Abstract
Background:
Vaccination is a fundamental tenet of public and population health. Several barriers to vaccine uptake exist, exacerbated post-COVID-19, including misconceptions about vaccine efficacy and safety, vaccine hesitancy, vaccine inequity, costs, religious beliefs, and insufficient education and guidance for healthcare professionals. Vaccine uptake may be aided using microarray patches (MAPs) due to reduced pain, no hypodermic needle, enhanced thermostability, and potential for self and lay administration.
Objectives:
This protocol outlines the development of a scale that aims to accurately measure the perceived safety, usability, and acceptability of MAPs for vaccination among laypeople, MAP recipients, clinicians, and parents or guardians of children.
Methods and analysis:
This study will follow three phases of scale development and validation, including (1) item development, (2) scale development, and (3) scale evaluation. Inductive (interviews) and deductive methods (literature searches) will be used to develop scale items, which experts from target populations will assess through an online survey. Cognitive interviews will be conducted to observe their processes of answering the draft survey. Thematic analysis will be conducted to analyse qualitative data. Lastly, four surveys will be administered online to our target populations over two time points to determine their repeatability. Exploratory and confirmatory factor analyses, Cronbach’s alpha, and construct validity will be performed.
Ethics:
This study was approved by Metro South Health (HREC/2021/QMS/81653) and Western Sydney Local Health District (2023/ETH00705) Human Research Ethics Committees.
Discussion:
The scale will support a standardised approach to assessing the social and behavioural aspects of MAP vaccines, enabling comparison of outcomes across studies. Once validated, this scale will assist vaccination programmes in developing effective strategies for integrating MAPs and overcoming barriers to vaccination. This includes improving vaccine equity and accessibility, especially in lower- and middle-income countries and rural or remote locations.
Introduction
Vaccination is a fundamental tenet of public and population health. There are several well-established barriers to vaccine uptake, such as concerns and misconceptions about vaccine efficacy and safety, vaccine hesitancy, vaccine inequity, costs, religious beliefs, and insufficient education and guidance for healthcare professionals.1–3 Additional factors have arisen post-COVID-19, including growing vaccine fatigue and hesitancy, costs, community cohesion, trust in vaccine providers, communication about booster vaccines, and legal requirements or mandates. 4 The COVID-19 pandemic saw a large decline in routine childhood vaccination, with 25 million children missing out on vital immunisations in 2021, such as measles, polio, diphtheria, tetanus, and pertussis. 5 Adult vaccination rates are also suboptimal and require addressing. 6
Several studies have identified microarray patches (MAPs) as the preferred delivery option compared to needles and syringes in healthy adults, thus increasing willingness to be vaccinated.7–11 However, MAP vaccines are currently only in clinical development, and no MAP vaccine is available for use. MAPs are medical devices containing several thousand micro-projections, invisible to the human eye, designed to penetrate the stratum corneum and deliver vaccines to the epidermis and upper dermis. 12 MAPs deliver vaccine into dermal dendritic cells evoking a strong immune response. 13 Several types of MAPs are designed to effectively deliver vaccines to the skin; these include coated MAPs, biodegradable or dissolving MAPs, solid MAPs, hollow MAPs, hydrogel-forming MAPs, porous MAPs, and hybrid MAPs.14,15 MAP technology has several benefits, including the absence of sharps, reduced need for cold-chain management, the potential for self-administration or trained user administration, and reduced clinical waste.16–20
Safety, usability, and acceptability are important social and behavioural outcomes to consider in achieving success in vaccination programmes. First, safety refers to eliminating or reducing potential hazards a product may present when in use (i.e. adverse events following immunisation). 21 Second, usability is described as the extent to which a product can achieve its desired goal by users. 22 Finally, acceptability refers to the receiver of a product by users (i.e. willingness to be vaccinated) and the requirements of an organisation.23,24 MAPs may overcome barriers to vaccination, such as needle-related anxiety, accessibility, cost, and time, in ways that are safe, usable, and acceptable. 25 Moreover, MAPs could reduce other barriers to vaccination programmes, including vaccine equity through enhanced thermostability and reduced vaccine hesitancy. 25 However, it is important to note that MAPs will not overcome all barriers to vaccination, and further research is needed to sustain vaccine uptake.
