Abstract
Study Design:
Case Report.
Objective:
The Temporalis muscle (TM) Flap is a reliable, versatile flap with adequate bulk and flexibility. However, temporal hollowing is the most common postoperative complication after harvesting the flap. We describe a surgical modification for harvesting the split TM flap to prevent postoperative temporal hollowing and report 2 cases with up to 18 months of follow-up.
Method:
This study discusses the posterior ⅔ split temporalis muscle flap in preventing temporal hollowing. We report 2 well-documented cases with up to 18 months of postoperative follow-up. Also, the authors discuss the key points regarding the technique’s indication and limitation.
Results:
The posterior ⅔ split temporalis muscle flap is reliable in both benign and malignant disease processes. The results demonstrate the flap’s reliability with minimum donor site morbidity.
Conclusion:
The use of the posterior ⅔ of the TM flap is a viable technique to prevent the flap’s harvest’s temporal hollowing.
Introduction
Since its first introduction in the late 1800s, the use of the Temporalis Muscle (TM) Flap has evolved in the craniomaxillofacial region. The TM Flap is a reliable, versatile flap with adequate bulk and flexibility to be mobilized to adjacent defects. Additionally, surgical modifications extended its uses in maxillofacial reconstruction, including coronoidectomy and zygomatic arch osteotomy, to facilitate flap rotation. Other modifications include splitting the muscle into 2 halves and combining the flap with bone or skin as a composite flap. 1 –3
Cadaveric studies have shown that the blood supply to the temporalis muscle is derived from the maxillary artery that will give the two-deep temporal arteries, the anterior (ADTA) and posterior(PDTA). It is also supplied by the middle temporal artery, which is a branch of the superficial temporal artery. The viability of the muscle during harvest and transfer will depend on the ADTA and the PDTA, thus it has 2 primary blood supplies and a Mathes and Nahai type III vascular pattern. 4 –6 In his leading article, Cheung has found that the PDTA is the most abundant blood supply to the temporalis muscle, and it provides blood supply to the middle 40%. On the other hand, the ADTA occupies only 20% of the muscle anteriorly, and the middle temporal artery occupies the remaining posterior region. Furthermore, he confirmed that the venous drainage system runs close to the corresponding arteries, with double veins associated with 1 artery. 2
Due to the unique vascular blood supply to the temporalis muscle, the TM flap can be divided into anterior ⅔ and posterior ⅓. Most authors have recommended using the anterior segment for reconstruction and then mobilize the posterior portion anteriorly to prevent the temporal hollowing. 7,8 Others have also suggested the use of alloplastic material such as silicon and porous polyethylene. 9,10
Since the temporal hollowing is the most commonly cited donor site morbidity following TM flap, we have used the posterior 1/3 of the temporalis muscle and preserved the anterior portion to prevent the temporal hollowing. The rationale is the reliability of the middle and posterior segments of the temporalis muscle as well as the assurance that the anterior ⅔ will keep the contour of the temporal region undisturbed.
This article will describe the surgical technique for harvesting the split TM flap and report 2 cases with up to 18 months of follow-up for both cases. Also, we will discuss the advantages and limitations of this surgical technique.
Report of Cases
Case 1
A 49-year-old female with a history of cT4aN0M0 squamous cell carcinoma of the floor of the mouth and anterior mandible. Six months before her presentation, she had a composite resection of the oral cavity, bilateral neck dissection, and reconstruction with the right anterior lateral thigh. The pathological staging was pT4N0MNA.The patient elected not to have postoperative radiation therapy. During her 6-month follow-up, she presented with pain and left retromolar trigone ulcers that measured about 2 cm by 1 cm in its greatest dimension—Figures 1 and 2. The lesions were biopsied, and the result came back as a moderately differentiated squamous cell carcinoma.
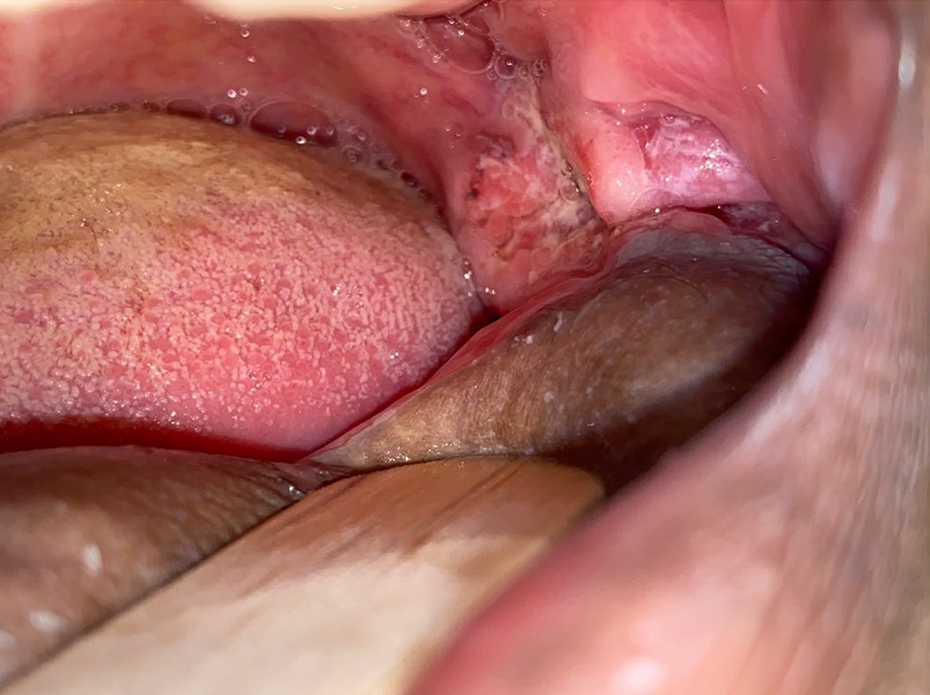
Intraoral picture of the recurrent squamous cell carcinoma of the retromolar trigone.
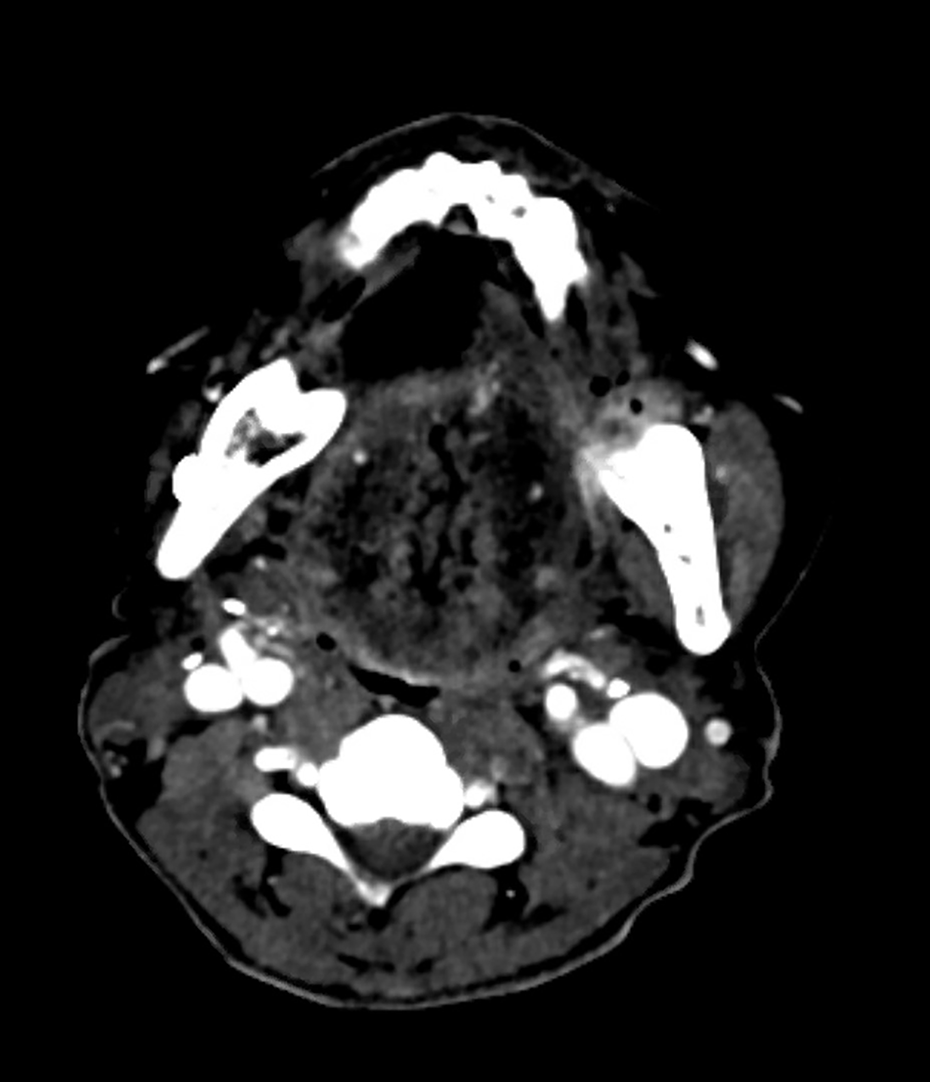
Contrast-enhanced CT scan demonstrating the lesion at the retromolar trigone. No bone invasion was detected.
