Abstract
Plastic pollution is the accumulation of plastic objects in the Earth’s environment and is a global problem of increasing importance. The laboratory and health care industries contribute to this problem by the widely accepted single use of plastics, including microtiter plates used for compound testing. At AstraZeneca, we predict the use of more than 45,000 384-well and more than 11,000 1536-well microtiter plates per year. IonField Systems has developed a microplate cleaning system (MCS) powered by PlasmaKnife technology that uses cold plasma to clean microtiter plates. AstraZeneca proposed the use of this system for standard ANSI (https://slas.org/resources/information/industry-standards/) microtiter plate regeneration. Here we present the results of an evaluation using a model system involving the cleaning of plates following an enzyme-based biochemical assay, as well as the software and hardware enhancements that were incorporated into the production PlasmaKnife MCS. The method involved determining the level of inhibition achieved by residual compound following different cleaning protocols and showed that cleaning achieved in about 2 min was sufficient to remove trace compound contamination. Future work will focus on assessing the number of regeneration cycles that can be reliably achieved.
Introduction
Plastic pollution is an increasingly serious problem for today’s society, with most of Earth’s environments affected by the presence of plastic, from micro- to macro-scale particles.1–4 About 8.3 billion tons of plastic has been produced since the 1950s, and only about 9% of this plastic has been recycled, 12% has been burned, and the remaining 79% has ended up in a landfill or the environment. 5 Within the pharmaceutical industry, there has been a steady rise in the amount of plastic biohazard waste produced, as operations within laboratories are largely built on the accepted use of disposable products. Every day, vast amounts of plastic, including pipette tips, microplates, cell flasks, and other items, are used and thrown away after a single use, only to be incinerated or sent to a landfill. This adversely contributes to the high levels of plastic pollution with a negative impact on the environment. In 2014, it was estimated that 5.5 million tons of laboratory plastic waste was generated by some 20,500 research institutions worldwide. 6
As the problem is becoming increasingly recognized within the industry, scientists are trying to address the issue by applying the 3 Rs strategy—Reduce, Reuse, Recycle—to develop solutions aimed at reducing the amount of plastic waste generated within laboratories. These actions include adapting protocols to use less labware, universities setting up recycling programs preventing plastics from going to landfill, and novel solutions offered by the companies that manufacture labware.
Within AstraZeneca, we have been exploring practical solutions to help us reduce our plastic footprint. Currently, our high-throughput screening (HTS) center performs about 20 HTS campaigns per year, testing up to 2 million compounds per campaign, and using more than 40,000 plastic microtiter plates in the process. Process efficiency and cost, as well as the impact on the environment around us, have led us to continually explore the best technological advances to progress the screening field, using a wide range of commercial equipment for screening purposes,7,8 including reagent dispensers, plate washers, 9 and multiparameter plate readers. 10 In this report, we have worked with IonField Systems (IFS; Moorestown, NJ) to evaluate and develop technology to clean plastic microtiter plates so that they can be reused multiple times. IFS has developed a microplate cleaning system (MCS) powered by PlasmaKnife technology that uses a combination of liquid wash and cold-plasma exposure 11 to clean used assay plates. For microtiter plates to be reused, they must be functionally indistinguishable from new plates when in repeated use within and between assays. Here we present data demonstrating success in cleaning microtiter plates used in biochemical assays, allowing them to be reused in subsequent HTS campaigns. Optimization and integration of this methodology will help us to take a significant step toward operating in a more environmentally conscious manner while continuing to produce reliable, high-quality data.
