Abstract
Background:
Clear and trustworthy information is crucial to improving public acceptance of COVID-19 vaccinations, especially among pregnant women. Given the increased risk of severe viral pneumonia in pregnant women, it is critical to foster confidence in the vaccine’s safety and understand any potential adverse events (AEs). So, we did this study in Jeddah, Saudi Arabia (SA), from March to May 2022 to compare women who did not get any doses of the COVID-19 vaccine during pregnancy (Group A) to women who did get at least one dose during pregnancy (Group B) regarding (1) the frequency, types, AEs, and management of its AEs of the COVID-19 vaccination; and (2) exploring pregnancy, delivery, and fetus-related complications (e.g., miscarriage, birth defects, and preterm labor)
Methods:
A cross-sectional study targeted 438 women who gave birth or were pregnant within the previous 8 weeks. Data was collected through face-to-face interviews with skilled nurses in 13 randomly selected primary healthcare facilities, using a validated, well-structured questionnaire that included the Centers for Disease Control (CDC) COVID-19 vaccination-related AEs. We analyzed the collected data using SPSS version 27.
Results:
Most participants were aged 25 to less than 35 (58.8%), and 287 (61.3%) were university graduates. There was no statistically significant difference among the studied groups regarding demographics. However, women in Group B had a significantly higher rate of abortions, oligohydramnios (24.4%), abnormal placentas (size and location), 103 (42.7%) abnormal fetal growth, 122 (53.7%) problems breastfeeding, blood pressure problems, and more cases of malaise, headaches, chest pain, breathing problems, and sleep problems than women in Group A. After the second and third doses, the confirmed post-vaccination COVID-19 rates in Group B were lower than those in Group A.
Conclusion:
The COVID-19 vaccine significantly reduces post-vaccination COVID-19. Although COVID-19 vaccine-related AES are prevalent, analgesics and antipyretics effectively treat most of them.
Plain language summary
Background:
Clear and trustworthy information is essential for increasing public acceptance of COVID-19 vaccinations, particularly among pregnant women who face higher risks of severe illness. This study, conducted from March to May 2022 in Jeddah, Saudi Arabia, aimed to explore issues related to COVID-19 vaccination in pregnant women. The study focused to compare those who did not receive any vaccination doses during pregnancy (Group A) and those who received at least one dose or more during pregnancy (Group B) regarding 1) the frequency, types, AEs, and management of the COVID-19 vaccination among moms; and 2) exploring pregnancy, delivery, and fetus-related complications (e.g., miscarriage, birth defects, preterm labor, etc.).
Methods:
We conducted the study on 438 women who had given birth within the past 8 weeks. Data were collected through face-to-face interviews with skilled nurses at 13 randomly selected primary healthcare facilities using a validated questionnaire.
Results:
In this study, it is observed:
• Most participants were aged 25 to less than 35 years old (58.8%), and 61.3% were university graduates.
• No significant demographic differences were found between groups.
• The study groups showed a statistically significant difference in the occurrence of spontaneous abortions, oligohydramnios, abnormal placentas (size and location), abnormal fetal growth, blood pressure problems, breastfeeding problems, malaise, headaches, chest pain, breathing problems, and sleep problems. However, most reported AEs required only analgesics and antipyretics.
• Post-vaccination COVID-19 rates were lower in vaccinated women after the second and third doses.
Conclusions:
The COVID-19 vaccine significantly reduces post-vaccination COVID-19. Although COVID-19 vaccine-related AES are prevalent, analgesics and antipyretics effectively treat most of them.
