Abstract
The use of microplates (for bioassays, immunoassays, and general research) that are manufactured from plastic materials has proved problematic due to issues with accuracy, repeatability, and specificity of the results generated. The cause of these issues has been identified as leachables present in the plastic materials. This article presents an extractables study performed with available microplates manufactured with plastic. Common microplates from five different vendors were obtained, including plates from SiO2 Medical Products (SIO) containing a plasma treatment designed to produce an ultra-low protein-binding surface. The microplates were solvent extracted, and the resulting extracts were analyzed for organic extractables. The extractables profiles were examined and compared among the five different plate types. Detected extractables were identified in each of the extracts, and the potential effect on protein binding is discussed.
Introduction
Plastic materials are omnipresent in laboratory consumable products such as pipette tips, centrifuge tubes, flasks, microplates, and petri dishes. Widespread use of plastic lab consumables compared to borosilicate glass has been perpetuated by their improved safety, lower cost, and convenience. A growing body of literature has exposed extractables and leachables as a key limitation of plastics for applications, including clinical immunoassays,1 –4 drug discovery, 5 food and beverage, 6 and basic research.7,8 Specifically, for drug discovery bioassays or immunoassays, the presence of leachable contaminates can lead to erroneous or inconclusive results that hinder lab productivity and waste limited drug product. This is further complicated by the desire to analyze drug proteins in lower concentrations by using more sensitive analytical instrumentation. Accordingly, there is a clear, unmet need for clean, low-protein-bind laboratory consumables such as microplates.
The objective of this study is to screen commercially available microplates for extractables that could affect protein binding during use in bioassays and the storage of biologics. The study was designed to compare the full extractables profiles of the plates, and was not specific to a particular bioassay or group of assays. Among the different microplates compared in the current study are those produced by SiO2 Medical Products (SIO). SIO developed a proprietary microplate technology that combines a clean engineered polymer with a plasma treatment, resulting in an ultra-low protein-binding surface. Higher recoveries were reported for five model proteins in 1.5 nM solutions and 72 h incubation on SIO plasma-treated microplates as compared to a commercial low-protein-bind microplate on the market today. 9 Furthermore, gas chromatography–mass spectroscopy (GC/MS) and liquid chromatography–mass spectroscopy (LC/MS) analysis was conducted after 72 h extraction with acetonitrile and after 72 h extraction with isopropanol, respectively. The total ion chromatograms indicated that the plasma treatment introduced no new extractables and that the chromatograms of SIO microplate extracts were identical to the isopropanol blank. Chromatograms of extracts from other commercial low-protein-bind microplates exhibited peaks corresponding to common additives such as Irgafos 168 and other impurities.
The current extractable study expands on the scope of the prior study 9 by including four more commercial microplates and applying more sensitive LC and GC methods with mass spectral detection. This study used a 24 h extraction with isopropanol, followed by injection of a 1 µL aliquot into GC/MS and LC/MS analytical instruments for extractable compound detection.
Materials and Methods
Solvents and Additives
Isopropyl alcohol (IPA), methanol, and ammonium acetate were of LC/MS grade and purchased from Fisher Scientific (Thermo Fischer Scientific, Waltham, MA). Irganox 1076 and Irgafos 168 were obtained from MilliporeSigma (St. Louis, MO). Purified water was generated in-house from a Millipore Advantage A10 water purification system (MilliporeSigma).
Microplates
Low-protein-binding microplates supplied by SIO, hereinafter referred to as SIO microplates, were molded from polypropylene and plasma treated with a proprietary process to minimize protein binding. 9 Commercial microplates with and without low-protein-binding characteristics were purchased from various commercial sources, including: (1) Porvair Sciences 96-well, 0.5 mL plates (cat. no. AWLS-X1001; Arctic White, Bethlehem, PA); (2) Greiner Bio-One 96-well, 0.5 mL plates (cat. no. Z667234; MilliporeSigma); (3) Eppendorf LoBind 96-well, 1.0 mL plates (cat. no. 951032107; Eppendorf, Hamburg, Germany); and (4) Nunc 96-well, 0.5 mL plates (cat. no. 267245; Thermo Fischer Scientific). The SIO plasma-treated and Eppendorf LoBind plates exhibit lower protein binding than standard polypropylene plates. 9 The Porvair Sciences, Greiner, and Nunc microplates have protein-binding character typical of polypropylene. 9 The commercial microplates underwent no additional treatment or coating.
