Abstract
Approximately 2.3 million people are suffering from human immunodeficiency virus (HIV)/hepatitis C virus (HCV) co-infection worldwide. Faster disease progression and increased mortality rates during the HIV/HCV co-infection have become global health concerns. Effective therapeutics against co-infection and complete infection eradication has become a mandatory requirement. The study of small non-coding RNAs in cellular processes and viral infection has so far been beneficial in various terms. Currently, microRNAs are an influential candidate for disease diagnosis and treatment. Dysregulation in miRNA expression can lead to unfavorable outcomes; hence, this exact inevitable nature has made various studies a focal point. A considerable improvement in comprehending HIV and HCV mono-infection pathogenesis is seen using miRNAs. The prominent reason behind HIV/HCV co-infection is seen to be their standard route of transmission, while some pieces of evidence also suspect viral interplay between having a role in increased viral infection. This review highlights the involvement of microRNAs in HIV/HCV co-infection, along with their contribution in HIV mono- and HCV mono-infection. We also discuss miRNAs that carry the potentiality of becoming a biomarker for viral infection and early disease progression.
Introduction
In 1983, a French virologist Luc Montagnier and his team first reported about a virus, which later became identified as human immunodeficiency virus type 1 or HIV-1 in the field of medical virology.
1
Just after which another two groups of researchers collected enough evidence, from isolated samples, that claimed HIV to be the causative reason behind the acquired immunodeficiency syndrome, acronym as AIDS.2,3 Characteristics like consisting of two positive strands of RNA encoding for numerous proteins and viral microRNAs (vmiRNAs) make HIV stand out among the other viruses. Researchers have, so far, discovered five HIV-1 microRNAs and 15 proteins, which facilitate various viral processes
On the contrary, HCV is also a blood-borne virus, like HIV, belonging to the family
Both HIV and HCV are infectious and can spread through blood, hence sharing a common transmission route. Usage of contaminated needles, syringes, or drug injection equipment is always seen to be the primary reason behind getting an infection of HCV or HIV. This leads to a higher chance of getting infected by HIV and HCV co-infection. Currently, available data show that about 36.7 million people are HIV infected globally, among which 2.3 million reported having HCV infection, past or present. Interestingly, 1.36 million co-infection patients were ones who were treated with intravenous drugs. 10 The natural history of both the viruses significantly alters during the co-infection. While the HCV viral RNA count, liver fibrosis, and other clinical features of hepatic dysfunction develop more rapidly in HIV-infected patients, the disease progression toward AIDS also observed much faster in HIV/HCV co-infection than in HIV alone. 11
MicroRNAs: the game-changer in viral pathogenesis
Till now, about 3000 miRNAs have been identified in mammalian genome.
12
microRNAs are 18–22 nucleotide long, non-coding RNAs. They play a vital role in different cellular activities like proliferation, apoptosis, and differentiation by controlling gene expression. They are highly conserved in human genomes. When required, RNA polymerase II transcripts form primary miRNAs (pri-miRNAs). This Pri-miRNA consists of 5′-end cap and poly-A tail, further refined by ribonuclease Drosha to form precursor miRNA (pre-miRNA). This hairpin structured Pre-miRNA is then transported to the cytoplasm with the help of Exportin-5. There it is cleaved by Dicer to form mature miRNA (Figure 1).
13
Although miRNAs form not more than 1% of the human genome but are capable of targeting approximately 60% of mRNA present in the genome.
13
miRNA recognizes the target mRNA
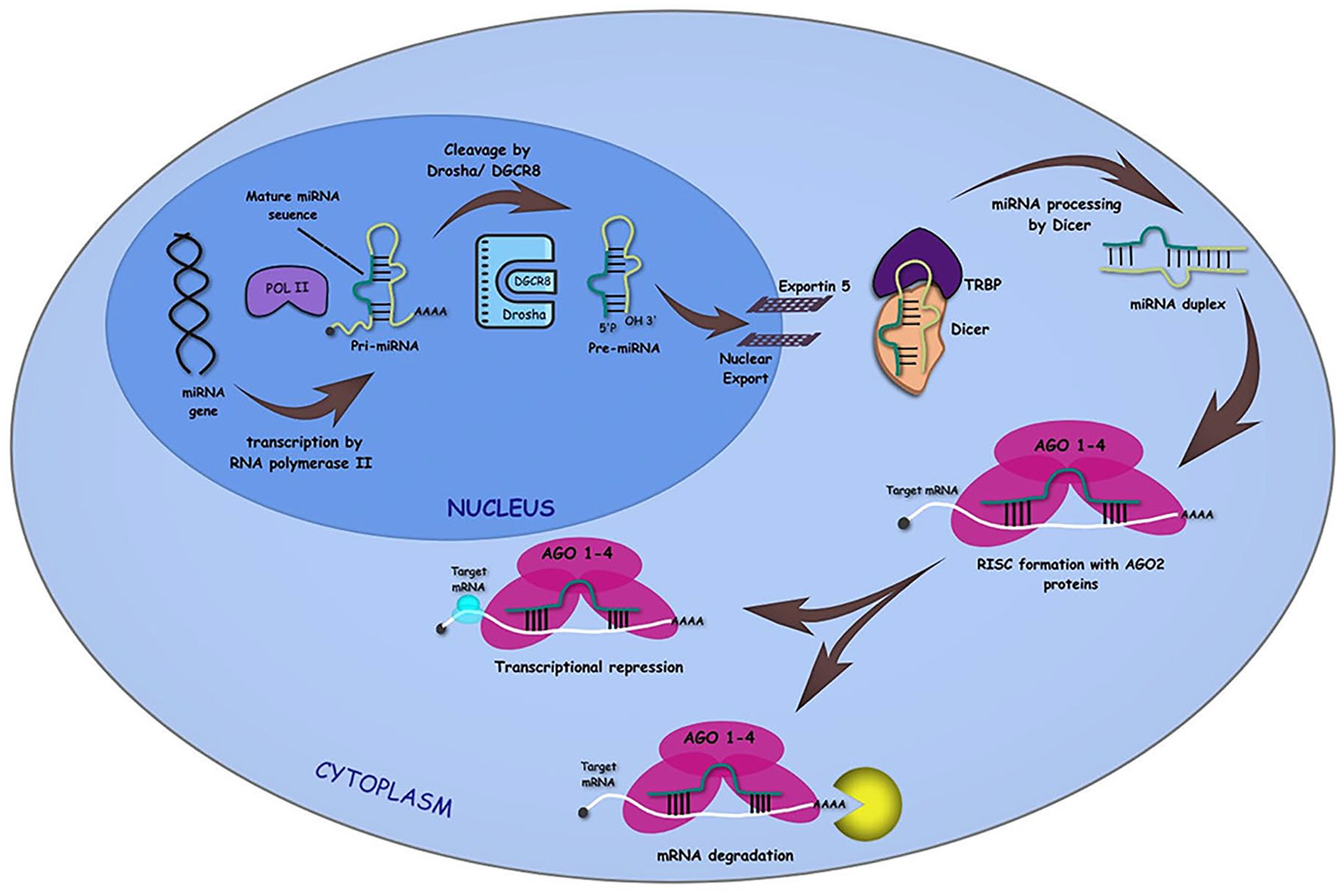
The Biogenesis of microRNAs by Canonical Pathway: Maturation of miRNA sequence by transcription of primary miRNAs or Pri-miRNAs from the miRNA gene by RNA polymerase II followed by the cleavage of Pri-miRNAs to precursor or Pre-miRNAs through Drosha/DGCR8. The Pre-miRNAs are then exported from nucleus to cytoplasm with the help of Exportin 5. In the cytoplasm, Dicer cleavage of the hairpin structure takes place, where one of the strands of the miRNA duplex is loaded onto the RISC
miRNAs in HIV lifecycle
HIV replication
For decades, HIV has successfully kept scientists busy with their research on finding its every possible cure. One of the properties that make HIV stand out is its ability to encode its own microRNAs. One such viral miR, hiv1-miR-H1, is seen to promote HIV replication in macrophages successfully.
