Abstract
Many important vaccines use bacterial capsular polysaccharides, or shorter polysaccharides or oligosaccharides, derived from the capsular polysaccharides, conjugated to protein. It is imperative that manufacturers understand the carbohydrate composition of these vaccines and deliver a product with a consistent polysaccharide or polysaccharide conjugate composition and content. High-performance anion-exchange chromatography with pulsed amperometric detection (HPAE-PAD) is a major technique used to understand the carbohydrate composition of these vaccines and ensure product quality. HPAE-PAD separates and detects carbohydrates without analyte derivatization. This paper describes the basics of the HPAE-PAD technique and then reviews how it has been applied to Haemophilus influenzae type b, pneumococcal, meningococcal, group B streptococcal, and Salmonella polysaccharide and corresponding conjugate vaccines.
Introduction
In the late 18th century Edward Jenner developed a vaccine for smallpox, which was the first successful vaccine. Today there are several vaccines that are important for preventing the occurrence and spread of debilitating and often life-threatening diseases. Some of these vaccines are developed using a polysaccharide isolated from a strain (serotype or serovar) of the targeted disease-causing bacteria. While the vaccine can sometimes be developed using only the polysaccharide, often a more effective vaccine is created by conjugating the polysaccharide, or an oligosaccharide derived from that polysaccharide, to a protein. These are classified as glycoconjugate vaccines.
As vaccines are medical products for human use, they require rigorous characterization and assays to ensure final product quality and consistency. For a glycoconjugate vaccine this includes quantifying the amount of polysaccharide conjugated as well as checking for polysaccharide impurities. High-performance anion-exchange chromatography with pulsed amperometric detection (HPAE-PAD) is a common carbohydrate analysis technique, and it has been applied to assaying the quality and consistency of polysaccharide and glycoconjugate vaccines as well as the development of these vaccines, which include Haemophilus influenzae type b (Hib), Streptococcus pneumoniae, and Neisseria meningitidis vaccines.
HPAE-PAD was first described more than 35 years ago.1,2 Since then it has been applied to the determination of a wide variety of carbohydrates in an even wider variety of samples. HPAE-PAD separates and detects carbohydrates without analyte derivatization. Carbohydrates determined by HPAE-PAD include monosaccharides, disaccharides, sugar alcohols, sugar phosphates, amino sugars, sialic acids and other sugar acids, oligosaccharides, and small polysaccharides. The separation is by HPAE. While most carbohydrates are not anions, uncharged carbohydrates are anions at high pH (>12). Therefore, separations use sodium or potassium hydroxide mobile phases. For many carbohydrates this is sufficient for separation, but if the carbohydrate is large, or negatively charged at pH 7, a stronger mobile phase is needed. For these carbohydrates, typically sodium acetate is added to the hydroxide mobile phase. These separations are high resolution with the ability to separate structurally similar carbohydrates. The type of carbohydrate being determined dictates the choice of the HPAE column, commonly one of the CarboPac column family.
After separation the carbohydrate must be detected. Because most carbohydrates lack a good chromophore, they were traditionally difficult to detect with good sensitivity without creating either a volatile derivative for gas chromatography or a UV-absorbing or fluorescent derivative for liquid chromatography. That changed with the introduction of PAD, which detects the native carbohydrate with good sensitivity. PAD works by oxidizing a small amount of the eluting carbohydrate on a gold working electrode in strong alkaline solution, which is ideal because the mobile phase required for HPAE is alkaline. The current generated by the carbohydrate over a given time period is integrated to yield charge that is plotted versus time to produce the HPAE-PAD chromatogram. More details on HPAE-PAD and its application to a wide variety of carbohydrates and samples from diverse industries and areas of scientific study can be found in review articles.3–9
The first published account of applying HPAE-PAD to vaccine characterization was in 1992. 10 The authors from Merck Research Laboratories were focused on developing the proper acid hydrolysis conditions for the capsular polysaccharides from Hib as well as S. pneumoniae types 6B, 14, 18, and 23F. Table 1 shows the repeating unit of these polysaccharides and other polysaccharides discussed in this review. They were able to find hydrolysis conditions that yielded the component sugar alcohols and monosaccharides from each polysaccharide, while also removing the phosphate from the sugar phosphates. These component carbohydrates were then quantified by HPAE-PAD to determine the composition, purity, and concentration of each polysaccharide. The carbohydrates separated from this set of polysaccharides were glycerol, ribitol (Rib), rhamnose (Rhm), glucosamine (GlcN), galactose (Gal), glucose (Glc), and ribose (Ribo). Figure 1 shows a typical determination of monosaccharides by HPAE-PAD. Monosaccharides are typically separated with hydroxide mobile phases using either a CarboPac PA1, PA10, or PA20 column. More details on HPAE-PAD monosaccharide analysis as applied to vaccines are discussed under the sections dedicated to individual vaccines.
