Abstract
Vitamin D belongs to the fat-soluble vitamins and is an integral part of bone metabolism. In the human body, a decreased vitamin D level can be an additional risk factor for diseases like cancer, diabetes, and mental diseases. As a result, an enormous increase in the demand for vitamin D testing has been observed in recent years, increasing the demand for powerful methods for vitamin D determination at the same time.
Automation is the key factor in increasing sample throughput. This study compares three fully automated sample preparation methods for the determination of 25(OH)D2 and 25(OH)D3 in plasma and serum samples. Starting from a semiautomated reference method, the method is tested manually and subsequently fully automated on the Biomek i7 Workstation by integrating a centrifuge and a positive pressure extractor into the workstation. Alternatively, the centrifugation for the separation of protein aggregates and supernatant is replaced by a filter plate. Finally, the sample throughput is further increased by using phospholipid removal cartridges. The results show that phospholipid removal significantly increases the recovery rates in liquid chromatography–mass spectrometry. With the phospholipid removal cartridges, recovery rates of 97.36% for 25(OH)D2 and 102.5% for 25(OH)D3 were achieved, whereas with the automated classic automated preparation method, the recovery rates were 83.31% for 25(OH)D2 and 86.54% for 25(OH)D3. In addition to the technical evaluation, the different methods were also examined with regard to their economic efficiency. Finally, the qualitative and quantitative performance of the developed methods is benchmarked with a selected semiautomatic reference method.
Keywords
Introduction
Vitamin D, which belongs to the group of secosteroids, plays a vital role in the human body, including the metabolism of calcium. 1 Vitamin D is a generic term for a set of fat-soluble vitamins, the most important compound of which is vitamin D3 (cholecalciferol). 2 25-(OH)-Cholecalciferol is formed in the human body in two ways. The first formation mechanism is the conversion of 7-dehydrocholesterol into cholecalciferol (calciol; vitamin D3) (via the intermediate-level previtamin D), which takes place in the skin by UV radiation.3–5 In the liver, the cholecalciferol formed is hydroxylated to 25-OH-cholecalciferol (calcidiol; 25-hydroxy-vitamin D3). Then it is synthesized in kidneys to form the metabolically active hormone 1-alpha, 25-(OH)2-cholecalciferol, which stimulates calcium absorption in the bones.6–9
Although 25(OH)D3 is only an intermediate stage in the synthesis of the physiologically active form, the 25(OH)D3 concentration is the most important parameter for diagnosing a vitamin deficiency, since 25(OH)D3 is the main storage form of vitamin D in the human body.10,11 If the 25(OH)D3 concentration is too low, the metabolically active form cannot be sufficiently synthesized, which would lead to a disruption of calcium resorption in the bones. The reference range is between 35 and 60 ng/mL. 12 In addition to the synthesis of cholecalciferol in the skin, it can also be absorbed through food. Vitamin D from plant nutrients and fungi is called ergocalciferol or 25-(OH)-vitamin D2 and is derived from ergosterol. 4 Ergocalciferol is similar to cholecalciferol except for a structure in the side chain. It is metabolically activated just like cholecalciferol and is also present in the blood since it can be ingested via plant food. 10
When food intake is insufficient or a metabolic disorder leads to a persistent vitamin deficiency, diseases such as rickets in children and osteomalacia or osteoporosis in adults may occur,13–15 whereas acute or chronic overdose can lead to hypervitaminosis with sometimes life-threatening effects. 16
In addition to the importance of the vitamin D level for the calcium balance, a chronic vitamin D deficiency can also lead to mental illness or mental impairments such as depression, difficulties in concentrating, and increased tiredness.17,18 The influence of vitamin D status in other serious diseases such as certain cancers or type 2 diabetes is currently being investigated.19–21 For example, a study showed a 50% reduction in colorectal cancer risk when 25-(OH)-vitamin D serum levels were greater than 33 ng/mL compared with the reduced status of 12 ng/mL. 22 For this reason, the amount of vitamin D tested has increased enormously in recent years.23–26
The main transport medium of vitamin D in the human body is blood. For this reason, both plasma and serum, as noncellular, fluid parts of the blood, are suitable as matrix for determining the vitamin D level. 27 Apart from cells, numerous other components of the blood in addition to vitamin D make plasma and serum a complex matrix and require time-consuming sample preparation. 28 For example, due to its lipophilic nature, the main storage form of vitamin D, calcidiol, is not transported freely in the blood but is attached to transport proteins (vitamin D binding protein [DBS] or albumin).29,30 Consequently, an important step in sample preparation is to resolve the protein binding.
The classic sample preparation usually consists of a protein precipitation followed by an extraction of the compound of interest. Protein precipitation is used to detach vitamin D from its transport protein and to remove proteins from the sample. Methanol, acetonitrile, methanol–acetonitrile mixtures, 2-propanol, and sodium hydroxide are used as solvents.31,32 Zinc sulfate can also be used to improve the release from protein binding. 33 The supernatant is then separated from the protein aggregate by means of centrifugation, whereby the integration of powerful centrifuges into an automation system is challenging.
A consecutive extraction step leads to enrichment of the analyte and further isolation, which reduces matrix effects in the subsequent detection system. For extraction, both solid-phase extraction (SPE) and liquid–liquid extraction (LLE) are available. With LLE, two immiscible phases are brought together to allow the transfer of the analyte from one phase to the other. 34 The SPE is based on the adsorption principle, in which an analyte in the stationary “solid” phase is retained on the surface of the sorbent bed as the liquid phase passes through. Hydrophilic or hydrophobic interactions and also anion–cation exchange mechanisms are used for retention. 35 A solvent, for example, methanol, acetonitrile, ethanol ethyl acetate, or methyl tert-butyl ether, is then applied to break the interactions and to return the isolated analyte to the solution.36–38 A sensitive equilibrium must be maintained in which the analyte is completely dissolved from the sorbent bed, but interfering substances are still retained. Positive or negative pressure is required to move solutions through the sorbent bed, whereby the application of positive pressure is also technically demanding. The flow rate must not be overly high and must be constant across all columns to prevent some columns from running dry. 39
In addition, the detection system can require a solvent exchange or further concentration. This can be done, for example, by evaporation using nitrogen followed by reconstitution.40,41
For vitamin D detection, immunological techniques such as radioimmunoassays (RIAs), chemiluminescent immunoassays (CLIAs), and enzyme-linked immunosorbent assays (ELISAs) or physical techniques such as high-performance liquid chromatography (HPLC), liquid chromatography–mass spectrometry (LC-MS), and liquid chromatography–tandem mass spectrometry (LC-MS/MS) are available. 42 LC-MS is the most commonly used technique due to its high sensitivity and specificity. 43 By using MS/MS, matrix effects, caused by sample components that are not removed, are further reduced.44,45 In plasma and serum samples, limits of detection (LODs) achieved with a protein precipitation, an SPE, and an LC-MS/MS detection for 25(OH)D2 and 25(OH)D3 range from 0.2 to 3 ng/mL and limits of quantification (LOQs) range from 0.8 to 6.2 ng/mL.33,38,46–49 Some authors reported even lower LOQ values of 0.0252 ng/mL.38,49
Nevertheless, detection methods require the cleanest possible sample; thus, the effort for sample preparation is high. Consequently, solutions have been developed by various manufacturers to simplify and improve the sample preparation process by combining sample preparation steps, removing additional sample components such as phospholipids, or eliminating the need to perform a centrifugation step.50,51
One option to skip the centrifugation step is to replace the separation of protein aggregate and supernatant by a mechanical filter in lieu of centrifugation. Instead of using gravity to separate heavier protein molecules, the size of the molecules is used to separate them from smaller analytes. 52 To accelerate the separation, either vacuum or positive pressure can be applied to the filter, and instead of removing the supernatant, the filtrate is collected, which also facilitates automation. This principle is used by the manufacturers Phenomenex and Biotage, among others. In the method presented by Lensmeyer et al., 53 the impact protein precipitation plate (Phenomenex, Torrance, CA) is used with an acetonitrile–water–zinc sulfate solution to precipitate proteins prior to SPE and subsequent evaporation and reconstitution for the determination of vitamin D from human serum by using an HPLC-MS/MS method. In this method, the sample preparation is performed manually. Margaryan et al. 54 used ISOLUTE PPT+ plates (Biotage, Uppsala, Sweden) for the manual determination of doxepin and nordoxepin from serum samples. The 96-well microtiter plate format of these two consumables allows an increased sample throughput.