The case was discussed in the Head and Neck Tumor Board and, due to the dismal prognosis; the decision was made to perform a second tumor ablative procedure with the reconstruction of the defect with a regional flap. This would allow the patient to start adjuvant chemoradiation therapy as soon as possible. After discussing multiple options with the patient, the decision was made to proceed with the split TM flap to prevent the temporal hollowing.
Postoperatively, the patient progressed very well and discharged from the hospital on postoperative day 3. The flap’s complete epithelialization was achieved by the fourth-week postoperatively, and the adjuvant concurrent chemoradiation therapy started on the fifth week after the surgery. Figures 3 and 4 show her last follow-up 18 months after the surgery.
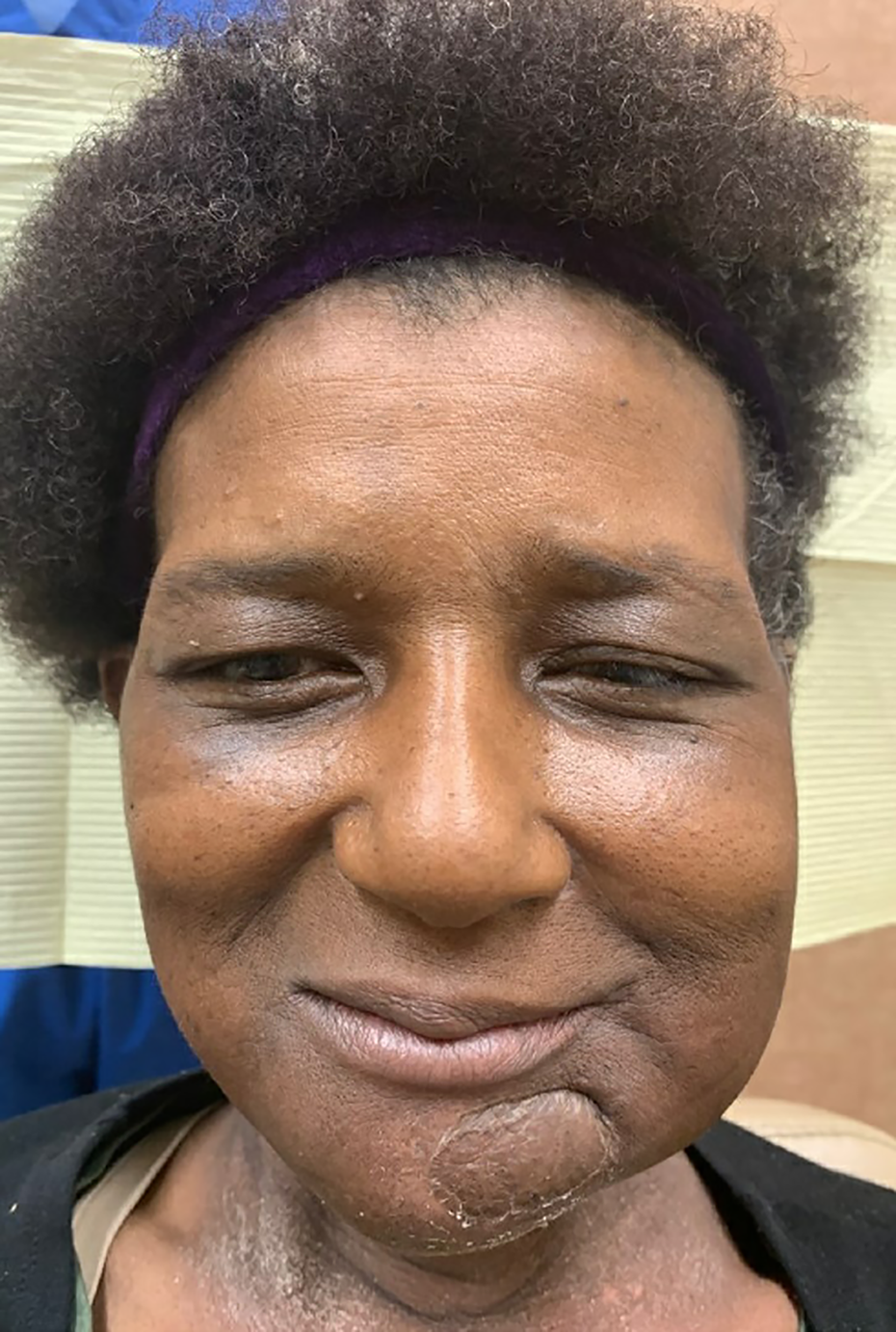
18 months follow-up after reconstruction of the left retromolar trigone with split temporalis muscle flap.
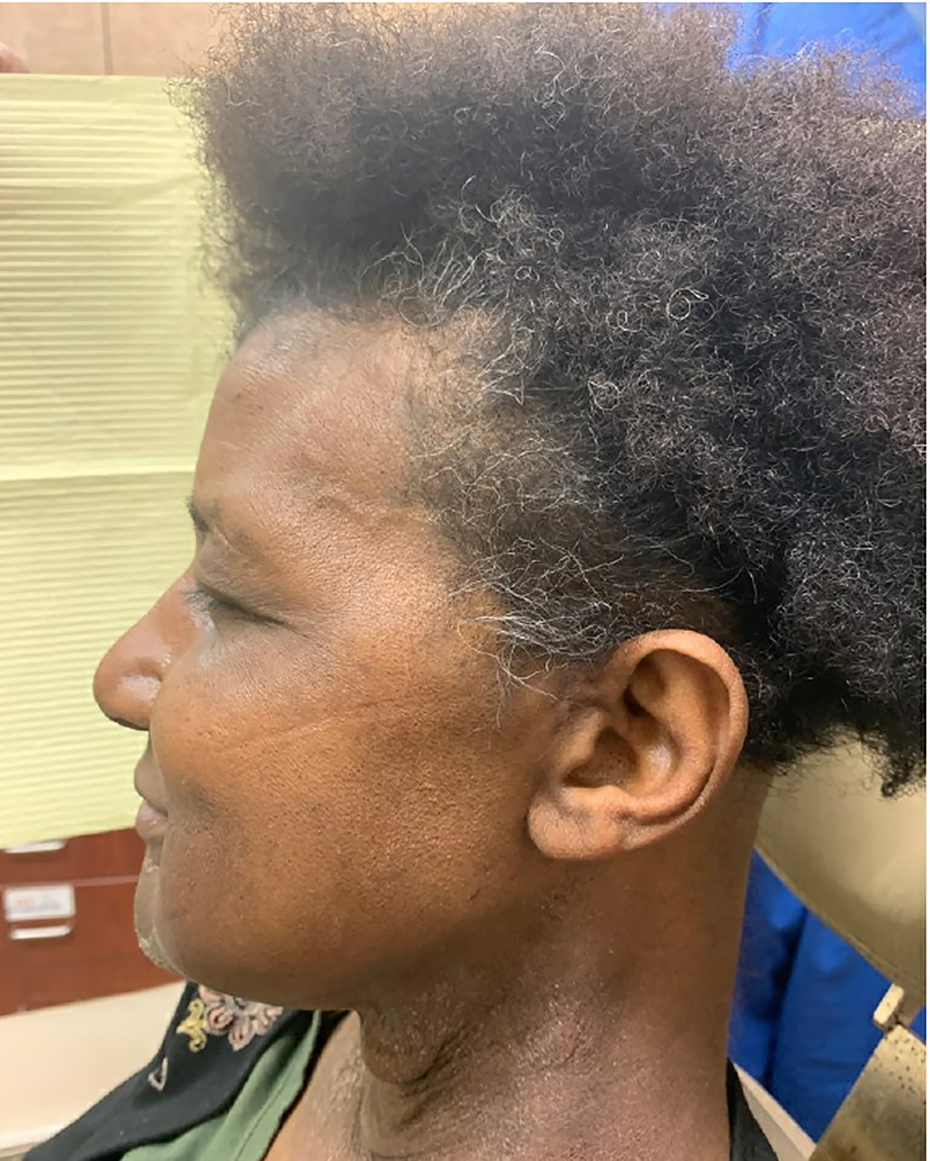
Healed surgical incision.
