Abstract
Ocular drusen are extracellular deposits that form between the retinal pigmented epithelium (RPE) and Bruch's membrane. Although the presence of large and/or numerous drusen in the macula is a significant risk factor for development of age-related macular degeneration (AMD), a major cause of irreversible blindness, little is known about their origin or composition. We have expanded on our previous investigations related to drusen-associated glycoconjugates by examining lectin binding patterns after removal of terminal sialic acid residues. Strikingly, intense and distinct labeling of drusen subdomains is revealed by Arachea hypogea agglutinin (PNA) after neuraminidase treatment. PNA binding is confined to discrete domains within both hard and soft drusen. These “cores” are positioned centrally within drusen and are typically juxtaposed to Bruch's membrane. Only one core per druse is observed. PNA labeling of drusen cores does not co-localize with associated lipids and is abrogated by digestion with O-glycosidase but not N-glycosidase. The association of cores with small drusen suggests that they may participate in drusen biogenesis.
A number of studies have focused on characterizing the structure of drusen, largely in attempts to classify their phenotype. However, relatively little is known about their composition and/or genesis. One commonly held view, supported by a number of clinicopathological studies, is that small “hard” drusen are precursors of large “soft” drusen (Sarks et al. 1980, 1994). It has been proposed that drusen form after expulsion of RPE cytoplasm, organelles, and/or basal laminae into Bruch's membrane, possibly as a mechanism for removing damaged cytosol (Farkas et al. 1971; Burns and Feeney Burns 1980; Ishibashi Patterson et al. 1986a,b). Other mechanisms of drusen biogenesis, including formation of drusen from lipoidal degeneration of the RPE (Fine 1981; El Baba et al. 1986) or from vascular components (Friedman et al. 1963), have been proposed. It is likely that the deposition of drusen, which may reach sizes of hundreds of micrometers or more in diameter, is initiated by an early “nucleation” event that is followed by aggregation of macromolecules, resulting in their growth (Sarks et al. 1994; Hageman et al. 1999).
One impediment encountered in efforts to determine the origin of drusen relates to our overall lack of knowledge about their composition. To date, only a few studies have sought to identify and/or characterize drusen-associated proteins (Newsome et al. 1987; van der Schaft et al. 1993; Hageman et al. 1999; Hageman and Mullins in press), lipids (Pauleikhoff et al. 1992; Sheraidah et al. 1993), glycosaminoglycans (Kliffen et al. 1996), and other carbohydrates (Kliffen et al. 1994; Mullins et al. 1997). To better understand the composition of different drusen phenotypes, we previously characterized drusen-associated glycoconjugates using lectin histochemistry (Mullins et al., 1997). We identified a number of carbohydrate moieties in drusen and have found that drusen contain sialic acid, β-galactose, N-acetylglucosamine, and glucose/mannose residues but do not appear to contain galactosamine-containing saccharides. The study presented here was directed towards further characterization of sialic acidcontaining glycoconjugates within drusen. Distinct carbohydrate-rich subdomains were identified within drusen after exposure of hard and soft drusen to neuraminidase. This is the first observation of compositional and substructural heterogeneity within drusen. We refer to these previously undescribed domains as “drusen cores” and report some of their characteristics.
Materials and Methods
Reagents
Reagents and supplies were purchased from the following sources. From EY Laboratories (San Mateo, CA) and Vector Laboratories (Burlingame, CA): fluorescein isothiocyanate (FITC)- and rhodamine-conjugated Limax flavus agglutinin (LFA), Triticum vulgaris agglutinin (WGA), Canavalia ensiformis agglutinin (ConA), and Arachea hypogea agglutinin (PNA). From Boehringer-Mannheim (Indianapolis, IN): neuraminidase (EC3.2.1.18; cleaves α2,3-, α2,6-, and α2,8-ketosidic bonds) derived from Clostridium perfringens. From Genzyme (Cambridge, MA): endo-α- N-acetylgalactosaminidase. From Sigma Chemical (St Louis, MO): peptide: N-glycosidase F and globulin-free bovine serum albumin (BSA). From Molecular Probes (Eugene, OR): 4′,6-diamidino-2-phenylindole (DAPI). From Poly Scientific (Bay Shore, NY): Sudan black B solution. From Bethesda Research Laboratories (Bethesda, MD): acrylamide and other reagents used for embedding. From Miles (Elkhart, IN): Optimal Cutting Temperature (OCT) compound. From Fisher Scientific (Pittsburgh, PA): Superfrost Plus slides.
