Abstract
Background and Aims:
The peak of the third wave of COVID-19 infection was in the summer (August–September) of 2021, dominated by the Delta variant. Florida was the epicenter of the third wave with more than 151,449 cases in the first week of August with a positivity rate of 20%. The purpose of this study is to identify the percentage of COVID-19 infection in vaccinated patients in a minority population in south Florida and to elucidate the relationship, if any, between demographics and breakthrough infections, the rate of vaccine hesitancy, as well as the willingness to receive the monoclonal antibody REGEN-COV for the treatment of COVID-19.
Methods:
This cross-sectional study was performed at the Emergency Department, Larkin Community Hospital Palm Spring Campus, located in Hialeah, the fourth largest city in Florida. Hialeah is dominated (94.7%) by Hispanics and Latinos. This city represents a cross-sectional sample of US cities in general and Florida in specific. We enrolled 127 COVID-19 PCR-positive patients.
Results:
The infection in vaccinated patients (breakthrough) was found to be about one in three (34%). Despite the high infection rate and mounting death toll, about 73% of our unvaccinated patients answered no to the question ‘knowing the consequences of being infected with COVID-19 and the fact that you are positive, would you have chosen to be vaccinated earlier?’ However, about 27% of these patients agreed to receive the vaccine and 20.5% received the monoclonal antibody REGEN-COV.
Conclusions:
Our study revealed that vaccine hesitancy in South Florida continues to be a major challenge, especially with the emergence of mutations including Delta plus and Omicron.
Keywords
Introduction
The Center for Disease Control and Prevention (CDC) defines a vaccine breakthrough infection as “the detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) RNA or antigen in a respiratory specimen collected from a person ⩾ 14 days after they have completed all recommended doses of a U.S. Food and Drug Administration (FDA)-authorized COVID-19 vaccine.”
1
In the United States, as of 15 May 2022, over 257 million people (78% of the population), have received at least one dose of the US emergency use authorization (EUA) vaccine, while over 220 million people (66% of the population) are EUA fully vaccinated. 2 Slightly above 102 million US population (31%) have received a booster vaccine. 2
Currently, the FDA has approved three vaccines under Biologics License Application (BLA) or EUA, which includes Pfizer-BioNTech, Spikevax by Moderna, and Janssen’s by J&J. Pfizer-BioNTech vaccine is used as two primary doses starting at 5 years old with a third primary dose for 5 years and above in immunocompromised individuals. 3 The FDA has approved a single booster for individuals between 5 and 49 years old, while a second booster is approved for 50 years old and above. 3 Spikevax by Moderna was approved on 31 January 2022 for individuals 18 years old and above. 4 The FDA has approved two doses of Spikevax, with a single booster dose for individuals between 18 and 49 years old, and a second booster dose for 50 years old and above. 4 A third vaccine to get limited authorization under EUA as on 5 May 2022 is Janssen’s single-dose vaccine, with a single booster for 18 years old and above. 5 J&J COVID-19 vaccine is only authorized to be used for the individuals who cannot get previous two approved vaccines due to the medical condition or inaccessibility. 5
An exhaustive list of mutations of SARS-CoV-2 strains has been identified. The World Health Organization (WHO) has classified five variants as variants of concern (VOC); Alpha, Beta, Gamma, Delta, and Omicron, 6 and other WHO variants of interest (VOI) include Lambda and Mu. SARS-CoV-2 and its mutations have gripped the world since December 2019 and are responsible for over 326 million infections and over 5.5 million deaths, as of January 2021. 7 While there are over two dozen approved COVID-19 vaccines, 8 the mutations in the virus topology, viral load, as well as duration of viral RNA shedding play a role in the transmission of the disease. 9
