Abstract
Using artificial intelligence (AI) to enhance chimeric antigen receptor (CAR)-based therapies’ design, production, and delivery is a novel and promising approach. This review provides an overview of the current applications and challenges of AI for CAR-based therapies and suggests some directions for future research and development. This paper examines some of the recent advances of AI for CAR-based therapies, for example, using deep learning (DL) to design CARs that target multiple antigens and avoid antigen escape; using natural language processing to extract relevant information from clinical reports and literature; using computer vision to analyze the morphology and phenotype of CAR cells; using reinforcement learning to optimize the dose and schedule of CAR infusion; and using AI to predict the efficacy and toxicity of CAR-based therapies. These applications demonstrate the potential of AI to improve the quality and efficiency of CAR-based therapies and to provide personalized and precise treatments for cancer patients. However, there are also some challenges and limitations of using AI for CAR-based therapies, for example, the lack of high-quality and standardized data; the need for validation and verification of AI models; the risk of bias and error in AI outputs; the ethical, legal, and social issues of using AI for health care; and the possible impact of AI on the human role and responsibility in cancer immunotherapy. It is important to establish a multidisciplinary collaboration among researchers, clinicians, regulators, and patients to address these challenges and to ensure the safe and responsible use of AI for CAR-based therapies.

Keywords
Introduction
An innovative method for curing cancer patients involves employing immune system components called cancer immunotherapy. In this context, several treatment approaches include antibodies that attach to proteins produced by cancer cells and prevent them from performing their intended functions. 1 Alternative strategies include vaccinations, native T-cell injections, and the use of immune cells that have been altered, such as natural killer (NK) or chimeric antigen receptor (CAR)-T cells. 2 Tumor-reactive T-cell adoptive cell transfer (ACT) is a successful treatment strategy for solid and hematological cancers. 3 ACT causes tumor cells to become more powerful, enabling them to successfully eliminate cancerous cells in vivo. 4 The acquisition of tumor-infiltrating lymphocytes, their extensive in vitro culture to achieve an appropriate frequency by the use of cytokines, and ultimately their transplantation into the patient were all necessary according to ACT guidelines. 5 To specifically identify and eradicate tumor cells, the use of CAR-based treatments (such as CAR-T cells and CAR-NK cells) has gained interest recently.6,7
The tumor microenvironment (TME) contains both human T cells and NK cells, which play a major role in tumor immune surveillance. While NK cells produce cytolytic granules, inflammatory cytokines, and chemokines that stimulate both the innate and adaptive immune systems, they also have strong antitumor effects. T cells normally provide long-lasting, antigen-specific, effector, and memory immune responses. 8 It is now easier to genetically alter T and NK cells to produce tumor antigen-specific CARs because of advancements in cellular engineering methods.9,10
Artificial intelligence (AI) has undergone decades of intense development, and now it refers to a wide range of technologies, including DL, machine learning (ML), and artificial neural networks.11–13 AI’s DL is a significant area 13 and can automatically extract features from enormous datasets. Furthermore, DL may identify information in pictures that the human eye is unable to detect.14–16 This has major implications for early tumor diagnosis using imaging data. AI can aid in tumor diagnosis and treatment as well. With its robust capacity for logical reasoning and learning, AI frequently uses multilayer neural network structures to imitate human thought processes exceptionally well.17,18 AI can make the quickest and most intuitive decisions to address issues, like the human brain. It is not simple to infer that AI has the potential to significantly optimize current investigations into cancer models.
AI is a remarkably sophisticated approach in the quest for computer-assisted cancer immunotherapy. Although the technology is still distant from being widely used in clinical practice, it can expand its practical uses in the response to immunotherapy with the growing amount of clinical data and better AI techniques. We anticipate a bright future in which AI will probably change the way cancer immunotherapy is performed, ultimately leading to better patient safety and healthcare.
Landscape of cancer immunotherapy
Various approaches to cancer immunotherapy and its current state
CAR T-cell therapy
An increasing number of people are turning to CAR T-cell therapy as a means of treating cancer and other diseases. 19 Medical treatment for multiple myeloma, acute lymphoblastic leukemia, and B-cell lymphoma may now include CAR T-based medications. Furthermore, their potential use is also being investigated in the management of non-small-cell lung cancer, 20 glioblastoma tumors, 21 human immunodeficiency virus, 22 heart damage, etc. CAR T treatments can mobilize the patient’s immune system to begin an intentional assault on cancer cells that gives them the edge. 23 Although CAR T-cell treatments show potential as cancer therapies, their efficacy is limited because the TME contains signals that inhibit the immune system. 24 It has been shown that the programmed cell death protein 1 (PD-1) signaling pathway considerably reduces T-cell activity in the TME, particularly in adoptive CAR T-cell therapy. The immunological checkpoint receptor PD-1 (CD279) is more highly expressed in activated T cells. 25
An immunosuppressive response is triggered when programmed cell death ligand 1 (PD-L1) or programmed cell death ligand 2 (PD-L2) binds to PD-1. This response reduces regulatory T-cell activity and enhances apoptosis of effector T cells, hence suppressing inflammatory T-cell activity. 26 Although PD-L2 expression is low, PD-L1 is abundant across many cancer types, contributes to immune evasion, and decreases the efficacy of CAR T cells. 26 To reestablish the efficacy of CAR T cells in TMEs rich in PD-L1, it is of great importance to suppress the interactions between PD-1 and PD-L1. As a result, preserving CAR T efficacy in PD-L1-rich TMEs requires investigating whether preventing PD-1 and PD-L1 interactions is a feasible approach. 27 While blocking antibodies in conjunction with CAR T-cell therapy has several benefits, using them alone might cause autoimmune responses and systemic reduction of PD1 activation. 28
Since the present method of CAR T therapy involves producing autogenic T cells that express the exogenous CAR, it is feasible to include targeted PD-1 suppression to produce CAR T cells with reduced PD-1 signaling while preserving the PD-1 pathway in some manner. To disrupt the PD-1 pathway, researchers have investigated several techniques, including CRISPR-Cas9-mediated ablation of PD-1 and CAR engineering, with encouraging results,29,30 using a combination of a soluble anti-PD-1 single-chain variable fragment (scFv) and the creation of a short hairpin RNA that targets PD-1. However, most of these methods rely on changing the genome in some way, either by introducing new genes via viral vectors or by CRISPR-Cas9 genome editing. Because T cells continue to become sensitized to this anti-inflammatory signaling pathway, this suggests that the consequences of PD-1 inhibition are long-lasting and might cause autoimmune reactions. T cells possess an inherent mechanism called the RNA-induced silencing complex that can temporarily mute genetic transcripts. Thus, a fascinating and mostly unexplored alternative to traditional approaches is the transitory interruption of the PD-1 signaling pathway by RNA interference at the transcriptome level. 30
Immune checkpoint inhibitors
Immune checkpoint inhibitors, also known as ICIs, are monoclonal antibodies (mAbs) that target the inhibitory checkpoint molecules that are expressed on the cell membranes of CD4+ T cells and antigen-presenting cells.31,32 The discovery of ICIs has opened a new front in the fight against various types of cancer, including melanoma, kidney cancer, and lung cancer. It is expected that this will lead to a change in the current standard treatments for many cancers. 33 Table 1 shows some of the important ICIs.