Human Donor Eyes
Eyes with drusen from 61 human donors, 35 to 101 years of age, were obtained from the Iowa Lions Eye Bank (Iowa City, IA). Eyecups were preserved either in 4% paraformaldehyde in 100 mM sodium cacodylate, pH 7.4, or embedded directly in OCT compound and frozen in liquid nitrogen, without fixation, within 6 hr post mortem. Institutional Review Board approval was obtained from the Human Subjects Committee for the use of all donor tissues.
Fixation and Embedding
After 2–4 hr in fixative, eyecups were washed twice for 10 min in 100 mM cacodylic acid, pH 7.4, and wedges spanning from the macula to the ora serrata were dissected and embedded in acrylamide embedding media, as described previously (Johnson and Blanks 1984; Mullins et al. 1997). Tissues were then embedded in OCT, snap-frozen in liquid nitrogen, and stored at −80C.
Glycosidase Treatment
Sections of fixed and unfixed tissues were incubated overnight with 1 U/ml neuraminidase in 30 mM sodium acetate buffer, pH 5, at 37C in a humidified chamber. Sections from 61 eyes, 52 fixed and acrylamide-embedded and nine unfixed and OCT-embedded, were treated with neuraminidase and then labeled with FITC- or rhodamine-conjugated PNA. Adjacent control sections were incubated with buffer alone or with FITC. Serial sections from two eyes (one fixed and one unfixed) were treated with neuraminidase and labeled with LFA, ConA, and WGA to determine the effects of neuraminidase on labeling of drusen with other lectins. Fixed tissue sections from two additional donors were pretreated with neuraminidase and then treated with endo-α- N-acetylgalactosaminidase (O-glycanase, 1 U/ml in 15 mM sodium cacodylate, pH 6) or peptide: N-glycosidase F (PNGase F, 1 U/ml in PBS) for 72 hr at 37C. Control sections were treated with buffer alone.
Lectin Histochemistry
For lectin labeling, 6–8-μm-thick cryostat sections were cut, mounted on Superfrost Plus slides, labeled with PNA, WGA, ConA, or LFA as described previously (Mullins et al. 1997). Unlabeled adjacent sections were used as controls to distinguish between lectin binding and drusen autofluorescence. Sections were viewed on Olympus BH-2 or VH-2 fluorescence microscopes, using BP545, BP495, or UG-1 filter sets.
To compare labeling of enzyme- and buffer-treated sections, intensity and binding patterns on serial sections containing the same drusen were compared. Identical exposure times for experimental pairs were used during photomicrography and photographic processing.
Quantitation
The percentage of drusen cores was determined in eyes obtained from three donors. Stepped sections were collected at 60-μm intervals to avoid sampling the same core more than once and were pretreated with neuraminidase and labeled with FITC-PNA as described above. The numbers of cores in a total of 501 drusen were counted.
Results
Neuraminidase Treatment
Incubation of drusen-containing tissue sections with neuraminidase completely eliminated LFA labeling of drusen and other structures in the chorioretinal complex compared with controls. This loss of drusen labeling was used throughout the study to control for the efficacy of the enzyme treatment (Figure 1A and 1B) as were changes in labeling of the interphotore ceptor matrix, as described previously (Johnson and Hageman 1987; Kivela 1990).
Labeling of drusen with WGA and ConA was not significantly diminished after neuraminidase treatment (Figure 1C and 1D), demonstrating that the binding of these lectins with drusen-associated glycoconjugates is not primarily due to sialic acid. In some eyes, the intensity of WGA labeling of the choroidal stroma and neural retina decreased after neuraminidase treatment without a concomitant loss of WGA binding to drusen in the same section (Figure 1D).
