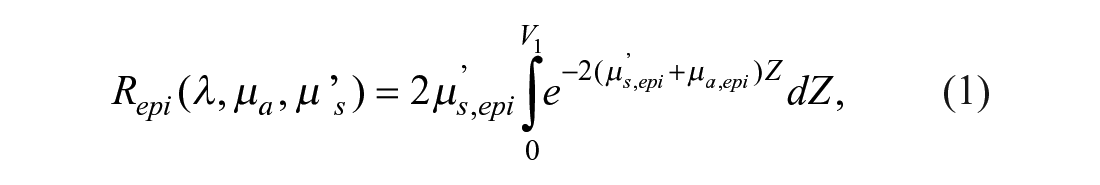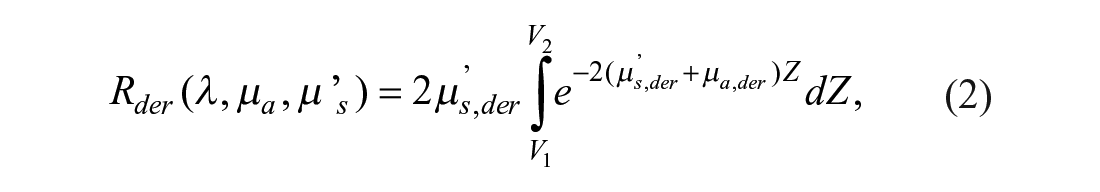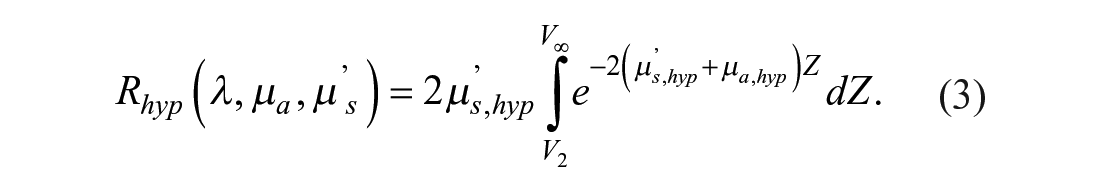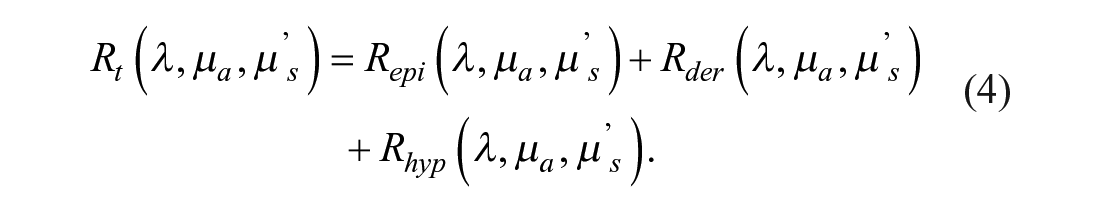Abstract
Cutaneous leishmaniasis (CL) is a parasitic disease that produces chronic skin ulcers. Although it has a worldwide presence, it is a neglected disease that still requires novel tools for its management. In order to study the use of optical tools in CL, this article presents a preliminary study of the correlation between CL histopathological and optical parameters. Optical parameters correspond to absorption and scattering coefficients obtained from diffuse reflectance spectra of treated CL in golden hamsters. Independently, histopathological data were collected from the same hamsters. As a result, after Spearman correlation and the Kruskal–Wallis test, inverse correlation was found between absorption/scattering optical parameters and inflammatory histopathological values, such as the scattering parameter related to the diameter of fibroblasts with the histopathological parameters of fibrosis, polymorphonuclear neutrophils, lymphocytes, plasmocytes, hyperplasia, and Leishmania, and the absorption parameter oxygen saturation showed a relation with the granulation tissue histopathological parameter. These correlations agree with the expected behavior of tissue composition during the healing process in CL. The results correspond to a proof of concept that shows that optical diffuse reflectance-based tools and methods could be considered as an alternative to assist in CL diagnosis and treatment follow-up.
Introduction
Leishmaniasis is a parasitic disease that produces chronic skin ulcers. Although it is present in 99 tropical countries around the world and close to 1.5 million people are at risk, including in both in-development and developed countries,1,2 cutaneous leishmaniasis (CL) is a neglected disease and few efforts have been made toward the development of novel tools for its understanding, diagnosis, and treatment follow-up. 3 Thus, the clinical criteria to determine the success of treatments is mainly based on a visual inspection and the measurement of the perimeter of the ulcer.4,5 These techniques show neither the behavior of the internal components of the skin nor the persistence of the parasite. Based on the clinical criteria of healing, a CL ulcer can be classified as cured but the parasite could still be present. In such cases, the inflammatory cells may respond to the presence of the parasite, leading to reactivation of the skin ulcer. 6 This fact limits the quality of life of patients, as well as produces delays and high costs in the health system. 7 The presented problem shows that there is a need for novel tools that allow a better understanding of the CL physiology, avoiding misdiagnosis. 3
Spectral systems measure the reflected energy from a surface illuminated by several spectral bands. Changes in a spectral signature reveal the internal composition and physiology of the surface, which changes the manner as light is absorbed, scattered, and reflected. Mathematical models and inverse modeling procedures can describe this relation. Among these models are the Kubelka–Munk model with the Fresnel equations, the radiative transfer equation, the modified Beer–Lambert law, the photon-diffusion model, and exponential models with Levenberg–Marquardt and genetic algorithms.8–15
In biomedical research, spectral systems have been used for the analysis of skin pathologies such as cancer or cutaneous ulcers due to diabetes.16–19 In these cases, the changes in a spectral signature could be associated with the number of skin chromophores and structural cells such as melanin, hemoglobin, and collagen. The quantification of those components allows a better understanding of the illness, leading to better treatment follow-up and better selection of an applied drug. Moreover, research around the use of spectral systems in biomedical applications could lead to the development of low-cost systems that could be used in low-income remote areas. As an example, it is possible to observe the works conducted by He and Wang, in which a hyperspectral smartphone-based system has been used for the monitoring of hemodynamics. 20
Despite all the advantages that spectral systems present for biomedical applications, their use in cutaneous ulcers caused by leishmaniasis has not been so much explored. This fact opens the opportunity to explore the potential use of spectral systems in CL treatment follow-up analysis. The use of optical systems in CL management can lead to the development of a prototype that could be used in primary level hospitals and remote areas, which are where most of the human population suffering from CL present.
In this article, we propose to use an optical spectrometer-based system to acquire a set of reflectance spectra from treated CL ulcers in an animal model. Spectral signatures in the visible near-infrared (VIS-NIR) region are obtained from golden hamsters (GHs) inoculated with CL in different states of healing. The GH is recommended for CL studies due to the similarity of the structure of its skin to that of humans.4,21 The processing of the acquired spectral signatures is conducted by means of an inverse three-layer diffuse reflectance model based on a genetic algorithm. As a result, optical parameters related to absorption and scattering coefficients are obtained. These parameters represent biological components in skin tissue. Also, biopsies were obtained from these hamsters, from which 24 histopathological parameters were measured. An analysis based on the Kruskal–Wallis test and Spearman correlation was conducted to identify the optical parameters related to both different states of clinical criteria of healing and histological parameters. The results show an inverse correlation between the calculated absorption/scattering parameters and inflammatory histopathological ones, such as the scattering parameter representing the diameter of fibroblasts with the histological parameters of fibrosis, polymorphonuclear neutrophils (PMN), lymphocytes, plasmocytes, hyperplasia, and Leishmania, and the oxygen saturation absorption parameter with the granulation tissue histological parameter.