Some important ICIs and their description.
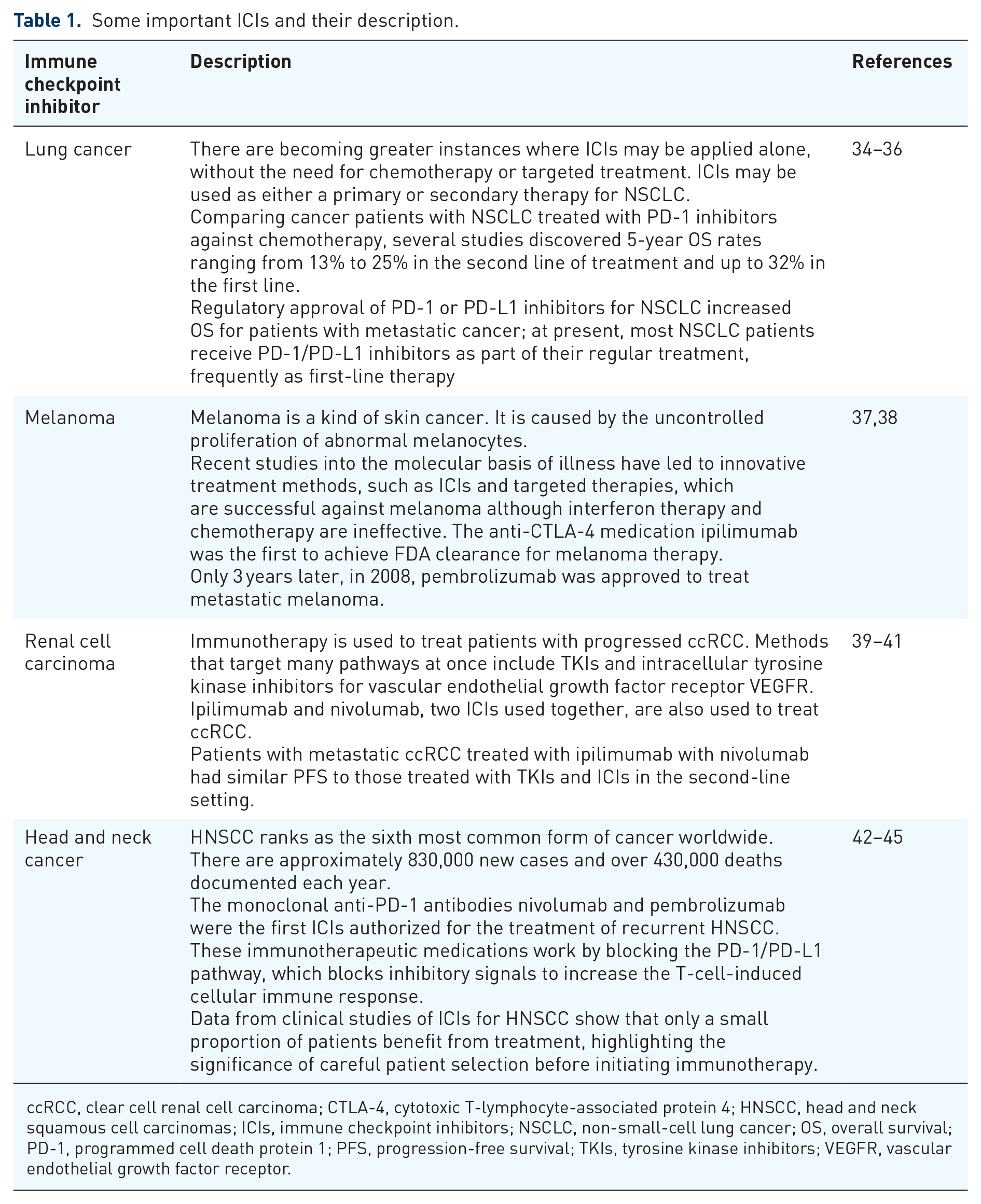
ccRCC, clear cell renal cell carcinoma; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; HNSCC, head and neck squamous cell carcinomas; ICIs, immune checkpoint inhibitors; NSCLC, non-small-cell lung cancer; OS, overall survival; PD-1, programmed cell death protein 1; PFS, progression-free survival; TKIs, tyrosine kinase inhibitors; VEGFR, vascular endothelial growth factor receptor.