Another approach to avoid extensive centrifugation and filtration steps was developed by Tecan Trading AG (Männedorf, Switzerland) with the Tecan AC Extraction Plate. The Tecan AC Extraction Plate is a deep-well microtiter plate whose inner walls are coated with a special sorbent material. The coating extracts small molecules such as vitamin D. Sample preparation is based on the “pipette and shake” principle (Tecan Trading AG). Since no filtration is carried out, solutions added for protein precipitation, washing, and elution are subsequently pipetted out and discarded or, in the case of the eluate, supplied for analysis. An example of a sample preparation method for vitamin D determination with an LC-MS/MS is described in Tecan 55 . This fully automated method achieves LOQs of 4.3 ng/mL for 25(OH)D3 and 14.1 ng/mL for 25(OH)D2. Recoveries are 84% for 25(OH)D3 and 80% of 25(OH)D2, with a coefficient of variation (CV) of less than 7%. In addition, no significant ion suppression effects or enhancements were observed.
The removal of phospholipids is a further area of interest. Phospholipids are part of food and are frequently found in blood. 56 Phospholipids are formed from fatty acids and a glycerol and phosphate moiety. Common phospholipid groups are glycerophosphatidylcholines and phosphatidylcholines like lecithin. 57 In electrospray ionization (ESI), phospholipids ionize in both positive and negative modes and interference caused by them cannot be avoided by the choice of ionization. However, by the selective use of sorbents to eliminate polar groups, phospholipids can be removed from the sample and matrix effects can be reduced.50,58 In the method presented by Tulipani et al., 59 the Ostro protein precipitation and phospholipid removal plate (Waters, Milford, MA) and the Phree phospholipid removal plate (Phenomenex), as well as the classic protein precipitation, are compared. Between extraction technologies of Waters and Phenomenex, no significant differences in the amount of phospholipid residues could be found. Still, selective phospholipid removal is more successful than deproteinization alone. This is also evident in the determination of metabolome and for the semiautomated determination of budesonide. 60 Pucci et al. 58 compared an automated method for phospholipid removal by using a phospholipid removal plate without subsequent SPE with a classic protein precipitation. The results of this study show both cleaner extracts and a significant shortening and simplification of the method. The automation was performed with the Hamilton StarPlus liquid handler robot (Hamilton Robotics, Reno, NV).
In the (partially) automated methods from Nordstrøm et al. 61 and Fernau et al., 62 a high throughput of up to 600 samples/day is achieved with LLE. The Hamilton Robotics Microlab Starlab Automated Pipetting Workstation is used for automation. The addition of the protein precipitation reagent; subsequent mixing; and the addition of 600 µL of hexane for extraction, mixing, and centrifugation are fully automated by Nordstrøm et al. In contrast, the method of Fernau et al. can be interrupted for mixing and centrifugation, allowing both the integration of a centrifuge into the deck of the workstation and the use of an external centrifuge. In contrast to SPE, the use of LLE eliminates the need for pressure application steps, which leads to a shortening of the method. However, the method of Nordstrøm et al. requires the provision of large amounts of solvent (600 µL of hexane per sample) as well as additional evaporation and reconstitution prior to the introduction onto an LC-MS system. The LOD achieved is 4 ng/mL. The CV of the measurement precision ranges from 3.5% to 9.1% for 25(OH)D3 and 3.1% to 13.6% for 25(OH)D2 in the concentration range of 4–104 ng/mL. The recovery ranges from 97% to 129% for 25(OH)D3 and from 100% to 116% for 25(OH)D2. 61 In the method established by Fernau et al., the technique of process automation using the Hamilton Microlab Starlab Workstation, which resembles the Biomek i7 Workstation (Beckman Coulter Life Sciences, Indianapolis, IN), is described. The Hamilton Microlab Starlab Workstation provides gateways for the integration of third-party devices, for example, devices for pressure application (SPE), centrifuges, or shakers (LLE). Features such as labware sensing, liquid-level detection, antidroplet control for highly volatile substances such as methanol or hexane, and barcode and sample tracking are shown. The subsequent comparison of the (partially) automated method with a manual method shows a correlation coefficient of 0.9911. 62
In comparison with high-throughput automated methods, a manually performed classic sample preparation procedure may cause increasing costs in addition to the high effort involved. Therefore, we present three fully automated, SPE-based methods for the preparation of plasma and serum samples for the determination of vitamin D. In addition to the automation procedure and the technical evaluation, the different methods were also examined with regard to their economic efficiency.
Materials and Methods
Automation Approach
Automation Methods
A procedure corresponding to the classic preparation of plasma and serum samples for the determination of vitamin D was used in the semiautomatic method of Knox et al., 63 which serves as a reference method here. They removed proteins from the sample manually before the SPE was automated. Pure methanol was used for protein precipitation. After manual loading of the centrifuge, the samples were centrifuged for 5 min at 3000 rpm in the method of Knox et al. The automated SPE consisted of six steps. First, cartridges used for the SPE were conditioned with methanol and then equilibrated with water. The purified sample was then added to the SPE cartridge and washed with a methanol–water solution. The elution was performed with methanol. Finally, water was added to the sample to adjust the polarity.
