Abstract
The localization of ubiquitin (UB) signals in the acrosomes of rat spermiogenic cells was investigated by immunoelectron microscopy using two anti-UB antibodies: UB1, reacting with ubiquitinated proteins and free UB; and FK1, recognizing polyubiquitinated proteins but not monoubiquitinated proteins or free UB. Labeling of UB by UB1 (UB1 signal) was detected in the acrosomes at any stage of differentiation. In step 1 spermatids, UB1 signals were detected on the cytoplasmic surface and in the matrix of transport vesicles located between the trans-Golgi network and the acrosome. Weak signals were detected in acrosomal granules within acrosome vesicles that had not yet attached to the nucleus. In step 4–5 spermatids, the acrosome vesicles had enlarged and attached to the nucleus. Strong gold labeling was noted in a narrow space between the outer acrosomal membrane and the developing acrosomal granule, where a dense fibrous material was observed on routine electron microscopy, whereas the acrosomal granule was weakly stained by UB1 antibody. In step 6–8 spermatids, UB1 signals were detected in the fibrous material that expanded laterally to form a narrow electronless dense zone between the acrosomal granule and the outer acrosomal membrane. Labeling in the acrosomal granule increased. In step 9–11 spermatids, UB1 signals were confined to the narrow zone from the tip of the head to the periphery of the ventral fin. The matrix of the acrosome was weakly stained. In epididymal sperm, UB1 labeling in the acrosome decreased without any pretreatment, whereas staining was noted in a spot in the neck region and in the dorsal fin after trypsin digestion. On the other hand, the staining pattern with FK1 was quite different from that with UB1. The trans-Golgi network was weakly stained but the cis-Golgi network was strongly stained. The dense fibrous material just beneath the outer membrane was never stained with FK1. The results suggest that UB on the surface of transport vesicles is involved in anterograde transport from the Golgi apparatus to the acrosome. The physiological role of UB in acrosomes is not clear. Two candidates for monoubiquitinated proteins in the acrosome, which have a UB-interacting motif, were found by cyber screening.
Keywords
D
The formation of the acrosome is thought to involve many proteins, which, having completed their roles, are degraded via the intracellular proteolysis system. Intracellular proteolysis involves two systems, a lysosomal system and a proteasome system. In the former, proteolysis occurs in membrane-bound compartments, called lysosomes, into which cytosolic proteins are incorporated through a process using the KFERQ sequence and binding to chaperon protein (Dice et al. 1986) or through microautophagy (de Duve and Wattiaux 1966). In the latter proteolytic system, target proteins are tagged with a ubiquitin (UB) chain in a series of ATP-dependent enzyme reactions involving E1, E2, and E3 (Hershko et al. 1983). The polyubiquitin-conjugated proteins are recognized and degraded by the 26S proteasome (Hershko and Ciechanover 1992), a barrel-shaped structure consisting of one or two regulatory particles and a core particle (Ikai et al. 1991; Loewe et al. 1995). In the UB proteasome system, target proteins are conjugated with polyubiquitin chains and recognized and cleaved by the regulatory particles (Thrower et al. 2000). Thus, UB has a very important role in the tagging of proteins to be degraded.
Recently, other functions of UB have been discovered: a single molecule of UB was found to modify proteins (Dupre et al. 2001; Hicke 2001; Raiborg et al. 2003). This monoubiquitination is involved in at least three distinct cellular functions: histone regulation, endocytosis, and vesicular transport between the trans-Golgi network (TGN) and endosomes, and in the budding of retroviruses from the plasma membrane.
During the formation of the acrosome, acrosomal proteins are thought to be transported to acrosomal vesicles from the TGN, where they are sorted from other lysosomal proteins and membrane proteins. It is not known whether UB is used for this sorting.
In this paper, we investigated the localization of UB signals in the acrosome during the spermiogenesis of rat testis using two anti-UB antibodies.
Materials and Methods
Male Wistar albino rats (200–250 g, 9 weeks old) and male Japanese white rabbits (3.0–3.5 kg) were fed appropriate standard diets and water ad libitum until used. The animal experiments were performed in accordance with the Guidance for Animal Experiments of the University of Yamanashi.
Antibodies
Anti-UB antibody against bovine erythrocyte UB was prepared by the method of Hershko et al. (1982). Briefly, 10 mg of bovine red cell UB (Sigma-Aldrich; St Louis, MO) and 16 mg of keyhole limpet hemocyanin (Sigma-Aldrich) were dissolved in 1.2 ml of 0.14 M potassium phosphate (pH 7.0). To this solution, 40 μl of 3% glutaraldehyde was added by stirring at room temperature. The same amount of glutaraldehyde was added three more times at 10-min intervals. The mixture was allowed to stand for 90 min and was dialyzed overnight in the cold against 2 liters of PBS. SDS was added at a final concentration of 2% to the mixture, which was then boiled for 5 min. After cooling, AlCl3 was added to the mixture at a concentration of 1% (w/v) and slowly neutralized with 1 M NaOH (Lazarides and Hubbard 1976). The resulting Al/SDS/protein precipitate was emulsified with the same volume of Freund's complete adjuvant. The emulsion containing 0.1 mg of UB was injected four times every 2 weeks into rabbits of 3.0-3.5 kg. Blood was collected 2 weeks after the last injection. The reactivity of antisera to UB was tested by dot blot analysis. All antisera reacted with 0.02 μg of UB. Specific antibody was purified on an affinity column of UB-coupled Sepharose 4B (Pharmacia; Uppsala, Sweden) and named UB1. In addition to our antibody, we used another anti-UB antibody, mouse monoclonal antibody, recognizing only polyubiquitinated proteins (FK1; Affiniti Research Products Ltd., Exeter, UK). Horseradish peroxidase (HRP) -conjugated swine anti-rabbit IgG and HRP-conjugated rabbit anti-mouse IgG and IgM were obtained from DAKO Japan (Tokyo, Japan). Cy3-conjugated goat anti-rabbit IgG was acquired from Chemicon International (Temecula, CA). Alexa 546-conjugated goat anti-rabbit IgG was purchased from Molecular Probes (Eugene, OR).