Therapy based on ICIs is also emerging as an appropriate choice due to its significant antitumor effects. 46 However, the therapeutic value of ICIs is limited by tumor resistance to ICIs and ICI-mediated toxicity. Overall, approximately 60% of patients do not have a clinically significant response to therapy with ICIs. 47 Changes in the TME that maintain angiogenesis and prevent immune cell antitumor responses are mostly responsible for the development of resistance to ICIs and other cancer immunotherapies. The genes, inflammation, unusual neovascularization, and other variables involved in tumor immune resistance are all part of the problem. 47 Combining chemotherapy with inhibitors of angiogenesis and immunotherapy for cancer has solid evidence due to the essential role of angiogenesis in tumor progression and opposition to immunotherapies.48,49
Challenges and limitations of immunotherapy in clinical practice
The trial design must identify significant results with regard to the medicine’s particular features, pharmacokinetic (PK)/pharmacodynamics (PD) profile, and clinical behavior if the drug is to be considered a success.50,51 Clinical studies that are not optimized may not pose the appropriate questions; therefore, the results may not reflect the agent’s true potential. Many patients do not react to ICIs, although their appeal depends on their ability to provide long-term and even full responses. Whenever thoughts such as pseudoprogression (PP) or hyperprogression (HP) develop in relation to ICI treatment, it may be difficult for clinicians to determine whether a patient has a real therapeutic impact. 52 When compared to traditional oncology components, the adverse effects of ICIs tend to be more variable in terms of their duration, frequency, and severity. Immune-related adverse events (irAEs) might be different depending on the modality class; however, the adverse reactions of ICIs are generally the same yet have a wide frequency range. 53 First, irAEs caused by ICIs tend to damage a single organ instead of many organs simultaneously. 54 Second, toxicity often does not start until much later in treatment (about the fourth week for ipilimumab 55 and around the tenth week for nivolumab). 56 The toxic effect caused by ICIs is not inversely proportional to the dose; therefore, reducing the dosage may not prevent future adverse effects. 57 Since ICI combination techniques more accurately represent immunological damage, they are often used to stop therapy permanently. Thus, it has been shown that 42% of melanoma patients receiving chemotherapy combined with nivolumab and ipilimumab had to stop treatment due to any drug-related adverse events. 58 These occurred at a rate of 13% in the nivolumab arm and 15% in the ipilimumab arm. 58
Innovation and personalized approaches
Biomarkers, personalized medicine, imaging technologies, and wireless monitoring devices have all advanced greatly because of the enormous amount of data obtained from each cancer patient’s unique physiological and clinical features (i.e., genomics, radiomics, metabolomics, and other omics). AI has become a useful tool for doctors to provide higher-quality treatment. 59 Its enormous data analysis abilities may help provide personalized therapy utilizing its recommendations. AI may have a major effect on a variety of processes, such as cancer prevention, medication development, and genomic-based therapies. 60 Molecular biologists and computer scientists are working together to acquire new insights into tumor biology due to advances made possible by AI. Since cancer is a genetic illness, oncology would obtain the most significant benefits from advances in AI. Analyzing DNA methylation in tumors may help with categorization and prognosis. The potential for machine-determined DNA methylation to classify over 70% of tumors classified by humans holds great promise for revolutionizing cancer prognosis and therapeutic choices. In a seminal study from Capper et al., 61 82 different types of brain cancers could be accurately classified with a 93% success rate using whole-genome methylation studies conducted on tumor specimens using the Illumina HumanMethylation450 (450 k) or Methylation EPIC (850 k) array platforms. The authors asserted a much greater level of accuracy when compared to pathologists. Watson for Oncology, a particular assistant-decision system, has shown satisfactory concordance with the choices made by multidisciplinary groups. This may facilitate quick, less resource-intensive decision-making at the patient level. 62
The role of AI in cancer immunotherapy
Figure 1 shows molecular oncology and the role of artificial intelligence in cancer immunotherapy.

Molecular oncology and AI.
AI in predicting immunotherapy responses
The ability to program computers to carry out particular tasks and then provide decision support systems is an important way forward for the field of AI medicine; early findings show that it can correctly differentiate between immunohistochemically scored subtypes of cancer, tumor biomarkers, and prognostic indicators. 63 In addition, the use of AI techniques in cooperation with modern technology holds enormous potential for optimizing healthcare by reducing the prevalence of misdiagnoses and enhancing the individualized nature of patient care. 64 With immunotherapy’s impressive advancement and widespread use in cancer treatment, it remains a challenge to choose the patients most likely to benefit from treatment. Effective immunotherapy is increasingly probable now that AI can help anticipate a patient’s susceptibility to immune checkpoint blockade (ICB) treatments by the creation of immunotherapy-associated scores. 65 In future AI practices, there is still a shortage of integrated ICB response prediction models, which is a barrier caused by the undetermined impact of solo predictive biomarkers, the difficulty of integrating different biomarkers into one system, and the fact that there were previous challenges.
Using AI, it is possible to recognize major histocompatibility complex (MHC) features related to immunotherapy response with an accuracy of 90% or higher. 66 In addition, AI may be applied to perform consistent evaluations within institutions prior to the clinician’s assessment. Thus, patients may benefit more from cancer immunotherapy if AI is applied to the field. 67
AI predicting the efficacy of immunotherapy
Acquiring, filtering, segmenting, extracting, and selecting features from multiscale medical data from the training cohort is the first step in using AI to predict the efficacy of immunotherapies. Then, features are fed into the AI for learning and modeling.68,69 Genomics, proteomics, pathology tissue, computed tomography (CT)/MR imaging-omics, and many other types of multiscale medical data are all within the boundaries of possibility. Training AI to accurately identify which patients need further testing, such as whole-genome sequencing, or to determine whether immunotherapy will be beneficial for a certain patient is the ultimate aim. It also helps in determining whether an immunotherapy treatment will be effective.
Existing approaches to predict the outcomes of immunotherapy
The existing approaches that are being used for the prediction of immunotherapy are summarized in Table 2.
Existing approaches that are currently in use to predict cancer immunotherapy.

AI, artificial intelligence; ICIs, immune checkpoint inhibitors; NGS, next-generation sequencing; WGS, whole-genome sequencing.
Evidence suggests that PD-L1 expression, TME, tumor immunology locus, TP53 mutations, anaplastic lymphoma kinase (ALK) rearrangements, gut microbiota, Fc gamma receptor polymorphisms, serum complement levels, and other anomalies in microRNAs all have a role in immunotherapy response.81,82 It is possible to greatly improve prediction accuracy by merging these data with data from diseased sections and imaging histology to obtain multiomics integrated data. Organs that resemble tumors are useful for testing immunotherapies, as they can reproduce tumor conditions in vitro while still producing immune-tumor interactions. 83 By creating a practical instrument for tumor in vitro culture, growth analysis, medication screening, and tissue collection, AI in organoids is anticipated to tackle the safety and personalization concerns linked to traditional prediction tools. 84 By analyzing the cell necroptosis index and antigen presentation pathways, AI may be able to forecast ICB reactions. 85 Table 3 shows different methods that use AI to forecast the results of immunotherapy.
