Abstract
Background:
Due to the COVID-19 situation, vaccination is a key factor in reducing the severity and transmission of the disease, especially in the vulnerable population, which includes pregnant women. Currently, various policies are in place to promote the vaccination of pregnant women against COVID-19; however, some pregnant women decline vaccination.
Objectives:
To study pregnant women’s knowledge, attitude, and acceptance regarding the COVID-19 vaccine.
Design:
A cross-sectional study was conducted among pregnant women who received antenatal care at King Chulalongkorn Memorial Hospital, Chulalongkorn University, Bangkok, Thailand.
Methods:
Pregnant women who received antenatal care at King Chulalongkorn Memorial Hospital, Chulalongkorn University from November 2021 to April 2022 were included. The participants answered an online questionnaire through their electronic devices. Univariate and multivariate logistic regression analyses were performed to analyze the data.
Results:
A total of 500 pregnant women participated in this study. Among them, 67.4% and 81.4% had great knowledge and a positive attitude toward the COVID-19 vaccine, respectively, with scores of 80% or higher in each section. Of the 500 participants, 468 (93.6%) accepted to receive the COVID-19 vaccine. After adjusting for certain variables through multivariate analysis, the factor associated with the decision to receive the COVID-19 vaccine was having great knowledge about the vaccine (adjusted odds ratio (OR) 13.25, 95% confidence interval (CI) 2.45–71.61). However, the most significant factor associated with the decision to reject the COVID-19 vaccine was the recent COVID-19 infection (adjusted OR 0.11, 95% CI 0.02–0.62).
Conclusion:
The COVID-19 pandemic presents severe and life-threatening conditions for both pregnant women and their fetuses. The majority of the pregnant women in this study had great knowledge and acceptance of the vaccine and a positive attitude toward it. The pregnant women who had prior knowledge of the vaccine tended to accept to receive it during pregnancy, whereas those who recently contracted COVID-19 were hesitant to receive the vaccine.
Plain language summary
Due to the COVID-19 situation, vaccination is a key factor in reducing the severity and transmission of the disease, especially in the vulnerable population, which includes pregnant women. Currently, various policies are in place to promote the vaccination of pregnant women against COVID-19; however, some pregnant women decline vaccination. This study aimed to investigate pregnant women’s knowledge, attitude, and acceptance regarding the COVID-19 vaccine. A total of 500 pregnant women were recruited. The study found that 67.4% and 81.4% had great knowledge and a positive attitude toward the COVID-19 vaccine, respectively, with scores of 80% or higher in each section. 93.6% accepted to receive the COVID-19 vaccine. The factor associated with the decision to receive the COVID-19 vaccine was having great knowledge about the vaccine. However, the most significant factor associated with the decision to reject the COVID-19 vaccine was the recent COVID-19 infection. In summary, the majority of the pregnant women in this study had great knowledge and acceptance of the vaccine and a positive attitude towards it.
