 and VGLUT2
Rat VGLUT2 cDNA, as previously described (Aihara et al. 2000), was subcloned into the EcoRI site of expression vector pcDNA3.1 (Invitrogen; San Diego, CA). The resultant construct, VGLUT2-pcDNA3.1, was used to transfect COS7 cells by lipofection using Trans IT reagent (Mirus; Madison, WI). The expression vector for VGLUT1, VGLUT1–pcDNA3.1, was kindly supplied by Drs. S. Takamori and R. Jahn of Göttingen University (Germany). The COS7 cells were grown in DMEM containing 10% fetal calf serum, penicillin, and streptomycin in a humidified incubator at 37C with 5% CO2. After incubation for 24 hr in 10-cm dishes, VGLUT1–pcDNA3.1, VGLUT2–pcDNA3.1, or the pcDNA3.1 vector alone was transfected into COS7 cells by adding 12 μg of the plasmid DNA per dish. After further incubation for 48 hr, the cells were rinsed with 10 ml of buffer comprising 20 mM MOPS-Tris (pH 7.0), 0.3 M sucrose, 2 mM Mg-acetate, 4 mM KCl, and protease inhibitors (leupeptin and pepstatin A at 10 μg/ml each), and then disrupted by brief sonication for 30 sec at output 4 with a Tomy Ultrasonic Disruptor UD-200. Then the mixture was centrifuged at 10,000 × g for 5 min and the resultant supernatant was centrifuged at 266,000 x g for 30 min in a Beckman Optima TLX ultracentrifuge. The precipitate was suspended in the same buffer, and used for Western analysis.
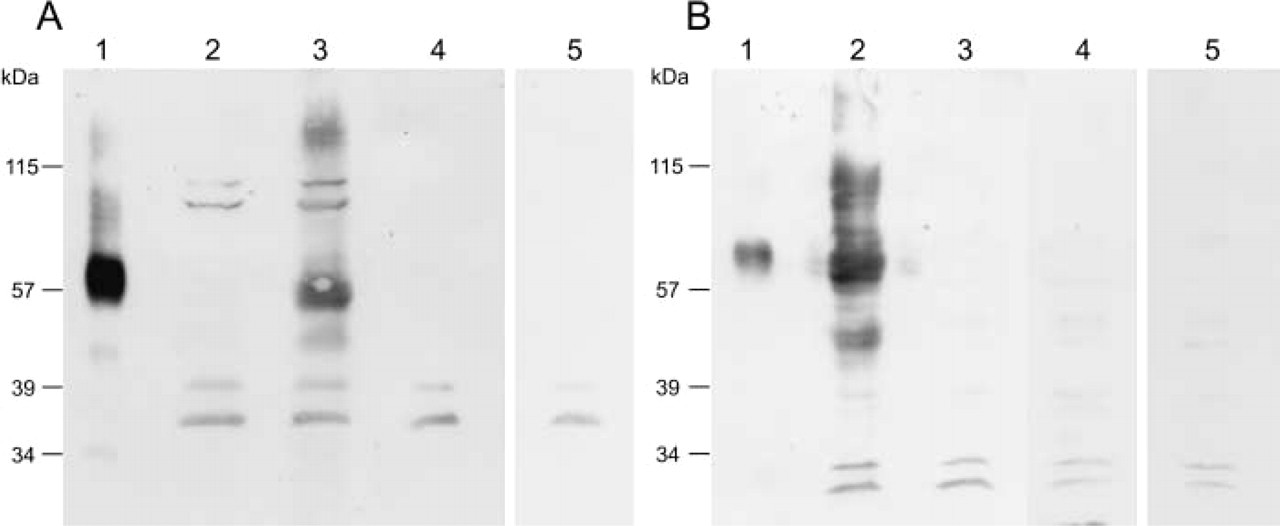
Immunological specificities of antibodies against VGLUT1 and VGLUT2. COS7 cells expressing VGLUT1 or VGLUT2 were cultured. The membrane fraction was solubilized with SDS sample buffer and aliquots (50 μg protein) were subjected to SDS-PAGE and then immuno-blotting was performed with anti-VGLUT1 antibodies (
Specimens, Immunohistochemistry, and Immunoelectron Microscopy
Indirect immunofluorescence microscopy was performed as described previously, using an Olympus FV-300 confocal laser microscope (Hayashi et al. 2001,2003). For immunoelectron microscopy, the LR White embedding immunogold method was used (Hayashi et al. 2003). Rats were anesthetized with ether and then perfused intracardially with saline, followed by 0.2% glutaraldehyde and 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4). Then each pancreas or testis was cut into small pieces, washed with 0.1 M cacodylate buffer (pH 7.4), stained with uranyl acetate for 2 hr, dehydrated, and then embedded in LR White for 2 days at −20C. Ultrathin sections on nickel grids were incubated with PBS containing 2% goat serum and 0.5% bovine serum albumin for 15 min, and then treated with either antibodies against VGLUT2 (diluted 1:50) or a mixture of antibodies against VGLUT2 (diluted 1:50) and pancreatic polypeptide (diluted 1:200) for 30 min. The sections were washed and treated with the secondary antibodies conjugated with colloidal gold. The sections were washed with 0.1 M cacodylate buffer, postfixed with 5% glutaraldehyde, stained sequentially with uranyl acetate for 30 min and lead citrate for 1 min, and observed under a Hitachi H-7100S electron microscope.
RT-PCR and Northern Analysis
Total RNA extracted from brain, islet of Langerhans, testis, and other tissues (1 μg) was transcribed into cDNA as described previously (Hayashi et al. 2001,2003). For PCR amplification, the 100-fold diluted synthesized cDNA solution was added to the reaction buffer containing 0.12 μM dNTPs (30 μM each dNTP), 25 pmole of primers, and 1.5 U of Gold Taq DNA polymerase (PE Biosystems; Urayasu, Japan). In the case of VGLUT1, the following primers were used: sense primer, AGTGAAATGGAAGACGAGGTT (1687–1717); antisense primer, TTCGGCACGAGCTTGAAACT (2002–2022) (GenBank no. U07609) (Ni et al. 1994). In the case of VGLUT2, the following primers were used; sense primer 2, AACACATCAACCAAGCAAGTC (2304–2324); antisense primer, AGGTAGTGAATGGGAGAGCA (2896–2915) (GenBank no. AF271235) (Aihara et al. 2000). Thirty temperature cycles were conducted as follows: denaturation at 94C for 30 sec, annealing at 58C for 30 sec, and extension at 72C for 1 min. In the case of KA1 and GluR5, the primers used and conditions for amplification were described previously (Yatsushiro et al. 2000), which were ATGACAAGACAGCTACTATC (703–722) as a sense primer and CCATTCCTGGTGAACTGTAA (1212–1231) as an antisense primer for KA1 (Werner et al. 1991), and GGTCCTCTATTACAACTGGA (523–544) as a sense primer and ATGCCATCCAGAAGACCAGT (959–978) as an antisense primer for GluR5 (Bettler et al. 1990). The amplification products were finally analyzed by polyacrylamide gel electrophoresis. DNA sequencing was performed by the chain-termination method (Sambrook et al. 1989). For Northern analysis, mRNA (25 μg) was purified with an OligoTexdT30 Super mRNA purification kit (Takara; Kusatsu, Japan) from total RNA (500 μg) isolated from testis or other tissues. The mRNAs (5.0 μg per lane) were separated on a formaldehyde agarose gel (1%) and then transferred to a nylon membrane (Amersham). The immobilized RNA was probed with cDNA fragments of VGLUT1 (bases 1687–2022), VGLUT2 (bases 2304–2915), KA1 (bases 703–1231), and GluR5 (bases 523–978), and labeled with [32P]-dCTP (3000 Ci/mmol; Amersham) random priming. After extensive washing, the membrane was subjected to autoradiography using BAS 1000 film (Fuji Film; Fuji, Japan).