Capsular Polysaccharide Repeating Unit and Carbohydrate(s) Measured by HPAE-PAD.
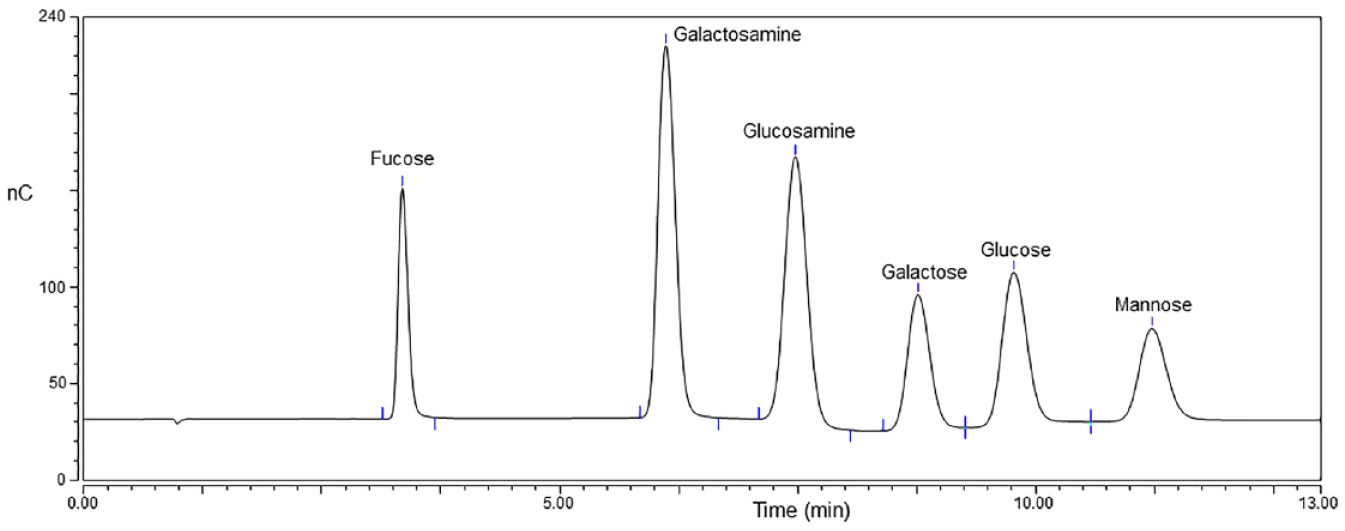
Monosaccharides separated and detected by HPAE-PAD.