Western Blot Analysis of Testis Homogenate and Epididymal Sperm
Rat testis and epididymal sperm were homogenized in 0.1 M phosphate buffer containing 0.1% Triton X-100 and protease inhibitors (all from Sigma-Aldrich), 10 μM phenyl-methylsulfonyl fluoride, 4 μM leupeptin, 4 μM chymostatin, 4 μM pepstatin, and 4 μg of bestatin (Wilson and Goulding, 1986) as well as 10 mM iodoacetamide to prevent deubiquitination (Baarends et al. 1999) and centrifuged at 100,000 × g for 1 hr. The supernatants were stored at −70C and used. Protein concentrations were determined by the bicinchoninic acid method (Pierce Chemical; Rockford, IL) with BSA as a standard. The protein concentrations of the extracts were adjusted to 2 mg/ml, and the extracts were mixed with 1 volume of sample buffer for SDS-PAGE and heated in boiling water for 2 min. Five μg of each sample was analyzed by Western blotting.
Tissue Preparation
Testes were dissected out from rats and sliced in an ice-cold fixative consisting of 4% paraformaldehyde, 0.1-0.2% glutaraldehyde, 0.01% CaCl2, and 0.2 M Hepes-KOH buffer (pH 7.4). For immunofluorescence microscopy, the tissue was fixed with the same fixative containing 0.1% glutaral-dehyde for 1 hr at 4C. For immunoelectron microscopy, the tissue slices were fixed in the same fixative containing 0.2% glutaraldehyde for 1 hr at 4C and then cut into small tissue blocks. The tissue blocks were dehydrated in a graded ethanol series and embedded in LR White resin at −20C. Sperm from caudal epididymis was fixed in 4% paraformaldehyde plus 0.2 M Hepes-KOH buffer (pH 7.4) for 30 min. After a brief wash in PBS, the fixed sperm was suspended in PBS containing trypsin (1 mg/ml; Difco Laboratories, Detroit, MI) and allowed to stand for 3 min at 37C. After another wash in PBS, the sperm was dehydrated and embedded in LR White resin as described above.
Smear Preparation of Sperm
Epididymal sperm was collected from the caudal epididymis and suspended in P-1 Medium (Irvine Scientific; Santa Ana, CA). After a wash with the same medium, sperm was smeared on clean glass slides coated with polylysine and fixed in 4% paraformaldehyde buffered with 0.2 M Hepes-KOH to pH 7.4 for 15 min at room temperature. After a wash with a solution consisting of 0.05 M phosphate buffer (pH 7.2) and 0.9% NaCl (PBS), the preparations were used for immunofluorescence staining.
Immunofluorescence Microscopy
The fixed tissue was embedded in Tissue-Tek (Sakura Fine Technical; Tokyo, Japan) and frozen at −20C. Frozen sections (10 μm thick) were cut with a Coldtome (Sakura Fine Technical) and mounted on clean glass slides. Sections were treated with 0.05% SDS for 30 min and then with 0.05% sodium borohydride for 30 min at room temperature (Robinson and Vandre, 2001). After a brief wash with PBS, sections were incubated with the primary antibodies, as described above, followed by Cy3- or Alexa 549-labeled secondary antibodies. For the immunocytochemical control, the incubation with the primary antibodies was omitted. Some of the smear preparations were stained in the same way but without pretreatment with SDS and sodium borohydride, whereas the others were stained after they had been treated with trypsin (1 mg/ml) for 2 min at 37C. Sections and smear preparations were examined using a Y-FL fluorescent microscope (Nikon; Tokyo, Japan) or a TCS 4D con-focal laser scanning microscope (Leica Microsystems; Mannheim, Germany).
Postembedding Immunoelectron Microscopy
Thin sections of rat testis and epididymal sperm embedded in LR White resin were cut with a diamond knife equipped with a Reichert Ultracut R (Leica; Vienna, Austria), mounted on nickel grids, and incubated overnight with the primary antibodies (5 μg/ml) at 4C, followed by protein A-gold probes 15 nm in diameter. For the immunocytochemical control experiment, the incubation of sections with the primary antibodies was omitted. Sections were stained with 2% uranyl acetate for 8 min and lead citrate for 30 sec and examined with a H7500 electron microscope (Hitachi; Tokyo, Japan) at an acceleration voltage of 80 kV.
Routine Electron Microscopy
Tissue slices of rat testis were fixed in 2% glutaraldehyde for 1 hr at 4C and cut into small tissue blocks. After a brief wash in PBS, the tissue blocks were postfixed in 1% reduced osmium tetroxide for 1 hr at room temperature. The tissue blocks were dehydrated in a graded ethanol series and embedded in Epon 812. Thin sections were stained with lead citrate and examined with an electron microscope.
Quantitative Immunoelectron Microscopy
Seven micrographs of spermatids at the steps mentioned below, which contained a Golgi region, acrosomes, and multivesicular bodies, were taken at a magnification of × 15,000. The images were of spermatids at steps 1-3, 4-7, 8-11, 12-15, and 16-19. They were enlarged twofold into positive pictures. The cis-Golgi network and the trans-Golgi network were traced on the images. The areas of the cis-Golgi network, the trans-Golgi network, and the multivesicular bodies and the length of the outer membrane to which dense fibrous material was attached were estimated using a Sigma-Scan scientific measurement system equipped with a computer (Jandel Scientific; San Rafael, CA), and the numbers of gold particles in those areas and the length were calculated. The labeling density was expressed as the number of gold particles per square micrometer of the estimated areas or per micrometer of the estimated length.
Results
Western Blot Analysis
The Western blots of rat testis homogenate and epididymal sperm extract are shown in Figure 1. Our UB1 antibody produced many discrete bands over a broad range of molecular masses in the homogenate (Figure 1, lane 2). Many of these bands were weakly stained or eliminated when the UB1 antibody was absorbed by free bovine erythrocyte UB (Figure 1, lane 3). FK1 antibody produced smeared broad bands at higher molecular masses in the homogenate (Figure 1, lane 4). Most of these bands were not abolished with the absorption by free UB (Figure 1, lane 5). Both UB1 and FK1 antibodies showed a few bands in sperm extract (Figure 1, lanes 6 and 8). The most prominent band was produced with both antibodies, and its molecular mass was 61 kD (Figure 1, arrow). This band was eliminated on the absorption of UB1 by free UB (Figure 1, lane 7).