Case 2
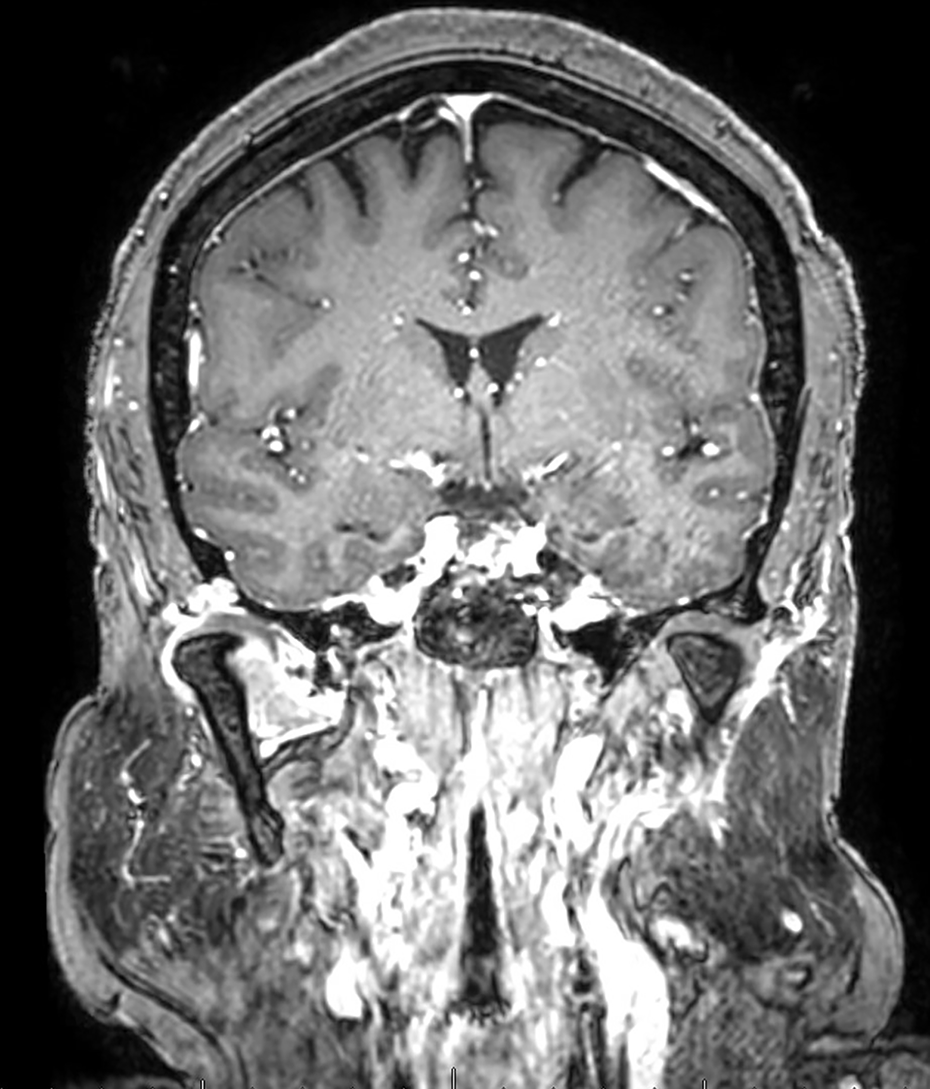
A 63-year-old female presented with a 3-year history of bilateral preauricular pain and progressive left sided deviation of the mandible. The patient underwent multiple non-surgical and surgical therapies at an outside facility without improvement. Upon clinical exam, the patient had tenderness around the left preauricular region with an anterior open bite. The patient reported that her occlusion was normal before her symptoms started. She also attempted orthodontic treatment of her malocclusion 3 times, with no resolution of her symptoms. MRI imaging was done and revealed a 3 cm right TMJ mass eroding through the skull base with a likely synovial etiology. Due to the erosion of the glenoid fossa and skull base, CT maxillofacial imaging was completed. The results showed a hypodense soft tissue mass anterior to the right mandibular condyle with associated extensive focal erosion of the glenoid fossa and widening of the temporomandibular disk space. Both CT and MRI showed severe erosion of the glenoid fossa with possible intracranial extension (Figures 5 and 6).
Coronal CT scan with contrast showing the amount of osteolysis at the level of the glenoid Fossa.
MRI with contrast showing the possible intracranial extension. Also, note the hyperintense lesion around the medial pterygoid.
The working diagnosis was determined to be a synovial chondromatosis. The case was discussed and reviewed with the Neurosurgery team, and the plan was to proceed with excision of the lesions and preserve the articular disc. During the surgery, it was noted that the lesions were firmly adherent to the disc, and the separation of the 2 was not possible (Figure 7). Arthroplasty and discectomy were performed, and the disc was replaced with a split TM flap. During the arthroplasty procedure, numerous large, round nodules were attached to the synovium, and several were free-floating within the joint space. The reconstruction was completed with a posteriorly based temporalis myofascial flap. Part of the muscle was used to cover the eroded area of the glenoid fossa; another part of the muscle was sutured retro-discal tissues (Figure 8).