Materials and Methods
Animals and CL Wound Healing
For this work, the GH was used as an animal model of CL as described previously. 4
A total of 11 GHs were used. Hamsters were inoculated with Leishmania braziliensis in their dorsal area. One month after Leishmania inoculation, hamsters were treated with 200 µg of intralesional meglumine antimoniate three times per week. After a 3-month posttreatment period, 12 diffuse reflectance spectra were acquired from 12 different points of the central part of the infected area (center of the current or previous skin ulcer). After that, hamsters were euthanized and skin samples of 8 mm in diameter and less than 5 mm thick were taken using a biopsy punch—one biopsy from one hamster. Each skin sample included both healthy and affected tissue (scarring or ulceration, depending on the case), as specified in international guidelines. 22 The samples were preserved in 10% neutral buffered formalin, labeled, and embedded in paraffin. Then, 5 µm thick sections from each biopsy were stained with hematoxylin and eosin (H&E) for analysis under a microscope. A single biopsy allows characterization of the biological changes inside the affected area. 22
For data acquisition, hamsters were anesthetized with a 9:1 mixture ratio of ketamine (50 mg/mL) and xylazine (20 mg/mL). Each hamster was positioned in dorsal decubitus, tilting it cranially to favor the displacement of the viscera and prevent its puncture. A total of 300 µL of anesthetic was slowly injected intraperitoneally with a 27-gauge needle. Then, the hair in a 3 × 3 cm area on the dorsal skin was removed using a shaving device, avoiding laceration. This procedure was approved by the Animal Ethics Committee of the Universidad de Antioquia and follows the national law of animal protection. 23
A cutaneous ulcer caused by leishmaniasis is formed according to four phases: acute phase, silent phase, active phase, and ulcerative phase. 24 In the ulcerative phase, after the inflammatory response caused by macrophages and dermal necrosis, an ulcerative lesion appears. This lesion is covered by epidermal keratinocytes killed by apoptosis and necrosis, 25 dry exudate, and dead cell development. At this stage, some T cells infiltrate mainly the injury site.26,27 In this phase, the dermis is transformed into dermal granulomas. 28
After the ulcerative phase, either a healing or chronic phase can be the next stage. In the case of the healing phase, the epidermal tissue is remodeled with a new epidermis, 29 the dermis is transformed into fibrosis tissue (scarring), and the number of parasites decreases. 30 The inflammatory reaction is chronic and forms a granulomatous infiltrate that contains lymphocytes, multinucleated giant cells, and epithelioid cells. 29 More information about CL healing can be found in the article by Nylén and Eidsmo. 24
Experimental Setup and Procedure for Spectral Data Acquisition
Diffuse reflectance spectra from hamster dorsal skin (HDS) were acquired with a typical spectrometer-based system as presented in Figure 1 . In our case, the system was composed of a 150 W QTH lamp (66088 Fiber Optic Illuminator, 150 W QTH, Oriel Instruments, Newport Corporation, Irvine, CA) with a liquid light guide (LLG) (OSL1–High-Intensity Fiber Light Source from THORLABS, Newton, NJ), an optical fiber (OF) with a core diameter of 300 µm (P300-1-SR from Ocean Optics, Largo, FL) connected to a spectrometer with a charge-coupled device (CCD) detector (HR4C3337 from Ocean Optics), and a computer with dedicated software installed (Ocean View from Ocean Optics). 31 The LLG and OF were coupled to a single probe to accomplish requirements as suggested by the American Society for Testing and Materials. 32 In this probe, in order to collect the reflected light, the OF was connected to a collimator lens. The collimator lens has a numerical aperture of 0.15 and a focal distance of 18.07 mm. The probe was in contact with the hamsters’ skin.
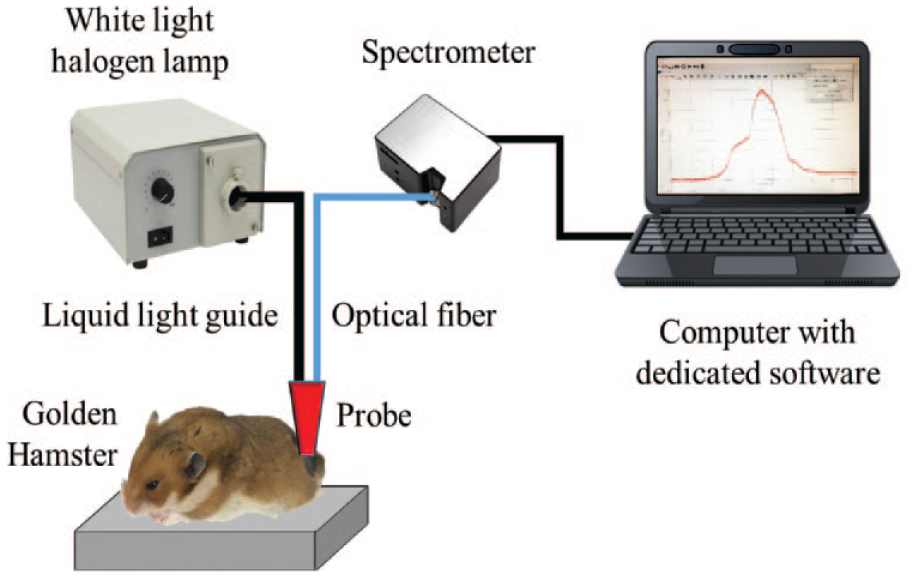
System for the acquisition of diffuse reflectance spectra from animal model skin.
The halogen lamp illuminated the HDS through the LLG, in a collimated spot area of around 3 mm diameter. After illumination, a diffuse reflectance spectral signature was acquired and guided to the spectrometer through the OF. For each hamster, 12 diffuse reflectance spectral signatures were acquired from the central part of the infected area (center of the current or previous skin ulcer). Spectra acquisition was carried out in the VIS-NIR spectral region (480–800 nm).
Spectral signatures were calibrated and filtered according to the procedure described by Botina et al. 31 For calibration, we used white and black diffuse reflectance standards, which reflect more than 99% and less than 2% of the light, respectively. Then, calibrated spectra were compared against an acceptance range to eliminate outliers and obtain only 10 diffuse reflectance spectra per hamster. The acceptance range is based on the 3 sigma rule (mean spectrum ± 3 times the standard deviation of the measured signatures). After that, the chosen spectra were filtered through a digital median filter to reduce noise. More information about this procedure can be found in the article by Botina et al. 31
Exponential Model for Diffuse Reflectance
For the purposes of this article, a diffuse reflectance model31,33,34 has been used. In this model, the diffuse reflectance of a semi-infinite turbid medium is expanded into three parts, each one representing the three main layers of the skin: epidermis, dermis, and hypodermis. These reflectances are expressed by eqs 1–3, respectively:
As presented in eq 4, the total diffuse reflectance
In these equations, µ’s,Li represents the reduced scattering coefficient in layer Li, µa,Li the absorption coefficient in layer Li, and V1, V2 the epidermis and epidermis plus dermis thicknesses, respectively.