Exploring the perceptions of potential consumers and users of MAP vaccines could help to understand human factors specifically safety, usability, and acceptability. This could be useful in determining the potential of MAPs to enhance vaccine uptake and the cost-effectiveness of vaccination programmes. The field of vaccination requires a validated scale to accurately measure the safety, usability, and acceptability of MAP vaccines. In all, 13 studies have assessed or explored the perceived safety, usability, and acceptability of MAPs for vaccination, but no validated scales have been used. There are no standard approaches; thus, findings across the studies cannot be directly compared and the measured outcomes may be less accurate. 25 A validated scale will provide more reliable and precise assessments of these underlying themes. 26
Aim
This study will develop a validated scale to measure the safety, usability, and acceptability of MAP vaccines for vaccination in various settings. It will assess participants’ perceptions of effectiveness, safety, ease of use, views of new technology, cost, and delivery method preferences. This scale will be validated for use in key target populations, including (1) laypeople, (2) MAP recipients, (3) clinicians (i.e. vaccine providers), and (4) parents and guardians of children.
Methods
Study design
The SPIRIT statement informed this study protocol. Although this is not a clinical trial, all relevant items have been documented on the 33-item checklist (Supplemental Material). 27 The development and validation will be guided by the three phases described by Boateng and colleagues: (1) item development, (2) scale development, and (3) scale evaluation. 26 The expert advisory group and cognitive interview phases of this study will be conducted in Australia. The survey administration phase will be conducted across Australia, Canada, New Zealand, and the United Kingdom. Various types of recruitment will occur throughout the phases, including cognitive interviews, expert assessments, and survey participants. The scale will be separated into four independent surveys administered over two time points, (1) laypeople perceptions of MAP for vaccination, (2) MAP recipient perceptions, (3) clinician perceptions of MAP for vaccination, and (4) parent or guardian perceptions of MAP vaccination for their child/children. Each survey will consist of three domains with tailored questions, (1) safety and efficacy, (2) usability, and (3) acceptability. The domains were developed, and key end users were identified based on a literature review and cognitive interviews that were conducted as part of a clinical study. This scale validation study will assess outcomes broadly to allow use between MAP products (e.g. wear times, application sites, and type of administrator) and vaccine types. Participants will be provided with an information sheet with the instructions for use and an application video using the Vaxxas High-Density Microarray Patch currently in development as an example. 28 The study flowchart is illustrated in Figure 1.
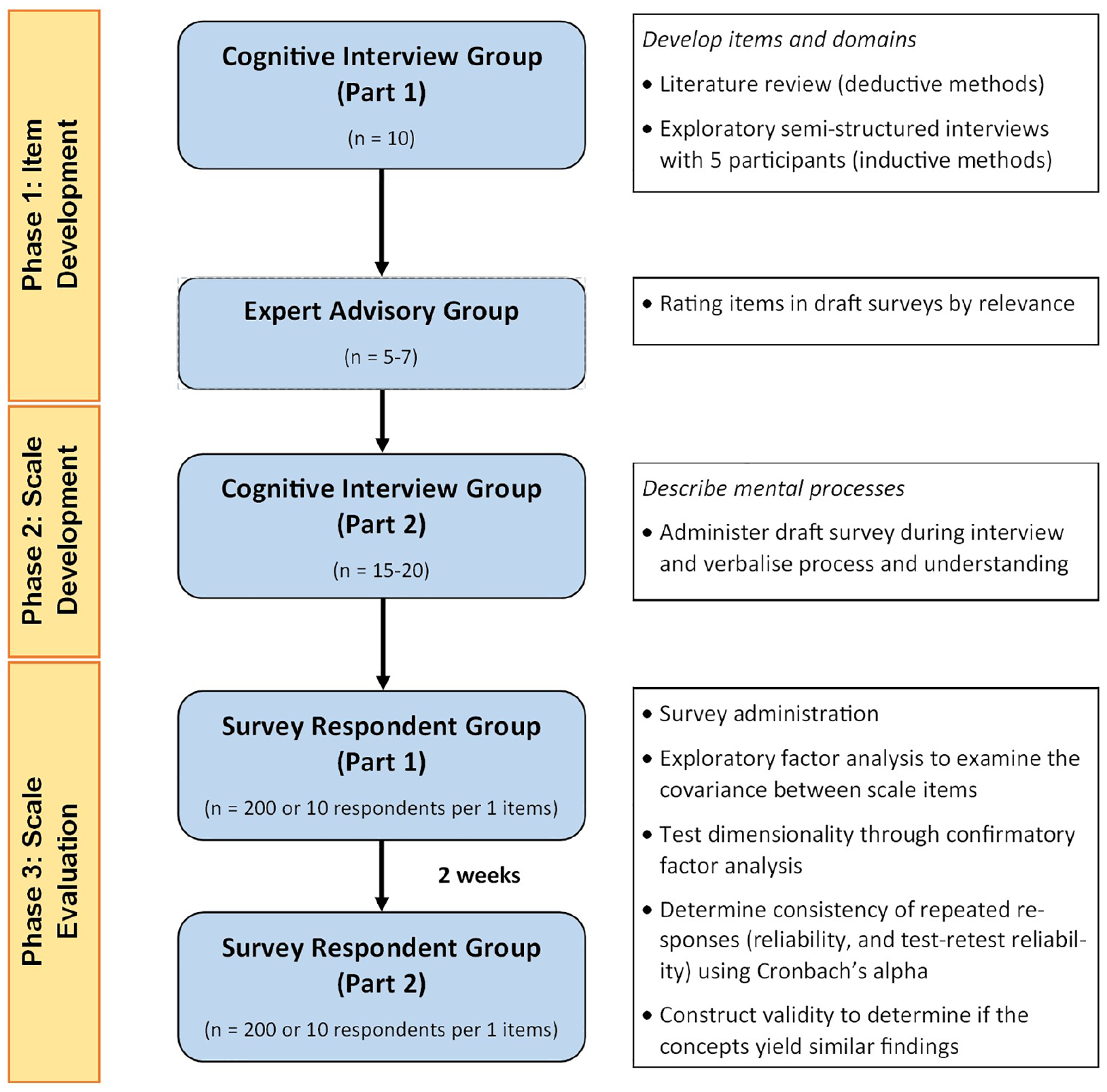
Study flowchart using best practices for developing and validating scales.