Introduction
In December 2019, the World Health Organization announced that SARS-CoV-2, a new virus, was the cause of the pandemic. It is a single-stranded, capsulated RNA virus that can be spread through droplet transmission or airborne transmission by aerosol-generating procedures. This viral infection can cause severe pneumonia that leads to respiratory failure or even death.1,2
Pregnant women who have COVID-19 infection are at greater risk of being admitted to hospital intensive care units and mortality. COVID-19 infections tend to cause more severe respiratory illnesses in pregnant women than in the general population, mainly due to the decrease in residual lung volume and pulmonary reserve during pregnancy.3,4 Moreover, pregnant women are also at higher risk of developing thromboembolic events as a result of an altered hypercoagulable state during pregnancy. 4
For fetuses and newborns, there is no serious complication other than preterm birth, which only occurs in severe cases of maternal COVID-19 infection.4–6 Congenital COVID-19 infection is rare, as the SARS-CoV-2 viral RNA level is usually undetectable in newborns and asymptomatic. 4
From the latest update on December 2022, the COVID-19 pandemic has caused a major effect on our global health system, with more than six million deaths from a total of 600 million infections worldwide. 2 Meanwhile, in Thailand, there have been approximately four million reported cases of infection, with 30,000 deaths. 7
Several vaccines have been invented to prevent and mitigate the severity of COVID-19. Currently, the Food and Drug Administration of Thailand has approved six vaccines: AstraZeneca, CoronaVac, Johnson & Johnson, Moderna, Sinopharm, and Pfizer-BioNTech. 8 The Pfizer-BioNTech and Moderna vaccines, which contain a part of the mRNA virus, have good efficacy in preventing COVID-19 infection, at 95% and 94.1%, respectively.9,10 The AstraZeneca and Johnson & Johnson vaccines, both adenovirus vector vaccines, have preventive efficacy rates of 70.4% and 66.9%, respectively. 11 The CoronaVac and Sinopharm vaccines, made of dead SARS-CoV-2 viruses, have a preventive efficacy rate of 50%–84% and 79%, respectively.12,13 The World Health Organization strongly recommends the vaccination of pregnant women against COVID-19 as the benefits outweigh the potential risks. 14
In a literature review examining the acceptance of the COVID-19 vaccines, a cross-sectional study conducted in Libya in 2020 interviewed 15,087 people above18 years old via electronic mail and social media. The study revealed that 79.6% of the respondents expressed willingness to receive the vaccine with an efficacy rate of over 90%, 60.6% were willing to receive the vaccine with an efficacy rate of over 70%, and only 41.2% were willing to receive the vaccine with an efficacy rate exceeding 50%. 15 Another cross-sectional study conducted in 2020 interviewed 17,871 people from 16 countries via an online questionnaire. The participants were pregnant women, non-pregnant women aged 18 years and above, and mothers of children under 18 years old. The results of the study revealed that 52% of the pregnant women expressed a preference for receiving the COVID-19 vaccine during pregnancy. 16 Previous study regarding COVID-19 vaccine acceptance in general population in Thailand found that 88.4% of the participants accepted the vaccine. Males and high education significantly correlated with vaccine acceptance. 17
Some studies demonstrated hesitancy in the COVID-19 vaccine in pregnant women18,19 and parents.20,21 The vaccine hesitancy rate in pregnant women was 40.7%–57%.18,19 Women with vaccine hesitancy were more likely to fear adverse reactions, safety, and efficacy compared to those without vaccine hesitancy. 19 The vaccine hesitancies were largely based on the lack of knowledge about the impacts of vaccination on pregnancy, fetal development, and later child well-being. 18 Regarding the hesitancy of the COVID-19 vaccine in parents, behavior, safety and efficacy, and general attitudes of parents toward childhood vaccines were the risk factors associated with the hesitancy in the parents from Zhejiang. 21 Previous systemic review found that vaccine hesitancy rates ranged from 26% to 57% among different studies. Fear of adverse events and lack of knowledge were shown to be the main drivers of hesitancy. 22
Due to the COVID-19 situation, vaccination is a key factor in reducing the severity and transmission of the disease, especially in the vulnerable population, which includes pregnant women. Currently, various policies are in place to promote the vaccination of pregnant women against COVID-19; however, some pregnant women decline vaccination. This study aimed to examine Thai pregnant women’s knowledge, attitude, and acceptance regarding the COVID-19 vaccine. The research questions of this study were as follows: (1) What is the level of knowledge regarding COVID-19 vaccination among pregnant women? (2) What is the COVID-19 vaccine acceptance associated with pregnancy?
Materials and methods
This was a cross-sectional study carried out at the Department of Obstetrics and Gynecology, King Chulalongkorn Memorial Hospital, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand. The research protocol was approved by the Institutional Review Board of the Faculty of Medicine, Chulalongkorn University (IRB no. 622/64). King Chulalongkorn Memorial Hospital is a large tertiary care hospital and a university hospital, located in Bangkok, capital city of Thailand. It has 1500 beds for inpatient admissions.
