Abstract
Cytokines play a critical role in regulating host immune response toward cancer and determining the overall fate of tumorigenesis. The tumor microenvironment is dominated mainly by immune-suppressive cytokines that control effector antitumor immunity and promote survival and the proliferation of cancer cells, which ultimately leads to enhanced tumor growth. In addition to tumor cells, the heterogeneous immune cells present within the tumor milieu are the significant source of immune-suppressive cytokines. These cytokines are classified into a broad range; however, in most tumor types, the interleukin-10, transforming growth factor-β, interleukin-4, and interleukin-35 are consistently reported as immune-suppressive cytokines that help tumor growth and metastasis. The most emerging concern in cancer treatment is hijacking and restraining the activity of antitumor immune cells in the tumor niche due to a highly immune-suppressive environment. This review summarizes the role and precise functions of interleukin-10, transforming growth factor-β, interleukin-4, and interleukin-35 in modulating tumor immune contexture and its implication in developing effective immune-therapeutic approaches.
Concise conclusion
Recent effort geared toward developing novel immune-therapeutic approaches faces significant challenges due to sustained mutations in tumor cells and a highly immune-suppressive microenvironment present within the tumor milieu. The cytokines play a crucial role in developing an immune-suppressive environment that ultimately dictates the fate of tumorigenesis. This review critically covers the novel aspects of predominant immune-suppressive cytokines such as interleukin-10, transforming growth factor-β, interleukin-4, and interleukin-35 in dictating the fate of tumorigenesis and how targeting these cytokines can help the development of better immune-therapeutic drug regimens for the treatment of cancer.
Keywords
Introduction
Immune-suppressive cytokines are well known to play an essential role in controlling effector immune responses required to prevent autoimmune and inflammatory reactions.1,2 The functions and roles of immune-suppressive cytokines in autoimmune/infectious diseases are well characterized. These cytokines are beneficial in one arm and control the severity of autoimmune and inflammatory reactions; in the second arm, they play a negative role by damaging host effector antitumor immunity and making the environment favorable for tumor growth and metastasis.3,4 The altered epigenetic and growth kinetics in cancer cell leads to the extracellular secretion of several factors that control the activity of immune cells and makes them favorable for tumor growth.5,6 Among these, secretion of immune-regulatory cytokines is the primary factor for recruiting immune-suppressive cells. 7 The immune-regulatory cytokines in the tumor microenvironment recruit other immune-suppressive cells and are responsible for phenotypical and functional switch of effector immune cells, and make them supportive for tumor development.8,9 The phenotypically switched immune cells further elevate the level of immune-suppressive cytokines and make the tumor environment resistant to the activity of antitumor immune cells.10,11 These immune-suppressive cells mainly include MDSCs (myeloid-derived suppressor cells), TAMs (tumor-associated macrophages), regulatory T (Treg) cells, regulatory B (Breg) cells, and TANs (tumor-associated neutrophils).12,13 Although several other types of immune cells such as Th17, Th2, and dendritic cells may act as immune-suppressive in certain cancers, however, MDSCs, TAMs, Treg, and Breg cells are the prominent source of regulatory cytokines and dominate the tumor milieu.14–17 These cells exert their suppressive functions via several mechanisms, but the secretion of immune-regulatory cytokines is a significant contributor to immune suppression among several other mechanisms.2,9 These immune-regulatory cytokines show tumor-promoting characteristics by regulating intracellular signaling in cancer cells and immune cells present within the tumor milieu by an autocrine or positive feedback loop.13,18
The immune-suppressive interleukin (IL)-10, transforming growth factor-β (TGF-β), IL-4, and IL-35 have distinct physiological effects. These cytokines alter the fate of innate and adaptive immune cells and regulate the growth and proliferative capacity of cancer cells. Furthermore, the regulatory IL-10 and TGF-β drive and maintain the immune-suppressive phenotype of T cells by diminishing the effector Th1, Th2, and Th17 phenotypes,19,20 while IL-4 predominantly favors the Th2 phenotype of T cells. 21 The IL-35 was shown to have major effects on T cells and B cells, where it converts conventional T cells into IL-35, producing immune-suppressive T cells by inhibiting Th1 and Th17, and effector phenotype of T cells.22,23 These cytokines are secreted by diverse immune populations, including cells from innate and adaptive immune systems. In certain scenarios, tumor cells might secret the IL-10, TGF-β, IL-4, and IL-35, and drive the immune-suppressive fate of tumor niche.24–26 Generally, IL-10, TGF-β, and IL-4 share similar receptor patterns across cancer cells, and immune cells, except IL-35 uses different receptor patterns depending on the cell type. IL-10 signals through IL-10Rα and IL-10Rβ, TGF-β signals through TGF-βRI and TGF-βRII, IL-4 signals through IL-4Rα, and IL-35 signals through IL-12Rβ2 and gp130 in case of T cells and IL-12Rβ2 and IL-27Rα in case of B cells.27–31 The downstream signaling of these receptors activates phosphorylation of signal transducer and activator of transcription (STAT) proteins and different transcription factors, which further dictates immune suppression and tumorigenesis.
