Abstract
Background:
Two monovalent mRNA vaccines, available in December 2020, were demonstrated to have high efficacy against both the original SARS-CoV-2 strain and variants circulating through the summer and into the fall of 2021. In the context of the Omicron/BA.1 variant, which was predominant from late fall 2021 into winter of 2022 in the United States, and subsequent Omicron subvariants that have been predominant thereafter, vaccine effectiveness of the monovalent mRNA vaccine option is attenuated.
Objectives:
We aim to investigate the relative effectiveness of the bivalent booster compared to the monovalent booster against SARS-CoV-2 infection in the 60 days following administration in Shelby County, TN.
Design:
This observational population-based cohort study utilizes COVID-19 surveillance data to identify adults who were vaccinated with a monovalent booster dose between August 1, 2022 and August 30, 2022 or a bivalent booster dose between September 1, 2022 and September 30, 2022. Both groups were followed for COVID-19 status for 60 days from their administration date.
Methods:
We calculated incidence rates with 95% confidence intervals and propensity-adjusted hazard ratios with 95% confidence intervals of COVID-19 diagnosis in the 60 days following administration of the booster dose between the bivalent group and the monovalent group. Stratified analysis was conducted by age group (18–34, 35–64, and 65+ years old).
Results:
The incidence of reported SARS-CoV-2 infection was substantially higher for those who received the monovalent booster, across age groups. Overall, we observed a 51% lower hazard of infection during the study period among those who received the bivalent booster, compared to the monovalent booster.
Conclusion:
These results support and extend prior findings that the bivalent booster dose may be more effective in preventing infection against the Omicron sub-variants of SARS-CoV-2.
Introduction
Two monovalent mRNA (messenger ribonucleic acid) vaccines, available in December 2020, were demonstrated to have high efficacy against both the original strain of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes COVID-19 disease, and variants circulating through summer and into the fall of 2021.1,2 However, vaccine effectiveness of the monovalent mRNA vaccines against infection and symptomatic infection is attenuated, compared to levels seen during the Delta period, in the context of the Omicron/BA.1 variant, which was predominant from late fall 2021 into winter of 2022 in the United States (US), and subsequent Omicron subvariants that have been predominant thereafter.3–5
On August 31, 2022, the Moderna and Pfizer-BioNTech Omicron BA.4/BA.5 bivalent booster vaccines were authorized by the US Food and Drug Administration (FDA) under an emergency use authorization (EUA) for use at least 2 months after completion of the primary vaccine series or after the first booster dose. 6 Concurrent with the authorization of these bivalent boosters, the EUA for the monovalent mRNA booster options was rescinded. Therefore, monovalent booster doses were no longer authorized for those aged 12 and older as of August 31, 2022. 6
Side effects of COVID-19 mRNA vaccinations are well documented and similar across doses, including primary series and booster doses. 7 Evidence that supported its FDA authorization showed that side effects associated with the bivalent booster are not expected to differ substantially, compared to the monovalent booster. 6
An early immunologic study of a Moderna candidate bivalent vaccine booster formulated to include the Omicron BA.1 subvariant showed evidence that the bivalent booster produced an increased mean titer response compared to the monovalent booster dose. 8 In addition, a recent epidemiological study of bivalent booster dose effectiveness by the Centers for Disease Control and Prevention (CDC) showed that the bivalent booster offered additional protection to those who had previously received at least two monovalent booster doses. 9 Several studies have reported increased vaccine effectiveness using the bivalent booster doses compared to that of the monovalent dose.10–13 This study aims to investigate the relative effectiveness of the bivalent booster compared to the monovalent booster against SARS-CoV-2 infection in the first 60 days following dose administration among those age 18 and older.