This study was conducted between November 2021 and April 2022 and included 500 Thai pregnant women aged 18 years and above who visited the antenatal care clinic at King Chulalongkorn Memorial Hospital. Pregnant women who were unable to read or write in Thai and those without access to electronic devices as the participants were to use their own electronic devices were excluded from the study. The researchers recruited the participants which were pregnant women who visited antenatal care clinic of King Chulalongkorn Memorial Hospital. Details of the questionnaire were explained, and confidentiality was confirmed. Informed consent was performed before the questionnaire was given. The recruitment period took 6 months from November 2021 to April 2022.
The eligible pregnant women at the antenatal care clinic were provided with the online Google Form questionnaire via QR code. They were assured of the confidentiality of their responses and that they would only be used for the study. Moreover, the responses to the questionnaire were anonymous and could not be traced back to the participants. There was no patient’s specific identity that could trace back to the participant on the Google Form. Only the researchers had password to enter to the answers of the Google Form questionnaire.
After agreeing to participate in the study, the participants were required to provide informed consent. Agreement was provided in written form of informed consent. They were not incentivized to participate in the study. Following this, they received the QR code to access and respond to the questionnaire. It took 20 min for participants to complete the questionnaire. The researchers provided the Google Form questionnaire and assured the confidentiality issues. It was not the same person. They did not take care of the participants.
The sample size was calculated based on the formula of single proportion (n = Zα/22 pq/d2) with p as an acceptance rate of 0.5 from the expected rate of acceptance. With an α of 0.05 and d = 0.05, the total number of participants needed for this research to have an adequately powered study was 500 after adding 30% of incomplete data.
The primary objective of this research was to study the acceptance of the COVID-19 vaccine in pregnant women. The secondary objectives were to study pregnant women’s knowledge of and attitudes toward the COVID-19 vaccine and the factors associated with their acceptance of the vaccine.
The questionnaire was divided into five major sections:
Demographic data: This section comprised of questions regarding the participants’ age, marital status, parity, gestational age, underlying disease, educational level, hometown, occupation, treatment rights, monthly income, past and current COVID-19 infection, family members or friends infected with or deceased from COVID-19, and main sources of information about the COVID-19 pandemic and vaccine.
Perception of the COVID-19 pandemic and vaccination concerns: This section comprised of questions about the government comments on the expected duration of the pandemic used to control it, trust in the government and healthcare personnel’s advice, belief in the reported number of COVID-19 cases, confidence in the effectiveness and safety of the vaccines and awareness of their complications, concern about a shortage and unequal distribution of vaccines, and preference for COVID-19 vaccines.
Knowledge, attitude, and practice related to COVID-19 infection during pregnancy: This section covered the participants’ knowledge of preventive measures when in contact with infected people, awareness of the guidelines on breastfeeding during COVID-19 infection, knowledge about the SARS-CoV-2 virus and the importance of social distancing, knowledge of good hygiene practices, and coping methods when possibly infected with COVID-19. This section consisted of a total of 21 questions, with five, seven, and nine questions allocated to the knowledge, attitude, and practice subsections, respectively. Participants received one point for each correct answer, and a total score of 80% or higher in each subsection was determined as the cut-off score for the participant’s great knowledge, attitude, and practice regarding COVID-19.
Knowledge, attitude, and acceptance regarding the COVID-19 vaccine: This section comprised of questions on the importance of vaccination, which individuals should be vaccinated, initial gestational age at which to begin vaccination, teratogenicity and adverse effects of the vaccine on pregnancy, trust in the benefits of the vaccine, the belief that COVID-19 vaccines can be equally distributed, the participants’ opinion on the cost of the COVID-19 vaccine, recommendations to friends or family to receive the vaccine, the decision to accept vaccination based on its effectiveness, the habit of receiving flu vaccines vaccination status, and finally, the specific type and number of shots of the vaccine taken. A total of 21 questions contributed to the scores, with seven, six, and eight in the knowledge, attitude, and acceptance subsections, respectively. The last two questions in the acceptance section, regarding the type and dose of the vaccine received, did not contribute to the total score. As in the previous section, each correct answer received one point. A total score of equal to or more than 80% was determined as the cut-off score for the participant’s great knowledge, attitude, and acceptance regarding the COVID-19 vaccine.