Introduction
The Royal College of Obstetrics and Gynecology strongly recommends COVID-19 vaccines during pregnancy, as pregnant women are more likely to get severe viral pneumonia than others. 1 Vaccination is the best way to protect against the known risks of a SARS-CoV-2 infection during pregnancy, which may increase the risk of various pregnancy problems, admission to intensive care, premature birth (before 37 weeks), or stillbirth compared to the infants of unvaccinated women. 2
On December 11, 2020, The Food and Drug Administration (FDA) approved the Pfizer-BioNTech vaccine for emergency use authorization (EUA) in individuals older than 16 years of age. In Saudi Arabia (SA), the SFDA approves vaccination use. The Specialized Scientific Committee recommended the COVID-19 vaccine in April 2021 for pregnant women. 3 On December 16, 2021, the Joint Committee on Vaccination and Immunization (JCVI) announced that pregnant women are now considered a “vulnerable” group within the COVID-19 vaccination program, emphasizing the urgency of their receiving COVID-19 vaccination and booster doses. 4 Less than one-third of pregnant women accepted the COVID-19 vaccination during pregnancy, and they experienced similar pregnancy outcomes as unvaccinated pregnant women. Younger women, non-white ethnicities, and people from lower socioeconomic backgrounds had lower uptake. 5
Despite its limitations, knowledge about the safety and effectiveness of the COVID-19 vaccination during pregnancy is expanding. Getting the COVID-19 vaccine appears to have more benefits than any known or potential risks during pregnancy. Pregnant women, women who are breastfeeding, trying to get pregnant now, or may become pregnant in the future should get the COVID-19 vaccine. 6 The Centers for Disease Control and Prevention (CDC)’s guidelines and early evidence, including those from the American College of Obstetricians and Gynecologists, the Society for Maternal-Fetal Medicine, the American Society for Reproductive Medicine, and many others that serve pregnant women, recommend Pfizer-Bio-NTech or Moderna (mRNA COVID-19 vaccines) to start the immunization series or get a booster dose. However, the J & Janssen COVID-19 vaccine may be considered in specific instances. 7
The COVID-19 mRNA vaccine is a safe option for pregnant women and their unborn fetuses, as it effectively reduces the severity of the SARS-CoV-2 infection and lowers the risk of their infants COVID-19. 7 The first COVID-19 vaccination showed quick antibody responses, produced antibodies while pregnant, and worked to create a humoral immune response in pregnant women. However, SARS-CoV-2 immunoglobulin G (IgG) levels were lower than those seen in vaccinated women who were not pregnant.7,8 Two vaccination doses are associated with more robust maternal and fetal antibody levels. Higher levels of fetal IgG antibodies and a lower proportion of antigens transmitted are associated with longer times between the first dose of immunization and birth. After booster vaccination, adaptive immunity becomes more significant and improves placental antigen transmission. 9
The most common COVID-19 vaccine adverse events (AEs) include fatigue, changes at the injection site, myalgia, headache, arthralgia, and, less frequently, allergy. Serious AEs are extremely rare and cause long-term health disorders such as thrombosis events, myocarditis, and pericarditis. 10 COVID-19 AEs usually manifest 6 weeks after the initial dose and fade in a few days in adults, so health institutions should continuously monitor, and report AEs associated with vaccines and medications to the relevant authorities for 8 weeks following the last dosage. 11
The CDC and the US FDA use the Vaccination Adverse Event Reporting System (VAERS) as a passive post-COVID-19 vaccination licensure surveillance system to collect, monitor, and assess reports of AEs. Even though adequate vaccine safety and efficacy are essential for their licensure, fast-tracking vaccine development processes compared to traditional vaccinations may increase the risk of AEs.11–19
Several studies worldwide have evaluated the side effects, effectiveness, and safety of COVID-19 vaccines in pregnancy. In November 2021, a review summarized the effects of COVID-19 vaccination in pregnancy, including the rate of SARS-CoV-2 infection, maternal antibody responsiveness, placenta antibody transmission, and AEs. There was no effect on gestation, childbirth, or perinatal mortality. Injection-site discomfort, tiredness, and migraines are the most prevalent side effects, but these are temporary. 9 The negative neonatal outcomes include The VAERS reported no neonatal deaths, the most common pregnancy-related AE, despite preterm delivery, small size for gestational age, and negative neonatal outcomes. 16
The computed percentage of unfavorable pregnancy and newborn outcomes in people who completed a pregnancy after receiving the COVID-19 vaccine was like the incidences reported in studies involving pregnant women conducted before the COVID-19 pandemic, albeit not exactly comparable. While the BNT162b2 vaccine at any stage of pregnancy does not exhibit any safety concerns based on its profile and short-term obstetric and neonatal outcomes, reporting of the COVID-19 vaccination in the preconceptions phase (within 30 days after the last menstrual period) or during pregnancy is mandatory. The v-Safe Vaccine Pregnancy Registry. If people who have signed up for v-safe say they were pregnant at the time of vaccination or afterward, the registry staff may call them to find out more. 17
To our knowledge, SA researchers have completed various studies on COVID-19 vaccinations and their AE effects, but they have always focused on the general population and have not conducted any research on pregnant women. There is a need for clear communication about vaccine safety among pregnant women and healthcare providers, as well as efforts to combat vaccine hesitancy. These efforts include post-vaccination surveillance to collect data, particularly after first-trimester vaccination and long-term newborn follow-up, as well as guide policymakers in making effective recommendations based on evidence-based, updated information. These efforts aim to enhance the general health condition of expectant mothers and their infants.
Clear and trustworthy information is crucial to improving public acceptance of COVID-19 vaccinations, building confidence in vaccine safety, and comprehending any possible AEs. More investigation on the impacts of the COVID-19 vaccine is recommended by the National Institutes of Health (NIH). Therefore, we conducted this cross-sectional study from March to May 2022 in Jeddah, SA (1) to investigate the frequency, types, and self-reported or clinically diagnosed AEs associated with the COVID-19 vaccine, including those that occurred post-vaccination (COVID-19); (2) exploring the clinically and self-reported antenatal, natal, and pregnancy outcomes and (3) comparing women who did not receive any doses of the COVID-19 vaccine during pregnancy (Group A) with those who received at least one dose of the COVID-19 vaccine (Group B).
Methods
Study design and participants
From March to May 2022, we conducted this cross-sectional study that included women who gave birth or were pregnant within the previous 2 months in SA, either Saudi or non-Saudi, and were more than 18 years old or less than 49 years old.