Methods
This observational population-based cohort study utilizes surveillance data as of January 2, 2023 including vaccination history and positive COVID-19 case status to identify adults (age 18+) who had received a primary vaccine series and were most recently vaccinated with a monovalent booster dose of at least 60 days after their previous dose between August 1, 2022 through August 30, 2022 and those who had received a primary vaccine series and were most recently vaccinated with a Moderna or Pfizer-BioNTech Omicron BA.4/BA.5 bivalent booster dose at least 60 days after their previous dose between September 1, 2022 and September 30, 2022. Both vaccination groups were followed for confirmed or probable case status for 60 days from the date that they received their applicable booster dose, where confirmed case status was defined by a positive polymerase chain reaction (PCR) test, and probable case status was defined by a positive clinically assessed antigen test.
Recent research suggests that the incubation period for the Omicron variant of COVID-19 is shorter than that of previously circulating variants, at about 3.4 days. 14 To account for the possibility that those testing positive for COVID-19 shortly after administration of the booster dose were exposed prior to its administration, follow-up time began 4 days after administration of the booster dose. Those who tested positive within 4 days following booster administration (n = 11) were excluded from this analysis. All data analysis was conducted at the Shelby County Health Department (SCHD) in Memphis, TN. Shelby County was designated as having a low level of community spread by the CDC, based on case counts and hospitalization metrics, for 18 of the 19 weeks between July 28, 2022 and December 7, 2022, 15 containing the entire follow-up period for both groups. Daily surveillance data reported to SCHD includes resident cases identified via lab or provider-reported positive antigen or PCR tests or death report as well as all resident COVID-19 vaccination history captured in the Tennessee Immunization Information System (TennIIS). COVID-19 vaccination surveillance data include all reported information about administration dates, locations, products, and doses received. In our data-cleaning process, we excluded records that indicated more than six total doses, records that indicated erroneous dates for the most recent or prior dose, as well as records that indicated a bivalent booster was administered before completion of the primary series.