Materials and Methods
Biochemical Absorbance Assay
Enzyme activity for human recombinant arginase (His-TEV full-length arginase 1 protein in 50 mM HEPES, pH 8.0, 100 mM KCl, 100 µM MnCl2, 2 mM TCEP, 5% glycerol, custom synthesis supplied by Viva Biotech, Shanghai) generated a thiol group from the substrate thioarginine; this was detected by Ellman’s reagent (5,5′-dithiobis-[2-nitrobenzoic acid] or DTNB [D218200]; Sigma-Aldrich, St. Louis, MO), and product was measured by absorbance at 405 nm. Briefly, compounds in DMSO (100 % v/v) were acoustically dispensed in 10-point concentration format (each compound was tested at 18.3 nM to 200 µM in a single well for each concentration) using an Echo 555 (Labcyte, San Jose, CA) into 1536-well polystyrene clear microtiter plates (Greiner Bio-One, Kremsmünster, Austria). A Multidrop Combi (Thermo Scientific, Waltham, MA) was used to add 3 µL/well enzyme/DTNB, and plates were then incubated at ambient temperature for 30 min. Thioarginine (custom synthesized by Pharmaron, Shanghai, China), 3 µL/well, was then added and plates incubated for a further 50 min at ambient temperature before measuring absorbance at 405 nm using a PHERAstar (BMG, Ortenberg, Germany) plate reader. Final buffer conditions were 50 mM HEPES (H3375, Sigma-Aldrich), pH 7.5, 0.0005% Brij (B4184, Sigma-Aldrich), 100 µM MnCl2 (M3634, Sigma-Aldrich). The final enzyme concentration was 5 nM, and the final concentrations of thioarginine and DTNB were 0.72 mM and 0.4 mM, respectively. Plates that had been used in the assay were then cleaned using the PlasmaKnife MCS. To test the efficiency of this process, cleaned plates were retested in the biochemical absorbance assay by adding enzyme, DTNB, and thioarginine, followed by measuring absorbance.
PlasmaKnife MCS Components
The PlasmaKnife MCS consists of six hardware components. The three main components are the rinse station, plasma station, and the human machine interface (HMI) with an integrated touchscreen (

The PlasmaKnife microplate cleaning system. (
The secondary components include an enclosure for the rinse station’s high-pressure pump, a TipCharger controller used to generate plasma, and an optional automated ethanol/water mixing system. The PlasmaKnife system includes two software modules. The first module is the programmable operating system running on the control module. The second module is the application programming interface needed for robotic integration. The hardware for the application programming interface communications is integrated in the control module.
Standard Plate-Cleaning Process Using Prototype PlasmaKnife MCS
Used assay plates were cleaned using prototype rinse and plasma stations supplied by IFS. Following a 5 s spin by the rinse station to centrifugally empty the well assay contents, plates were washed using 14% ethanol by passing under the water knife for 30 s. The water knife sprays a thin (2/1000th inch) layer of wash fluid over the upper surface of the microplate. The pressure used is up to 150 PSI (or 10.34 Bar). The volume dispensed is user programmable, with a typical volume per microplate of 0.3 to 0.4 L. The water knife is positioned across the breadth of the microplate at 6.5 mm above the top surface. Following the wash step, plates were centrifuged at 1500 rpm (relative centrifugal force = 254g) for 5 s before being treated with plasma for up to 60 s. The prototype unit used an eight-channel plasma-generation head that spans the breadth of the microplate and evenly blows the plasma over the surface. Plasma-treated plates were then left to air dry before being retested in the biochemical absorbance assay as empty cleaned plates (with no further compound additions).
Data Analysis
All assay plates (1536-well format) included control wells. Negative control wells (0% inhibition; 18 wells in total) contained a matched concentration of DMSO (1% v/v) to compound wells. Positive control wells (100% inhibition; 18 wells in total) contained an assay-specific control compound. All screening data were analyzed using Genedata Screener (Basel, Switzerland). The activity of each test compound was normalized to the median values of negative control wells. IC50 values were calculated by nonlinear regression fitting to a four-parameter logistic model using the automated curve-fitting technology in Genedata Screener. GraphPad Prism (GraphPad Software, San Diego, CA) was used to analyze the percentage inhibition data exported from Genedata Screener to compare the effect of modifications to the plate cleaning process.
