Abstract
Aims:
Viral hepatitis and tuberculosis are major public health concerns with shared risk factors, especially in marginalized communities. Despite this, routine hepatitis B and C viruses screening in tuberculosis patients is uncommon. This study, conducted in Golestan Province, where tuberculosis and hepatitis B virus have high incidence rates, aimed to assess the rates and prevalence of hepatitis B and anti-hepatitis C viruses testing among active tuberculosis patients.
Materials and methods:
Our cross-sectional study was conducted between March 2018 and March 2023 and included patient records of 2283 tuberculosis cases registered in the database of Golestan University of Medical Sciences. Hepatitis B and anti-hepatitis C viruses were tested among patients with confirmed tuberculosis. Clinical and demographic data were collected by taking patient records and performing structured interviews. Exclusions were limited to patients with a confirmed tuberculosis diagnosis. Patients who did not consent to participate and had incomplete information were excluded from the study.
Results:
Among 2280 tuberculosis patients, 50.1% were male, with a mean age of 46.22 years. Hepatitis B virus surface antigen was detected in 10.57%, and 2.32% tested positive for anti-hepatitis C virus antibodies. Men were more frequently tested for anti-hepatitis C virus positivity than women (62.15% versus 37.85%,
Conclusions:
Tuberculosis patients were more likely to test positive for hepatitis B and anti-hepatitis C viruses than the general population. These results emphasize the need for regular screening and coordinated care for co-infected patients.
Introduction
Globally, chronic hepatitis B virus (HBV) and hepatitis C virus (HCV) infections are major contributors to morbidity and mortality. 1 Hepatitis is liver inflammation, often caused by viral infection. HBV and HCV account for a significant proportion of liver diseases, leading to damage ranging from mild conditions to cirrhosis and hepatocellular carcinoma (HCC).2,3
Despite intensive vaccination efforts that have been continued since the 1990s, HBV continues to be a frequent cause of the worldwide burden of liver disease. According to current epidemiological data, one-third of the world’s population (about two billion individuals) is infected with HBV. Among them, 5%–10% of the adults and as much as 90% of the children become chronic carriers, confining more than 350 million chronic infections throughout the world.4,5 Most HBV infections in high-prevalence regions occur during pregnancy or early infancy. At the same time, in moderate-to-low endemic areas, the disease mainly affects young people and spreads via sexual or parenteral routes of transmission.6,7 On the other hand, HCV infection serves as a primary etiological factor in persistent hepatic disorders and may progress to HCC. 8 Over 80 million individuals worldwide have contracted HCV, which is associated with high morbidity and mortality rates.9,10 The economic and healthcare burden imposed by both hepatic and extra-hepatic complications significantly hinders progress in treatment and recovery. 11
The
Patients in high-prevalence regions often present with dual HBV and TB infections. These coinfections substantially amplify liver toxicity risks from TB medications, frequently demanding therapy cessation. 17 The hepatotoxic effects are three- to five-fold greater than in TB-only cases, with HIV/HCV coinfected individuals facing a dramatic 14-fold increased risk. 18 Although HBV and HCV infections share similar clinical presentations during initial and prolonged phases, HCV demonstrates a markedly higher propensity for chronic progression. 19 Among TB patients, the frequency of persistent HBV infection ranges greatly from 0.5% to 44%.20,21 Among TB patients, the prevalence of HCV infection ranges from 1.0% to 44.6%, with variations depending on geographic location, population characteristics, and risk factors associated with co-infection.22,23 Drug-induced liver injury (DILI) is known to be caused by HBV and/or HCV infections. However, during anti-TB therapy, additional factors such as high alcohol consumption, malnutrition, advanced age, and female sex have also been identified as significant contributors to DILI development, further complicating patient management.24,25 Co-occurring HBV infection and TB are associated with worse treatment response and patient outcomes. 26 The presence of HBV in TB patients is capable of exacerbating DILI from antitubercular medications, creating clinical scenarios where treatment cessation becomes medically necessary to prevent severe hepatic complications. 27 Individuals with HBV infection and concurrent TB are more prone to the hepatotoxic effects of medications. Preexisting hepatic injury from viral hepatitis exacerbates medication sensitivity, rendering the liver more prone to toxic damage from anti-TB drugs.28,29 Guidelines issued by the WHO consequently advise viral hepatitis screening for TB patients displaying HIV coinfection, hepatic enzyme abnormalities, or recognized exposure risks.30,31
The presence of TB alongside HBV or HCV co-infection is crucial for managing patients, as it impacts the selection of TB medications and raises the risk of adverse treatment effects. Furthermore, diagnosing hepatitis during patient care can prompt HCV treatment initiation, potentially preventing future complications from chronic viral hepatitis. 32 Limited research has been conducted on the association between TB and HCV and/or HBV infections. However, this relationship is physiologically plausible, as recent studies suggest that HCV infection may increase the risk of developing active TB due to its immunomodulatory effects. 33
This study aimed to assess the prevalence of HBV and anti-HCV positive infections among TB patients in Golestan Province, located in northeastern Iran, which has a higher TB incidence compared to the national average. However, data show that the incidence of HBV cases in Golestan is significantly higher than in other regions, positioning the province as a hotspot for HBV infection in the country. Golestan also reports the highest prevalence of both HBV and HCV infections in Iran.