Exposure groups in this analysis are defined by the most recent dose type received, either a monovalent booster dose or a bivalent booster dose. For ease of reporting and interpretation, incidence rates per group are reported per 100 person-years of follow-up time. We calculated incidence rates and 95% confidence intervals with the R package epiR (R version 4.2.1, R Core Team 2022) and propensity-adjusted hazard ratios (HR) and 95% confidence intervals with the R package survival (R version 4.2.1) of COVID-19 diagnosis in the 60 days following administration of the booster dose between the bivalent group and the monovalent group (reference). Analysis was conducted stratified by age group (18–34 years old, 35–64 years old, and 65+ years old) and for all adults (overall). Propensity scores were calculated using patient gender, ethnicity, race, and residential zip code as well as the dose count of the most recent COVID-19 vaccine dose they received and the time between their most recent dose and the previous dose they received. Vaccine effectiveness was calculated as (1 − HR)*100 for each age group and for all adults, overall. Results are reported using STROBE guidelines, 16 and this study was overseen by the University of Memphis IRB (PRO-FY2022-317).
Patient and public involvement
Patient/public involvement was not feasible for this study.
Results
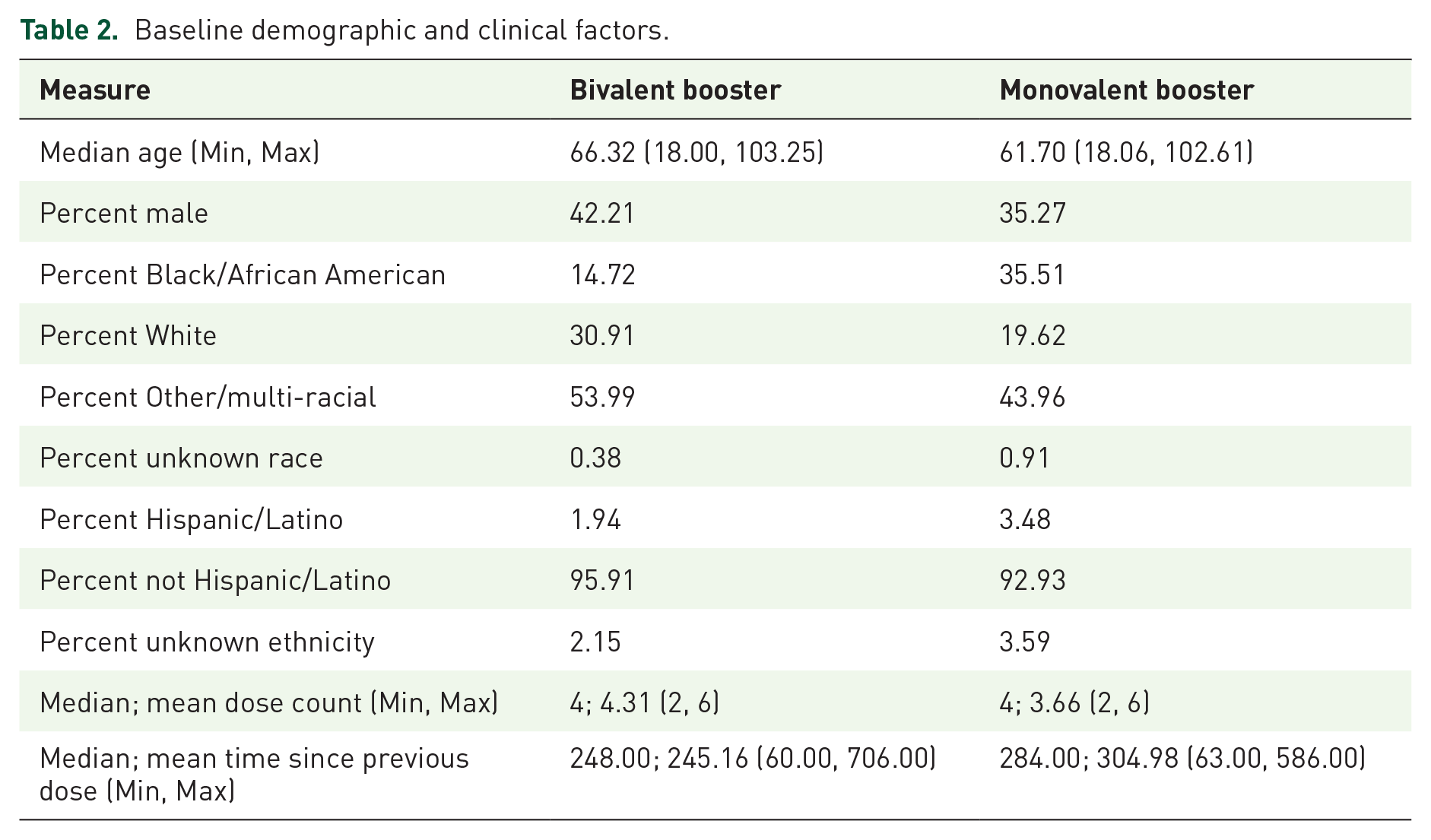
This study included 20,712 adults, of whom 16,201 (78.2%) received a bivalent booster dose between September 1, 2022 and September 30, 2022, and 4511 (21.8%) received a monovalent booster dose between August 1, 2022 and August 30, 2022, as shown in Table 1. Among those 18–34 years old, 1203 (73.4%) received the bivalent booster and 437 (26.6%) received the monovalent booster, in the 35- to 64-year-old age group, 6281 (73.9%) received the bivalent booster and 2219 (26.1%) received the monovalent booster, and in the 65+ age group, 8717 (82.5%) received the bivalent booster and 1855 (17.5%) received the monovalent booster. Table 2 demonstrates the baseline demographic and clinical factors of both groups. Age patterns were similar between the bivalent and monovalent groups. The bivalent group had a slightly higher percentage of males compared to the monovalent group (42.21% vs 35.27%), and differences with respect to racial makeup were also seen between the groups, where a higher percentage of Black or African American people were seen in the monovalent group compared to the bivalent group (35.51% vs 14.72%), and a higher percent of white people and people of other or multiple races were seen in the bivalent group compared to the monovalent group (30.91% vs 19.62% and 52.92% vs 42.30%, respectively). In both groups, the median dose count was 4 with a minimum of 2 and a maximum of 6. The median time since the most recent previous dose was higher for those in the monovalent group (284 days) compared to the bivalent group (248 days).