The immune-suppressive cytokines alter the epigenome and transcriptional network in naïve immune cells and favor immune-suppressive cell lineage commitment.8,17 Furthermore, these cytokines can directly act on cytotoxic CD8 T cells or natural killer (NK) cells and restrain their effector functions and proliferation into the tumor microenvironment.32,33 In addition, immune-suppressive cytokines directly modulate the tumor cell growth and favor their survival and proliferation.2,34,35 The immune-suppressive cytokines are classified into a broad range and secreted by various cells, including tumor cells, epithelial cells, innate immune cells, and adaptive immune cells. Among the wide range of immune-suppressive cytokines, IL-10, TGF-β, IL-4, and IL-35 dominate the tumor environment in multiple cancers and are mainly responsible for immune suppression36–41 (Figure 1). Herein we reviewed the recent updates on essential functions of these cytokines in modulating tumor microenvironment and how targeting these cytokines can limit cancer cell survival and activate the antitumor immunity that helps to control tumor growth and metastasis.
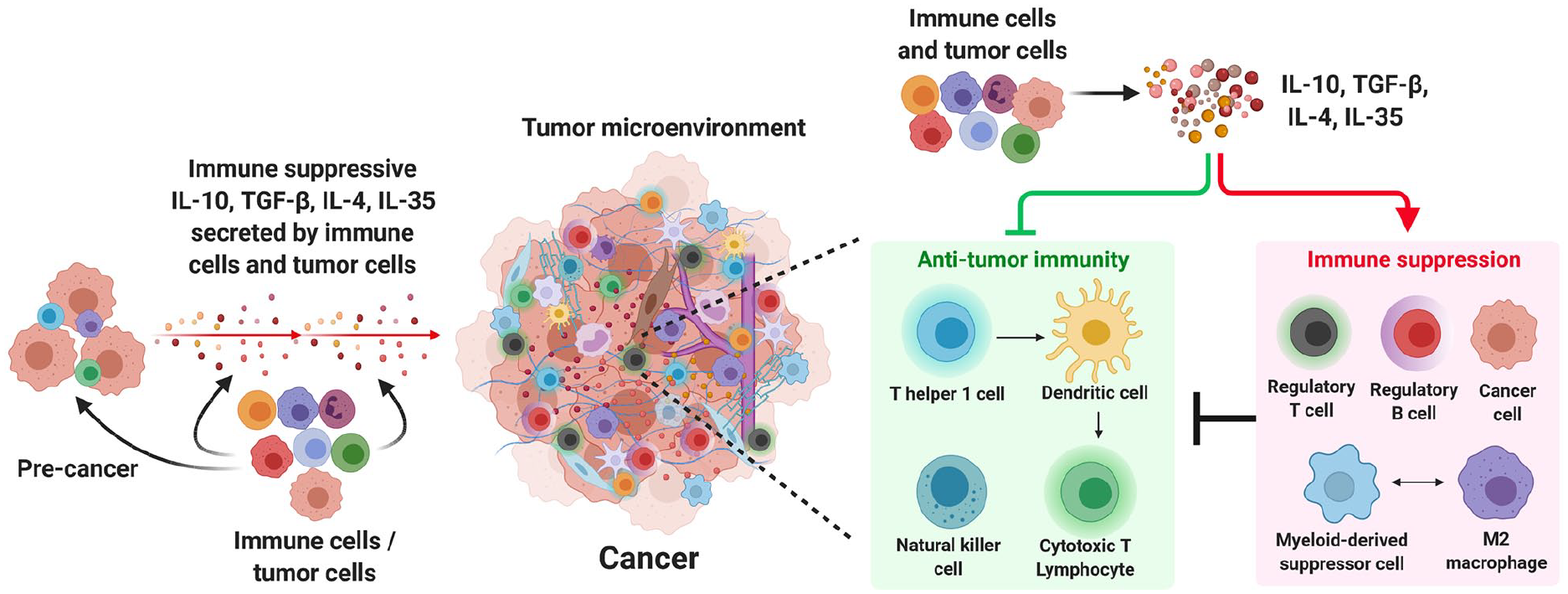
Immune-suppressive IL-10, TGF-β, IL-4, and IL-35 in promoting tumor growth.