Methods
Patients
We used a cross-sectional approach to estimate the seroprevalence of HBV and HCV among TB patients in Golestan Province, Iran. The study examined clinical differences between patients with only TB and those co-infected with viral hepatitis. Data spanning 5 years (March 2018–2023) were sourced from Golestan University’s TB surveillance system. The registry included 22,183 diagnosed TB cases from outpatient and inpatient settings. All records contained verified demographic and clinical information, with TB diagnoses confirmed according to Iran’s national standards. Data collection involved trained clinical nurses conducting detailed reviews of medical records and structured patient interviews. The study received ethical approval from Golestan University’s review board prior to commencement, ensuring adherence to human research ethics. Participants aged 18 or older provided written consent; minors under 18 required written permission from a Legally Authorized Representative (e.g. parent or guardian), along with verbal assent when possible. Consent covered access to medical records, structured interviews, and use of anonymized data for research purposes. No participant was enrolled without proper consent, demonstrating our commitment to ethical standards regardless of infection status. Confidentiality was rigorously maintained, with protocols in place to safeguard data privacy. Inclusion criteria required a confirmed TB diagnosis in line with WHO standards. After TB confirmation, we evaluated HBV infection status, anti-HCV antibody presence, and demographic data from patient records. Participants who refused participation, had incomplete records, or lacked a confirmed TB diagnosis were excluded.
Data analysis
Statistical analysis was performed using IBM SPSS Statistics version 22.0 (IBM Corporation, Armonk, NY, USA). For continuous variables, independent samples
Results
The demographic characteristics of the 2283 confirmed TB patients are summarized in Table 1, stratified by HBV and anti-HCV infection status. The study population comprised patients with TB, with an average age of 49.78 ± 20.32 years, predominantly male (53.39%), and the majority were married (65.87%). Of the TB cases screened during the 5 years, 64.34% (
Basic demographic and clinical characteristics of participants.
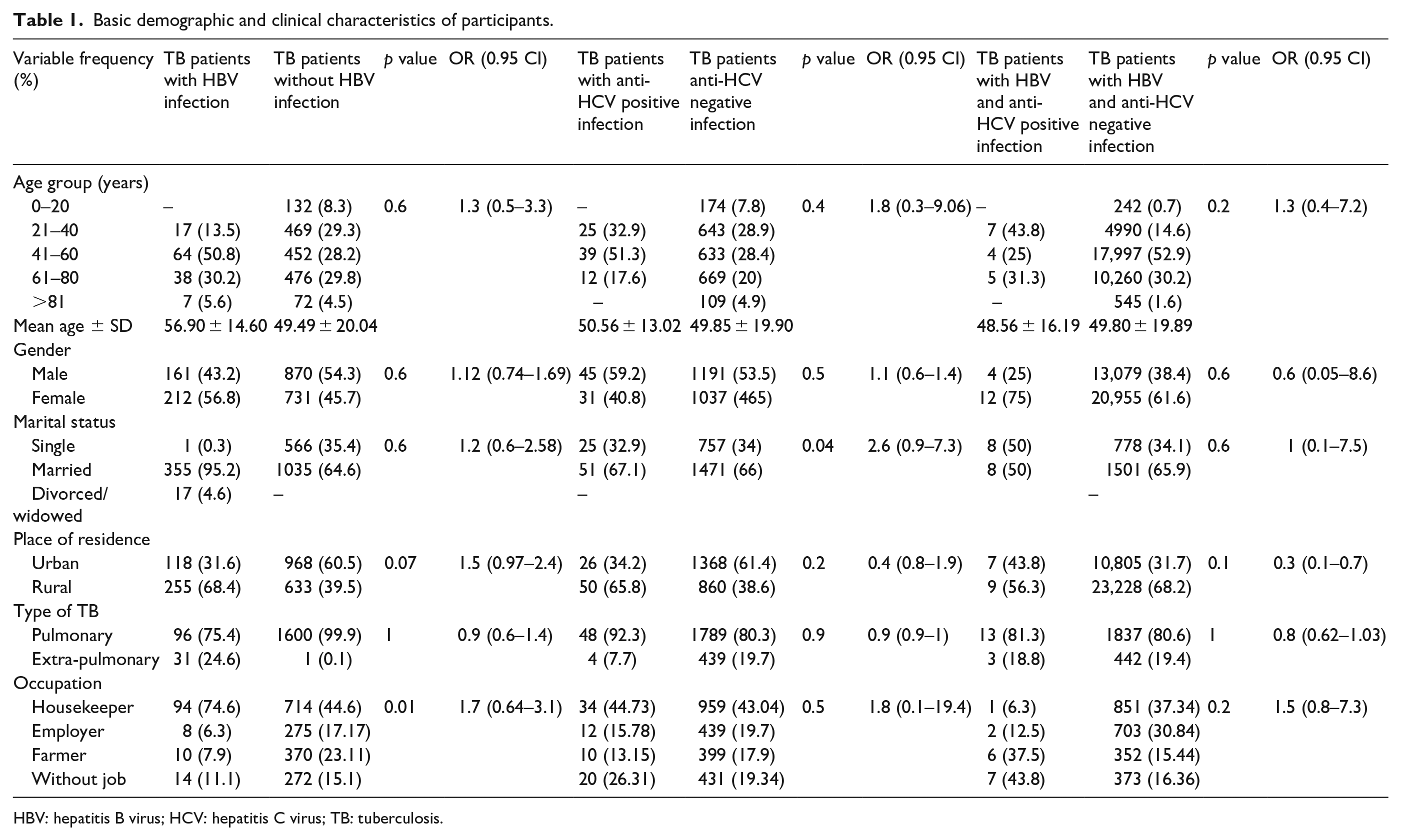
HBV: hepatitis B virus; HCV: hepatitis C virus; TB: tuberculosis.
Among TB patients with documented HBV infection, 50.8% were in the 41–60 age range, with a median age of 56.90 ± 14.60 years. Of these patients, 75.4% had pulmonary TB, 68.4% resided in rural areas, and 56.8% were female. The following factors showed a positive correlation with HBV/TB co-infection: place of residence (
Of the co-infected patients, 68.2% resided in rural areas, and 61.6% were female, suggesting potential regional and demographic patterns in the incidence of the condition. Pulmonary TB was the most common form, affecting 80.6% of the co-infected individuals. Correlation analysis revealed a positive relationship between age group and anti-HCV positive/HBV/TB co-infection (