In the development process of the method, this basic procedure was adopted and first tested manually, before fully automating the method. An overview of operation for all three developed methods is given in Figure 1 , while Figure 2 shows method workflows. Adjustments in method 1 compared with the reference method were made with regard to volumes used and adding zinc sulfate solution to methanol to enhance protein precipitation. Furthermore, the dilution of the sample after SPE was eliminated. 64
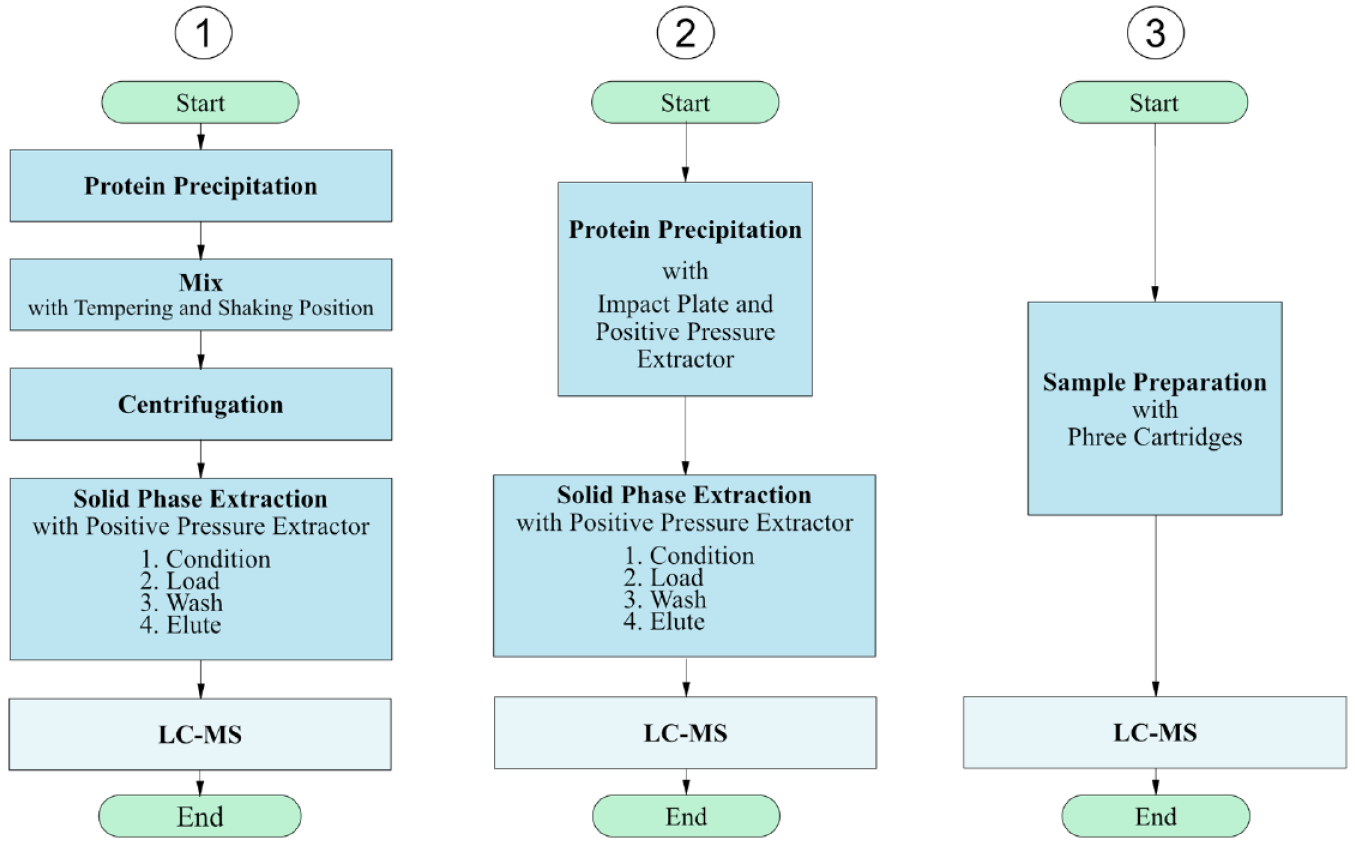
Comparison of the processes of method 1 using the Microplate Centrifuge VSpin, method 2 using the impact plate, and method 3 using the Phree cartridges.
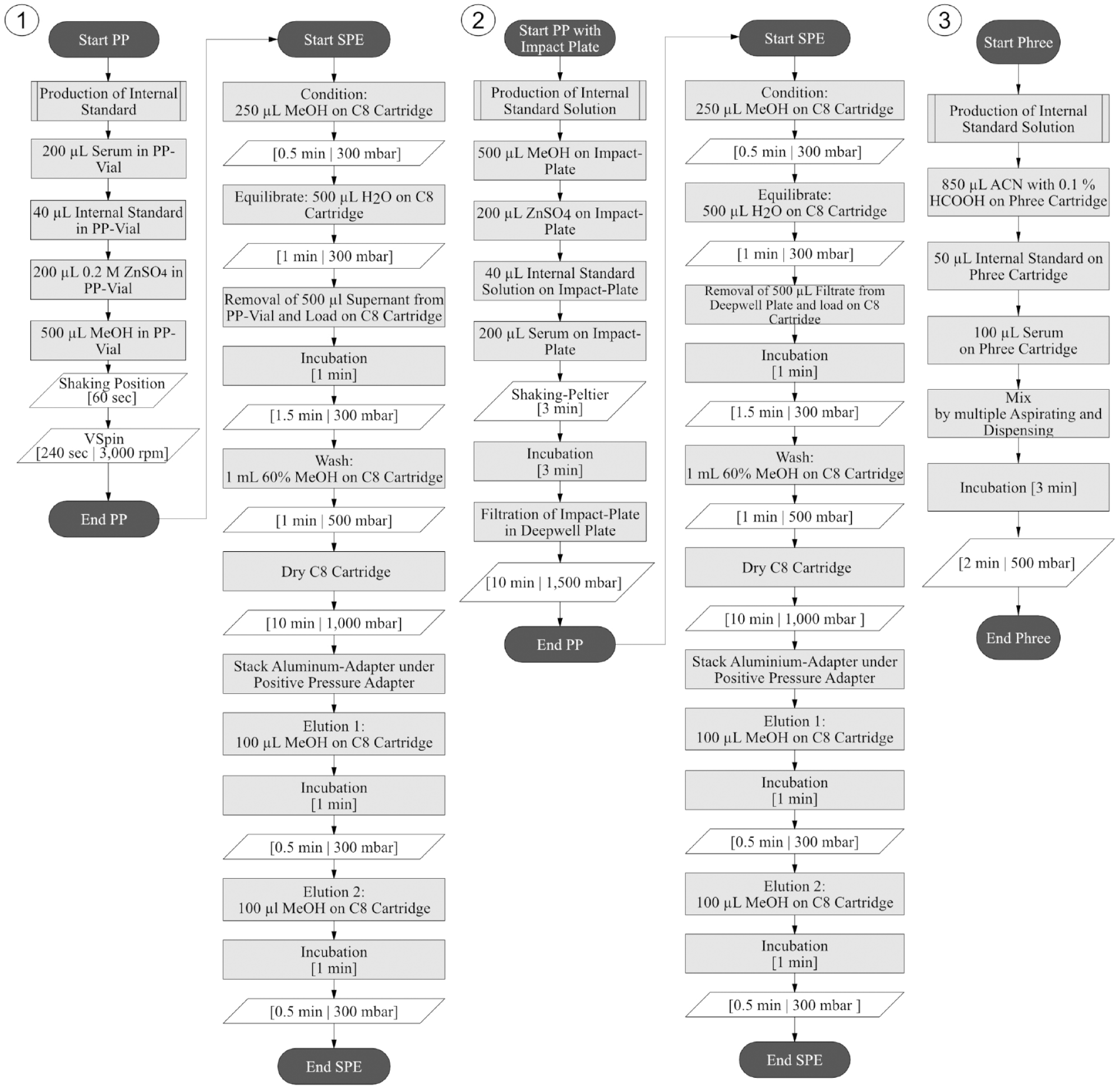
Program flowcharts of method 1 using the Microplate Centrifuge VSpin, method 2 using the impact plate, and method 3 using the Phree cartridges.