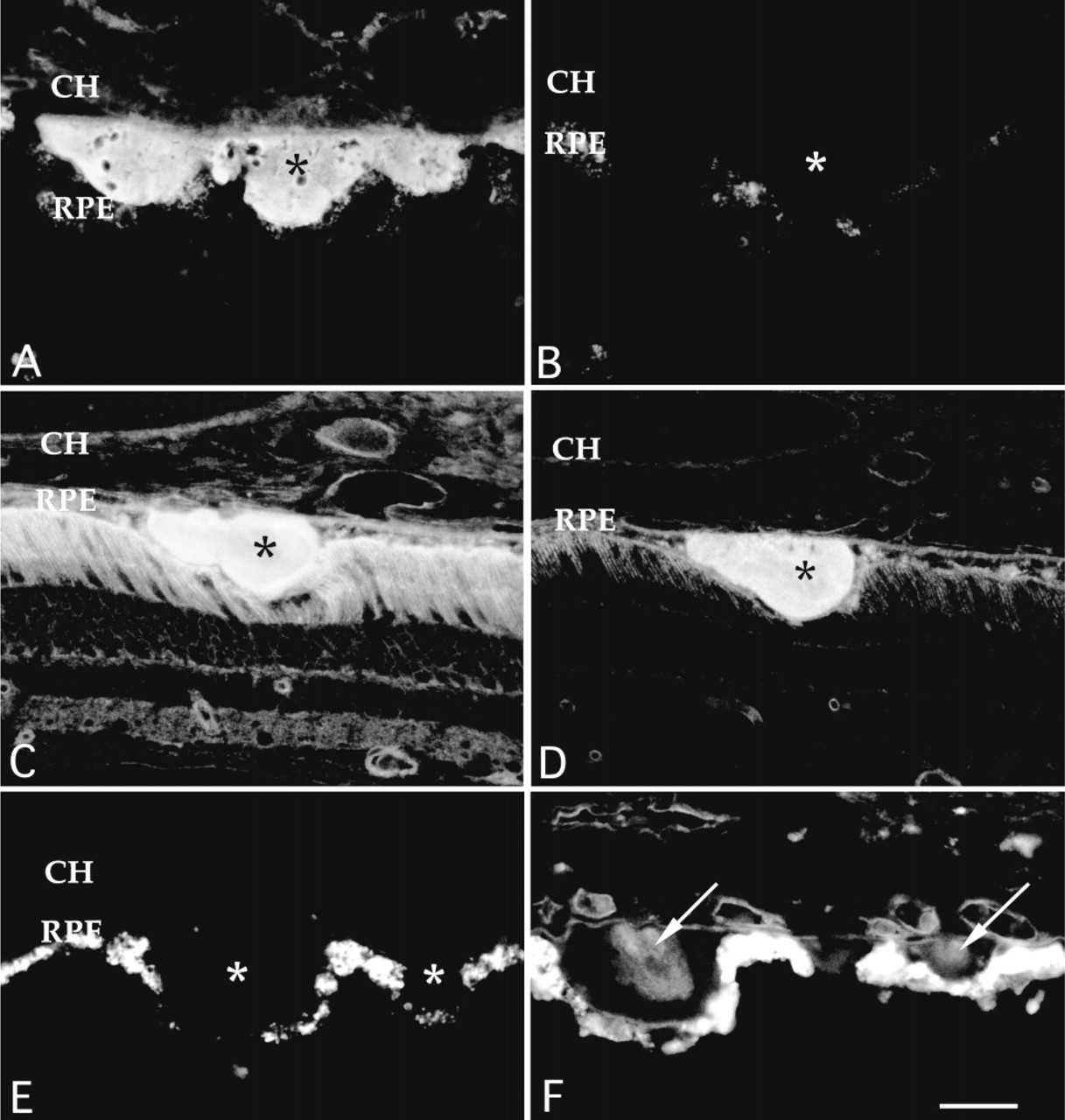
Cryostat sections of human donor eyes depicting the effects of neuraminidase treatment on lectin labeling. Buffer (A,C,E)- and neuraminidase (B,D,F)-treated tissue sections were incubated with LFA (A,B), WGA (C,D), or PNA (E,F). Neuraminidase pretreatment (B) completely removes the normally intense labeling of drusen glycoconjugates by LFA (A), a sialic acid binding lectin. This treatment has little or no effect on labeling of drusen with WGA (C,D). PNA, which normally does not bind drusen (E), binds to regions within drusen that are unmasked after neuraminidase treatment (F, arrows). These sites are localized to unique subdomains, termed drusen cores (arrows). RPE, retinal pigmented epithelium; CH, choroid; asterisks, drusen. Bar = 30 μm.