Controlled Extraction and Analysis
A controlled extraction study was conducted by filling the wells of the microplates with the extraction medium. One microplate each of the SIO microplates and four types of commercial microplates were sampled. Twenty wells of the sample microplate were each filled with IPA, covered with a clean microscope slide, and incubated at room temperature for 24 h. The resulting extracts from each well were pooled into precleaned vials and stored at 5 ± 3 °C until analysis.
Gas Chromatography–Mass Spectrometry
The IPA extracts were directly injected into an Agilent 7890B Gas Chromatograph (Agilent, Santa Clara, CA) coupled with a 5977B MS detector (Agilent) for analysis of semivolatile compounds. A 1 µL aliquot was injected at a 10:1 split ratio onto an Agilent HP-5ms ultra-inert column with dimensions of 30 m × 250 µm × 0.25 µm. The inlet and detector temperatures were 280 and 300 °C, respectively. The oven temperature program started at 40 °C for 1 min, ramped to 280 °C at a rate of 25 °C per minute and further to 315 °C at a rate of 5 °C per minute, and held at 315 °C for 8 min.
Data analysis was performed using Agilent Masshunter Qualitative Analysis software. Compounds were first extracted via the chromatogram deconvolution function or conventional peak integration, and then identified by searching against the 2014 NIST (National Institute of Standards and Technology) Mass Spectral Library. No specific reporting limit was set for this qualitative study. Before injecting samples, a standard mix containing Irganox 1076 and Irgafos 168, prepared in IPA at 10 µg/mL each, was analyzed to evaluate system suitability and general compound response factors.
Liquid Chromatography–Mass Spectrometry
The IPA extracts were directly injected into an Agilent 1290 UHPLC coupled with a 6550 iFunnel Q-TOF MS detector for analysis of nonvolatile compounds. A 1 µL aliquot was injected onto an Agilent Zorbax RRHD Eclipse Plus C8 column with dimensions of 2.1 mm × 50 mm × 1.8 µm. The column was held at 40 °C. The mobile phases A and B were 0.01% ammonium acetate in purified water and 0.01% ammonium acetate in methanol, respectively. The gradient started at 30% B, ramped to 80% B within 7 min and further to 100% within 19 min, and held at 100% B for 4 min. The flow rate was 0.6 mL/min. The MS scanned from 50 to 1700 m/z in both positive and negative ion modes. The ion source was an electrospray source. A reference solution was infused by an isopump at 0.6 mL/min for mass correction.
Data analysis was performed using Agilent Masshunter Qualitative Analysis software. Compounds were first extracted via the molecular feature extraction function and then identified by searching against the Boston Analytical Extractables & Leachables (E&L) database (built from the Agilent E&L PCDL B07.00 database). Formulae based on accurate masses were generated for unmatched compounds. No specific reporting limit was set for this qualitative study. Before injecting samples, a standard mix containing Irganox 1076 and Irgafos 168, prepared in IPA at 0.1 µg/mL each, was analyzed to evaluate system suitability and provide response factors for these two compounds.
Results and Discussion
Controlled Extraction
Under normal use conditions, these polypropylene microplates are designed to hold aqueous biological fluids for a short period of time. This study was designed as an extractables study, in which exaggerated conditions were used to generate an extractables profile for each material. This methodology followed the guidelines in the United States Pharmacopeia’s Assessment of Extractables Associated with Pharmaceutical Packaging/Delivery Systems. 10 The resulting extractables profiles represent potential leachables that could negatively affect protein binding during use of the plates. In this extractables study, IPA was selected as the extraction medium to exaggerate the polar organic environment created by biomolecules such as proteins in aqueous solutions. The goal was to extract the plates under exaggerated but not exhaustive conditions. The contact time was extended to 24 h as a worst-case scenario. The extraction medium contacted only the well surfaces, similar to normal use. The temperature also simulated normal use temperature (i.e., room temperature). The overall extraction conditions represented a slightly exaggerated scenario without going into the overaggressive realm.