Since that 1992 publication, HPAE-PAD has been applied to a few polysaccharide and glycoconjugate vaccines. The remainder of this review describes how HPAE-PAD has been applied to commercial vaccines, and some vaccines in development. Applications include carbohydrate compositional analysis of the bacterial polysaccharide, while also determining its purity and concentration. HPAE-PAD has also been used to assay the production of oligosaccharides from the polysaccharide. These oligosaccharides are used to make the glycoconjugate vaccine, and HPAE-PAD is also used to determine their composition. Other common applications include determining the amount of polysaccharide/oligosaccharide conjugated and the free or unconjugated polysaccharide/oligosaccharide in a vaccine, assessing contamination, and determining vaccine stability. The application of HPAE-PAD and other ion chromatography methods to Hib, S. pneumoniae, and N. meningitidis vaccines was the subject of an earlier review. 11
Haemophilus influenzae Type b Vaccines
HPAE-PAD has been extensively applied to Hib vaccines. These vaccines are based on the capsular polysaccharide polyribosyl ribitol-phosphate (PRP), made of 5-
In 2000 scientists from what was then Chiron in Siena, Italy, reported a different approach to quantifying polysaccharide in Hib conjugate vaccine. 15 This new approach was necessary because when their Hib conjugate vaccine was used in combination with the DPT vaccine there were interferences preventing quantification of the Hib subunit. Therefore, they used acid hydrolysis (trifluoroacetic acid [TFA]) to yield Rib and quantified it using a CarboPac MA1 column and a 650 mM sodium hydroxide mobile phase. They used this method to quantify both unconjugated and conjugated saccharide in the combination vaccine after an ultrafiltration step. Since their publication this has become an accepted method to assay Hib conjugate vaccine, and it was one of the assays used in two collaborative studies involving Hib glycoconjugate vaccines.16,17
The first of the two collaborative studies, which was organized by the World Health Organization (WHO), evaluated an international standard for the Hib capsular polysaccharide. 16 The evaluation included determining the polysaccharide content as well as its stability. Six of the 10 participating laboratories used HPAE-PAD for those evaluations. Five assayed the Hib subunit after alkaline hydrolysis, and one lab assayed Rib after acid hydrolysis. The study established the international PRP standard. The second more recent study was also organized by the WHO, but in collaboration with the Biological Standardisation Programme of the Council of Europe/European Directorate for the Quality of Medicines and HealthCare and of the European Union Commission. 17 In this study the aim was to establish a single method to determine the PRP content in a liquid combination vaccine protecting against five diseases. The method was acid hydrolysis and determination of Rib using a CarboPac MA1 column. By evaluating five liquid vaccine preparations, the method was found to be suitable for its intended purpose of determining conjugated and unconjugated PRP after an RP cartridge sample preparation.
Some more recent studies have expanded on prior work. The Hib subunit assay was used to determine the progress of PRP preparation and demonstrated that the HPAE-PAD method did not have the interferences of other methods that the authors tried for following the progress of PRP preparation. 18 Another study optimized the chromatography of the Hib subunit assay to improve peak shape and reduce analysis time. 19
In addition to these applications of HPAE-PAD to the Hib vaccine, the same group that designed the Rib assay used HPAE-PAD to evaluate the oligosaccharides resulting from the partial acid hydrolysis of the capsular polysaccharide. 20 These oligosaccharides are used for conjugation to produce the vaccine. This work used a column typically used for traditional ion chromatography, the IonPac AS11 column, with a mobile phase gradient of sodium hydroxide to separate the oligosaccharides.
Neisseria meningitidis Vaccines
Publications concerning the application of HPAE-PAD to these vaccines first appeared in the year 2000.20,21 The same publication that evaluated the oligosaccharides produced by partial acid hydrolysis of the Hib capsular polysaccharide also evaluated the oligosaccharides produced by partial acid hydrolysis of the N. meningitidis serotype A and C capsular polysaccharides. 20 There are glycoconjugate vaccines produced to protect against single serotypes of N. meningitidis as well as multivalent vaccines to protect against multiple serotypes. The serotypes targeted by commercial vaccines are A, C, Y, and W135. For serotype A, the capsular polysaccharide is poly-α1,6-N-acetylmannosamine-6-phosphate. The polysaccharide for serotype C is poly-α2,9-N-acetylneuraminic acid. For serotypes Y and W135, the polysaccharides are poly-α2,6-glucose-α1,4-N-acetylneuraminic acid and poly-α2,6-galactose-α1,4-N-acetylneuraminic acid, respectively. These polysaccharides each have varying levels of O-acetylation, a modification lost under the alkaline conditions of HPAE-PAD. Authors from what was then Aventis Pasteur in Swiftwater, Pennsylvania, used HPAE-PAD to determine the amount of free polysaccharide compared with conjugated polysaccharide in various glycoconjugate vaccines of the four serotypes. 21 Deoxycholate was used to precipitate conjugated polysaccharide from free polysaccharide, and each solution was hydrolyzed in TFA and analyzed for released sugars using HPAE-PAD with a CarboPac PA10 column and a GlcN-1-P internal standard. The authors make no mention of the sugars measured to quantify each serotype polysaccharide. Their method starts with a sodium hydroxide mobile phase and changes to a sodium hydroxide with a sodium acetate mobile phase. This suggests that they are measuring both neutral (uncharged at pH 7) and charged monosaccharides. Subsequent publications by other groups and the nature of the polysaccharides indicate that is what they did.