The scattering contributions given by the epidermis, dermis, and hypodermis are calculated employing the Mie theory. 35 These values are estimated as functions of the diameter and volume fraction (VF) of the scattering particles, which in skin tissue correspond to the cells presented in Table 1 . The refractive index was set to the constant value of 1.38 for all the scattering particles and 1.36 for the media. These values are based on those reported by Lister et al. 36
Absorption and Scattering Parameters in Human Skin and Related Variables in the Light-Tissue Exponential Model.
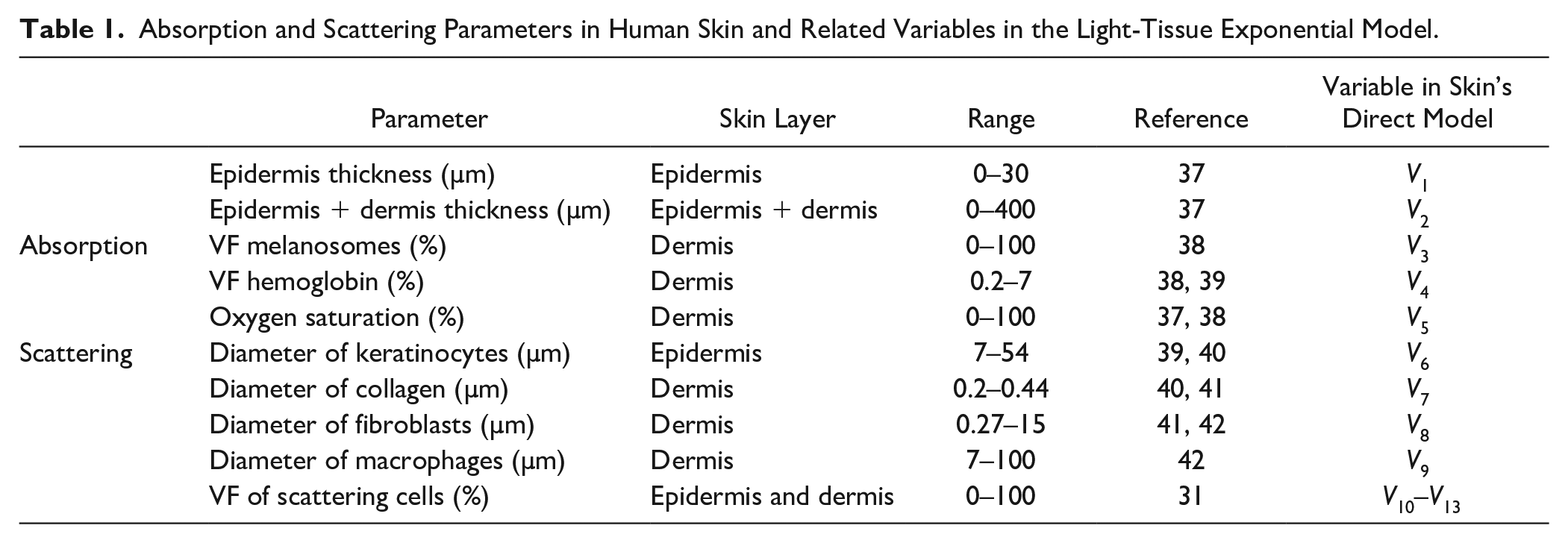
Absorption parameters are represented by the main chromophores present in the skin. Table 1 presents the main chromophores used in this model.
The absorption contributions given by the epidermis and dermis are represented by eqs 5 and 6, respectively:34,38
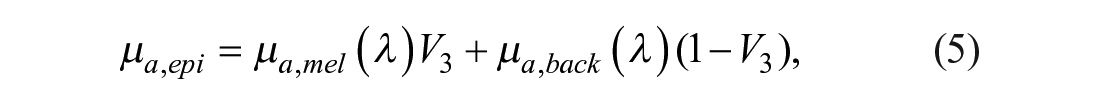
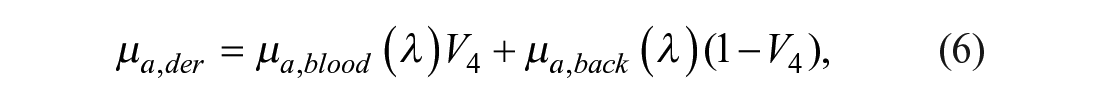
where µa,mel (λ) corresponds to the absorption spectrum of melanin, 40 V3 is a variable related to the VF of melanosomes, and µa,back is related to the background absorption, which can be described by eq 7: 34
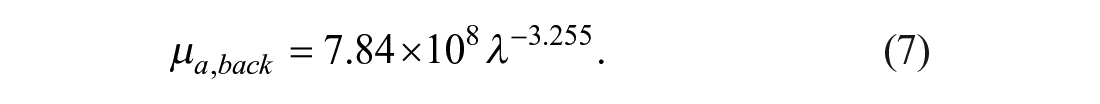
µa,blood (λ) is the blood absorption due to oxyhemoglobin µa,oxy and deoxy-hemoglobin µa,deoxy (eqs 8–10): 34
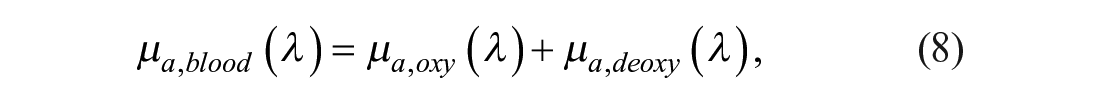
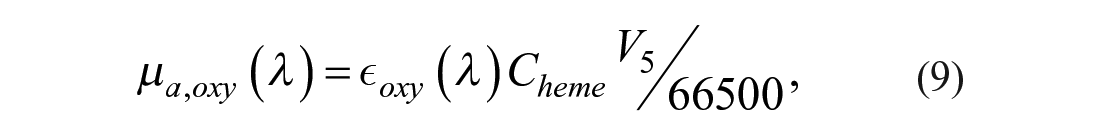
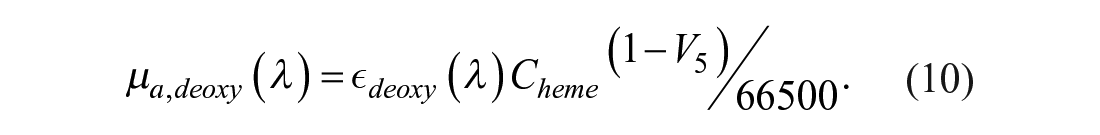
µa,hyp is the absorption component of hypodermis calculated by Van Veen et al. 45
Inverse Model Based on a Genetic Algorithm Approach
The inversion model uses the measured reflectance for the estimation of skin scattering and absorption parameters considered in the previous section. The inversion model employs an optimization algorithm to adjust the skin parameters in the diffuse reflectance model until the simulated spectral signature is very close to the measured reflectance. In the literature, several optimization algorithms are used for inversion of light-tissue interaction models, such as Levenberg–Marquardt,16,46 k-nearest neighbors, 47 Newton–Raphson, 48 genetic algorithms,49–51 and random forests. 52 In this study, we use a genetic algorithm for the estimation of skin parameters. A genetic algorithm is a robust approach that offers good performance in several applications. 53
In the implemented genetic algorithm, first a random population of 20 individuals was obtained per each absorption and scattering variable, according to the valid range presented in Table 1 .54,55 As the second step, simulated spectra (Rs) were generated by evaluating the diffuse reflectance model in the parameter values randomly selected; then, Rs was compared with the measured signatures (Rm) computing the MSE according to eq 11: 56
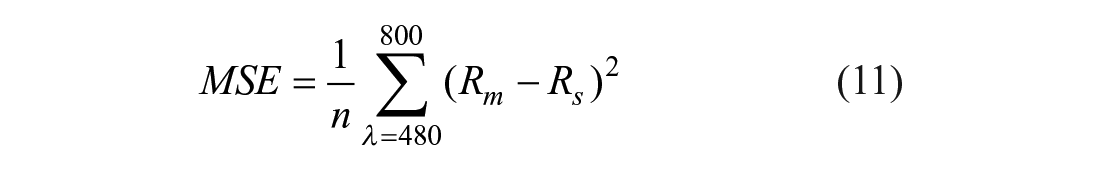
where n corresponds to the number of points in one diffuse reflectance spectrum. In our case, n corresponds to 321 points (800–480); Rm is the measured diffuse reflectance, and Rs is the simulated one. MSE corresponds to the cost function of the algorithm.
