Abstract
Objective
Determine risk factors for failure to receive surgical treatment among patients with obstructive sleep apnea.
Study Design
Population-based observational longitudinal cohort study.
Setting
Population-based database.
Methods
Multivariate analysis of 500,792 individuals with obstructive sleep apnea from Optum’s deidentified Clinformatics Data Mart database (2004-2018).
Results
Black race, increased age, diabetes, atrial fibrillation, obesity, and congestive heart failure were independently associated with a decreased rate of surgery for obstructive sleep apnea. Asian race, hypertension, arrhythmias other than atrial fibrillation, pulmonary disease, and liver disease were independently associated with an increased rate of surgery for obstructive sleep apnea.
Conclusion
Racial disparities in health outcomes related to health care access and in economic resources have an enormous impact on public health and social equity. We found differences in rates of surgery for obstructive sleep apnea based on race. These data are consistent with others demonstrating disparities in medical treatment of sleep apnea with positive pressure and underline a need for a change in awareness and treatment in these populations.
Racial and ethnic disparities in health were brought recently to the forefront of public consciousness due to the disproportionate toll that the COVID-19 pandemic has taken on African American individuals1,2 and the Black Lives Matter movement. However, these disparities are deep rooted in American society, originating from extensive social and structural inequities. Over hundreds of years, racism and discrimination have resulted in vast economic and educational disadvantages for African American individuals that result in decreased health care access and quality, ultimately leading to poorer health outcomes. We are now beginning to understand the scope of the issue.
Obstructive sleep apnea (OSA) is an enormous public health challenge that disproportionately affects African American individuals. Moderate to severe OSA (apnea-hypopnea index ≥15) affects approximately 10% of US adults and is strongly associated with increased cardiovascular disease, diabetes, neurocognitive disease, and death. These health consequences are mitigated or prevented with treatment 3 ; however, an alarming 80% to 90% of those who have OSA remain undiagnosed. 4
Disturbingly, African American adults and children have a higher prevalence of OSA5-12 and a decreased rate of treatment as compared with White people.13-20 A recent study demonstrated moderate or severe OSA in 24% of African American adults, while just 5% of those had a prior diagnosis. 21 It is well known that the path to diagnosis and treatment for OSA can be tedious and fraught with opportunities to fall through the cracks, and African American patients are especially susceptible. One study found that only 38% of African American patients referred by their primary care providers eventually received an evaluation by a sleep clinician; 91% of those evaluated were diagnosed with OSA. 22 African American patients self-refer for sleep-disordered breathing at lower rates vs White patients 23 and have increased OSA severity at diagnosis. 24 Treatment does indeed reduce mortality among African American patients with OSA. 19 However, once a diagnosis has been made, data show decreased continuous positive airway pressure (CPAP) usage among African American individuals13-19 (although others found no difference23,25).
There is no organic, scientifically valid explanation for these disparities across race. In contrast, socioeconomic factors account for many disparities. It has been proposed, with limited data, that the increased prevalence of OSA among African American adults vs White adults can be explained by a higher rate of obesity.6,7,26,27 The cause of the latter trend, though, is clear. Abundant data demonstrate that these increased rates of obesity are explained by socioeconomic factors, such as decreased access to affordable and healthy dietary options, access to health care, vocation, and walkability of neighborhoods.28-31 While data exist showing disparities in diagnosis and treatment of OSA, to date there are limited data on demographic variables or comorbidities affecting rates of surgical treatment for OSA.
Methods
This study was approved by the Stanford Institutional Review Board (IRB-40974).
Data Source
All data were sourced from Optum’s Clinformatics Data Mart administrative claims database, which contains the longitudinal health care records of commercially insured enrollees covered by qualifying health care plans from a health care insurance provider. Diagnoses can be queried through ICD-9/10 codes (International Classification of Disease, Ninth and Tenth Revision), while procedures were inferred from Current Procedural Terminology (CPT) codes. As this data source parsed race/ethnicity as White, Black, Asian, and Hispanic, we refer to Black race rather than African American individuals for the remainder of this article. We acknowledge that White, Black, and Asian are races, while Hispanic vs non-Hispanic refers to ethnicity; we nevertheless present the variables set by our database.
Cohort Identification
Adult patients (>18 years) with OSA were identified from Optum’s Clinformatics Data Mart database (2014-2018), which is derived from a database of deidentified administrative health claims for members of large commercial and Medicare Advantage health plans.