For serotype A, scientists at Chiron used TFA hydrolysis and then HPAE-PAD with an IonPac AS11 column to measure mannosamine-6-phosphate (ManN-6-P). 22 This phosphorylated monosaccharide would become the compound used to measure serotype A polysaccharide in multivalent vaccines. At the same facility, this method was used to measure the stability of the serotype A polysaccharide conjugation in a tetravalent vaccine. 23 They chose the AS11 column rather than the CarboPac PA1 column because the ManN-6-P was less tightly bound and only required a sodium hydroxide mobile phase. They did use the PA1 column with a hydroxide and acetate mobile phase to separate mannosamine, N-acetylmannosamine, ManN-6-P, and mannose-6-phosphate to optimize hydrolysis. Their method was used to measure the polysaccharide during production (fermentation) prior to its use in vaccine production and the development of serotype A vaccine. The single serotype vaccine is produced because the multivalent vaccine cannot be given to infants.
Around the same time as the Chiron publication a group in the Bacteriology Division of the National Institute for Biological Standards and Control (NIBSC) evaluated the stability of serotype C vaccines from two manufacturers, with HPAE-PAD used as part of their evaluation. 24 They used the CarboPac PA1 column and a sodium hydroxide/sodium acetate mobile phase to measure N-acetylneuraminic acid (Neu5Ac) after acid hydrolysis employing glucuronic acid as the internal standard. Quantifying Neu5Ac would become the way to measure serotype C polysaccharide in multivalent vaccines. Figure 2 shows HPAE-PAD chromatography of Neu5Ac together with another sialic acid, N-glycolylneuraminic acid. One measure of stability was to determine the loss of polysaccharide conjugation. Unconjugated polysaccharide was separated from conjugated polysaccharide by ultrafiltration. As with Hib glycoconjugate vaccines, vaccines containing aluminum phosphate were treated with sodium hydroxide (to dissolve the aluminum phosphate) before ultrafiltration.
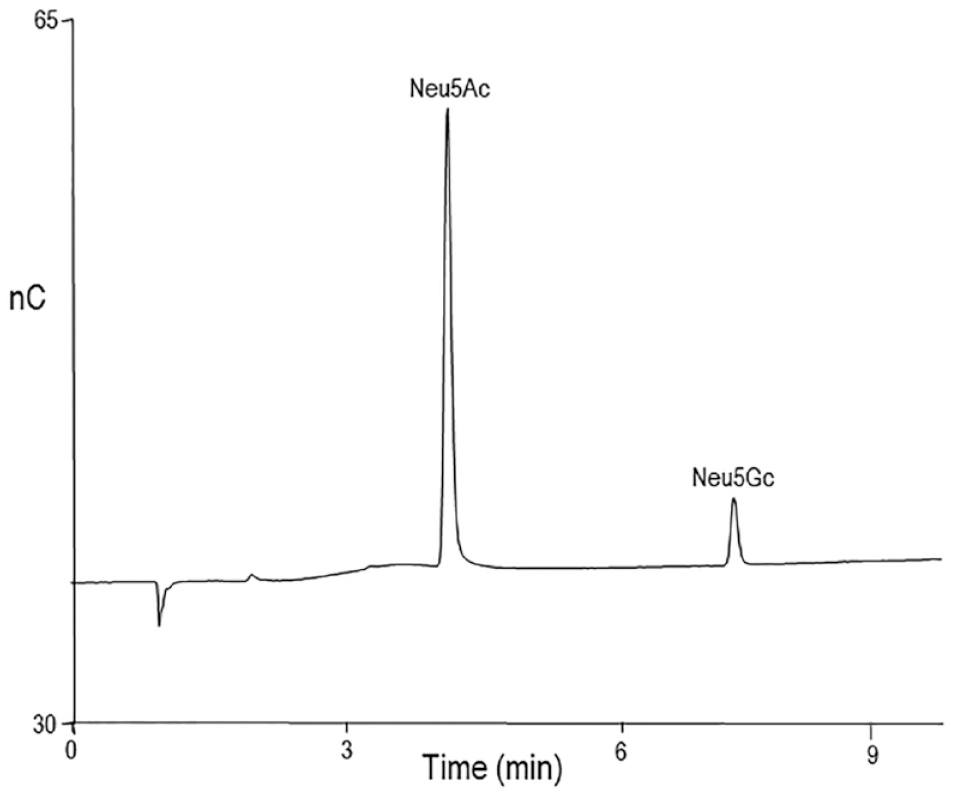
Sialic acids separated and detected by HPAE-PAD.