25
It is found near NF-κB sites in the LTR region. It enhances the infection and cell damage by targeting apoptosis antagonizing transcription factor (AATF), leading to apoptosis. It also targets a few cellular proteins like c-Myc and Dicer.
25
Likewise, the viral genome also encodes for transacting responsive hairpin (TAR) consisting of a terminal repeat region. After asymmetrical slicing by Dicer, the miRNAs produced from these regions are claimed to be distinctively crucial because of their varied role in the HIV life cycle.
26
They participate in various processes like dimerization, replication, transcription, and translation. In 2008, researchers first established the location of HIV-1 TAR to be present at 5′ end in a stem-loop structure.
26
The same group later established another unique characteristic of TAR miRs, where they use AGO proteins to manipulate cellular mRNA expression.
27
In addition to other studies, bioinformatics showed the higher possibility of TAR having complementary sites within genes of apoptosis-like Caspase 8, Aiolos, and Nucleophosmin (NPM)/B23. In conclusion, HIV-1 TAR is completely capable of causing an imbalance between apoptosis and cell viability in T cells by influencing gene expression.
27
Some studies also claim its presence in monocytes-derived macrophages, responsible for monitoring viral replication rate
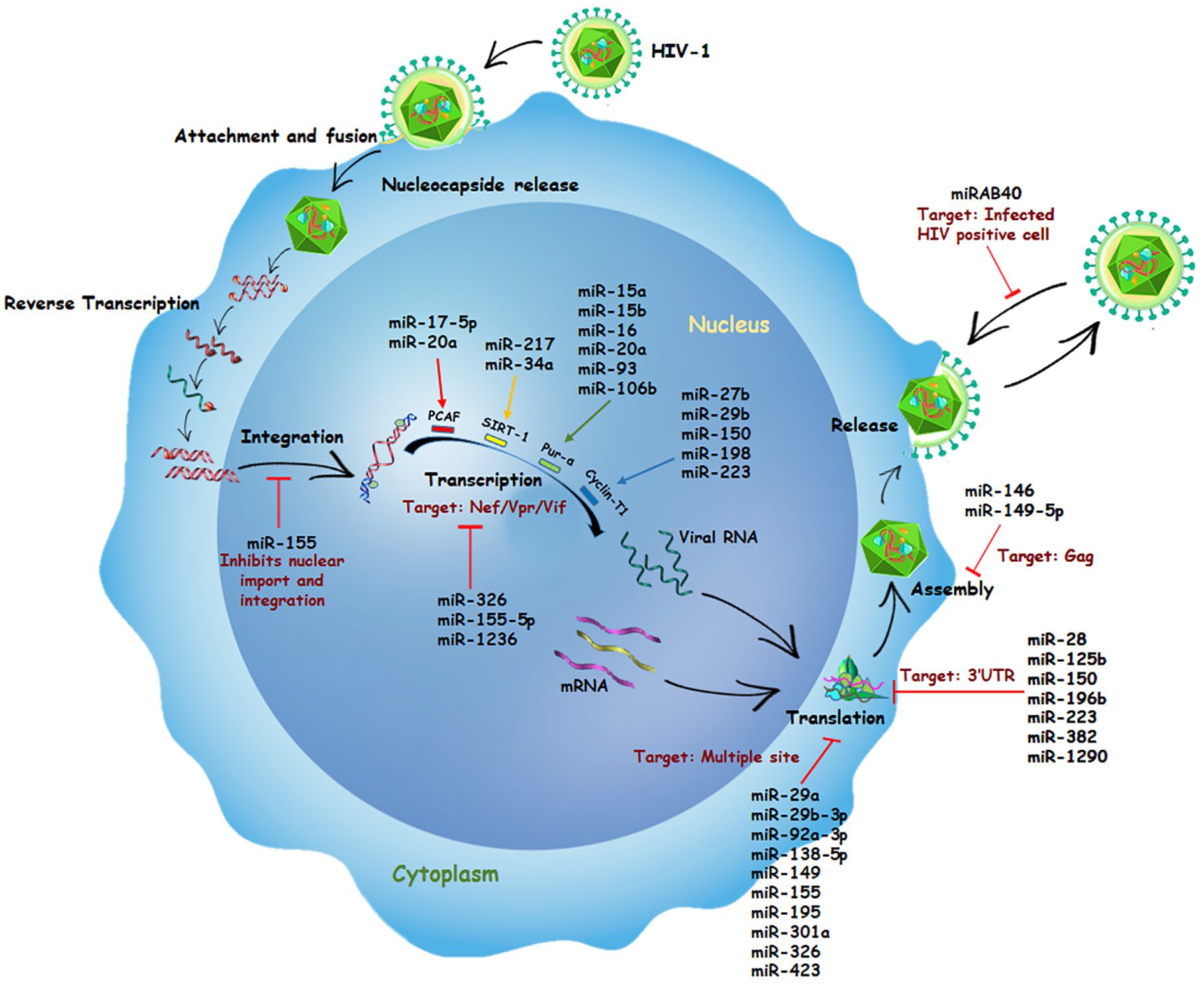
Role of miRNAs in HIV replication: The figure describes the mode of action of various cellular and viral miRNAs that regulate the HIV replication. While the cellular miRNAs, miR-28, miR-29, miR-92a, miR-125, miR-138, miR-146, miR-149, miR-150, miR-155, miR-195, miR-196b, miR-223, miR-301, miR-326, miR-382, miR-423, miR-1236, and miR-1290, and the viral miRNA, miRAB40 block the HIV replication, the cellular miRNAs, miR-15, miR-16, miR-17, miR-20a, miR-27b, miR-34a, miR-93, miR-106b, miR-198, and miR-217 are reported to be promoting the viral replication. Both the viral and the cellular miRNAs regulate the host and the viral gene expression or HIV-positive cells to modify the replication process either as a host response trying to fight the infection or to block or establish, or both, the infection within the host at different stages of virus lifecycle.