Individuals included for analysis by age group and dose type.

Baseline demographic and clinical factors.
Individuals included in the analysis contributed 738.7 person-years to the monovalent group and 2656.5 person-years to the bivalent group (Table 3). Overall, 114 cases were reported in the 60-day follow-up period, 82 among the bivalent group and 32 among the monovalent group. The highest crude number of cases was seen in the bivalent group among those aged 65+. The lowest crude number of cases was seen in the 18- to 34-year-old age group, with three cases in each booster group. Crude incidence rates per 100 person-years of follow-up time in the 60 days following the administration of the bivalent or monovalent booster dose by age group are shown in Table 3. Across all age groups, the incidence of reported SARS-CoV-2 infection was substantially higher for those who received a monovalent booster, compared to a bivalent booster. Incidence differences by age group ranged from 28.6% to 180% higher among those who received the monovalent booster compared to the bivalent booster in the 35–64 and 18- to 34-year-old age groups, respectively.
Cases, person-time contributed, and crude incidence rates per 100 person-years of follow-up time in the 60 days following booster dose administration.

Among all adults, individuals with the bivalent booster had 0.51 (0.32, 0.80) times the propensity-adjusted hazard (95% CI) of reported SARS-CoV-2 infection compared to the monovalent group. Small numbers of reported cases in both booster groups among those aged 18–34 lead to a wide confidence interval around the HR estimate: 0.38 (0.06, 2.35). However, among those aged 35 and older, the age-stratified HR (95% CI) ranged from 0.52 (0.26, 1.07) among those aged 35–64 to 0.53 (0.28, 0.99) among those aged 65+ (Table 4). Differences between the bivalent booster and the monovalent booster are significant at the 0.05 level for those aged 65+ and are significant at the 0.10 level for those aged 35–64. Overall, for those aged 18 and older, the bivalent booster was 49.1% more effective in preventing reported infection in the first 60 days following administration, compared to the monovalent booster. In stratified analysis, results were similar between those aged 35–64 and those aged 65+ (Table 4).
Hazard ratios and comparative vaccine effectiveness estimates, comparing the bivalent group to the monovalent group (reference).

Vaccine effectiveness is calculated as (1 − hazard ratio) × 100.
Discussion
In this population-based cohort study, we identified a 49.1% lower hazard of reported SARS-CoV-2 infections among adults who had at least completed a primary vaccination series and had received a bivalent booster as their most recent dose between September 1, 2022 and September 30, 2022 at least 60 days after their previous most recent dose compared to those who received a monovalent booster as their most recent dose between August 1, 2022 and August 30, 2022, in the 60 days following administration.
We found very few reported infections among those aged 18–34 in the 60 days following administration of either the bivalent booster or the monovalent booster. No significant differences in the hazard of infection were observed in the stratified analysis of this age group. However, in the analysis of all individuals aged 18 and older, and in the stratified analysis of those aged 65+, we found significant reductions in the hazard of reported infection between those who received the monovalent booster and those who received the bivalent booster. We also found evidence that comparative vaccine effectiveness may be similar across age groups, as estimated comparative vaccine effectiveness in those aged 65 and older was nearly the same as those aged 35 to 64, 47.5% compared to 47.9%.