The best individuals are those with MSE close to zero, which are selected during the evolutionary stage of the genetic algorithm in step 3. Finally, in a fourth step, crossover of 70% and mutations of 80% are performed to obtain a new generation of individuals. This procedure is repeated by 200 generations until convergence to a minimum value, which in our case corresponds to an MSE value of less than 0.2 × 10−3. More information about this inverse modeling procedure can be found in the literature.50,53–55
Clinical Phenotype of Healing and Histopathologies
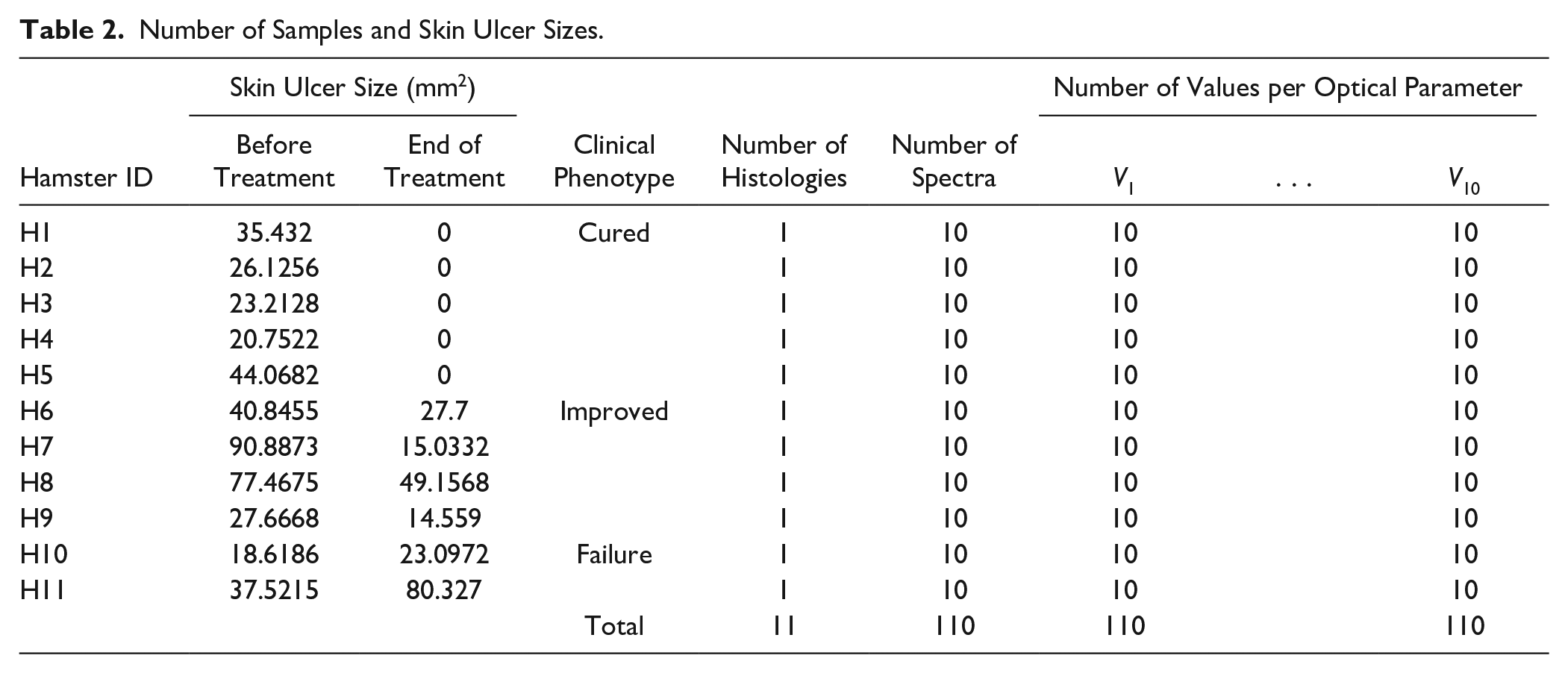
The size of the skin ulcer is measured with a digital caliper at the beginning and at the end of the treatment. From these two measurements, a difference is calculated: the measurement at the end of the treatment is subtracted from the measurement at the beginning. Based on this difference, three clinical phenotypes are established: cure, improvement, and failure. 4 A skin ulcer is cured when the measurement at the end of the treatment is zero; it is improved when the difference is higher than zero; and it is a failure when the difference is lower than zero. It is important to bear in mind that before treatment, skin ulcers should have a size of at least 4 × 4 mm. Table 2 presents the hamsters’ skin ulcer sizes measured before treatment.
Number of Samples and Skin Ulcer Sizes.
Histopathologies were obtained from the biopsies acquired from the dorsal area of the GH. The biopsies covered both healthy and affected skin areas. Biopsies were fixed in 10% formalin and embedded in paraffin. The fixed tissues were stained and examined under an optical microscope to study the microarchitecture, the characteristics of the cellular infiltrate, and the presence or absence of parasites. 4 Based on these observations, the pathologist could compare the tissue and determine whether there was an increase or decrease in 24 histological parameters of the tissue under evaluation. These 24 histological parameters correspond to vacuolar degeneration, fat and cloudy degeneration, atrophy, fibrosis, hyperplasia, edema, congestion, pigment, protein cylinders, necrosis, cardiomegaly, binucleation, minerals, apoptosis, hemorrhage, thrombi, PMN, eosinophils, macrophages, lymphocytes, plasmocytes, granulation tissue, and Leishmania. These parameters are rated based on an arbitrary unit scale from 1 to 6, where grade 1 means without injury; grade 2, mild injury; grade 3, between mild and moderate injury; grade 4, moderate injury; grade 5, between moderate and severe injury; and grade 6, severe injury. 57
Data Processing and Analysis Procedure
Hamster data segregation: Both hamsters’ histopathological and optical data were grouped into three groups according to the clinical phenotype of skin ulcer healing. The first group consisted of cured hamsters; group 2, hamsters showing improvement as a result of treatment; and group 3, those hamsters for which the treatment finished in failure. The number of samples is summarized in Table 2 .