Immune-suppressive cytokines IL-10, TGF-β, IL-4, and IL-35 in cancer and immunotherapy
IL-10
IL-10 is a multifaceted immune-suppressive cytokine and possesses immune-regulatory and angiogenic functions. 42 IL-10 enhances tumor cell survival, proliferation, and metastasis by controlling antitumor immunity. 43 IL-10 is secreted by cancer cells and several immune cells, including myeloid and lymphoid lineages. IL-10 exerts its suppressive effects on other effector immune cells, including potent antitumor cytotoxic NK and CD8 T cells.20,44 NK cells and CD8 T cells are essential for killing cancer cells and controlling tumor growth. Recent studies showed that the neutralization of the effects of IL-10 induces the tumor-specific cytotoxic immune response. The Cetuximab-based IL-10 fusion protein (CmAb-(IL10)2) demonstrates potent antitumor effects by preventing dendritic cell–mediated apoptosis of tumor-infiltrating CD8 T cells. Combination of CmAb-(IL10)2 with immune checkpoint blockade showed improved antitumor immunity in mice with advanced tumors. 45
Further studies by Naing et al. showed that PEGylated IL-10 induces CD8 T-cell-mediated immune response in cancer patients by elevating their proliferation and tumor infiltration, and their interferon-gamma (IFN-γ) and granzyme B production. In addition, a combination of PEGylated IL-10 with immune checkpoint blockade enhances intratumoral expansion of CD8 T cells. 46 Similar studies confirmed that the PEGylated IL-10 treatment was shown to restore tumor-specific CD8 T cell response and attenuates tumor growth. 47 In agreement with the role of IL-10 in inhibiting tumor-specific cytotoxic immune response, therapeutic blockade of IL-10 has been shown to have a positive effect on antitumor immune response mediated by CD8 T cells and NK cells. In human breast cancer, therapeutic blockade of IL-10 receptor signaling augments CD8 T-cell-dependent primary tumor response to chemotherapeutic agents such as paclitaxel and carboplatin. 48 In addition, several studies have demonstrated the inverse correlation between CD8 T cells, poor prognosis, and elevated levels of IL-10 in the tumor microenvironment. In esophageal cancer patients, IL-10 suppresses activation of CD8 T cells, and the level of IL-10 in the tumor microenvironment is elevated in the advanced tumor stage. 49
In addition, B-cell-secreted IL-10 in patients with differentiated thyroid carcinoma (DTC) suppresses the effector functions of CD4 and CD8 T cells. It increases the frequency of tumor-infiltrating Treg cells. 50 Similar studies by Hu et al. 51 demonstrated that the B-cell-secreted IL-10 reduces the effector CD4 and CD8 T-cell-mediated immunity by attenuating production of IFN-γ, TNF-α (tumor necrosis factor alpha), and IL-17 and converts these effector T cells into IL-10-producing T cells in patients with gastric carcinoma. In contrast, Zhang et al. 52 showed the TAM-secreted IL-10 drives an immunoevasive microenvironment that causes poor prognosis and inferior therapeutic responsiveness to adjuvant chemotherapy in gastric cancer patients, indicating IL-10 TAMs can be used as a potential target for gastric cancer. In addition, IL-10 limits CD8 T cell activation and function via modulation of cell surface glycosylation. 53 Likewise, Li et al. reported the T follicular regulatory (Tfr) cell-secreted IL-10 was negatively correlated with tumor-infiltrating CD8+IFN-γ+ T cells in ovarian cancer (OC) patients. The in vitro co-culture of CD8 and Tfr cells revealed Tfr cells suppress the activation of CD8 T cells in IL-10 but not TGF-β-dependent manner. The frequency of Tfr cells was enhanced in peripheral blood samples of OC patients. 54
Besides the effects of IL-10 in controlling CD8 T-cell-mediated immune response, IL-10 can have tumor-promoting effects by augmenting regulatory CD4 and CD8 T cell phenotypes. Mechanistic studies in multiple myeloma patients showed IL-10 augments immune-suppressive activities by favoring generation and expansion of CD8+CD28−CD57+LFA-1high Treg. These CD8 Treg cells contribute to the immune escape mechanism of tumors in multiple myeloma patients. 55 Furthermore, IL-10 was also shown to regulate negative receptor expression on CD8 T cells. Liu et al. observed the high infiltration of T cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain (TIGIT) expressing CD8 T cells resulted in poor survival of muscle-invasive bladder cancer (MIBC) patients. Mechanistically, TIGIT+CD8+ T cells impaired cytotoxic activity of effector CD8 T cells by producing immune-suppressive cytokine IL-10 and overall promotes suppressive tumor immune contexture. 56 Likewise, IL-10 was shown to upregulate the expression of lymphocyte activation gene-3 (LAG-3) and programmed cell death protein-1 (PD-1) on CD8 T cells and impaired the production of effector cytokines IFN-γ and TNF-α in patients with epithelial OC. Dual blockade of IL-10 with LAG-3 and PD-1 induces the proliferation and cytokine production of tumor-specific CD8 T cells. 57 In addition, Tannir et al.58,59 revealed pegilodecakin (pegylated recombinant human IL-10) with immune checkpoint blockade induces intratumoral effector IFN-γ+ CD8 T cell response and showed promising clinical activity in patients with metastatic renal cell carcinoma. This study demonstrates the combinational treatment targeting IL-10 with immune checkpoint blockade has notable clinical benefits.