Criteria for inclusion were initiation of CPAP therapy with either a formal diagnosis code corresponding to apnea or a polysomnogram within 1 month of CPAP initiation ( Figure 1 ). Development of this search strategy was required given the previously discovered low sensitivity of using only diagnosis codes for OSA for case identification. 32 The index diagnosis date was defined by the first OSA-related encounter (the first occurrence of CPAP initiation, polysomnogram within 1 month of CPAP initiation, or an apnea diagnosis within 1 month of CPAP initiation). Additionally, patients were required to have at least 1 year of continuous preindex follow-up. Comorbidities were defined per the Charlson Comorbidity Index. Additional comorbidities included hypertension, atrial fibrillation, increased body mass, and other cardiac arrhythmias. The primary outcome of interest was time to surgical intervention following OSA diagnosis. Eligible procedures were uvulopalatopharyngoplasty, septoplasty, tonsillectomy, turbinate reduction, hyoid myotomy, and genioglossus advancement. Maxillofacial surgery was not included. Other features in our analysis were demographic (age, sex, and race) and socioeconomic (income, education, and number of dependents in the household). An overall 500,792 patients were included in analysis, with demographic data for this cohort presented in Table 1 .

Inclusion criteria. Algorithm for inclusion of patients with OSA through available administrative data; adapted from McIsaac et al. 32
Demographics. a
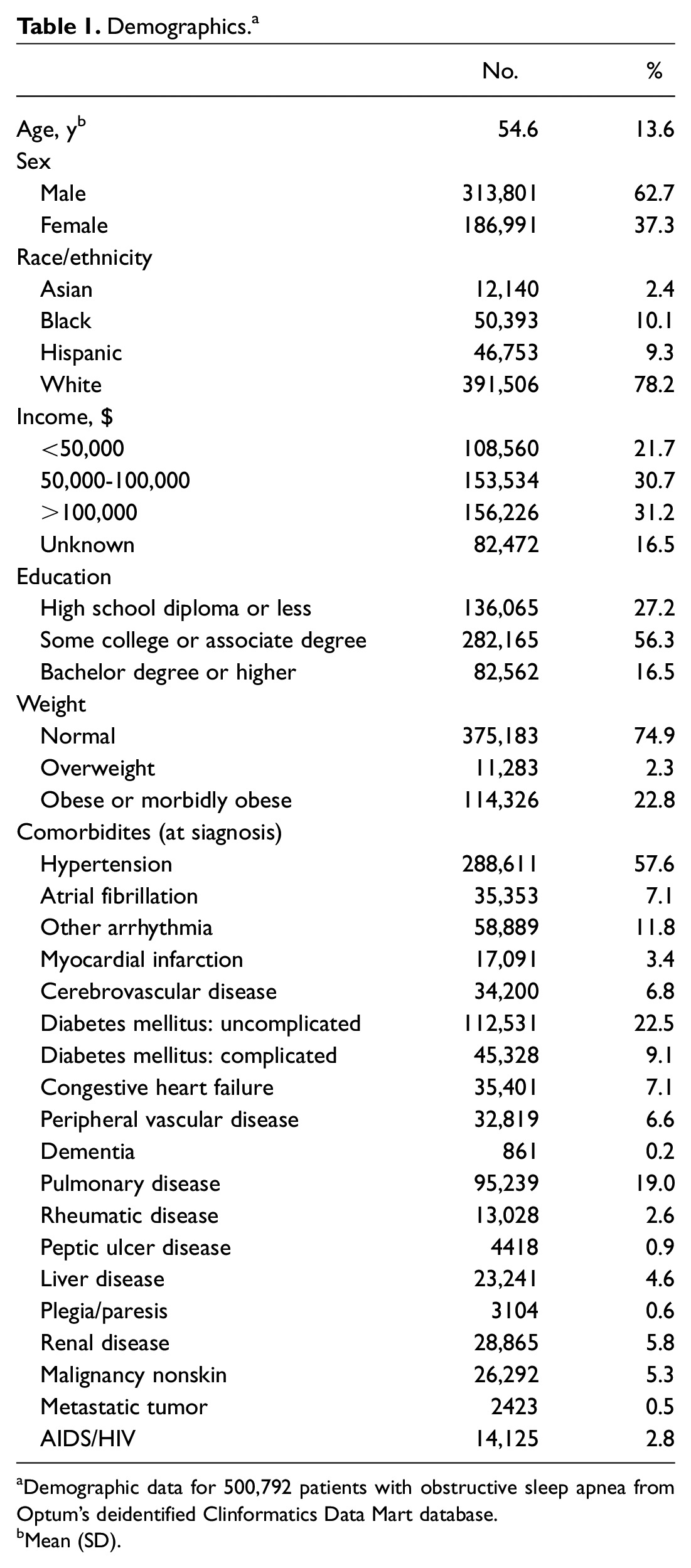
Demographic data for 500,792 patients with obstructive sleep apnea from Optum’s deidentified Clinformatics Data Mart database.