We excluded women with a history of hormonal or menstrual disturbances, chronic complicated diseases, or those taking medications prior to pregnancy; women who had a previous pregnancy with complications; and women with complicated medical, mental, or psychotic disorders, such as schizophrenia. These factors may complicate the determination of whether a pregnancy disturbance stems from the COVID-19 vaccine or preexisting health issues, interfere with participation, and potentially affect pregnancy outcomes. We also excluded Internet nonusers and illiterate women.
Sample size estimation and techniques
The Open Epi-Info program calculates the sample size. From the 1,368,958 Saudi women (18–49 years old) in Jeddah, where about 4500 babies are born every year, to the same months in 2020 in the second cluster, where 1763 babies are born alive, we assumed that 50% of pregnant women in Jeddah received vaccinations either during their pregnancy or afterward. 18 We calculated a sample size of 210 women, assuming a type I error of 0.05 and a type II error of 0.20 (a power of 0.80%) at a 95% confidence level. With a degree of freedom of 2 to represent the whole Jeddah region, the required sample was 420 women who had recently given birth or were pregnant within the previous 2 months.
We used the cluster-sampling method to collect a representative sample of the 100 primary health care centers in Jeddah. To do this cross-sectional study, we randomly selected 13 primary health care centers, especially the baby clinic or immunization clinic, in the Jeddah region of SA.
Data collection tools
After obtaining informed consent, we collected the data through a face-to-face interview with well-trained nurses using a well-structured and validated questionnaire (Cronbach’s alpha = 0.78). The data collection tool was prepared based on earlier studies.15,16,20–23 It comprises the following five sections:
● Sociodemographic and health-related factors include age, residence, nationality, educational level, occupation, and consanguinity. In addition, the history of COVID-19 pre-vaccination and comorbidities.
● Pregnancy-related issues current pregnancy planning, folic acid intake, germinal measles vaccines, vitamin and supplement intake, and antenatal clinical visits.
● Antenatal or pregnancy-related symptoms include delayed gestation, menstrual changes, abortion, or abortion threats. The symptoms of gestational diabetes include pre-eclampsia, hyperemesis gravid arum, severe hypersensitivity, mood swings, hyperpigmentation, recurrent infections, and weight changes.
● Delivery-related issues include premature membrane rupture and decreased amniotic fluid. Preterm labor (less than 37 weeks), difficult or prolonged labor, decreased amniotic fluid, and abnormal placenta size or location are some of the delivery issues. There were reports of postpartum hemorrhage, mood swings, puerperal sepsis, and embolism, among other complications.
● Fetus-related issues: normal fetal growth, normal vital signs, abnormal birth weight These conditions include intrauterine fetal death (IUFD), stillbirth, congenital anomalies, ICU admission, pneumonia, and neonatal COVID-19, Breastfeeding: Did the vaccination affect breastfeeding or not?
● Adverse event following COVID-19 immunization (AEFI) which is defined as is any adverse health result refers to any unfavorable or unexpected sign, aberrant laboratory result, symptom, or illness that arises after vaccination. That does not necessarily have a causal relationship with the vaccination usage. 22
Include the types of vaccinations, the number of doses administered during pregnancy, and COVID-19-related AEs. We classified vaccine AE according to CDC updates on January 12, 2022, either no AEs or the other 30 symptoms. 15 And utilized the adverse event of special interest (AESI) list obtained from the Safety Platform for Emergency Vaccine (SPEAC) Project work at Brighton Collaboration. This project’s AESI list has established three categories for AESIs19,20:
✓ General symptoms may include malaise, fatigue, bone pain, body pain, hypersensitivity, headaches, a runny nose, visual disturbance, hair loss or whitening, skin disorders, loss of taste or smell, auditory changes, sleep disturbance, and water retention.
✓ Laboratory results: include thrombocytopenia, abnormal liver function, abnormal kidney function, anemia, and post-vaccination COVID-19. 21
✓ Self-reported clinically diagnosed AEs include thrombosis, convulsions, cardiac-related AEs, meningitis, neurological symptoms, changes in blood pressure, and lymphadenopathy.
Statistical analysis
We coded and analyzed the collected data using the SPSS program version 27.0. We expressed the categorical variables as frequency (F) and percentage (%) and used the chi-square test (χ2) for analysis between the two studied groups, Group A, which are the pregnant women who didn’t receive any vaccination dose during pregnancy, and Group B, which is the vaccinated group when pregnant women received at least one dose or more during pregnancy. We showed quantitative variables as mean and standard deviation (SD). First, we used the Kolmogorov–Smirnov test to see if the continuous data was skewed or not normally distributed. A value of p < 0.05 was used to determine statistical significance.