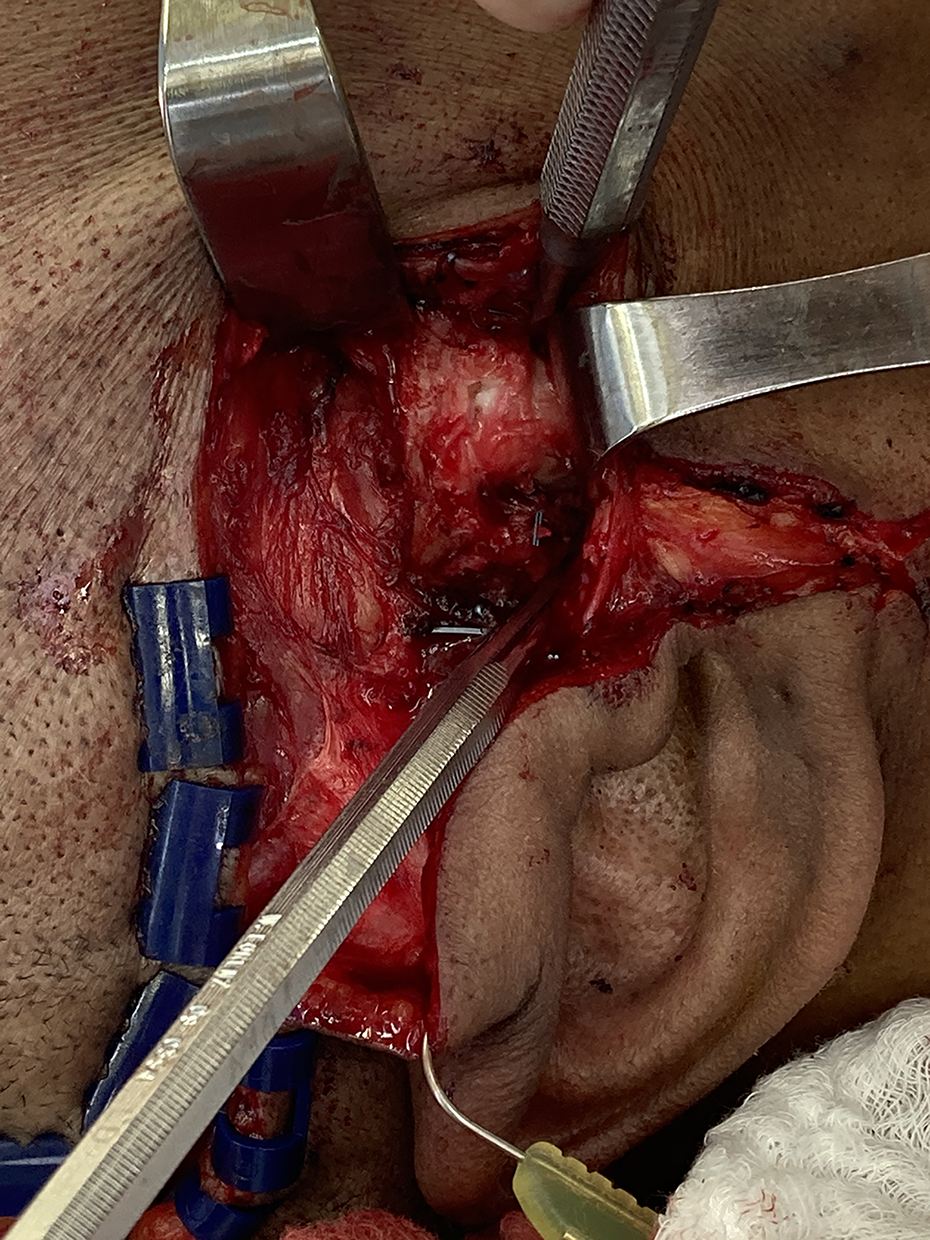
Intraoperative picture showing the adherence between the lesion and the articular disc.
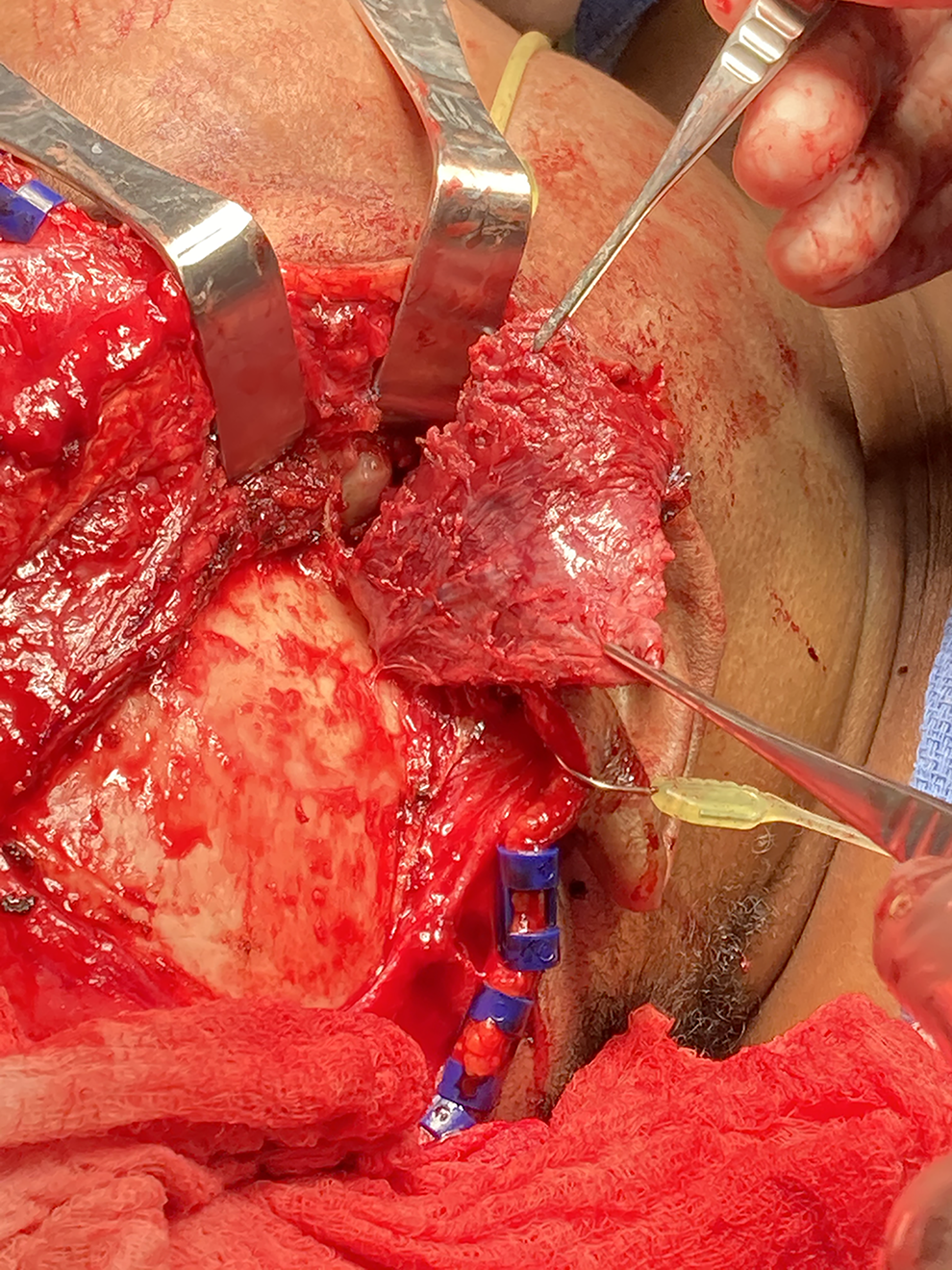
Intraoperative picture showing the split of the temporalis muscle based on the Posterior deep temporal artery.
The final pathology confirmed the diagnosis of synovial chondromatosis with pseudogout crystals. The prominent histopathological feature was the presence of chondrocytes with atypia (binucleated) within the synovium. The presence of the calcium pyrophosphate dihydrate crystals was also confirmed with the polarized light, but it was located only at the medial part of the lesion. Postoperatively, the patient remained in intermaxillary fixation for 1 week, and then transitioned to elastic occlusal guidance for additional 2 weeks. The patient’s premorbid occlusion re-established, and the patient was thoroughly satisfied with the results. Figures 9 and 10 showed the 1-year follow-up.

One-year follow-up after reconstruction of the right TMJ with split temporalis muscle flap.