Results and Discussion
Initial Data Using Prototype PlasmaKnife MCS
The biochemical absorbance assay described in the Materials and Methods section was used for screening chemical inhibitors of a target enzyme, ARG1, and using this experimental model, we generated a set of used assay microtiter plates that tested a set of 78 active known pharmacology compounds, with a pIC50 of 3.7 to 7.5. Using a 10-point concentration format, 780 (plus 36 control wells) of 1536 wells were used per plate for all experiments, as can be seen in the heat map (
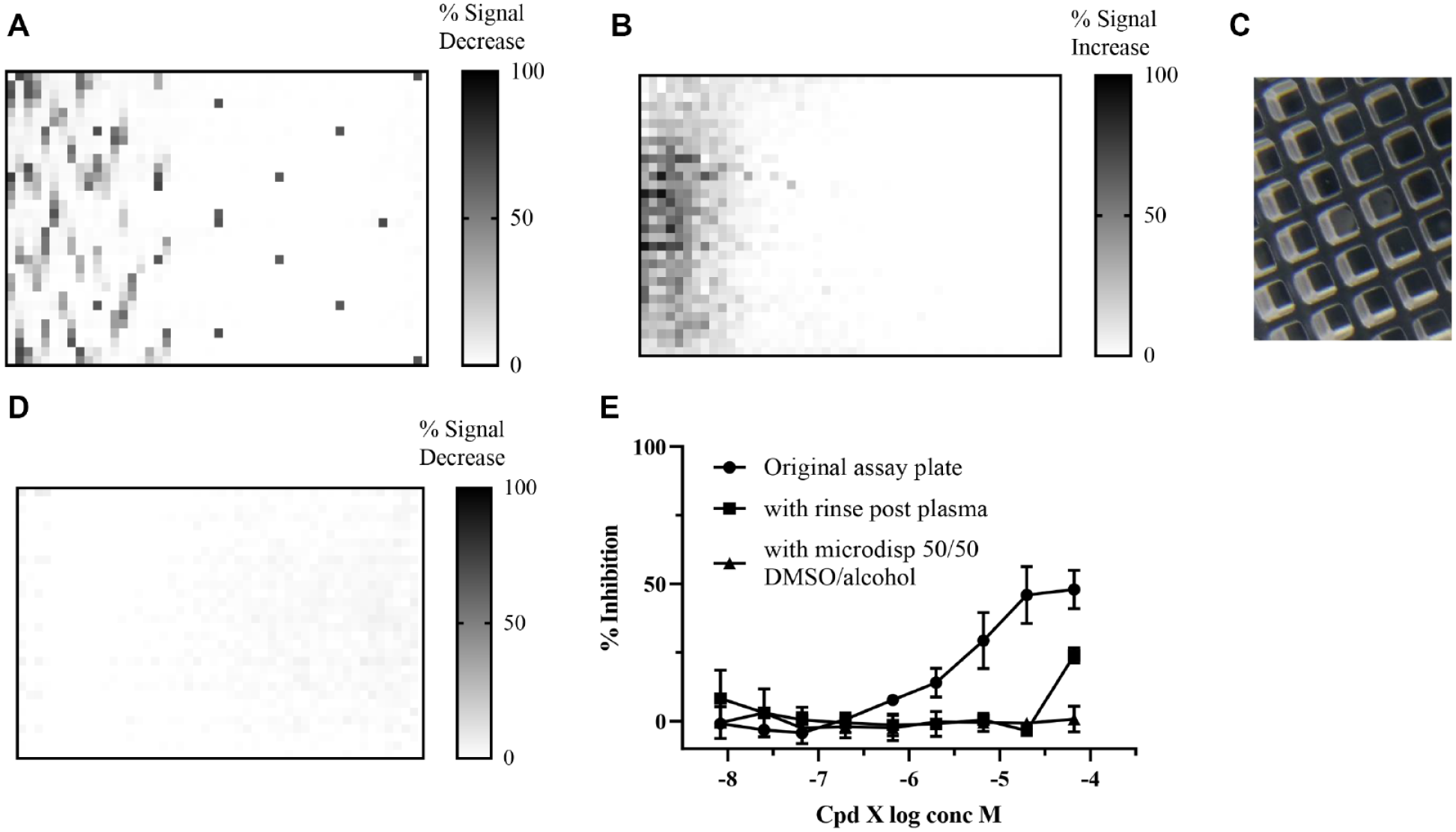
Effect of increasing plasma concentration and addition of DMSO/ethanol microdispense prior to the main wash step to solubilize any precipitated compounds. (
Initial data using the prototype PlasmaKnife microplate cleaning system (MCS) to clean used assay plates. (
Effect of Including Water Prewash and a More Focused Cold Plasma Source
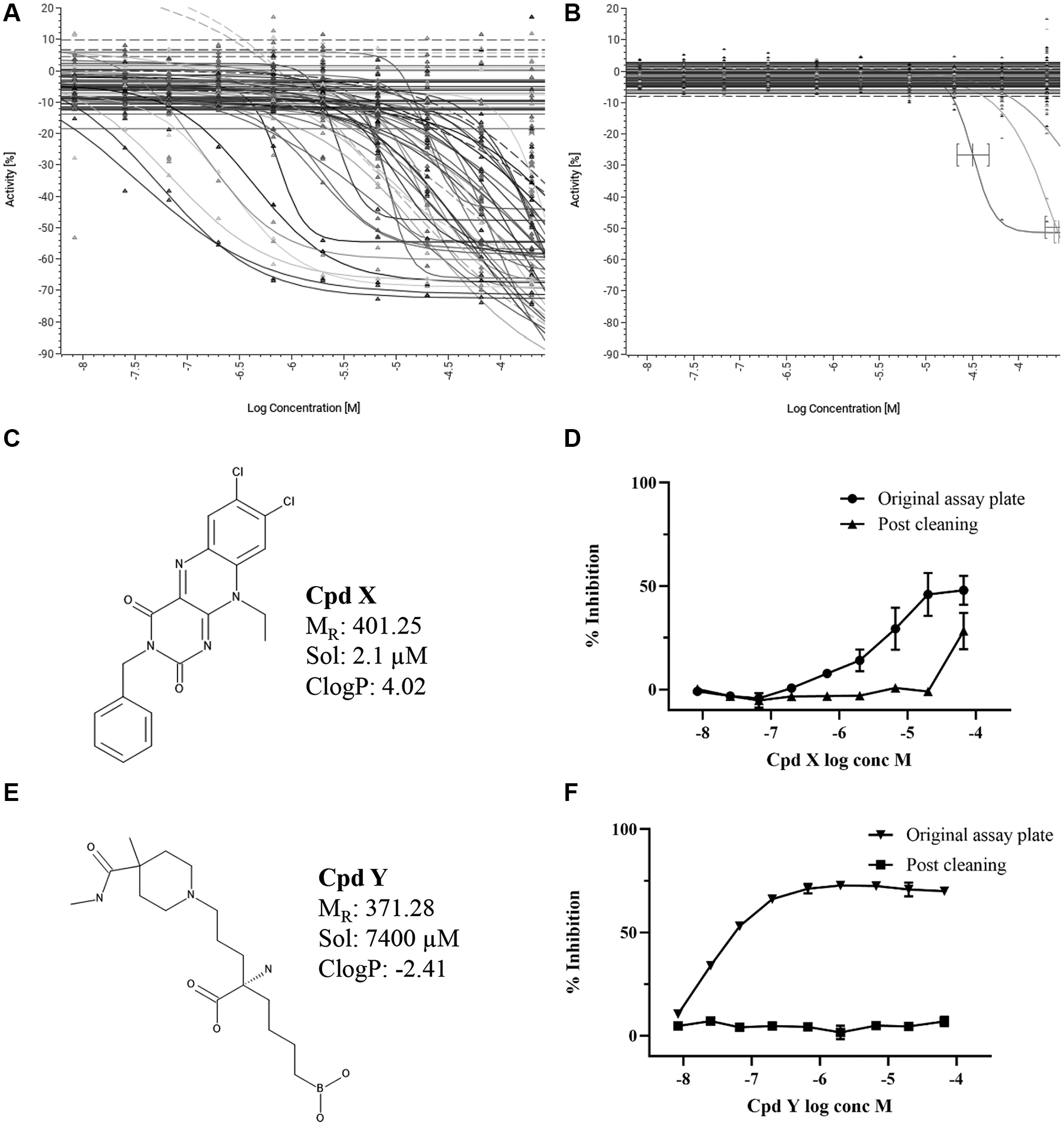
Following these initial results, we worked with IFS to improve the prototype instrument to deliver a more robust and reliable plate-cleaning process. Hardware improvements included the use of more hydrophobic materials to reduce the wetness of the components, such as the rinse station plate holder after processing plates through the device. Parts were also upgraded to focus the plasma more directly onto the microtiter plate, by increasing the pumping pressure of the plasma gas from 90 to 150 PSI, through a narrow slot (the bottom gap of