HIV latency
The period in HIV infection, about 6 weeks later, with no signs and symptoms is also called asymptomatic HIV infection. Here, the virus is still active and replicating at a meager rate, yet fully capable of progressing the infection to its 3rd stage, that is, AIDS. These latent proviruses manage to escape immune responses, and even current ART treatments fail to target them. Like any other process, this also employs various viral and cellular miRNAs. Numerous studies investigated miRNA differences between elite controllers and chronic HIV samples in order to target essential miRNAs for diagnostic or therapeutic uses. One such study found concentrated amounts of miR-125 and miR-150 present in cells undergoing latent infection, possibly because of their notable role as HIV protein suppressors. 29 At the same time, another study showed contrary results where they found these same miRNAs to be downregulated in HIV elite controllers compared with chronically infected samples. 30 A similar study done by Reynoso R. revealed increased expression of miR-146a-5p, miR-33a-5p, and miR-29b-3p in those HIV-positive patients who controlled viral replication in the absence of ART. 31 A lot of miRNAs like miR-28, miR-223, miR-382, miR-125b, and miR-150 exhibit their role in maintaining latency period, while other studies have also added another two miRNAs to that list, miR-196b and miR-1290.29,32 These miRNAs simply bind to 3′ UTR of HIV mRNAs and contribute to HIV latency.29,32 Furthermore, a recent study showed the contribution of host miRNAs in dysregulation of the p53 signaling pathway during HIV infection that drives latent infection. 33 Along with cellular miRNAs, HIV encoded viral miR-TAR, miR-N367, and miR-H1 have also established their roles in HIV latency, and on the contrary, miR-H3 has proved its role in activation of the latent reservoirs. 34 miR-N367 is located within Nef gene region in the HIV genome. 35 Nef protein is responsible for the easy production and release of virions from the infected cell. 25 Hence, its degradation led by hiv1-miR-N367 helps in viral latency. 25 The presence of such transcriptionally silent virus in resting CD4 + cells increases the chances of reoccurrences. Hence, other than ART treatments, it has become essential to target factors like miRNAs that are responsible for viral latency. This new therapeutic approach might help us find a definite cure for HIV.
miRNAs directly target HIV-1 genome
As previously discussed, miRNAs regulate viral processes either by interfering with cellular mechanisms or by directly targeting the viral genome (Figure 3).16–18 Likewise other viral proteins, HIV Vpr protein is crucial in promoting viral infection
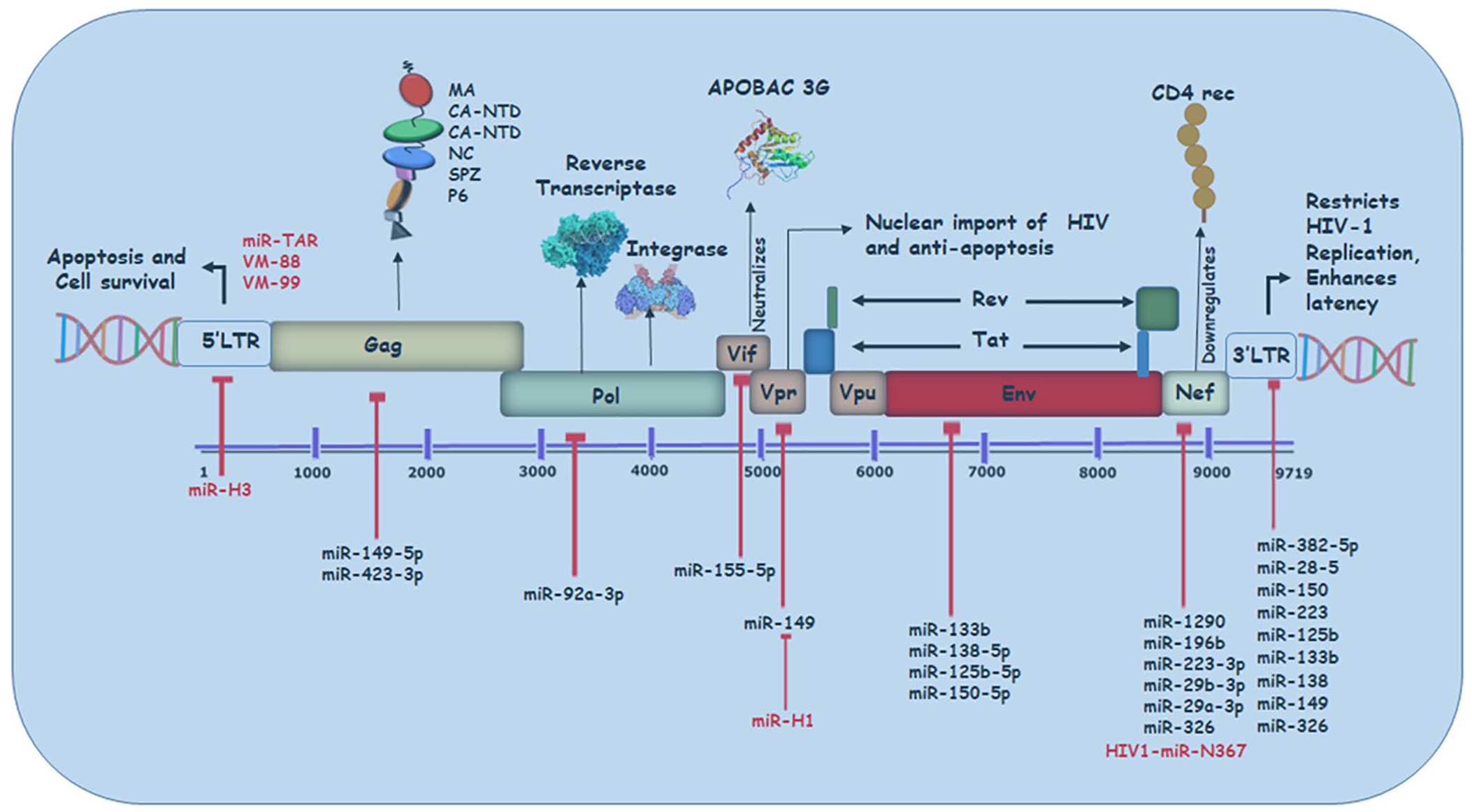
miRNAs targeting HIV genome: Various miRNAs have been reported to interact directly with the HIV genome; however, they impede viral replication upon binding. The binding of those cellular (black) and viral (red) miRNAs within the 5′ or 3′ LTR or the HIV ORF has been depicted along with their respective viral gene targets.