Death from COVID-19 was reported in fully vaccinated; however, several groups have higher likelihoods of COVID-19 hospitalizations, such as non-Hispanic blacks and Hispanics, and COVID-19 vaccine breakthrough have infections, as have in those with substance use disorders and solid organ transplant.10–12
Moderna’s vaccine effectiveness is found to be about 93%, and Pfizer-BioNTech’s is 88% effective. 13 While in a study published in The Lancet, the risk of SARS-CoV-2 infection and SARS-CoV-2-related hospitalization is 26% and 37% lesser in Moderna’s vaccines as compared with Pfizer, respectively. 14
Factors such as ethnicity, race, gender, and age can also be contributing factors for infection post-vaccination or serious complications.15,16 For example, in Washington state, as of December 2021, infection surveillance reports reveal that Hispanics are leading second to whites in the number of COVID-19 breakthrough cases with 4868 cases of infections. 17 According to the Colorado vaccine breakthrough data as of December 2021, Hispanics had a three-time higher rate of COVID-19 vaccine breakthrough compared with other minority groups (blacks or African American, Asian, and native Hawaiian).18,19 Women compared with men, also, have a higher likelihood of a breakthrough infection in Colorado. 20
CDC reported the Delta variant to be behind most of the vaccine breakthrough infections but also emphasized that vaccines protect against severe illness and death, including from the Delta variant. 21 Recent reports showed that a third booster shot of the mRNA vaccine with at least a 5-month gap between the second and the third booster shot can improve the efficacy of the vaccine against the COVID-19-related severe illness and fatality. 22
During the pandemic, there has been extensive research into treatment options for COVID-19 infection and monoclonal antibodies have emerged as a promising option. Regeneron’s REGEN-COV is an antibody combination treatment consisting of monoclonal antibodies.23,24 Casirivimab and Imdevimab have received FDA authorization for emergency use for the treatment of mild-to-moderate COVID-19 and post-exposure prophylaxis in the adult and pediatric population at risk for a severe infection. 25 Other monoclonal antibody treatments with FDA EUA include Sotrovimab, Tocilizumab, and the combination treatment Bamlanivimab and Etesevimab. Among the 50 states, Florida and Texas are leading in monoclonal antibody administration. 26 As of 17 September 2021, in Florida, there have been 27,850 doses of REGEN-COV and 3100 doses of Bamlanivimab and Etesevimab administered, and in Texas 21,270 doses of REGEN-COV and 2370 doses of Bamlanivimab have been administered. 26
This study aims to establish the percentage of breakthrough infections based on demographics, type of vaccine received, the rate of vaccine hesitancy, and the factors affecting the willingness to receive the monoclonal antibody REGEN-COV for the treatment of COVID-19. These goals are achieved through analysis of a cross-sectional study of data arising from a survey during the third peak of COVID-19 at a community hospital in Hialeah, Florida.
Subjects and methods
Study setting
This study, conducted from 28 July to 1 September 2021, during the peak of the third wave of COVID-19 (Delta variant) in Florida, at the Emergency Department (ED) of Larkin Community Hospital, Palm Springs Campus, Hialeah, Florida. 27 Florida’s highest one-day total of 21,683 cases of COVID-19 was reported on day two of the study. 28 Since the beginning of the pandemic, Larkin Community Hospital has received all patients in its ED regardless of COVID-19 status.
The study was approved by Larkin Community Hospital Institutional Review Board Protocol #: LCH-3-112020.
Study design and population
The purpose of the study is to identify the percentage of COVID-19 infection in vaccinated patients in a minority population in south Florida, and to elucidate the relationship between demographics and breakthrough infections, the rate of vaccine hesitancy, as well as the willingness to receive the monoclonal antibody REGEN-COV for the treatment of COVID-19.