A group at Novartis (previously Chiron) published a characterization of a multivalent meningitis vaccine (A, C, Y, W135). 25 They used HPAE-PAD to assay the amounts of Y and W135 polysaccharide. After TFA hydrolysis, they separated the monosaccharides on a CarboPac PA1 column using Glc to quantify serogroup Y and Gal for serogroup W135. Assaying the polysaccharide content of each of the four polysaccharides in the tetravalent meningococcal vaccine using only HPAE-PAD was described by the Centre for Vaccine Evaluation, Biologics and Genetic Therapies Directorate of Health Canada. 26 They determined optimal acid hydrolysis conditions for each serotype polysaccharide, and then they measured one monosaccharide for each serotype. Optimization involved testing with multiple monovalent and polyvalent vaccines in different formulations. For formulations with carbohydrate excipients (e.g., lactose) they designed ways to remove the excipient. To measure serotypes A, C, Y, and W135, they used ManN-6-P, Neu5Ac, Glc, and Gal as previously discussed. To simplify the analysis, they designed a single multistep sodium hydroxide/sodium acetate mobile phase gradient on a CarboPac PA10 column to determine all four carbohydrates. They then applied their method to quantifying the serotype polysaccharides in multivalent meningococcal vaccines from China, including evaluating multiple lots of the same vaccine for product variation. 27 A group at JNI Medical Corporation in Omaha, Nebraska, used essentially the same approach with a CarboPac PA1 column and a different mobile phase gradient method to quantify each of the polysaccharides conjugated in the tetravalent vaccine in vialed vaccine and formulated final bulks. 28 The group at the NIBSC also used this approach with a variety of mobile phase conditions and either the CarboPac PA1 or PA10 column as part of their quality and stability studies of multivalent meningococcal vaccines. 29
The group at Chiron developed an interesting application of HPAE-PAD to determine the size of the oligosaccharides, which were derived from the serotype poly-saccharides Y and W135 by partial acid hydrolysis, used for conjugation to make a glycoconjugate vaccine. 30 The polysaccharide sample was divided in half. One half was hydrolyzed in acid and analyzed for total Neu5Ac. The other half was first reduced with sodium borohydride and then hydrolyzed in acid. The reduction will only reduce the terminal Neu5Ac. The degree of polymerization (size) was determined by measuring the reduced Neu5Ac in the second sample and ratioing it to the total Neu5Ac in the first sample. Reduced Neu5Ac and Neu5Ac are separated on a CarboPac PA1 column with a sodium hydroxide/sodium acetate mobile phase.
While most of the publication activity has concerned serotypes A, C, Y, and W135, there are other serotypes that have been subject to vaccine development. This includes serotype B. In 2004 a group from Baxter Healthcare in Beltsville, Maryland, published an HPAE-PAD assay to judge the integrity of a vaccine targeting serotype B. 31 The polysaccharide is an α2,8-linked linear homopolymer of Neu5Ac. They discovered that this polysaccharide was not amenable to acid hydrolysis when conjugated to the carrier protein. This led them to develop a different type of HPAE-PAD assay to assess free and conjugated polysaccharide. In their assay, they perform acidic methanolysis, which yields the de-N-acylated methylglycoside of Neu5Ac. This compound is determined by HPAE-PAD using a CarboPac PA1 column and a 500 mM NaOH mobile phase and a 2-keto-3-deoxynonulosonic acid internal standard added prior to methanolysis.
