Abstract
Objective
Unilateral hypoglossal nerve stimulation (uniHNS) is an effective treatment for obstructive sleep apnea. Bilateral hypoglossal nerve stimulation (biHNS) is a novel therapeutic option and a different approach to hypoglossal nerve stimulation. The aim of this study was to analyze the clinical outcome of the first 10 biHNS cases vs the first 10 uniHNS cases ever implanted.
Study Design
Prospective data analysis.
Setting
International multicenter comparative clinical trial.
Methods
The first 10 patients in 2020 who received a biHNS device (Genio System; Nyxoah) and the first 10 patients in 2014 who received a uniHNS system (Inspire II; Inspire Medical Systems) were included. Treatment outcome was evaluated at 3 months after surgery. Data collection included demographics, apnea hypopnea index (AHI), oxygen saturation and desaturation index, Epworth Sleepiness Scale, and adverse events.
Results
The mean ± SD age was 52.1 ± 9.6 years (biHNS) and 58.3 ± 8.6 years (uniHNS). The mean body mass index was 26.4 ± 5.6 kg/m2 (biHNS) and 26.2 ± 2.2 kg/m2 (uniHNS). The mean preimplantation AHI (biHNS, 39.9 ± 14.8/h; uniHNS, 32.2 ± 11.0/h) decreased in both groups after 3 months (biHNS, 19.2 ± 14.0/h, P = .008; uniHNS, 13.1 ± 16.8/h, P = .037) with no significant difference between groups (P = .720). The mean preimplantation Epworth Sleepiness Scale (biHNS, 11.8 ± 6.2; uniHNS, 11.1 ± 4.9) decreased as well after 3 months (biHNS, 9.4 ± 6.3; uniHNS, 6.0 ± 5.0).
Conclusion
Preliminary postmarket data suggest that biHNS may be as safe and effective as uniHNS. Long-term follow-up in a larger sample size is required to assess the stability of biHNS.
Keywords
Obstructive sleep apnea (OSA) is the most prevalent sleep-related respiratory disorder, characterized by upper airway narrowing and collapse during sleep, causing obstructive hypopneas or apneas. Untreated sleep apnea results in reduced quality of life and is a risk factor for cardiovascular and metabolic diseases, such as insulin resistance, dyslipidemia, vascular disease, and increased mortality.1-4 The effectiveness of first-line therapy, continuous positive airway pressure (CPAP), is dependent on patients’ adherence to the treatment, which varies from 50% to 70%,5,6 resulting in poor therapy efficacy (34.1%) as estimated in a recent study. 7
In the treatment of CPAP-refractive OSA, short- and long-term research has suggested that unilateral hypoglossal nerve stimulation (uniHNS) is efficacious in reducing subjective and objective parameters, is associated with long-term compliance and few adverse events, and is well tolerated. 8 As a consequence, uniHNS has been adopted as standard of care in everyday clinical practice. The surgical procedure, implanted device, results, and complications have been reported extensively.9,10 Since the first successful implantation of a uniHNS system in 2001, 3 uniHNS systems have been CE marked (European Commission), all differing slightly.
Recent developments have seen the introduction of the first bilateral hypoglossal nerve stimulation (biHNS) system. It differs favorably from uniHNS devices, as it does not require any leads to connect the electrodes to an implantable pulse generator and only 1 incision is required without any tunnelling, instead of 2 or 3. Furthermore, stimulation is delivered bilaterally and controlled from an externally worn unit that activates a small implanted battery-free submental stimulator at a predetermined adjustable rate and duty cycle.
The pilot study (BLAST OSA) of the first biHNS system reported similar results, namely a reduction of OSA severity and improvement in quality of life without device-related complications. 11 The BLAST OSA trial results demonstrated the safety and efficacy of biHNS therapy, which can be replicated in commercial real-world settings. 11
The aim of this study was to analyze the clinical outcomes of the first 10 biHNS cases with the first 10 uniHNS cases with moderate to severe OSA in our clinical centers in Munich (Germany), Antwerp (Belgium), and Amsterdam (the Netherlands).