Mean (SD).
Statistical Analyses
Broadly, nonparametric approaches were used for 2-sample comparisons of continuous and categorical variables (Mann-Whitney U test and chi-square test of independence, respectively). A time-varying multivariable Cox regression approach, including all demographic and medical covariates ( Tables 2 and 3 ), was used to interrogate the primary outcome of interest (presence of surgical treatment for OSA). Comorbidities were analyzed either as static variables or with time-varying analysis. Whereas static variables are associated with a patient only if they have been entered in the patient’s chart at the time of initial OSA diagnosis, time-varying analysis incorporates any new diagnosis during the time between OSA diagnosis and the endpoint (surgery).
Demographic Variables. a
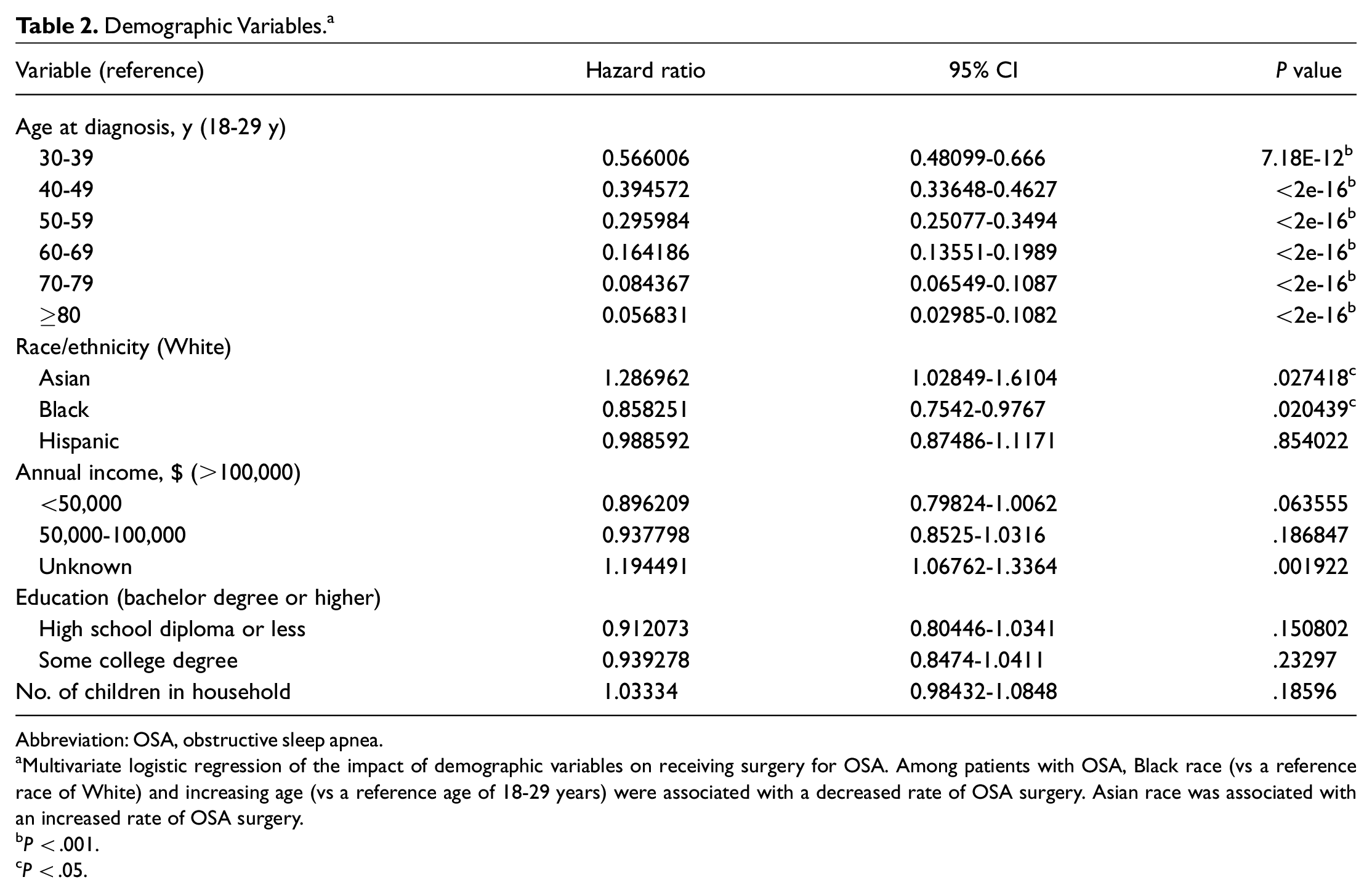
Abbreviation: OSA, obstructive sleep apnea.
