Abstract
Messenger ribonucleic acid-based vaccines that target severe acute respiratory syndrome coronavirus 2 have shown high effectiveness. While these Coronavirus Disease 2019 vaccines have a favorable safety profile, there can be rare adverse drug reactions that should be understood. Here, we report the case of a 65-year old male who displayed polymyalgia rheumatica soon after receiving the first dose of the BNT162b2 messenger ribonucleic acid Coronavirus Disease vaccine. Symptoms such as high fever and severe general pain occurred 10 days after vaccination. After administering more than 30 mg/day prednisolone, those symptoms persisted and inflammation continued until 90 days after vaccination. However, those symptoms disappeared over time, following vaccination, and the patient finally achieved complete remission from polymyalgia rheumatica without any additional treatment.
Introduction
The outbreak of Coronavirus Disease 2019 (COVID-19) rapidly spread internationally. 1 One desirable preventative method to overcome the COVID-19 pandemic was to develop a vaccine. Conventional vaccines were unable to effectively target severe acute respiratory syndrome coronavirus 2, whereas messenger ribonucleic acid (mRNA)-based vaccines promised to be a reliable solution to the COVID-19 pandemic response due to their flexible immunogen design. 2 A novel, lipid-nanoparticle-encapsulated mRNA vaccine expressing the prefusion-stabilized spike glycoprotein was developed independently by Pfizer BioNTech and Moderna.3,4 Both mRNA vaccines showed high effectiveness and safety in clinical trials involving large populations, so these mRNA vaccines were mass manufactured. Despite these successes, data on mRNA vaccines need to be accumulated to appreciate if there are any adverse drug reactions, even if they are rare cases. Here, we report the case of a 65-year old male who had no past significant medical history but developed polymyalgia rheumatica (PMR) after inoculation with the first dose of the Pfizer-BioNTech BNT162b2 mRNA COVID-19 vaccine. His PMR status was refractory in the initial phase after receiving the vaccine, but he achieved complete remission from PMR without any therapy almost 1 year after the vaccine was administered.
Case
A 65-year old Japanese healthy man was vaccinated in his left upper arm with the first dose of the BNT162b2 mRNA COVID-19 vaccine. He had no past medical history nor did he receive any medical treatments before inoculation. However, he developed pain in his right shoulder 10 days after receiving the first dose. He developed a fever soon after that pain, which spread throughout the body. He was admitted twice to a local clinic but was not diagnosed with any condition nor did he receive any treatment. He was admitted to our hospital 19 days after the onset, or 29 days after the first dose. Due to the severity of his symptoms, he did not receive the second dose of the COVID-19 vaccine.
Upon admission, his body temperature reached 38.8°C and he claimed to have severe general pain, which caused overall body discomfort. Due to its severity, he was unable to determine whether that pain was occurring in joints or muscles. However, inflammation of peripheral joints, including the metacarpophalangeal or proximal interphalangeal joints, were lacking, and his pain showed an asymmetrical distribution. A manual muscle test revealed that his muscle strength decreased to four in limb muscles, specifically the bilateral deltoid, biceps, triceps, wrist extension, wrist flexion, extensor digitorum, flexor digitorum, gluteus maximus, iliopsoas, hamstrings, quadriceps femoris, and tibialis anterior muscles. A laboratory examination upon admission showed an elevated white blood cell (WBC) count of 9,500/µL, an erythrocyte segmentation rate (ESR) of 84 mm/h, and C-reactive protein (CRP) content of 12.2 mg/dL. Serum ferritin level was 128.4 ng/mL (normal range; 13–277 ng/mL). The results of other laboratory tests, including for rheumatic factor and auto-antibodies, showed normal values. Chest X-ray and chest to abdominal computed tomography examinations showed no abnormal findings. Considering both clinical and biological findings, we diagnosed the patient with PMR based on the 2012 EULAR/ACR classification criteria of PMR, 5 with a score of 5, differentiating it from other autoimmune diseases including rheumatoid arthritis, 6 infectious diseases, and tumors.
Considering the severity of his symptoms, we initiated prednisolone at 50 mg/day (1 mg/body weight kg/day), then gradually decreased the dosage but maintained it at 30 mg/day until 40 days after the first dose of the vaccine had been administered. Our expectation was that his symptoms would dramatically improve, but his symptoms and laboratory data did not improve sufficiently. Although his fever disappeared and pain improved only partially, he continued to suffer from general pain in joints. A laboratory examination at 40 days after vaccination showed an elevated WBC of 18,500/µL, ESR of 34 mm/h, and CRP of 4.9 mg/dL. Although the elevated WBC could be due to the effect of prednisolone therapy, the elevated ESR and CRP values were accompanied by residual inflammation. Prednisolone at 30 mg/day was thus continued until 95 days after vaccination. Even though a higher dose of prednisolone and longer administration, relative to usual PMR cases, was used, his symptoms lingered and a laboratory examination showed an elevated WBC of 13,400/µL, ESR of 26 mm/h, and CRP of 2.8 mg/dL at 68 days after the vaccination.
We considered that one possibility was that the BNT162b2 mRNA COVID-19 vaccine may have been associated with progression of the illness and waited for the vaccine’s effect to disappear. Although the dosage of prednisolone had not changed, his symptoms disappeared suddenly 90 days after vaccination. The laboratory data 95 days after vaccination indicated a decrease in ESR to 9 mm/h and in CRP to 0.4 mg/dL. Prednisolone was gradually decreased to 15 mg/day, and both ESR and CRP values at 120 days after vaccination improved, reaching a normal range, 6 mm/h and 0.2 mg/dL, respectively.