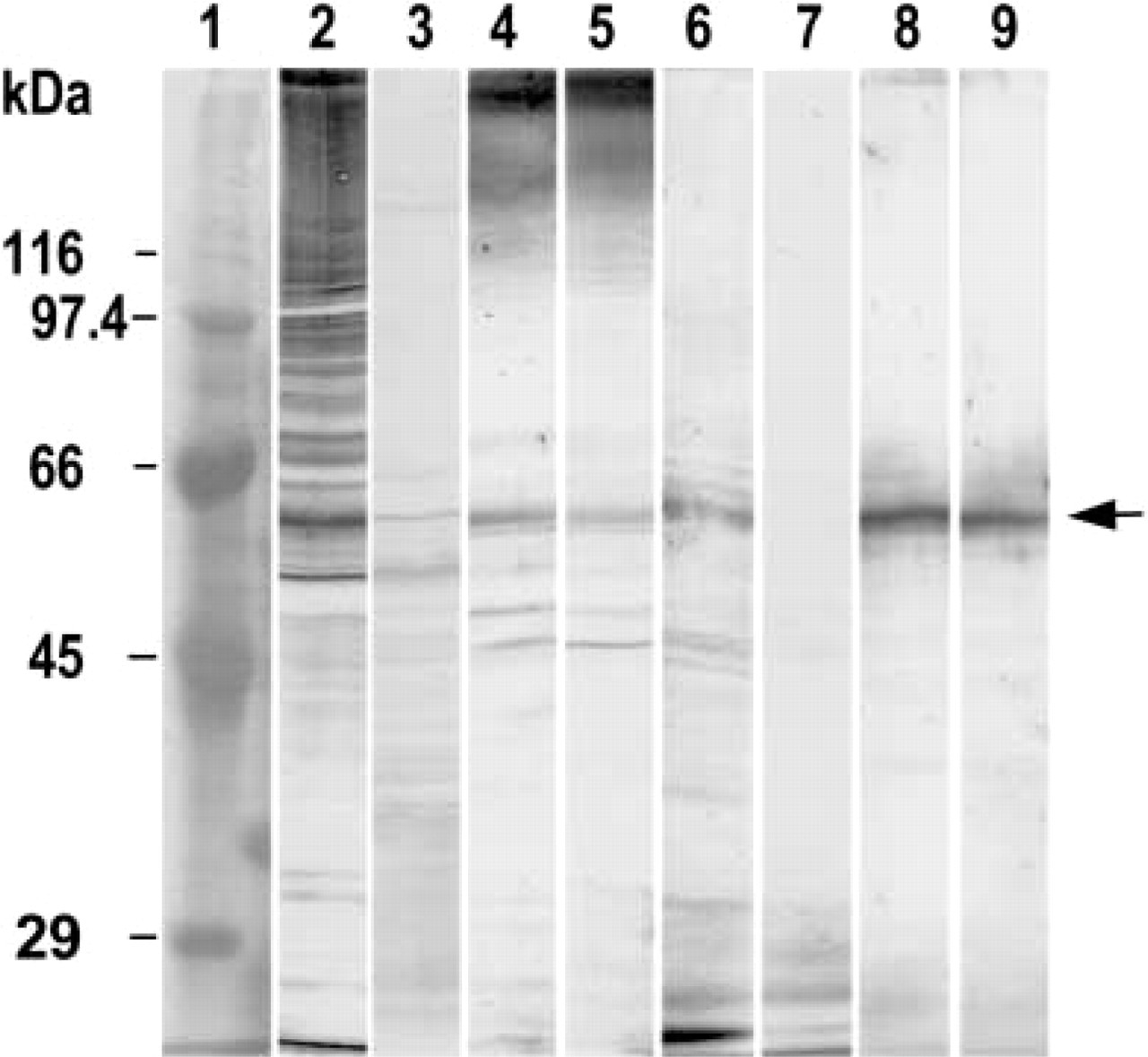
Western blot analysis of rat testis homogenate and epididymal sperm. Lane 1; molecular markers. Arabic numerals indicate molecular mass of marker proteins. Lanes 2–5, testis homogenate; lanes 6–9, epididymal sperm. Lanes 2 and 6, incubation with UB1 antibody; lanes 3 and 7, incubation with UB1 absorbed with bovine erythrocyte UB; lanes 4 and 5, incubation with FK1 antibody; lanes 8 and 9, incubation with FK1 absorbed with bovine erythrocyte UB. A band with a molecular moss of 61 kD (arrow) was detected in testis homogenate and sperm extract with both UB1 and FK1 antibodies.
Immunofluorescence Staining for UB
When the frozen sections of rat testis were stained with UB1, recognizing ubiquitinated proteins and free UB, fluorescence signals were observed in spermatids and interstitial cells, including Leydig cells, fibroblasts, and macrophages. The staining intensity in the seminiferous tubules greatly changed depending on the stage of development (Figures 2A–2C). The seminiferous tubules at stages IV to XII were most heavily stained (Figure 2B). In the seminiferous tubules at stages VII and VIII, acrosomes were stained (Figure 2A). At stage VIII, sperm heads were stained (Figure 2C). No staining was observed in the sections when the UB1 antibody was absorbed by UB (Figure 2D). When the sections were incubated with FK1, recognizing specifically polyubiquitinated proteins, elongated nuclei at stages VIII to XII were strongly stained (Figure 2E). At stages IV to XII, residual bodies were strongly stained by FK1 antibody (Figure 2F). In the smear preparation of epididymal sperm, the middle piece was heavily stained by UB1 antibody, whereas the head, the principal piece, and the end piece were very weakly stained (Figure 2G). But a spot strongly stained by UB1 appeared in the neck region, and intermediate staining was noted in the dorsal region of the head (Figure 2H) when the formaldehyde-fixed sperm smear preparation was digested with trypsin (1 mg/ml) for 3 min.