Multivariate logistic regression of the impact of demographic variables on receiving surgery for OSA. Among patients with OSA, Black race (vs a reference race of White) and increasing age (vs a reference age of 18-29 years) were associated with a decreased rate of OSA surgery. Asian race was associated with an increased rate of OSA surgery.
P < .001.
P < .05.
Comorbid Variables. a
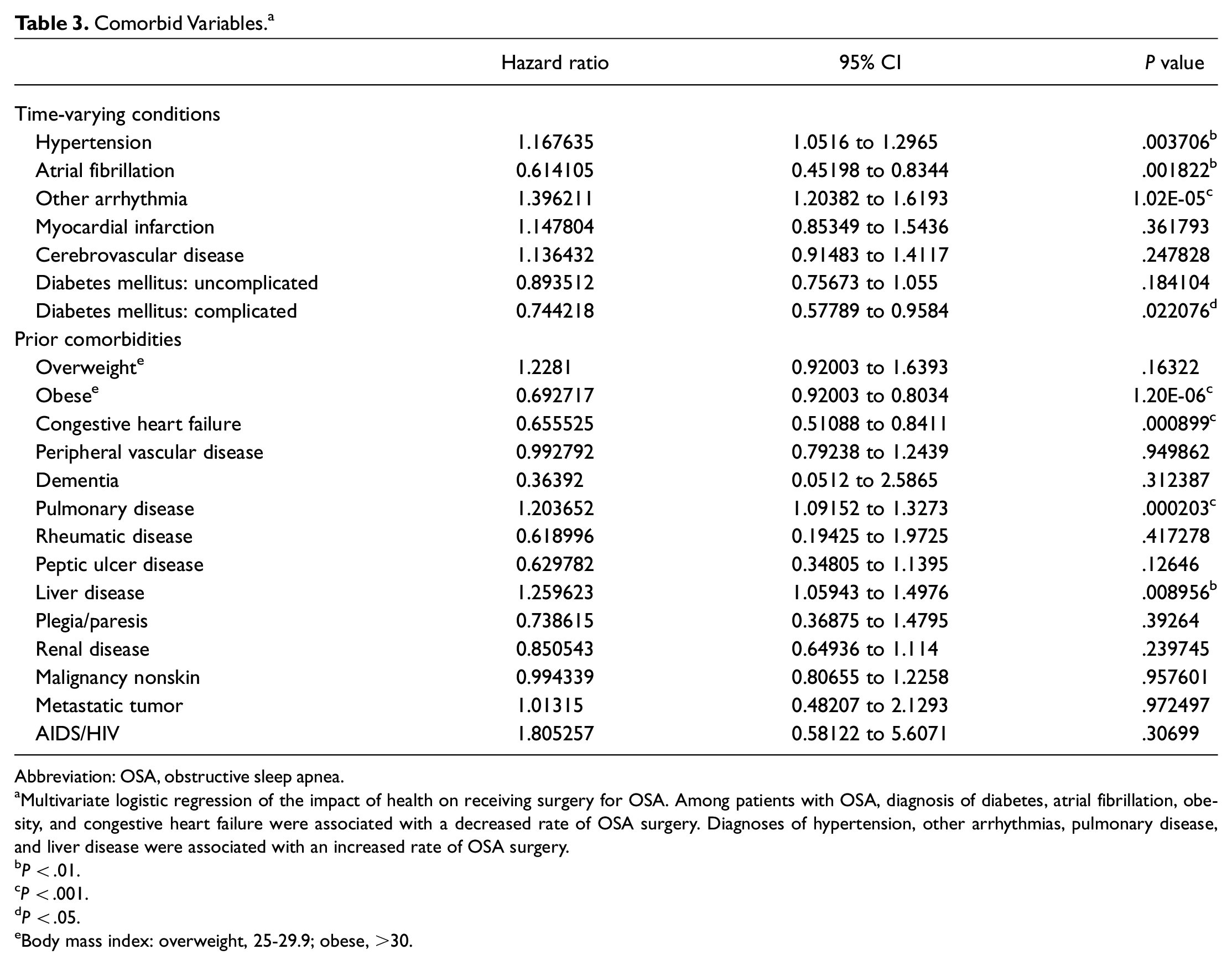
Abbreviation: OSA, obstructive sleep apnea.