Different methods use AI to forecast the results of immunotherapy.

ALK, anaplastic lymphoma kinase; CT, computed tomography; EGFR, epidermal growth factor receptor; LDH, lactate dehydrogenase; PD-1, programmed cell death protein 1.
Precision medicine and AI: Future synergies
AI and precision medicine are merging to aid in resolving some of the most difficult challenges in patient-specific treatment. Five examples of personalized healthcare convictions that present inherent difficulties, however, may be responsive to improvement with the help of AI 88 as shown in Figure 2.
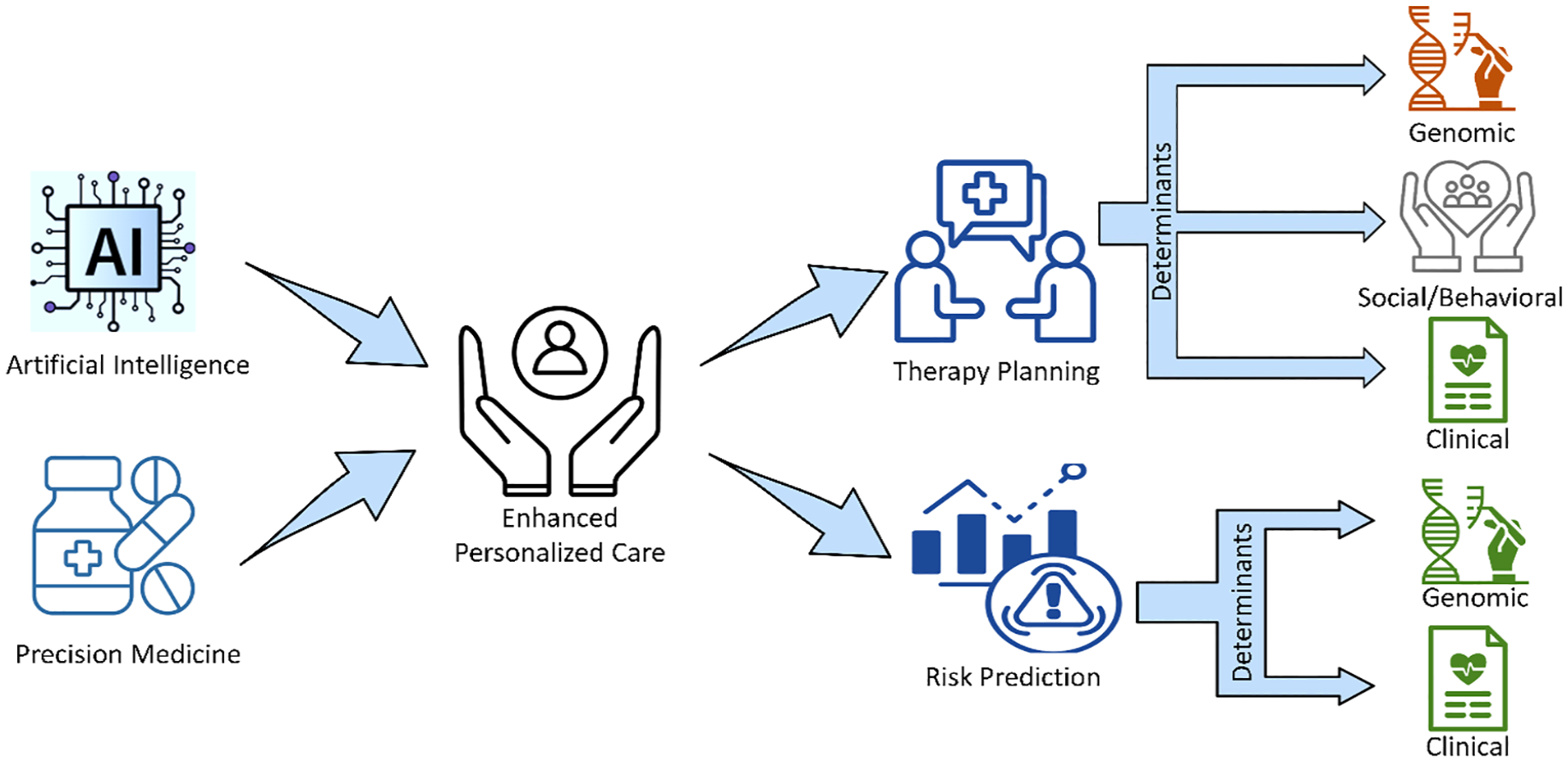
An overview of how precision medicine and AI combined help in enhancing personalized care.
Therapy planning and considerations
Table 4 shows the therapy planning and considerations (genomic, environmental, and clinical) with their possible outcomes and references.
Genomic, environmental, and clinical considerations.

AI, artificial intelligence.
Biomarker discovery and personalized immunotherapy
AI methods allow computers to become better over time at conducting a certain activity; the result is systems for decision-making that show potential in their ability to differentiate between, for example, immunohistochemistry scores, cancer subtypes, and biomarkers. 92 In addition, as Thompson et al. 64 investigated, AI, with its superior computational technology, may have the capacity to rebuild the specialty by minimizing mistakes and increasing parameters such as the specificity of patient therapy and dosage control. Although immunotherapy represents a major advancement in the treatment of cancer, it may be difficult to determine whether some patients will benefit from it. Effective cancer immunotherapy was previously improbable, but the emergence of AI has raised the probability due to its ability to anticipate treatment impact through the development of immunotherapy prediction scores such as immunoscore and immunophenoscore. 65 Both scores were designed to assist doctors in determining the probability of recovering from ICB treatment.
However, several restrictions need more studies, such as the absence of ICB response prediction models that can include multiple biomarkers and the unknown predictive ability of specific biomarkers. Integrating an AI-based diagnostic algorithm with clinicians’ interpretations was shown to improve the precision of diagnoses for difficult-to-identify cancer subtypes in prior research. 92 AI achieves a recognition accuracy of 91.66% for the MHC, patterns linked to immunotherapy response. 66 In addition, AI may be used to standardize tests across institutions, which reduces the requirement to depend on the interpretation of physicians, which can be naturally subjective occasionally.67,93 Thus, the use of AI in cancer immunotherapy might result in beneficial outcomes for patients such as the chronology of major breakthroughs of AI applications in cancer immunotherapy is shown in Table 5.