These results support the findings of other recent studies showing evidence that the bivalent booster dose may be more effective in preventing infection against Omicron sub-variants of SARS-CoV-2,8,9 in a racially diverse, real-world population in the mid-south US. An observational matched cohort study conducted in the Republic of Korea assessing the effectiveness of the Omicron BA.1 and BA.4/BA.5 bivalent boosters against infection in the 2022–2023 winter season found 12.2% (95% CI: 6.5%–17.7%) additional protection conferred, compared to that of the monovalent vaccine among adults aged 18 and older. 13 Similarly, a study in the same region and timeframe investigating SARS-CoV-2 infection, critical infection, and death in a matched cohort between those receiving the monovalent compared to bivalent vaccine (primarily mRNA-1273.214, the Moderna bivalent vaccine containing Omicron BA.1) found that incidence proportions were substantially higher among the monovalent group than the bivalent group, across all outcomes, by a factor of 3 for infection, 9 for critical infection, and 11 for death. 12 A matched cohort study conducted in the spring of 2022 among those age 60+ in France also found 8% (95% CI: 0%–16%, p = 0.045) additional protection against symptomatic disease conferred by the Pfizer-BioNTech Omicron BA.4-5 bivalent vaccine compared to that of the monovalent vaccine. 11 However, results comparing the effectiveness of bivalent to monovalent boosters have been mixed. For example, a test-negative design study from Ontario, Canada in a similar timeframe among adults aged 50 or older reported similar levels of initial protection between the bivalent (Moderna BA.1 and Pfizer BA.4-5) and monovalent boosters against severe outcomes. 17 Nonetheless, the current study’s findings support the importance of continued public health efforts to improve COVID-19 vaccination uptake.
A major strength of this analysis is that it is propensity-adjusted by key demographic and clinical factors that have the potential to confound the relationship between booster type and reported infection due to SARS-CoV-2. However, this analysis is unable to account for potentially confounding factors such as behavioral patterns, prior infection, access to testing, or unreported home testing, which may have impacted results. In Shelby County, vaccine uptake has historically been highest among those age 65+ compared to the other age groups represented in this analysis. In addition, in previous waves of the pandemic, universal indoor masking requirements were in place. During the timeframe of this analysis, no such mitigation measures were active. Infection mitigation behaviors are another factor that may differ across age groups and could confound results. Further analysis considering the confounding impact of such behavioral factors would be useful to further assess the comparative vaccine effectiveness of the bivalent booster compared to the monovalent booster.
The non-randomized observational design of this study limits our ability to draw definitive conclusions. Underreporting of COVID-19 cases among the monovalent and bivalent groups is a factor that could have introduced bias, given the nature of the data used for this study, and a factor that limits our ability to accurately adjust for prior infection. However, we do not believe that there is reason to expect the rate of under-reporting of cases or the rate of prior infection would differ substantially between those who received a monovalent booster dose from August 1, 2022 through August 30, 2022 and those who received a bivalent booster dose between September 1, 2022 and September 30, 2022. Because this study is not randomized, and because people are included according to their vaccination status (bivalent booster vs monovalent booster) based on when they received the respective vaccine, an inherent limitation of this study is that though the groups are followed for the same amount of time, they are not followed at the same time. The 1-month lag between the monovalent group and the bivalent group means that those who were vaccinated with the monovalent booster in early- to mid-August were followed through mid- and late-August when newly reported case counts were higher in Shelby County than they were in the period from mid-September through late-November. However, the timeframe most affected by increased case counts in August is relatively small, and Shelby County’s Community levels were designated as low by the CDC for 18 of the 19 weeks between July 28, 2022 and December 7, 2022. A strength of this population-based cohort study design is the ability to evaluate the full population of Shelby County vaccinated with a monovalent or bivalent booster during the timeframe studied, suggesting these data could be more representative of real-world impact compared to results from a randomized clinical trial.
Limitations notwithstanding, these findings validate the short-term comparative utility of the bivalent mRNA booster vaccination in preventing infection due to Omicron sub-variants of SARS-CoV-2 in a diverse real-world population in the mid-south US.
Supplemental Material
sj-docx-1-tav-10.1177_25151355241278852 – Supplemental material for Comparative 60-day effectiveness of bivalent versus monovalent mRNA vaccines in Shelby County: a population-level analysis
Supplemental material, sj-docx-1-tav-10.1177_25151355241278852 for Comparative 60-day effectiveness of bivalent versus monovalent mRNA vaccines in Shelby County: a population-level analysis by Allison P. Plaxco, Jennifer Kmet, Matthew P. Smeltzer, Yu Jiang, Michelle Taylor and Vikki G. Nolan in Therapeutic Advances in Vaccines and Immunotherapy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