A simplification of the first method is realized in method 2 ( Fig. 1 ). In this method, the centrifugation step is replaced by a filtration step with the positive pressure extractor, eliminating the need to integrate a centrifuge into the deck layout. This is achieved by using the impact protein precipitation plate (Phenomenex). After protein precipitation with the impact plate, the SPE is performed in analogy to method 1. As a result, the investment costs are reduced.
In method 3, the protein precipitation and SPE are combined into one step using the Phree cartridges. This led to a further shortening of the method ( Fig. 1 ). Phree cartridges contain a mechanical filter for the retention of larger particles and a sorbent bed, which is specially designed for the adsorption of phospholipids. Since phospholipids cause interference in a subsequent LC-MS, their targeted removal allows us to abandon additional SPE. Figure 1 compares the procedure of the methods.
Labware
The samples are provided in 1.5 mL Eppendorf safe-lock vials (Eppendorf, Hamburg, Germany). Eppendorf vials are provided in a specially designed and manufactured adapter (CELISCA, Rostock, Germany) ( Fig. 3b ) on the deck of the Biomek i7 Workstation.

(
Protein precipitation is performed in method 1 in standard 2 mL vials (Agilent Technologies, Santa Clara, CA), which are also stored in an adapter developed at CELISCA ( Fig. 3d ). In all methods, standards are provided in standard 2 mL vials, stored in an aluminum adapter (CELISCA) ( Fig. 3e ), and closed with a Greiner Multiwell Plate Lid (VWR International, Radnor, PA) to ensure the stability of the standards through the closure and additional cooling. The final elution is also performed in standard 2 mL vials that are stored in the aluminum adapter. The positive pressure adapter (amplius, Rostock, Germany) ( Fig. 3c ) is used to position Strata C8 1 mL cartridges (Phenomenex) for SPE.
Deck Design and Automated Methods
In order to integrate protein precipitation and centrifugation into the automation system and to achieve a fully automated sample preparation, the Biomek i7 Workstation (Beckman Coulter Life Sciences) ( Fig. 3a ) is used. In the Biomek i7 Workstation, the Microplate Centrifuge VSpin (Agilent Technologies) ( Fig. 3h ) can be integrated, allowing the workstation to supply the automated centrifuge loader (Agilent Technologies) ( Fig. 3h ) and perform pipetting steps necessary for protein precipitation. The Microplate Centrifuge VSpin is operated at 1000g for 240 s, which also corresponds to the top speed of the centrifuge of 3000 rpm. The integration of the SPE is achieved by embedding the positive pressure extractor (amplius) ( Fig. 3i ) into the deck of the Biomek i7 Workstation in the same way. The positive pressure extractor is used for the automated application of positive pressure for filtrations or extractions, for example, as part of an SPE. The positive pressure extractor is loaded by the gripper of the Biomek i7 Workstation and can provide pressures of up to 100 psi (approximately 7 bar) on cartridges in an adapter or filter plates. The minimum required clamping pressure is 17.4 psi (1.2 bar). An important feature is the flow reduction, which prevents drying of a single cartridge or well.
In addition to the positive pressure extractor and the Microplate Centrifuge VSpin, the deck of the Biomek i7 Workstation is equipped with a number of special solutions. The self-refilling quarter reservoir (amplius) ( Fig. 3g ) is used to provide liquids that are needed in large quantities, such as acetonitrile (see method 3). It consists of four individual liquid containers with a filling volume of 40 mL, which are filled by a pump from corresponding supply bottles. An ultrasonic sensor measures the liquid level and actuates automatic refilling when the liquid level decreases with a liquid aspiration. In methods with large solvent consumption, the use of the self-refilling quarter reservoir saves deck positions and reduces the evaporation of volatile solvents. Furthermore, the Peltier tempering position is used for cooling standard solutions to prevent evaporation, as well and the tempering and shaking position (Inheco, Martinsried, Germany), which is used for mixing steps. To remove small amounts of liquids and to reduce the amount of residual solvent volume, labware can be beveled up to 15 degrees in a three-dimensional way with the 3D Tilt ALP (amplius) ( Fig. 3f ), which saves material and costs.
The deck design is created with the aim of achieving the highest possible throughput while ensuring a high degree of user-friendliness, so that the same labware is placed next to each other. Furthermore, the deck design and the setup of the methods allow a flexible adjustment of the number of samples to be prepared. In the first method in Figure 4 , the Microplate Centrifuge VSpin is integrated on the left side of the deck. To avoid an imbalance in the centrifuge, a counterweight is placed. On the right side of the deck, the positive pressure extractor for the subsequent SPE is integrated. With this deck, up to 96 samples can be prepared in one run. Due to the specific method, higher volumes have to be delivered. The processing of 96 samples requires 96 mL of methanol/water (60/40, v/v) for column washing, 48 mL of water to equilibrate the column, 19.2 mL of 0.2 M zinc sulfate for protein precipitation, and 98.995 mL of methanol in the overall process. To limit the number of deck positions, the self-refilling quarter reservoir is integrated to provide these high solvent quantities. The same pipette tips in 1070 µL, 230 µL, and 90 µL are utilized in all methods. There are differences in the number of pipette tips necessary due to the number of pipetting steps (11 steps with methods 1 and 2, 3 steps with method 3). These are higher in the first two methods because protein precipitation and SPE are performed one after the other. Due to the omission of a separate SPE when using Phree cartridges, significantly fewer pipetting steps and therefore also pipette tips are required for preparation than in methods 1 and 2.
