Abstract
Background:
Vaccination is a safe and effective way to prevent disease and save lives, but it may also produce some undesirable adverse events (AEs)which may affect healthy individuals. Therefore, the monitoring of AE following immunization (AEFIs) is necessary. The objective of this study was to assess the AEs following COVID-19 vaccinations in a tertiary care hospital.
Methodology:
The study was conducted as active vaccine safety surveillance for a period of 6 months among the COVID-19 vaccine beneficiaries of the study site. Active surveillance was conducted via initiating two telephone contacts. The first surveillance was conducted in 8 days and the second surveillance after 28 days of post-vaccination. All identified AEs following immunizations (AEFIs) were reported and analysed by the AEFI investigation team at the study site. The causality assessment of each identified AEFI was performed using the World Health Organization’s causality assessment algorithm.
Results:
A total of 2927 enrolled study population completed the study with a response rate of 80.85%. The study identified 902 AEFIs from 614 study populations with an incidence rate of 20.97%. Of which 794 and 79 AEFIs were associated with COVISHIELD™ and COVAXIN®, respectively. The majority of the events were reported among the age group of 18–29 years. Overall, only three events were serious and no deaths were reported among the study population. A total of 75.59% of events had a consistent causal association with vaccination and were categorized as vaccine product-related reactions. The study identified various factors such as gender (p = 0.019), age (p < 0.05), co-morbid status (p = 0.032) and dose number (p = 0.001) as potential predictors for development of AEFI.
Conclusion:
The study identified only 0.33% of events as serious, and 99.67% of the study population recovered from the AEFIs, which reveals that COVISHIELD™ and COVAXIN® have a generally favourable safety profile. However, close monitoring is required to identify the potential signals, as the safety data from the clinical trials are limited.
Keywords
Introduction
Vaccination has been regarded as a major tool by many health authorities and the community of medicine for the achievement of public health success rates, to quote an example of smallpox eradication. They have been recommended for all age groups, starting from infants to geriatrics. For a large group of individuals, the concept of vaccination has not been accepted wholeheartedly, which might be due to their safety aspects and the adverse events (AEs) they might supposedly cause. 1
The process of licensing and marketing the vaccines occurs only after the approval by the regulatory agencies of the country with utmost care and evidence. The important aspect begins during the post-marketing surveillance studies, wherein the goal is to detect the occurrence of any AEs that have not been reported during the clinical trial phases. The World Health Organization (WHO) recommended the surveillance of AEs following immunizations (AEFIs) in the year 1991. The proclamation of these data could elevate the reliability and awareness among the general public regarding the safety of the vaccines and their consent to take any decisions regarding vaccination.2,3
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a novel coronavirus that first appeared in China in December 2019 and has since spread over the world and become a worldwide pandemic with high morbidity and mortality. With the appearance of this menace, a massive push to invent vaccines against it began. In less than a year, several manufacturers have produced and assessed vaccines; some are now being used widely in numerous countries under emergency or conditional use authorizations.4,5
COVID-19 vaccines were given emergency use authorization before the regular rounds of clinical testing were completed. However, there is no complete analysis of safety data from vaccination trials, which gives crucial information for developing strategies to promote COVID-19 vaccine uptake and minimize risk aversion associated with COVID-19 vaccine AEs. 6 COVID-19 vaccine was introduced by the Government of India on 16th January 2020 with active surveillance of AEFIs, and information gathered is submitted to databases such as the CoWIN (a database in India), which was evaluated to determine the acute immunization against COVID-19 in the country for the monitoring of the vaccine delivery. It carries a risk regrettably because vaccination was approved for emergency use and there is limited data to determine the long-term and chronic AEs of this newly developed COVID-19 vaccine. 7
The accelerated development and introduction of these new vaccines created many safety concerns among the public. Appropriate measures should be taken to address such concerns to avoid hesitancy among the public. This study was conducted to evaluate the AEs following COVID-19 vaccinations in a tertiary care hospital.
Methodology
Study method
An active surveillance study was conducted in the immunization centre of JSS Hospital, Mysuru, India for a duration of 6 months (September 2021 to March 2022). The study included a population of any gender aged ⩾18 years, who received any dose of COVID-19 vaccination at the vaccination centre of the hospital during the study period.