Other Procedures
Crude synaptic vesicles were prepared as described (Hayashi et al. 2001). Islets of Langerhans were isolated from male Wistar rats at 7–8 postnatal weeks by the collagenase digestion method combined with discontinuous Ficoll gradient centrifugation (Gotoh et al. 1987). A membrane fraction of islets of Langerhans or testes was prepared as follows: 500 islets or 2 testes were homogenized with a Dounce homogenizer in 20 mM MOPS–Tris (pH. 7.5) containing 0.3 M sucrose, 5 mM EDTA, 5 μg/ml pepstatin A, 5 μg/ml leupeptin, 5 μg/ml chymostatin, and 5 μg/ml antipain. After removing nuclei and unbroken cells by centrifugation at 3000 × g for 8 min, the supernatant was centrifuged at 100,000 × g for 40 min and the resultant pellet was washed twice, suspended in the same buffer, and finally dissociated with the sample buffer containing 10% SDS and 10% β-mercaptoethanol. Western blotting was performed as described previously (Hayashi et al. 2001).
Results
Specific Antibodies to VGLUT1 and VGLUT2
In this study we used site-specific polyclonal antibodies against VGLUT1 and VGLUT2, which were prepared using GST-fusion proteins containing the carboxyl terminals of these transporters. The regions used for the antibodies are different from each other (Ni et al. 1994; Aihara et al. 2000). The VGLUT1 antibodies recognized a single polypeptide of ∼62 kD in brain membranes (Figure 1A, Lane 1) and VGLUT1-expressing COS7 cell membranes (Figure 1A, Lane 3), but not any polypeptide in the membrane fraction prepared from COS7 cells (control vector) (Figure 1A, Lane 2) or VGLUT2-expressing COS7 cells (Figure 1A, Lane 4). The immunoreactivity was blocked when the nitrocellulose sheet was treated with the antigenic peptide used for the preparation of anti-VGLUT1 antibodies during immunostaining (Figure 1A, Lane 5). Conversely, antibodies against VGLUT2 specifically reacted with a 67-kD polypeptide in synaptic vesicle membranes and VGLUT2-expressing COS7 membranes (Figure 1B, Lanes 1 and 2). No crossreactivity was observed in control COS7 membranes (Figure 1B, Lane 3) or VGLUT-1-expressing COS7 membranes (Figure 1B, Lane 4). The immunoreactivity was blocked when the nitrocellulose sheet was treated with the antigenic peptide used for the preparation of anti-VGLUT2 antibodies during immunostaining (Figure. 1B, Lane 5). These results indicated that these antibodies specifically recognized VGLUT1 and VGLUT2, respectively.
Expression and Localization of VGLUTs in the Gastroenteropancreatic System
We explored the expression of VGLUT1 and VGLUT2 in peripheral tissues by indirect immunofluorescence microscopy and immunoelectron microscopy, and found that the major sites of VGLUT1 and VGLUT2 expression were in the gastroenteropancreatic system (see below).
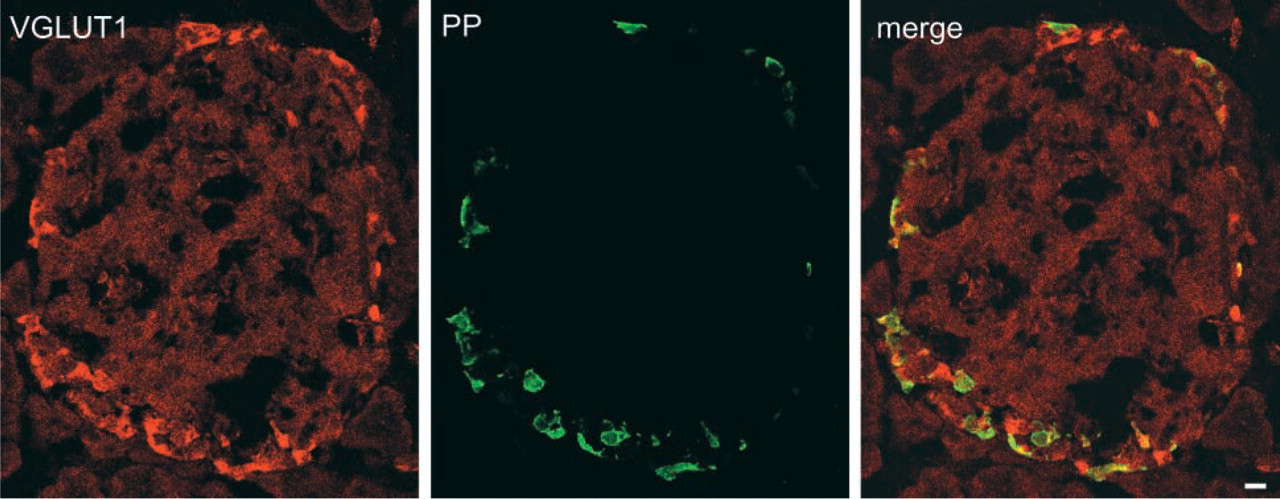
Indirect immunofluorescence microscopy revealed that VGLUT1 and VGLUT2 are associated with pancreatic polypeptide-containing secretory granules in F-cells of islets of Langerhans. Sections of pancreas were doubly immunostained with antibodies against VGLUT1 (green) and pancreatic polypeptide (red) and then observed under a confocal microscope. A merged picture is also shown. Bar = 10 μm.
As in the case of stomach, no VGLUT1 immunore-activity was detected in the small intestine (data not shown). On the other hand, VGLUT2 immunoreactivity was frequently observed in the ileum. Figure 5 indicates that the VGLUT2 immunoractivity is always co-localized with that of glucagon, which was obtained from 80 cells. Preabsorbed VGLUT2 antibodies failed to immunostain glucagon-containing cells (data not shown). In contrast to islets of Langerhans and stomach, pancreatic polypeptide-containing cells (33 cells observed) did not contain any immunoreactivity for VGLUT2. Serotonin-containing cells (more than 100 cells) also did not contain any immunoreactivity for VGLUT2. Among enteroendocrine cells, L-cells showed glucagon immunoreactivity (Solcia et al. 1987). Proglucagon is processed to glicentin in L cells (Holst 1997). Pancreatic polypeptide-like peptide (PYY), which differs from authentic pancreatic polypeptide, was also present in secretory granules in most L-cells (Solcia et al. 1987). Consistently, VGLUT2 immunoreactivity was also co-localized with PYY, which was obtained from 48 cells observed (Figure 5). The VGLUT2-positive cells are located in the mucosal epithelium region near muscles and show a morphology distinct from the enteric neurons, and do not coincide with VAchT and synaptophysin, markers of glutamatergic enteric neurons (Tong et al. 2001) (Figure 6). Considering these findings, we concluded that the VGLUT2-expressing cells are L-cells. Co-association of VGLUT2 with glucagon and PYY with particles suggests that VGLUT2 is associated with glicentin-containing secretory granules in these cells, as in the case of glucagon-containing secretory granules in islet α-cells.