Methods
We performed a prospective multicenter observational study. Data were collected retrospectively from the first 10 patients who received a uniHNS implant in 2014 (Inspire II Upper Airway Stimulation System; Inspire Medical Systems), while data were collected prospectively from the first 10 patients who received a biHNS implant in 2020 (Genio System; Nyxoah). Data were pooled and analyzed from 3 participating European centers: Klinikum rechts der Isar, Technische Universität München, Munich, Germany; OLVG, Amsterdam, the Netherlands; and Antwerp University Hospital, Antwerp, Belgium. Following the ethical guidelines, written informed consent was obtained from the subjects to collect and analyze clinical data, and the study was approved by the ethics committee and institutional review board of Klinikum rechts der Isar. Data were pooled and analyzed from the first 10 patients who received a uniHNS implant in 2014 (Inspire II Upper Airway Stimulation System; Inspire Medical Systems).
For both systems, the eligibility criteria for enrollment were an apnea-hypopnea index (AHI) between 15/h and 65/h, as well as nonadherence to CPAP therapy and <25% central and mixed AHI of the total AHI. Patients were nonadherent if they were unwilling to proceed with CPAP despite multiple attempts to use it for several days. Patients with a body mass index >35 kg/m2 prior to surgery were excluded. For both systems, preoperative screening included an 18-channel in-patient polysomnography (PSG), clinical examination, and drug-induced sleep endoscopy 12 to exclude patients with complete concentric collapse of the soft palate. For biHNS, the cricomental space—defined as the distance between the neck and the bisection of a line from the chin to the cricoid membrane when the head is in a neutral position—should be >0 cm. For assessing daytime sleepiness, the Epworth Sleepiness Scale was used. Patients were excluded if they had pronounced anatomic abnormalities identified during clinical examination that could prevent the effective use of HN stimulation (eg, enlarged tonsils).
Surgical Procedure and Follow-up for UniHNS
The selected patients underwent surgery under general anesthesia to implant the uniHNS according to the earlier described surgical technique.9,13 Briefly, a horizontal upper anterior neck incision is used to place the stimulation lead around the protruding fibers of the hypoglossal nerve (HN) on the right-hand side of the neck. A sensing lead for breathing cycle–dependent simulation is placed on the lateral chest wall. Finally, the stimulation and sensing leads are tunneled to the implantable pulse generator, which is placed in a pocket through an incision inferior to the clavicle on the right chest wall. 12
The device was activated after 1 month postsurgery. Standard settings were used to perform the nocturnal acclimatization, and patients had a titration PSG 2 months postsurgery. A second PSG was carried out at 3 months postsurgery to gain insights into therapy effectiveness 1 month after the polysomnographically controlled titration.
Figure 1 summarizes and compares the different timelines of the investigations for both systems.
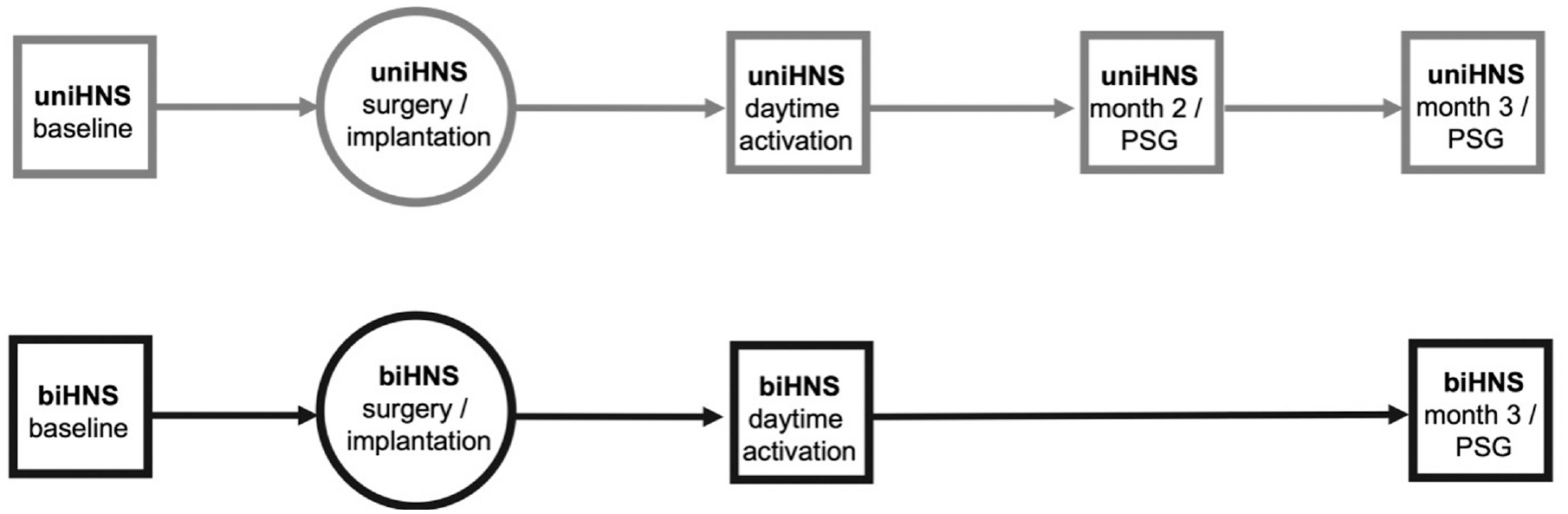
The treatment pathways for patients with uniHNS and biHNS. biHNS, bilateral hypoglossal nerve stimulation; PSG, polysomnography (in sleep laboratory); uniHNS, unilateral hypoglossal nerve stimulation.