Processing of the diffuse reflectance parameters: 11 hamsters were used in this study. From each hamster, 10 spectral signatures were obtained from different, randomly selected places as explained in previous sections. From each spectral signature, 10 optical parameters were calculated using the genetic algorithm so that, for each hamster, 10 values of each optical parameter were obtained. Taking all 11 hamsters into account, this resulted in 110 different values for each optical parameter. Next, two sets of analysis of the optical parameters were processed as follows:
Set 1: The 110 values of an optical parameter were grouped according to the clinical phenotype of healing as follows: the “cured” group, 50 values; the “improved” group, 40 values; and the “failure” group, 20 values. After that, the Kruskal–Wallis nonparametric test was carried out. The Kruskal–Wallis test was conducted to identify the optical parameters whose values differed among clinical healing phenotypes. Since there were 10 different optical parameters, the set of analyses was carried out 10 times—one per parameter.
Set 2: For one hamster, the mean value of each of the 10 optical parameters was calculated. Since there were 11 hamsters, this resulted in 11 mean values for one optical parameter. Then, these 11 mean values of one parameter were classified according to the clinical phenotype of healing as follows: the “cured” group, five mean values; the “improved” group, four mean values, and the “failure” group, two mean values. After that, all mean values, independent of group, were correlated against the histopathological parameters.
Optical versus histopathological parameters: The relation between the mean optical and histological parameters was evaluated utilizing the Spearman correlation test. From the correlation test, two parameters were obtained: p value and rho. From this, first, the most significant relations were identified with a p value around 0.05 and an absolute rho value higher than 0.6. Then, an analysis was conducted by a physician to infer if the identified optical–histological relations had reasonable biological meaning.
Results and Discussion
Distribution of Histopathological Parameters According to the Clinical Phenotype of CL Healing
Each of the 24 histopathological parameters obtained from the 11 hamsters was assigned to a proper group according to the skin ulcers’ clinical phenotype of healing: five hamsters were classified as cured, four as improved, and two as a failure. These groups of data were evaluated employing the Kruskal–Wallis nonparametric test. The most relevant results (p ~ 0.05) of this test are presented in Figure 2 . The parameters not presented in the figure correspond to those histopathological parameters whose values were the same among all three clinical phenotypes.
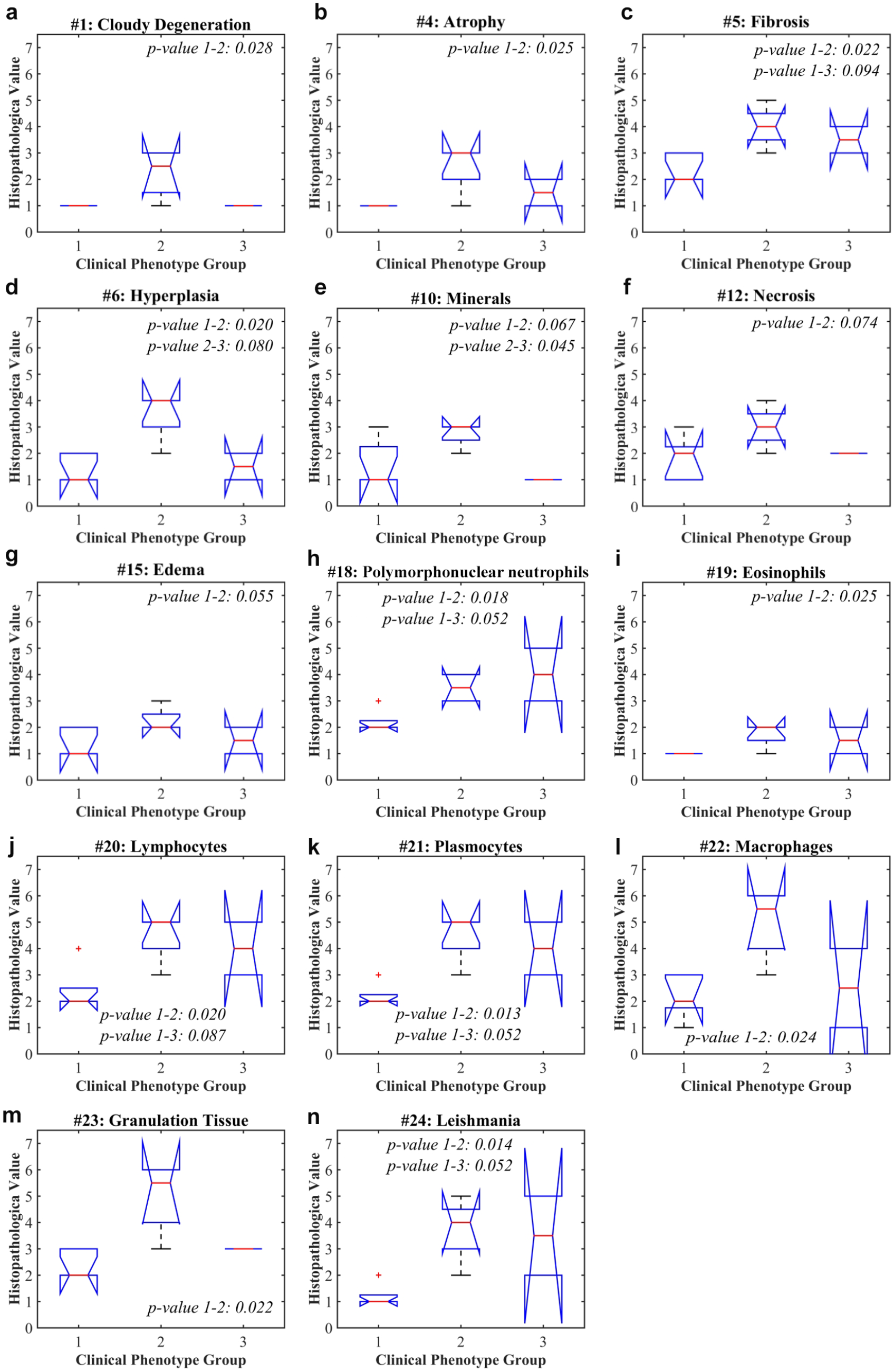
Kruskal–Wallis nonparametric test of histopathological parameters grouped according to the clinical phenotype of healing. Group 1, cure; group 2, improvement; group 3: failure.
In
Figure 2
, it is possible to observe that among the 24 histopathological parameters, 13 present differences between clinical phenotype of healing: 5 parameters present differences between cure and failure (fibrosis [
Fig. 2c
], PMN [
Although the presented histopathological parameters showed differences among skin ulcer clinical phenotypes of healing, it is possible to observe that some hamsters present histopathological values out of the mean value of a specific phenotype group. For example, we can observe lymphocyte values for the cure clinical phenotype group (
Hence, it is important to have a tool that not only allows phenotype classification but also allows estimation of changes in biological skin parameters. The evaluation of changes in biological skin parameters can lead to a greater understanding of CL and better treatment management and follow-up.