Expression and Localization of VGLUT2 in Testis
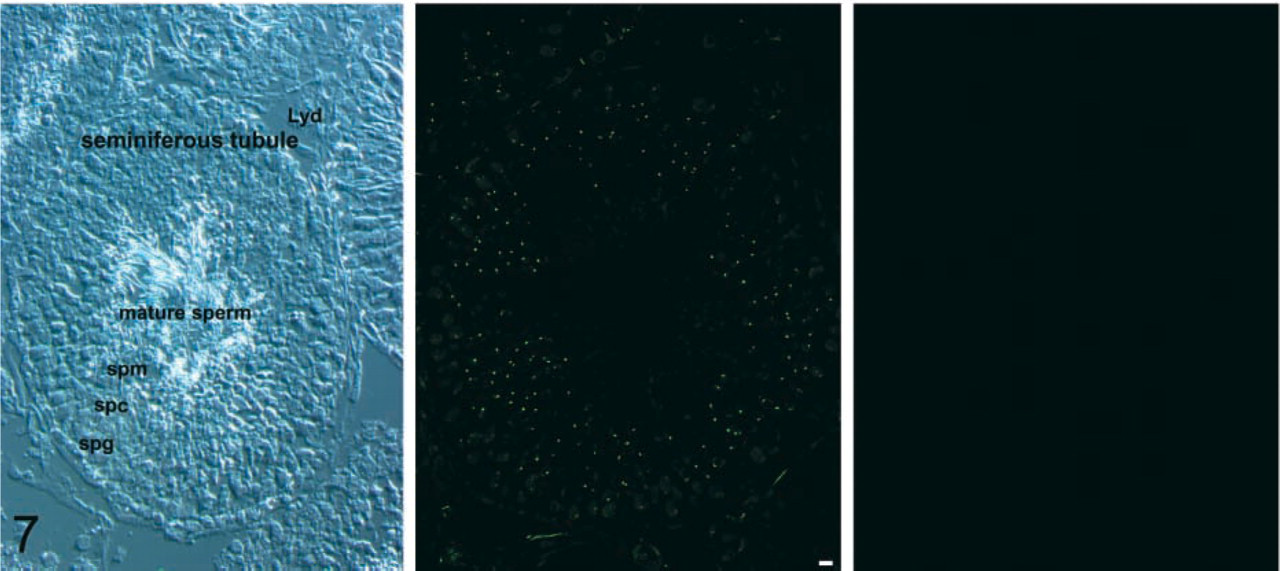
Mammalian testes consist of many seminiferous tubules, in which the epithelial cells called Sertoli cells anchor and provide nourishment for the developing spermatozoa. The germ stem cells in each tubule reside at the tubule periphery and give rise to proliferating spermatogonial cells. Only the primitive spermatogonia undergo a series of synchronous cell divisions and differentiation, leading to mature sperm (see Figure 7). Immunohistochemical evidence suggests that the immunoreactivity for NMDAR1, GluR2/3 or mGluR2/3 antibodies is distributed in various sites, including the sperm heads of spermatids and spermatozoa (Gill et al. 2000), and that for mGluR5 and mGluR1 antibodies is found inside semiferous tubules and in mature spermatozoa, respectively (Storto et al. 2001). These observations prompted us to explore the expression of VGLUTs in testis.
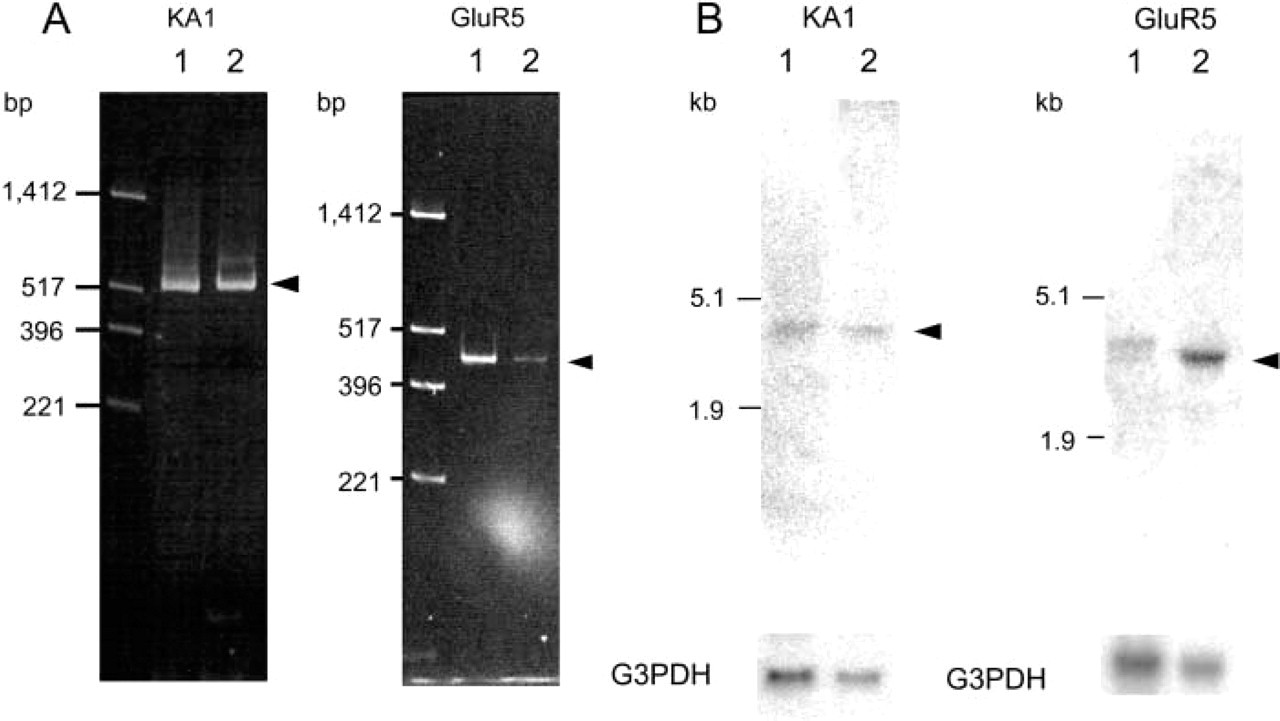
RT-PCR and Northen blotting analysis clearly indicated the presence of a VGLUT2 gene transcript in testis (Figure 8), while the level of transcription of the VGLUT1 gene was below the detection limit of our assay (not shown). Western blotting analysis indicated the presence of VGLUT2 protein in the particulate fraction of testis (Figure 8). These results indicated the expression and localization of VGLUT2 in testis. Immunohistochemistry indicated that VGLUT2 immunoreactivity was specifically located in spermatids (Figure 7). This immunoreactivity disappeared when the antibodies were pre-absorbed with antigenic polypeptide (Figure 7). No VGLUT2 immunoreactivity was observed in primitive spermatogonia or mature sperm. These results indicated that VGLUT2 is expressed in spermatids. Developmentally, the immunoreactivity appears at 3 postnatal weeks, reaches a plateau at 7 weeks, and then gradually decreases, although some immunoreactivity remains even after 1 year. Consistent with the results of RT-PCR and Northern blotting analysis, little immunoreactivity for VGLUT1 was observed throughout a cross-section of testis (data not shown).