Extractables Profiles of Commercial Microplates
Comparisons of the extractables profiles of the SIO plasma-treated and Eppendorf LoBind plates are included in Figures 1 – 3 . These plates are designed to exhibit low protein binding. Four extractables compounds were detected in the IPA extract of the Eppendorf LoBind plate by GC/MS, and 13 compounds by LC/MS (both positive and negative). A series of esters formed between glycerol and long-chain fatty acids was observed in the LC/MS chromatogram, with the most prominent being glycerol monopalmitate and glycerol monostearate. Both were also observed in the GC/MS analysis. These compounds are commonly used as lubricants in polymers 11 and speculatively provide some degree of reduced protein binding in this case. In the GC/MS analysis, isopropyl palmitate and isopropyl stearate were detected, both of which may be attributed to the side reaction products of palmitic acid and stearic acid with the extraction medium IPA. Besides these esters, a nucleating agent bis(3,4-dimethyl-dibenzylidene) sorbitol (DMDBS) and the antioxidant Irgofos 168 were also detected in the IPA extract of the Eppendorf LoBind microplate.
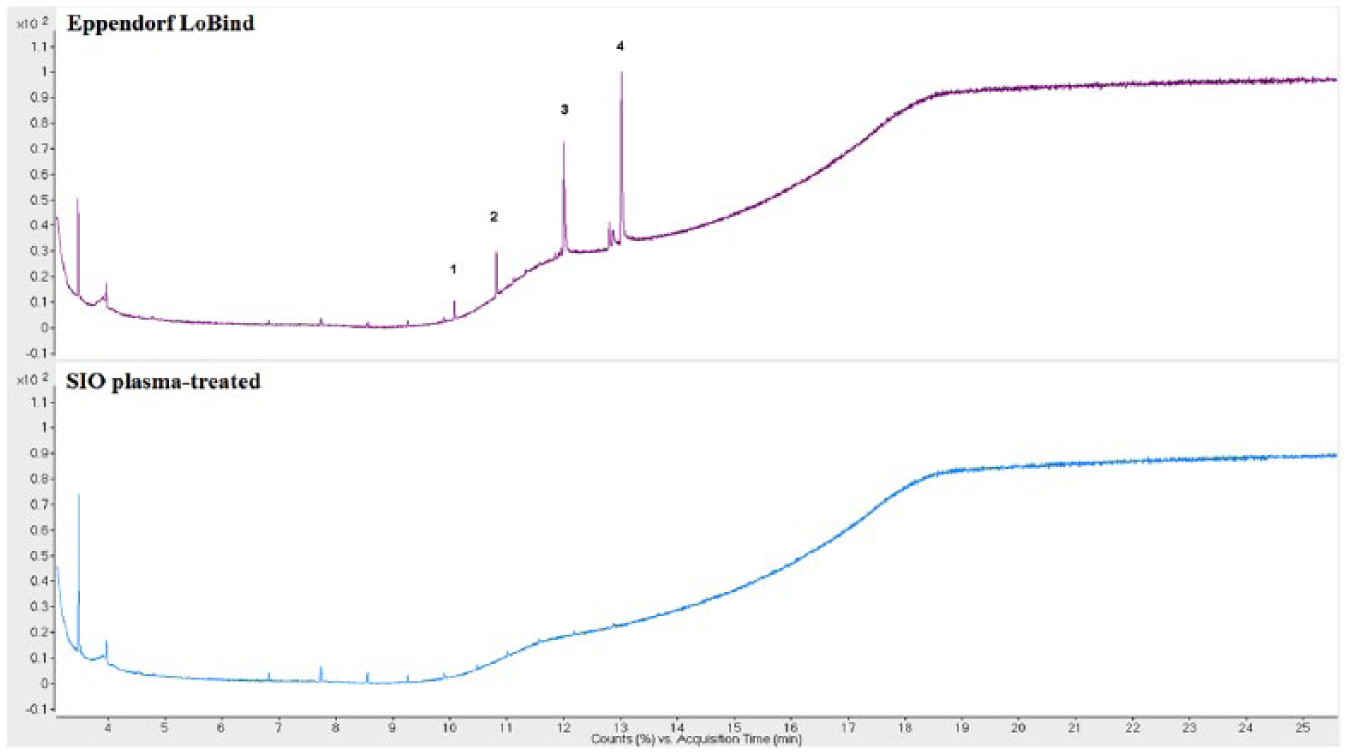
Gas chromatography–mass spectroscopy (GC/MS) chromatograms of Eppendorf LoBind (top) and SiO2 Medical Products (SIO) plasma-treated microplates (bottom). (
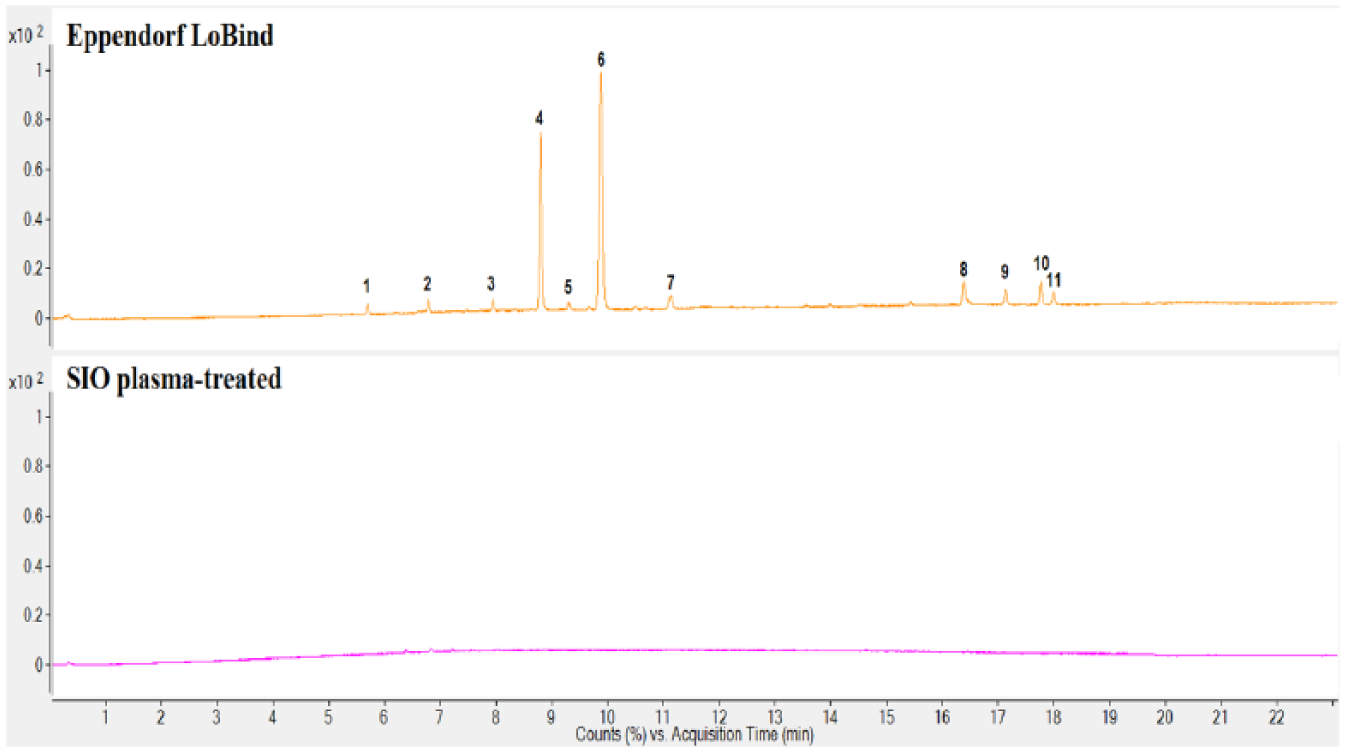
Liquid chromatography–mass spectroscopy (LC/MS) positive ion total ion chromatograms of the isopropyl alcohol (IPA) extract of the Eppendorf LoBind microplate (top) and IPA extract of the SiO2 Medical Products (SIO) plasma-treated microplate (bottom). (
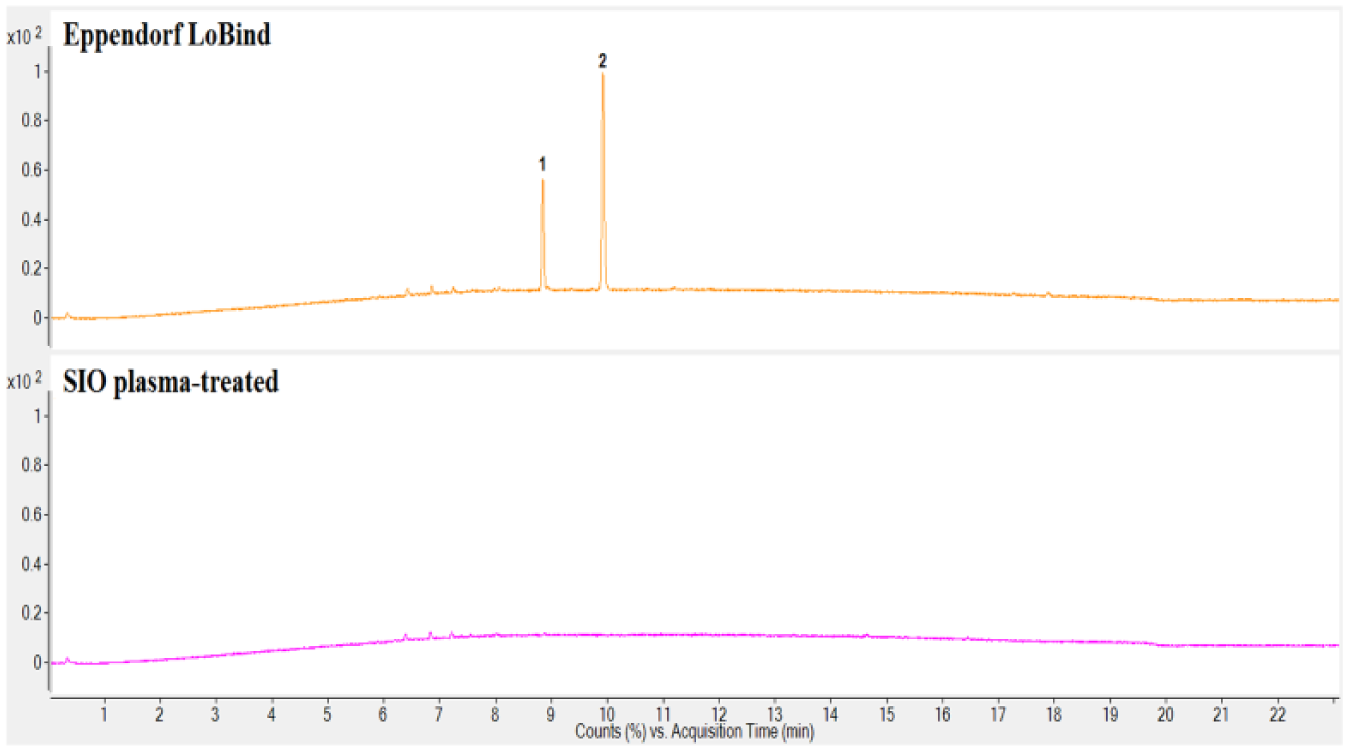
Liquid chromatography–mass spectroscopy (LC/MS) negative ion total ion chromatograms of the isopropyl alcohol (IPA) extract of the Eppendorf LoBind microplate package (top) and IPA extract of the SiO2 Medical Products (SIO) plasma-treated microplate (bottom). (
A comparison of the extractables profiles of all the plates included in this study is shown in Figures 4 – 6 . The SIO plasma-treated and Porvair plates had the lowest amount of extractables detected. Extractables were detected in the extraction profiles of the other three microplates. For the Greiner microplates, Irgafos 168, Irgafos 168 phosphate (an oxidized form of Irgafos 168), the nucleating agent DMDBS, and a number of branched alkanes were detected. For Nunc microplates, Irgafos 168, Irganox PS800, oxidized Irganox PS800, Irganox 3114, and a number of branched alkanes were prominent in the chromatograms. These antioxidants are added as part of the additives package included with the polymer resin, and the branched alkanes are likely residual oligomers from the polymerization of the polymer material.