Consistent with the multigenerational hepatitis study by Naderi et al.,
33
which evaluated HBV infection and anti-HCV seropositivity across family units (including 30 three-generation and 40 two-generation cohorts), our investigation extended this approach to examine concurrent TB infections within similar demographic parameters. The analysis revealed TB co-infection rates of 6.7% (
Additionally, 7.5% (3/40) of intrafamilial HBV patients and 20% (8/40) of mothers and children among these Chronic hepatitis B (CHB) patients had concurrent anti-HCV testing results. Furthermore, 12% (5/40) of patients with anti-HCV/HBV were from two generations, and 5% (2/40) of intrafamilial patients also had TB infection.
Our findings demonstrated that all patients co-infected with HBV and TB tested positive for HBc-Ab, suggesting either a resolved infection or prior exposure to HBV. Additionally, these patients tested negative for HBs-Ab, indicating susceptibility to HBV infection. The presence of HBsAg indicated that all subjects had been infectious for at least 6 months prior. Laboratory research showed a consistent HBeAb+/HBeAg− serological pattern among HBV–TB coinfected cases. Comparative analysis between HBV-infected groups with and without TB coinfection showed no significant variation in these marker profiles (
Liver function test findings revealed that mean LFT levels were higher in HBV/anti-HCV positive/TB co-infected patients compared to HBV/anti-HCV/TB negative patients. A statistically significant correlation was observed (
Discussion
The global burden of viral hepatitis continues to affect hundreds of millions of patients, maintaining its position among the world’s most pressing health concerns. The prevalence of liver infections caused by the HBV and HCV is rapidly increasing in the general population, particularly among TB patients.
33
HBV co-infection increases the likelihood of treatment failure in TB patients,
34
may trigger the reactivation of latent TB, and can lead to drug-induced liver damage.24,35 Given the potential coexistence of HBV and TB in endemic regions, some studies have proposed that screening for HBV in TB patients could improve treatment outcomes.
36
Sometimes, interruptions in treatment may be required due to the heightened risk of hepatitis caused by the hepatotoxicity of anti-TB medications.
37
The majority of individuals who were co-infected with TB resided in rural regions. Among TB patients, 10.57% tested positive for HBV, and 2.32% tested positive for anti-HCV. A statistically significant increase in viral loads (
According to a study conducted by Ahmadi Ghezeldasht et al., who identified a single HBV genomic region, the overall prevalence of occult HBV infection (OBI) among TB patients was 33.3%. More than half of the OBI/TB patients had both HBc and HBs antibodies, with 61.5% being negative for HBc-Ab and 38.5% being seropositive.