Surgical Procedures and Follow-up for BiHNS
All patients underwent surgery under general anesthesia to implant biHNS through a single submental incision. 14 A nerve integrity monitoring system (NIM 3.0; Medtronic) with 2 electrodes in the tongue (1 on each side) and 2 electrodes in the floor of the mouth (paramedian left and right) was used to perform selective placement of the paddle electrodes. 13 To capture activation of the genioglossus muscles intraoperatively, an electromyographic electrode was placed on either side of the floor of the mouth. To exclude retractor branches of the styloglossus and hyoglossus muscles, an electrode was placed 5 cm from the tip of the tongue on each side around the corner of the tongue body. A submental midline incision was performed to place the implantable stimulator. A skin incision was followed by a subplatysmal flap from mentum to hyoid bone to divide the mylohyoid muscle in midline and to identify and lateralize the vertical fibers of the geniohyoid. In the next step, the genioglossus muscles were visualized, and the lateral border of the genioglossus and hyoglossus were identified to explore the HN. A NIM-controlled separation of the protrusion and retrusion nerve fibers was performed to form a pocket for the paddle electrodes of the implantable stimulator on each side. Once the implant was placed into position, the implantable stimulator was sutured to the genioglossus muscle bellies on either side. With the help of an external stimulator, the correct placement of the paddle electrodes on the protruding nerve fibers of the genioglossus was tested intraoperatively, and tongue motions were visualized. Once a satisfactory response was obtained, skin sutures were used to close the incision. Six weeks after surgery, the device was activated with standard settings, and an acclimatization period for the patients ensued in the following weeks. After 3 months postsurgery, a titration PSG was performed.
For biHNS, an activation chip is placed under the chin with an adhesive patch to externally provide battery and control electronics to the passive implant ( Figure 2 ). The activation chip needs to be charged on a daily basis.
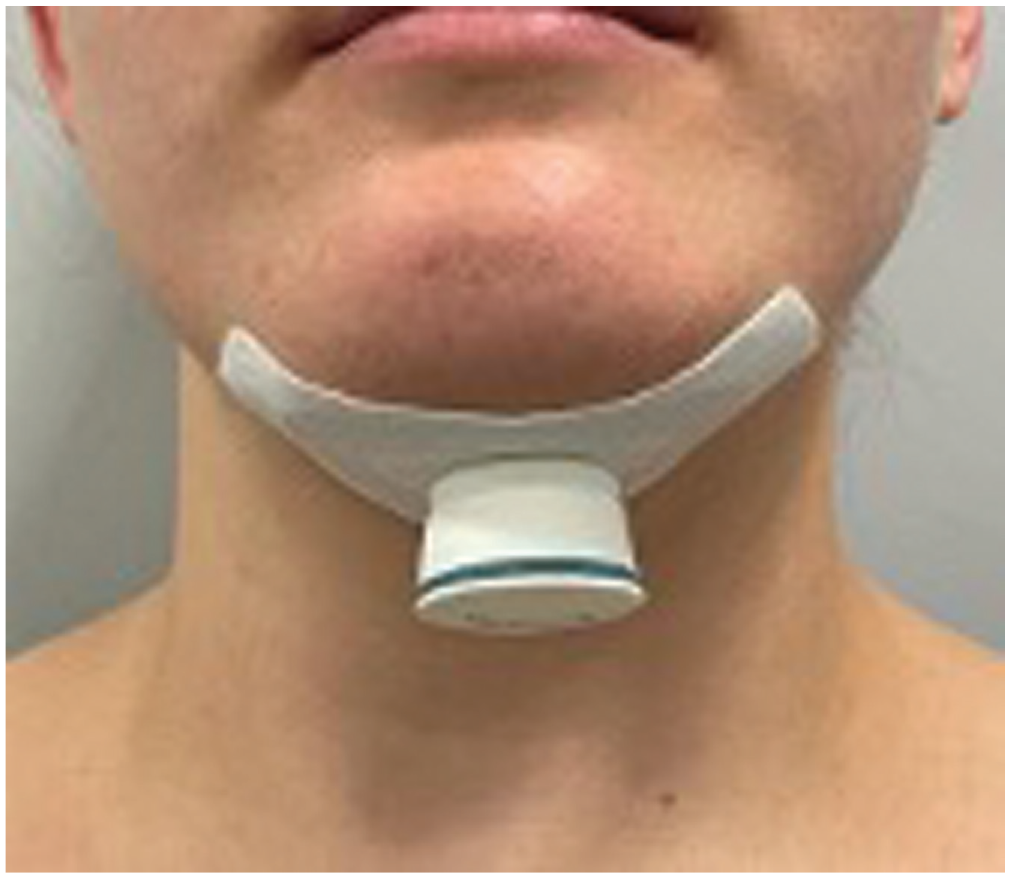
A submental stimulator without battery has been implanted to receive the energy and signals from the external unit, which includes a battery and electronic chip.