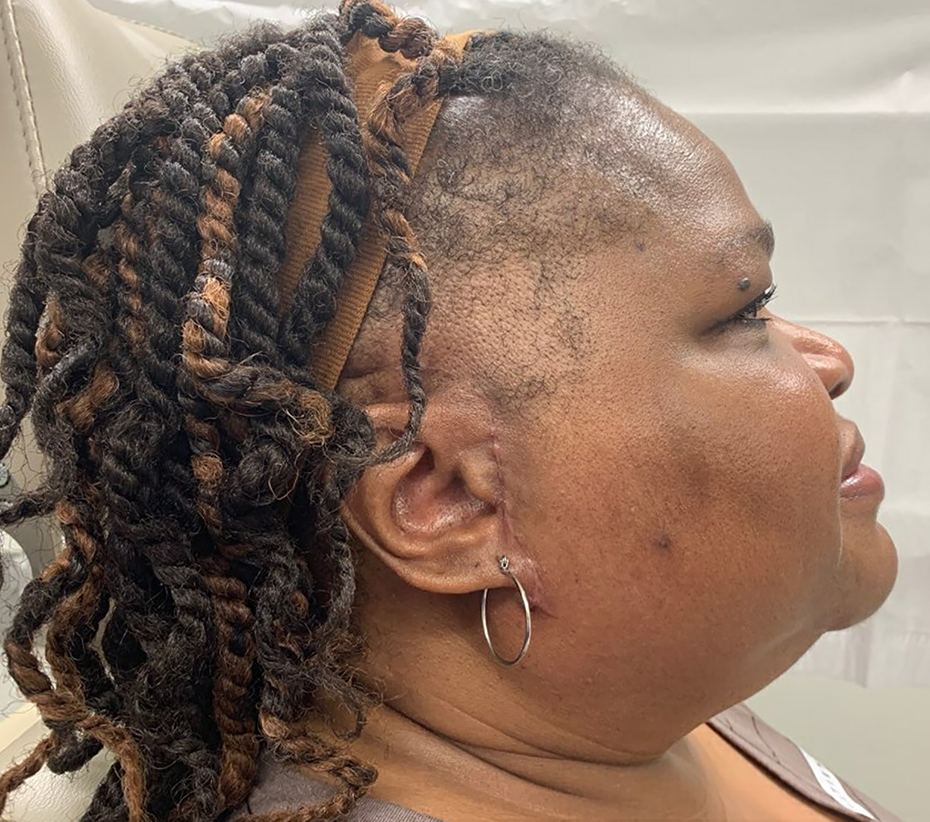
Lateral profile picture after reconstruction of the right TMJ with split temporalis muscle flap.
Surgical Technique
The surgical anatomy of the temporal region is demonstrated in Figures 11 and 12. There are 4 dissection planes described to access the temporal region and dissect through the zygomatic arch. 11 We use a modification of the supratemporalis approach. This approach has the lowest risk of facial nerve injury and temporal hollowing.
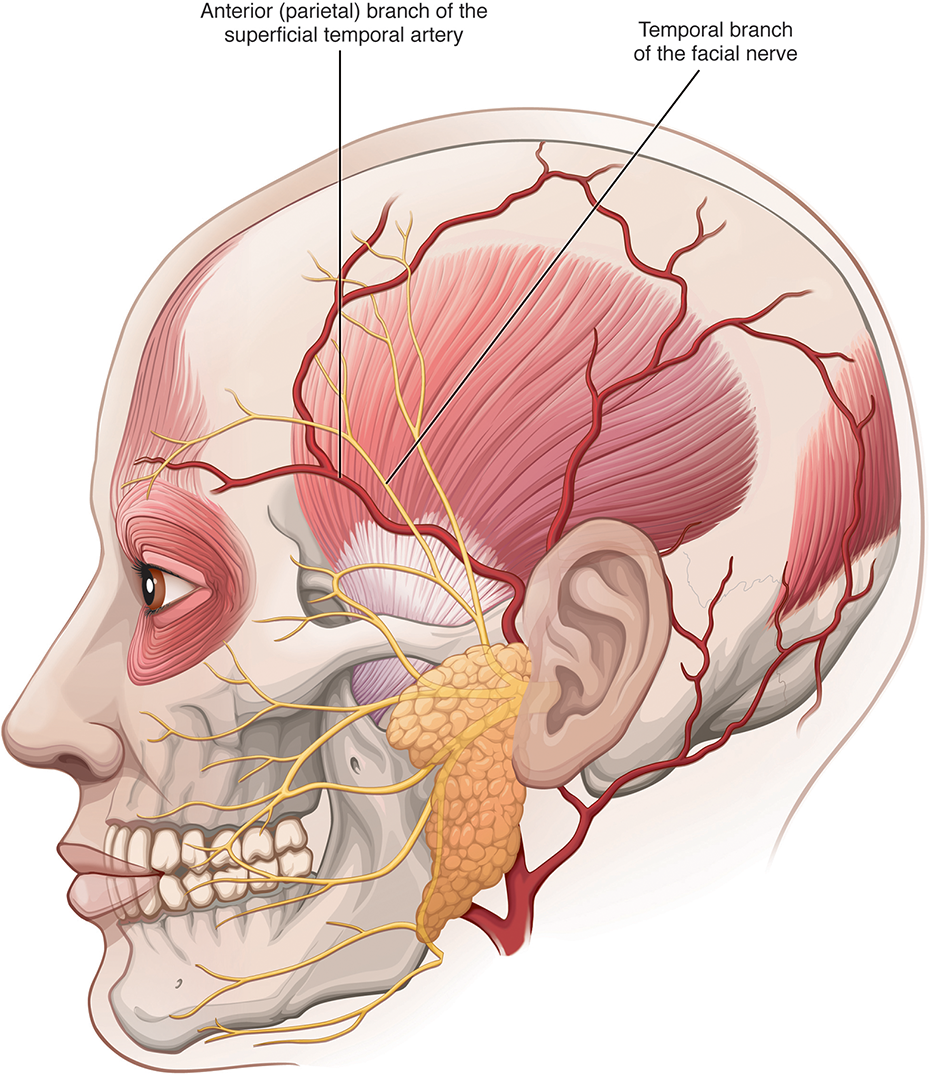
Surgical anatomy of the temporal region. Notice the relationship between the temporalis muscle and the facial nerve.
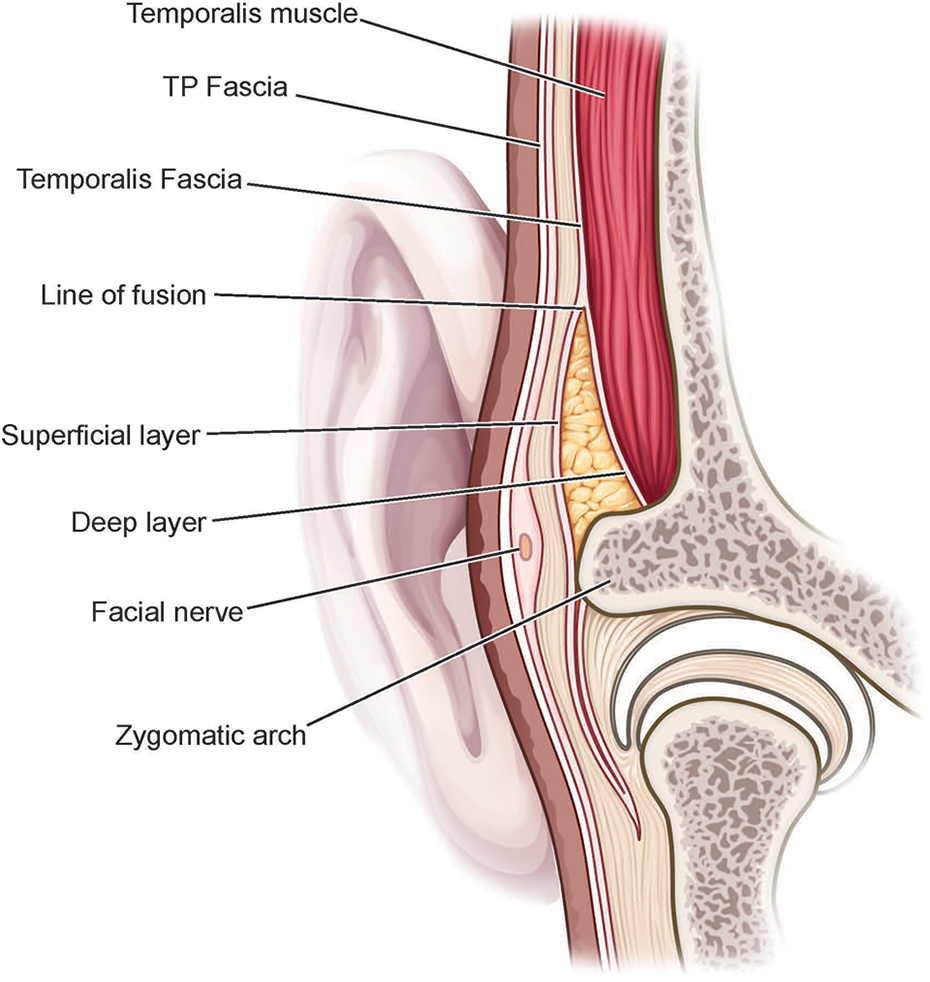
Surgical anatomy of the temporal region. Notice the location of the facial nerve at the level of the temporoparietal fascia.
