Abstract
Bullous pemphigoid is recognized as the most prevalent autoimmune blistering skin disease. This report presents a case of a 54-year-old male in Canada who developed new-onset bullous pemphigoid 4 weeks after receiving his fifth COVID-19 vaccine booster (Pfizer-BioNTech). The patient initially presented with a generalized erythematous rash, which progressed despite treatment with topical and systemic corticosteroids. He later developed tense blisters and peripheral eosinophilia, with a skin biopsy confirming the diagnosis of bullous pemphigoid. Although bullous pemphigoid has previously been linked to various vaccinations, the underlying mechanisms remain uncertain. Notably, cases of bullous pemphigoid flare-ups have been reported in patients with existing bullous pemphigoid after receiving mRNA COVID-19 vaccines. This report underscores the importance of considering new-onset bullous pemphigoid in patients presenting with blistering skin conditions following COVID-19 vaccination.
Introduction
Bullous pemphigoid (BP) is considered the most common autoimmune blistering skin disease. It is characterized by the formation of autoantibodies, most commonly IgG, against structural basement membrane antigens. 1 The main BP antigens are BP antigen 1 (BP230) and BP antigen 2 (BP180). These hemidesmosomal proteins are crucial for maintaining the dermal‒epidermal junction. The binding of autoantibodies to BP antigens results in separation of the epidermis and dermis through an inflammatory process. 2
BP most commonly affects elderly individuals, with the majority of cases occurring in patients over the age of 60. 3 The typical clinical presentation of BP includes the formation of tense blisters on normally appearing skin or, less commonly, on an erythematous base. Other associated cutaneous abnormalities, such as urticaria and erythema multiforme, can also be observed in patients with BP. Almost all patients present with severe pruritus. Mucosal involvement has been reported but is not a common feature. 4 The diagnosis of BP is suspected on the basis of the clinical presentation and confirmed by obtaining a skin biopsy via direct immunofluorescence, which reveals linear deposits of IgG or complement component 3 (C3) at the dermal‒epidermal junction. Circulating autoantibodies can also be detected in some patients via enzyme-linked immunosorbent assay (ELISA).
The exact etiology of BP is largely unidentified. Certain factors have been linked to the occurrence or exacerbation of the disease, referred to as “trigger factors,” including medications, trauma, burns, vaccines, infections, and others. 1 Vaccine-induced BP is rare, with a few case reports of new-onset BP triggered by a variety of vaccines, including herpes zoster, tetanus toxoid, influenza, rabies, and meningococcal.
Multiple adverse cutaneous reactions to mRNA-based COVID-19 vaccines, including BP have been reported. 5 In addition, patients with underlying autoimmune blistering skin disease have shown evidence of disease flares after receiving the Pfizer-BioNTech (Pfizer Inc., New York, NY and BioNTech SE., Mainz, Germany) vaccine during remission. 6
We report the first case in Canada of new-onset BP in a male following the administration of the fifth COVID-19 mRNA vaccine with Pfizer-BioNTech.