Changes in the Diffuse Reflectance Spectral Parameters According to the Clinical Phenotype of CL Healing
As with the histopathological parameters, each one of the diffuse reflectance parameters obtained from the 11 hamsters was assigned to a proper group according to the skin ulcers’ clinical phenotype of healing. These groups of data were evaluated using the Kruskal–Wallis nonparametric test. The most relevant results (p ~ 0.05) of this test are presented in Figure 3 . The parameters not presented in the figure correspond to those optical parameters, whose values were the same among all three clinical phenotypes. Figure 3 also presents a table with the corresponding p values.
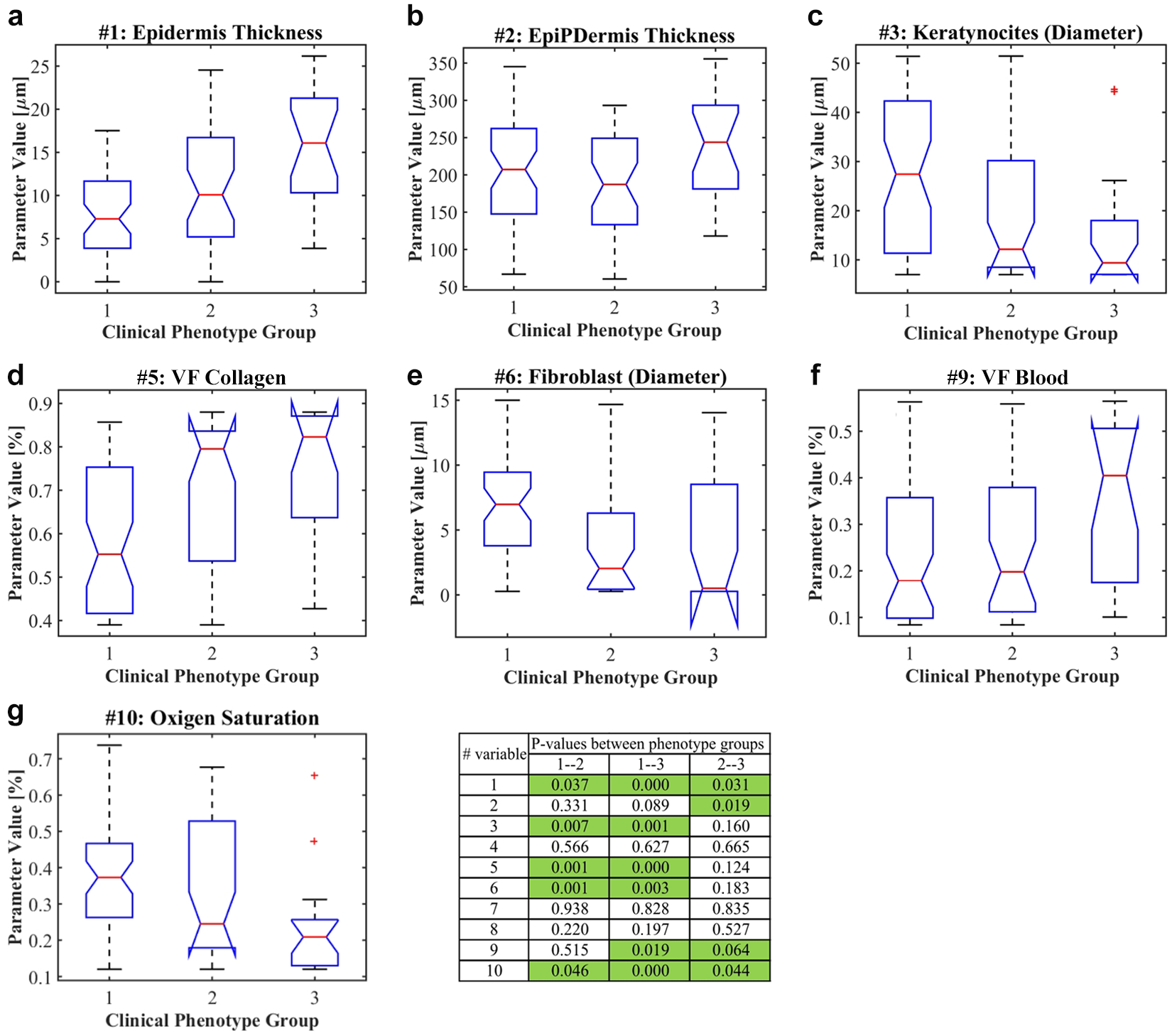
Kruskal–Wallis nonparametric test of diffuse reflectance parameters grouped according to the clinical phenotype of healing. Group 1, cure; group 2, improvement; group 3, failure.
In
Figure 3
, it is observed that the following diffuse reflectance parameters present differences between cured and failure hamsters: epidermis thickness (
Fig. 3a
), keratinocytes (
Distribution of the Optical Diffuse Reflectance Spectral Parameters According to the Histological Parameters
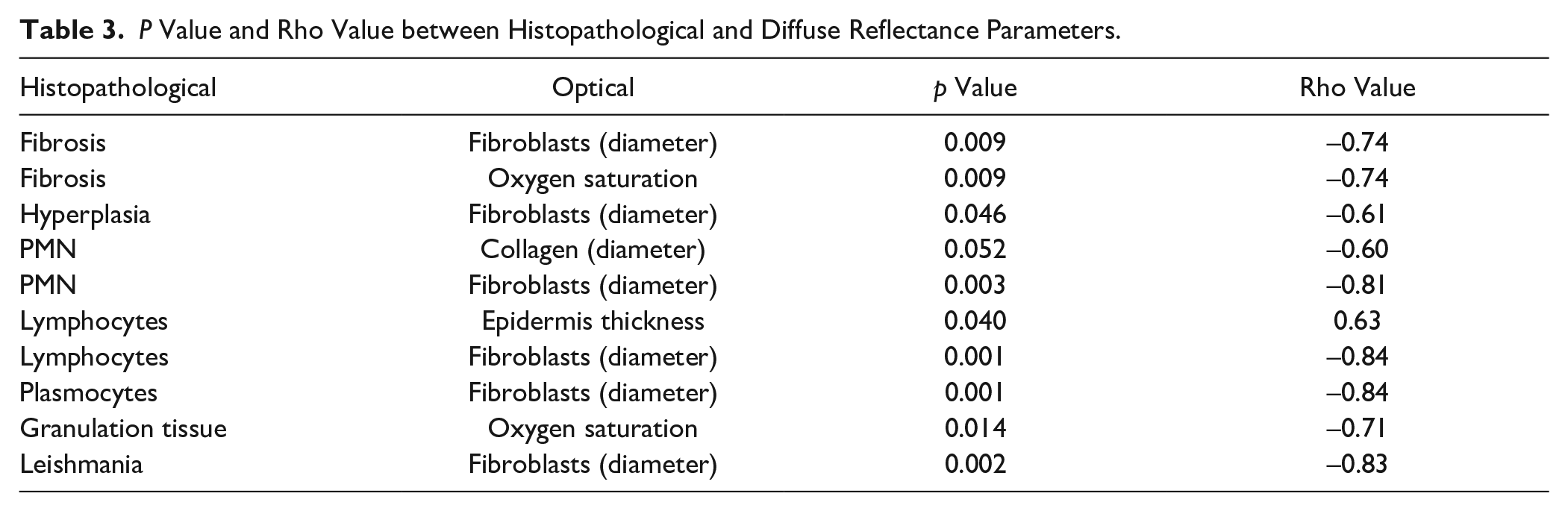
Table 3 and Figure 4 show the most significant results of the correlation analysis carried out between the histopathological and optical parameters (p < 0.05 and |rho| > 0.6).