Results
Sociodemographic and health-related factors and their differences among the studied groups
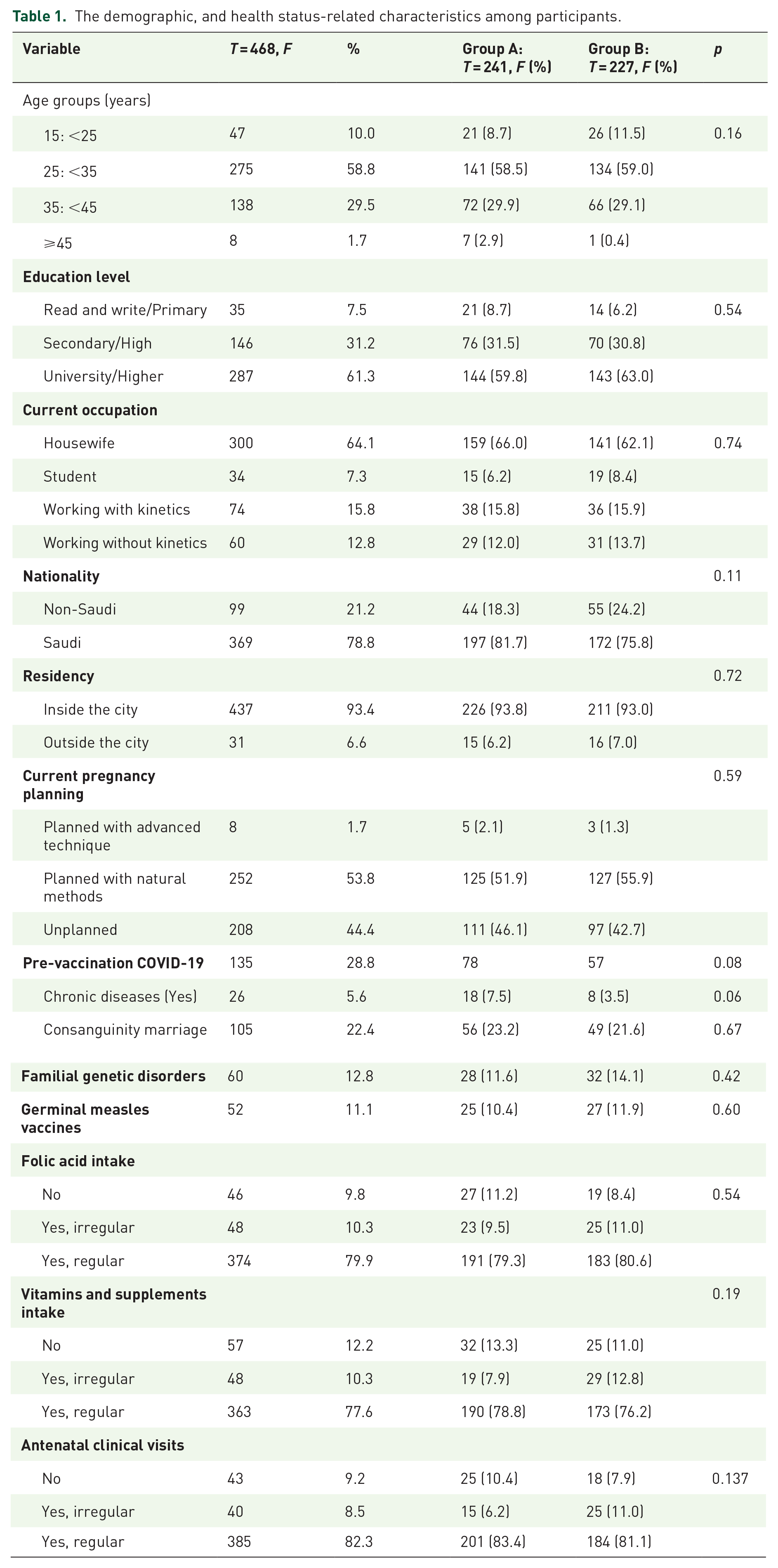
Most of the participants were aged 25 to less than 35 (58.8%), 287 (61.3%) were university graduates, 300 were housewives (64.1%), 369 were Saudi citizens (78.8%), and 437 were residents of the city (93.4%). They regularly attended antenatal care visits, took folic acid, and consumed vitamins and minerals. There was no statistically significant difference (p > 0.05) among the studied groups regarding demographic and health status-related characteristics (Table 1).
The demographic, and health status-related characteristics among participants.
Preconception, and antenatal pregnancy symptoms and their differences among the studied groups
The most reported preconception symptoms among the recruited women were menstrual changes (82, 17.5%), history of spontaneous abortion or miscarriage (48, 10.3%), and antenatal and pregnancy symptoms: mood swings (69, 14.7%), significant weight changes (63, 13.5%), recurrent infections (42, 9.0%), and hyperemesis gravidarum (36, 7.7%).
Among the studied groups, we found that Group B (which received at least one COVID-19 vaccine dose during pregnancy) was significantly (p < 0.05) associated with higher frequencies of spontaneous abortion (miscarriage) than Group A. Severe hypersensitivity compared to pregnant women in Group A who did not receive any COVID-19 vaccination dose during pregnancy, the rate of severe hypersensitivity was 10.4% (4.4%; Table 2 and Figure 1(a)).