The temporalis muscle is accessed through a coronal incision with a preauricular extension. This will provide the best exposure and refine the aesthetic outcome. The incision’s coronal extension should be placed about 2 cm above the temporal line and behind the hairline, while the preauricular extension should be within 8 mm of the tragus to avoid facial nerve injury (Figure 13).We recommend using a local anesthetic with epinephrine to help reduce the bleeding and aid in the dissection. The incision and dissection are carried out superiorly through the skin, subcutaneous tissues, the temporoparietal fascia (TPF) until the identification of the glistering temporalis fascia. The facial nerve’s temporal branch runs within the TPF fascia. Careful identification of the nerve is crucial since it travels relatively superficially in the temporal region. The skin flap with the TPF is elevated from superior to inferior until the zygomaticotemporal junction inferiorly and the lateral orbital wall medially are encountered. The temporal branch of the superficial temporal artery is identified and serves as a demarcation that should not be crossed to avoid injury to the facial nerve’s frontal branch (Figures 11, 12, and 14).
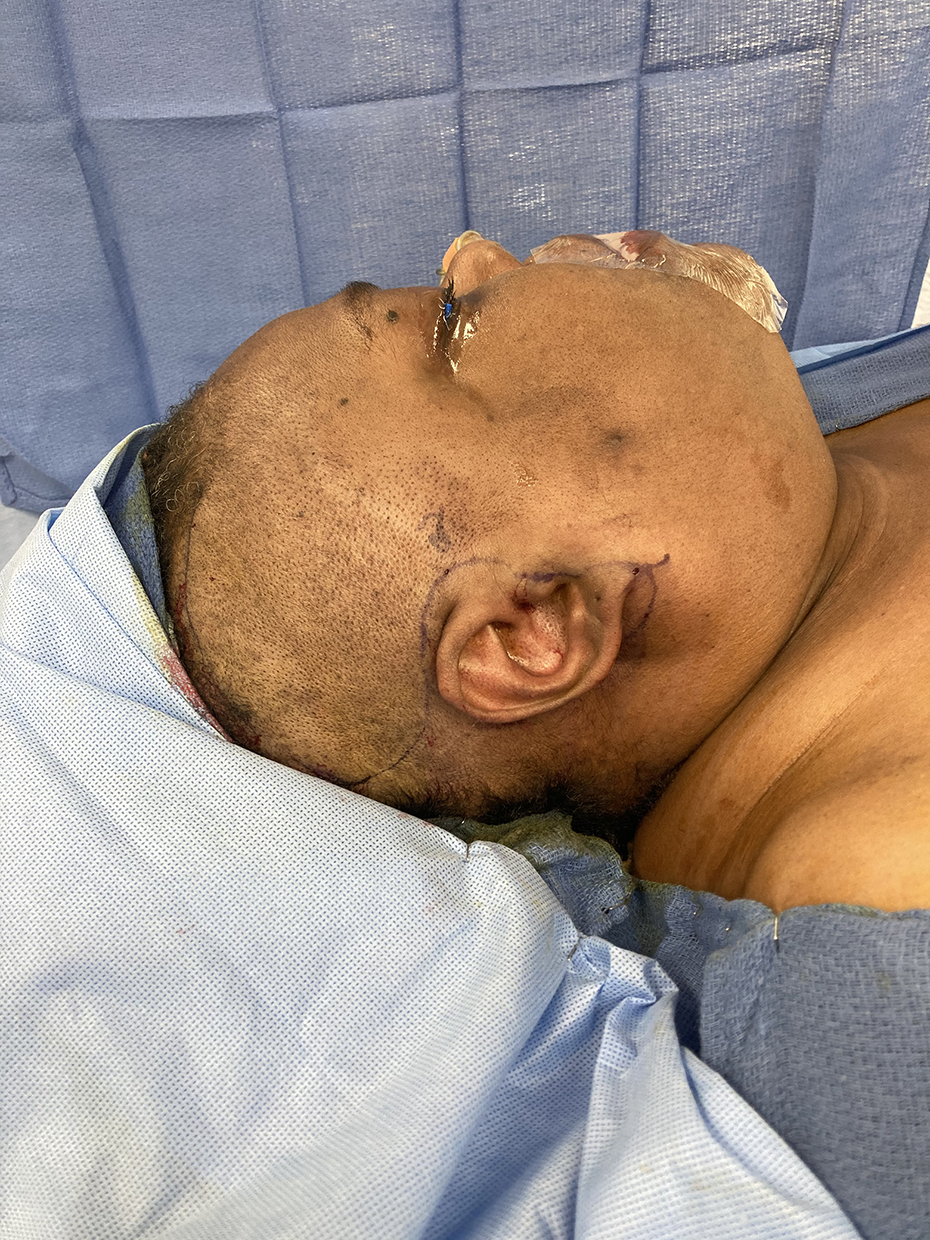
Marking of the planned incision. Notice the superior extension of the incision above the temporal line.
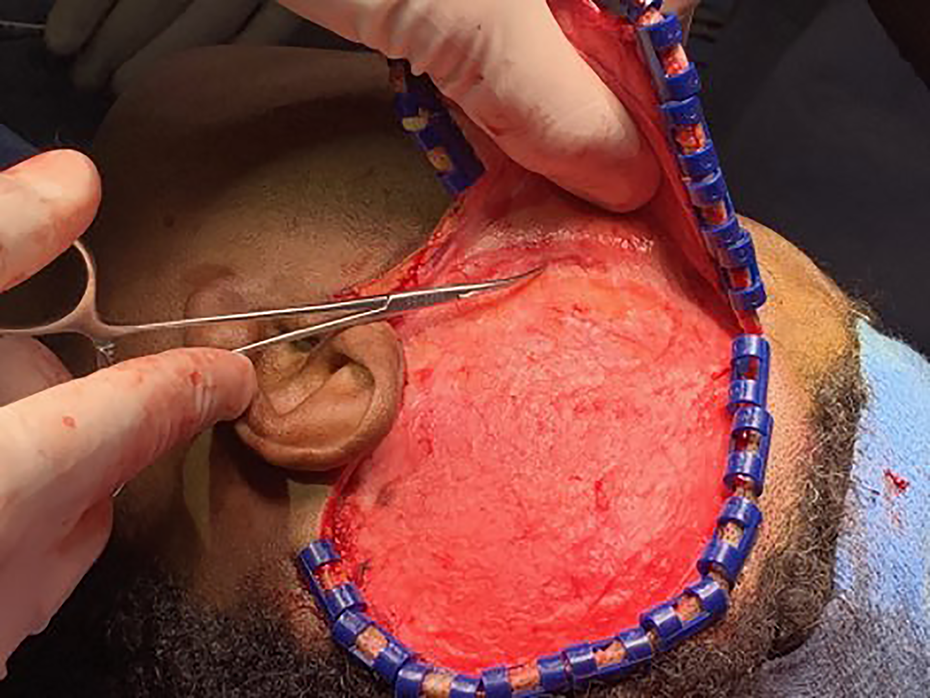
Elevation of the skin flap with the temporoparietal fascia to expose the underlying temporalis fascia. Hemostat pointing at the temporal branch of the facial nerve.