(
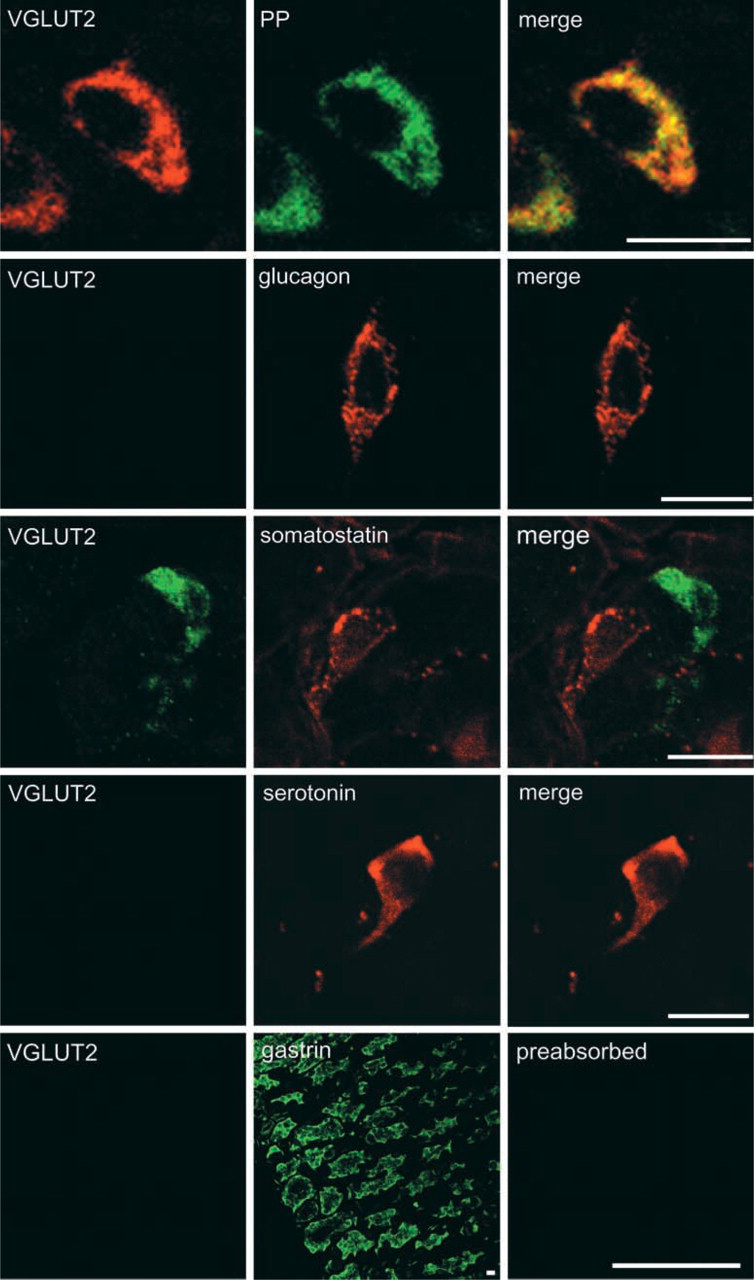
VGLUT2 is co-localized with pancreatic polypeptide in stomach mucosa. Sections of stomach were doubly immunostained with antibodies against VGLUT2 (green) and pancreatic polypeptide (red), VGLUT2 (green) and glucagon (red), VGLUT2 (green) and serotonin (red), and VGLUT2 (green) and somatostatin (red), and then observed under a confocal microscope. Merged pictures are also shown. For gastrin (G)-cells, consecutive stomach sections were immunostained for gastrin (green) and VGLUT2 (green), respectively. Preabsorbed VGLUT2 antibodies did not immunostain pancreatic polypeptide-containing cells (preabsorbed). Bars = 10 μm.
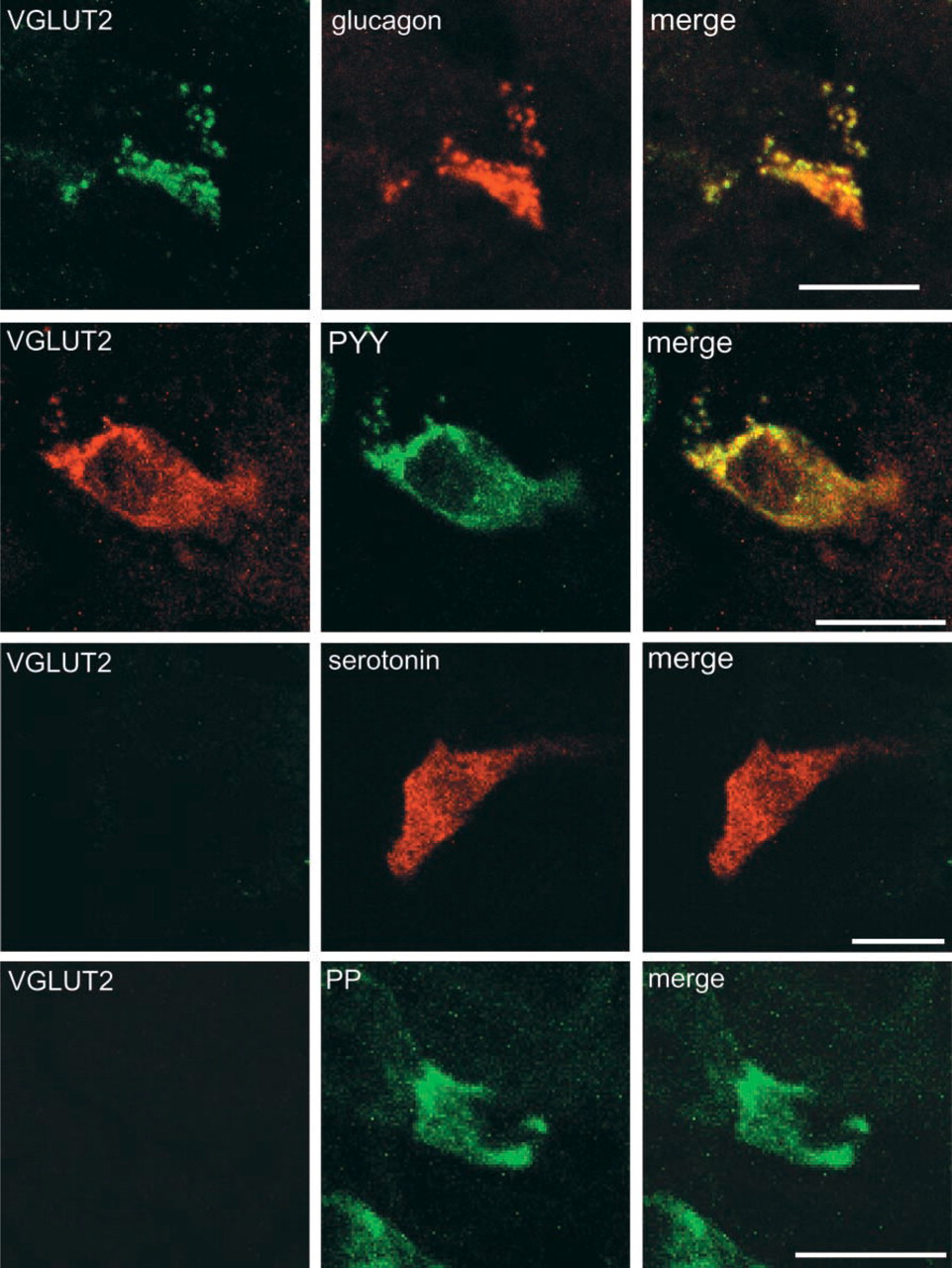
VGLUT2 is expressed in L-cells in the mucosa of ileum. Sections of ileum were doubly immunostained with antibodies against VGLUT2 (green) and glucagon (red), VGLUT2 (green) and PYY (red), VGLUT2 (green) and serotonin (red), and VGLUT2 and pancreatic polypeptide (PP) (red). Merged pictures are also shown.