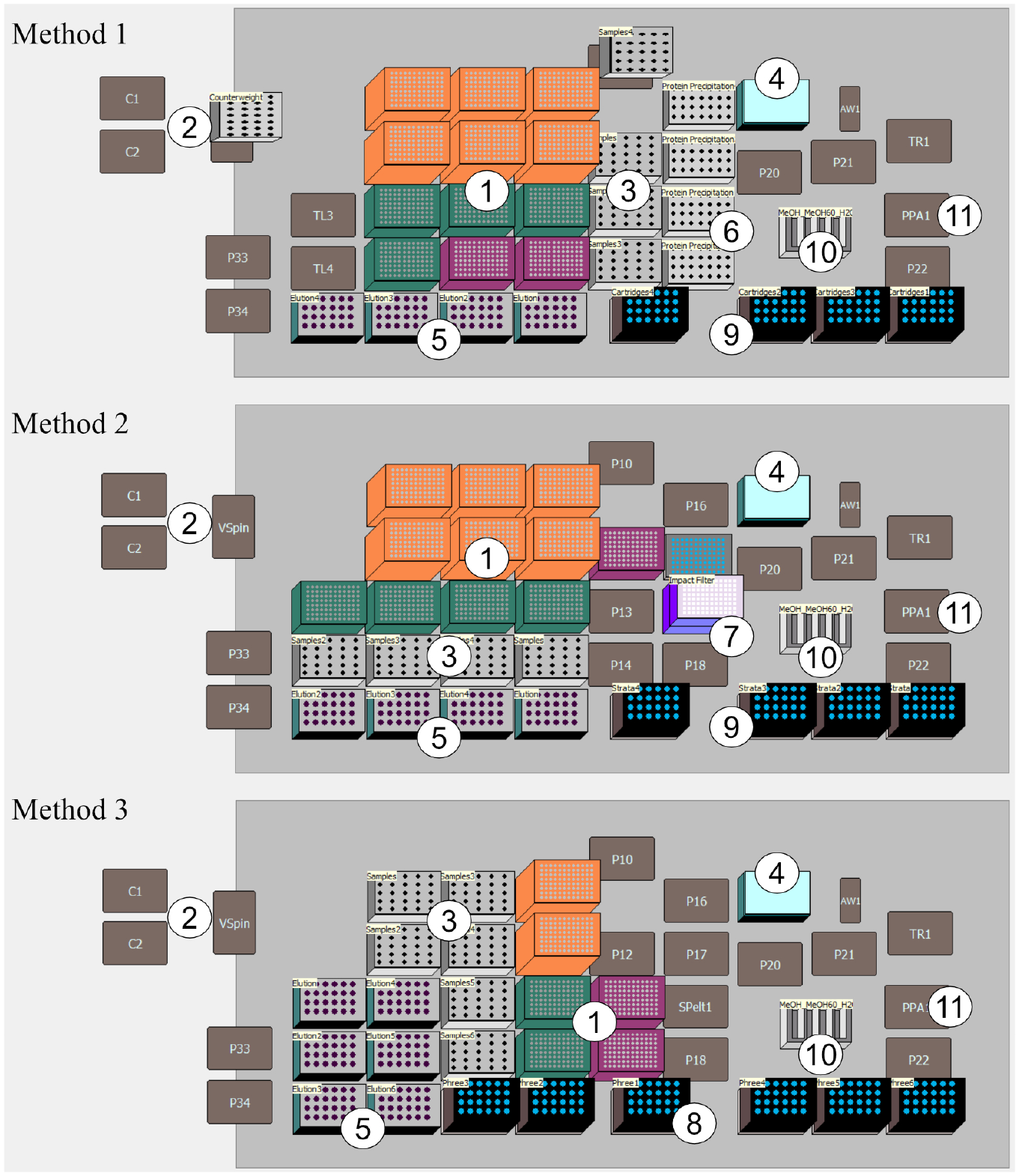
Deck layouts for vitamin D determination: (1) tip boxes, 1070 µL, 230 µL, and 90 µL; (2) centrifuge (in centrifuge supplementary counterweight); (3) samples; (4) internal standard (ISTD) in adapter made of aluminum; (5) adapter for elution vials made of aluminum; (6) vials for protein precipitation; (7) impact protein precipitation plate with deep-well collecting plate; (8) Phree phospholipid removal cartridges; (9) adapter for SPE cartridges; (10) self-refilling quarter reservoir; and (11) position of positive pressure extractor.
As with method 1, up to 96 samples can be prepared in one run with method 2. In order to ensure that the protein binding is disrupted prior to separation of the protein aggregate and supernatant, the sample and solvent are thoroughly mixed with the tempering and shaking position and then separated by positive pressure using the positive pressure extractor. The integration of a centrifuge in the deck design is therefore not necessary. With method 3, up to 144 samples can be prepared in one run. By replacing the classic steps of protein precipitation and SPE with Phree cartridges, fewer pipetting steps and labware are required, leaving space on the deck for further runs. This is also reflected in the shortening of the method in
Chemicals and Consumables
The following chemicals and standards were used for method development and subsequent testing: methanol (MeOH; HPLC gradient grade) and acetonitrile (ACN; HPLC gradient grade) were purchased from Carl Roth (Karlsruhe, Germany). Zinc sulfate solution (ZnSO4 [2 M]), porcine serum (500 mL), and standard solutions of D6-25-hydroxy-vitamin D3 (50 µg/mL), 25-hydroxy-vitamin D3 (100 µg/mL), and 25-hydroxy vitamin D2 (50 µg/mL) were purchased from Sigma-Aldrich (Steinheim, Germany). Furthermore, real pig serum samples provided by the Leibniz Institute for Farm Animal Biology (FBN, Dummerstorf, Germany) and the State Office for Agriculture, Food Safety and Fishing (LALLF, Rostock, Germany) were used.
Strata C8 (55 µm, 70 Å), 30 mg, 1 mL cartridges (Phenomenex) were used for SPE (methods 1 and 2). In method 2, the Impact Protein Precipitation 2 mL 96-Square Well Filter Plate (Phenomenex) was utilized to separate protein aggregates and supernatant before SPE. In method 3, Phree phospholipid removal 1 mL tubes (Phenomenex) were exclusively applied.
Instrumentation
All sample preparation steps (methods 1–3) were performed using the Biomek i7 Workstation (Beckman Coulter Life Sciences), including the previously described positive pressure extractor, the Microplate Centrifuge VSpin, and integrated special solutions. Samples were analyzed by injecting 10 µL of the sample into an LC/time-of-flight (TOF)-MS system (Agilent Technologies) with a flow rate of 0.5 mL/min. To increase the lifetime of the reverse-phase column Zorbax Eclipse PAH (Rapid Resolution HT, 4.6 × 50 mm, 1.8 µm, 600 bar), a 1260 Infinity LC in-line filter (4.6 mm) was installed before the chromatographic system. In addition to this measure, regular flushing with methanol and acetonitrile was performed. The mobile phase consisted of 92% methanol with 0.1% formic acid and 8% water with 0.1% formic acid. To avoid peak carryover, the run time of the method was 20 min. A dual ESI in positive ionization mode was used. The system was calibrated according to the internal standard method in the range of 0.01–2 ppm 25(OH)D2 and 25(OH)D3. Calibration curves for both analytes are shown at the top of Figure 5 . Typical chromatograms and spectra of analytes 25(OH)D2 and 25(OH)D3 are also given in Figure 5 .