Chronology of major breakthroughs of AI applications in cancer immunotherapy.

AI, artificial intelligence; CNNs; convolutional neural networks; CT, computed tomography; ICB, immune checkpoint blockade; ML, machine learning; TNBC, triple-negative breast cancer.
Application of AI in current challenges of immunotherapy
Immunotherapy has made significant contributions to the development of cancer treatment in the clinic. The number of immunotherapy medication approvals, particularly in the class of ICIs, has been rising in conjunction with the advancement of therapies in the clinical and preclinical context.100,101 Therapies targeting T-cell immunoreceptors with immunoglobulin as well as ITIM domains, activation of lymphocyte gene 3 and T-cell immunoglobulin and mucin-domain 3, are currently in clinical trials or development for cancer immunotherapy, among many other new therapies targeting other prospects. Pembrolizumab, avelumab, and ipilimumab are three FDA-approved medications that target the inhibitory molecules PD-1, PD-L1, and CTLA-4. Although many molecular targets and treatments are very efficient, approximately 20% to 50% of patients truly react to therapy. There has been an increase in interest in the processes causing immune checkpoint resistance. Arlauckas et al. 102 discovered that macrophages present in tumors rapidly removed anti-PD-1 mAbs from T cells, reducing the effectiveness of the immune system’s cytotoxic T-cell responses. Impaired establishment of T-cell memory and inadequate production of antitumor T cells are two more possibilities. 103 As a result, there is an immediate requirement to establish methods for determining which patients may benefit most from immunotherapy. However, irAEs have emerged as persistent effects of checkpoint blockade, highlighting a significant clinical issue in ensuring the safe administration of these inhibitors.
Some irAEs, such as colitis or rash, appear quickly after using ICIs, whereas others, such as hypophysitis or liver damage, arise steadily. Some irAEs, such as dermatitis or pneumonitis, are easily treated because of the organ’s intrinsic regeneration capacity, while others, such as adrenal corticosteroids and insulin insufficiency, cause permanent tissue damage because of the death of endocrine organs. 104 In addition, the severity and frequency of irAEs greatly increase when combination therapies are used. Wolchok et al. 105 found that anti-CTLA-4 with anti-PD-1 treatment increased the risk of significant irAEs, such as inflammation of the heart and brain, in up to 60% of patients. These symptoms are most likely due to checkpoint networks’ physiological role in regulating adaptive immunity and preventing autoimmunity. Therefore, identifying the causes of irAEs and developing strategies for reducing their effects are becoming serious issues. To fully recognize and understand the biological processes of irAEs, a thoroughly accurate, standard database of irAEs has been developed. 106 The identified irAEs may be used as a benchmark against which to evaluate the accuracy of automated irAE extractions from other data sources, and they may lead the way for the development of computational techniques to learn about irAEs and, ultimately, ensure the safety of cancer therapy. Finally, expensive treatment might cause significant individual costs for patients as well as pressures on the national healthcare system. The potential use of AI technology in cancer research and therapy has become clearer.
AI approaches face challenges in the same way that a resident physician would: by first observing the patient, then using algorithms to filter variables and look for combinations that accurately predict results. Several areas of medicine have used AI-based strategies, the most common being pathology and radiology, and in certain instances, these techniques have exceeded the performance of human experts. 107 For example, the integration of AI into pathology would provide enhanced diagnostics and a better workflow, permitting physicians to evaluate and exchange pictures rapidly and utilize computational algorithms to assess significant findings to obtain a more enlightened and thorough cancer diagnosis. Medical images include an abundance of information that could assist in diagnosis, response to therapy, and prognosis, and these algorithms may be able to use this information in ways that would be invisible to a human investigator. In a backward study, Wang and Xu 106 established a semisupervised DL approach to data extraction from CT scans for predicting high-grade serous ovarian cancer recurrence. Searching for diagnostic and prognostic biomarkers and describing tumor cell phenotypes are only two examples of the expanding use of AI in immunotherapy. Effland et al. 108 used a DL-based variation network for joint reconstruction of images and segmentation to show how immune cells communicate with melanoma cells. By studying the antigen-presenting pathway, AI may be able to predict how the body would react to ICB. The low patient response rate, irAEs, and high hospitalization costs associated with cancer immunotherapy are only a few of the problems that AI provides as a possible solution. Houy and Le Grand 109 discovered that immunotherapy might benefit from the use of AI tools to improve the therapy schedule, which could lead to a reduction in irAEs by enabling the use of lower dosages and more cost-effective therapies.