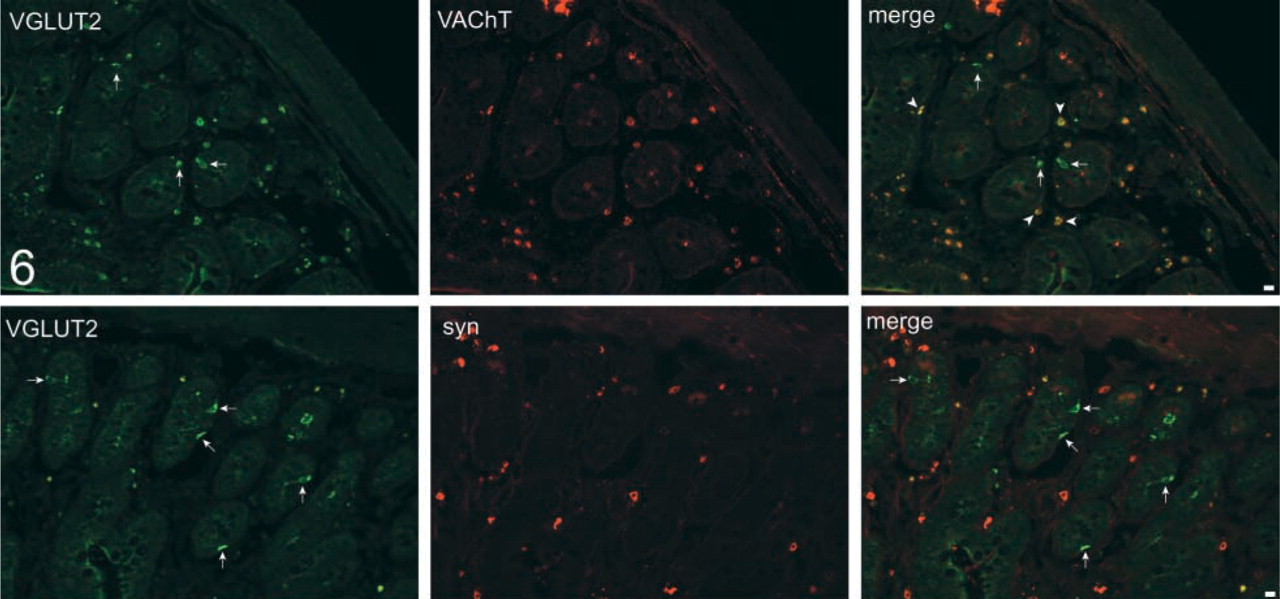
Low-power views of sections of ileum doubly immunostained with antibodies against VGLUT2 (green) and vesicular acetylcholine transporter (red), and VGLUT2 (green) and synaptophysin (red). The specimens were observed under a confocal microscope. Merged pictures are also shown. VGLUT2-containing endocrine cells are indicated by arrows. Enteric neurons expressing vesicular acetylcholine transporter (VAchT) are indicated by arrowheads. Bars = 10 μm.
VGLUT2 is expressed in testicular spermatids. (
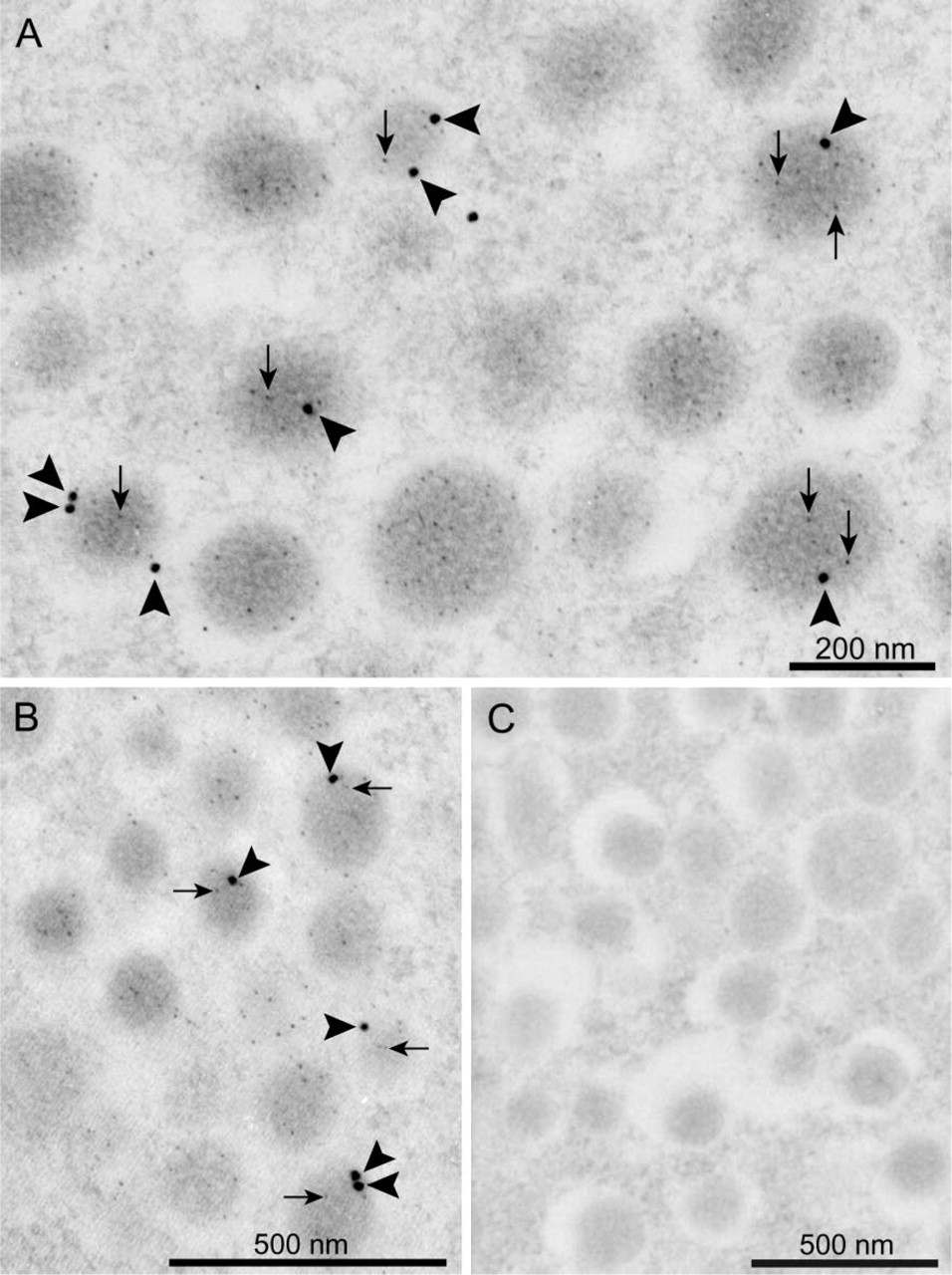
More detailed localization of VGLUT2 at the sub-cellular level was conducted as follows. We found that VGLUT2 immunoreactivity is always closely localized with acrosin, a marker for acrosomal protein in spermatids (Peknicova and Moos 1990), indicating that VGLUT2 is in some way associated with the acrosomal membrane (Figure 9). Consistently, immunoelectron microscopy demonstrated that gold particles for VGLUT2 are associated with the outer acrosomal membrane (Figure 10A, arrows). A lower level of labeling by the VGLUT2 gold particles was observed in the acrosomal membranes of late spermatids (Figure 10B). Furthermore, only background labeling was observed in acrosomes of mature sperm (Figure 10C) compared with the labeling with control IgG (Figure 10D). From these results, we conclude that VGLUT2 is present in the outer acrosomal membrane.