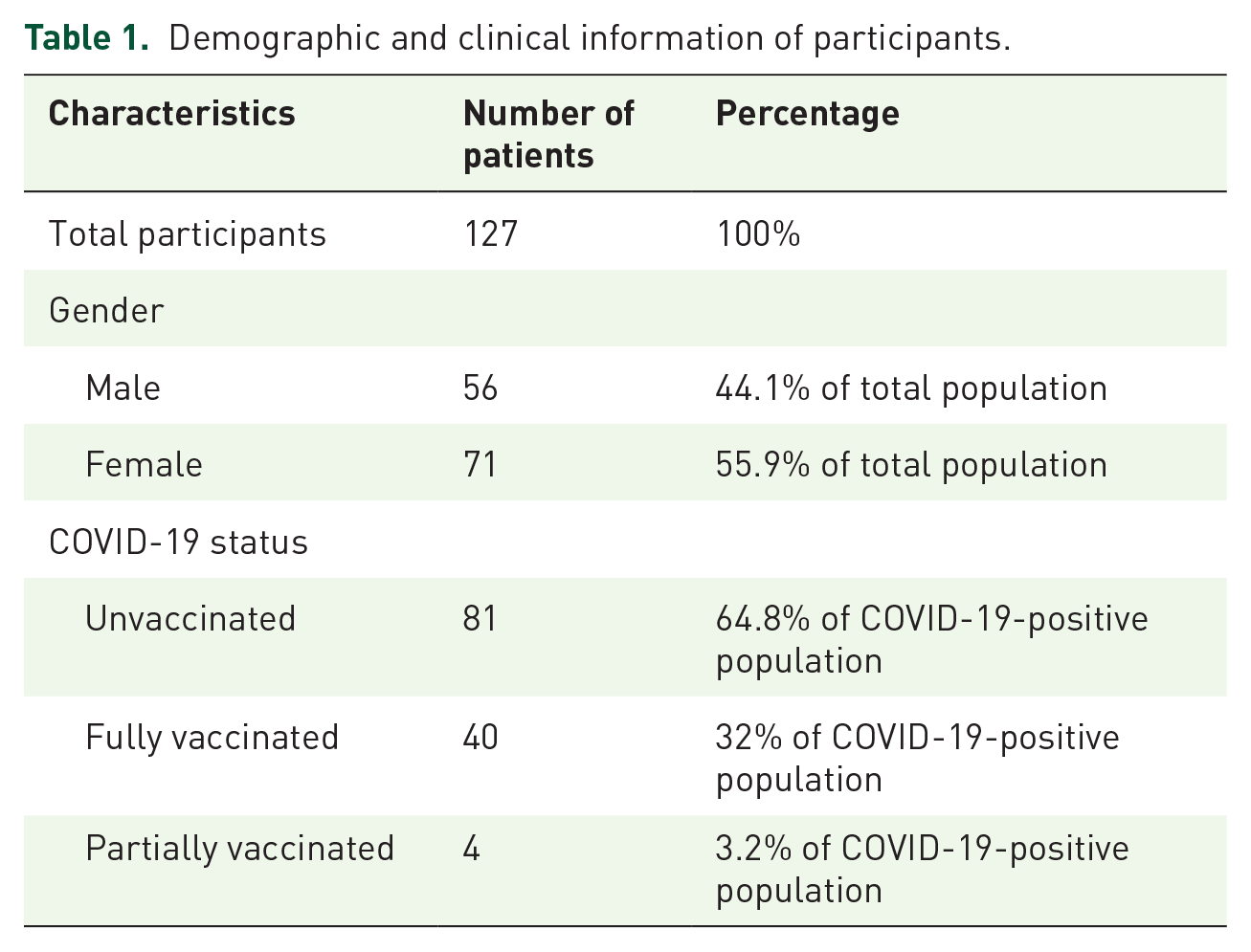
In this cross-sectional study, we recruited 127 patients (Table 1) from our ED. The study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 29 The population under study consisted of both vaccinated and unvaccinated, and 125 patients identified as Latino or Hispanic adults, who were confirmed to be COVID-19 positive by PCR in the ED. According to the 2010 US Census, Hialeah, Florida, where our ED is located, is 94.7% Hispanic or Latino. 30
Demographic and clinical information of participants.
Eligibility criteria
This study included only adults who presented to the ED and tested positive for COVID-19 RNA using PCR. After a brief explanation from a trained physician, participants were given the option to consent to participate in the study.