AI in clinical diagnostics
The early detection and diagnosis of cancer patients, as well as their consequent treatment, depend on the development of highly accurate AI algorithms for the early identification of the disease. 17 The use of AI in clinical diagnostics has the potential to improve the treatment of patients. Clinical diagnostics could become more effective with the use of cancer screening methods, including mammography, radiography, and image processing. 110 AI algorithms have already been established with enormous datasets, and they demonstrate enhanced diagnosis compared to medical professionals. The efficacy of AI-assisted diagnostics in identifying cancer at diverse and complicated stages has been demonstrated across a variety of clinical datasets. 110 The Food and Drug Administration of the United States has given its approval to the use of a number of AI systems that are currently under development for use in some aspects of cancer treatment, such as the detection of suspicious lesions in cancer and the interpretation of magnetic resonance imaging or CT. 111 Several AI algorithms exist for cancer detection, tumor classification, treatment trend analysis, and dataset assessment. 111 For instance, AI algorithms exist to identify breast abnormalities and to determine lung nodules in patients with lung cancer.112,113
AI and new emerging technologies
AI and other cutting-edge technologies are currently changing patient care as they expand across the healthcare system. The amount of data that is now available has increased at an exponential rate, which may be used in the process of early diagnosis and making clinical decisions. 114 The change brought about by AI in the field of biomedical research is essential for the development of the idea of precision medicine. Alongside the growth of the field of precision medicine has come an even more significant change in the knowledge of the processes involved in the early identification of cancer through the use of digital technologies. In the field of cancer research, AI has mostly focused on predicting risk with the expectation that using risk information would impact both health habits and treatment results. 115 Insights and techniques for translating AI knowledge into effective treatment may be obtained by studying the science of early death in cancer. AI has already been used in a variety of healthcare contexts. For instance, AI has created a smartphone app called Diagnosis for evaluating and annotating medical pictures and videos based on strong connections between cancer prognosis and the efficacy of subsequent therapy. 114
AI has the potential to accelerate the creation of advanced medicines, innovative designs, and cutting-edge therapeutic approaches. The predictive prediction of biological processes enabled by advanced big data analysis with AI has the potential to speed up the transformation of discoveries into new therapies and improve the precision with which complicated diseases are treated. 114 For instance, DeepMind, a firm supported by Google, has developed a tool that can make instant diagnoses of a variety of diseases. Diabetic retinopathy, age-related macular degeneration, and cancer may all be detected in their earliest stages.114,116 The National Institute of Health has also created the Big Data to Learning program to fund the study and creation of methods for incorporating big data and data science into the field of biomedical research. Clinical care that is led by AI may have a significant impact on cancer prevention, detection, and treatment. 114 The use of AI in cancer treatment has the potential to increase diagnostic precision and treatment efficiency, leading to improved patient outcomes. To analyze images of patients with prostate, breast, and brain cancers, scientists developed computer algorithms. 117 It may be used in clinics as a tool to aid with diagnosis, clinical decision-making, and the prediction of patient outcomes. Commonly altered gene predictions, biomarker detection, picture interpretation, and cancer diagnosis are all within AI’s abilities. 117
Drug discovery and development
The process of drug discovery and development is extremely time-consuming, expensive, and difficult. It can often take more than 10 years from the discovery of molecular targets until a pharmacological product can be licensed for sale and made accessible to the public. Any rejection that occurs throughout this period has a significant influence on the organization’s profitability; in fact, a large number of medication candidates fail at some point during the development process and never make it to market. In addition, there are growing regulatory challenges, as well as the problems of constantly identifying medication compounds that are far better than what is already being marketed. Because of this, the process of developing new drugs is difficult and inefficient, and any new medication products that are effective in accessing the market come with an expensive cost. 118 In the most recent few years, there has been considerable growth in the quantity of data that can be accessible to evaluate the activity of medicinal compounds and biological data. This is because an increasing number of tasks are being computerized, as well as because new experimental methods such as parallel synthesis and speech-to-text synthesis that are based on hidden Markov models have been developed.
However, mining of large-scale chemistry data is needed to efficiently classify potential drug compounds, and ML techniques have shown great potential. 119 Since the 1990s, numerous methods, including support vector machines, neural networks, and random forests, have been employed to build models that assist in the process of drug development. In recent years, DL has started to be implemented in practice as a result of an increase in the quantity of data as well as a continual increase in computer power. During the process of discovering new drugs, several steps may be made more efficient with the help of ML. This involves the prediction of drug molecule properties and activities, the development of new pharmacological compounds through de novo design, the analysis of drug–receptor interactions, and the estimation of drug reactions. 120
New applications of AI in developing cancer drugs and in making precision medicine
AI is the demonstration of intelligence by machines designed by humans. Cybernetics, computer engineering, neuroscience, and linguistics are all included in this field of study. Many people put the origins of AI at the 1956 Dartmouth Conference. As the field of AI has continued to grow rapidly over the last several decades, its definition has broadened to include not only ANNs and DL but also other technologies. 11 DL is a type of AI that may automatically extract features from enormous datasets. In addition, DL is capable of interpreting information from images that a person’s vision cannot. 14
Anticancer drug development with the help of AI
AI is used to predict the effectiveness of anticancer treatments and to assist in the creation of new anticancer drugs. Large-scale screening data frequently show an association between cancer cell genetic diversity and therapeutic efficacy; however, responses between various malignancies and drugs may differ significantly. Based on the present mutation position of a malignant cell genome, the model is used to forecast the efficiency of anticancer drugs. Another team of researchers, Wang et al. 48 developed an ML method termed elastic net regression to generate a model of drug susceptibility. In patients with gastric cancer, ovarian cancer, and endometrial cancer, ML algorithms have been found to effectively predict treatment susceptibility. 121 The framework predicts resistance among people who have recently been prescribed tamoxifen, people who have been treated for stomach cancer with 5-FU, and those who experienced endometrial cancer treated with paclitaxel. All these patients had a dismal prognosis.
This research shows that AI has great potential for determining which cancer drugs will be effective. AI is also essential in the fight against resistance to cancer treatments. The use of AI is also important in the battle against cancer drugs that have become resistant. 122 AI can rapidly comprehend how cancer cells build resistance to cancer treatments by studying and analyzing data on large drug-resistant tumors, which may assist in improving pharmaceutical development and use. 123 AI has the potential to improve several aspects of cancer treatment, including imaging, treatment, screening, detection, and medication. AI has the potential to improve cancer diagnostics and treatment. Currently, cancer imaging represents the cutting edge of AI application across all areas of cancer research. Medical imaging and AI can complement one another to help find cancer therapies. 123 Table 6 describes how AI can be used with different types of cancer therapies.
Combination of AI and different cancer therapies.

AI, artificial intelligence; HR, homologous recombination; PARP, Poly (ADP-ribose) polymerase.
Development of AI tools for cancer management
Incorporating AI into health data might lead to the development of valuable clinical tools that aid in cancer care. A growing number of AI approaches based on health imaging are showing excellent promise as practical therapeutic aids in many contexts. 13 A few examples include risk classification of patients, medication response prediction, multidimensional heterogeneous data prediction of recurrence and survival, and prediction of tumor genetic traits and association with tumor spread. 128 The development of AI solutions based on images still requires access to massive, quality-controlled datasets, even with these notable advancements. 129 Overcoming that problem remains difficult. To construct these imaging biological databases, several technical and operational challenges must be resolved, including image and data harmonization, data curation and annotation, picture preprocessing and annotation, and various legal and ethical constraints.130,131 Therefore, the number, quality, and representativeness of datasets continue to be significant limiting factors in the process of developing prediction tools for cancer management.