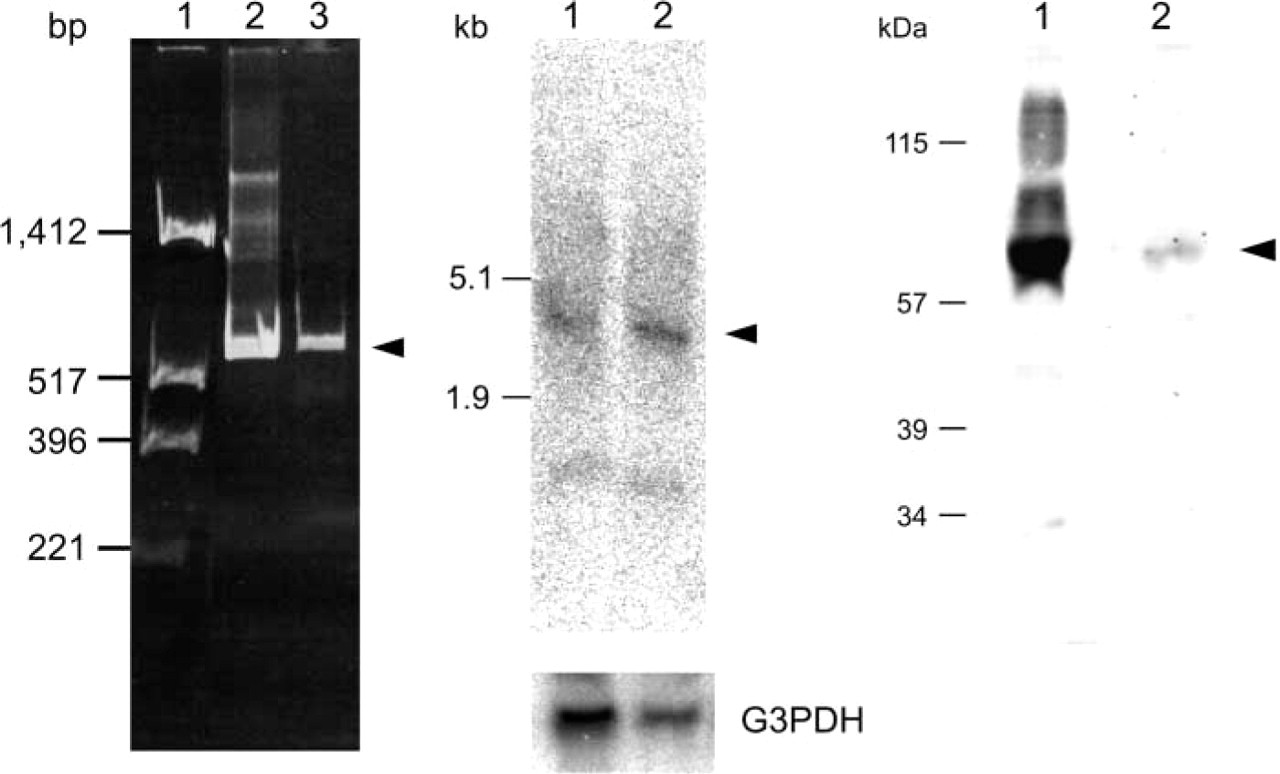
Detection of transcript of VGLUT2 gene and VGLUT2 protein in testis. (
Western blotting was performed using the indicated antibodies and the immunoreactivity was visualized by ECL. The apparent molecular masses are also shown.
The presence of VGLUT2 in the outer acrosomal membrane suggests that the acrosome is the site of
Discussion
VGLUT confers the glutamatergic phenotype on neurons and some endocrine cells. Either VGLUT1 or VGLUT2 or both are expressed in almost all glutamatergic neurons. Although VGLUTs are greatly homologous to each other, their primary amino acid sequences exhibit 90% identity, the properties of the transporters, e.g. substrate specificity, chloride anion-sensitivity, and driving force, are almost identical (Bellocchio et al. 2000; Takamori et al. 2000,2001,2002; Fremeau et al. 2001; Hayashi et al. 2001; Kaneko and Fujiyama 2002; Varoqui et al. 2002). Recent results from our laboratory constituted the evidence for the functional occurrence of VGLUT1 and VGLUT in peripheral tissues. To completely characterize peripheral glutamatergic systems, in the present study we explored the expression and localization of VGLUT1 and VGLUT2 in peripheral tissues.
We found that VGLUT1 and VGLUT2 are expressed in the gastroenteropancreatic tract and testis. In pancreas, VGLUT2 is present in glucagon-containing secretory granules in α-cells (Hayashi et al. 2001,2003) and in pancreatic polypeptide-containing secretory granules in F-cells (Figure 2). VGLUT1 is also expressed and localized in pancreatic polypeptide-containing secretory granules in F-cells (Figure 2) and in glucagon-containing secretory granules in a subset of α-cells (Hayashi et al. 2003). The localization of VGLUT1 and VGLUT2 in these secretory granules implies that
VGLUT2 is localized in the acrosomes. Sections of testis were doubly immunostained with antibodies against VGLUT2 (green) and acrosin (red) and then observed under a confocal microscope. The boxed area was enlarged. A merged picture is also shown. Note that the immunologically positive areas for VGLUT2 and acrosin do not completely match each other. Bars = 10 μm.