Recently, the immune-modulatory effects of IL-10 on antitumor NK cells were well characterized. In pancreatic ductal adenocarcinoma (PDAC) patients, CD16hiCD57hi NK cells produce high-level IL-10, hindering their cytotoxic activity and IFN-γ expression. The NK cells secreting IL-10 were abundantly found in the blood of PDAC patients, indicating possible options for NK-based immunotherapy for the treatment of PDAC. 60 Sun et al. observed that in patients with hepatocellular carcinoma (HCC), IL-10 enhances expression of NKG2A in NK cells and the NKG2A ligand, HLA-E, in intratumor HCC tissues. These NKG2A expressing NK cells were functionally exhausted and associated with a poor prognosis. Moreover, IL-10 blockade specifically inhibits NKG2A expression in NK cells and restores immune response against tumor cells by reversing NK-cell exhaustion. 61 Similar studies in HCC patients showed CD96+ NK cells were functionally exhausted with diminished IFN-γ and TNF-α secretion due to high expression of IL-10 and exhibit poorer clinical outcomes. 62 Ostapchuk et al. 63 showed IL-10 activates immune-regulatory functions of NK cells, which impair the efficiency of antitumor immunity in breast cancer patients. These observations indicate IL-10 has diverse suppressive effects on antitumor immune cells, required for tumor cell clearance. Therefore, targeting IL-10 in the tumor microenvironment alone or combined with conventional chemotherapeutic or immunotherapeutic agents has substantial clinical benefits for cancer treatment.
TGF-β
The TGF-β is an immune-suppressive cytokine that mainly exerts tumor immune-suppressive effects, resulting in tumor development.28,64 TGF-β is required for the development and lineage-specific differentiation of Treg cells that ultimately leads to developing a highly immune-suppressive tumor environment.65,66 Recent reports showed the different mechanisms of TGF-β overexpression in the tumor environment that restrains effector antitumor immunity. By blocking the activity of histone demethylase LSD1 (lysine-specific demethylase) in tumor cells, Sheng et al. found that the intensity and percentage of TGF-β were significantly higher, inhibiting the cytotoxic activity of intratumoral CD8 T cells. Next, the synergistic depletion of LSD1 and TGF-β in combination with immune checkpoint blockade ameliorates intratumoral CD8 T cell infiltration and effector functions, resulting in eradication of tumor and long-term protection from tumor re-challenge. 67
In parallel, Marwitz et al. showed the TGF-β downstream-activated SMAD3 phosphorylation in CD8 T cells and Treg cells negatively impacts the overall survival of lung cancer patients. In a rapidly progressing group of lung cancer patients, the Treg cells expressing high pSMAD3 are near CD8 T cells, which indicates TGF-β pathway activation in tumor-infiltrating immune cells influences survival of lung cancer patients. 68 Similarly, a recent study reported that the ablation of TGF-β downstream signaling in CD4 T cells restricts tumor progression by enhancing Th2 cell phenotype and causing the death of cancer cells in distant avascular regions. 69 Given that TGF-β is essential for sustained immune-suppressive Treg cells, enhanced immune suppression in the tumor niche can resist the immune checkpoint blockade treatment. An argument with this de Streel et al. revealed TGF-β secreted by GARP+ (glycoprotein-A repetitions predominant) Treg cells in human metastatic melanoma induces resistance to anti-PD-1 immunotherapy. The selective blockade of TGF-β isoform with anti-GARP:TGF-β1 monoclonal antibodies (mAbs) overcomes resistance to PD-1/Programmed death-ligand 1 (PD-L1) blockade in cancer patients. 70
In patients with esophageal squamous cell carcinoma, MDSC-secreted TGF-β augments PD-1 expression on CD8 T cells and develops resistance toward PD-1/PD-L1 blockade. Combinatorial therapy of PD-1/PD-L1 and TGF-β blockade restored tumor-antigen-specific CD8 T cells. 71 Consistently, TGF-β blockade and immune checkpoint blockade boost Th1 polarization and increase the clonal expansion of CD8 T cells, improving the survival of patients with metastatic castration-resistant prostate cancer. 72 Similar studies show the combinatorial blockade of PD-L1 and TGF-β enhances the intratumoral infiltration of CD8+ T cells and improves the survival of mice with colon and pancreatic adenocarcinoma. 73 An alternative study by Liu et al. 74 shows dual blockade of TGF-β1 and GM-CSF (granulocyte-macrophage colony-stimulating factor) improves the effects of chemotherapy treatment by preventing polarization of TAMs toward immune-suppressive M2 phenotype and induces response of CD8 T cells in pancreatic cancer patients. The TAN polarization was known to regulate by TGF-β. TGF-β promotes the immune-suppressive N2 phenotype of TANs, and blockade of TGF-β results in enhanced infiltration and activation of the antitumor phenotype of TANs. 75
Recent reports showed the relationship between TGF-β and the expression of negative receptors on immune cells. Li et al. observed osteopontin-induced TGF-β and upregulated PD-1 expression on immune cells. In patients with advanced triple-negative breast cancer (TNBC), the inhibition of PD-1 signaling impedes TGF-β expression and improves the treatment responses. 76 In addition, TGF-β downstream-activated SMAD2/3 induces PD-1 expression on intratumoral CD8 T cells that resulted in CD8 T cell dysfunction, exhaustion, and increased tumor growth in gastric cancer patients. 77 In addition, Yang et al. demonstrated Notch1 signaling in B16 melanoma cells enhances tumor-induced immune suppression by promoting the expression of TGF-β. TGF-β hinders the proliferation and intratumoral infiltration of effector CD4 and CD8 T cells and reduces the level of IFN-γ by upregulating the expression of PD-1. 78 Thus, targeting the TGF-β-PD-1 axis may be beneficial specifically in combination with immune therapies for cancer treatment.