Data and sample collection
Per CDC infection control recommendations, 31 upon arrival to the ED, patients were promptly screened for symptoms of COVID-19 and triaged into well-ventilated areas accordingly. Testing for the presence of COVID-19 with a PCR test was performed on all patients, regardless of the presence of symptoms or vaccination status. Nasopharyngeal swabs were collected by trained personnel, either ED physicians or nurses. In addition, all COVID-19 positive patients were contacted by phone about a week after their initial visit and given a questionnaire to complete. The questionnaire assessed the patient’s demographics, vaccination status, the type of vaccine received (Pfizer, Moderna, or J&J), and whether Regeneron’s REGEN-COV (monoclonal antibody) treatment was received. The patient’s willingness to be vaccinated was assessed by asking: (1) ‘knowing the consequences of being infected with COVID-19 and the fact that you are positive, would you have chosen to be vaccinated earlier?’ and (2) ‘now that you are positive, would you want to be vaccinated?’ Patients who were Spanish speaking were interviewed after being discharged from the hospital in Spanish by Spanish-speaking staff. The study participants were both English and non-English speakers, which may introduce a bias in the way the questions are understood and answered.
Statistical analysis
The study used descriptive statistics to report on the frequency of various demographic and clinical characteristics. Given the impacts of age, gender, immunization status, and willingness to be treated with Regeneron were used. An assessment was performed on how likely patients would want to be vaccinated once their present infection had resolved.
Results
This study was conducted during the peak of the third wave of the COVID-19 Delta variant in the ED of our community hospital in Hialeah, Florida. The virus uses the spike protein to attach and enter human cells, replicating and overwhelming the immune system of affected individuals, especially the immunocompromised. The third wave of the COVID-19 was dominated by the Delta variant where the dominant mutations occurred due to mutations on the virus’s protein. In our study, the cross-sectional analysis was performed following STROBE criteria and included all 127 patients who visited our ED and who had a PCR-positive test for COVID-19 infection. Except for two patients, the population under study were 100% Latinos/Hispanics.
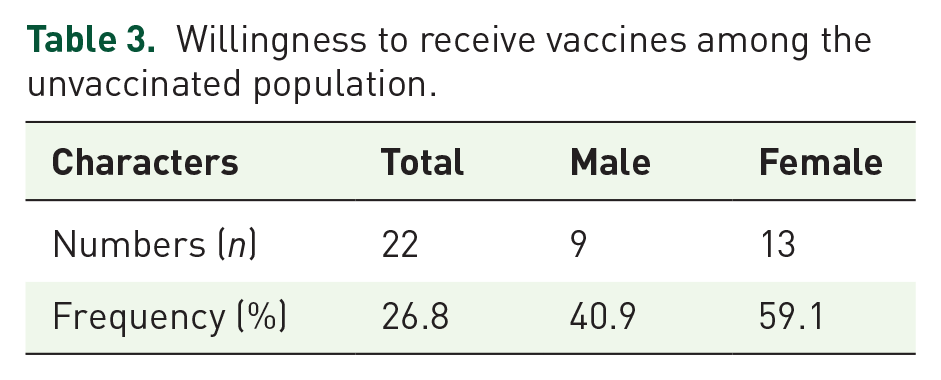
Table 1 shows all 127 participants’ demographics and clinical information. About 64.8% of the patients enrolled were unvaccinated with about 57% females and 43% males. The overall gender distribution consisted of about 44.1% male and 56% females. The average age of the patient population was 46.7 years. While 77.5% of the patients had received Pfizer, 12.5% had received Moderna, and 10% had received J&J (Table 2). Among the 82 unvaccinated patients, 42.7% were male and 57.3% were females. Of those unvaccinated, about 27% expressed an interest in getting vaccinated (Table 3). Regeneron was administered to 26 patients, and 13 patients were admitted with severe symptoms for further management. Among the three different vaccinated groups, Pfizer-vaccinated patients had more frequent admission than Moderna or J&J (Figure 2). Vaccine hesitancy for all patients enrolled in this study was assessed and varied across individuals. However, a common finding was a lack of trust in the potency of the vaccine and a fear of the unknown. In addition, the patient’s decision to receive the vaccine before falling ill or at their current ED visit was evaluated using the appropriate native language (English or Spanish) and documented accordingly. Table 4 shows vaccine hesitancy among unvaccinated participants. About 73% of unvaccinated remain hesitant to receive vaccines.
Vaccination status and vaccine type of participants.