Multivariate logistic regression of the impact of health on receiving surgery for OSA. Among patients with OSA, diagnosis of diabetes, atrial fibrillation, obesity, and congestive heart failure were associated with a decreased rate of OSA surgery. Diagnoses of hypertension, other arrhythmias, pulmonary disease, and liver disease were associated with an increased rate of OSA surgery.
P < .01.
P < .001.
P < .05.
Body mass index: overweight, 25-29.9; obese, >30.
Time-varying covariates were selected a priori and included diabetes mellitus (with or without chronic complications), myocardial infarction, cerebrovascular disease, hypertension, atrial fibrillation, and other arrhythmias. Particularly over an extended period of follow-up, time-varying models can account for changes in a patient’s current condition and their effects on the outcome of interest (in this case, undergoing surgery for OSA relief). Medical treatment of OSA has been shown to reduce morbidity and mortality and/or improve biomarkers associated with these conditions; thus, a new diagnosis of OSA may indicate a need for earlier surgery.33-38 We limited the time-varying feature set to avoid increasing model complexity, which limits the model’s applicability to new data by overfitting on the existing data. The time interval for the analysis began at the date that patients met inclusion criteria. These patients were followed until a surgical intervention for OSA or for as long as data were continuously available in our database, with a minimum interval of 1 year. All demographic and medical comorbidities were combined in the same analysis.
All analyses were conducted on all variables at the same time (survival package, R version 4.0.0; R Project for Statistical Computing). Statistical significance was met below a 2-tailed P value of .05.
Results
Our analysis comprised 500,792 insured patients with a diagnosis of OSA based on our inclusion criteria. All variables in our multivariate analysis are presented in Tables 2 and 3 . We included patient data from 2004 to 2018. The age range was 18 to 90 years at diagnosis, with a median of 54. Median follow-up postdiagnosis was 872 days. As reported by the database, this population had a racial/ethic breakdown of White (391,506), Asian (12,140), Black (50,393), and Hispanic (46,753). Our multivariable Cox regression included all demographic and medical covariates.
Among demographic variables, increased age above the 18- to 29-year range (P < .001) and Black race (P = .020) were independently associated with decreased likelihood for receiving surgery for sleep apnea. Asian race was associated with increased likelihood of receiving surgery for sleep apnea (P = .027). Neither annual income nor education level was significantly associated with surgery for OSA, although yearly income <$50,000 trended toward an association with a lower likelihood for receiving surgical treatment (P = .064; Table 2 ).
Among prior comorbidities—which, for the purpose of our analysis, were necessarily present at the time of OSA diagnosis—obesity (P < .001) and congestive heart failure (P < .001) were independently associated with decreased likelihood for receiving surgery for sleep apnea. Pulmonary disease (P < .001) and liver disease (P < .01) were associated with increased likelihood of receiving surgery for sleep apnea. No association was found between surgical treatment for OSA and those who are overweight or those diagnosed with diseases associated with OSA, such as dementia and pulmonary disease ( Table 3 ).
Among comorbidities analyzed in a time-varying analysis, which accounted for new diagnosis of a disease between the time of OSA diagnosis and the endpoint, diabetes associated with chronic complications (P = .022) and atrial fibrillation (P < .01) were associated with decreased likelihood of surgery for sleep apnea. Hypertension (P < .01) and other arrythmias (P < .001) were associated with increased likelihood of surgery for sleep apnea. Neither diagnosis of uncomplicated diabetes nor having a heart attack or stroke during the follow-up period was associated with subsequent surgery to treat OSA ( Table 3 ).
Limitations
Our data were derived from a large database of insured patients, which introduces several limitations. All demographic and health information was extrapolated from insurance codes. Furthermore, the study population includes only insured persons, which limits its applicability to the general population. However, as Black Americans are at higher risk of being uninsured than their White counterparts and as uninsured people have reduced access to health care, our findings likely underestimate any disparity in care.