Several studies have shown the elevated levels of TGF-β associated with cancer patient’s survival, and it regulates tumor-specific cytotoxic immune cells. Yu et al. 79 showed tumor-cell-secreted TGF-β was inversely correlated with the frequency of CD8 T cells in the tumor niche, and it is associated with a worse prognosis of breast cancer patients. The higher expression of TGF-β was also shown to be inversely correlated with CD8 T cell infiltration in lung cancer patients, and altered expression of TGF-β can predict the therapeutic response to nivolumab. 80 In parallel, Wu et al. demonstrated TGF-β could induce the tumor-promoting suppressive phenotype of CD8 T cells in OC environment. Moreover, OC patients express an elevated level of TGF-β, and it is positively correlated with the frequency of CD8+ Treg cells. 81 Besides, TGF-β was shown to alter the lineage plasticity of CD8 T cells in the tumor environment and make CD8 T cells more susceptible to TGF-β-induced immune suppression. 82 In addition, in gastric cancer patients, CD8 T cell dysfunction was positively correlated with elevated levels of TGF-β, which can be used as a potential therapeutic marker. 83 Alternatively, TGF-β was shown to regulate effector functions of NK cells. SMAD2/3/4 activation downstream of TGF-β signaling suppresses T-bet, a positive regulator of IFN-γ, and plays a vital role in regulating NK-cell-mediated immune functions. 84 TGF-β is also known to regulate epithelial-mesenchymal transition (EMT) and regulates cancer metastasis.85,86 EMT plays an essential role in tissue repair, and remodeling and acquiring the metastasis phenotype of cancer cells87,88 indicates the importance of TGF-β in promoting tumor growth independent of immune cells.
The inhibitory effect of TGF-β on effector functions and cytotoxic activity of NK cells were well characterized. Fujii et al. showed TGF-β-induced suppression of NK-cell-cytotoxic function could be inhibited via promoting the IL-15 signaling pathway. Mechanistically, the IL-15 superagonist/IL-15 receptor α fusion complex (IL-15SA/IL-15RA) suppresses the Smad2/3-induced transcription and rescues cytotoxic gene expression of NK cells. 89 Recently, the efficacy of Chimeric antigen receptor (CAR) T cells was shown to be affected in the presence of TGF-β. Stüber et al. showed the negative impact of TGF-β on the efficiency of CAR T cells against TNBC. TGF-β reduces effector cytokine production, proliferation, viability, and cytotoxic activity of ROR1-CAR T cells. Furthermore, the kinase inhibitor SD-208 impedes the TGF-β-receptor signaling, protects CD8+ ROR1-CAR T cells, and sustains their antitumor function. 90 These findings highlight the importance of TGF-β in generating and maintaining a highly immune-suppressive tumor environment that restricts the activity of effector immune cells and promotes tumor growth. Therefore, targeting TGF-β alone or with other combinatorial therapies has notable clinical benefits for the treatment of cancer patients.