Immunogold Staining for UB
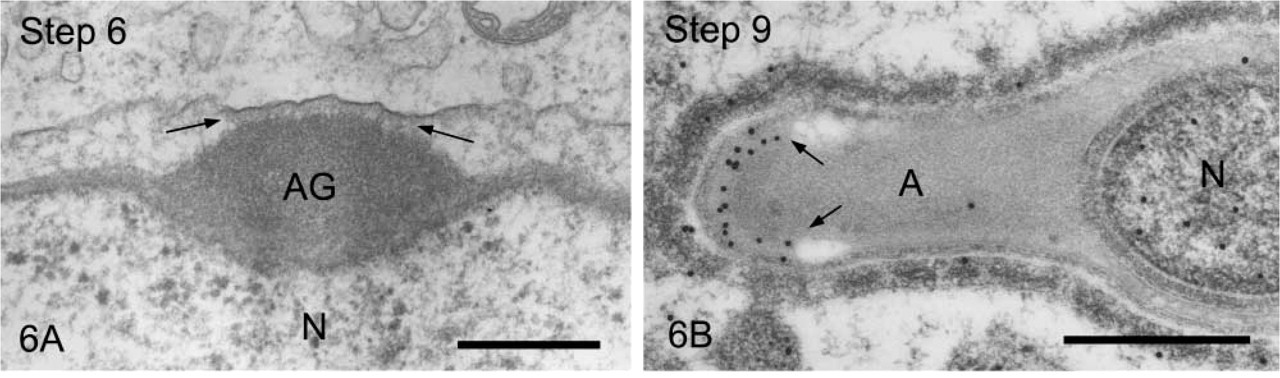
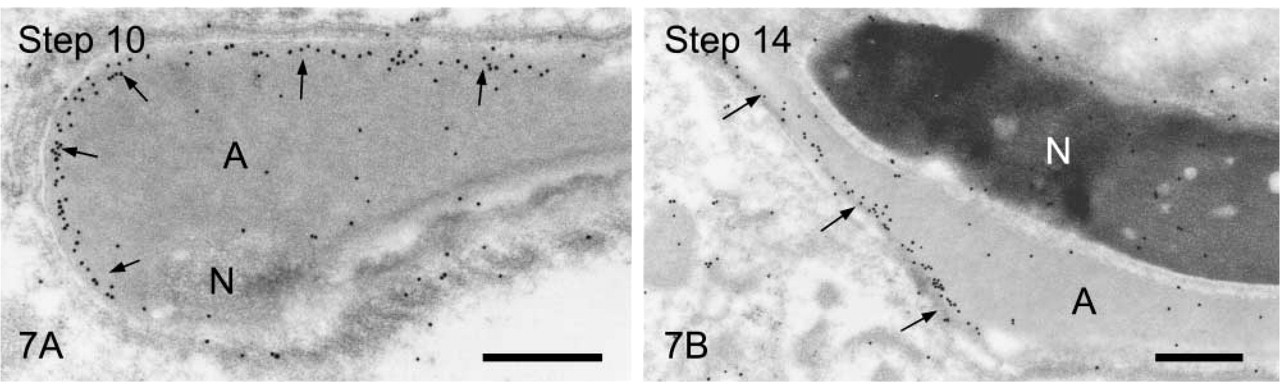
We first observed the staining patterns for UB1 antibody and then compared those for UB1 and FK1. In the sections incubated in the unabsorbed UB1 antibody, gold particles were observed throughout the differentiation, whereas no UB signals were detected in the sections incubated in UB1 antibody absorbed with free bovine erythrocyte UB (Figure 3A). Labeling was observed in the chromatoid body, the nucleus at specific steps, the cytoplasm of the residual body, and the mitochondria of the middle piece (data not shown). The stage of differentiation of spermatids was determined using morphological criteria proposed by Russell et al. (1990). In step 1–2 spermatids, an acrosomal vesicle appeared in the juxtanuclear area that contained a small acrosomal granule in the middle of the matrix. In immunoelectron microscopic control sections incubated with free UB-absorbed UB1, no gold particles were observed (Figure 3A). A few gold particles showing UB were associated with the acrosomal granule, whereas heavy gold labeling was present in Golgi vesicles surrounding the acrosomal vesicle (Figure 3B). Gold particles were closely associated with the outer surface of vesicles in the trans-Golgi network or those surrounding the acrosomal vesicle (Figure 3B). In step 3–4 spermatids, the acrosomal vesicle flattened and attached to the nuclear membrane. Labeling of UB1 was observed in Golgi vesicles in the vicinity of the developing acrosome (Figure 4A). In the acrosome, gold particles were present in the acrosomal granule and in a narrow space between the outer acrosomal membrane and the acrosomal granule (Figure 4A). The matrix of the acrosome containing fine fibrous materials was mostly negative for UB (Figure 4A). In a high-power view of the trans-Golgi area, some gold particles were found to be closely associated with the electrondense material contained in coated vesicles and others with the membranes of uncoated vesicles (Figure 4B). In step 5–6 spermatids, the acrosomal vesicle further expanded over the apical pole of the nucleus and the acrosomal granule was tightly attached to the nuclear membrane. At step 5, the Golgi complex was still located near the acrosome and labeling for UB was present in Golgi vesicles (Figure 5A). In the acrosome, gold particles for UB were observed in the acrosomal granule and in the narrow space between the outer acrosomal membrane and the acrosomal granule (Figure 5A). In step 6 spermatids, gold labeling of the outer acrosomal membrane was confined exclusively to this narrow space, where the particles were closely associated with an electron-dense fibrous material (Figure 5B). This material was also observed in the space between the outer acrosomal membrane and the acrosomal granule on routine electron microscopy (Figure 6A). In step 7–8 spermatids, the acrosome continued to expand and covered almost half of the spermatid nucleus. The Golgi complex moved away from the acrosome and migrated caudally to the basis of the developing flagellum. Labeling of UB was noted in the acrosomal granule and in the narrow space between the outer acrosomal membrane and the acrosomal granule (data not shown). In step 9–10 spermatids, the spermatid nucleus was elongated and the acrosome expanded to cover most of the nucleus. Heavy labeling for UB was detected in a specific portion of acrosome that seemed to correspond to the region between the outer acrosomal membrane and the acrosomal granule (Figure 6B). This strongly stained portion extended along the outer acrosomal membrane in some sections (Figure 7A), suggesting that the heavily stained portion formed a belt stretching from the apex to the caudal portion of the nucleus. The acrosomal matrix derived from the acrosomal granule was weakly stained. In spermatozoa obtained from the caudal epididymis, weak labeling for UB was noted in the acrosomes but not in the nuclei (Figure 8).