As with Hib glycoconjugate vaccines, the number of vaccines targeting meningitis has sparked efforts to develop reference standards.32,33 The NIBSC successfully developed an international standard for meningococcal type C polysaccharide. 32 Twelve labs participated in the collaborative study of that standard, with four labs using HPAE-PAD with either a CarboPac PA1 or PA10 column to quantify the polysaccharide by measuring Neu5Ac after acid hydrolysis. Their methods varied in terms of the hydrolysis method, internal standard, and mobile phase conditions. More recently, the NIBSC led a collaborative study to establish international standards for meningococcal serotype A and serotype X polysaccharides. 33 The serotype X polysaccharide is composed of repeating units of N-acetylglucosamine-4-phosphate residues linked by α1–4 phosphodiester bonds. Nine labs participated in the collaborative study, with three using HPAE-PAD. These three used either a CarboPac PA1 or PA10 column to measure ManN-6-P and glucosamine-4-phosphate (one lab measured GlcN) for serotypes A and X, respectively. As for the serotype C standard, methods varied in terms of the hydrolysis method, internal standard, and mobile phase conditions.
Streptococcus pneumoniae Vaccines
Many vaccines have been made from free and conjugated capsular polysaccharides from S. pneumoniae. As discussed earlier, the authors from Merck investigated acid hydrolysis conditions for S. pneumoniae serotype 6B, 14, 18, and 23F polysaccharides. 9 They found that when the polysaccharide contained phosphate, to generate phosphate-free monosaccharide it was necessary to treat it with hydrofluoric acid (HF) before TFA. These conditions were necessary for serotypes 6B, 18, and 23F, but not for serotype 14 because it lacks phosphate. Each serotype had an identifying set of monosaccharides. A group at what was then Aventis Pasteur in Marcy l’Étoile, France, took a different approach. 34 They developed acid hydrolysis conditions for serotypes 1, 3, 4, 5, 6B, 7F, 9V, 14, 18C, 19F, and 23F. For polysaccharides containing uronic acids (1, 3, 5, and 9V) they used methanolysis followed by TFA hydrolysis, and only TFA for the other polysaccharides. Following these treatments of free or conjugated polysaccharide, they determined the monosaccharides by HPAE-PAD using a CarboPac PA10 column and sodium hydroxide/sodium acetate mobile phases to quantify the amount of polysaccharide. The same group also developed an HPAE-PAD assay to determine C-polysaccharide contamination of polysaccharides isolated and used to make pneumococcal vaccines. 35 It is difficult to isolate these polysaccharides without some C-polysaccharide contamination, and it is highly immunogenic. They used the two-step acid hydrolysis procedure from Merck (i.e., HF followed by TFA) to yield Rib, which was quantified by HPAE-PAD using a CarboPac MA1 column and a 480 mM sodium hydroxide mobile phase. To optimize hydrolysis, they used their CarboPac PA10 method. 34 They tested their method with 23 serotype polysaccharides and could successfully detect C-polysaccharide contamination in 21. The other two serotype polysaccharides contain Rib.
In addition to quantifying pneumococcal polysaccharide and determining the amount of C-polysaccharide impurity, HPAE-PAD has been used to separate oligosaccharides derived from the partial acid hydrolysis of serotype 3 polysaccharide. 36 The authors used a similar approach as they used for meningococcal oligosaccharides derived in the same manner. 30 Namely, the oligosaccharides were separated with an IonPac AS11 column with a sodium hydroxide mobile phase gradient and detected by PAD. They isolated individual oligosaccharides for structural analysis.
Group B Streptococci Vaccines
The group at Novartis in Italy reported application of HPAE-PAD to group B streptococcus (GBS) vaccines. 37 They developed methods to quantify the serotype polysaccharides used for conjugation to make the vaccine. Specifically, they developed methods for serotype 1a and V polysaccharides during the different stages of vaccine production. Additionally, they developed a method to determine group B carbohydrate impurity. Group B carbohydrate is a common process impurity as noted by authors investigating the linkage of GBS capsular polysaccharides to the cell wall. 38 Both serotype 1a and V polysaccharides along with other GBS capsular polysaccharides have repeating units of Gal, GlcNAc, Glc, and Neu5Ac with differentiation provided by the linkages.