Fully vaccinated comprises two doses Pfizer-BioNTech, two doses Moderna, and one dose Johnson & Johnson.
Provided exclusively at the emergency room to patients not admitted to the hospital.
Willingness to receive vaccines among the unvaccinated population.
Vaccine hesitancy among unvaccinated participants.

Discussion
The COVID-19 virus is transmissible via respiratory droplets caused by SARS-CoV-2. 32 The virus uses the spike protein to attach and enter human cells, replicating and overwhelming the immune system of affected individuals. 33
Although the historical relevance of vaccines in preventing infectious diseases such as measles, Ebola, and polio have been shown to be significant over the years, the recent RNA-based COVID-19 vaccines posed a lot of controversies, mainly because it was the first of its kind, with little or no data to support its efficacy. 34 This may explain the ‘fear of the unknown’ expressed by most of the patients who visited Larkin Hospital ED and were hesitant about receiving the vaccine. Recently, it has been reported that RNA-based vaccines had the highest efficacy of all vaccines with 94.29% compared with 70% efficacy of the other vaccines. The adverse drug reactions (ADR) experienced with the RNA vaccines were pain, fatigue, and headache. 35 Furthermore, effective communication of these findings to patients may help reduce some of their doubts, and perhaps hesitancy toward receiving the various COVID-19 vaccines.
The vaccine may prove to be effective in not only controlling the pandemic but also preventing a future outbreak. As of June 2022, over 12.02 billion doses of vaccines have been administered globally covering about 66.4% of the worldwide population. 36 According to the CDC, a vaccine breakthrough infection happens ‘when a fully vaccinated person gets infected with COVID-19’. Fully vaccinated people with a vaccine breakthrough infection are less likely to develop serious complications than those who are unvaccinated. In this study, about 32% of patients were fully vaccinated (Table 1 and Figure 1). The risk of getting severe illness and hospitalization encounters is 96% lower in the fully vaccinated population versus the unvaccinated. 37 About 11% of our patients had complications that necessitated admission for treatment (Table 1 and Figure 1). As vaccines are not 100% effective, as the number of people who have fully vaccinated increases, the number of vaccine breakthrough infections is also bound to increase. However, the risk of infection remains much higher for unvaccinated than vaccinated people and the symptoms tend to be more severe in them as well. As seen in our findings documented in Table 1, 64.8% of Covid-19-infected patients at Larkin ED were unvaccinated.

Demographics of the study population comparing the vaccinated versus unvaccinated.
As of 17 September 2021, in Florida, there have been 27,850 doses of REGEN-COV and 3100 doses of Bamlanivimab and Etesevimab administered, and in Texas 21,270 doses of REGEN-COV and 2370 doses of Bamlanivimab have been administered. 26 Currently, this therapy is available for use in all states and approved for emergency use under the EUA by the FDA for certain patient populations including black and Hispanic populations that are at risk for severe disease because of COVID-19 infection. 38 The four main monoclonal antibodies utilized are Casirivimab–Imdevimab, Sotrovimab, Tocilizumab, and Bamlanivimab–Etesevimab. The mode of administration is intravenous (IV) and is commonly administered in an outpatient setting. Research revealed that when monoclonal antibodies are used within 7–10 days of symptoms onset, the maximum benefit of the drug is attained. 38 In our study, it is important to note that about 20% of the studied sample was prescribed with Regeneron in the ER for active COVID-19 infection and the majority had symptoms improved within 3–4 days (data not shown), while only about 11% required hospitalization for further management. Patients who received the Regeneron monoclonal antibodies within 10 days of COVID-19 infection showed a rapid overall improvement with little or no sequelae. Nonetheless, more research is needed to substantiate the efficacy of monoclonal antibodies in the treatment of COVID-19 infection. It is important to stress that monoclonal antibody treatments are not an alternative to vaccine administration, which should take precedence in the fight against COVID-19 infection.