Case report
We describe the first case in Canada involving a 54-year-old male with a known history of dyslipidemia who was managed with rosuvastatin for 5 years and who presented to the emergency department 4 weeks after his fifth COVID-19 mRNA vaccine booster with a widespread erythematous rash. He had no associated fever, chills, joint aches, or any other systemic symptoms. There was no new medication exposure, including antibiotics and nonsteroidal anti-inflammatory drugs. His family history was negative for significant diseases, autoimmune conditions, and skin disorders. The patient had no history of autoimmune diseases and reported no adverse skin reactions to the previous four doses of the COVID-19 mRNA vaccine or other vaccinations. On physical examination, numerous nontender erythematous annular plaques were observed on the back (Figure 1). There were no blisters or mucosal lesions. The patient was afebrile and hemodynamically stable. He was initially treated as an outpatient with topical and systemic corticosteroids (prednisone 0.5 mg/kg); however, the patient returned to the emergency department as the rash progressed to involve the perineum and inguinal folds. In addition, he developed generalized tense blisters on both arms and peripheral eosinophilia (Figure 2). The patient was hospitalized, and two skin biopsies suggested a diagnosis of BP. Direct immunofluorescence showed a linear band of C3 along the basement membrane, but the diagnosis could not be confirmed definitively. This was due to the unavailability of tests like ELISA for BP180 NC16A, salt-split immunofluorescence, and immunoblotting, which are crucial for ruling out other conditions such as epidermolysis bullosa acquisita and anti-p200 pemphigoid. The patient’s eosinophil count peaked at 2.33 × 109/L. Due to clinical progression, dermatology and allergy specialists recommended increasing the prednisone dose to 1 mg/kg orally. Wound care and topical steroids were continued during his hospital stay. His condition was complicated by a bacterial infection, with Staphylococcus aureus detected in blood cultures, requiring intravenous (IV) cefazolin treatment. An echocardiogram showed no signs of infective endocarditis. After consultation with the infectious disease team, he completed a 4-week course of IV cefazolin.

Erythematous coalescing targetoid annular plaques on the back.
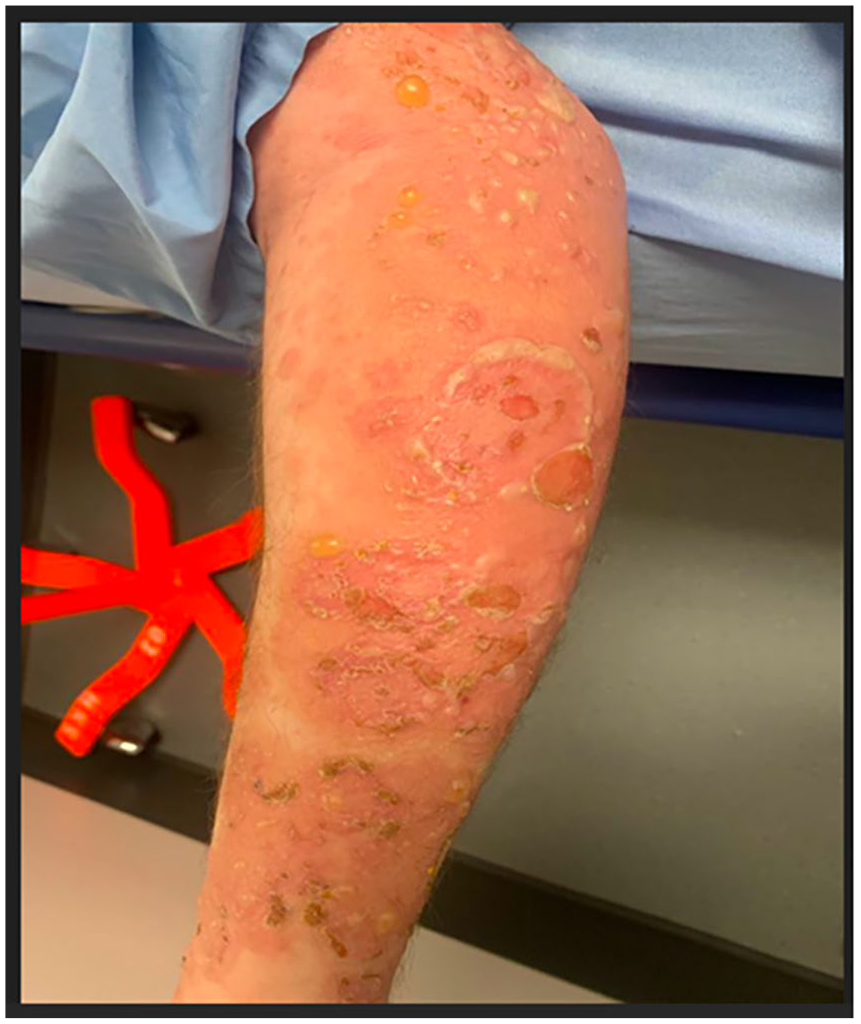
Tense bullae associated with erosions and crusting on the left arm.