The goal of this project is to aid in the development, testing, and preliminary clinical validation of AI technologies that may assist oncologists in the estimation of crucial clinical endpoints in the field of cancer. 132 It includes tools for manipulating images, extracting data from radiomics, arranging treatments, and making prognostic forecasts. After these have been verified, Open Challenges will be held to encourage other outstanding developers to make use of CHAIMELEON’s tools to create their models. This project will use a methodological technique termed continuous learning, which will enable seamless updating of the models, including the addition of new descriptions and instruction, to gradually enhance performance throughout the project’s time frame. It is thought that this continuous learning approach would provide the repository’s structure with considerable flexibility in terms of algorithm performance and dataset management. However, a great number of AI models that predict parameters associated with disease identification as well as therapy outcomes have been produced in recent years.133–135
Challenges and ethical considerations
As a result of its complexity and multidimensionality, medicine has almost dropped behind other sectors about the impact of big data and AI technology. This has led to technological obstacles in the process of designing and evaluating solutions that apply to a wide variety of populations.136,137 In fact, intrinsic biases and miseducation of algorithms are becoming more significant problems in the context of the fact that they are being used in regular clinical practice. Concerns have been expressed about the generalizability and accuracy of AI, as well as methodological restrictions that might arise during algorithm development. In addition, the current situation of AI is becoming more fragmented in the field of medicine. In addition, inadequate verification might restrict clinical translation. 138
Methodological challenges
Trust in the algorithms of AI depends on validation
Problems with AI generalizability and repeatability, as well as methodological constraints during algorithm creation, make the current AI ecosystem incredibly fragmented, despite AI’s indisputable capacity to improve cancer patient care and substantially impact oncology as a whole. 138 Most AI research to date has been retrospective, and randomized controlled studies comparing treatment outcomes with and without an AI-based clinical decision support system are very rare in cancer. 137 Few investigations have been conducted, despite significant advancements in internal validation methodologies, which has led to a decrease in trust in the validity and applicability of the research. 139 A further issue is that AI-based models have a biased geographic distribution, which limits their applicability to populations with different demographics and different healthcare systems. The algorithms’ collider bias and inaccurate association production are both caused by these variables, which impact the characteristic distribution directly between the training and validation cohorts. 137 Benchmark datasets that represent different patient cohorts should be utilized extensively to improve algorithm generalizability and accuracy, as shown in certain international collaborations. 137
Data supervision: Data distribution change and calibration deviation
Aligning a bidirectional data flow from patients to algorithms for training and from learned algorithms back to patients is necessary to achieve the aim of enhancing cancer therapy, highlighting the need to integrate data science with clinical practice. 140 ML algorithms are very contextual and time-sensitive; their responses change based on the specifics of each dataset’s development. We are unable to take the effectiveness of one paradigm and apply it to another age or organization since clinical settings are diverse and unpredictable. By improving its characterization using independent data and calibrating the model, common cause variation, the known and predictable portion of the system’s variability may be reduced. A change in the dataset’s shared percentage of the inputs and the target variables is the root cause of special cause variations, which are unexpected declines in model performance. For organizations to make any changes that are needed to the model or data collection and development, it is crucial to monitor AI for things such as calibration drift and special-cause variability.141,142 The purpose of model updating, in contrast to AI monitoring, is to either maintain or improve upon a model’s prior performance by incorporating new data or changing conditions. 137
Critical assessment and gaps in research
The study of programming computers to act in ways that resemble human intellect is known as AI. For decision-making and other activities, it uses computers programmed to follow algorithms designed by humans or acquired automatically. 69 ML, a branch of AI, is the process by which a computer may train itself to perform better by adding information to an already existing model in a repetitive way. 92 In DL, a specialization of ML, mathematical algorithms are used to create systems with many interconnected “brains,” or computer layers, to mimic human intelligence. This category includes neural networks with a variety of topologies, including recurrent, convolutional, and long-term short-memory networks. Artificial neural networks, which can apply mathematical rules to data in a number of ways, may be very useful for evaluating unstructured data. 143
In the medical industry, unstructured data are often used to record qualitative and subjective information gathered using patient–provider interactions or picture collecting. Applying AI to unstructured text often benefits from DL algorithms such as natural language processing approaches and recurrent neural networks. The most popular and effective AI architectures for exploring image data are convolutional neural networks. Essential components of building and validating ML models include selecting the right problem, collecting data, preprocessing (including anonymization), training, testing, optimization, assessment, and external validation. 144 Developing a trustworthy ML model that may be used in clinical practice involves several steps (Figure 3). Model consistency requires ongoing monitoring of results and applications after deployment to detect and prevent performance degradation due to model drift. Furthermore, prospective clinical studies with problem-specific metrics are needed to evaluate the therapeutic value of ML models. The receiver operating characteristic (ROC) curve is the gold standard for classification tasks in medicine. An indication of a test’s accuracy in predicting the frequencies of true positives and false positives is the area under the ROC curve. Sensitivity, specificity, and accuracy may also be evaluated with the use of the confusion matrix.

Steps involved in AI-based models to be used for diagnosis in healthcare.