Preconception, and antenatal symptoms among the studied participants.
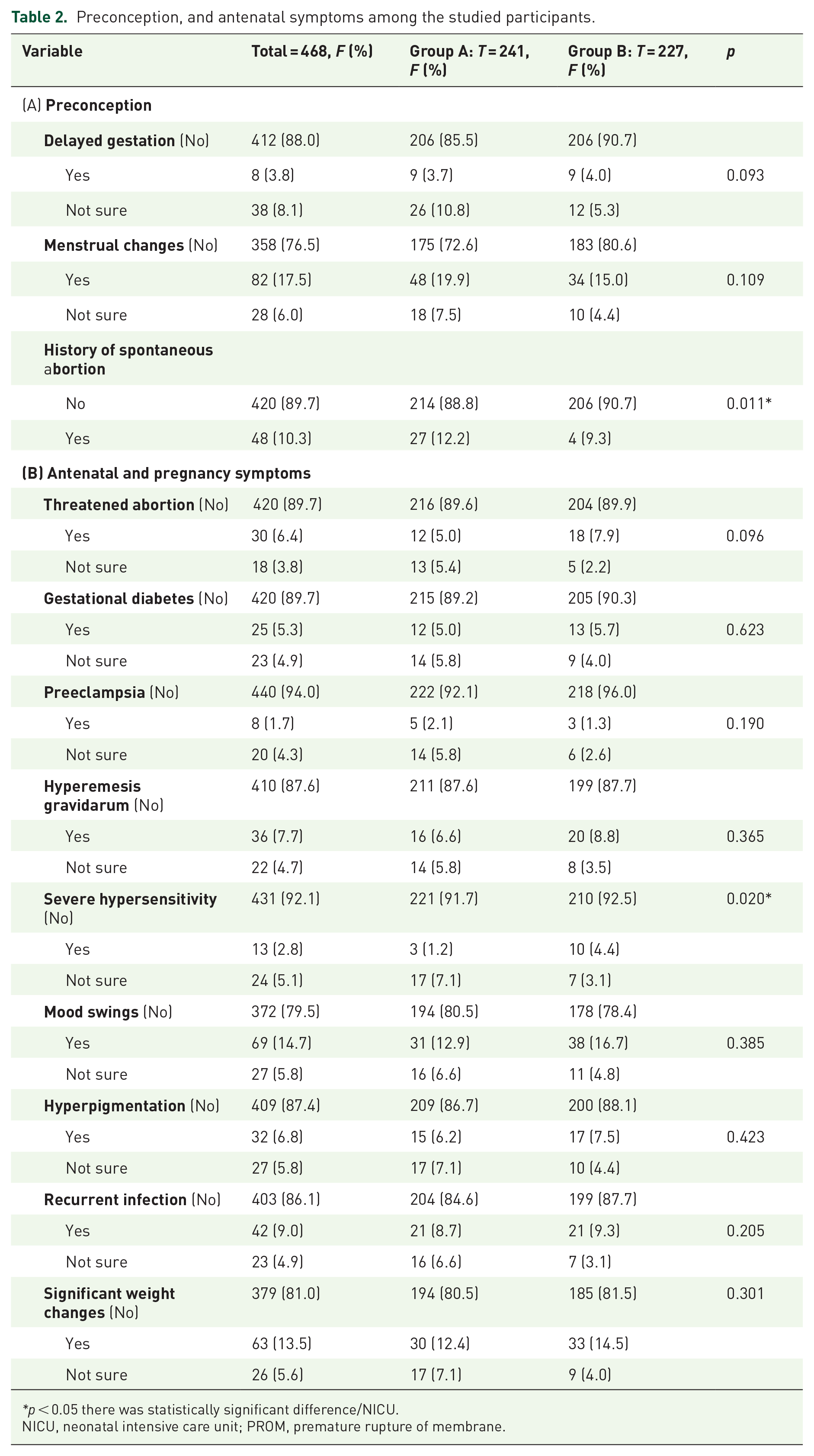
p < 0.05 there was statistically significant difference/NICU.
NICU, neonatal intensive care unit; PROM, premature rupture of membrane.

(a) Antenatal pregnancy symptoms and differences between the studied groups. (b) Delivery-related issues and their differences among the studied groups.
Delivery- and fetus-related issues and their differences among the studied groups
Only 124 (26.5%) of the women who were recruited had a cesarean section. Another 120 (25.0%) had an abnormal placenta (size and location), 87 (18.6%) had oligohydramnios, 77 (16.5%) had mood swings after giving birth, and 34 (7.3%) were in labor before they were due (Figure 1(b)).
Among the studied groups, we found that the COVID-19 vaccine during pregnancy was significantly (p < 0.05) associated with higher frequencies of 55 oligohydramnios (24.4%) and 70 (30.8%) having abnormal placentas (size and site) compared to pregnant women who didn’t receive any COVID-19 vaccination dose during pregnancy: 32 (13.3%) and 50 (20.7%), respectively (Table 3).