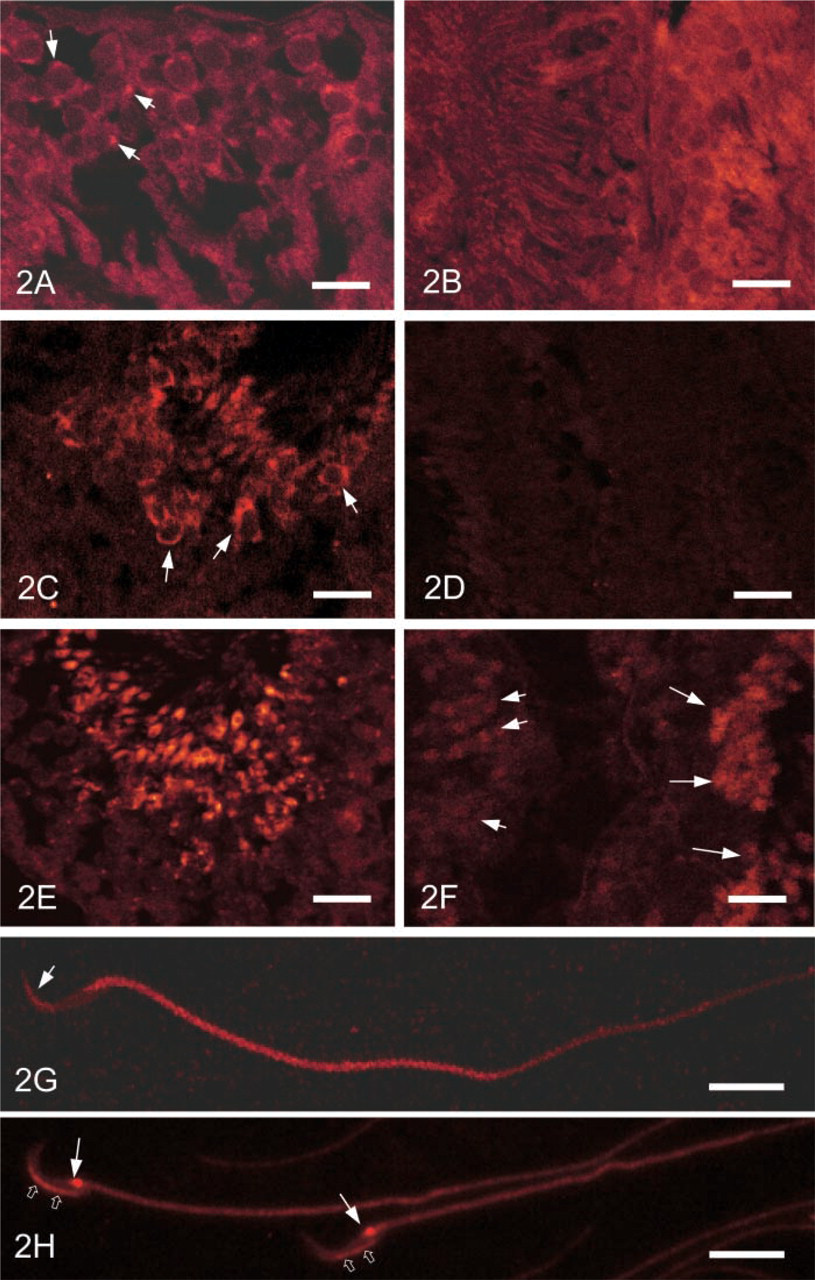
Immunofluorescence staining of rat testis. (
Next we compared the staining patterns obtained using antibody UB1 with those for antibody FK1. The most remarkable difference was observed in the staining of the fibrous material between the outer acrosomal membrane and the acrosomal granule; UB1 stained this structure heavily, but FK1 did not stain it at all (Figures 9A and 9B). In the Golgi apparatus, UB1 stained the trans-Golgi network strongly but the cis-Golgi network very weakly (Figure 9C). FK1 labeled the cis-Golgi network heavily and the trans-Golgi network lightly (Figure 9D).