miRNAs in HIV antiviral activities
Given the fact that miRNAs are an intrigued part of cellular mechanisms, hence, viruses tend to hijack and use them for their benefit. 16 However, some miRNAs are found to exhibit antiviral properties by interfering with several pathways inhibiting protein expression or by targeting HIV dependency factors (HDFs).37,39 Multiple studies demonstrated how the dysregulation of miR-7-3p, miRNAs-30, miRNA-125b, miRNA-150, miR-186, miR-210, and miR-222 has a negative impact on viral infection.40–43 These miRNAs were found to target genes of Dicer, NTNG1, EFNB2, CXCL12, or HRB (HIV-1 Rev-binding protein), and HIV-EP2 (HIV-1 enhancer-binding protein 2).40,42 The downregulation of these genes eventually leads to reduce viral expression.40–42 Few other microRNAs exhibiting antiviral activities are listed here (Table 1).25–27,29,31,35,38,44–51
miRNAs exhibiting anti-HIV-1 activity.
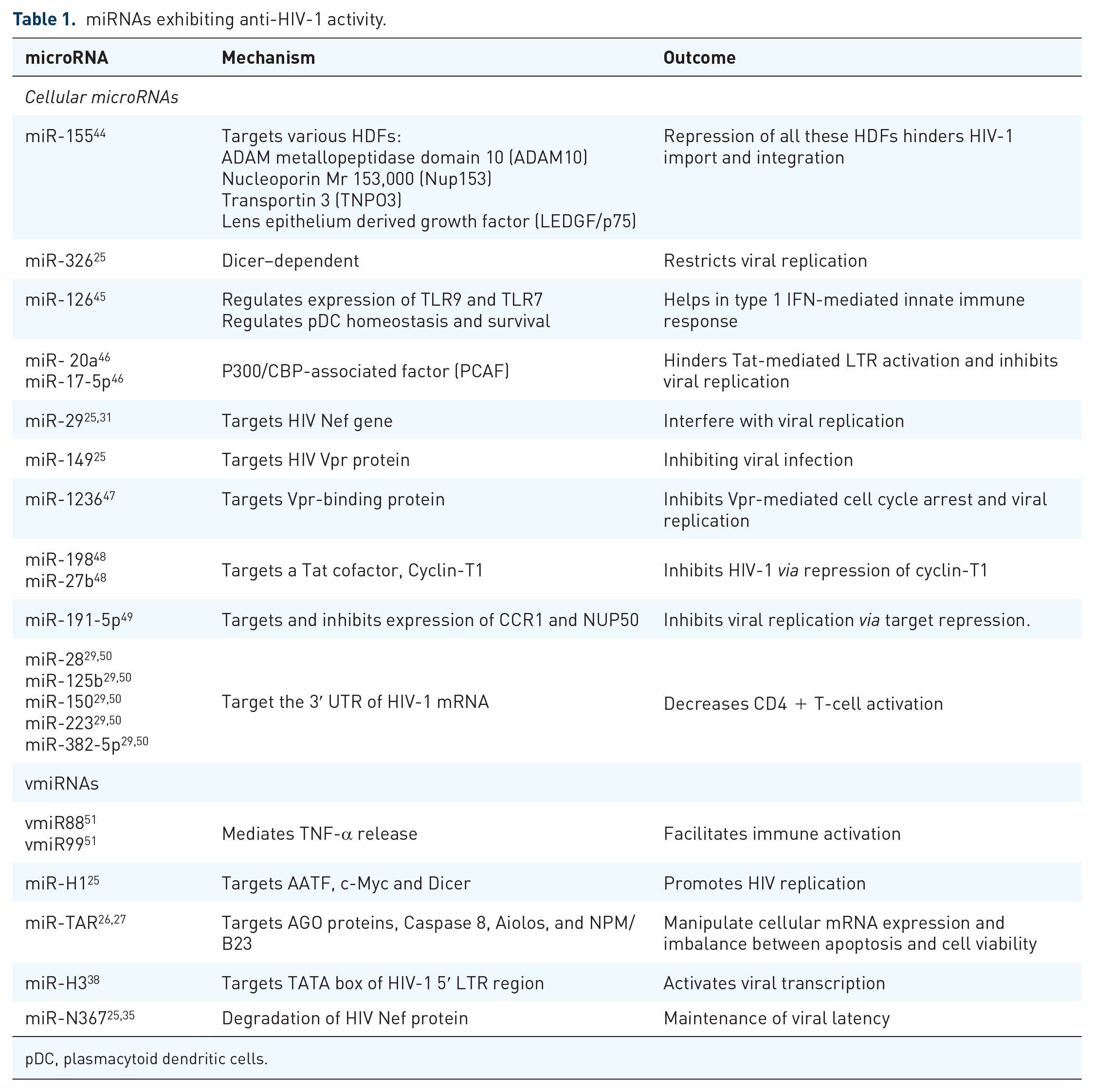
pDC, plasmacytoid dendritic cells.