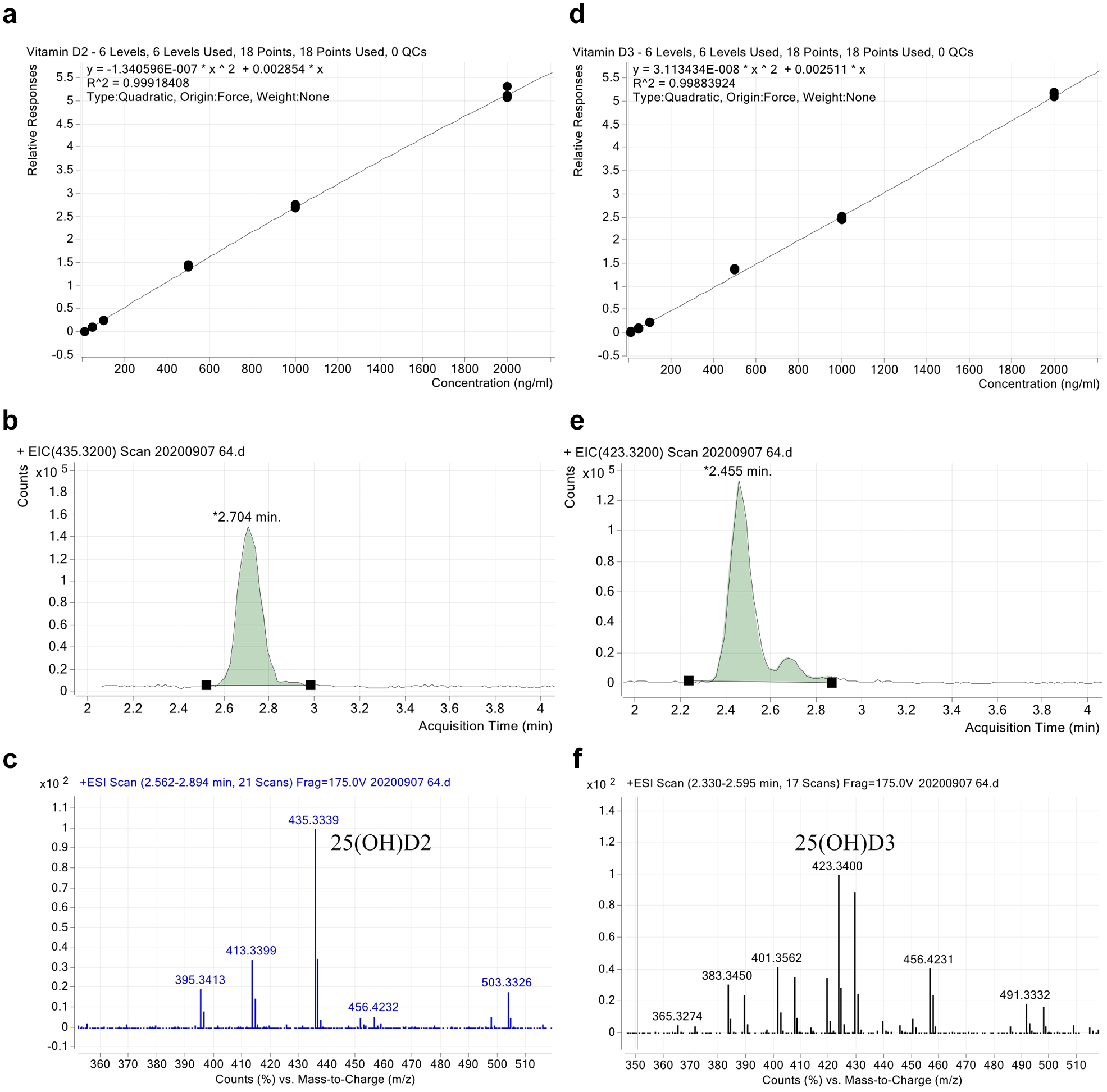
Calibration curves, chromatograms, and spectra for (
Results
Method Evaluation
For the comparative evaluation of methods, the performance characteristics LOD, LOQ, measurement precision, within-laboratory precision, and recovery were determined. To determine the LOD and LOQ, 10 blank samples containing solely the internal standard were prepared and analyzed. The analytical LOD was calculated from the mean value of the blanks plus three times the standard deviation of the blank. In analogy, the analytical LOQ is calculated from the mean value of the blanks plus 10 times the standard deviation of the blank value. Methodical LODs and LOQs were calculated from the analytical LODs and LOQs using the dilution factor of each method to determine the serum concentrations that would be detectable. To determine the measurement precision, a sample was measured 10 times in series, and the CV was calculated. To evaluate the within-laboratory precision, 10 samples with added 25(OH)D2 and 25(OH)D3 and four blank samples with serum were prepared and measured on five different days. In addition, the recovery was determined from 20 samples prepared and measured on the same day and four blanks with serum, by indicating the mean value of the measured concentration normalized to the measured value of the internal standard of a calibration solution. 65
The results of the method evaluation are presented in Table 1 . The recovery of the methods for 25(OH)D2 ranges from 77.76% to 97.36% and for 25(OH)D3 from 83.53% to 102.5%. The highest recovery was achieved with method 3. An influencing factor could be the assumed analyte loss during SPE with methods 1 and 2. In addition, the higher recovery in method 3 is particularly attributed to the targeted removal of phospholipids by Phree cartridges used, which reduce ionization disturbances at the ion source. This conclusion is consistent with the results reported from Carmical and Brown 50 and Tulipani et al. 59
Evaluation Results.
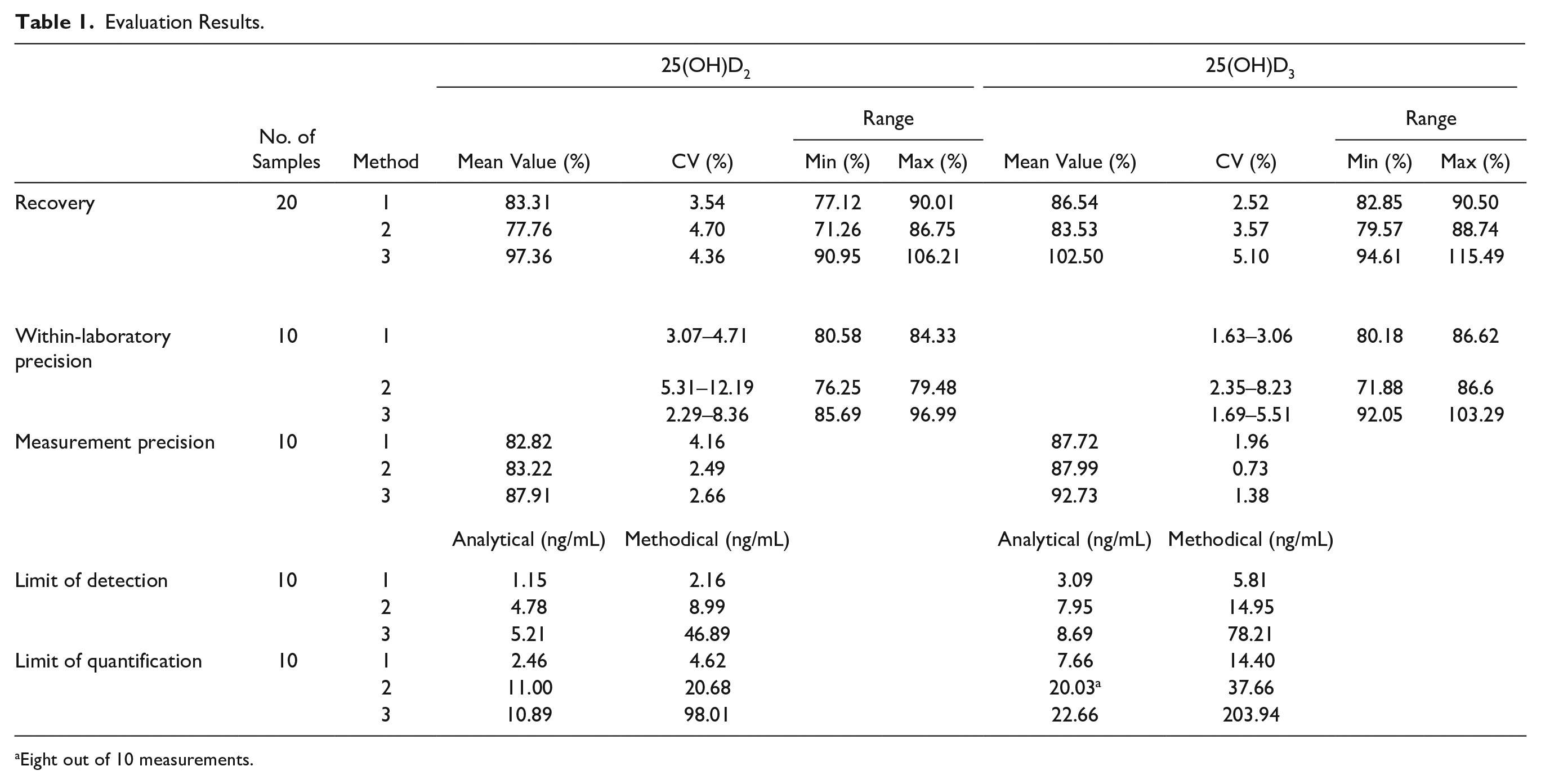
Eight out of 10 measurements.