Data collection
The study population was given a validated patient information leaflet with information about COVID-19 immunization, probable AEFIs and the study team’s contact information. Beneficiaries were informed to report back to the immunization centre or notify the research team in case of any AEs occurring following the immunization. The study team also conducted active surveillance by initiating two telephone calls from the study site such as surveillance I: 8 ± 2 days of post-vaccination and surveillance II: 28 ± 2 days of post-vaccination. The study population who does not respond to any of the surveillance was considered as dropouts from the study. Each beneficiary was interviewed for any AEFIs during the surveillance period and the details were documented.
All demographic details such as age, gender, contact information and any pre-existing medical issues of the subject were meticulously captured, as well as vaccine details and the time and date of vaccination of each participant were documented in the data collection form. In case of any AEFI reported among the study population, a suitably designed data collection form was used to collect the required data. The data collection form had a provision to document demographic details, allergic status, past medical history and AEFI details. The AEFI section of the data collection form was developed based on WHO’s AEFI core variables and had a provision to collect details of the vaccine, description of AEFI, date and time of the start of AEFI, date and time of stop of AEFI, duration, severity, seriousness, details of medical attention sought due to AEFI, management of AEFI and the outcome of the developed AEFI. In case of any serious AEFIs reported among the study population, the medical records were thoroughly evaluated and the details such as diagnosis, laboratory findings (if any), treatment and the outcome were recorded. Serious AEFIs cases were discussed with the treating medical team to get clarification on the unavailable or doubtful information. The reported instances were subjected to their causal association with the vaccine using the WHO causality assessment algorithm.
Causality assessment
Reported occurrences were classified as minor, severe or serious, depending on their severity. The causality assessment was performed by the AEFI causality assessment team of the study site, composed of two senior professors of Paediatrics, a clinical pharmacist and a clinical pharmacologist who have interest and experience in the area of vaccine safety. Each reported event was confirmed with a valid diagnosis with an appropriate case definition (such as the definition given by Brighton Collaboration, standard literature). The causality assessment of each reported AEFI was performed by considering various principles such as the strength of association, specificity, temporal relationship between the event and vaccine, consistency of the event and biological plausibility. Finally, the AEFIs were categorized as per the causality assessment classification. 8
The comparison of quantitative characteristics such as age group of the study population with or without AEFI was done using an independent t-test. Bivariate analysis was performed to identify the predictors of AEFI among the study population.
Results
During the study period, 3620 eligible populations were enrolled in the study from the site and 19.15% (n = 693/3620) were dropped out due to no response to any of the surveillance conducted by the study team. The number of population who completed the study was 2927, with a response rate of 80.85% (n = 2927/3620) and all of them belong to the Asian ethnic group. The majority of the study population received or utilized COVISHIELD™ (n = 2466) as compared to COVAXIN® beneficiaries (n = 461) among the study population. Among the responders, the majority of them were females that is 54.26% (n = 1576/2927) and 43.08% (n = 1261/2927) of the vaccine beneficiaries belonged to the age group of 18–29 years, followed by 28.93% (n = 847/2927) in 30–39 years. The demographic details of the study population are detailed in Table 1.
Details of the vaccine beneficiaries.

Time–temporal relationship of the vaccines
During the study period, a total of 614 COVID-19 vaccine beneficiaries reported 902 AEs, with a calculated incidence rate of 20.97% (614/2927 × 100). Of which COVISHIELD™ vaccine alone accounted for an AEFI incidence rate of 18.82% (n = 551/2927) and COVAXIN® contributed 2.15% (n = 63/2927). A majority of the female population reported a high number of AEFIs (59.77%, n = 367/614), while the male population had a low incidence of AEFIs (40.22%, n = 247/614). The age group 18–29 years (51.6%, n = 317/614) has the highest prevalence of AEFIs, followed by 30–39 years (25.7%, n = 158/614) and 40–49 years (14.1%, n = 87/614). Identified AEFIs were reported in various time sequences such as 87.62% (n = 538/614) of them developed AEFIs in the first 24 h, 22.28% (n = 14/614) in the next 48 h, 2.76% (n = 17/614) in the next 72 h, 2.76% (n = 17/614) in the next 96 h and 4.71% (n = 29/614) in the next 4 days of post-vaccination. Table 2 gives a full account of the vaccine’s time–temporal association with AEFIs.