As described previously (Mullins et al., 1997), PNA does not normally bind to drusen or any other tissue within the RPE-choroid complex (Figure 1E). After preexposure to neuraminidase, the endothelial cells and/or the endothelial cell basal laminae of the choriocapillaris and other choroidal vessels, as well as the basal lamina of the RPE, were labeled intensely with PNA.
Strikingly, some drusen were labeled intensely by PNA after exposure to neuraminidase. This labeling was restricted to distinct subdomains or “cores” within drusen (Figure 1F). These cores were not observed in adjacent sections treated with buffer alone (Figure 1E). Drusen cores were typically spherical, centrally located within the druse, and juxtaposed to the inner collagenous layer of Bruch's membrane. Only one core was observed within any given solitary druse. Cores were not regularly observed within larger diffuse or confluent drusen. The sizes of drusen cores ranged from 5 to 38 μm in diameter, with a mean diameter of 14 μm. Both hard and soft drusen possessed cores, although large soft drusen typically had PNA binding cores that were larger and less centrally localized than those of hard drusen (e.g., Figure 2C and 2D). Cores were found in donors exhibiting a variety of ultrastructural drusen phenotypes (Hageman and Mullins in press). Significantly, cores were observed in the smallest of drusen. Cores were observed in both macular and peripheral drusen.
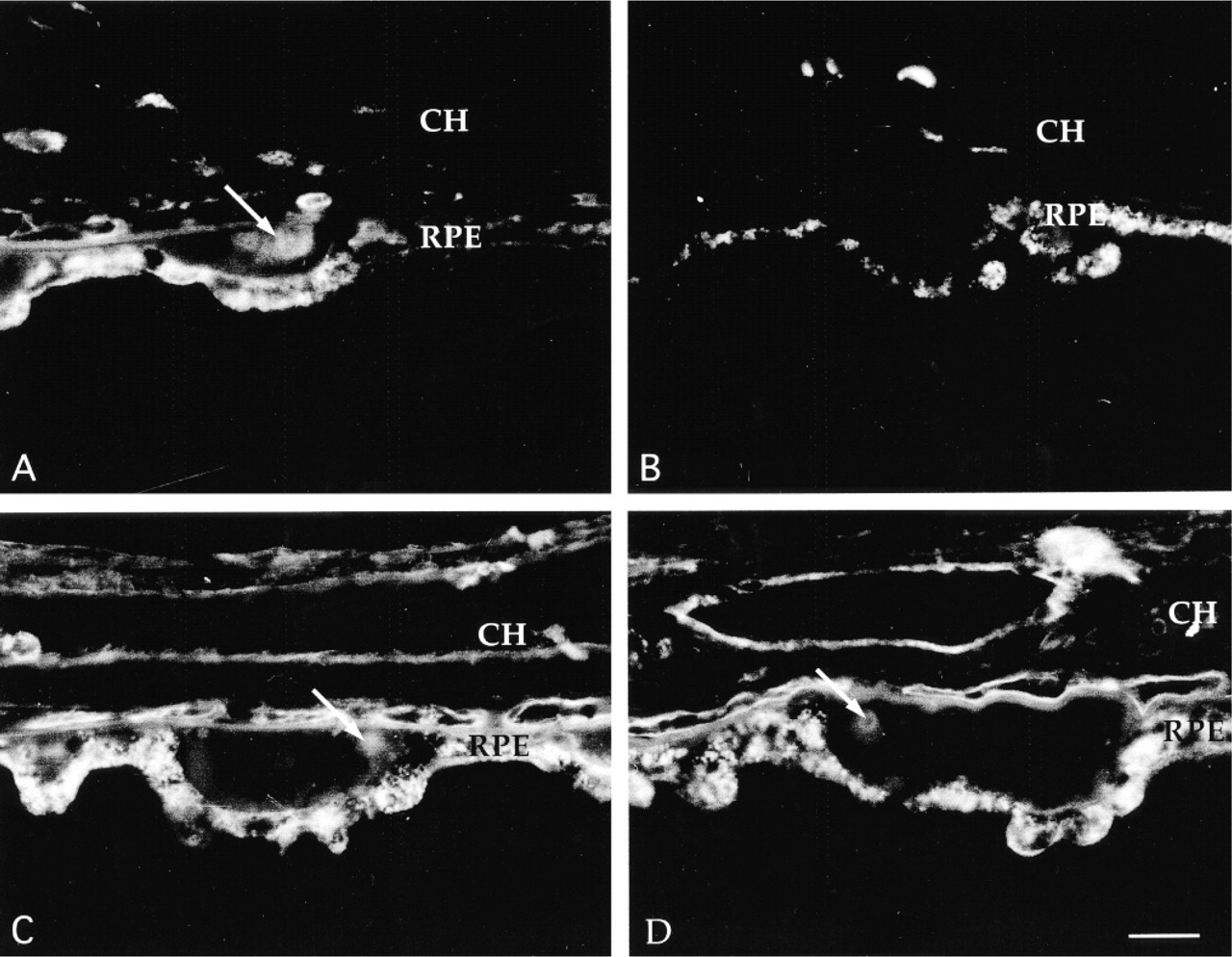
Enzymatic characterization of PNA binding drusen cores. Buffer-treated controls (A,C) and glycosidase-treated cryostat sections (B,D) of human donor eyes are depicted. Treatment with O-glycosidase (B) but not N-glycosidase (D) abrogates PNA binding to drusen cores (B) as well as to the neural retina and choroid, suggesting that these carbohydrates are covalently bound to O-linked glycoproteins. RPE, retinal pigmented epithelium; CH, choroid. Bar = 30 μm.
Drusen cores were observed frequently; in this study, cores were observed in 45 of 61 eyes examined. No differences in the appearance, relative size, and/or frequency of these deposits was noted with respect to fixation and embedding regimens. Intense labeling of drusen cores was observed using both FITC-PNA and rhodamine-conjugated PNA, demonstrating that this observation is not strictly due to the interaction of drusen with the fluorophore. Although drusen fluoresced when incubated with FITC alone, this binding was not associated with core subdomains. The percentage of cores was determined in three donors. Cores were observed in 22.2, 29.7, and 33.7% of drusen in each of the three donors. From a combined total of 501 drusen, 134 exhibited cores (26.7%). Twelve small (<60 -μm) drusen were bound in their entirety by PNA (2.4%).