Factors influencing the acceptance or rejection of the COVID-19 vaccine: This last section of the questionnaire consisted of two questions regarding the main factors influencing the decision to either accept or reject the COVID-19 vaccine.
The researchers created the questionnaire and took 1 month to create it. All items included in this questionnaire were modified from previous study. 15 The English questionnaire was translated to Thai, retranslated back to English, and then compared to the first English version by different translator to ensure translation accuracy. Content validation was ensured by two obstetricians. Content validity index was 0.9. A pilot study was performed in 30 participants to assess the reliability of the questionnaire. The questionnaire had an internal consistency, with Cronbach’s alpha values of 0.743.
Statistical analysis was performed using the SPSS software version 22.0. Categorical variables were presented as frequencies and percentages, while continuous variables were presented as means and standard deviations. The independent t-test was used to compare continuous variables, whereas the Mann–Whitney U test was used to compare nonparametric variables. The chi-squared test and Fisher-exact test were used to compare categorical variables. Multivariate logistic regression analysis was used to evaluate the association between factors and the acceptance of the COVID-19 vaccine. The factors with a p value greater than 0.05 in the univariate analysis were entered into the multivariate logistic regression analysis. The odds ratio (OR) and 95% confidence interval (CI) were calculated. A p value of 0.05 was considered statistically significant.
Results
Demographic data
A total of 500 pregnant women were eligible for inclusion in the study and consented to complete the questionnaire. The demographic data of the study population and acceptance of the COVID-19 vaccine are shown in Table 1. The age distribution ranges of the participants were as follows: 20–34 years (68.2%), ⩾35 years (31.2%), and <20 years (0.6%). Nearly half of the participants (45.4%) were in the third trimester of pregnancy, most of them had no underlying disease (79.0%), and 11.8% had previously been infected with COVID-19. Most of the participants had a bachelor’s degree or higher (80.2%). The participants’ information about the COVID-19 pandemic and vaccine mainly came from more than one source, with the internet and social media being the leading ones.
Demographic data and acceptance to receive COVID-19 vaccine.

Perceptions of the COVID-19 pandemic and vaccination concerns
Table 2 shows the participants’ perceptions of the COVID-19 pandemic and vaccination concerns and their association with COVID-19 vaccine acceptance. Most of the participants (81%) had concerns about serious complications from the vaccine. For most of the participants (59.2%), the most preferred vaccine for future vaccination was the Pfizer-BioNTech vaccine.
Association between perception toward the COVID-19 pandemic and vaccination concerns and acceptance to receive COVID-19 vaccine.

Knowledge, attitude, and practice related to COVID-19 infection during pregnancy
Table 3 represents the association between pregnant women’s knowledge, attitudes, and practices related to COVID-19 infection during pregnancy and their acceptance to receive the COVID-19 vaccine. The table also depicts the scores received from each subsection and their association with the decision to accept the COVID-19 vaccine.
Association between knowledge, attitude, and practice toward COVID-19 infection in pregnant women with scores received from each section and acceptance to receive COVID-19 vaccine.