the holder is 0.060 inches) to accelerate the flow rate and minimize the dilution of the energy intensity (the electron density is in the range of 1014 to 1015 cm−3 using factory settings) by the surrounding room air. The gap size provides the appropriate flow speed based on the pressure of the air feed into the plasma generator for optimal plasma generation. Plasma exposure is not determined by time under the gap, and the microtiter plate movement involves multiple passes under the plasma generator. Both the speed of the plate movement and the number of repeat passes have been optimized to yield the maximum cleaning effectiveness and is independent of plate format for standard-height microplates.
The rinse station centrifuge spin speed was reduced to 1000 rpm (relative centrifugal force = 113g) to retain more liquid in the wells, because a damp environment ensures the most effective plasma treatment. We also tested the effect of a 30 s prewash with MilliQ (MerckMillipore, Darmstadt, Germany) water (after the pulse spin step emptying the assay well contents), prior to rinsing plates with 14% ethanol to test whether this would improve plate cleaning. The test set of 78 active compounds was used to generate a set of used microtiter plates in the biochemical absorbance assay, and these were then cleaned using the modified PlasmaKnife MCS prototype. Figure 4A shows that despite these various changes, the cleaning process was still not completely effective, because wells that had contained the highest concentrations of compound X were still inhibiting the assay signal on the cleaned plates when retested in the assay as empty plates. Figure 4B shows that these changes to the cleaning protocol still resulted in successful cleaning of compound Y, as no inhibition could be detected after PlasmaKnife MCS cleaning.
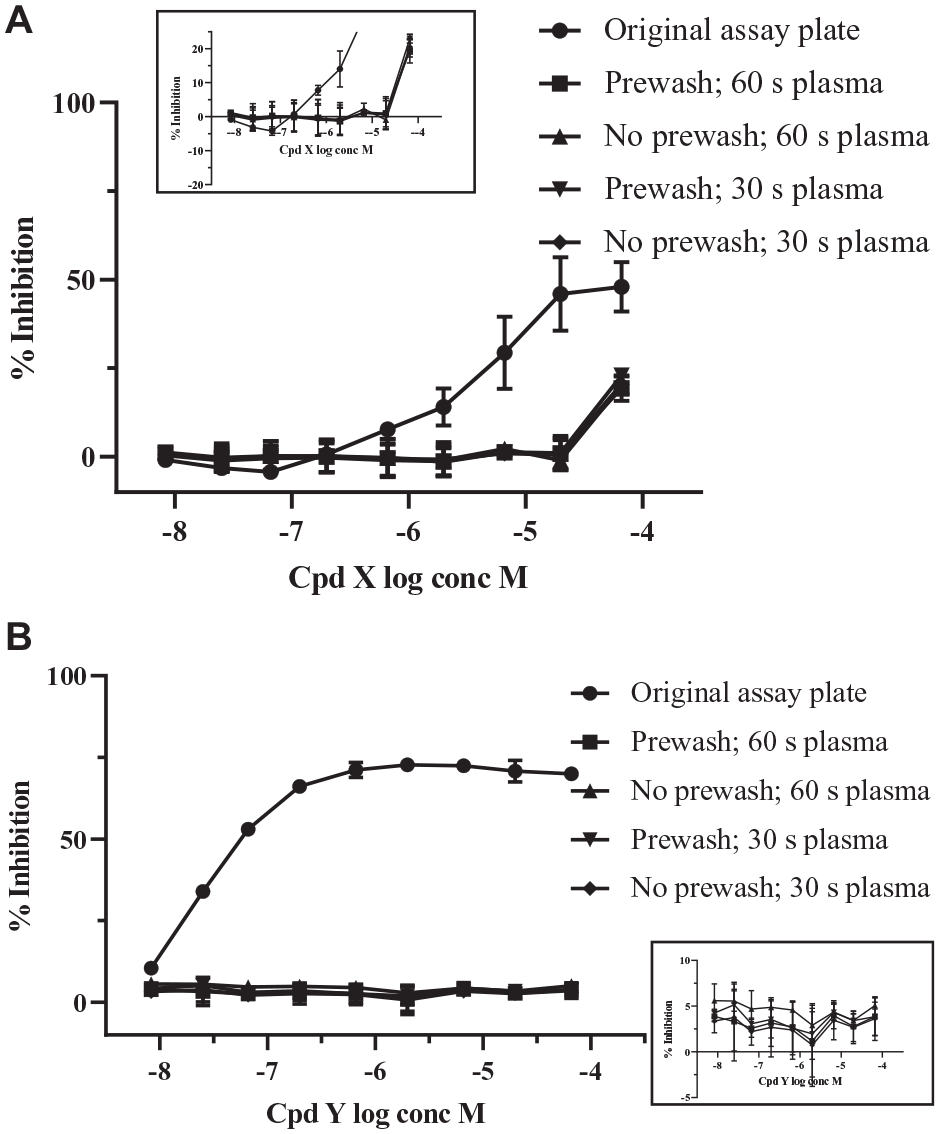
Effect of water prewash and length of plasma treatment on the plate-cleaning process. Optimizing the workflow using the prototype PlasmaKnife microplate cleaning system (MCS) to clean the used assay plate by including a water prewash step and different lengths of cold plasma treatment time. All PlasmaKnife MCS cleaned plates were retested in assay as empty plates. (
Plasma Controller Modifications
During this prototype development, IFS also implemented several hardware enhancements. The original plasma generator produced a single band of plasma under which the microplate moved. As described in the previous section, this was replaced with a full microplate-sized focused plasma generator to increase plasma density and energy and thereby reduce heat. Used assay plates were cleaned as before using the new plasma source. Representative heat maps of assay data from the original used assay plate (
We found that the patterning seen on these cleaned plates was due to uneven air flow during plasma treatment with the full microplate-sized plasma generator; there was no air dilution of plasma in the plate center, but this occurred at the plate edges. The production PlasmaKnife MSC now has a single row-type plasma generator, with plasma applied in two passes over the microtiter plate, which is more effective than one pass and eliminates uneven plasma treatment.
Removing Residual Compound Contamination Using Solvent Microdispense
The biochemical absorbance assay used clear microtiter plates, and visual inspection after the wash and plasma treatment process showed white residues in wells 12C and D (
To implement this procedure, an additional microdispenser was added in the rinse station, which could be programmed to optionally dispense DMSO, solvent, or a mix into the wells to help remove any dried or precipitated compounds. It is standard practice in most HTS labs to use acoustic dispensing of compound stocks in DMSO into empty microplates to create assay-ready microtiter plates. 14 From a review of the literature, dried or precipitated compounds are most commonly associated with acoustic dispensing onto dry plates, 15 and the PlasmaKnife MCS needed to address this issue. We routinely use a 2 s microdispense, which adds 1.5 to 2 µL per well in a 1536-well microtiter plate (hence, four times this volume for 384-well plates), but the dispense time can be adjusted in 1 s increments if another volume is found to work better. The solvent mix is left in the wells for 5 s before it is washed out with the standard 14% ethanol wash. The low volumes of DMSO/alcohol mix will not materially alter the waste stream generated.