Subsequently, the temporalis fascia is incised at least 2 cm above the arch, parallel to the temporal line, until the underlying temporalis muscle is identified. After that, the temporalis fascia is elevated with the skin flap, leaving behind the temporalis muscle. Although blunt dissection is generally recommended, we have found that sharp dissection using the back of the 15-blade will help dissection and make for easier separation of the temporalis fascia from the temporalis muscle. The middle temporal artery will be encountered at this step, and careful identification and early ligation are prudent to prevent unnecessary bleeding. As described earlier, sacrificing this artery will not jeopardize the TM flap. This dissection is carried down inferiorly until the superficial temporal fat pad is identified. Once the fat pad is exposed, a retractor is used to identify the zygomatic arch, and the superficial layer of the deep temporal fascia is incised and further dissected anteriorly and inferiorly. This will lead the dissection directly to the zygomatic arch and protect the facial nerve branches. The zygomatic arch is then exposed using a periosteal elevator. At this juncture, if the arch is planned to be osteotomized, a 1.5 mm miniplate is predrilled, and the arch’s osteotomy is performed as far posteriorly as possible to help in rotating the muscle flap. Care should be taken to protect and preserve the blood vessels entering the muscle at its inferior aspect. Once the muscle is fully exposed, a handheld Doppler is used to identify the ADTA and the PDTA. The temporalis muscle is divided into anterior ⅓ and posterior ⅔ based on the distribution of the vessels (Figure 15). After determining the extent of the muscle and the line of division, the posterior ⅔ is incised and elevated off the underlying temporal fossa. Monopolar electrocautery is used to cut the more firm attachments. Particular care should be taken to early identify and protect the PDTA, which arises on the deep aspect of the muscle (Figure 16). The dissection should be carried down inferiorly to the level of the coronoid process. If extra length is deemed necessary, a transoral coronoidectomy can be performed. This will gain about 1.5 cm of pedicle length. 12,13 This will also help in creating a wide enough tunnel intraorally for the muscle to pass through. However, a minimum of 2 fingerbreadth is required to allow the muscle to pass easily and prevent pedicle compromise. A skin graft can be applied, but it is not necessary since the muscle will mucosalize nicely over time (Figure 17). 13,14
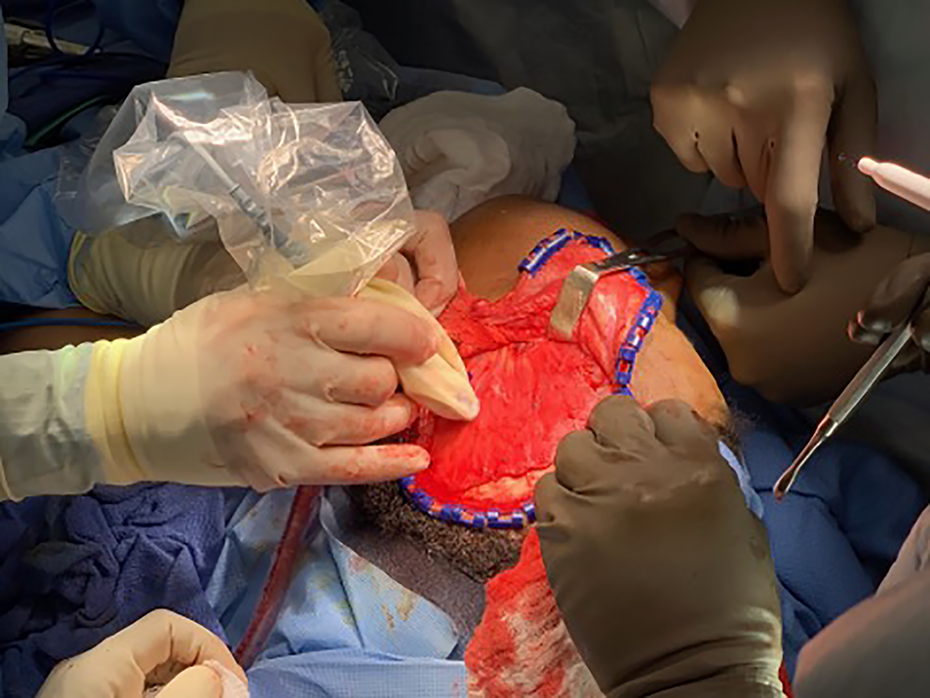
The use of the handheld Doppler to identity the anterior and posterior deep temporal arteries before splitting the temporalis muscle.
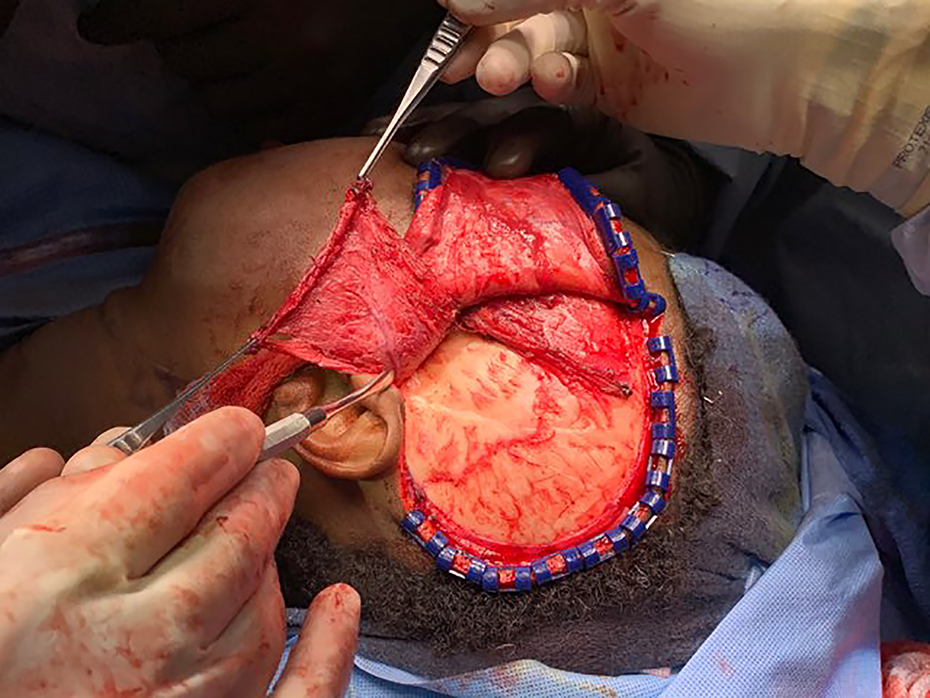
The elevation of the posterior ⅔ of the temporalis muscle and the identification of the Posterior deep temporal artery.