Delivery and pregnancy outcome among the studied groups.

p < 0.05 there was statistically significant difference/NICU.
IFUD, intrauterine fetal death; NICU, neonatal intensive care unit.
Most pregnancy outcomes were: 272 (58.1%) normal fetus growth, 219 (46.8%) normal vital signs, 241 (51.5%) normal birth weight, 37 (7.9%) admissions to the neonatal intensive care unit (NICU), 16 (3.4%) of participants had pneumonia, as well as neonatal COVID-19, IUFD 17 (17.3%), and stillbirth 13 (2.8%; Figure 2). There were statistically significant differences among the studied groups regarding abnormal fetal growth and the frequency of impaired breast feeding to be significantly (p < 0.05) higher in Groups B 103 (42.7%) and 122 (53.7%) compared to Groups A 74 (32.6%) and 101 (41.9%), respectively.

Fetus-related issues and their differences among the studied groups.
The COVID-19 vaccine-related issues and their differences among the studied groups
The vaccination history among the studied groups.

p < 0.05 there was statistically significant difference
Boldfaced values signify the highest or the lowest frequency.
AE, adverse event.
Self-reported general AEs (symptoms) among the studied groups following the COVID-19 vaccine.

p < 0.05 there was statistically significant difference.
AE, adverse event.
COVID-19 vaccination-related laboratory changes or confirmed COVID-19 among participants.

p < 0.05 there was statistically significant difference.
Clinically diagnosed COVID-19 related AEs among participants.

p < 0.05 there was statistically significant difference.
Boldfaced values shows signify the highest or the lowest frequency.
AE, adverse event.
Management of COVID-19-related AEs of different types and different doses of vaccines.

AE, adverse event.
The most common self-reported general AEs (symptoms) among the studied groups following the COVID-19 vaccine were malaise, body ache, bony ache, headache, running nose, smell or hearing impairments, skin disorders, and hair whitening and falling. Group B had significantly higher frequencies of malaise, bony ache, headache, chest pain, breathing difficulties, and sleep disturbances than group (Table 5).
Regarding the COVID-19 vaccination, there are AEs related to laboratory changes or post-vaccination COVID-19. The most reported laboratory changes associated with COVID-19 were anemia in 64/349 vaccinated women, followed by thrombocytopenia in 25/349, and liver function disturbance in 22/349. In terms of laboratory-confirmed post-vaccination COVID-19, it was highest after the second dose, 79 (25.6%), the first dose only, 73 (20.9%), and the third dose, 26 (8.3%). The confirmed post-vaccination COVID-19 rates in Group B were lower than those in Group A after the second dose (23.4%) and the booster dose (12.21%), compared to 32 (31.4%) and 14 (24.1%), respectively (Table 6).
The most reported clinically diagnosed COVID-19-related AEs were: 29/349 lymphadenopathy as well as neurological or psychological impairment; 28/349 convulsion as well as blood pressure disturbance; 24/349 carditis; and 22/349 immunity disorders (rapid myopathy) as well as thrombosis. There was a statistically significant relationship (p < 0.05) between the studied groups as regards the blood pressure disturbance that was higher in Group A and the neurological or psychological impairment that was higher in Group B (16/227) compared to Group A (Table 7).
Regarding the management of COVID-19 vaccination-related AEs, after the first dose of different types and doses of vaccines, most of the participants (131/62.7%) needed painkillers and fever reducers from Pfizer, 34/58.6% from AstraZeneca, and 4/40 from Moderna. Only one case (10 after Moderna) required hospital admission, while 11 cases (4/251 after Pfizer, 2/70 after AstraZeneca, and 1/10 after Moderna) required medical attention.
After the second dose, most participants required analgesics and antipyretics; medical consultation was required in 11 cases (5/209 after Pfizer, 4/58 after AstraZeneca, and 1/23 after Moderna); and hospital admission was required in only one case (58 after AstraZeneca). After the boss dose, most participants required analgesics and antipyretics; medical consultation was required in 5 cases (1/23 after Moderna and 4/105 after Pfizer), and no one required hospital admission (Table 8).
Discussion
During pandemics and emerging pathogen outbreaks, pregnant women were among the most vulnerable groups, and their disproportionate impact appears to have influenced the inclusion of pregnant women’s concerns in the development and implementation of vaccinations against emerging diseases. In these cases, the public’s concerns about the safety of the new vaccines and rumors about unfavorable events that may occur during the present and future pandemics are recurring elements. 22 We conducted this study to investigate the COVID-19 vaccine AEs among pregnant women, revealing a significant reduction in the risk of post-vaccination COVID-19 and a significant increase in the frequency of symptoms such as nausea, headaches, chest pain, breathing issues, and sleep disturbances compared to Group B.