Decreasing the Duration of Main Wash Step
After successful complete cleaning of the used microtiter plates, we investigated the effect of decreasing the length of the main wash step with 14% ethanol. This would reduce the length of time for plate processing through the rinse station, speed up the overall process, and positively affect the waste stream by reducing the amount of solvent waste generated. We tested reducing the wash step from 30 s to 20, 10, or 5 s using assay plates generated in the biochemical absorbance assay using the same set of 78 test compounds that had been used in previous experiments. Data in Figure 5 show that using compound X as an example of a compound that had been difficult to clean, we were able to reduce the main wash step to 5 s and still maintain the ability to clean plates successfully. The high-pressure wash uses about 60 mL per second, so reducing the wash from 30 to 5 s results in an ~80% waste reduction, which has a positive impact on the waste stream disposal. The system can be set for shorter wash times (minimum 1 s), which could further reduce the waste stream.
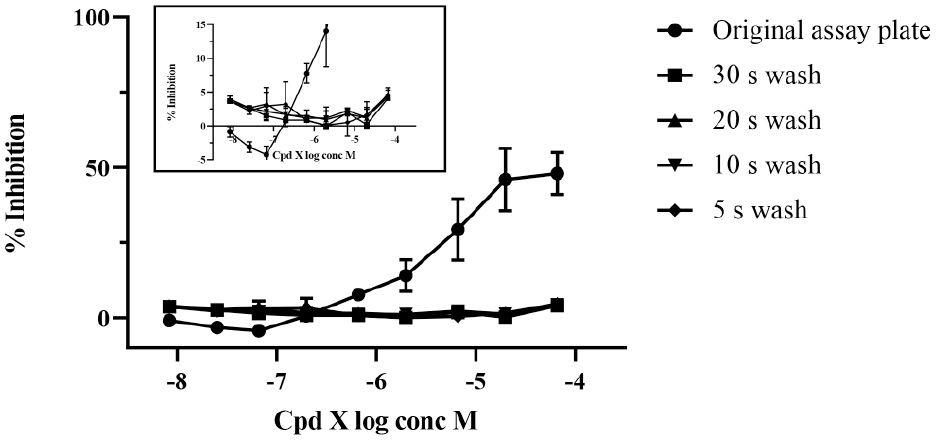
Effect of decreasing the length of wash time with 14% ethanol. Compound X was successfully cleaned by reducing the main wash time to 5 s. The inset shows an expanded data set at the lower percentage inhibition levels to more easily visualize the overlaid data shown in the main graph. Mean and standard deviation of n = 2 data are shown.
Standard Plate-Cleaning Process
All steps in the cleaning process have variables, such as length of dispense time, centrifuge speed, dwell time after all dispense steps, and time of cold plasma treatment, which allow the user to optimize the wash and plasma treatments and create bespoke cleaning protocols for different assay and plate types. We selected a colorimetric detection endpoint as the test assay to develop the PlasmaKnife MCS, as we believe this to be the most sensitive to interference from compounds that have not been successfully cleaned. Typically, compounds contained within pharmaceutical company compound collections interfere more with absorbance assays than fluorescence endpoints. 16 From this work, we have now developed a standard cleaning protocol using the production PlasmaKnife MCS, which has been successfully used to clean a range of standard polystyrene microtiter 1536-well plates (clear, white, and black) used in absorbance, fluorescence, and luminescence biochemical assays within the HTS center, demonstrating that the MCS can be more broadly applied to typical conditions or assays. The standard cleaning process employs a 5 s pulse spin to empty assay contents, a 2 s addition of DMSO/alcohol, followed by 5 s dwell time to aid solubilization of any dried/precipitated compounds. Plates are spun for 5 s and then washed using 14% ethanol for 5 s, before being centrifuged at 1500 rpm (relative centrifugal force = 254g) for 5 s. Plates are then treated with plasma for 60 s, followed by a 5 s rinse with 14% ethanol and left to air dry. It is possible for liquid droplets to be on the vertical edge and/or skirt of the microplate, and so these should be allowed to dry before stacking the cleaned microplates.