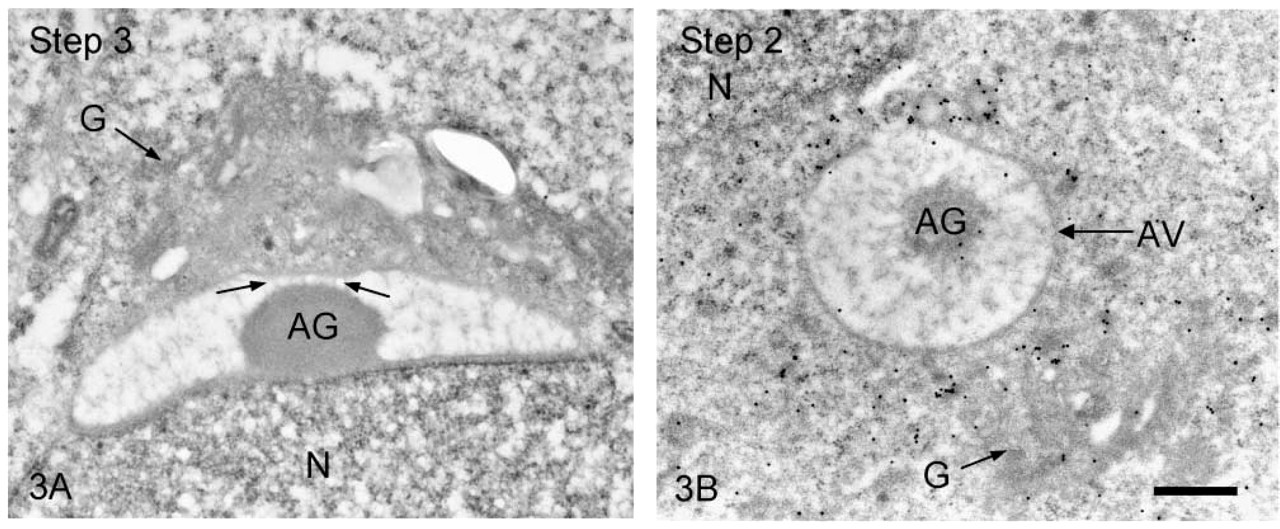
(
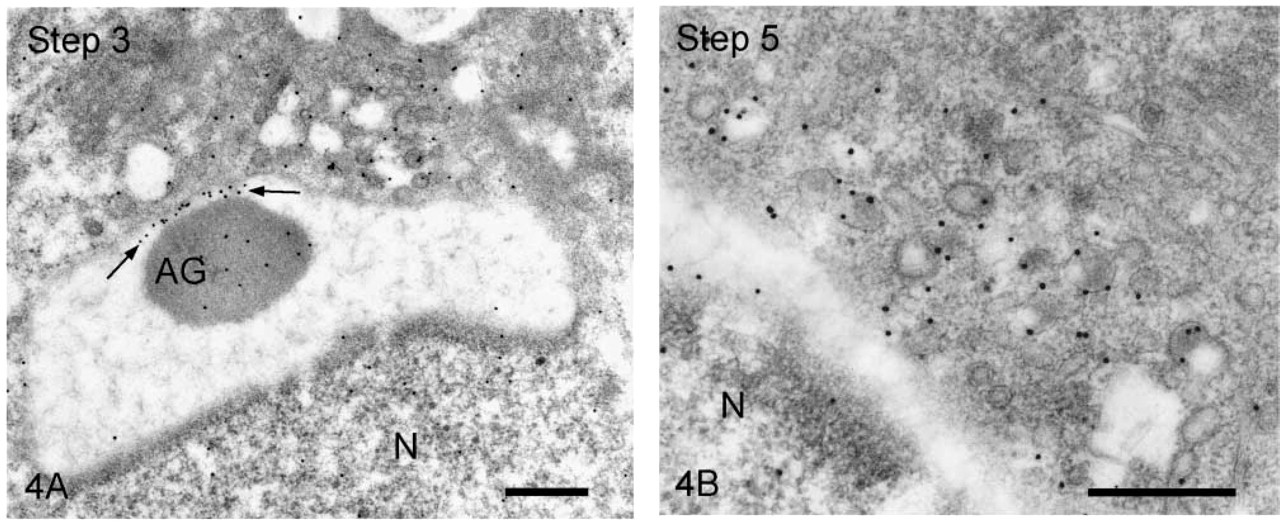
Spermatids stained with UB1. (
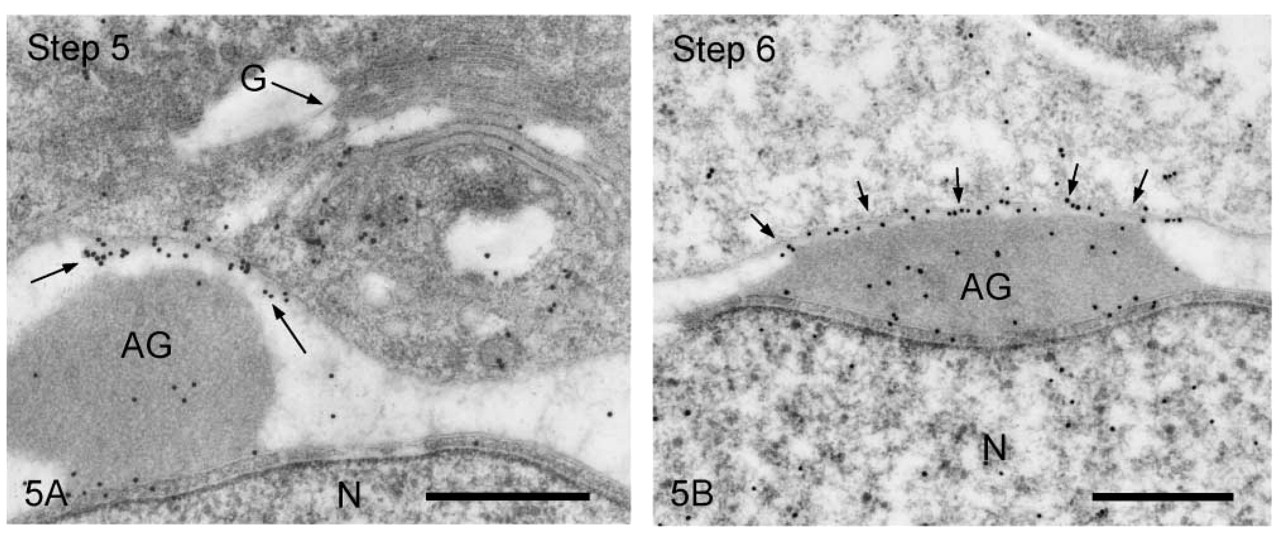
Early spermatids stained with UB1. (
(
(
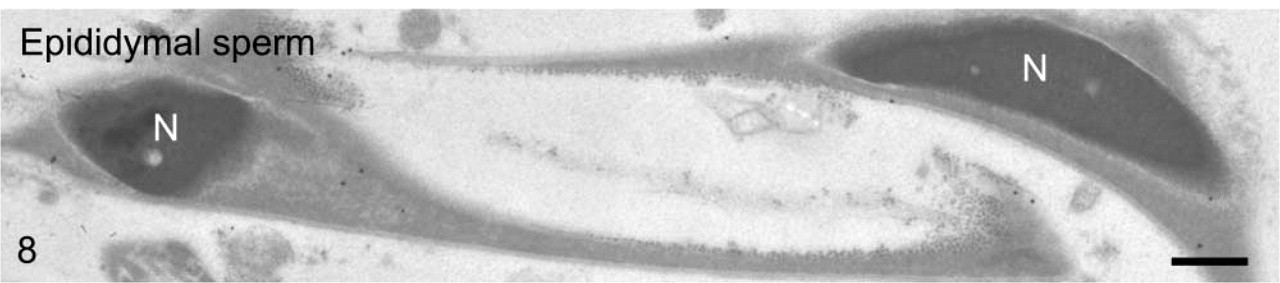
Epididymal sperm stained with UB1. The acrosomes are stained very weakly but the nuclei are not stained. Bar = 0.5 μm.