In addition, there are some other miRNAs, which show antiviral properties by sharing a common target that is Purine-rich element-binding protein alpha. This protein plays a vital role in Tat-mediated LTR activation and hence in viral replication. It has been observed to be targeted by nearly six miRNAs, that is, miR-15a, miR-15b, miR-16, miR-20a, miR-93, and miR-106b. 52
HCV infection and role of miRNAs
HCV entry
HCV tropism is generally found in human and chimpanzee liver cells, allowing the researchers to be suspicious about various intracellular factors favoring HCV entry and infectivity. Few studies had shown the importance of SCARB1, CD81, claudin-1 (CLDN1), and occludin (OCLN) in facilitating viral entry. 53 This was again proven when scientists were successful in creating a mouse model for HCV infection. 54 These factors allowed the HCV to enter, yet no sustained chronic infection was observed later. On the contrary, when scientists allowed miR-122 to be abnormally over-expressed in mouse embryonic fibroblasts, it showed increased subgenomic HCV replication (Figure 4). 55 In contrast, another study unveiled an antiviral property of miR-122 due to its binding efficiency with OCLN. Over-expression of mimic miR-122 resulted in downregulation of OCLN protein by ~80%. Hence, a notable decrease in expression of OCLN in chronic HCV patients can be blamed upon inhibiting the property of miR-122. 56 Along with miR-122, miR-200c is also observed to efficiently target 3′ UTR of OCLN and repress its activity. 57 However, different miRNAs exhibit different effects on HCV entry receptors like CLDN1. The role of miR-155 and miR-182 showed conflicting results in terms of interaction with claudin-1 (Figure 4). miR-182 is claimed to inhibit CLDN-1 mRNA expression in the infected cell line, hampering HCV infection. 58 Nevertheless, a positive correlation between miR-155 expression and CLDN1 is seen in diseases like colorectal cancer. 59 The role of their interaction in HCV infection requires further research. A study on 28 HCV liver biopsies and bioinformatic analysis revealed the inhibitory effect of miR-194 on the CD81 receptor. Successful treatment of miR-194 mimics in different HCV-infected cell lines resulted in decreased expression of CD81 protein (Figure 4). 60 CD81 is an essential tool during the process of HCV viral entry and infection; hence, its inhibition by miR-194 might open up new therapeutic approaches in HCV treatment. In addition, epigallocatechin gallate (EGCG), one of the components found in green tea extract, is known for its antiviral effects against HCV. 61 Along with its other activities, EGCG hinders HCV infection by repressing CD81 expression with the help of miR-194.60,61 Similarly, EGCG is also found to promote expressions of other miRNAs like miR-548a. A recent publication stated that miR-548a consists of 2 binding sites on CD81, providing it a tool to inhibit protein expression. Hence, ECGC and miR-548a can be linked with the host cellular defense mechanism. 62 HCV infection also enhances miR-130a expression, which in turn inhibits endogenous interferon-induced transmembrane protein 1 (IFITM1) expression in a hepatoma cell line (Figure 4). 63 However, on the other side, enhanced expression of the same miR-130a was observed as a result of IFN treatment and claimed for restoring the act of IFN α/β. 64 Future research is needed to study the contradictory nature of this miRNA.
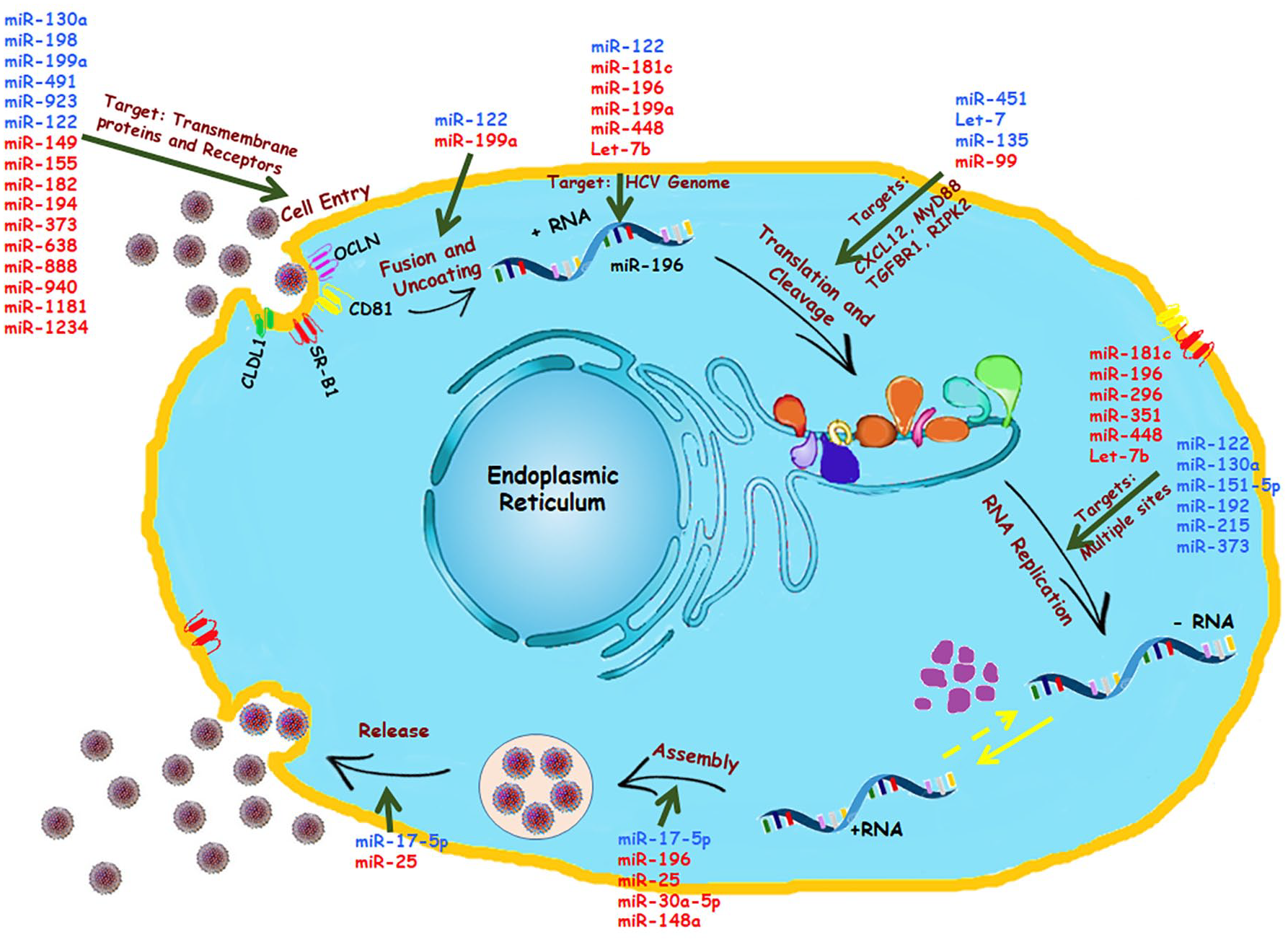
Cellular miRNAs controlling HCV lifecycle: Several miRNAs have been identified for controlling HCV replication and pathogenesis, as illustrated. These miRNAs are known for their targets in the various phases of viral lifecycles: Cell entry, Fusion and Uncoating of viral RNA, Translation and Cleavage of the viral polyprotein, RNA replication, Assembly of the viral genome, and Release of progeny viruses. The upregulated miRNAs are shown in blue, and the downregulated miRNAs are shown in red.