IL-4
IL-4 is mainly required for lineage-specific differentiation of Th2 cells and regulation of humoral immune responses. In addition to basophils and mast cells, IL-4 is predominantly secreted by Th2 cells via autocrine signaling.91,92 IL-4 plays a diverse role in the tumor microenvironment and exhibits immune-suppressive and antitumor activities.38,41 Here, we primarily focused on the immune-suppressive role of IL-4 in controlling antitumor immunity and supporting tumor growth. In a mouse model of primary lung carcinogenesis, IL-4 downstream STAT6-activated signaling in CD11b+ myeloid cells promotes M2 immune-suppressive phenotype, and triggers IL-4 expression in M2 cells by a positive feedback loop and enhances lung cancer progression. 93
Further studies from Ito et al. revealed that blockade of IL-4 decreases the generation of immune-suppressive M2 phenotype and MDSCs in a specific tumor microenvironment and ameliorates tumor-specific CD8 T cell response in a mouse model of colon and breast cancer. In addition, blockade of IL-4 improves the responsiveness of anti-OX40 immunotherapy. 94 Similar studies in a mouse model of non-small-cell lung cancer showed blockade of IL-4 augments IL-12 production by tumor-antigen-specific dendritic cells that enhance infiltration and proliferation of effector T cells and reduce tumor burden. 95 In addition, Joshi et al. 96 demonstrated depletion of IL-4 augments the antitumor immunity and overall survival without any visible toxicity in mouse models of anaplastic thyroid cancer (ATC).
Several recent reports critically illustrated the significance of IL-4 signaling in the survival and proliferation of cancer cells. Nappo et al. 97 showed IL-4 downstream-induced STAT6 signaling is required for increased survival and proliferation pathways, including Akt and NF-κB (nuclear factor-κB) pathways in prostate cancer cells. Likewise, IL-4 triggers the mitogen-activated protein kinases (MAPKs) and specifically induces proliferation of A253 (head and neck), MDA-MB-231 (breast), and SKOV-3 (ovarian) cells by c-Jun N-terminal kinase (JNK) activation. 98 Moreover, Li et al. observed enhanced growth of MCA205 fibrosarcoma in the presence of IL-4. Mechanistically, IL-4 upregulates the expression of IL-4R and anti-apoptotic genes in tumor cells, which is required for the survival of tumor cells. Furthermore, blockade of IL-4 by 11B11 mAb showed a reduction in tumor growth. 41 Besides, Zhao et al. 99 showed IL-4-TBX21 signaling induces expression of cancer stemness markers and proliferation in lung cancer cells, indicating IL-4 signaling is an essential factor for cancer development in lung adenocarcinoma patients. The CD44hi expressing tumor cells have been revealed to have tumor-initiating characteristics. Erb et al. 100 showed the expression of CD44 on prostate cancer cells was upregulated by IL-4 and increased their proliferation, migration, and clonogenic potential. These studies indicate the role of IL-4 signaling in the survival and proliferation of cancer cells.
The effects of IL-4 gene polymorphisms on cancer risk were well characterized. Jia et al. 101 found that IL-4 rs2243250 and rs79071878 polymorphisms are associated with cancer sensitivity. Likewise, Shamoun et al. 102 showed that the IL-4 SNP rs2243250 genetic polymorphism could be a useful prognostic marker for stage III colorectal cancer patients. The IL-4 rs2243250 and variable number tandem repeat (VNTR) polymorphisms were also associated with lung cancer and breast cancer susceptibility;103,104 this observation shows the critical relationship between IL-4 polymorphism and tumorigenesis. In contrast, IL-4 was induced upon radiation therapy and contributed to tumor growth and metastasis. Formentini et al. 105 revealed that radiation-induced IL-4 upregulates the expression of IL-4Rα in human colorectal cancer cells and facilitates colorectal cancer metastasis. Similarly, Kim et al. 106 observed expression of IL-4 is highly elevated upon radiation in metastatic carcinoma tissues, and it is correlated with poor survival of cancer patients, indicating IL-4 can be a promising target to prevent radiation-induced immune suppression.