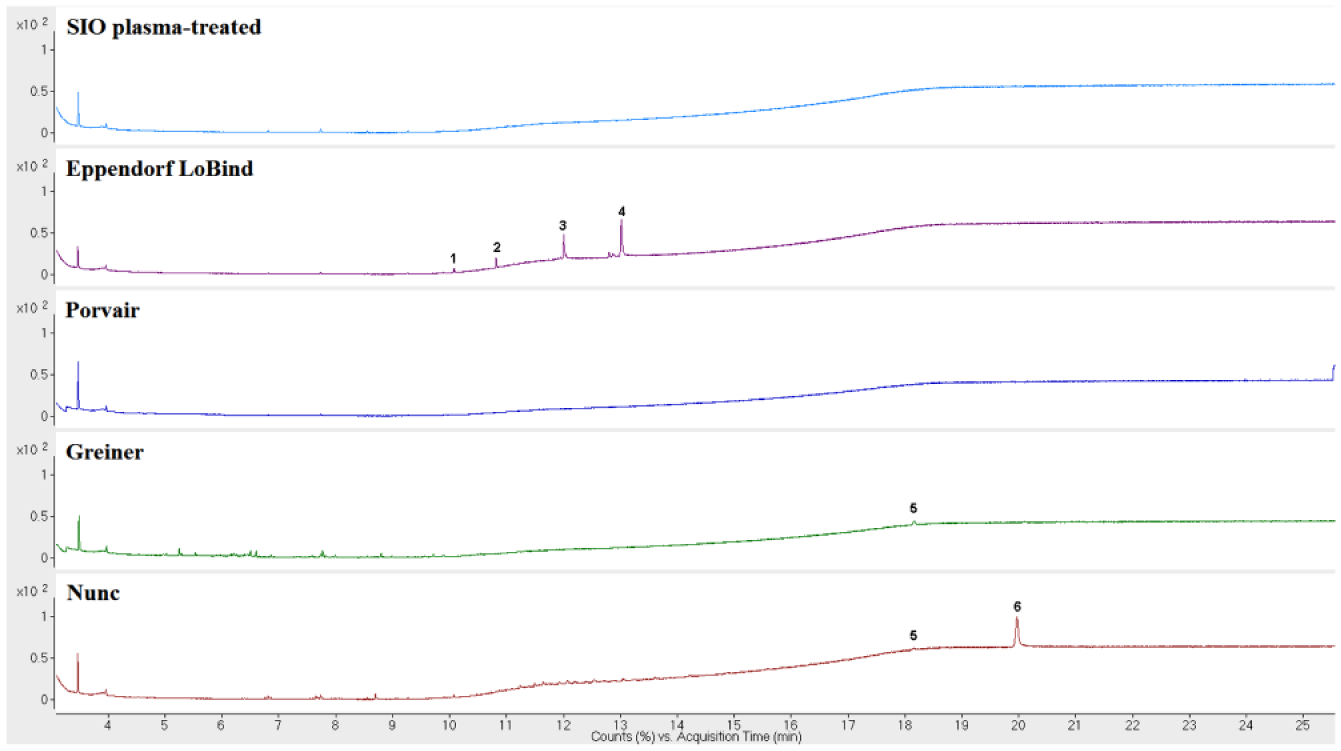
Gas chromatography–mass spectroscopy (GC/MS) chromatograms of SiO2 Medical Products (SIO) plasma-treated, Eppendorf LoBind, Porvair, Greiner, and Nunc microplates. (
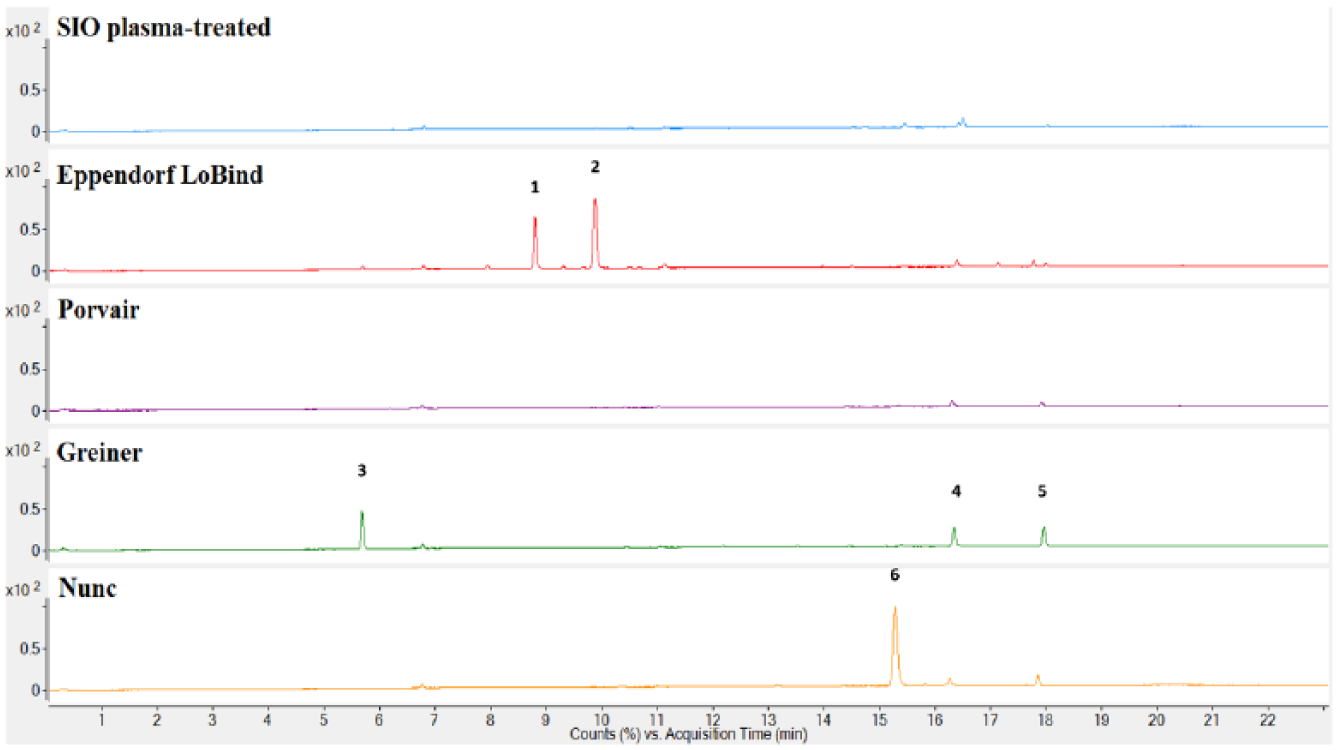
Liquid chromatography–mass spectroscopy (LC/MS) positive ion total ion chromatograms of the isopropyl alcohol (IPA) extracts of the SiO2 Medical Products (SIO) plasma-treated, Eppendorf LoBind, Porvair, Greiner, and Nunc microplates. (
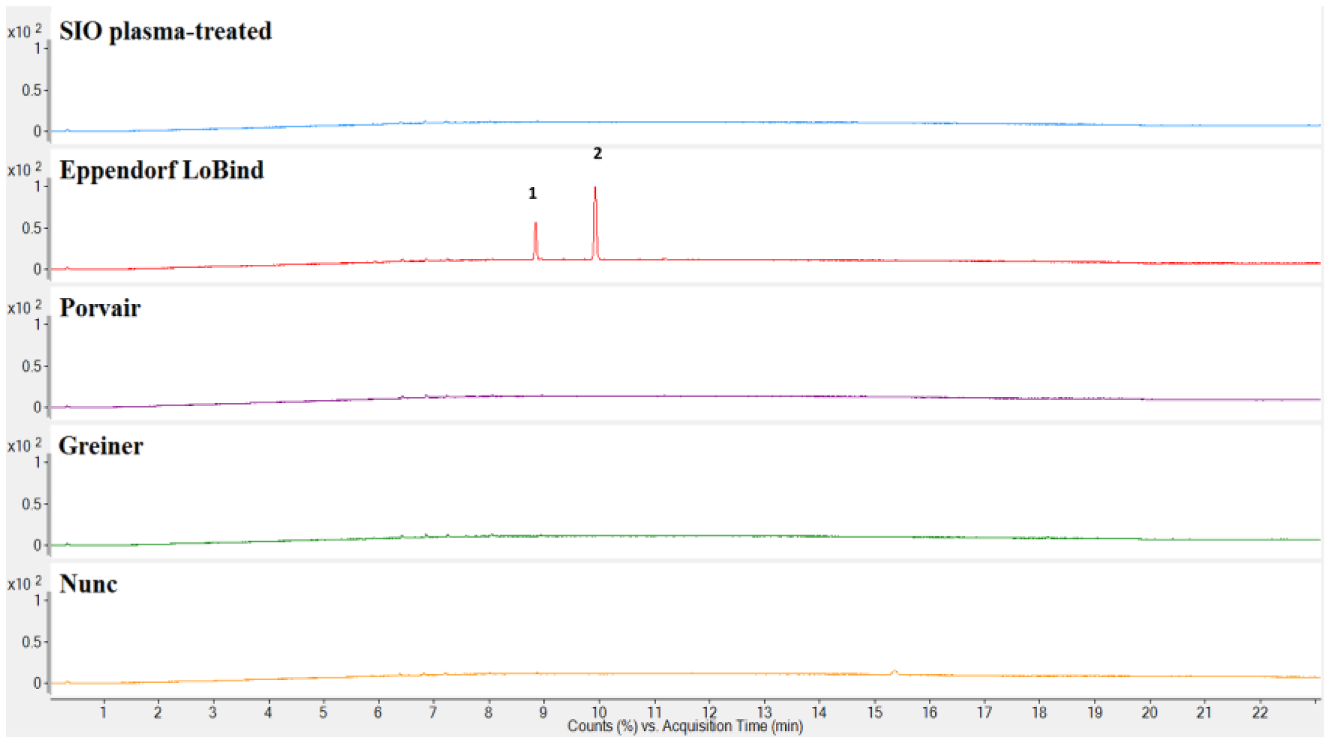
Liquid chromatography–mass spectroscopy (LC/MS) negative ion total ion chromatograms of the isopropyl alcohol (IPA) extracts of the SiO2 Medical Products (SIO) plasma-treated, Eppendorf LoBind, Porvair, Greiner, and Nunc microplates. (
All compound assignments were made using the 2014 NIST Mass Spectral Library for the GC/MS analysis, and the Boston Analytical E&L database for the LC/MS analysis. Compound identifications were not confirmed with standards and therefore are considered tentative. Standards of common polymer additives were analyzed with the plate extracts to verify the method suitability.