Data Collection and Questionnaire for UniHNS and BiHNS
All patients who underwent uniHNS or biHNS surgery received an 18-channel PSG according to the American Academy of Sleep Medicine guidelines from 2012 at screening and baseline. The titration nights for uniHNS at month 2 postimplant and for biHNS and uniHNS at month 3 were also performed as PSGs. The same scoring criteria were used for all sleep studies: hypopneas were scored on 30% airflow reduction and 4% oxygen desaturation, whereas apneas were scored on 90% reduction in airflow. 15 The data for respiratory parameters such as AHI and oxygen desaturation index were collected not as titrated parameters but as parameters from the whole night. Patient-reported outcomes included the Epworth Sleepiness Scale.
Statistical Analysis
SPSS 28.0 software (IBM) was used to perform data analysis. Descriptive statistics and Mann-Whitney U test were used for calculation and comparison of demographic variables and parameters between uniHNS and biHNS. A Wilcoxon test was used to compare baseline and postimplantation values for uniHNS and biHNS. Data are reported as median (interquartile range) and mean ± SD. P≤ .05 was considered statistically significant.
Results
Patients’ characteristics are summarized in Table 1 . In both groups, patients had moderate to severe OSA with daily symptoms such as sleepiness as measured with the Epworth Sleepiness Scale. The CPAP therapy of all patients had failed. There were no female patients in the