Interestingly, several reports demonstrated the altered expression of IL-4 is associated with the severity of cancer, and it regulates diverse immune infiltrates in the tumor milieu. In patients with papillary thyroid carcinoma (PTC), the increased serum level of IL-4 is associated with persistent disease. It is positively correlated with the production of antithyroglobulin antibodies. 107 In addition, tumor-cell-secreted IL-4 was shown to significantly impact effector tumor infiltrates, including TAMs and CD8 T cells. IL-4 was shown to enhance pancreatic-cancer-promoting functions of TAMs by inducing cathepsin protease activity in macrophages. 108 Furthermore, Apte et al. 109 observed tumor-cell-secreted IL-4 elevates expression of GATA3 and type 2 cytokines, which reduces the antitumor immune response mediated by CD8 T cells in a mouse model of thymic lymphoma. Likewise, Noble et al. 110 showed IL-4 upregulates the expression of CD39 and IL-10 that induces peripheral tolerance and suppressive activity of tumor-infiltrating CD8 T cells. Moreover, treatment of CD8 T cells with IL-4 downregulates expression of CD127 and contributes to impaired CD8 T cell function and antitumor immune response. 111 In addition, Olver et al. showed the suppressive effects of tumor-cell-secreted IL-4 on intratumoral CD8 T cells. IL-4 alters the cytotoxic activity of tumor infiltrated CD8 T cells that resulted in delayed primary tumor clearance and increases possibilities for the development of secondary tumors indicating inhibition of IL-4 in the tumor milieu can improve the efficacy of CD8 T cells. 112 The role of IL-4 in modulating NK-cell functions is well characterized. Ohayon et al. 113 showed a reduction of IFN-γ and TNF-α by IL-4-treated human NK cells and reduced cytotoxicity toward K562 cells. Likewise, human NK cells exposed to IL-4 differentially regulate and prime naïve T cells toward their regulatory phenotype via monocyte-derived dendritic cells and modulate the NK-cell interaction with Dendritic cells (DCs). 114 In addition, IL-4 can reduce the functional ability of NK cells by secreting type-2 cytokine expression among innate cells and positively regulates tumor progression.34,115
These findings indicate IL-4 has a dual role in the tumor microenvironment; it regulates the tumor cell survival, proliferation, and migration, and modulates antitumor immunity. It shows that IL-4 possesses an emerging therapeutic target to reduce tumor-induced immune suppression and boosts the efficacy of conventional cancer therapies. Thus, similar to other immune-suppressive cytokines, IL-4 is a critical regulator of tumor initiation and metastasis and may represent an attractive immune therapy target alone or in combination with currently available treatment regimens for the effective treatment of patients with cancer.
IL-35
IL-35 is a heterodimeric and relatively newly discovered cytokine compared to IL-10, TGF-β, and IL-4, belonging to the IL-12 family cytokine and made up of p35 and EBi3 subunits.116,117 IL-35 is well known to regulate adaptive immune responses and control effector T-cell-mediated immune responses in autoimmune diseases.118,119 In addition, IL-35 induces immune-suppressive iT35 cells from conventional T cells that control T cell proliferation promote exhaustion of CD8 T cells.22,23 Treg-cell-secreted IL-35 in tumor microenvironment enhances intratumoral T cell exhaustion by activating several inhibitory receptor expression and exhaustion–associated gene expression in tumor-infiltrating CD8 T cells and inhibits effective antitumor immunity. 120 Similar studies show the role of Treg-cell-derived IL-35 promotes exhaustion and suppression in non-Treg cells. 121 The expression of IL-35 is highly elevated in mouse and human pancreatic tumor samples and specifically inhibits the activity of CD8 T cells in STAT3-dependent mechanism.122,123
Higher IL-35 levels were also detected in several tumor types, linked with tumor growth and overall survival. Zhu et al.124,125 demonstrated enhanced expression of IL-35 in serum samples of prostate cancer patients was positively associated with metastasis and disease progression. Moreover, IL-35 contributes to prostate cancer progression via increasing cancer cell proliferation and tumor angiogenesis. Mechanistically, IL-35 blocks the infiltration of antitumor immune cells by favoring proliferation and intratumoral infiltration of Treg cells and MDSCs.124,125 The expression of IL-35 was also elevated in tumor tissues of patients with non-small cell lung cancer (NSCLC). It is correlated with overall and progression-free survival. 126 Likewise, the increased level of IL-35 was observed in children with acute lymphoblastic leukemia (ALL), and it is associated with the development of cancer. 127
The overexpression of IL-35 is associated with the progression of human gastric cancer. Fan et al. 128 revealed IL-35 is involved in the proliferation of gastric cancer cells by upregulating the expression of Ki67 and reducing apoptosis. Similarly, Gu et al. 129 observed that the enhanced serum level of IL-35 correlates with disease prognosis in gastric cancer patients. Furthermore, Chen et al. demonstrated the elevated level of IL-35 in serum and tumor tissues of breast cancer patients is associated with tumor progression. IL-35 facilitates the growth of breast cancer cells by augmenting the expression of epidermal growth factor receptor (EGFR), Ki67, and p53 suggests IL-35 is an essential factor for the progression of breast cancer. 130 Moreover, in colorectal cancer patients, high expression of IL-35 is directly correlated with the severity of malignancy and the clinical stage of the tumor. Mechanistically, the cancer-cell-secreted IL-35 recruits Treg cells in the tumor microenvironment and promotes immune suppression. 131 In addition, the elevated level of IL-35 in diffuse large B-cell lymphoma (DLBCL) is correlated with overall survival in DLBCL patients. 132 These observations showed that the elevated level of IL-35 is associated with tumor progression, and it can be served as a better prognostic marker. Overall, these findings uncover the potential role of IL-35 in the modulation of host innate and adaptive immune responses that promote tumor growth. Therefore, targeting or blockade of IL-35 has significant therapeutic benefits for the successful treatment of cancer patients.