Conclusion
Extractable profiles were compared for microplates marked as “low-protein-binding” as well as for standard microplates. Low-protein-binding SIO microplates exhibited low levels of extractables for both types of microplates. The SIO plasma-treated microplates gave extractable profiles that were lower in total extractables than the other low-protein-binding microplates included in this study. This is the result of the polymer resin selection, molding process, and plasma treatment process used in the production of these plates. In a comparison of extraction profiles for all microplates in this study, the SIO plasma-treated and Porvair microplates contained the lowest amount of extractables. Microplates with the lowest level of extractables are the most favorable to conduct bioassays and store biologic drugs. The SIO plasma-treated microplate has the added benefit of a high protein recovery due to its low binding surface characteristics.
Supplemental Material
ELP_-_2017_-_fillable_PDF – Supplemental material for Characterization of Extractable Species from Polypropylene Microplates
Supplemental material, ELP_-_2017_-_fillable_PDF for Characterization of Extractable Species from Polypropylene Microplates by Eric J. Hill, Steve J. Martin and Christopher M. Weikart in SLAS Technology
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Eric J. Hill is employed by Boston Analytical and Steve J. Martin and Christopher M. Weikart are employed by SiO2 Medical Products Inc. Their research and authorship of this article was completed within the scope of this employment.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Eric J. Hill is employed by Boston Analytical and Steve J. Martin and Christopher M. Weikart are employed by SiO2 Medical Products Inc. Their research and authorship of this article was completed within the scope of this employment.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