uniHNS cohort, while 2 female patients were implanted in the biHNS group. Patients in the uniHNS group were slightly older, while the OSA tended to be more severe in the biHNS group before surgery. However, both observations were not statistically significant.
Patients’ Characteristics at Enrollment and Implantation in Both Groups. a
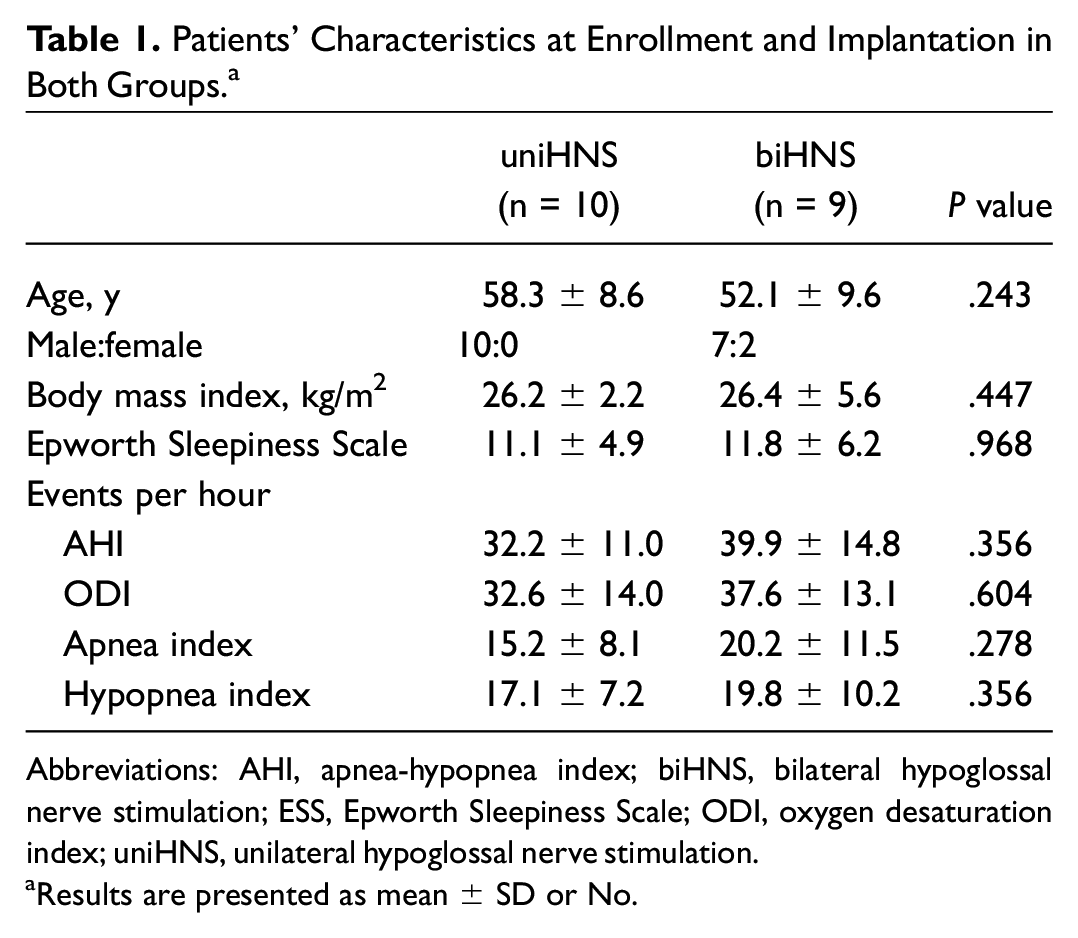
Abbreviations: AHI, apnea-hypopnea index; biHNS, bilateral hypoglossal nerve stimulation; ESS, Epworth Sleepiness Scale; ODI, oxygen desaturation index; uniHNS, unilateral hypoglossal nerve stimulation.
Results are presented as mean ± SD or No.
One patient in the biHNS group dropped out due to COVID-19 infection.
Surgical Implantation
In both groups, no adverse and severe adverse events were reported during and after implantation. The surgery was successfully performed with good perioperative stimulation testing in all patients.
Respiratory Outcome Parameters
A significant reduction in AHI was observed in the uniHNS and biHNS groups. Coincidentally, both groups showed the same mean reduction of 52% ( Table 2 ). Apneas as well as hypopneas were reduced significantly within the groups without any statistical significant difference between them.
Results From All 19 Subjects in Both Groups at Month 3. a
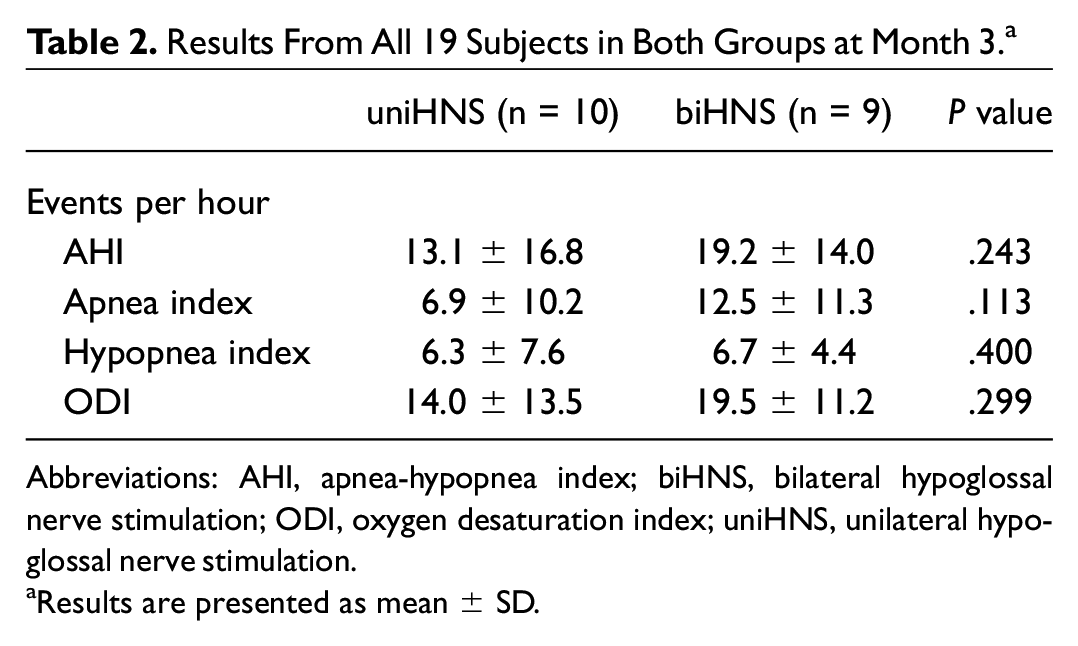
Abbreviations: AHI, apnea-hypopnea index; biHNS, bilateral hypoglossal nerve stimulation; ODI, oxygen desaturation index; uniHNS, unilateral hypoglossal nerve stimulation.