Conclusion and future perspectives
Cytokines play a crucial role in shaping and dictating the tumor immune microenvironment that decides the fate of tumorigenesis. Several studies showed that the highly immune-suppressive tumor environment is mainly responsible for tumor growth and tumor resistance to multiple chemotherapeutic and immunotherapeutic drugs. For example, the acquired resistance to conventional immune-therapeutic treatment such as immune checkpoint blockade is predominantly due to elevated immune-suppressive cytokines in the tumor milieu.133,134 These immune-suppressive cytokines restrict the proliferation and effector functions of antitumor immune cells, alter their plasticity, and convert them into suppressive phenotypes. Due to restricted infiltration or absence of effector immune cells, the immune checkpoint blockades cannot exert optimum effects135–137 (Figure 1).
Similarly, multiple radiation therapies are well known to induce immune suppression, primarily due to the secretion of regulatory cytokines either by tumor cells or by immune cells in the tumor niche.138–140 Therefore, targeting or depleting these cytokines has significant clinical benefits. Several cytokines play an immune-suppressive role in tumorigenesis depending on the type and tumor immune contexture. However, in this review, we mainly focused on IL-10, TGF-β, IL-4, and IL-35, since several recent studies described above showed their role in limiting antitumor immunity and progression of tumor growth. In addition, the cytokines secreted by tumor cells as well as immune cells such as IL-23, IL-27, IL-39, IL-1β, IL-13, IL-21, and IL-17 are well known to regulate tumor immune contexture and can participate in immune suppression; however, their precise role and importance in contributing tumor growth are out of the scope of this review.
The critical challenge in developing better immune-therapeutic drugs is to combat immune suppression within the tumor milieu. The tumors typically resist the infiltration of immune cells, considered immunologically “cold tumors.” 141 The immune-suppressive cytokines play a vital role in developing the “cold” nature of the tumor. 142 In addition, tumor-cell-expressed markers and secreted factors such as soluble inhibitory factors, inhibitory receptors/ligands, and immune-suppressive cytokines/chemokines further facilitate the “cold” nature of the tumor.143–145 The overreaching goal is to target these factors alone or combine them with available conventional therapies to achieve maximum tumor clearance. A blockade of immune-suppressive cytokines such as IL-10, TGF-β, IL-4, and IL-35 might play an essential role in making tumors sensitive to conventional therapies. Blockade or neutralizing the activities of these cytokines might convert the immunological “cold” nature of the tumor to a “hot” tumor. The immunological “hot” tumor further facilitates the infiltration and proliferation of antitumor immune cells and makes the tumor more responsive toward conventional immune therapies such as immune checkpoint blockades (Figure 2). Future studies should delineate these cytokines’ detailed mechanism of action in regulating immune-suppressive cell phenotype and tumor growth. Additional studies are required to monitor their systemic versus localized effect and check whether these cytokines can induce any adverse effects like toxic or inflammatory reactions. Thus, targeting highly abundant immune-suppressive cytokines such as IL-10, TGF-β, IL-4, and IL-35 in the tumor microenvironment considerably helps to revert immune suppression, enhancing the efficacy of conventional cancer therapies and activating antitumor immunity, and controlling tumorigenesis.

Targeting the immune-suppressive IL-10, TGF-β, IL-4, and IL-35 reduces tumor growth.
Limitations of the study
The initiation and development of cancer is a multistep process. Although immune cells and their secreted immune-suppressive cytokines play a vital role in dictating the fate of tumorigenesis, several other important factors can modulate tumor growth. Several growth factors, chemokines, and tumor-cell-secreted extracellular factors can significantly influence immune-suppressive cytokines. In addition, this review mainly focused on immune-suppressive IL-10, TGF-β, IL-4, and IL-35 cytokines; however, several other cytokines exist, such as IL-13, IL-5, IL-1β, IL-2, and IL-6, and they can play immune-suppressive roles, which controls antitumor immunity and promotes tumor growth. Apart from the immune-suppressive functions of IL-10, TGF-β, IL-4, and IL-35, these cytokines might act directly on tumor cells and control their growth and proliferation. Nevertheless, this review predominantly revealed their immune-suppressive functions of pro-tumorigenic effects. Therefore, further detailed studies are necessary to elucidate distinct functions of IL-10, TGF-β, IL-4, and IL-35 in tumor milieu and their role in maintaining equilibrium between pro-tumor and antitumor activities (see Table 1).
Clinical implications of IL-10, TGF-β, IL-4, and IL-35 in cancer immunotherapy.

IL: interleukin; TGF-β: transforming growth factor-β; PD-1: programmed cell death protein-1; NSCLC: non-small cell lung cancer; IFN: interferon; HCC: hepatocellular carcinoma; TAMs: tumor-associated macrophages; CAR: chimeric antigen receptor.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank UNC Chapel Hill Library for providing financial support for publication of the article.