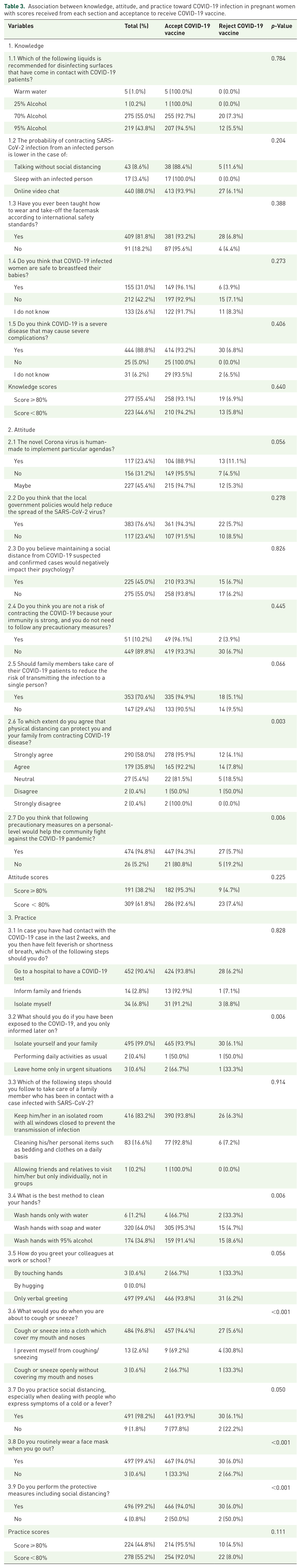
More than half of the participants had great knowledge about protecting themselves from COVID-19 patients, with 55.4% scoring 80% or higher.
In the attitude subsection of the questionnaire, 38.2% of the participants achieved a score of 80% or higher. However, in the practice subsection of the questionnaire, less than half of the pregnant women (44.8%) received a score of 80% or higher.
Knowledge, attitude, and acceptance regarding the COVID-19 vaccine
The association between the participants’ knowledge, attitude, and acceptance regarding the COVID-19 vaccine, with the scores received from each section, and the decision to receive the COVID-19 vaccine is shown in Table 4. Most of the participants (67.4%) had great knowledge about COVID-19 vaccination, with a score of 80% or higher. More than half (78.8%) of the participants thought that the COVID-19 vaccine could not cause potentially adverse effects on their fetuses and newborns, and 73.0% perceived that the vaccine have no adverse effects on their pregnancy.
Association between knowledge, attitude, and acceptance of COVID-19 vaccine in pregnant women with scores received from each section and decision to receive COVID-19 vaccine.
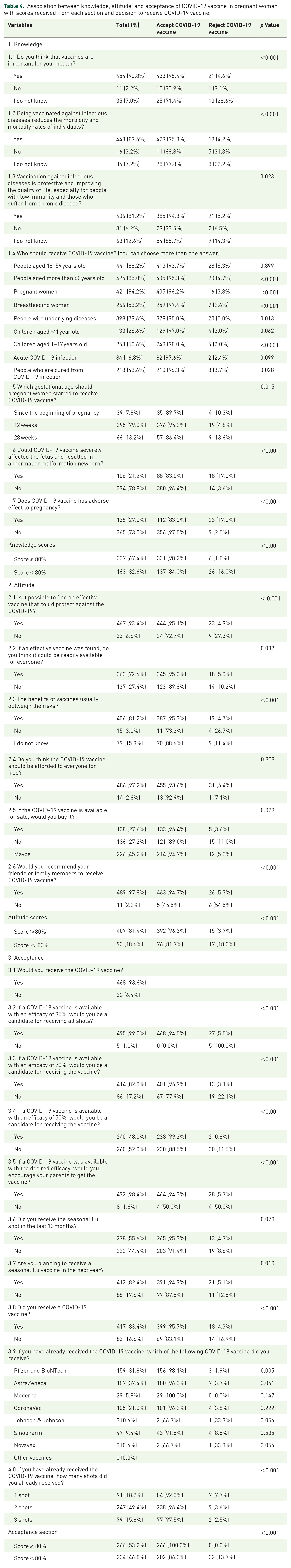
A great attitude toward the COVID-19 vaccine was demonstrated by 81.4% of the participants, with a score of 80% or higher. The vaccine acceptance rates were significantly higher in the groups with great knowledge and great attitudes, at 98.2% and 96.3%, respectively. Regarding the acceptance of the COVID-19 vaccine during pregnancy, 53.2% of the participants had a score of 80% or higher, and 93.6% expressed their choice to receive the vaccine. When stratified by efficacy, it was observed that a vaccine with an efficacy rate of 95% gained acceptance from 99.0% of the participants, while a vaccine with an efficacy rate of 70% rate gained acceptance from 82.8%. In contrast, vaccines with an efficacy rate of 50% gained acceptance from only 48.0% of the participants.