HCV replication
Involvement of various cellular miRNAs in viral replication has always been a field of interest for researchers (Figure 4). Utilizing Bioinformatics to study and identify different miRNAs and their role in HCV infection has also been proven beneficial. With the help of this resource, scientists identified miR-199a-3p to have complimentary seed sequences with domain II of HCV internal ribosomal entry site, which is conserved among all HCV genotypes. Hence, its expression can possibly suppress HCV replication. 65 Similarly, binding sites on NS5B and 5′ UTR of HCV genome for let7-b were also established. This allowed let7-b to act against HCV replication and restrict its accumulation. 66 Ectopic expression of miR-196 is linked with decreased viral load with two possible explanations (1) by targeting Bach1 (2) by targeting NS5a region of HCV genome. 67 Interestingly, Bach1 is believed to be responsible for HMOX-1 downregulation by managing its enhancer availability. 67 Heme oxygenase 1 is a cytoprotective protein with antioxidant properties. It is seen to be underexpressed in patients with chronic HCV infection. However, it has an important role against the infection as its overexpression leads to a subsequent decrease in viral replication.5,67 Similarly, another miRNA induced by IFN, miR-448, targets core sequences of the viral genome and hence holds a negative correlation with viral replication in the cell. 67 In addition, miR-181c levels are observed to be inversely proportional to the proliferation rate of HCV due to its compatibility with E1 and NS5a regions. It inhibits the infection by altering CCAAT/ enhancer-binding protein β (C/EBPβ). 68
miR-122: protagonist or antagonist?
microRNAs are seen to participate in viral infections
Can dysregulation in miRNAs alter crucial pathways for HCV infection?
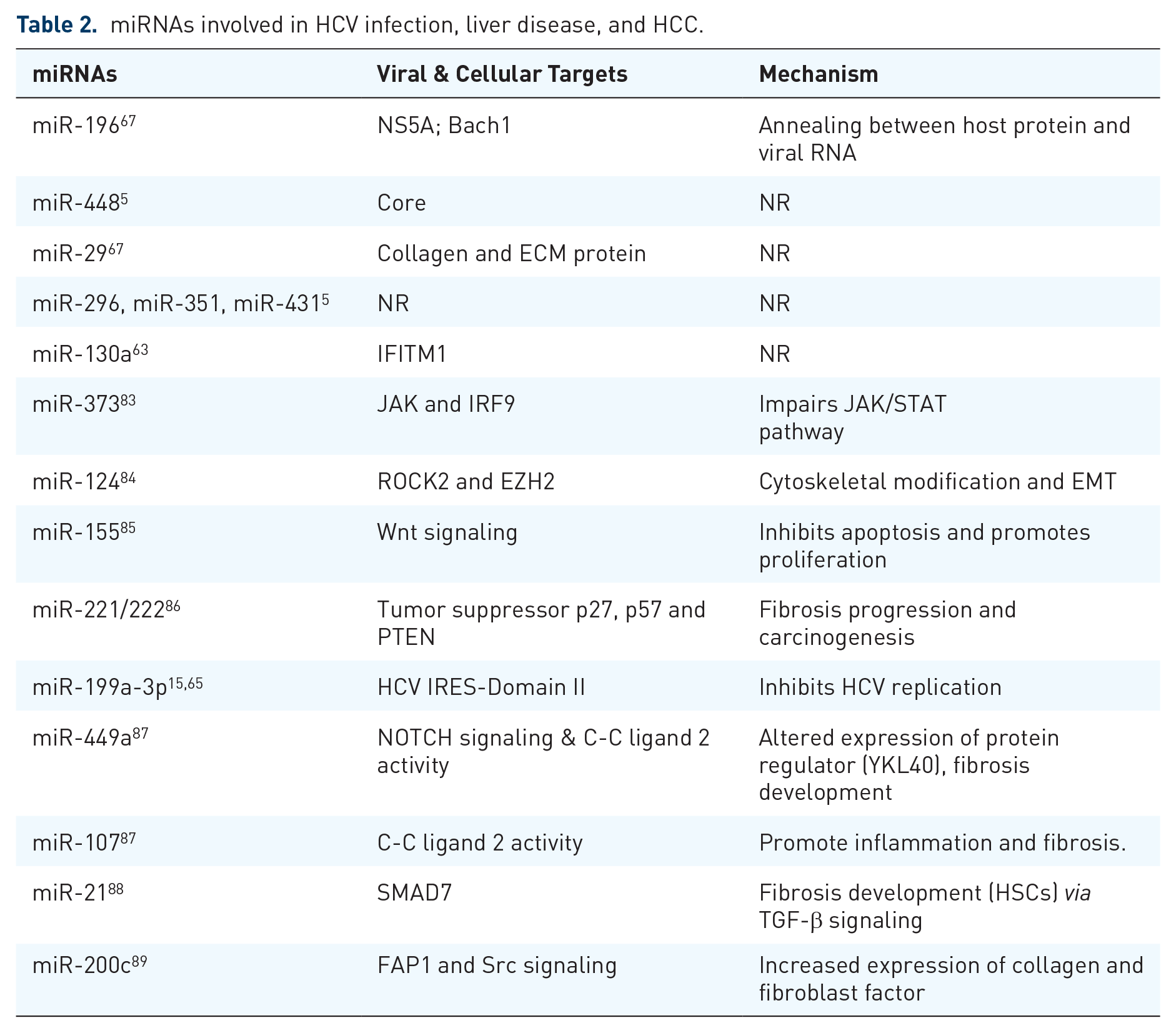
Some miRNAs particularly target host cellular pathways to enhance HCV replication and infection. One such pathway that plays a role in cell proliferation, survival, and growth is PI3k/Akt pathway, targeted by miR-491, miR-320c, and miR-483-3p. Dysregulation of these miRNAs helps elude the immune system and HCV enhancement. 78 IFN system and innate immune response go hand in hand, and miRNAs are also seen to be an essential part of this circle. One of the mechanisms cell uses to fight viral infection involves type-1 interferons, and HCV is seen to successfully defeat this mechanism by using several miRNAs (Table 2).5,67 For instance, miR-122 inhibits IFN signaling pathway to favor HCV replication. Hence, using antisense oligonucleotides of miR-122 to carry out miRNA silencing induced methylation at SOCS3 gene promoter, resulting in a decrease of SOCS3 levels. This decrement then intensified IFN-induced ISRE activity in the host. 79 In addition to miR-122, upregulation of some other miRNAs like miR-758 in infected cells is also responsible for reduced IFN signaling. They carry out this by eliminating ongoing activities of toll-like receptors 3 and 7, 80 while some miRNAs favor immune responses induced by IFN like miR-221 and miR-30 cluster either by targeting SOCS1/3 proteins and genes, respectively.81,82 A study revealed that nearly 30 miRNAs were differentially expressed in response to IFN α, β and γ treatment given to HCV-infected cells. Among which, eight are almost complementary to the seed sequence of the HCV genome.5,67,68 Further studies showed that some miRNAs like miR-196, miR-296, miR-351, miR-431, and miR-448 exhibited antiviral effects by inhibiting viral replication (Table 2).5,15,63,65,67,83–89
miRNAs involved in HCV infection, liver disease, and HCC.