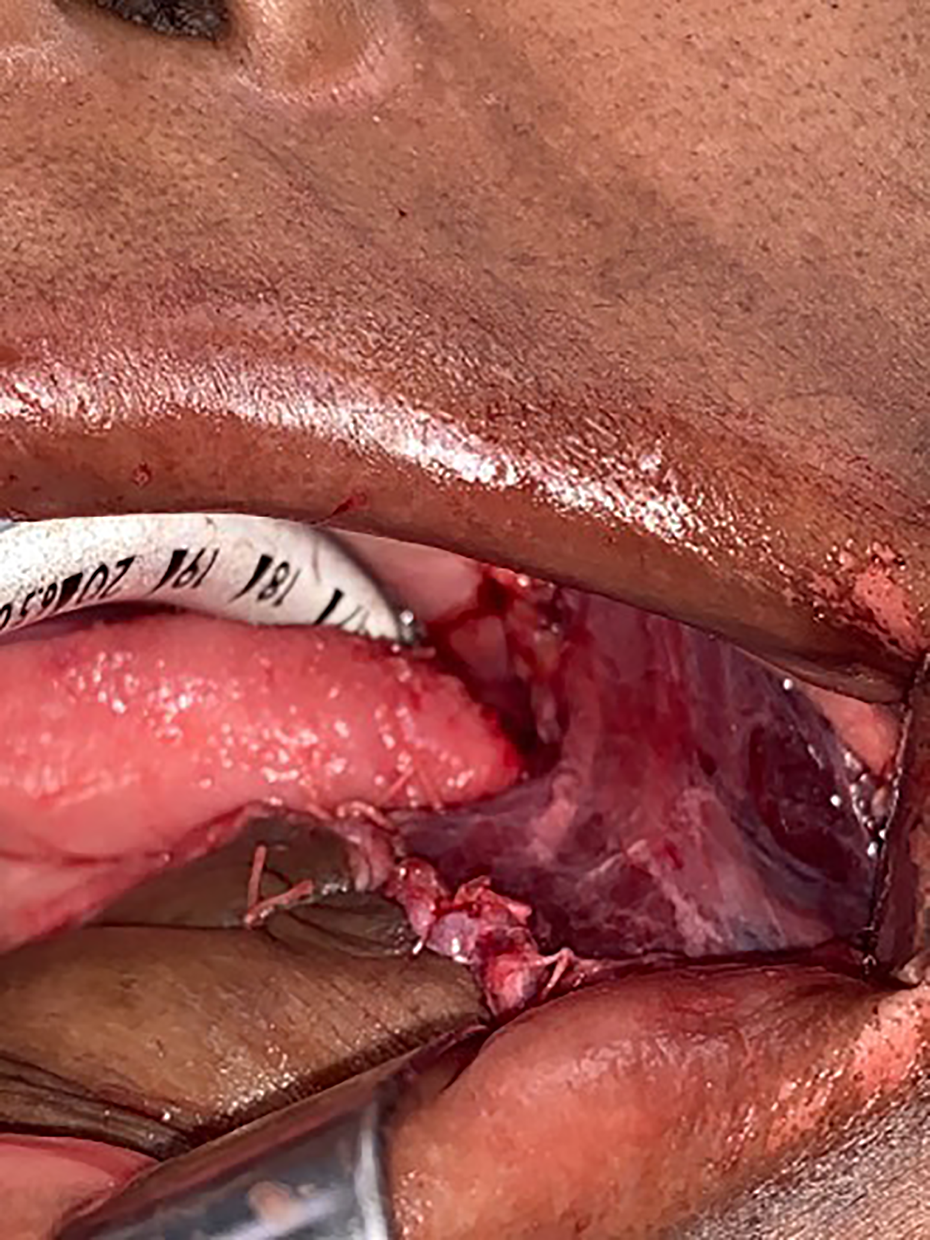
Insetting of the flap intraorally to cover the retromolar trigone region.
Careful attention is then directed toward the closure. Suspension of the temporalis fascia is recommended using 2-0 absorbable sutures or unidirectional barbed sutures. A suction drain is placed at the subgaleal plane, and the incision is closed in layers. A prophylactic antibiotic is recommended for the first 24 hours after the surgery.
Discussion
Several donor tissues can be harvested from the temporal region due to its multiple blood supplies and different tissue layers. The Temporalis Muscle (TM) flap has a robust blood supply, and therefore, it is a very reliable flap for maxillofacial reconstruction. However, one of the TM flap’s main disadvantages is the donor site morbidity, mainly the temporal hollowing. In this article, we described our surgical technique to prevent the temporal hollowing by using the posterior ⅔ of the temporalis muscle, leaving the anterior ⅓ undisturbed to maintain the lateral contour. This modification was used in 2 patients, and here we report the cases with up to 18 months of follow-up.
Temporal hollowing is a relatively common complication after surgery in the temporal region. The reported incidence varies depending on the performed procedure but, in general, it ranges between 0 and 70%. Disruption of the temporalis muscle and the fat pad are the most common causes of postoperative temporal hollowing. Cadaveric dissection has shown that the anterior inferior fossa is the deepest part of the temporal fossa and it contains the thickest portion of the temporalis muscle and fat pad. Therefore, the technique of harvesting the anterior portion of the temporalis muscle and replacing it with the posterior (thin) part of the muscle will not prevent temporal hollowing. 11 Additionally, another complication of the mobilization of the posterior part of the muscle is the risk of devascularization and denervation to the muscle that will lead to subsequent hollowing. 12
Several reports suggested using alloplastic materials such as silicon and porous polyethylene implants to prevent temporal hollowing deformity. 3,9 Others also used polymethyl methacrylate cement and hydroxyapatite cement to correct the donor site defect. 13,15 Although the results of these reports show long-lasting results, the risks of infection and foreign body reaction cannot be underestimated, particularly with the use of the cement. 16 This will result in another surgery to remove the implants or debride the chronic inflammatory tissues with a higher risk of facial nerve injury. Another alternative described is the use of secondary autologous fat grafting. 13,17 The main disadvantage of fat grafting is the unpredictable longevity and the need for multiple grafts to achieve satisfactory results. On the other hand, the use of the posterior ⅔ of the temporalis muscle described here mitigates the need for the placement of synthetic material, secondary surgery, and it showed satisfactory long term results even after radiation therapy.
Several complications are associated with the TM flap, including flap necrosis, facial nerve injury, and postoperative limited mouth opening. The TM flap has a robust and reliable blood supply; therefore, the rate of flap necrosis is less than 2%. 8,18 The main reason for flap failure is the tunnel’s inadequate size, resulting in the pedicle’s compression. Creating a large tunnel, at least 2 fingerbreadths, and removing the zygomatic arch will minimize the risk of vascular pedicle compression. In addition, with the splitting of the muscle the risk of damaging the vascular pedicle is high, therefore, careful identification of the main pedicle before splitting is mandatory to prevent catastrophic injury and loss of the flap. Another risk factor that can compromise the healing of the TM flap is the previous radiation therapy to the temporal region. Postoperative radiotherapy will result in loss of muscle volume but will not affect the flap’s survival, as demonstrated in our first case. 19
Facial nerve injury associated with the TM flap is relatively uncommon. The incidence of temporary nerve injury is approximately 10%, and permanent damage is about 3%. 3,8,18,19 Having a thorough understanding the anatomy of the temple region and careful dissection will minimize this risk. Also, we use the anterior branch of the superficial temporal artery as a landmark to identify the temporal branch of the facial nerve. In addition, performing a subperiosteal dissection at the level of the zygomatic arch and connecting the temporal region to the zygomatic region in a sub temporalis fascial plane will ensure the safety of the facial nerve. None of the 2 patients described in the article had any postoperative facial nerve weakness. After harvesting the TM flap, postoperative trismus is relatively common, with a reported incidence of 10%. 18 Physical therapy should be commenced as soon as possible to help alleviate this problem. Both patients reported here started the physical therapy within 4 weeks after the surgery. The first patient described in this article received multiple surgeries and postoperative radiotherapy, resulting in significant trismus during her postoperative course. In her last follow-up, 18 months after the surgery, she was able to open her mouth with a maximum incisal opening of 25 mm. On the other hand, the second patient did not suffer from trismus, and she can open her mouth with a maximum incisal opening of 35 mm.
Limitations of using the posterior ⅔ of the TM flap include the relatively smaller size flap and difficulty to extend the flap to cover an anterior defect such as the anterior palate or orbital exenteration defects. Therefore, the use of this modification is indicated for lateral defects and intraoral defects that do not extend to the midline. 14 Also, the described technique was performed on 2 cases and the follow-up period is relatively short. Further studies with more cases and longer follow-up is recommended
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