Factors influencing COVID-19 vaccine acceptance or rejection
Table 5 reveals the association between the factors leading to pregnant women’s acceptance or rejection of the COVID-19 vaccine and the decision to receive the vaccine. The most common factor of the participant’s acceptance of the vaccine was trust in the safety of the vaccine (36.2%). As for the participants’ rejection of the vaccine, almost half of the participants (49.2%) did not want to put their fetuses at risk from its possible adverse effects.
Association between factors leading to acceptance or rejection of COVID-19 vaccine in pregnant women and decision to receive COVID-19 vaccine.

The factors associated with vaccine acceptance and rejection from the univariate analysis are shown in Table 6. The participants who relied on the Thailand Department of Disease Control or National Center for Disease Control as their primary sources of information, took precautionary measures on a personal level, routinely wore face masks, implemented social distancing, and had great scores in the knowledge, attitude, and acceptance sections of the questionnaire were more likely to accept the vaccine. The negative factor that led to the participants’ rejection of the vaccine was previous COVID-19 infection.
Univariate analysis of factors associated with COVID-19 vaccine acceptance in pregnant women.

A multivariate logistic regression analysis for the association of factors leading to pregnant women’s acceptance of the COVID-19 vaccine found that the participants with great knowledge of the COVID-19 vaccine were more likely to accept it (OR 13.25, 95% CI 2.45–71.61, p = 0.003), whereas those who had previously been infected with COVID-19 were less likely to do so (OR 0.11, 95% CI 0.02–0.62, p = 0.013).
Discussion
Interpretation of results
This study demonstrated that 67.4% and 81.4% of pregnant women had great knowledge of and attitudes toward the COVID-19 vaccine, respectively, while 93.6% accepted to receive the vaccine. The factor associated with the decision to receive the vaccine was having great knowledge about it. However, the factor associated with the decision to reject the vaccine was recent COVID-19 infection. Vaccination is crucial for disease control, so persuading people to join the vaccination campaign will benefit our community great immensely. Not only will this limit the pandemic, but it will also reduce the severity of COVID-19 infection. It is important to promote the COVID-19 vaccination campaign to pregnant women by emphasizing that vaccination will not cause any negative consequences for the fetus or the pregnancy.
Comparison with existing literature
Our study demonstrated that 93.6% of pregnant women accepted to receive the COVID-19 vaccine. If the efficacy rate of the vaccine is greater than 95%, 99.0% of them would undergo vaccination. In the case of vaccines with efficacy rates of more than 70%, 82.8%, and more than 50%, 48.0% of them would undergo vaccination. In a Turkish study conducted with 300 pregnant women participants, only 37.0% accepted to receive the COVID-19 vaccine. 23 Furthermore, in a 2021 Libyan study conducted with 15,087 participants, 79.6% of the participants accepted to receive the vaccine if it had an efficacy rate of more than 90%, 60.6% accepted if the efficacy rate was above 70%, and 41.2% accepted if the efficacy rate was above 50%. 15 In California, Simmons et al. reported that the vaccine hesitancy rate in pregnant women was 57%. 18 They found that younger age, living in a less urban environment, and essential worker status were associated with vaccine hesitancy. 18 In Japan, Saitoh et al. reported that the vaccine hesitancy rate in pregnant women was 40.7%. They found that women with vaccine hesitancy were more likely to fear adverse reactions, safety, and efficacy compared to those without vaccine hesitancy. 19 In Ohio, Germann et al. performed a study in pregnant and postpartum women. They found 40% COVID-19 vaccine hesitancy upon enrolment, and 52% hesitancy of the vaccine in subsequent vaccination at follow-up. Those who identified as non-Hispanic black, were Medicaid beneficiaries, and were still pregnant at follow-up were less likely to be vaccinated. 24 In Italy, Miraglia del Giudice et al. found 86.4% highly hesitant in pregnant women. Respondents who did not get a graduate degree, those less concerned that they could be infected by SARS-CoV-2, and those trusting mass media/internet/social networks for information were highly vaccine hesitant. 25 In contrast to these previous studies, our study reported a higher acceptance rate. This may be attributed to the increasing promotion of vaccination campaigns and the assurance of vaccine safety.