Role of miRNAs in HIV/HCV co-infection
Developing treatments for HIV-1 efficiently promises a healthy life span to patients, while on the contrary, severe liver diseases in HIV/HCV co-infection patients have been proven fatal. 90 Researchers observed that HIV has a significant role in the prognosis of HCV infection. The average time span between HCV-mono-infection and occurrence of cirrhosis is estimated to be 25–30 years usually, while in co-infection, the time span dramatically decreases, and a faster occurrence of cirrhosis or HCC is observed. Clinical data show co-infected patients have increased HCV viral load compared with mono-infected patients. 91 HIV-1 infection is so far claimed to be a reason behind the increment of viral load in co-infected patients.92,93 Although the counter effect of HCV on HIV infection is not significantly seen yet, some studies found HCV to enhance HIV-1 infection in macrophages with the help of different pathways. 92 HCV seems to use TLR2-, JNK-, and MEK1/2-dependent pathways to induce increased HIV viral replication. 94 Along with this, slow recovery of the immune system in co-infected patients is commonly seen compared with mono-infected patients. 95 Alongside, the effect of ART on liver disease progression in co-infected patients is still controversial. Some studies show no significant effect of ART in causing liver damage, with a lesser chance of liver failure, while some claim ART is associated with increased liver transaminase or frank liver decomposition.96,97 As a result, the severity of liver illnesses such as fibrosis and cirrhosis has grown. HIV-1 is significantly different from HCV as it is a retrovirus that majorly targets CD4+ T cells and macrophages. It contains two copies of positive-sense, single-stranded RNA, which encodes nine genes. Unlike HCV infection, HIV RNA undergoes reverse transcription and forms DNA which is then integrated within the host cell genome, 94 while on the other end, HCV RNA directly undergoes replication and transcription without any DNA intermediate. 6 So far, both HIV-1 and HCV infections are seen to have some effect on one another. Along with this, toxicity caused due to various treatments or protein interactions can also be seen as effects that cause increased progression toward liver diseases in co-infection. However, to achieve reliable diagnostic or therapeutic outcomes, the study of underlining mechanisms is necessary. microRNAs are one such non-detachable part of cellular mechanisms. Hence, the role of miRNA in co-infection is an emerging field of research. The scientists have already established several miRNAs and their roles concerning HIV-1 and HCV mono-infection. Although sex-specific biasness has been observed in the miRNA expression of HIV/HCV-co-infected patients, role of some miRNAs is believed to be conserved in the mono-infections and also has their potential contribution in HIV/HCV co-infection 98 (Figure 5).
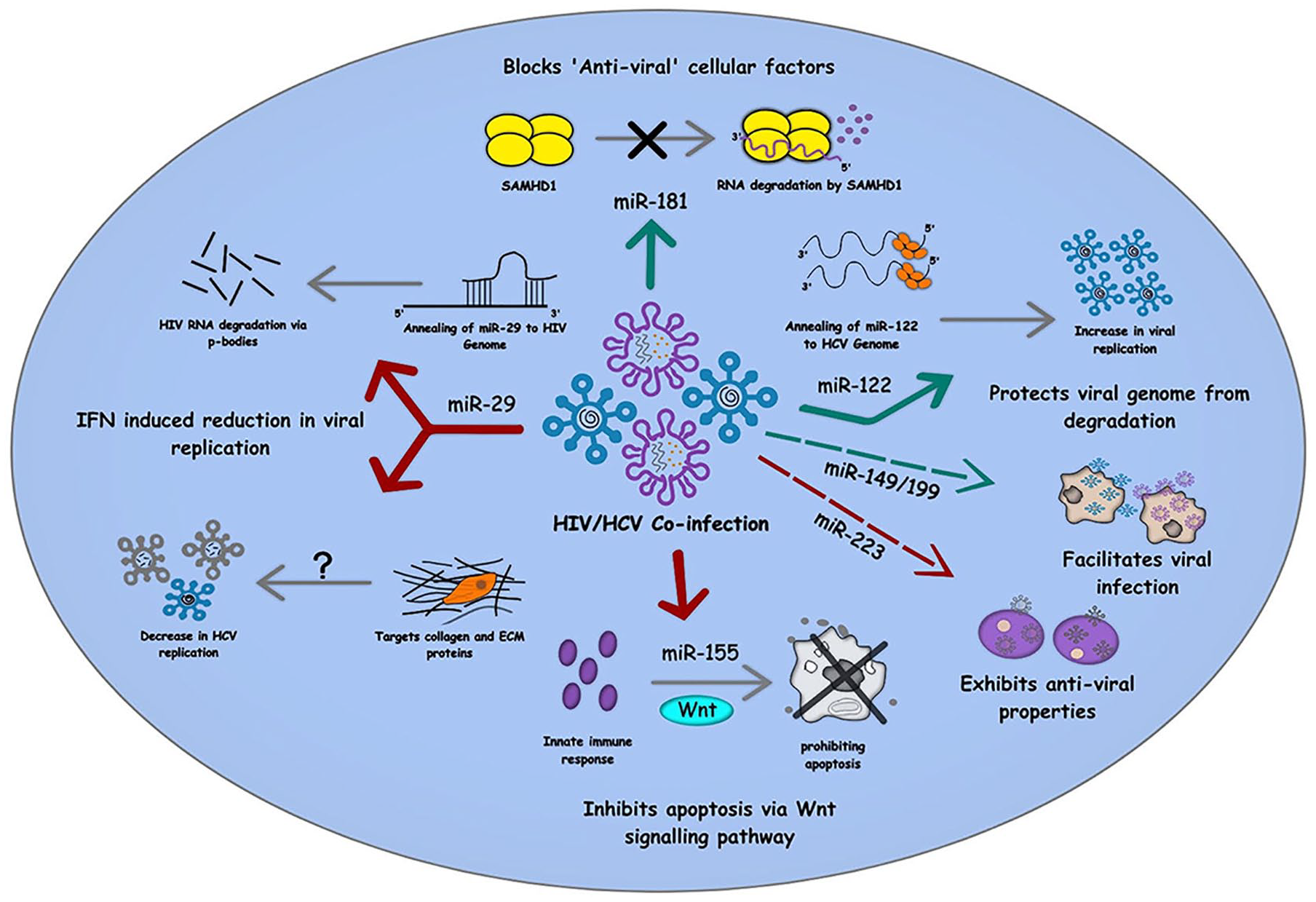
Role of cellular miRNAs in HIV/HCV co-infection: The figure summarizes the involvement of various miRNAs in the different cellular pathways commonly involved in HIV/HCV co-infection. While miR-29 and miR-155 involved in IFN-mediated suppression of viral replication, miR-122 and miR-181 inhibit apoptosis and other antiviral cellular factors to maintain a pro-viral environment within the infected cells. The red arrow denotes the inhibitory effect of miRNAs, and the green arrow represents pro-viral effects, whereas the dashed arrows signify the role of miRNAs that are yet to be examined mechanistically in HIV/HCV co-infection.