Enzymatic Characterization of Drusen Cores
After sequential incubation of tissue sections with neuraminidase and O-glycanase, PNA labeling of drusen cores was significantly reduced (Figure 2A and 2B). Labeling of the choroid and interphotoreceptor matrix was nearly or completely abrogated after O-glycanase treatment (not shown). O-Glycanase treatment had little effect on labeling of rod outer segments with ConA, suggesting that contaminating N-glycosidase activity was not present (not shown). In contrast to O-glycanase treatment, PNGase F pretreatment did not change the intensity of PNA labeling of drusen cores (Figures 2C and D). ConA labeling of rod outer segments was completely abrogated after incubation with PNGase F, demonstrating that the enzyme efficiently removed N-linked glycans (not shown).
When examined under UV fluorescence optics, drusen cores were frequently less autofluorescent than the rest of the druse in both PNA-labeled and unlabeled tissue sections (Figure 3B). DAPI, a DNA-specific probe, did not label cores (Figure 3B). In addition, the lipid stain Sudan black B did not bind to drusen cores (Figure 3 C), although the remainder of the druse was stained.
Discussion
We noted previously that some drusen possess heterogeneously distributed glycoconjugates (Mullins et al. 1997). For example, PHA-L and WGA occasionally bind to subdomains of drusen. In this study we demonstrate that PNA binds subdomains, termed “drusen cores,” within drusen after neuramindase treatment. One significant finding from the studies presented here is that drusen cores are common. In this study, at least some drusen in 74% of the eyes examined possessed PNA binding cores. In addition, 26.7% of all drusen examined possessed cores. These included ultrastructurally homogeneous drusen as well as heterogeneous drusen (Hageman and Mullins in press). Because cores represent a proportionally small domain within any given druse and because the section plane will not always pass through the core of any given druse, it is likely that cores are present in a greater frequency of drusen than we observed.
The observations that PNA binds discrete drusen subdomains (drusen cores) after neuraminidase treatment and that LFA, a sialic acid binding lectin, does not bind specifically to drusen cores suggest that at least two distinct classes of terminal sialic acidcontaining glycoconjugates are present in drusen and that they are distributed in different regions of drusen.