Discussion
Our analysis reveals insight into the management of OSA and the population of patients who receive surgical treatment. Our data do not shed light on CPAP adherence for which there are known disparities. 13 Surgery was avoided in patients with medical comorbidities that might increase the risk of general anesthesia, such as heart disease, obesity, and diabetes with chronic complications. In contrast, hypertension, pulmonary disease, and arrythmias (with the exception of atrial fibrillation) were associated with increased rates of surgery. Considering that treating OSA with CPAP is known to reduce blood pressure 37 and recurrence of arrythmias, 33 our results suggest that surgery for sleep apnea may be performed in the interest of optimizing health and reducing cardiovascular and pulmonary risk factors. The association seen in several other variables is more difficult to explain. The reduced incidence of surgery in patients with atrial fibrillation may be related to hesitance to stop the anticoagulation that may be prescribed, or it may be confounded by inconsistencies with coding (if coded as other arrythmia). Indeed, the main limitation of our analysis lies in the sourcing of data through insurance codes.
Asian race corresponded to an increased likelihood of surgical treatment of OSA. This may be explained by lower rates of obesity in Asian communities as compared with Caucasian communities. While obesity is a risk factor for OSA among all ethnicities, including Asian, surgical treatment may be more commonly offered in those with lower body mass index. 39 For example, Lam et al found that Asian individuals had more severe OSA, partly due to a more crowded posterior oropharynx, 40 suggesting that Asian patients with OSA may be more amenable to surgical intervention. There are limited data on the role of access to health care among Asian Americans.
The finding on which we are choosing to focus our discussion is that Black race is a risk factor for failure to receive surgical treatment of OSA, independent of all other demographic variables and comorbidities. In other words, insured Black individuals are less likely to receive surgery for sleep apnea than their White counterparts, independent of income, wealth, and comorbid health conditions. These findings raise the question of why.
One historical explanation pertains to craniofacial anatomy, in which there are known racial and ethnic variabilities, some of which have greater association with OSA.6,41 Whereas White individuals with OSA have differences in bony and soft tissue cephalometric measures as compared with White controls, differences in bony structures do not predispose to OSA in Black individuals.
In contrast, increased tongue area and increased soft palate length are associated with increased risk of OSA in Black individuals. 41 This may explain some of the disparities in rates of maxillofacial surgery. However, these surgical procedures are relatively rare and are typically performed after failure of less invasive surgery for OSA; moreover, maxillofacial surgery was excluded from our analysis.
We cannot discount mistrust of physicians as a factor explaining our result. The US medical system has a long history of mistreating Black people. Notable examples include forced sterilizations of Black women and the Tuskegee syphilis study, which withheld treatment from hundreds of Black men. As a result, this community has developed significant medical mistrust, which pervades the health care system. A November 2020 study by the Pew Research Center found that 42% of Black individuals intended to receive the COVID-19 vaccine, in contrast to 61% of White individuals (83% of Asian individuals). 42 This intention played out in reality, and as of December 13, 2021, just 51% of Black Americans have received at least 1 COVID-19 vaccine dose, in contrast to 58% of White Americans (77% of Asian Americans). 43 This mistrust developed and persisted because of systemic and overt racism, and it must be addressed rather than dismissed if we wish to decrease disparities in health. Indeed, OSA is emblematic of a much broader trend in public health.
To correct this and move toward a more equitable and healthy society, we must direct our attention and resources toward addressing racial and ethnic health disparities in our approach to future diagnostic and therapeutic interventions for OSA. Clinicians must educate and counsel patients in a tailored manner, sensitive to the justified mistrust as well as to the disparities in health literacy and exposure to the health system. Ultimately, this is a population health issue that is likely best addressed at the level of community outreach and policy. Research has demonstrated that culturally tailored approaches toward education, evaluation, and treatment of OSA, as developed with the input and approval of the targeted communities, can significantly improve outcomes. Specifically engaging community leaders, working through community centers and faith-based organizations, and developing tailored websites may help effectively reach minority populations.44-46 In 2010, the World Health Organization published a document outlining policy approaches toward achieving gender equality and health equity. 47 We assert that similar policy recommendations should be developed for race and ethnicity. Clinicians, companies, and policy makers must be ready and willing to open their eyes, ears, and minds to the perspectives of the underserved for us to dismantle the systemic barriers and gaps that perpetuate racial and ethnic health disparities in this country.