Time–temporal relationship of COVISHIELD™ and COVAXIN® with development of AEFIs.
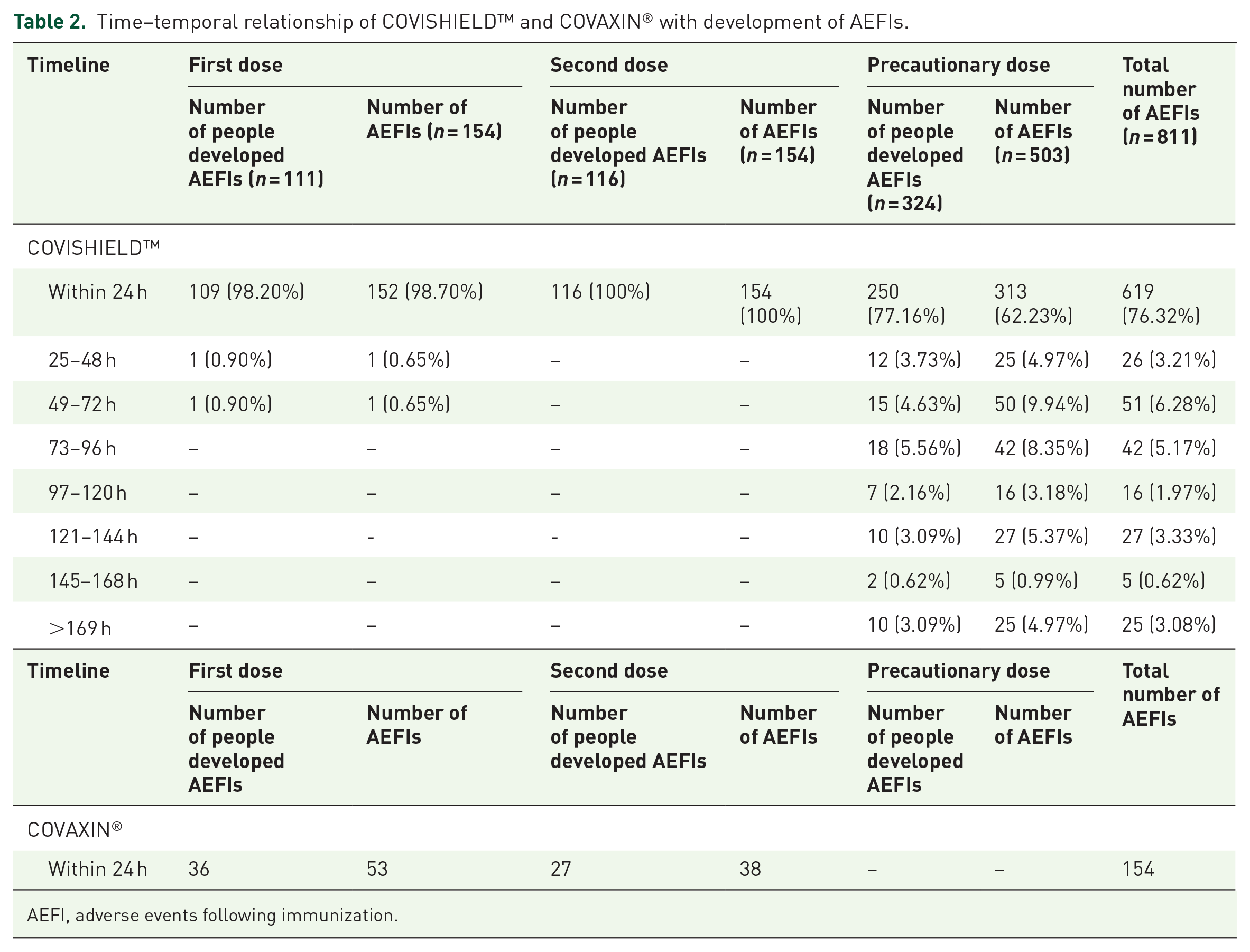
AEFI, adverse events following immunization.