Different HPAE-PAD assays are used for type 1a polysaccharide depending on the step of the vaccine production. For purified 1a polysaccharide they measured Neu5Ac after mild acid hydrolysis using a CarboPac PA1 column and a sodium hydroxide/sodium acetate mobile phase. The same assay was used to measure vaccine stability. In that application, free Neu5Ac is measured in the filtrate after ultrafiltration of the conjugated polysaccharide. To conjugate the 1a polysaccharide the Neu5Ac is activated by partial oxidation. Therefore, Gal is measured to determine free and total polysaccharide. Free polysaccharide is separated from conjugated polysaccharide by treatment with an RP cartridge. Gal is determined using a CarboPac PA1 column and a sodium hydroxide mobile phase. In the vaccine formulation, mannitol is added prior to lyophilization. The high concentration of mannitol interferes with Gal quantification on the CarboPac PA1 column, so they used a CarboPac MA1 column with a strong sodium hydroxide mobile phase to quantify the vaccine in the product.
Type V polysaccharide concentration is measured by 4 M TFA hydrolysis and quantifying GlcN (GlcNAc before acid hydrolysis). To have an assay that can be used at different stages of vaccine production, Neu5Ac cannot be used because it is removed for conjugation and Gal is not used because it is activated for conjugation by partial oxidation. GlcN separation is on a CarboPac PA column with a sodium hydroxide mobile phase. This method can be used to determine polysaccharide purity, free and total in the conjugate, and the amount in the lyophilized product. Group B impurity is determined in both purified polysaccharides by measuring Rhm on a CarboPac PA1 column with a sodium hydroxide mobile phase after 2 N TFA hydrolysis.
Salmonella Vaccines
Salmonella enterica serovar Typhi causes typhoid fever. In 2011, scientists at Novartis in Siena, Italy, reported on a glycoconjugate vaccine to prevent typhoid fever. 39 They conjugated the linear capsular polysaccharide from Salmonella Typhi (Vi) to a protein carrier. This polysaccharide is a linear homopolymer of α1,4-N-acetylgalactosaminouronic acid, the majority of which is O-acetylated at the C-3 position. The Novartis group designed an HPAE-PAD assay to quantify the amount of polysaccharide as well as the extent of conjugation. They found that by doing an alkaline hydrolysis they could obtain a single peak in the HPAE-PAD method using a CarboPac PA1 column and a sodium hydroxide/sodium nitrate mobile phase. Note that they use sodium nitrate rather than sodium acetate as the strong eluting anion. Acidic hydrolysis yielded a series of oligosaccharides. A more recent publication concerning a glycoconjugate vaccine based on the Vi polysaccharide used this HPAE-PAD method to determine the amount of free and conjugated polysaccharide. 40
In order to develop Salmonella vaccines, Novartis investigated purifying O-antigen from several Salmonella serovars.41,42 O-Antigen is a polysaccharide with a repeating unit that includes mannose (Man), Rhm, Gal, and Glc. This polysaccharide is linked to a core oligosaccharide that is linked to lipid A through 2-keto-3-deoxyoctonate (KDO) to form lipopolysaccharide. HPAE-PAD was used as part of the characterization of the purified O-antigen. After acid hydrolysis, Man, Rhm, Gal, and Glc were separated with a CarboPac PA10 column using a sodium hydroxide mobile phase to quantify O-antigen, a method described earlier to quantify the O-antigen from the Salmonella enterica serovar Paratyphi A in a glycoconjugate vaccine. 43 GlcN was separated from the other four monosaccharides and used to quantify the core oligosaccharide and ratio to Rhm to determine the number of repeats in the O-antigen. The same approach was used to characterize a bivalent vaccine targeting Paratyphi A and Typhi as well as O-antigen to produce conjugate vaccines for nontyphoidal Salmonella disease.44,45
Summary
This contribution reviewed the application of HPAE-PAD to polysaccharide and polysaccharide conjugate vaccines. Thus far HPAE-PAD has been applied to Hib, GBS, meningococcal, pneumococcal, and Salmonella vaccines. Applications include quantifying the polysaccharide, determining the amount of polysaccharide attached and free in a glycoconjugate vaccine, measuring vaccine stability, and determining polysaccharide impurities. Table 2 summarizes how HPAE-PAD has been applied to the five groups of vaccines reviewed here. These HPAE-PAD assays ensure vaccine quality and consistency.
Applications of HPAE-PAD to Polysaccharide and Glycoconjugate Vaccines.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