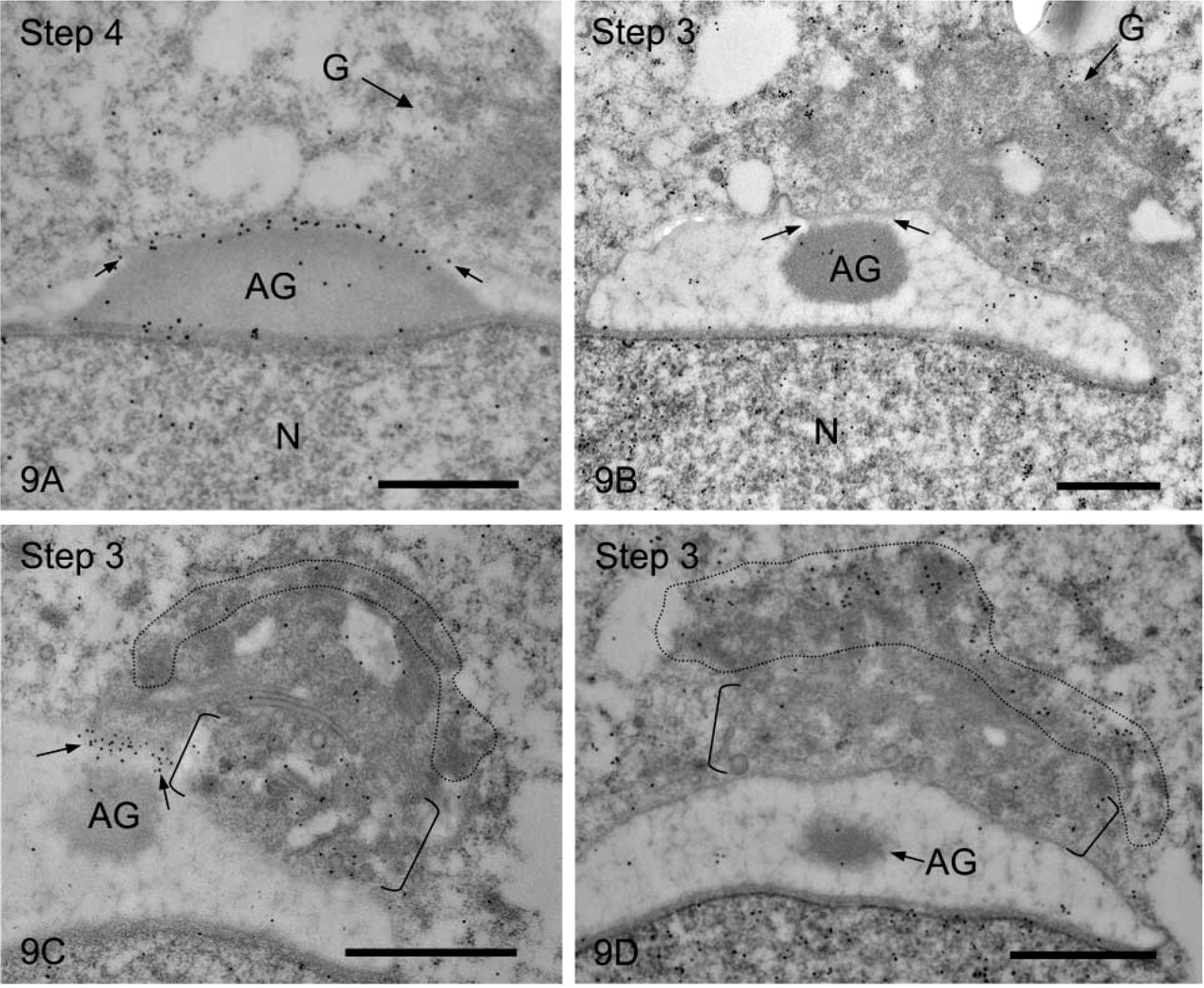
Comparison of immunostaining in acrosomes and Golgi regions obtained with antibodies UB1 and FK1. (

Multivesicular bodies of early and late spermatids stained with UB1. (
Next, we focused on the staining of multivesicular bodies. In pachytene spermatocyte, multivesicular bodies were weakly labeled by UB1 (Figure 10A), whereas in the elongating spermatids, strong UB signals were noted in multivesicular bodies (Figure 10B). Staining of the multivesicular bodies with FK1 was very weak (data not shown). Thus, it was shown that the staining patterns were markedly different between UB1 and FK1.
Quantitative Distribution of UB Labeling in Golgi Apparatus, Fibrous Material, and Multivesicular Bodies during Spermatogenesis
To confirm the differences of specificity for UB1 and FK1 antibodies, we carried out a quantitative analysis of the distribution of UB labeling. The distribution of ubiquitinated proteins in cis- and trans-Golgi networks, the outer membrane of the head cap, and the multivesicular bodies of spermatogenic cells during spermatogenesis is summarized in Table 1. The distribution revealed using antibody UB1 was quite different from that obtained with antibody FK1. The results obtained with each antibody were just the opposite. The cis-Golgi network of early spermatids was strongly labeled with FK1 but was not labeled with UB1. The trans-Golgi network of spermatids was stained heavily with UB1 but was not stained with FK1. The fibrous material between the acrosomal granule and the outer membrane of the head cap was markedly labeled with UB1 but was not labeled with FK1. Multivesicular bodies of spermatids at steps 4-15 were stained heavily with UB1 but not at all with FK1.
Discussion
In the testis homogenate, broad staining was observed after incubation with the UB1 or FK1 antibody. Staining with UB1 was almost eliminated by absorption of the antibody with bovine erythrocyte UB, whereas staining with FK1 was not abolished by the absorption with free UB. The results show that the FK1 antibody recognizes polyubiquitinated proteins as described previously (Fujimuro et al. 1994) and that the UB1 antibody reacts with monoubiquitinated proteins, the terminal UB of the polyubiquitin chain, and free UB. In epididymal sperm extract, signals produced by both antibodies greatly decreased, compared with those in testis homogenate. Signals produced by UB1 were eliminated by absorption of the antibody with free UB, but those produced by FK1 were not. A band with a molecular mass of 61 kD was present in the homogenate and sperm extract and was visualized with both UB1 and FK1 antibodies.