Reported AEFIs of the vaccines
There were a total of 902 AEFIs in 614 patients and an average of 1.4 AEFIs per beneficiary with a range of 1–6 AEFIs. Among the study population, a total of 68.72% (n = 422/614) of beneficiaries reported only one AEFI, and 20.52% (n = 126/614) of beneficiaries reported two AEFIs. Additionally, 7.81% (n = 48/614) of them reported three events, 1.30% (n = 8/614) with four AEFIs and 1.46% (n = 9/614) with five AEFIs. While a total of six different events (0.1%) were reported in a single individual during the surveillance period.
According to the fact sheet, 873 of the reported AEFIs were expected events, of which 794 AEFIs were developed after COVISHIELD™ and 79 were accounted for by COVAXIN® immunization. The study estimated a total of 18.04% (n = 143/794) AEFIs were reported after the first dose of the COVISHIELD™ vaccine, while 19.14% (n = 152/794) and 62.84% (n = 499/794) AEFIs were reported after the second and precautionary dose of the COVISHIELD™ vaccine, respectively. Similarly, 55.69% (n = 44/79) AEFIs were reported with the dose of COVAXIN® and 44.30% (n = 35/79) AEFIs with the second dose. The most commonly reported AEFI for both COVISHIELD™ and COVAXIN® was fever, followed by pain at the injection site, tenderness, redness/warmth at the injection site, swelling at the injection site, itching/bruising at the site of injection and body soreness. Tables 3 and 4 provide a detailed description of the AEFIs that have been documented.
Reported AEFIs of COVISHIELD™.

AEs, adverse events; AEFI, adverse events following immunization.
Reported AEFIs of COVAXIN®.

AEs, adverse events; AEFI, adverse events following immunization.
System organ classification associated with AEFIs
According to the System Organ Classification (SOC), 87.69% (n = 791/902) of AEFIs were categorized under ‘general disorders and administration site problems’. Whereas the least number of AEFIs which is 0.22% (n = 2/902) was reported under ‘cardiac disorders’ and ‘respiratory, thoracic, mediastinal diseases’. The details of the system organ classes reported with AEFIs are presented in Figure 1.

System organ classification associated with AEFIs.
Severity assessment of AEFIs
Overall, 0.33% (n = 3/902) of the events were severe, 92.24% (n = 832/902) AEs were mild and 7.53% (n = 68/902) were moderate. None of the AEFIs lead to death in the study population. Self-medication was used by 14.1% (n = 87/614) of the study participants. The majority of the patients took paracetamol tablets for symptomatic relief of adverse occurrences.
Causality assessment
The WHO’s causality assessment algorithm was used to examine the reported AEFIs’ causality. After the causality assessment of each reported AEFI, 75.59% (n = 613/811) events were determined as ‘vaccine product-related reaction’, 0.37% (n = 3/811) events were ‘immunization anxiety-related reaction’, 0.49% (n = 4/811) events were ‘immunization error-related reaction’ and 23.55% (n = 191/811) events were ‘coincidental event reaction’. All 91 AEs after COVAXIN® vaccination were classified as having a ‘consistent causal association with immunizations’, of which 97.8% (n = 89/91) events were ‘vaccine product-related reactions’ and 2.19% (n = 2/91) cases of ‘immunization anxiety-related reaction’. The types of AEFIs and causality assessment categories are depicted in Figures 2 and 3, respectively.

Types of AEFI.

Causality assessment of reported AEFIs.
Predictors of AEFI
The different possible predictors for developing AEFI among the study population were checked using bivariate analysis. The reported age, gender, doses and co-morbidities were considered as different potential predictors of AEFI development among the study population irrespective of the type of vaccine administered. Vaccine beneficiaries who received COVISHIELD™ identified the co-morbid conditions as a potential predictor for developing AEFI among the study population. Among these, hypertension (n = 7) was the most common concomitant condition, followed by hypothyroidism (n = 1), bronchial asthma (n = 1) and diabetes (n = 4). The details are collected in Table 5.
Predictors of reported AEFIs.