After 8 months, no symptoms recurred and results of examinations of all parameters were within a normal range. Consequently, we withdrew prednisolone at 12 months. After treatment withdrawal, the patient did not display any PMR symptoms, even after 18 months. Based on the disease progression, our diagnosis was that transient PMR was induced by a single dose of the BNT162b2 mRNA COVID-19 vaccine.
The patient’s clinical course, including PMR progression, vaccine, and laboratory data, are summarized in Figure 1.
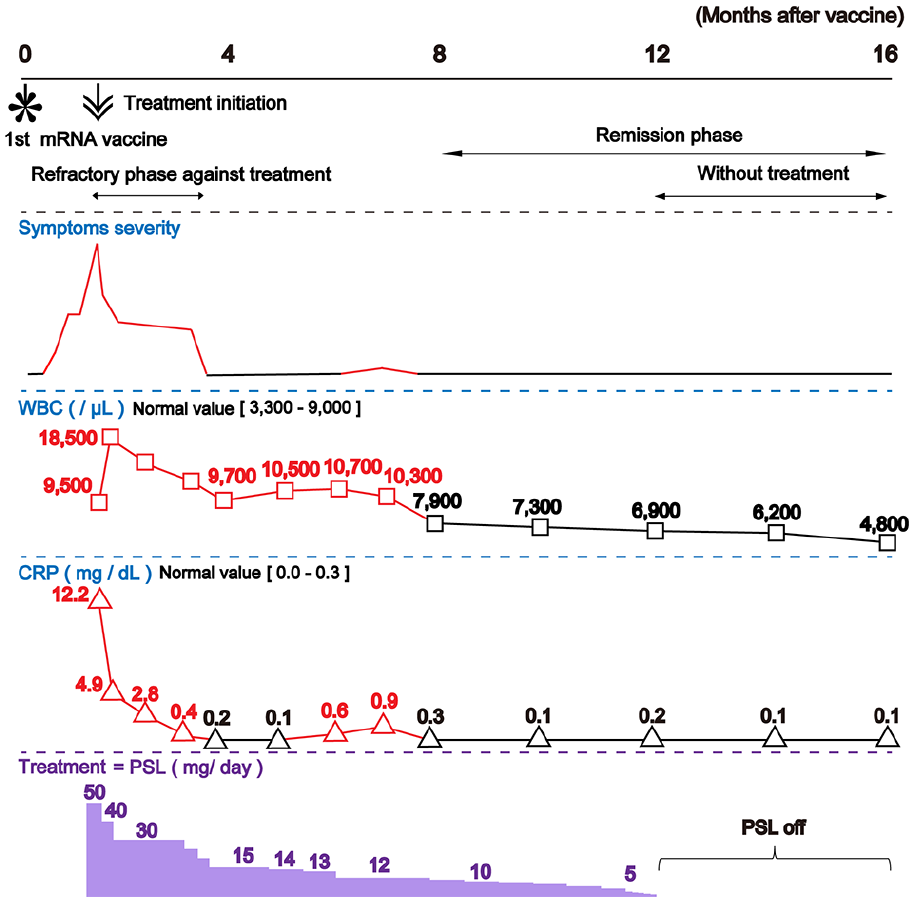
Patient’s clinical course (months 0–16). The relationships among the BNT162b2 mRNA Coronavirus Disease 2019 (COVID-19) vaccine, symptoms severity, laboratory data, and treatment are shown. Eight months after the first dose of the BNT162b2 mRNA COVID-19 vaccine, polymyalgia rheumatica promptly disappeared.
Discussion
The PMR case that we presented in this paper displayed a refractory response against high dose glucocorticoid therapy shortly after vaccination, but it was transient in the long term. Recent research has focused on the relationship between PMR and mRNA COVID-19 vaccines.7–10 Although the relationship between PMR and mRNA COVID-19 vaccines was unknown in the initial phase of the pandemic, cumulative reports suggest that PMR can follow the mRNA COVID-19 vaccine. 10 Thus, PMR pathology should be considered after the mRNA COVID-19 vaccine. The influenza virus vaccine can also induce PMR, and in its pathology, it is considered that the adjuvant induces inflammatory cytokine production such as interleukin-6 or tumor necrosis factor-α, leading to PMR.10,11 In contrast, mRNA COVID-19 vaccines do not contain any adjuvant, but they can stimulate innate immunity by activating Toll-like receptors. 3 This activation of Toll-like receptors is related to PMR following the mRNA COVID-19 vaccine. 10
A previous report indicated that both types of mRNA COVID-19 vaccine, BNT162b2 and mRNA-1273 (Moderna), were associated with PMR, which mostly occurred within 14 days after the first dose of the vaccine was received. 10 This outcome is the same as our presented case. In contrast, most PMR cases following the mRNA COVID-19 vaccine easily achieved remission when a certain amount of prednisolone was administered. 10 This is different from our presented case, which showed a refractory response against high dose glucocorticoid therapy shortly after vaccination. Given this difference, we focus on the relationship between genes and PMR. 9 Genetic factors can also play an important role in the pathogenesis of PMR. One report found an association between PMR and typing of human leukocyte antigen. 12 Genetic background might explain why certain individuals suffer adverse drug reactions, although no genetic analyses to assess links with PMR were conducted on our patient. We also emphasize that the adverse drug reaction, PMR, that was observed in our patient in response to one dose of the BNT162b2 mRNA COVID-19 vaccine, was both transient and ultimately reversible.
Conclusions
Some refractory but transient PMR can be induced after receiving the first dose of the BNT162b2 mRNA COVID-19 vaccine.
Footnotes
Acknowledgements
Authors’ contributions
M. K. was the attending doctor for the present case and drafted the manuscript. S.F. conceived the study, participated in its coordination, and helped draft the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