Compared with the reference method, recovery rates for both analytes in method 1 are slightly lower than in the method proposed by Knox et al. (R25(OH)D2 = 90% and for R25(OH)D3 = 93%), in which no ion suppression effects were observed. CVs of within-laboratory precision are in all three methods for 25(OH)D2 less than 13% and for 25(OH)D3 less than 9%; in method 1, CVs for both analytes are below 5%. This is valid for the measurement precision of the developed methods.
LODs in the developed methods range from 1.15 to 5.21 ng/mL for 25(OH)D2 and 3.09 to 8.69 ng/mL for 25(OH)D3, and LOQs range from 2.46 to 10.89 ng/mL for 25(OH)D2 and 7.66 to 22.66 ng/mL for 25(OH)D3. Method 1 is the most sensitive method, which corresponds to the classic sample preparation method in an automated way. With the reference method of Knox et al., serum concentrations of 1.32 ng/mL for 25(OH)D2 and 25(OH)D3 can be detected. With method 1, the lowest detectable serum concentration of 25(OH)D2 is 2.16 ng/mL and of 25(OH)D3 is 5.81 ng/mL.
Consequently, the sensitivity obtained by using the LC-MS is lower than reported sensitivities (0.2-3 ng/mL)33,38,46,47,49 by using MS/MS.
Nevertheless, since the reference range for adults is between 30 and 60 ng/mL, the sensitivity required for the quantification of 25(OH)D2 and 25(OH)D3 in serum samples is achieved with the developed method 1 and could further be enhanced by using an MS/MS.
Higher detection limits obtained for method 2 are attributed to the nonremoval of further matrix components by replacing classic protein precipitation with the impact plate in method 2, resulting in a stronger background noise. Method 3 showed lower LOD and LOQ values, which can be attributed to a lower concentration of the sample solution in the course of sample preparation.
This result, together with the results of David et al. 66 and Nilsson et al., 60 leads to the conclusion that a classic protein precipitation step with centrifugation combined with an SPE step with selective phospholipid removal would result in the cleanest extracts.
Profitability Assessment
In order to enable an economic comparison of sample preparation methods, costs per sample are calculated with the cost comparative calculation, due to the different throughput of methods. In the cost comparative calculation, operating costs, including material costs, wage costs, maintenance costs, and energy costs; capital costs, which consist of depreciation and interest rates; and the possible number of samples per year are bonded to calculate costs per sample.
Costs like material and investment costs are based on price information from providers. Material costs show a large influence on costs per sample due to their throughput coupling and are to be reduced. Investment costs, such as acquisition costs for equipment like the Biomek i7 Workstation, the centrifuge, and the 3D Tilt ALP, but also for reusable adapters, are included in the annual depreciation and the annual imputed interest rate. In contrast to material costs, the importance of the investment costs decreases with an increasing number of samples. Only the investment risk rises with increasing capital commitment. Further, unknown or varying costs are defined and used in all methods, such as costs of labor, with 72 USD/h for a lab technician, or the interest rate, which is assumed to be 1%, flowing into operating costs. The depreciation period is set to be 10 years. Costs for maintenance are also part of operating costs, with approximately 6000 USD/year. To calculate the number of possible samples that can be prepared, a working week of 5 consecutive days and 24 h operating time for 50 weeks/year is considered, to create, among other things, free slots for maintenance and repair work. The variable method run time is therefore implied by the number of possible samples. The fixed definition of the unknowns through all methods ensures the comparability of the methods. Finally, the total costs of a method and the possible number of samples per year result in costs per sample. A summary with major outcomes from the cost comparative calculation is given in Table 2 . The main cost groups and resulting costs per sample are shown. The lowest costs per sample offers method 3 with approximately 6.79 USD, due to the high sample throughput. Method 1 follows with approximately 9.99 USD/sample. With approximately 10.93 USD, method 2 has the highest costs per processed sample.
Summary of Profitability Assessment (10 Years’ Depreciation).
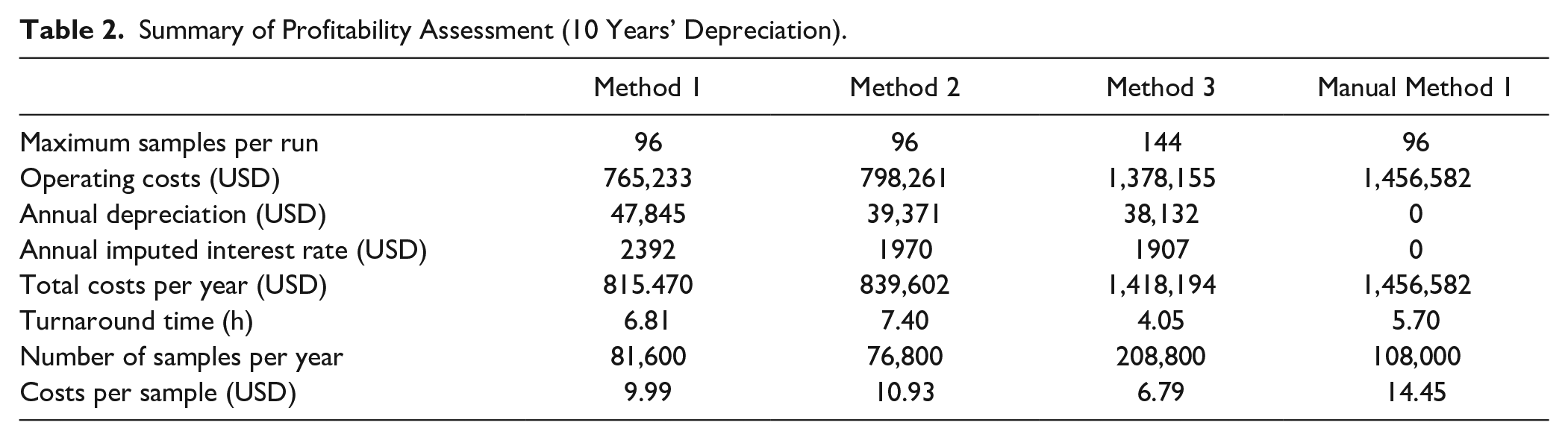
In order to support the previously made statement that manually performed sample preparation can lead to increased costs compared with automated methods, the economic analysis for a manual performance of method 1, which we had initially also tested manually, is calculated. First, we assume that no investment costs are incurred with the manual method and that, consequently, neither depreciation nor imputed interest is generated. However, since operating costs (1,456,582 USD) are significantly higher in relation to the throughput (108,000 samples/year) due to the labor costs now incurred, costs per sample increase to 14.45 USD. Since the breakeven point for all methods is 13.2 USD/sample, an increasing loss per period would be generated for sample preparation if method 1 was performed manually. In addition, the assumed time of 5.7 h for 96 samples is the ideal case of uninterrupted human work in the laboratory.