Quantitative distribution of UB signals at individual steps of spermatogenesis a

a+/−, <9 in labeling density (gold particles/μm2); +, 10-19; + +, 20-29; + + +, 30-39; + + + +, >40.
bLabeling density is shown as gold particles/μm.
cData obtained with antibody UB1.
dData obtained with antibody FK1 specific for polyubiquitinated proteins.
In the present immunofluorescence analysis, acro-somes were stained with the antibody UB1. FK1 stained mainly the nuclei of spermatids, especially elongated nuclei, which were strongly stained, but not acrosomes. Both antibodies stained residual bodies with similar intensity. In epididymal sperm, a strong signal from UB1 was detected in the middle piece but only a very weak UB signal was found in the acrosome. However, when sperm was digested with trypsin, the dorsal region of the head and a small spot in the neck were strongly stained. This suggests that UB molecules are masked during passage through the epididymal tract. It is well known that the maturation of sperm occurs in the epididymal tract (Bearer and Friend 1990; Yanagimachi 1994). A major event in this maturation is believed to be the change in plasma membrane components of sperm. Changes of immunofluorescence staining for acrosomal membrane surface antigens during the maturation of spermatozoa have been reported (Phillips et al. 1991). It was also reported that cholesterol in the membrane of spermatozoa was dramatically modified during the maturation process (Pelletier and Friend 1983). The present study suggests that the masking of ubiquitinated protein(s) occurs in the acrosome during maturation. Recently, it was suggested that the numbers of ubiquitinated spermatozoa decreased progressively between the caput and caudal epididymis (Sutovsky et al. 2001). Defective spermatozoa were ubiquitinated and might be phagocytosed by epididymal epithelial cells during the epididymal passage (Sutovsky et al. 2001). Thus, it was proposed that the ubiquitination of spermatozoa functioned as a quality-control system for sperm (Sutovsky 2003). The role of the masking of ubiquitinated protein(s) in the head and neck during maturation is not clear. It is likely that ubiquitinated protein(s) is used in the egg after fertilization.
Recently, a conserved UB-interacting motif (UIM), XEDEXLXXAXXXSXXEXXX, was proposed (Hofmann and Falquet, 2001), which was found in Eps15, Epsin, and HRS (Polo et al. 2002; Raiborg et al. 2002; Shih et al. 2002). Thus, we carried out a cyber screening of 295 acrosomal proteins and found two candidates, sea urchin sperm receptor and pig acrosomal protein (sp32), having sequences that nearly matched the motif. It is likely that these proteins are ubiquitinated. Although we reported that cathepsin H showed the same distribution as UB in the acrosome of differentiating rat spermatids in a previous study (Haraguchi et al. 2003), cathepsin H was not a candidate because it has no UIM motif.
In the present study, UB signals were present in the Golgi vesicles. Careful observation revealed two distribution patterns: gold particles were associated with the outer side of the vesicle membrane and were found on dense material within the vesicles. It was reported that several membrane proteins functioning as a receptor or a transporter are monoubiquitinated at the cytoplasmic tail and internalized through coated vesicles and then transported to endosomes to be degraded (Hicke 1999; Shih et al. 2000; Katzmann et al. 2002; Polo et al. 2002; Raiborg et al. 2002,2003; Haglund et al. 2003). Thus, evidence that UB is used for protein sorting into endosomes is accumulating. However, there has been no report so far that UB is used for the transport of proteins from the Golgi apparatus to the endoplasmic reticulum or to other compartments. In the present study, polyubiquitinated proteins revealed with the antibody FK1 were localized to the cis-Golgi network, but a few existed in the trans-Golgi network and in multivesicular bodies in which UB signals, including monoubiquitin, were detected using the UB1 antibody. In addition, no signals for polyubiquitinated proteins were detected in the fibrous material between the outer acrosomal membrane and acrosomal granules. These differences were confirmed by the quantitative analysis of ubiquitinated proteins stained with UB1 and FK1 (Table 1). Thus, the antibody FK1 recognized polyubiquitinated proteins as described (Fujimuro et al. 1994), whereas our antibody UB1 reacted preferentially with monoubiquitin on thin sections. It is likely then that the signals in both the Golgi vesicles and the fibrous material show monoubiquitinated proteins and those in the cis-Golgi show polyubiquitinated proteins. The results suggest that monoubiquitin is involved in the selective transport of proteins between the Golgi apparatus and the acrosome. It has been shown that the sorting and delivery of proteins between the Golgi apparatus and the acrosome are less precise and that Golgi-resident proteins are present in the acrosomes of round spermatids (Moreno et al. 2000). However, Golgi proteins mistransported to the acrosome are retrieved to the Golgi apparatus at later stages of differentiation and are completely absent in immature spermatozoa, suggesting that active anterograde and retrograde vesicular transport traffic pathways using β-coatmer protein-and clathrin-coated vesicles operate between the Golgi apparatus and the acrosome (Moreno et al. 2000). The present study showed that monoubiquitin-like signals in Golgi vesicles were more abundant in round than in elongating spermatids. This suggests that monoubiquitin is involved in anterograde transport between the Golgi apparatus and the acrosome. In addition, our results showed that in elongating spermatids, multivesicular bodies were heavily stained with UB1. It is likely that in the elongating spermatids, monoubiquitin is involved in the transport from the plasma membrane to multivesicular bodies. This suggests that in differentiating spermatids, monoubiquitin is involved in two different vesicular transport pathways: the transport from the Golgi apparatus to the acrosome, which occurs in early spermatids, and the transport from the plasma membrane to multivesicular bodies, which functions in late spermatids.
Footnotes
Acknowledgements
Supported in part by Grant-in-Aid 14580693 and the Center of Excellence Program from the Ministry of Education, Science, Culture, and Sport of Japan (SY).