From March to May 2022, the overall frequency of COVID-19-vaccinated mothers was 349/468 (74.5%). In Group A (mothers who did not receive any vaccination dose during pregnancy), 119 (49.9%) were unvaccinated at all. According to a poll in 16 countries, pregnant mothers were less likely to accept vaccinations for themselves. 23 This varies greatly depending on the current pandemic status (incidence of community infections), and other political, cultural, ecological, healthcare system, historical, and economic aspects that influence a person’s decision to accept vaccination. 24 Moreover, the Protection Motivation Theory posits that an individual’s motivation significantly influences their perceptions of vaccination (misinformation, the source, supply, cost, administration method, schedule of vaccination, and new vaccine introduction), and their general health status. 25 However, for both expectant mothers and clinicians, the safety of the COVID-19 vaccination and trust in the vaccine’s efficacy are the primary concerns and significant predictors of less vaccination uptake. 26 Vaccine hesitancy during pregnancy was understandable when COVID-19 vaccines were first authorized. During its initial trials, the authorities excluded pregnant females due to the risk of complications. 27 Trial participants’ inadvertent exposures early in pregnancy limited the initial data on pregnancy or birth outcomes after maternal COVID-19 vaccination.
Preconception symptoms: Menstrual changes (MCs) (82, 17.5%), which was relatively lower than what was reported in a multinational study including SA October 2021, 48 (57.1%) after more than one type of vaccine. The occurrence of MCs has been observed after the administration of both mRNA and adenovirus vectored COVID-19 vaccines. It was more likely due to the vaccination-triggered immune response than a specific element of the vaccine. 10 This can also explain the reported eight (3.8%) of delayed gestations.
Antenatal symptoms: Our analyses show a significant association between abortions and COVID-19 vaccines during pregnancy. We reported higher abortions (7.9%) in the vaccinated group compared to the unvaccinated group (5.0%), with a p-value of 0.011. In nearly identical results, 7.8% of women received one or more BNT162b2 (Pfizer-BioNTech) vaccines. Contrary to observational studies from Norway and the United States (US), mRNA COVID-19 vaccines administered during pregnancy were not associated with an increased risk for spontaneous abortions. 27
Regarding delivery-related issues: The analyses of our data showed nonsignificant differences among the studied groups as regards preterm labor, difficult labor, cesarean sections, postpartum hemorrhage, postpartum mood swings, puerperal sepsis, postpartum clots, and other complications. Observational studies from Norway and the US support our findings.28,29 In Iran, preterm labor was more than twice as high for women with COVID-19. It was alarming, as prematurity is the primary cause of mortality in children under 5 years old and carries a high morbidity rate. 30 The pooled analysis of a systematic review also shows that pregnant women who received vaccinations had a lower likelihood of non-reassuring fetal monitoring, lower gestational age at delivery, and a lower probability of premature delivery. 31
We reported marginally elevated incidences of IUFD (3.3%) in the unvaccinated group than in the vaccinated group (0.5%). Studies investigating the association of COVID-19 with IUFD have proved that it increases the IUFD ratio. Most likely, this is due to vertical transmissions. 32 According to a study in Paris, post-mortem analysis accounted for over half of the IUFD during the peak of the SARS-CoV-2 pandemic. Among them, 2.8% of the cases had a particular pattern of histological involvement associated with a placental SARS-CoV-2 infection. 33 A retrospective analysis showed that the COVID-19 vaccination did not lead to IUFD. 34 Thus, the COVID-19 vaccine is safe to receive while pregnant, but the SARS-CoV-2 infection puts expectant mothers and their unborn babies at serious risk. 35
Moreover, the percentage of abnormal birth weights in the immunized group was higher (49.3%) than in the non-vaccinated group (39.0%) in our study. A comprehensive original study from Egypt also agrees with our findings, as vaccinated pregnant females delivered a child with a slightly higher birth weight. 36
Regarding pregnancy outcomes: The frequency of congenital anomalies, stillbirth, NICU admission, pneumonia, and neonatal COVID-19 remained similar between the two groups in our data. Furthermore, in other studies, there was no proof that the COVID-19 vaccination during pregnancy increased the risk of unfavorable neonatal outcomes, such as NICU admission, preterm birth with a gestation of less than 34 weeks, low birth weight, extremely low birth weight, congenital abnormalities, and so forth.29,37,38
Regarding the effect of COVID-19 vaccinations on breast feeding: We observed that there was a decline in breastfeeding rates among unvaccinated (41.9%) mothers compared to vaccinated (53.7%) mothers. This shows that COVID-19 vaccinations did not affect milk production. This decline in breastfeeding could be due to exposure to formula milk by non-breastfeeding mothers. During COVID-19 in Pakistan, a study revealed that mothers who had previously breastfed their babies demonstrated higher breastfeeding rates compared to those who were familiar with formula milk or mixed feeding practices. 38
Regarding the types of vaccination: The Pfizer vaccine was multinational, followed by AstraZeneca, followed by Moderna, in agreement with another multination study. 21
In terms of laboratory-confirmed post-vaccination COVID-19: We found that it was highest after the second dose, 79 (25.6%), the first dose only, 73 (20.9%), and the third dose, 26 (8.3%). The confirmed post-vaccination COVID-19 rates in Group B were lower than those in Group A after the second dose (23.4%) and the booster dose (12.21%), compared to 32 (31.4%) and 14 (24.1%), respectively. The existence of the delta variation, which the WHO designated as a Variant of Concern on May 11, 2021, and other alarming variants during the data collection period in SA made evaluating the rates of SARS-CoV-2 infection after vaccination, challenging, especially considering the potential diminishing protection from earlier vaccinations. The gap in time between the first and second dosages is typically short, whereas the duration between the second and third doses can vary significantly among participants.21,29
The management of AEs differed among doses and vaccine types: This shows that the AEs following vaccinations may depend on the type of vaccine and the dosage administered. Most of our respondents received analgesics, antipyretics, and home rest. Medical consultations were less frequent. According to a review article, the COVID-19 vaccines are primarily safe during pregnancy; the most reported AEs were fever and pain on the injection site. Pregnant women had similar AEs to nonpregnant females. 39 A few individuals exhibited hypersensitivity reactions to COVID-19 vaccines. These reactions are rare and can occur with many other vaccines or medications, too.