Various endocrine cells may receive signals in the GI tract and transmit them to a local target, such as neighboring epithelial cells, subepithelial nerve endings, and smooth muscle fiber. Glicentin may act as an intercellular messenger in paracrine action (Solcia et al. 1987). Glucagon-like peptide also potently inhibits GI secretion and motility, and its physiological functions include mediation of the “ileal brake” effect, which regulates food intake (Holst 1997). Pancreatic polypeptide and its relative, PYY, have an effect similar to that of glucagon (Solcia et al. 1987). We have shown that VGLUT2 is expressed in a subpopulation of pancreatic polypeptide-containing cells in stomach and in glucagon-like immunoreactivity and PYY-containing cells in small intestine, which is obviously different from glutamatergic enteric neurons. The co-localization of VGLUTs with either glucagon, glicentin, pancreatic polypeptide, or PYY supports the possibility that
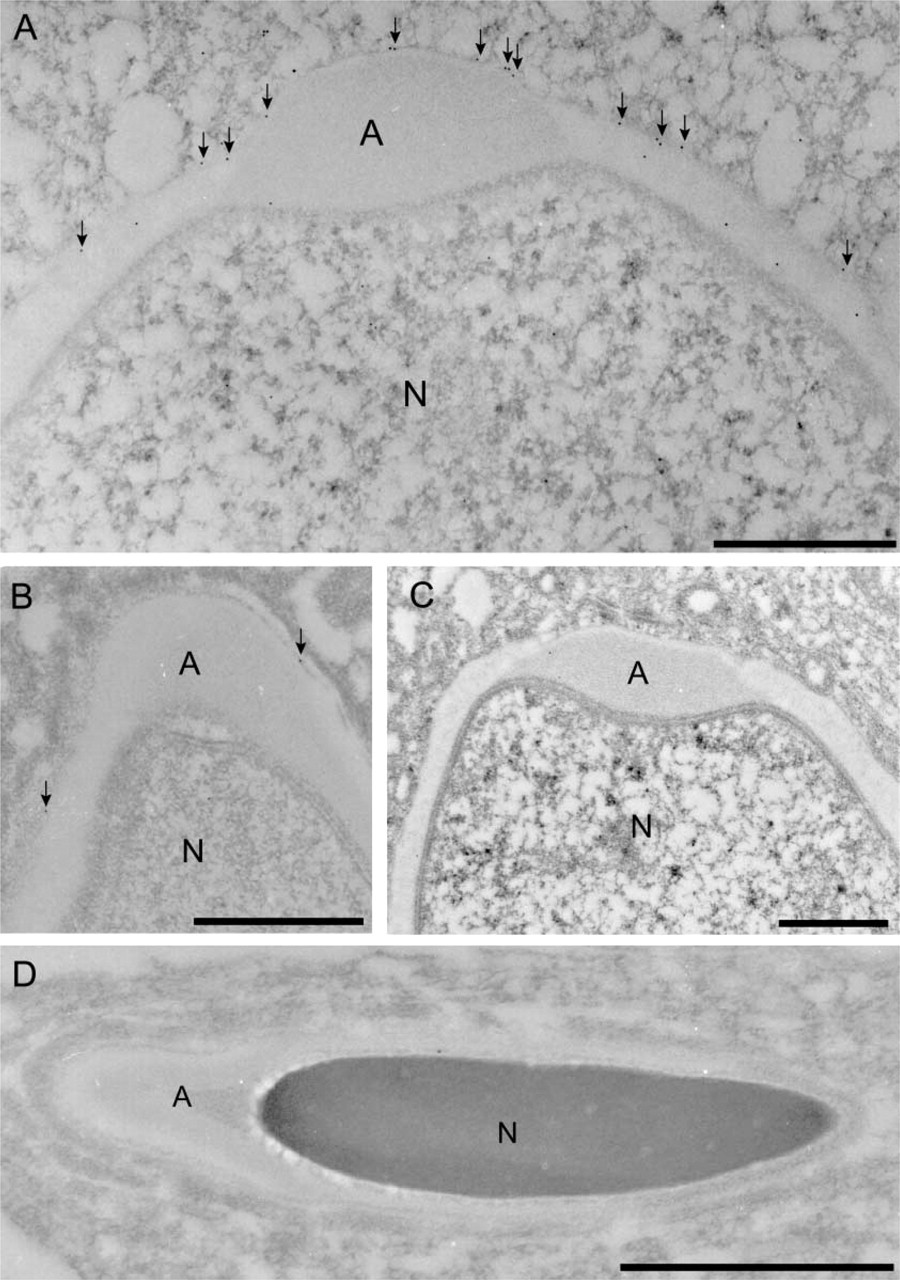
Immunoelectron microscopy revealed the localization of VGLUT2 with the outer acrosomal membrane in spermatids (
Expression and localization of KA1 and GluR5 glutamate receptors in testis. (
The mode of glutamate signal output can be classified into two major types, i. e., vesicle-dependent and vesicle-independent processes. The present study, and our or other recent studies, clearly indicated that vesicle-dependent glutamate signal output can be further classified into at least three groups, i.e., exocytosis of synapse vesicles in neurons, exocytosis of synapse-like microvesicles in pinealocytes, and exocytosis of hormone-containing secretory granules. Astrocytes are also known to secrete
In testis, VGLUT2 is present in the outer surface of the acrosomal membrane. This implies that
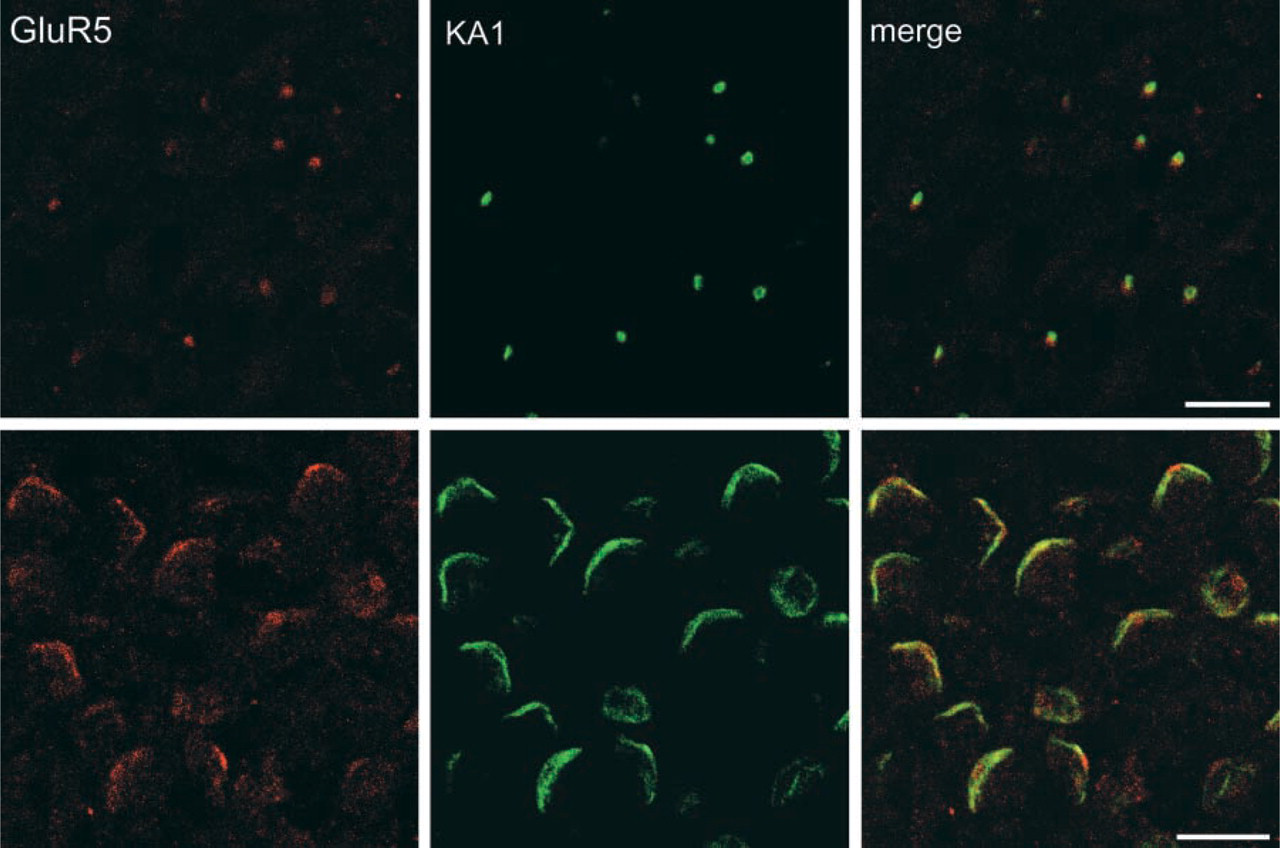
Co-localization of KA1 (green) and GluR5 (red) in early spermatids (upper panels) and late spermatids (bottom panels). Note that KA1 and GluR5 are elongated along with acrosomes in late spermatids and mature sperm. Bars = 10 μm.