Results are presented as mean ± SD.
Reduction of Respiratory Outcome Parameters
Calculating the absolute average AHI reduction (Δ= average AHI at baseline visit – average AHI at month 3) resulted in each group showing a statistically significant reduction for its therapy ( Table 3 , Figure 3 ). Regarding oxygen desaturation index reduction, biHNS seemed to be more effective than uniHNS.
Average Reduction: Difference Between Baseline AHI and Total-Night AHI at Month 3. a
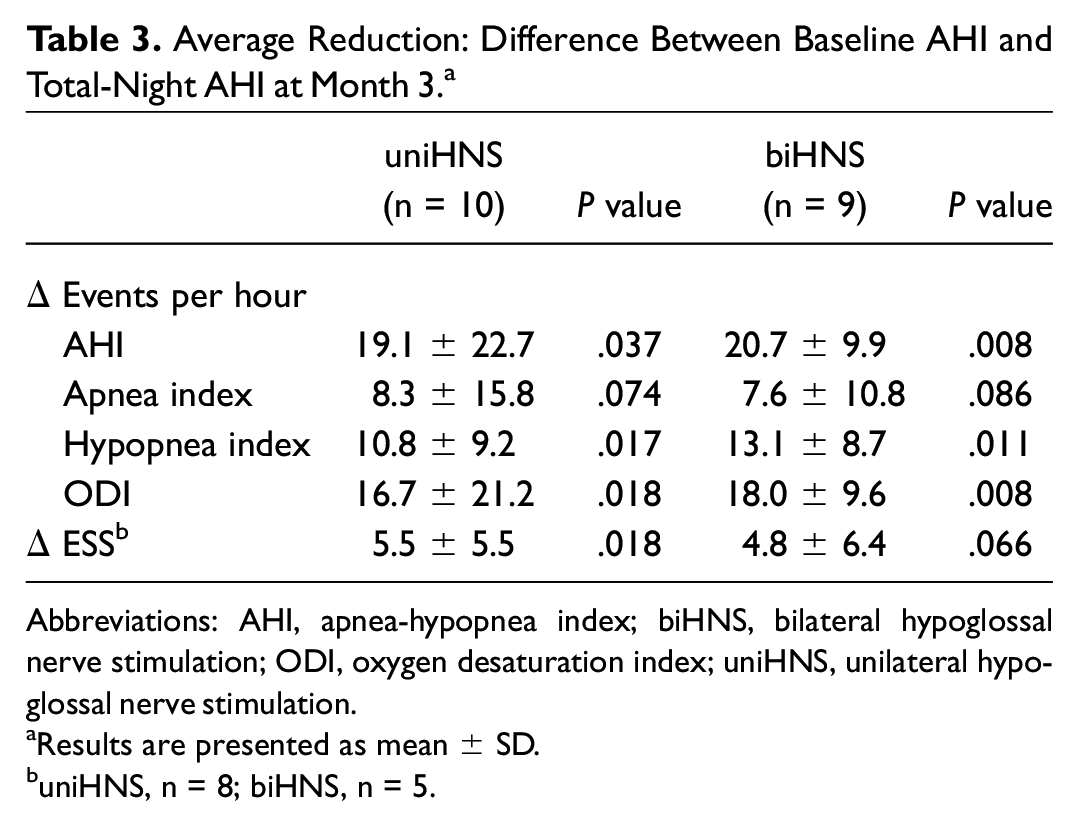
Abbreviations: AHI, apnea-hypopnea index; biHNS, bilateral hypoglossal nerve stimulation; ODI, oxygen desaturation index; uniHNS, unilateral hypoglossal nerve stimulation.
Results are presented as mean ± SD.
uniHNS, n = 8; biHNS, n = 5.