miR-122
As mentioned earlier, miR-122 is abundantly found in hepatic cells, which has a significant role in HCV RNA replication. miR-122 having a complimentary seed sequence with HCV gives it an advantage over other miRNAs and thus is suspected of promoting replication by stabilizing HCV genome.70,72,73,99 Hence, upregulation of miR-122 in HCV-infected cells is not new. Its upregulation is also observed in cells infected with HIV-1, though its connection with HIV-1 genome is not yet clear. 100 Thus, an assumption can be placed on the table that HIV-1 infection upregulates miR-122 in various cells like macrophages, which might give HCV an advantage to grow faster in cells other than liver cells during co-infection.100,101
miR-181
Many times, ‘antiviral’ cellular factors are noticed to be often blocked by miRNAs in order to promote viral infection. Likewise, miR-181 is also seen to anneal and inhibit cellular factor SAMHD1, promoting HIV-1 infectivity. It also holds a strong correlation with HCV viral load along with CD4 + T cells. Some studies claim miR-181 to enhance liver fibrosis. As it is found to be upregulated in HIV/HBV co-infection, hence their role in HIV/HCV co-infection can be a part worth exploring. 100
miR-155
As discussed above, its upregulation in HIV, HCV, and HBV can be linked with the defense mechanism of host cells. miR-155 is also reported to inhibit apoptosis
miR-29
Another miRNA with potential therapeutic advantage is miR-29. It is seen to inhibit HIV-1 infection
miR-223
Studies have shown miR-223 to be positively correlated with CD4 T-cell count and negatively correlated with HIV load and are responsible for HIV inactivity.100,104 Alongside, it is also downregulated in untreated HCV patients suffering through HCC. 101 Recently, miR-223 was found to be upregulated in HIV-HCV co-infection, but its significant role in co-infection is yet to be discovered. 100
Some miRNAs are seen to be significantly dysregulated in HIV and HCV mono-infection, but their expression and role in co-infection are not entirely studied (Figure 5). A high-throughput sequencing of smRNAs detected dysregulation of four miRNAs
Based on these data, such miRNAs can possibly have their own significant roles in HIV/HCV co-infection. Hence, allowing us to explore various advantages that they hold.
Biomarkers
All these studies reveal the indispensable role of miRNAs in viral infection and disease progression. Hence, many researchers have been working on the idea of using such miRNAs as diagnostic/therapeutic tools. Altering the expression of miRNAs for disease treatment has so far been a difficult task because of their varied nature in different situations. However, the same nature of being differentially expressed during different disease conditions allows miRNAs to be used as biomarkers. A study covering HIV-HCV co-infected patients serum with significant history of liver injury revealed upregulation of miR-22, miR-34a, and miR-122, 108 whereas another study disclosed miR-99a, miR-100, miR-125b, and miR-192 as the prime culprit for liver fibrosis and disease progression in plasma samples of HIV-HCV co-infected patients. 109 A recent study published in 2021 provided evidences for eight different miRNA that holds the potentiality to become a biomarker for HIV, HCV, or HIV/HCV co-infection. 110 The study included miRNAs like miR-122, miR-155, miR-223, miR-150, miR-199, miR-149, miR-29, and miR-let7.
Similarly, other studies were conducted on differential expression of miRNAs in HCV mono- and HIV/HCV co-infection after liver transplantation. They used liver biopsies of mono- and co-infected patients after 6 months of liver transplantation. These liver samples were affected by HCV but had not yet undergone any severe liver injury like cirrhosis but associated with the processes of viral reinfection, neurologic impairment, and coagulation disorders.111,112 Researchers believed this approach might become helpful in identifying different miRNAs that play a role in the early stage of disease progression.111,112 Results so far claimed miR29b, miR-200c, miR-222, and miR-338-3p can possibly act as biomarkers. Upregulation of miR-200c after transplantation can represent increased chances of reinfection, while downregulation of miR-338-3p signifies a lack of tumor-suppressing activities. 111 In addition, it was stated that the dysregulation of miR-29b and miR-222 might play the important role in impairing the immune response favoring fibrosis in HCV/HIV co-infected patients. 112
Likewise, many different microRNAs are identified as potential biomarkers in liver disease progression representing different pathways/mechanisms.
113
One such is miR-101 which in normal conditions is indulged in prohibiting hepatic stellate cells activation and pro-fibrogenic cytokines release
Conclusion
HIV and HCV are blood-borne infections; hence, this standard route of transmission plays a significant role in increasing cases of co-infection. Virus–host interactions play multiple different mechanisms involving microRNAs. miRNAs being an intrigued part of any cellular process, the study of their dysregulation during infection has always been the cynosure of researchers. Analyzing the role of differentially expressed miRNAs might allow us to comprehend various ongoing complex mechanisms and interactions in HIV/HCV co-infection. This prominent nature can further be used to develop an early-stage non-invasive diagnostic tool for co-infection. However, there are still many controversial questions that remain unanswered. What role does HCV hold in HIV infection? Do HIV-1 microRNAs play any role in HCV infection? What effects do ART or HCV treatment have on disease progression and pathogenesis during a co-infection? Indeed, a lot more research is required for in-depth study of co-infection and the role of miRNAs in it.
Footnotes
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Disclaimer
All the authors have contributed equally to this work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review work has been supported by the Indian Council of Medical Research and ICMR-National AIDS Research Institute, Pune.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Availability of data and materials
Not applicable.