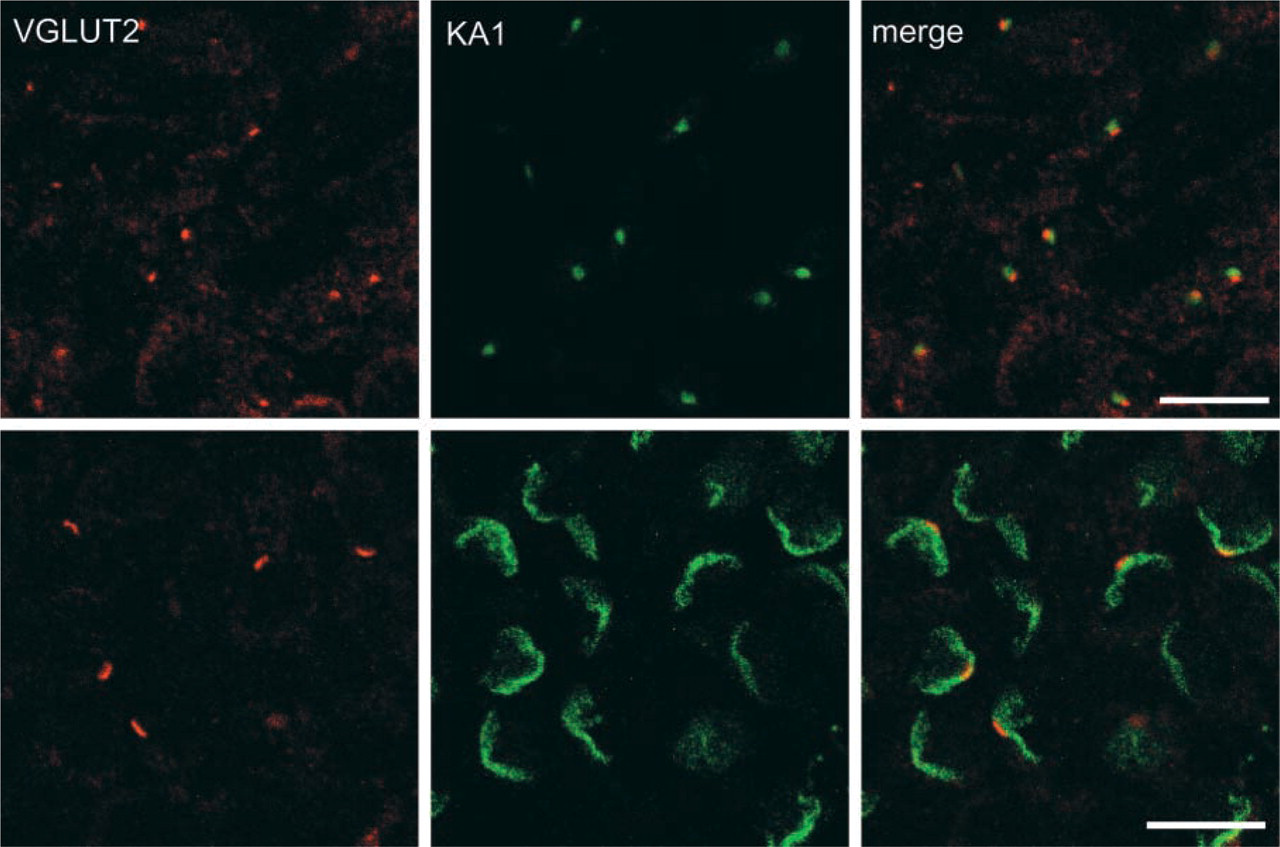
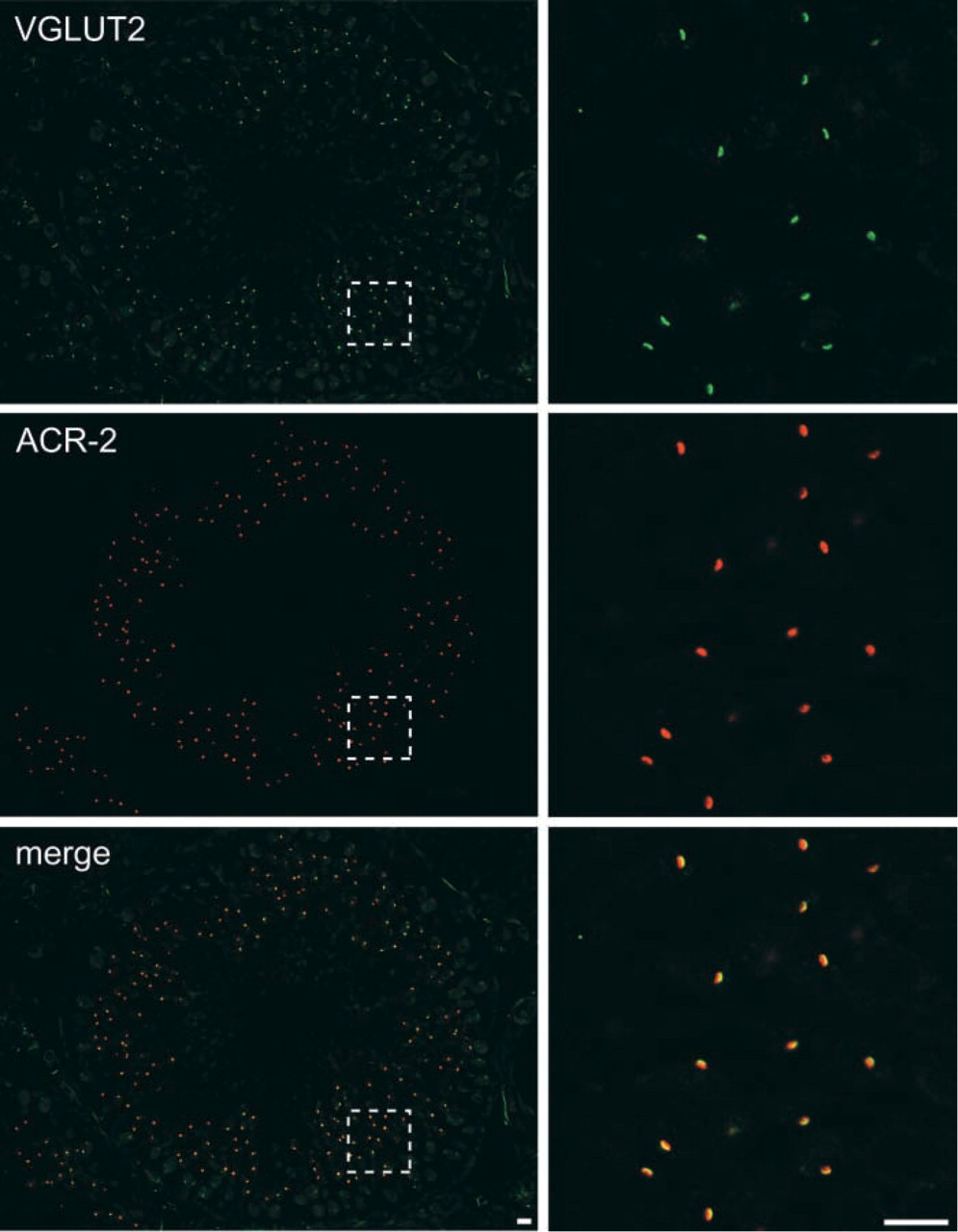
Immunohistochemical localization of KA1 and VGLUT2. Sections of testis were doubly immunostained with antibodies against VGLUT2 (red) and KA1 (green) and were observed under a confocal microscope. Merged pictures are also shown. Bars = 10 μm.
Alternatively, as to the function of acrosomal
Very recently, another VGLUT isoform, VGLUT3, has been identified (Fremeau et al. 2002; Gras et al. 2002; Schafer et al. 2002; Takamori et al. 2002). In central nervous system, VGLUT3 shows more restricted expression and is present in some populations of inhibitory neurons, cholinergic interneurons, monoamine neurons, and glia. VGLUT3 is also expressed in liver and kidney (Fremeau et al. 2002). It is therefore quite likely that VGLUT3 functions as a component of peripheral glutamatergic systems. Further studies, particularly expression and localization of VGLUT3, are required to elucidate the overall features of the peripheral glutamatergic system.
In conclusion, the present work has demonstrated the expression of VGLUT1 and VGLUT2 in the gastroenteropancreatic system and testis. The localization of VGLUT1 and VGLUT2 in secretory granules in the gastroenteropancreatic system suggests a widespread role of
Footnotes
Acknowledgements
Supported in part by grants from the Japanese Ministry of Education, Science and Culture, the NOVARTIS Foundation, grants from the Society for Research on Umami Taste, and the Kazato Research Foundation. M.H. was supported by research fellowships from the Japan Society for the Promotion of Science for Young Scientists.
We wish to thank Prof R. Jahn and Dr S. Takamori (Max Planck Institute, Germany) for providing us with the cDNA for VGLUT1. We are also grateful to Drs M. Otsuka, Y. Yatsushiro, and S. Ishio for their help in the initial stages of the study.