P Value and Rho Value between Histopathological and Diffuse Reflectance Parameters.
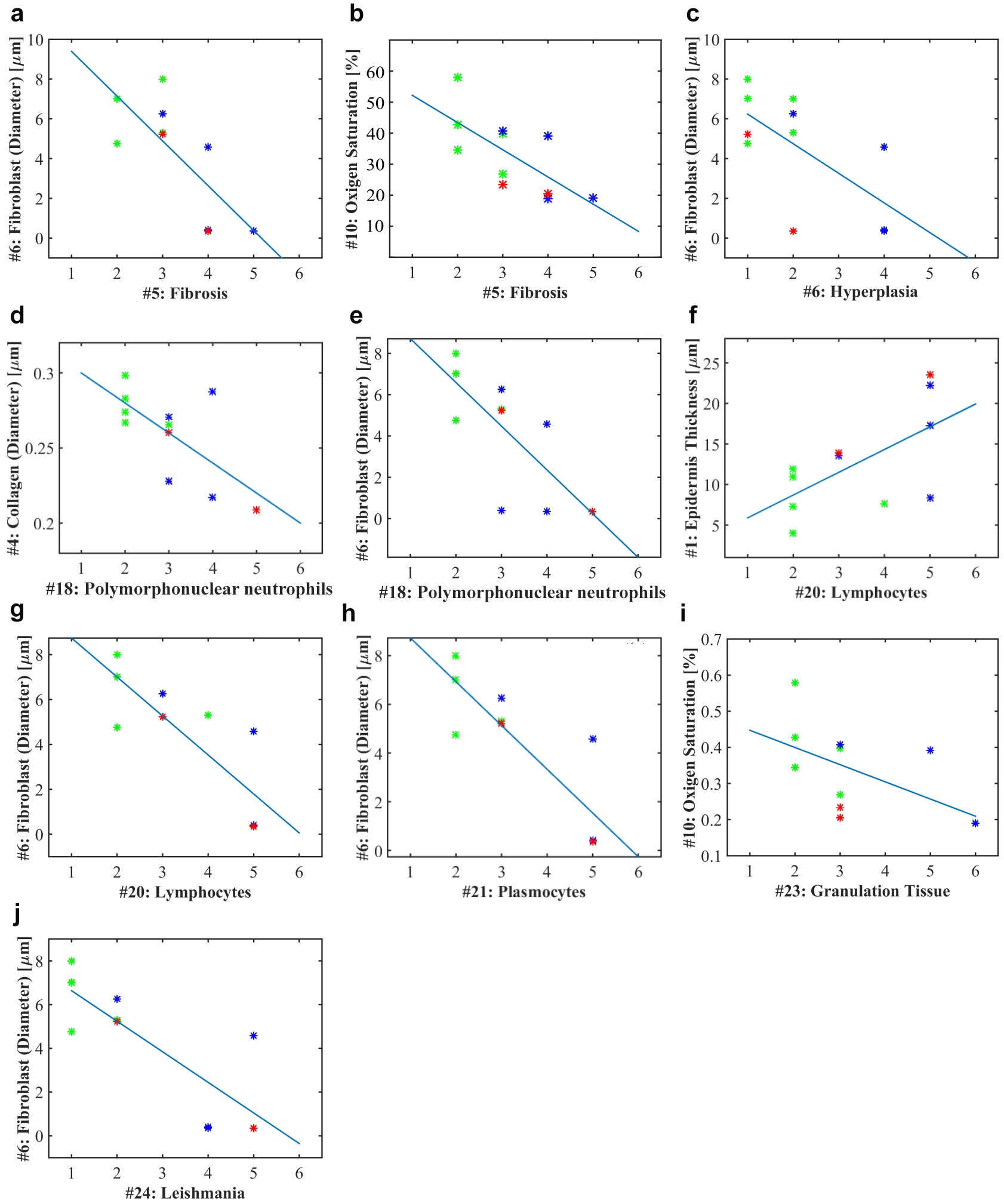
Results of the correlation test between histopathological parameters and absorption/scattering optical parameters. Green dots, cured hamsters; blue dots, improved hamsters; red dots, failure hamsters.
The diffuse reflectance parameter of fibroblasts (diameter) has an inverse correlation with the histopathological parameters of fibrosis ( Fig. 4a ), hyperplasia ( Fig. 4c ), PMN ( Fig. 4e ), lymphocytes ( Fig. 4g ), plasmocytes ( Fig. 4h ), and Leishmania ( Fig. 4j ). The oxygen saturation optical parameter shows an inverse correlation with fibrosis ( Fig. 4b ) and granulation tissue ( Fig. 4i ) histopathological parameters. The optical parameter epidermis thickness presents a direct correlation with the lymphocyte histopathological parameter ( Fig. 4f ). Finally, the collagen optical parameter (diameter) has an inverse correlation with the PMN histological parameter ( Fig. 4d ). An explanation of these relations is presented as follows:
1. PMN versus diameter of collagen: According to a previous study, 31 a higher diameter of collagen is related to the formation of granulomas. Collagen production allows connective tissue matrices to emerge. The appearance of granulomas is another characteristic of tissue pathology after healing and remodeling of the ulcer in scar tissue. In the case of PMN, some studies show that a decrease of PMN could have a beneficial effect on reepithelialization. 58 Therefore, the inverse relationship found between these two parameters has some meaning from the biological point of view.
2. A similar analysis could be carried out between PMN and fibroblasts; higher values of fibroblasts are related to newly formed granulation tissue that covers the wound area, which is characteristic in the curation phase of wound healing.58,59
3. Fibrosis versus fibroblasts: Fibrosis is related to the infection process. 4 Therefore, a higher process of fibrosis is consistent with lower values of fibroblasts. In the case of oxygen saturation, less fibrosis could be related to more oxygen saturation.
4. Lymphocytes versus epidermis thickness: According to Eming et al., “in the late inflammatory phase of wound repair, T lymphocytes appear in the wound bed.” 58 T cells are the main lymphocytic infiltrate in leishmanial skin lesions. Also, these cells are central in controlling parasite replication, tissue destruction, and tissue pathology. This inflammatory response is related to an active reparation of the ulcer. With respect to the “epidermis thickness” optical variable, this variable is related to the thickness of the first layer in the skin. 31 Higher values of epidermis thickness could be associated with the presence of a thicker scab, which is characteristic in skin ulcers that are still in an active ulcerative phase. Therefore, it is possible to infer that there could be a direct relationship between the presence of lymphocytes and epidermis thickness.