The PNA binding disaccharides (galactose β1,3 N-acetylgalactosamine) is frequently associated with O-glycosidically linked sugars but is also associated with some gangliosides (Zeller and Marchase, 1992) The observation that O-glycosidase eliminates PNA binding to drusen cores suggests that the PNA binding molecule(s) present in drusen cores consists of glycoproteins with O-glycosidically linked oligosaccharides, because this enzyme does not cleave carbohydrates from glycolipids (Umemoto et al. 1977). Furthermore, the exclusion of Sudan black B from drusen cores suggests that these domains are relatively free of lipid.
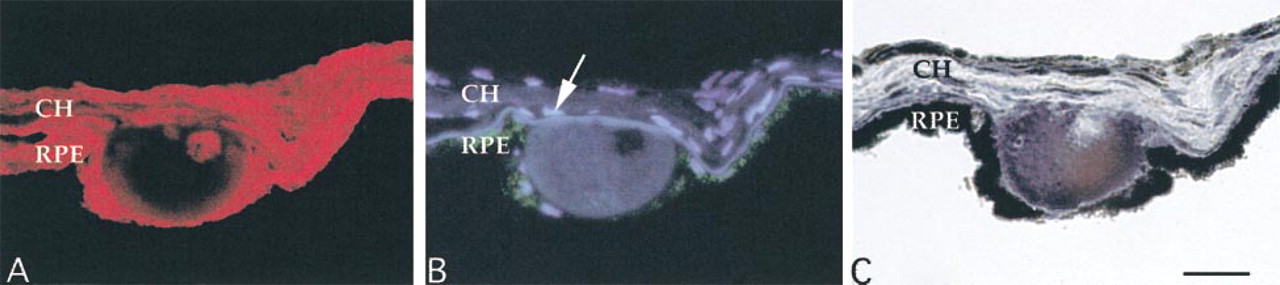
Serial sections of a druse labeled with PNA-rhodamine after neuraminidase treatment (A), DAPI/UV autofluorescence (B), and Sudan black B (C). Neuraminidase treatment of a druse exposes a PNA binding core (A). When the same section is viewed under UV filters, most autofluorescence of the druse is confined to the material around the core. Nuclear staining of the core with DAPI is not noted (B). Serial sections incubated with Sudan black B (C) reveal staining of the material surrounding the core, suggesting that drusen cores constitute relatively hydrophilic domains. Arrow, nuclear labeling of a choroidal cell; RPE, retinal pigmented epithelium; CH, choroid. Bar = 30 μm.
In summary, we have identified a carbohydrate moiety that, in contrast to other drusen-associated glycoconjugates that have been described, is localized to novel, discrete basal subdomains, or cores, within drusen. Although previously unrecognized, these cores are common components of drusen and appear to be composed of glycoproteins with O-glycosidically linked carbohydrate chains. Whereas sialic acid residues, as assessed by LFA binding, are distributed throughout drusen, at least one distinct glycoprotein possessing PNA binding epitopes that are masked by sialic acid is localized to these structures.
It is conceivable that drusen cores develop late in drusen ontogeny, although this is not strongly supported by our data. We propose that it is more likely that these subdomains are related to drusen biogenesis. The observations that solitary drusen have only one core and that these cores are typically located centrally and basally within drusen suggest that they may represent an early nucleation site around which other drusen-associated molecules are subsequently deposited. The remaining constituents of drusen, including lipids, would be deposited around the core. This contention is consistent with the observations that small PNA-positive structures are frequently visualized beneath the RPE and that larger confluent/diffuse drusen are less likely to have cores than smaller drusen.
We propose that the identification of the precise biological identity and source(s) of drusen core-associated glycoproteins may provide new insights into the genesis of drusen and into the etiology of age-related macular degeneration. Ongoing efforts are being directed towards this goal.
Footnotes
Acknowledgements
Supported in part by NIH grants EY06463 (GSH), EY11515 (GSH), and by an unrestricted grant to the University of Iowa Department of Ophthalmology and Visual Sciences from Research to Prevent Blindness, Inc. GSH is the recipient of the RPB Lew R. Wassarman Merit Award.
We thank Dr Stephen Russell for evaluation of donor eye tissues, Markus Kuehn for helpful discussions, and Bobbie Schneider, Cory Speth, Heidi Nienhaus, and Lisa Thayer for superb technical assistance.