Regarding the percentage of knowledge, attitude, and acceptance of COVID-19 vaccination, the percentages of pregnant women with scores above 80% in the knowledge, attitude, and acceptance subsections of our questionnaire were 67.4%, 81.4%, and 53.2%, respectively. In the 2021 Libyan study, the mean ± SD scores respondents received for the knowledge, attitude, and acceptance toward COVID-19 vaccine subsections were 2.35 ± 0.9 (ranging from 0 to 3), 3.2 ± 0.9 (ranging from 0 to 6), and 3.28 ± 1.7 (ranging from 0 to 6), respectively. 15
Compared to the Turkish study (100%) and the Libyan study (68.1%), almost all the participants in our study wore facial masks (99.4%).15,23 The percentage of pregnant women complying with social distancing was 99.2%. In comparison, the percentage of people implementing social distancing in the Turkish and Libyan studies was 100% and 77.3%, respectively. 23 This study and the Turkish study were both conducted on pregnant women; these women may implement protective measures more than ordinary individuals in order to protect their fetuses.
The main factors that influenced the participants’ decision to accept vaccination were trust in the safety of the vaccine (36.2%), belief in the effectiveness of herd immunity (21.4%), and belief in the effectiveness of the vaccine (17.2%). The negative factors that influenced the participants’ decision to reject vaccination were their concern about the adverse effects of the vaccine on their fetuses (49.2%), the need for more concrete data about the side effects and effectiveness of the vaccine (23.8%), and their concern about the side effects of the vaccine on themselves (11.2%).
After analyzing the results using the multivariate logistic regression method, we identified a significant association between the acceptance rate of the COVID-19 vaccine and having great knowledge about the vaccine, determined by a score of 80% or more (adjusted OR of 13.25, 2.45–71.61). Additionally, people who experienced prior COVID-19 infection had a lower tendency to receive another vaccine shot (adjusted OR 0.11, 0.02–0.62), probably because they assumed that they had an adequately protective immune system post-infection. Compared to the 2021 Turkish study, this study found a positive correlation between COVID-19 vaccine acceptance and the number of school-age children in the house. 23
Implications for practice
From this study, pregnant women who already have great knowledge about the COVID-19 vaccine are more likely to get the vaccine during pregnancy, while those who have recently been infected with COVID-19 will lose interest in getting the vaccine. Therefore, healthcare providers should educate pregnant women about the COVID-19 vaccine in order to prevent COVID-19 infection.
Strengths and limitations
The strength of this study is that the study was performed in a high-reputation university hospital for quality of care for pregnant women and/or pregnancy-related conditions. This study also had a larger sample of pregnant women than other studies. The limitation of this study was that it was a cross-sectional study. This study may have bias from recall bias and convenient sampling. Another limitation was that the facilitating questionnaire completion at a Department of Obstetrics and Gynecology where the benefits of COVID-19 vaccination are promoted and recommended was peculiar. The results generated here are likely hampered by social desirability on respondents’ parts, providing responses that they feel the researchers could want to hear. This may also help to explain the high COVID-19 vaccine acceptance rates routinely noted throughout the article. In the future, further studies should be conducted on breastfeeding women, as our present study results illustrate that people still believe that the COVID-19 vaccine is not safe for breastfeeding mothers.
In conclusion, Thai pregnant women who already have great knowledge about the COVID-19 vaccine tend to receive it during pregnancy, whereas those recently infected with COVID-19 lose interest in receiving the vaccine. Health authorities should educate people with a history of COVID-19 on the need to get a booster shot of the vaccine, as most of our study population still believes that it is not necessary; such an attitude is a major obstacle to disease control.