Finally, compared to pregnant women in Group A, Group B who received at least one COVID-19 vaccine dose during their pregnancy had significantly lower rates of post-vaccination COVID-19 and its severity. This is the primary rationale behind the CDC’s recommendation to receive the COVID-19 vaccine during pregnancy.
This can be explained by a multinational study published in April 2022, and another meta-analysis of more than 430,000 pregnant women revealed that COVID-19 during pregnancy doubled the risk of stillbirth, raised the risk of preeclampsia and preterm birth, and elevated the risk of ICU admission and mortality among pregnant women with COVID-19 in comparison to pregnant women who are not infected. Furthermore, COVID-19-associated pregnancies increased the likelihood of severe problems for neonates. The findings should prompt pregnant women and clinicians to strictly implement all recommended COVID-19 preventive measures and conduct thorough assessments. 40
Implications for practice and future research
There is not enough international data to make definitive claims given the few studies on the COVID-19 vaccine AEs particularly during pregnancy, so further prospective research is highly recommended. It is critical to implement a comprehensive and continuous safety assessment across the country, specifically to monitor and understand the incidence of uncommon side effects associated with vaccines. Additionally, it is critical to ensure that necessary services are accessible and available at vaccination locations. Depending on the findings of our study, program managers must be prepared to handle these problems by implementing proper vaccine safety surveillance and communication. Assessment of the potential risks of vaccination during pregnancy requires careful consideration of the timing of vaccination relative to gestational exposure windows. We recommend reporting any health issues that arise after immunization to VAERS. Healthcare practitioners can consult clinical considerations for additional guidance and suggestions.
Strengths and limitations
This study has many strengths, as we examine the COVID-19 vaccines administered to pregnant women using the AESI list from the SPEAC Project at Brighton Collaboration. This project’s AESI list has established three categories for AESIs. We used a relatively large sample size of more than 400 mothers through face-to-face interviews with their mothers and child health passports at their routine visits to the PHCCs within 8 weeks after their birth by well-trained nurses. We selected a representative sample from 13 randomly selected PHCCs in Jeddah. Thus, our study population represents the entire population.
However, there are many limitations, including the possibility of bias and confounding variables due to the observational nature of the study. The exclusion of women who took medication prior to pregnancy could potentially bias the sample, while the inclusion of women with better overall health could have led to lower rates of AEs in the vaccinated group. However, this study captures the existing data for future research. However, this study captures the existing data for future research. A further limitation is a lack of assessment of the health condition of participants before receiving the COVID-19 vaccine, which makes it difficult to distinguish between preexisting health problems and those caused by the vaccine. The timing of the vaccination was not thoroughly explored due to multiple vaccination doses and different vaccination AEs. Furthermore, the stress of the pandemic, particularly about breastfeeding concerns, was beyond our control.
Conclusion
Our study revealed a relatively low frequency of COVID-19 vaccination uptake during pregnancy. The Pfizer vaccine was the most used, followed by AstraZeneca, followed by Moderna. Pregnant women in Group A who did not receive any COVID-19 vaccination dose during pregnancy showed statistically significant higher frequencies of confirmed SARS-CoV-2 compared to pregnant women in Group B.
The study groups showed a statistically significant difference in the occurrence of spontaneous abortions, oligohydramnios, abnormal placentas (size and location), abnormal fetal growth, blood pressure problems, breastfeeding problems, malaise, headaches, chest pain, breathing problems, and sleep problems. However, most reported AEs required only analgesics and antipyretics.
Footnotes
Acknowledgements
The authors would like to express their special thanks to all the recruited participants in this study.
Declarations
List of Abbreviations
AEs adverse events
CDC Centers for Disease Control and Prevention
EUA Emergency use authorization
FDA Food and Drug Agency
IUFD intrauterine fetal death
JCVI The Joint Committee on Vaccination and Immunization
MCs menstrual changes
NICU neonatal intensive care unit
NIH The National Institutes of Health
US United States