Study phases and data analysis
Item development
Several studies have used questionnaires which helped to develop domains and questions from literature searches.7,16,29,30 We will seek to conduct individual interviews to explore and develop domains. 26 Subsequently, five to seven experts will be recruited to evaluate the scale using the content validity ratio to measure proportional agreement. 31 We will seek to recruit experts highly knowledgeable in vaccination, health behaviours, and health economics. Laypeople and parents or guardians will also be recruited to represent target populations and face validity.
Scale development
To ensure that questions and domains produce valid measurements and are meaningful, we will recruit 15–20 interviewees to attempt the scale and describe their processes in providing answers. 26 Interviews will be audio-recorded and transcribed verbatim, and analysed using thematic analysis. 32 The scale will be administered to 200–300 respondents via Research Electronic Data Capture (REDCap) survey. Item reduction analysis using item response theory 33 will promote the retention of items that are functional, parsimonious, and consistent. 26 Using an item difficulty index will allow for identifying individuals’ performance on specific items and developing questions for specified populations. Exploratory and confirmatory factor analyses will determine the ideal number of domains that fit a set of items. 26
Scale evaluation
Tests of dimensionality will be conducted to validate that the previous structure fits the items and estimate an independent cluster model, bifactor models, and measurement invariance. 26 Next, reliability and validity will be analysed by calculating scale scores. Cronbach’s alpha will be used to assess the scale’s internal consistency. 26 Participant performance will be tested and re-tested by determining the relationship between scales over two time points (3 months apart). 26 Multivariable regression will be constructed as part of construct validity to determine if the scale can predict outcomes; stronger associations will indicate predictive validity. 26 Lastly, we will determine the extent the scale assesses the safety, usability, and acceptability of MAP for vaccination and measure real-world criteria as part of construct validity. 26 Statistical analyses will be performed using Stata 17. 34 Findings from the survey administration will be analysed using chi-square tests and outcomes compared between groups and differences in responses from parents’ responses for themselves and their children. A senior researcher will double-check data analyses to ensure data quality.
Recruitment and sample size
Participants for cognitive interviews will be recruited from concurrent studies following the administration of online surveys. Five participants will be recruited prior to expert assessments to generate items, and up to 15–20 participants to trial the draft questions after expert assessments. Experts who are national experts or members of our target populations will be invited via email. Five to seven experts in the field will be recruited to evaluate scale items and domains. Recruitment will occur online for survey testing, targeting laypeople, MAP recipients, clinicians, and parents. Interview and expert participants will go into the draw to win a Coles & Myer gift card (one per group). Survey respondents will go into the draw to win one of five $100 (AUD) Tango gift cards. Sample sizes will be executed according to Boateng and colleagues’ best practice for scale development and validation; 10 responses per item (e.g. for five items, 50 participants would be required). 26 This study will aim to recruit 200–300 survey respondents. Paid social media advertisements will be sought on Facebook, Instagram, and X platforms. Email invites and advertisements will be shared with hospital networks and metropolitan medical centres to recruit clinicians.
Inclusion and exclusion criteria
All participants must (1) be aged 18 years or older, (2) have access to a computer and internet, and (3) communicate effectively with the research team and be willing to cooperate with the study requirements. Experts recruited for the expert assessments must be (1) a layperson, (2) a vaccine provider, (3) a healthcare professional including health economists and epidemiologists, or (4) a parent or guardian of a child/children. Survey respondents may only complete one survey for which they are eligible. Survey respondents must live in Australia, Canada, New Zealand, or the UK. Interview participants and survey respondents must meet the criteria listed in at least one survey from Surveys 1–4.