The patient was discharged on oral prednisone (1 mg/kg) and scheduled for outpatient dermatology follow-up. Despite being on systemic steroids, he continued to develop new blisters, so mycophenolate was added, and prednisone was gradually tapered. Three months later, he was off prednisone and remained in clinical remission with just mycophenolate. He has not received any further vaccinations since.
Discussion
BP has been associated with the administration of various vaccines, including COVID-19 mRNA vaccines. 5 The patient has no other risk factors for developing BP, including neurological conditions, autoimmune diseases, and high-risk medications. 1 The patient’s age and sex align with the typical characteristics affected by BP. In an Italian multicenter study, the median age at which BP was triggered by COVID-19 mRNA vaccines was 82, with a male-to-female ratio of 3:1. 5 The period between vaccine injection and the occurrence of BP varies from 1 day to 1 month. Reports of new-onset BP following the first dose of COVID-19 mRNA vaccines are consistent with this, with variable latency periods up to 1 month. 7 Additionally, some patients develop BP following the second booster dose of the COVID-19 mRNA vaccine as soon as 1 day 8 and up to 15 days after vaccination. 9
The pathogenesis of vaccine-triggered BP remains unclear. Given the structural differences between the vaccines and basement membrane proteins, a direct antibody response to the vaccines is unlikely to be the cause. 10 Some studies propose that patients may express high levels of BP antigens without displaying any clinical symptoms. 9 Experimental studies in mice have established that immunization could lead to the loss of tolerance to BP180, resulting in a subepidermal blistering disease similar to BP in humans, characterized by predominant eosinophilic infiltrate. 11 Another theory posits that the inflammation provoked by vaccination may disrupt the basement membrane zone, unveiling hidden antigens that lead to the production of anti-basement membrane-specific antibodies. 12
Flares of BP have also been described in patients with known BP who are in clinical remission, triggered by receiving mRNA vaccines for COVID-19. Following adequate treatment, most patients are able to receive booster doses without further exacerbations. 13 A minority of patients developed subsequent flares after their booster vaccinations; however, they were still able to resolve completely with systemic therapies. 14 Importantly, most BP patients do not experience exacerbations after receiving the COVID-19 mRNA vaccine.
Our findings align with the growing body of literature suggesting that while autoimmune blistering diseases (AIBDs) may occur following COVID-19 vaccination, such cases appear to be rare. Notably, a large population-based cohort study of over 112 million individuals demonstrated that COVID-19 infection itself is associated with a significantly increased risk of developing AIBDs, whereas vaccination was linked to a decreased risk. These results support the notion that vaccination may have a protective effect against autoimmune skin conditions, in addition to its well-established role in preventing severe COVID-19. This context highlights the importance of viewing post-vaccination autoimmune phenomena as rare exceptions rather than indications of broader risk. 15 Overall, the majority of patients who receive COVID-19 mRNA vaccines do not develop BP, and vaccination remains an effective method to mitigate the risk of acquiring severe acute respiratory syndrome coronavirus.
Conclusion
Diverse cutaneous reactions, including erythema multiforme-like eruptions, eczematous eruptions, psoriasis exacerbations, urticarial vasculitis, and BP, have been reported with COVID-19 vaccinations. It is important to consider new-onset BP in patients with blisters after mRNA vaccination. This report is not intended to question the safety of mRNA COVID-19 vaccines, but rather to increase awareness of this presentation and encourage early recognition and treatment.
Footnotes
Author note
This abstract was previously presented as a poster at the Clinical Society of Allergy and Clinical Immunology (CSACI) annual meeting in Quebec City, October 2023.
Ethical considerations
Our institution does not require ethical approval for reporting individual cases.
Consent for publication
Written informed consent was obtained from the individual for the publication of any potentially identifiable images or data included in this article.
Author contributions
Eman Badawod: conceptualization, data curation, investigation, resources, writing – original draft, and writing – review and editing.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