In our study, the incidences of vaccine breakthrough infections in Moderna are found to be lower than the Pfizer (Table 1 and Figure 2). 39 People who are vaccinated with Pfizer vaccines have greater susceptibility and are prone to breakthrough infection as compared with those who have received the Moderna.11,40 Similarly, the vaccine effectiveness was found to be less, or greater chances of vaccine breakthrough infections were found in J&J one-dose vaccine as compared with the Moderna and Pfizer two-dose vaccines. 41 Other risk factors involve male gender with immunocompromised status, and a weakened pulmonary system to be prone to vaccine breakthrough infections. 39
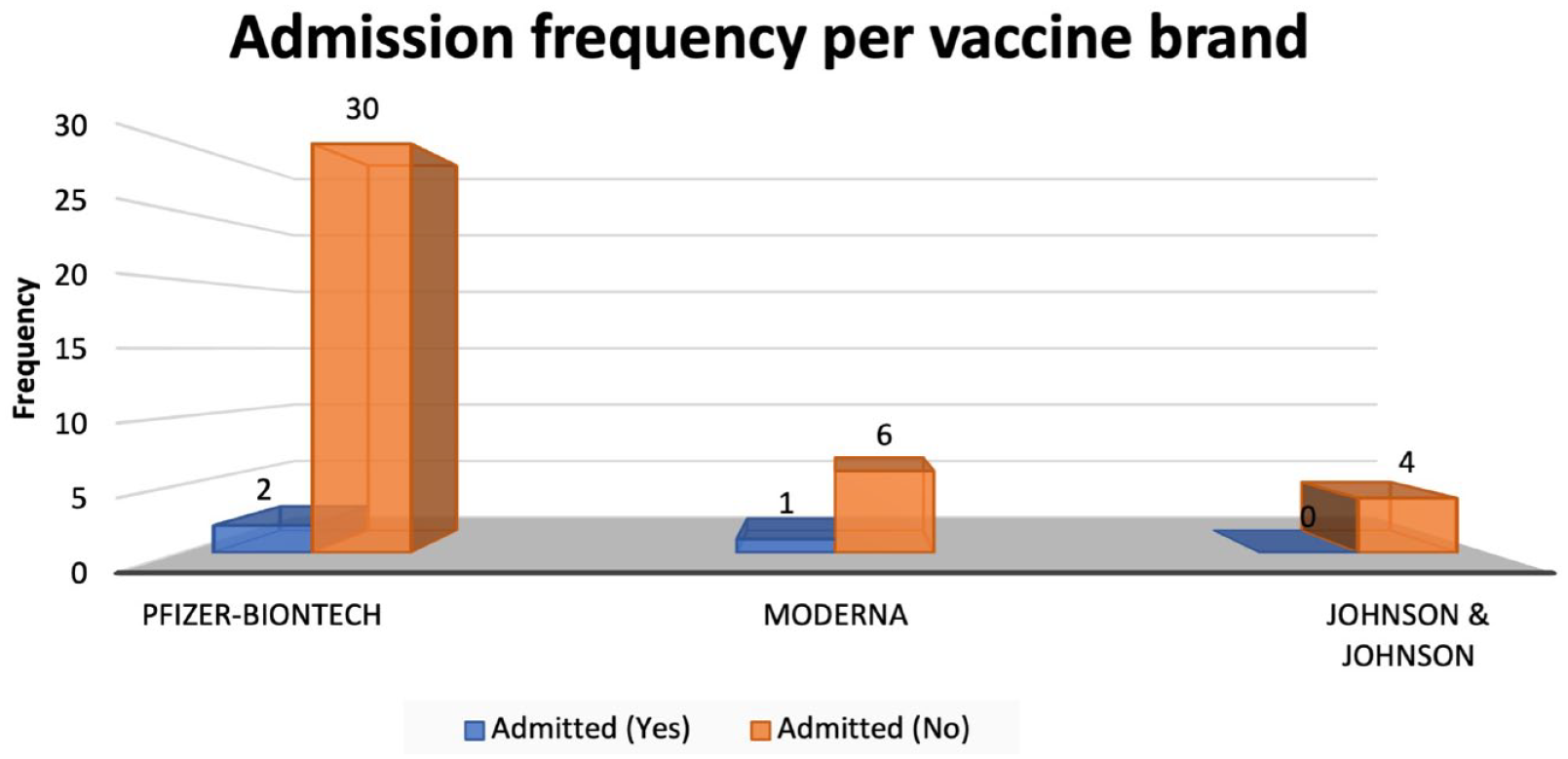
Admission frequency per vaccine brand.