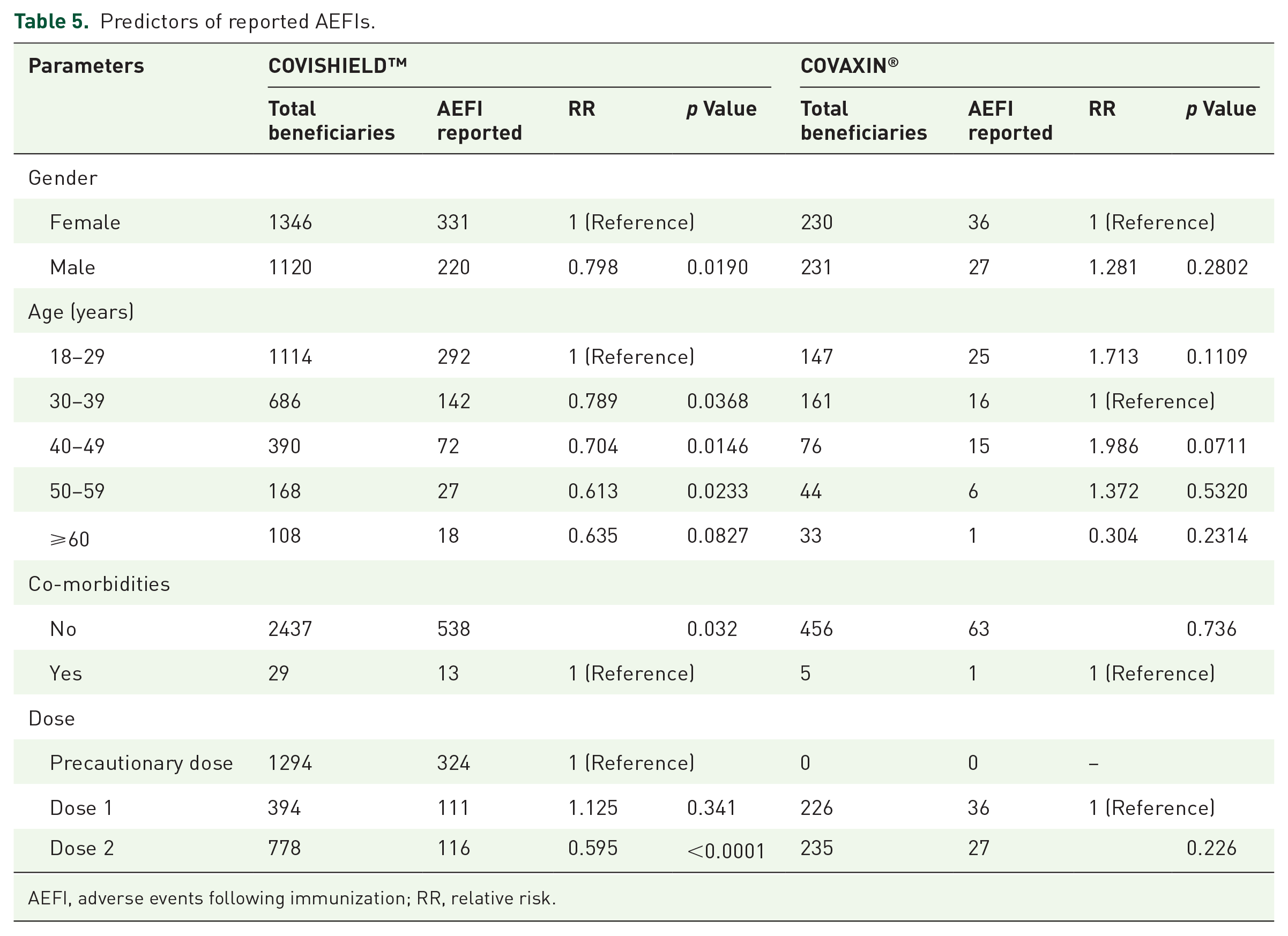
AEFI, adverse events following immunization; RR, relative risk.
Barriers to AEFIs
Incorrect numbers (1.73%, n = 12/693), non-response from the study population (95.95%, n = 665) and unwillingness to participate in the study (2.30%, n = 16/693) were the reasons for the 19.15% dropout rate. Telephonic call-related barriers such as non-response of the subjects, invalid numbers submitted while collecting the information and the inability of the subject to spare time. There was also difficulty in receiving all the data through phone calls. Mild AEFIs were not reported due to the lack of understanding of AEFI. Confidentiality issues such as not being willing to disclose the information were also major barriers.