5. Lymphocytes versus fibroblasts: The high inflammation process caused by T lymphocytes, which is characteristic for ulcers being in an active phase, could be related to less content of fibroblasts.
6. Plasmocytes versus fibroblasts: Plasmocytes are associated with antibodies related to the curation process (such as B cells); plasmocytes are present in active ulcers. Therefore, fewer plasmocytes could be related to a higher number of fibroblasts.
7. Hyperplasia versus fibroblasts: The inverse relationship between these two variables shows that in the connective phase all inflammatory processes finish in a reparation.
8. Granulation tissue versus oxygen saturation: Granulation tissue is present in the proliferation stage of a skin ulcer. This explains the inverse relationship with oxygen saturation: higher values of oxygen saturation are typical in healed skin ulcers. The proliferation stage consists of granulation tissue formation, angiogenesis, and extracellular matrix (ECM) synthesis. 60 Granulation tissues are made of elastin, hyaluronic acid, procollagen, and proteoglycans, 60 forming a structural repair scaffold to allow vascular growth. At the end of this stage, actin-rich myofibroblasts assist in the initiation of wound contraction. 60 The final phase of remodeling consists of wound closure, collagen synthesis, reepithelialization, and scar tissue formation. 61
9. Leishmania versus fibroblasts: More parasites are related to less fibroblasts. As seen from the previous relationships, high numbers of fibroblasts are associated with the curation phase of wound healing. Therefore, it is expected that in healed ulcers, where the presence of Leishmania is low, the fibroblast content is higher.
Other histopathological–optical correlation results that were considered present the following results: The optical parameter oxygen saturation shows an inverse correlation with PMN, lymphocytes, plasmocytes, and Leishmania. The optical parameter epidermis thickness presents a direct correlation with the PMN, plasmocytes, lymphocytes, and Leishmania. The collagen optical parameter (VF) has a direct correlation with the PMN histopathological parameter. Finally, the melanin (VF) optical parameter shows an indirect correlation with the fibrosis histological parameter.
Possible Changes in the Optical Spectra in Hamsters Infected with L. amazonensis
There could be differences from the histological point of view between hamsters infected with L. braziliensis and Leishmania amazonensis. The histopathological values vary depending on the species and strain of the animal species, as well as on the Leishmania species.
For example, in CBA mice infected in the skin of the footpad of an upper limb with L. amazonensis, a mixture of inflammatory cells, including macrophages, lymphocytes, neutrophils, and eosinophils, were observed as infiltrating cells. 62
In the work presented by de Oliveira Cardoso et al., 63 “the follow-up of CBA mice infected with L. amazonensis showed on the 60th day after infection that the mice developed ulcerative lesions filled with parasitized macrophages.” Necrosis, eosinophils, and fibroblasts were observed as well. In the case of C57BL/10 mice, the lymph nodes “were filled with plasma cells, eosinophils, and macrophages that contained amastigotes.” “DBA/2 mice also presented ulcerative lesions, but parasitized macrophages were rarely observed.” Also, in CBA mice, it is reported that at 70 days postinfection, “fibroblast proliferation and collagen formation appear throughout the lesion.” 64
In the case of the studies presented by Robledo et al., during a 10-day follow-up, “skin samples of hamsters infected with L. amazonensis showed a slight level of plasmocytes and PMN cells accompanied by moderate infiltration of lymphocytes; also a mild or severe infiltration of macrophages was found.” 4
According to our findings presented in Table 3 , the mentioned histopathological changes caused by L. amazonensis would make the following optical parameters vary: oxygen (related to the histological parameter necrosis), fibroblasts, macrophages, and collagen. Compared with Table 3 , all of these optical values are relevant in our study of L. braziliensis, so that it is not possible to immediately infer if a significant difference could be found between the optical parameters present in L. braziliensis with respect to L. amazonensis. However, further studies should be carried out in order to analyze if the correlation rho values could present any changes between optical and histological parameters.
Possible Histopathological and Optical Spectra Changes, Which Could Be Induced by Antimony in Hamsters Infected with L. amazonensis
As presented by Robledo et al.,
4
hamsters infected with L. amazonensis presented a decrease in PMN, macrophages, fibrosis, congestion, and lymphocytes, in comparison to untreated infected hamsters. From this study, it is possible to infer that the histopathological effect of meglumine antimoniate (in a dose of 120 mg/kg) is rather to decrease the values of the histopathological parameters, making them closer to the values obtained in normal skin. In this regard, as presented in the correlation graphics (
Conclusions
CL is a chronic disease in which follow-up is mainly clinical and subject to the variability of the observer. This problem shows that there is a need for novel tools that allow a better understanding of the changes in skin ulcers’ biological parameters that are crucial in the repair and scarring processes of wound healing. This fact would lead to a better approach to the illness with improved treatment follow-up.
In order to present the potential use that optical tools have for the assessment of cutaneous ulcer analysis, this article presents the relationship between histopathological characteristics of CL and optical parameters obtained from diffuse reflectance spectra. The study was conducted in GHs inoculated with L. braziliensis and treated for 1 month. Three months after treatment, both spectral data and biopsy samples of the affected skin area were acquired. Spectral data were processed using the inversion of a light-tissue interaction model. Absorption and scattering parameters were obtained from this inverse modeling procedure at each acquired spectrum. The biopsies were analyzed, and 24 histopathological parameters were obtained per hamster. Optical and histopathological results were processed through the nonparametric Kruskal–Wallis and Spearman correlation tests.
As a result, regarding the possible significant relationships, there was a direct relationship between the thickness of the epidermis and inflammatory parameters such as PMN, lymphocytes, and plasmocytes (rho > 0.6, p < 0.05); an inverse correlation between the diameter of the fibroblasts and the same inflammatory parameters (rho < –0.8, p < 0.009); and an inverse correlation between the oxygen saturation and inflammatory parameters such as PMN, granulation tissue, lymphocytes, and plasmocytes (rho < –0.6, p < 0.05).
These correlations reflect the expected behavior of the different cellular components of the tissue during the healing process in leishmaniasis, in which the inflammatory reaction mediated by PMN, lymphocytes, plasmocytes, and granulation tissue decreases, as does the presence of the Leishmania parasite due to activation of cytokines and growth factors that attract immune and nonimmune cells. Hyperplasia and fibrosis also decrease in the final stage of the healing process with the remodeling of a new epidermis; the synthesis of collagen and melanin; increased oxygen saturation; and the transformation of the dermis into fibrosis tissue that leads to scar formation and wound closure.
Based on these results, optical tools can be considered as a promising alternative to improve the management and follow-up of ulcers caused by leishmaniasis. Future work will seek temporal analysis and the use of machine learning techniques for data clustering.
Footnotes
Acknowledgements
The authors would like to acknowledge the support given by Dr. Carlos Muskus from PECET Laboratory in Universidad de Antioquia (UdeA), the histopathological laboratory of UdeA, the PECET bioterium Staff (Medellín-Colombia), and Dr. Jorge Arevalo from Universidad Peruana Cayetano Heredia (Lima-Perú).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge the financial support given by MINCIENCIAS (Colombia), Instituto Tecnológico Metropolitano, Universidad de Antioquia, Universidad Pontificia Bolivariana, and Kinetics Systems S.A.S. (Medellín-Colombia), under project number 115077757186.