Survey 1: General public
● Members of the general public with no medical or health qualification or experience.
Survey 2: MAP recipients
● Has received a MAP for vaccination, either a placebo, MAP with the vaccine, or simulated (e.g. applicator only).
Survey 3: Clinicians
● Is a clinician who administers and/or prescribes vaccines to patients (i.e. medical practitioner, registered nurse, nurse practitioner, or pharmacist).
Survey 4: Parents or guardians
● Willing to provide their opinion on behalf of their child.
Individuals will be excluded if they are (1) unable to communicate effectively with the research team or unwilling to cooperate with the study requirements or (2) unwilling to provide written consent to participate.
Ethics, consent, and confidentiality
This research will be conducted in accordance with the Declaration of Helsinki as adopted in the National Statement on Ethical Conduct in Human Research. 35 This study received ethics approval from Metro South Health (Queensland Health) (HREC/2021/QMS/81653) and Western Sydney Local Health District (2023/ETH00705) Human Research Ethics Committees. All participant responses will be de-identified and stored securely via the New South Wales Health online server. Only researchers listed on the project will have access to de-identified participant data. Prior to the initiation of the study, written Human Research Ethics Committee approval of the study protocol and participant information sheet and consent forms will be approved. Before its use, written HREC will approve information and advertisements for this study. Informed consent will be obtained as described in recruitment before the subject participates in the study. Return of a survey during the expert assessments and survey administration will be considered consent. The study’s aims, objectives, and methods in lay terms will be shared and explained to participants. The participant can withdraw from the study at any time, which will be detailed in the information and consent form and reiterated by the research team. Each participant will receive a copy of their information sheet and consent form (for interview groups). Following this process, if a participant meets the inclusion criteria and none of the exclusion criteria, they will receive a study identification number which will be included on all study documents. The ethics committee and investigators will be notified of any necessary modifications to the study.
Dissemination of findings
All investigators’ contributions will be acknowledged, including through the dissemination of study findings (e.g. conferences and journal articles). No publications shall take place without all investigators’ consent. Findings from this study will be disseminated to healthcare professionals, policymakers, and scientists through peer-reviewed publications and conferences.
Discussion
This study will be the first to develop and validate a scale to measure safety, usability, and acceptability for MAP vaccination. The aim is to provide a valid and reliable scale for MAP vaccination, thus reducing measurement errors in future studies. The scale will support a standardised approach to assessing social and behavioural aspects of MAP vaccines in future studies. This will enable comparison of outcomes across studies. This will assist with the effective implementation of MAPs in vaccination programmes by understanding the barriers to vaccine equity for consumers. We are validating a scale aimed at high-income countries where most early-phase trials are occurring.36–39 However, this scale could be adapted to address populations with lower vaccine uptake, such as low- and middle-income (LMIC) countries.
Moreover, MAPs may not address all barriers to vaccination but have implications for overcoming vaccine hesitancy and accessibility. By reducing logistic constraints, MAPs may overcome several barriers to vaccination in LMICs and rural or remote areas. This could include reducing reliance on cold-chain management and the potential for trained laypeople, making vaccines more accessible.19,40 Several MAP studies have reported reduced needle-related anxiety and pain and have the potential for self and lay administration.7,9,19 Effective strategies to improve vaccination coverage are imperative, as we have seen a large decline in childhood vaccination from COVID-19 and suboptimal vaccination coverage in adults, resulting in avoidable incidences of disease and outbreaks.5,6,41
This study will be limited due to using one example of a MAP vaccine. However, we will address common characteristics that can be modified to match the features of the MAP vaccine in question (e.g. wear time duration or application site). This scale will not use any specific vaccine as an example, thus allowing broad perceptions of MAP vaccines that are relevant to the individual’s lived experiences. The scale could be used to inform clinical studies to accurately measure perceived safety, efficacy, usability, and acceptability outcomes. This could also address human factors of MAP vaccine integration into clinical practice to inform policymakers, regulatory consultants, and vaccine providers. This validated scale will support future clinical MAP studies and possibly help vaccination programmes develop effective strategies and integrate MAPs.
Supplemental Material
sj-pdf-1-tav-10.1177_25151355241263560 – Supplemental material for Developing and validating a scale to measure the perceptions of safety, usability and acceptability of microarray patches for vaccination: a study protocol
Supplemental material, sj-pdf-1-tav-10.1177_25151355241263560 for Developing and validating a scale to measure the perceptions of safety, usability and acceptability of microarray patches for vaccination: a study protocol by Matthew N. Berger, Cristyn Davies, Erin Mathieu, Ramon Z. Shaban, Shopna Bag and S. Rachel Skinner in Therapeutic Advances in Vaccines and Immunotherapy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