Discussion
COVISHIELD™ and COVAXIN® are two of the highly utilized COVID-19 vaccines available in India against this pandemic. COVISHIELD™ has emergency use in India on 16 January 2021 COVAXIN® and was subsequently added to the vaccination programme from 13 March 2021. 10 In India, a total of 1,91,54,86,297 doses of COVID-19 vaccine were delivered through 29,818 immunization centres (first dose: 1,00,84,19,145; second dose: 87,69,97,020; and precaution dose: 3,00,70,132). 9 The highest COVID-19 vaccine utilized in the country was found to be COVISHIELD™ as compared to any other vaccines (79.28% of vaccine beneficiaries immunized with COVISHIELD™). 9 Similarly, the study site also accounted for increased utilization of COVISHIELD™ vaccines, that is, 84.25% (n = 2466) among the study participants. The indigenous vaccine COVAXIN® was the second most utilized COVID-19 vaccine in the country (16.49%), while the study site also observed 15.75% (n = 461) of utilization. 9
This study was conducted during the period of precautionary dose recommendation in the country and therefore the study population was seen to be high in the group of precautionary doses utilizers (44.21%, n = 1294). However, during this timeline, the study site observed no demand for precautionary doses of COVAXIN® vaccines, since the population who received first and second doses of COVAXIN® was out of the timeline for the precautionary dose. The previous study conducted in the same study site 11 observed a low utilization of COVAXIN® as compared to the COVISHIELD™ confirming that the demand for COVISHIELD™ vaccines was more in this locality. Hence, this study accounted for more number of AEFIs following COVISHIELD™ as compared with COVAXIN®.
The study reported an AEFI incidence rate of 20.97% from 2927 participants, that is, 902 AEFIs were reported from 614 COVID-19 vaccine beneficiaries. This was considered to be high when compared with the national AEFI incidence rate which accounted for 0.006% (according to the Indian government database for COVID-19 vaccination: Co-WIN portal). 9 A prior COVID-19 vaccine safety study using the spontaneous reporting method conducted in the same study site documented an AEFI incidence rate of 3.48%. 11 However, an increased incidence of AEFIs notified in this study can be explained by the difference in study design, here active surveillance was employed. A study conducted by Riad et al. 12 supports that the active surveillance method is the best tool to detect short-term and long-term side effects among post-authorized COVID-19 vaccines. 12
During the study period, a low frequency of AEFIs (16.67%) was reported among the geriatric population. This could be due to more enrolment of younger and middle-aged adults in the study as compared to the geriatric population. Moreover, our study period bypassed the priority timeline for vaccinating the elderly and people with co-morbidities (according to the country’s prioritization criteria) 13 against the COVID-19 infection. The patients aged 18–29 years (younger) made up 43.08% of our study and the age group with the most frequently reported AEFIs was also from the same age group (26.21%). This study identified young age as a potential predictor for developing AEFI, the incidence of AEFI among younger (18–29 years), middle (30–49 years) and late-middle-aged (50–59 years) population was high as compared to the geriatric (⩾60 years) population. However, rather than a cross-sectional study conducted in Nepal, 14 many of the contemporary studies observed a similar trend of high-risk factors for developing AEFIs among younger or middle-aged individuals as compared to the older age.15–17
Other than age, the study population was identified with few other predictors for developing AEFIs such as female gender, vaccine dose and co-morbid status. A prospective observational study conducted by Menni et al. supported that AEFIs were highly reported among females as compared to males irrespective of the vaccine received. 18 In this study, the female gender was considered as a potential risk factor for developing an AEFI among the COVISHIELD™ vaccine beneficiaries, while a similar trend among COVAXIN® beneficiaries was not observed. This gender variation can be explained by the theory stated by Fink et al. 19 that more vigorous immune response and build larger cell-mediated and humoral immune responses towards the antigenic stimulation resulting via vaccination or infection among women as compared with men. 19 By contrast, the research conducted by Kamal et al. 20 from South India found that no risk factor was associated with gender for developing an AEFI.