It is important to understand that Standard Operating Procedures (SOPs) formulated by the government and doctors around the world should still be followed, regardless of the vaccination status. One of the reasons for the breakthrough infections was the larger public gathering in the summer. As was witnessed in Barnstable County, Massachusetts when despite being fully vaccinated 74% out of 469 cases were detected with the COVID-19.42,43 As of 26 October 2021, the total number of cases for vaccine breakthrough in Massachusetts is 51,007 cases, which form 1.08% of the total vaccinated population. The total number of deaths in Massachusetts due to breakthrough infection is 406, which represents 0.009% of the total fully vaccinated population. 30
The ‘SAGE Working Group on Vaccine Hesitancy’ concluded that vaccine hesitancy refers to ‘delay in acceptance or refusal of vaccination despite the availability of vaccination services’. Vaccine hesitancy is a rapidly evolving global problem that requires adequate attention and monitoring. 44 Factors resulting in vaccine hesitancy include multiple family members living together, distrust in the information provided by the government due to political mistrust, not being forthcoming about vaccine status, and not believing in vaccine efficacy. Vaccine hesitancy is attributed to be a consequence of broader influences and the historical, political, and socio-cultural context. Our hospital is situated in a Latino/Hispanic predominant neighborhood, and we came across many such cases where people seem to exhibit hesitancy.
Studies showed that COVID-19 infection has disproportionately affected Black and Hispanic communities in the United States, which may be due to social factors including a lack of proper public health messaging and suboptimal adoption of prevention efforts. The results of a survey carried out in one of the big cities of the United States suggested that Hispanic individuals were at higher risk for infection due to a variety of reasons. 45 In a study designed to explore the heterogeneity in transmission dynamics of COVID-19, it was found that the risks and deaths among Latino populations differ by region and several structural factors place Latino populations and particularly Spanish-speaking individuals at an increased risk for infection.46,47
Limitations
Although Larkin community hospital receives a diverse patient population daily, this cross-sectional study was conducted using data from a limited period of 36 days and as a result, the sample size examined is small. Nonetheless, the aim of this article was achieved and can be inferred to a larger diverse patient population. Furthermore, information about the religious background of the patients is unknown, and this could explain some additional reasons for vaccine hesitancy among some patients. As data were collected in two languages, a methodological bias might be introduced.
Conclusion
In this study, 127 patients were enrolled, of which 44% were males, and 56% were females who were COVID-19 positive following the PCR test. While the infection rate in fully vaccinated patients in the United States was about 7%, in our hospital in south Miami we reached 35% at the peak of infection in late August 2021. Thirty-two percent of the patients testing COVID-19 positive after PCR testing were fully vaccinated with either Pfizer, Moderna, or J&J vaccines while 64.8% remained unvaccinated. Breakthrough infections in the fully vaccinated population were highest in those who took the two-dose Pfizer vaccine (77.5% of fully vaccinated). Pfizer was the highest vaccine among the available vaccines and thus most admissions of patients with severe symptoms were Pfizer vaccinated. As the population under study was Latino or Hispanic, a relationship was unable to be established between race/ethnicity and breakthrough infections. However, the highest rate of breakthrough infections was seen among females and in the age group > 60 (Figure 1). All enrolled unvaccinated patients were asked whether they would be willing to receive the vaccine at their ED visit and 26.8% were willing; however, 73.2% of the unvaccinated population were hesitant to receive the vaccine with the majority being female (56.7%). Factors relating to vaccine hesitancy and breakthrough infections need to be better understood among our population and addressed especially with the emergence of new mutations such as Delta plus and more recently, the Omicron variant. Monoclonal antibody treatment is an effective treatment for COVID-19 infections nationwide and about a quarter of the total population (20.5%) under study received the REGEN-COV antibody treatment.