Future Considerations
To significantly reduce the amount of microtiter plates going to plastic biohazard waste, there are areas that require further evaluation to widen the application of the PlasmaKnife MCS. During development of the prototype instrument, we investigated only the effect of one cleaning cycle; additional experiments are planned to investigate how many times plates can be cleaned and reused. It would also be interesting to develop and test cleaning protocols using assay plates containing more potent compounds (pIC50
Our evaluation was performed as a manual process, with scientists moving plates between the different devices in the standard workflow. Using the standard plate-cleaning process described earlier, the throughput is ~55 plates per hour. To realize the potential of the PlasmaKnife MCS to clean hundreds of plates, there is a clear requirement to automate the process, and IFS is developing software to facilitate this. Allowing for robotic transit time and data processing of sensor data, we estimated that the overall system could produce a renewed microplate every ~45 s for most assay plates. Handling different plate types is facilitated using custom three-dimensional-printed insets for the plate holders, allowing handling of different plate heights (96-, 384-, or 1536-well plates) as long as they have a standard American National Standards Institute footprint. The PlasmaKnife MCS has been designed so that most standard robot grippers can easily access the plate holder on both the rinse and plasma stations, and most materials used within the rinse station are hydrophobic in nature, although reliability testing would be required for robot handling of potentially wet/damp plates coming out of the rinse station. The automation of plate cleaning could also incorporate technology to monitor the process; for example, microplates could be scanned and recognized by high-resolution video recognition to ensure the correct software and hardware is present on the system to process that particular plate type. Cleaned plates could also be inspected by high-resolution video after plasma treatment for any anomaly and these plates sent to a quarantine location to prevent them from being included for use in future assays.
Cleaning and reusing plates at scale brings other practical considerations into play, such as the length of time between generating assay plates and cleaning them. Our finding generally is that the shorter the interval between use and cleaning, the easier the plates are to clean. Storage duration and conditions for cleaned plates also require consideration before plates are reused in assays. For example, should they be stored vacuum sealed to protect from moisture or dust ingress? Another aspect is the tracking of reused plates. Generally, plate barcoding is used to track information about the identity of compounds in each plate, with a requirement for single-use barcodes. Cleaning plates brings with it a need to rebarcode them, and information technology solutions may be needed to track the number of times plates are cleaned and reused. Another practical aspect to consider when cleaning hundreds of plates is disposal of the solvent waste generated during the process, to reduce the environmental impact. This could potentially be used as a biofuel to retrieve energy and offset the carbon footprint generated by the waste, but this needs to be carefully investigated.
The issue of plastic pollution is of increasing concern within today’s society, and sustainability is a clear focus for the pharmaceutical industry. Reducing single-use plastics within the laboratory is a key aspect for improving sustainability, with particular focus on the large number of plastic microtiter plates generated annually within screening centers such as ours. Initial data indicate that the development of the PlasmaKnife MCS delivers a potential solution to address this issue and highlight some areas for further consideration and development to deliver a solution at scale. It is important to consider all aspects of the plate-cleaning process workflow to ensure that all waste generated (plastic and solvent) is minimized in a manner that does not compromise data quality in future screening assays.
Footnotes
Acknowledgements
Many thanks to Clare Stacey, Johanna Kastl, Roz Brant, Darren Plant, and Elizabeth Mouchet within the HTS center for generating used microtiter assay plates and testing the PlasmaKnife MCS cleaned plates in assays to help evaluate the prototype instrument.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are employed by either AstraZeneca, IonFields Systems, or Talavera Science as appropriate, and their research and authorship of this article was completed within the scope of their employment with either AstraZeneca, IonFields Systems, or Talavera Science as appropriate.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