After comparing developed methods with one another based on the cost comparison calculation, we benchmark them against the reference method based on the throughput. In the reference method of Knox et al., with the CTC PAL (CTC Analytics, Zwingen, Switzerland), 80 samples can be prepared per working day. A time span of 8.5 h is assumed as a working day. To allow better comparability to the reference method of Knox et al., we shorten our time range from 5 days of 24 h each to a working day of 8.5 h and adjust the possible number of samples to this time range. Under these conditions, 96 samples are possible with methods 1 and 2, whereas with method 3 even 288 samples is possible in 8.5 h. This gain corresponds to an increase in throughput of up to 260% with regard to the reference method, using the fully automated sample preparation.
Discussion
In this work, we reported three methods for a fully automated sample preparation for the determination of vitamin D from plasma and serum samples. Liquid handling was performed using the Biomek i7 Workstation. The Microplate Centrifuge VSpin and the positive pressure extractor were integrated into the workstation to represent the classic procedure of sample preparation (method 1). In addition, other devices were used to improve liquid handling, such as the 3D Tilt ALP, to enable the smallest amounts of liquid to be removed from standard 2 mL vials, which have a concave bottom, making vertical removal of the liquid residue difficult. To avoid the integration of a centrifuge into the deck of the workstation, in method 2 the centrifugation step was replaced by a filtration step with the impact plate. Finally, in method 3, filtration and extraction were combined into one step by using the Phree cartridges, which led to a significant reduction in time and also simplification of the method in terms of, for example, the number of pipetting steps (from 11 to 3 steps comparing methods 1 and 3) required. A further optimization of methods consisted of the use of the self-refilling quarter reservoir, which can be used to provide large amounts of solvent, thus reducing solvent evaporation and effort needed, by eliminating the refilling process. Subsequently, the technical and economic evaluation of the methods was conducted.
Results of the evaluation and results of profitability assessment have shown strengths but also weaknesses of developed methods. Based on the throughput increase of 260% in comparison with the reference method, the effect of method simplification could be impressively demonstrated. Even the use of several special devices such as the 3D Tilt ALP or the quarter reservoir did not show any negative effects from an economic point of view, since higher acquisition costs are compensated by the higher sample throughput.
Around 30% of people in Germany have a deficiency in vitamin D in the winter months. 67 Early detection and subsequent therapy can alleviate or even prevent numerous diseases. This results in an increasing demand for corresponding diagnostics. The maximum number of samples of 570 per day is an entirely realistic value for the number of samples in large clinical diagnostic centers in large cities and metropolitan areas.
However, since the high throughput is enabled by the low turnaround time (manual procedure, 5.70 h; method with Phree Cartridges, 4.05 h), smaller laboratories can also consider automated methods for themselves by collecting samples, preparing them quickly, and having the time gained available for other test procedures. With this approach, values we calculated may cause an increase in costs, but they will still vary proportionally across all methods and the economic advantage is not lost. Moreover, values calculated in the price determination are only used as a tangible illustration of the influence of parameters used. It also has to be mentioned that the throughput for the manual sample processing was calculated with three lab technicians for 8 h each, processing exclusively vitamin D samples. In contrast, automated methods require the employee only during the pre- and postprocessing of a run, for example, to reload the deck. This factored-in time is about 0.5 h/run. The remaining working time of the employee can thus ideally be used to operate other instruments with other samples. In times when many laboratories are faced with an increasing shortage of skilled technicians, this relieving of staff can be a great benefit for the laboratories.
In terms of qualitative evaluation, the highest recovery rates of developed methods (97% for 25(OH)D2 and 103% for 25(OH)D3) were achieved with method 3. It is therefore concluded that the removal of phospholipids has a significant influence on the reduction of matrix effects and thus high recovery rates. Nevertheless, this method also reflects the shortcomings through the increase in the LOD of the LC-MS used. Due to the shortening of the method, other matrix components than phospholipids, which have been previously removed by the two-step sample preparation (protein precipitation followed by centrifugation and SPE), may have led to an increase in baseline noise. However, this effect can be compensated by using MS/MS.
Biomek i-Series Automated Workstations are not intended or validated for use in the diagnosis of disease or other conditions.
©2021 Beckman Coulter, Inc. All rights reserved. Beckman Coulter, the Stylized Logo, and Beckman Coulter product and service marks mentioned herein are trademarks or registered trademarks of Beckman Coulter, Inc. in the United States and other countries.
All other trademarks are the property of their respective owners.
This protocol is for demonstration only and is not validated by Beckman Coulter. Beckman Coulter makes no warranties of any kind whatsoever express or implied, with respect to this protocol, including but not limited to warranties of fitness for a particular purpose or merchantability or that the protocol is noninfringing. All warranties are expressly disclaimed. Your use of the method is solely at your own risk, without recourse to Beckman Coulter. Not intended or validated for use in the diagnosis of disease or other conditions.
Footnotes
Acknowledgements
The authors wish to thank Beckman Coulter (Beckman Coulter Life Sciences, Indianapolis, IN) for providing the Biomek i7 Workstation for this project. In addition, we would like to thank the Leibniz Institute for Farm Animal Biology (FBN, Dummerstorf, Germany) and the State Office for Agriculture, Food Safety and Fishing (LALLF, Rostock, Germany) for supplying the sample material generously.
Furthermore, we wish to gratefully acknowledge Dr.-Ing. Sebastian Neubert, Dr.-Ing. Thomas Roddelkopf, Dipl.-Ing. Lars Woinar, Heiko Engelhardt, Jan Hinnerichs, Sybille Horn, and Anne Reichelt, BSc, for valuable input, discussions, and technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is based on a bilateral cooperation contract between the Center for Life Science Automation/University Rostock (Germany) and Beckman Coulter Life Sciences (Indianapolis, IN). Both partners contributed equally to the research, authorship, and publication of this article.