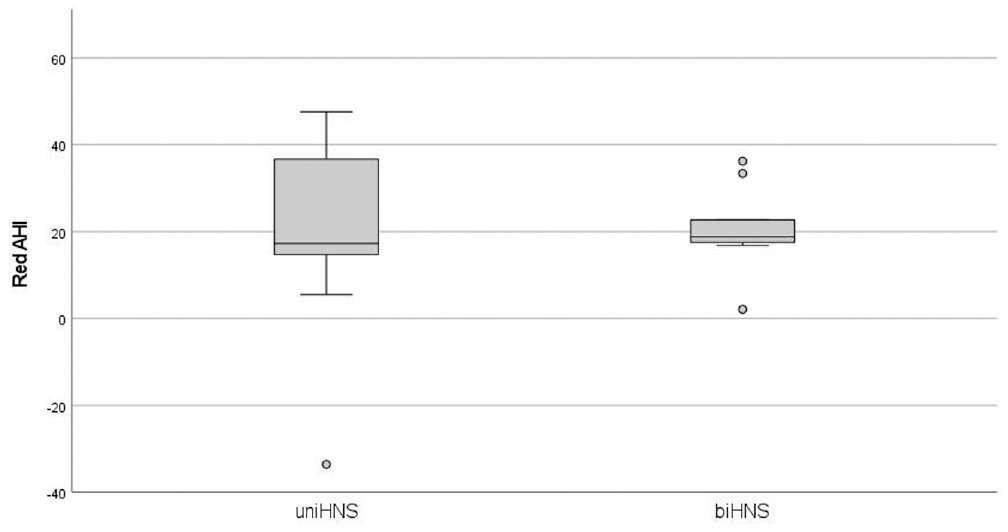
AHI reduction based on a box plot diagram in uniHNS and biHNS. No significant difference was observed (P = .842). Values are presented as median (line), interquartile range (box), 95% CI (error bars), and outliers (circles) AHI, apnea-hypopnea index; biHNS, bilateral hypoglossal nerve stimulation; uniHNS, unilateral hypoglossal nerve stimulation.
Reduction of Subjective Outcome Parameter “Sleepiness”
In both groups, sleepiness improved 3 months postsurgery and 2 months postactivation for uniHNS (11.1 ± 2.2 to 6.0 ± 5.2) and 1-month postactivation for biHNS (11.8 ± 6.2 to 9.4 ± 6.3). The Epworth Sleepiness Scale was reduced by 5.5 ± 5.2 (P = .018) in the uniHNS group and by 4.8 ± 6.4 (P = .066) in the biHNS group.
Responder Rates
Table 4 summarizes the responder rates for both groups at month 3 postsurgery. No statistical significance was detected between the groups.
Responder Rates at Month 3 per the Sher and Modified Sher Criteria. a

Abbreviations: AHI, apnea-hypopnea index; biHNS, bilateral hypoglossal nerve stimulation; uniHNS, unilateral hypoglossal nerve stimulation.
Results are presented as % (No.).
≥50% AHI reduction and AHI <20/h.
≥50% AHI reduction and AHI <15/h.
Discussion
In this small-sample study, we compared the early clinical safety and efficacy outcomes of biHNS therapy with that of uniHNS therapy. The preliminary data show no important differences between the results of the 2 systems. HN stimulation has shown to be effective, safe, and very well tolerated by patients with OSA.8,16,17 Over the last decade, multiple publications have shown that precise selection of patients, a highly standardized surgical procedure, a well-defined patient care pathway after implantation, and clear troubleshooting steps lead to good clinical outcomes.18-21
There are 5 major differences between the biHNS and uniHNS systems:
Bilateral stimulation of the HN instead of unilateral stimulation
Using paddle electrodes in biHNS instead of a cuff electrode in uniHNS
No electronic components and battery implanted in biHNS
Individually programmable duty cycle instead of a sensor that detects the breathing cycle
Stimulating the main protrusor “genioglossus muscle” instead of genioglossus and geniohyoid muscles to open the pharyngeal airway.
These differences make a biHNS system fundamentally different from a uniHNS system. First, it is conceivable that bilateral stimulation has a higher likelihood to result in improved response in OSA, although data to support that hypothesis are lacking. A previously published study of 19 patients who received a uniHNS system showed a greater response in PSG outcome parameters when a cross motor innervation of the HN could be detected. 18 This hypothesis can be important for patients who do not fully respond to HN stimulation therapy because of persisting obstruction at the level of the soft palate, since studies suggest that patients with a uniHNS implant and cross motor innervation demonstrated a greater opening on the soft palate. This could imply that bilateral stimulation of the HN, with stronger activation of the intrinsic transverse and vertical muscles, might have a stronger effect on the velum. 18 Preliminary results from an unpublished study from Australia (ClinicalTrial.gov: NCT03763682) showed that patients with complete concentric collapse of the soft palate can successfully be treated with biHNS, supporting this hypothesis. Patients with complete concentric collapse of the soft palate are contraindicated for uniHNS therapy. However, additional study trials are needed to establish the benefits of biHNS vs uniHNS due to the small sample size and absence of an active comparator group.