36
Individuals with both TB and HBV had worse outcomes compared to those without HBV, according to a study by Khan et al. Co-infected individuals were more likely to develop severe TB and required retreatment more frequently.
14
Consistent with a study performed by Khan et al., this study examined HBV blood markers in CHB patients who tested either positive or negative for TB in both three and two consecutive generations. The findings revealed that all patients with HBV/TB co-infection tested positive for HBc-Ab and negative for HBs-Ab. The HBV/TB cohort exhibited 1.5–2.3-fold higher mean liver enzyme values (ALT, AST, and ALP) versus HBV+/TB− participants, with all comparisons showing statistically significant differences (
Additionally, patients with HBV/anti-HCV positive co-infection and TB had the highest viral load compared to those with HBV infection alone (
Malden et al. reported that nearly one-quarter (23.1%, 461 cases) of 1997 chronic HBV patients screened positive for latent TB infection, corroborating earlier findings by Al-Khazraji et al. These findings further reinforce the need for enhanced HBV and TB screening, especially for individuals with both infections. 43 In another study by Getie et al., only 3.4% of the 145 TB patients tested positive for HBs-Ag, and none of them tested negative for anti-HCV. 11 Our recent screening of 2283 TB-positive individuals to determine the incidence of co-infections with HBV and anti-HCV revealed that 2.5% of TB patients tested positive for anti-HCV, suggesting a history of chronic HCV infection, while 12.31% of TB patients were also HBV-positive. Triple co-infection with HBV, anti-HCV, and TB was found in 7% of TB patients. These findings underscore the importance of frequent HBV and anti-HCV testing in TB patients to ensure prompt intervention and comprehensive care for co-infected individuals. Despite the detection of anti-HCV antibodies, the frequency of HBV among TB patients remained relatively high, suggesting a stronger epidemiological link between HBV and TB co-infection.
The analysis of mortality risk factors in TB–HBV coinfected patients revealed several significant associations. Cirrhosis was present in half of the deceased cases compared to only 15.4% of survivors (
Among 429 TB patients studied by Nooredinvand et al., 14.2% tested positive for isolated anti-HBc, 2.6% for HBsAg, and 1.6% for HCV antibodies. Active TB cases demonstrated significantly greater seroprevalence of chronic viral hepatitis markers (HBsAg+/anti-HCV+: 5.93%, 16/270) than latent TB infections (1.26%, 2/159), with this difference reaching statistical significance (
Limitations
Although I utilized all the information available in the Patient Detection Data Bank of Golestan University of Medical Sciences, which included 22,183 TB patients, no calculation or justification was provided for the sample size selected in this study. Moreover, the clinical database lacked sufficient details regarding the procedures or the names of the laboratory kits used for detecting HBV, TB, and anti-HCV.
Conclusion
The WHO has recognized TB as a significant global public health challenge over the past three decades. Despite extensive efforts to control its spread, TB remains prevalent, particularly in low- and middle-income countries where healthcare access and preventive measures are limited. Some reports suggest that TB patients should undergo HBV and HCV testing, as the prevalence of HBV and, in some cases, HCV infection is relatively high in TB patients, particularly in endemic regions. Such testing could enhance TB management and treatment outcomes. The findings of the aforementioned studies emphasize the importance of regularly monitoring liver function and HBV DNA levels in TB patients with chronic HBV infection during anti-TB therapy. This approach facilitates the early detection of liver complications and enables timely intervention. To optimize treatment safety, individuals with elevated HBV replication (DNA >104 IU/mL) should receive at least 2 weeks of antiviral therapy before starting anti-TB medications, creating a protective window against potential hepatotoxic effects. Also, screening for OBI in TB-positive individuals is recommended to provide a more comprehensive understanding of TB/HBV co-infection.
Footnotes
Acknowledgements
The authors thank all those who contributed to the study.
Ethical considerations
The Ethics Committee of Golestan University of Medical Sciences approved the current study with the Ethics code of IR. GOUMS.REC.1399.105.
Consent to participate
Informed consent was not sought for the present study for all participants because “participants were selected from the Patient Detection Data Bank of Golestan University of Medical Sciences, which included 22,183 TB patients with a clinical diagnosis, either from outpatient or inpatient facilities. The data bank contained clinical and demographic information, with tuberculosis diagnoses based on national protocols. Data were collected through document review. Clinical nurses were recruited to conduct interviews and gather the necessary information from medical records.” After that, participants who have tested positive for HBV and anti-HCV (we found via data bank) were referred to the appropriate healthcare providers. As a result, we prepared written informed consent from these participants before the study.
Author contributions
Writing the article: A.M., S.M.H., M.N.; conceiving and planning the experiments: A.M., S.M.H., M.N.; data analysis: V.H.S., S.A.S.; reading and approving the final version of the article: A.M., S.M.H., M.N.; revision: A.M., S.M.H., M.N.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available upon reasonable request from the corresponding author.