The study accounts for less number of people with co-morbid status; however, among the COVISHIELD™ vaccine beneficiaries, co-morbidity was observed as a significant predictor for developing AEFIs. Even though the study population was more concentrated with the precautionary dose utilizers, the COVISHIELD™ second dose receivers were found to be more prone to develop AEFIs (p value < 0.0001). A contemporary study conducted in North India by Kaur et al. also observed that dose is a significant predictor of AEFI, where 40% of the AEFI were prone to develop among the first-dose vaccine users and 16.67% AEFI was found in second-dose vaccine users. 15 However, this study could not identify potential predictors among COVAXIN® beneficiaries due to very low demand. This study could be compared with similar predictors identified by Parida et al., 27 where they could identify many more predictors such as past intake of steroids, past allergic reactions, past intake of drugs in the last 6 months and infection within 3 months. 27
During the study period, no deaths were reported while four serious AEs (SAEs) were reported following COVISHIELD™ immunization (breathing difficulty, autoimmune hepatitis, 21 autoimmune arthritis, leucocytoclastic vasculitis 22 ), which required hospitalization. Each reported SAEs during the study period underwent detailed investigation by the study team and was classified according to the WHO causality assessment algorithm. 8 These SAEs identified in this study were not included in the fact sheet while several similar case reports were accounted for globally.23–26 According to the SOC, the majority of the AEs were categorized under ‘general diseases and administrative site problems’. The most common local symptom was injection site soreness, while the most common systemic symptom was fever. The most prevalent AEFI was fever, followed by itching, discomfort, soreness, redness or bruising at the site of injection and body ache. This investigation’s findings are very comparable with those of Shrestha et al. 14 Majority of the events reported in the study were mild to moderate which implies that COVID-19 vaccinations have a higher safety profile.
The causal association of the event with the vaccination was consistent in 79.2% and 100% of events reported following COVISHIELD™ and COVAXIN®, respectively. A total of 639 (78.79%) events after COVISHIELD™ and 89 (97.80%) events after COVAXIN® are categorized as vaccine product-related reactions. Immunization anxiety-associated reactions were found in 0.33% (n = 3) of occurrences following COVISHIELD™ and two (2.1%) AEs following COVAXIN® immunization. Three of the events were classified as indeterminate reactions (Category B) 8 following COVISHEILD™ vaccination. Although the temporal relationship of these events was consistent, clear proof for vaccine-related incidents was lacking. These events may constitute warning signs or potential new signals, although they were not addressed in the fact sheets. There were 189 incidents classed as coincidental because they were not caused by vaccination, immunization error or immunization fear. A study conducted by Subedi et al. 28 analysed that more than half of the vaccines (57.1%) expressed some level of worry before getting vaccinated and the vaccine reluctance was due to fear of the newly introduced vaccine. Additionally, a total of 14.1% of the study population managed the AEFIs via self-medication, of which a majority of the patients were given paracetamol tablets to treat the symptoms of AEs. Most of the developed events were self-resolved and only three events persist (autoimmune hepatitis, autoimmune arthritis and leucocytoclastic vasculitis).
Limitation
As our study was based on follow-up through phone calls, there were certain barriers such as non-response and invalid numbers. It was difficult to get all the data through phone calls due to the inability to spare time. There have been subjects who were not willing to disclose the information which is attributed to confidentiality reasons. Additionally, more utilization of COVISHIELD vaccines was observed among the study population, which resulted in this vaccine safety study being more concentrated towards one vaccine (COVISHEILD).
Conclusion
Vaccine safety studies help healthcare professionals and the general population to realize that COVID-19 vaccines are safe to be administered. Active surveillance of vaccine safety studies could bring out most of the underreported and delayed AEs. The study could also identify a few risk factors for developing AEFI such as female gender, young to middle age and co-morbid status. Many of the non-serious immediate and rare delayed AEFIs spotted in this study could conclude that accelerated developed new COVID-19 vaccines are relatively safe to administer. This study could convince the population who are hesitant to immunization due to various concerns about COVID-19 vaccine safety. Hereby, more than worrying about mild AEFIs, achieving herd immunity against these COVID-19 infections through effective vaccination is essential for going back to normal life.
Footnotes
Acknowledgements
The authors would like to acknowledge the support received from the team at the COVID-19 immunization centre of the study site. We also thank the administrators and our colleagues for their encouragement throughout the study, which made the data collection and assessment possible.