Using a paddle electrode instead of a cuff electrode implies different effects on the electrical field. The cuff isolates the surrounding tissue from the energy while applying it along the entire surface area of the nerve segment. Yet, the paddle electrodes used in biHNS therapy are larger and hence result in a wider field of energy applied to the nerve. This is not a drawback of the biHNS system: it merely results in a different recruiting pattern of the HN. Additionally, because surgical access for the implantation differs between uniHNS and biHNS, different HN fibers could be targeted.9,11 Due to the submental midline approach, biHNS places the paddle electrodes a little farther anteriorly than in uniHNS. Both systems stimulate the HN branches, which are responsible for activating the genioglossus, intrinsic transverse, and vertical muscle fibers. 19 For biHNS, targeting the first cervical nerve (C1) can be challenging due to the anatomic variability. 19 In 7% of the patients, C1 branches off very proximally/early and leaves the main HN at a deep/acute angle.19,20 Accessing the HN fibers anteriorly can be challenging due to the difficulty in finding, mobilizing, and including this fiber under the paddle electrodes. To date, however, it is not entirely understood and resolved whether the larger and wider electrical field still stimulates C1. This needs to be proven in future clinical trials.
Another important difference, especially for the patient, is the fact that battery and electronic components are worn externally. This makes it mandatory to place the activation chip through a patch on the submental skin every night ( Figure 2 ). While concerns about skin irritation in the long term still exist, all patients who have been implanted and activated and who have been using their device for at least 2 months have thus far reported no problems. Prior to surgery, all participating centers offered patients a patch to take home for a few days to assess any potential allergy or skin irritation issues.
Both systems can be individually programmed to patients’ thresholds for sensing and motor stimulation. Concerning stimulation of the HN, uniHNS requires a sensing lead in the intercostal space, whereas biHNS simply uses a duty cycle. A duty cycle delivers stimulation at a fixed frequency, adjusted to overlap with the participant’s breathing frequency during unobstructed breathing when asleep. 11 In a previous clinical trial by Eastwood et al, stimulation duration was maintained at 70% of the total respiratory cycle time for most participants. 11 Whether an exact stimulation during inspiration is really needed is still unknown. Preliminary data from 3 patients with uniHNS and inversion of their sensing signals demonstrated an increase in AHI and oxygen desaturation index. 21 This pilot study included only 3 patients. To assess if a breathing cycle–dependent system is needed, additional studies are required to establish if the duty cycle matches the breathing cycle for maximum efficacy.
Early clinical data from the 3 advanced uniHNS centers using biHNS therapy suggest that the results may be comparable.
The study has several limitations. By design, the study was observational, and we did not randomize patients in 1 of the 2 groups. Also, the groups could not be matched or analyzed by propensity score matching due to the small sample size.
Sleep and associated respiratory events, measured through a total-night PSG, demonstrated the same effect in both patient groups. However, one must take into consideration that in the biHNS group, the reported AHI was a titration PSG; therefore, the reported AHI may be an underestimation of the AHI based on fixed therapeutic settings, since treatment is being optimized. Yet, it may also be an overestimation, since patients will start to practice with the optimized setting at home. If this setting causes too much discomfort, for example, the patient may opt to lower the setting, although this may lead to less therapeutic effectiveness.
These preliminary study results show that biHNS is a promising technique and to date not inferior to uniHNS. Further clinical trials are needed to distinguish the systems, as well as the long-term effects on respiration, sleep, inclusion criteria (phenotyping responders), compliance, and satisfaction.
Conclusion
Initial postmarket data show that biHNS may be as safe and effective as uniHNS. Furthermore, it could be successfully implemented in the routine clinical management of OSA. Long-term follow-up in a larger sample size is required to assess the stability of the biHNS outcomes in OSA.
Author Contributions
Disclosures
Footnotes
This article was presented at the AAO-HNSF 2021 Annual Meeting & OTO Experience; October 3-6, 2021; Los Angeles, California.