AI governance can be summarized as “a system of rules, practices, processes, and technological tools that are employed to ensure an organization’s use of AI technologies aligns with the organization’s strategies, objectives, and values; fulfills legal requirements; and meets principles of ethical AI followed by the organization.” As AI is being used in an increasing number of fields, both public and private, it has become a topic for governance.145,146 AI is an umbrella term for a variety of related but separate areas of study, including data interpretation, ML, and adaptive information systems. 147
Future prospects and unresolved questions
As an increasing number of medical records and images have become public, it is important to note that most of these datasets lack proper tagging, classification, quality control, or correlation to a particular diagnosis. One of the difficult aspects of creating an AI system is gathering and organizing the necessary data, which is a time-consuming process that requires the expertise of doctors. 148 This restriction is especially widespread in approaches that make extensive use of enormous amounts of information, such as convolutional neural networks. Methods that do not depend on labels or human oversight, such as uncontrolled and self-supervised methods, might be the answer. Experiments with unsupervised learning methods that do not need labels, such as variation autoencoders and generative adversarial networks, have shown positive results. 69 While a self-supervised method for segmenting and classifying lung diseases on chest X-rays and tested on the National Institutes of Health chest X-ray dataset yielded remarkable results, an unsupervised DL method for automatic segmentation of chest CT showed an accuracy of up to 98%. 149 We expect that research into therapeutic applications of AI will go into these areas. To incorporate AI into daily clinical practice, a number of requirements must first be fulfilled. Integrating AI technology into image archiving and communication systems is one of these, as well as the development of platforms that allow connectivity among various AI applications to construct a network of powerful tools. Screening for lung nodules and finding image-based biomarkers are two examples of the kinds of laborious and recurring duties that may be automated with the help of AI. This will be achieved with the help of AI, which will pave the way for the eventual automation of these processes. These discoveries will hopefully make it possible to characterize diseases in a manner that is noninvasive and reproducible, which will result in an improvement in treatment and bring personalized medicine one step closer to reality. 150
Challenges
The use of AI in biomedical imaging has the potential to revolutionize several fields, including lung cancer detection, prognosis, and treatment strategies. This will greatly enhance patient care once it is used in clinical practice. However, there currently exist a number of barriers that prevent the broad use of AI in regular work routines. AI tool development requires a massive volume of high-quality data. Despite the abundance of lung cancer datasets, there is a shortage of consistent imaging, clinical, and laboratory data that prevents the development of effective algorithms. 151 Therefore, it is important to gather information from a wide variety of sources, highlighting the significance of sharing information and working together in scientific research. The Cancer Imaging Archive is an example of an ever-growing open-access image database that has a large dataset on cancer and is a valuable resource for academics looking to confirm local work. 152 Collaboration is one of the most important aspects of AI research in the medical field. This is because multidisciplinary groups are necessary for the development of reliable models that may impact routine clinical practice. Radiologists, doctors, engineers, and programmers all work together in these groups, and they all learn from each other in a two-way, mutually beneficial process.
The study design is another significant restriction. In particular, a large portion of the literature on lung cancer outcome prediction has focused on very small patient populations.153–155 Results from AI models trained on a few case series are results that are difficult to generalize and hence useless in real clinical practice. Without further validation in other cohorts, the reliability and therapeutic utility of the findings cannot be confirmed. Prior to their use as clinical diagnostic tools, models built using historical data must be validated in a prospective scenario since they also have analogous constraints. It is important for researchers working on AI models to keep in mind that a comprehensive picture of lung cancer requires data from several sources. The recommendations suggest applying multivariate analysis with features other than imaging, such as family history and clinical and genetic data, to generate more comprehensive models. 156 One of the biggest obstacles AI must overcome before it can be used in the clinic is reproducibility since the radiomics process (from picture capture to model validation) may vary widely from study to study and institution to institution. 157
For instance, the signal-to-noise ratio and derived picture features may be affected by the diversity of image capture procedures between institutions. It also indicates that differences in imaging characteristics and values across patients are more likely the result of differences in capture parameters than in tissue biology. One way around this restriction is to avoid using features that are very sensitive to changes in the acquisition and reconstruction settings. Using open imaging standards, for instance, might help standardize image capture as another option.
Recent advancements in the use of AI for CAR design and CAR efficacy
Recently, Qiu et al. 158 created CAR-Toner, an AI-powered PCP (positively charged patch) computation tool. It also gives suggestions on how to improve the PCP score, but more research is needed to find out exactly what effect these changes have on the specificity and affinity of CAR antigen binding. However, this state-of-the-art technology is poised to advance the field of CAR-T design to the next level, paving the way for AI-driven advancements in CAR-T design.
One way to make an AI-based platform for choosing specific TCRs (T-cell receptors) that works well is to follow the steps suggested by Bujak et al. 159 Bujak et al. 159 recommended collecting patient samples and creating a database of pHLA (peptide-human leukocyte antigen) and TCR sequences. The multicenter observational trial included patients with stage II, III, or IV colon cancer adenocarcinoma from eight different hospitals. The patient recruitment process involved the registration of 100 participants. Bujak et al. 159 extracted and cryopreserved mononuclear cells from peripheral blood, collected samples of primary tumor tissue and peripheral blood, and carried out nucleic acid extraction (DNA and RNA) in 86 instances. Furthermore, they subjected 57 samples to RNA sequencing for gene expression profiling and whole exome sequencing to detect somatic mutations. According to Bujak et al., 159 the findings of their research may significantly impact the management of patients with colorectal cancer. A big database of pHLA:TCR sequences was made by the observational clinical study by Bujak et al. 159 This database will help make the AI-based platform for TCR selection. The outcomes so far show that patient enrollment and sample collection were effective, setting the stage for more research and the creation of a novel tool to speed up and improve TCR selection for precision cancer therapy.
Recently, Martarelli et al. 160 used AI to help them study the molecular dynamics and molecular docking of different anti-CD30 mAbs clones. They wanted to find the best scFv (single-chain variable fragment) binding before engineering CAR-T cells. The virtual computational scFv screening, surface plasmon resonance, and functional CAR-T cell tests all worked the same way to attach to and kill tumors in the lab and living things. Martarelli et al. 160 concluded that the creation of new CAR constructions might be advanced by the suggested quick and inexpensive in silico analysis, which would also significantly cut down on expenses, time, and the requirement for using lab animals.
Conclusion
Cancer is a complex and heterogeneous disease that poses significant challenges for diagnosis, prognosis, and treatment. AI, especially DL and radiomics, has shown great potential to improve the clinical management of cancer patients by providing data-driven insights from medical imaging. However, there are still many limitations and barriers that need to be addressed before AI can be widely adopted in routine practice. These include the quality, availability, and diversity of data, the validation and generalization of models, the reproducibility and standardization of methods, and the ethical and regulatory issues of AI applications. To overcome these challenges, multidisciplinary collaboration, open sharing, and transparent reporting are essential. Moreover, AI should not be seen as a replacement for human expertise, but rather as a complementary tool that can augment and enhance the decision-making process. Ultimately, the goal of AI is to improve the quality of care and outcomes for cancer patients and to support the development of precision medicine.
