Abstract
The oligosaccharide sequences of glycoconjugates in the human normal epididymis and the nature of linkages were studied with lectin histochemistry. The usual terminal sequences of oligosaccharide side chains in epithelial cell secretions were Neu5Ac2,3Galβ 1,3GalNAc; SO4 Galβ1,3GalNAc; and Galβ1,4GlcNAc, and they were mainly found in O-linked glycoproteins. The lectin pattern of mitochondria-rich cells differed from that of principal cells.
L
The testes and epididymides from 17 men aged 25-70 years, without reproductive disease and showing complete spermatogenesis, were obtained from autopsies 6-10 hr after death and processed for light and electron microscopy. To evaluate postmortem changes, three epididymides were obtained from testicular tumor surgery and processed in the same way.
The lectin histochemical procedures applied have been previously reported in detail (Leis et al. 1997). For light microscopy, either horseradish peroxidase (HRP)- or digoxygenin (DIG)-conjugated lectins were used. For HRP lectins, endogenous peroxidase was blocked with H2O2 and the sections were incubated with the lectins (Sigma; Barcelona, Spain) (Table 1): PNA (25μg/ml), ConA (20μg/ml), LTA (25μg/ml), WGA (10μg/ml), SBA (18μg/ml), UEA-I (20μg/ml), HPA (6μg/ml), and DBA (15μg/ml). Peroxidase was developed with diaminobenzidine (DAB) and H2O2. For DIG-lectins, endogenous peroxidase activity was destroyed by pretreatment with H2O2 and the sections were incubated with the lectin-DIG conjugates (Boehringer; Mannheim, Germany): DSA (10μg/ml), MAA (10μg/ml), AAA (20μg/ml), SNA (15μg/ml), and GNA (60μg/ml) (Table 1). Sections were incubated further with peroxidase-conjugated anti-DIG Fab fragments. Peroxidase activity was visualized with DAB and H2O2.
For electron microscopy, the samples were fixed in glutaraldehyde-paraformaldehyde and embedded in Lowicryl K4M. Ultrathin sections were incubated with: (a) ConA-gold lectin (10 nm); (b) unlabeled DSA and then floated on a drop with 15-nm gold-labeled ovomucoid; or (c) a lectin-DIG (MAA, SNA, AAA, SBA, PNA, WGA, UEA-I, HPA, or GNA), incubated further in sheep anti-DIG monoclonal antibody, and floated on a drop of donkey anti-sheep 15-nm gold-labeled IgG (BioCell; Cardiff, UK). To evaluate staining intensity of each lectin, the average number of colloidal gold particles per μm2 was calculated for each subject and epididymal zone in 8-16 electron micrographs of 5-12 ultrathin sections.
The carbohydrates that could be masked by sialic acid were exposed with HCl (paraffin sections) or neuraminidase (ultrathin sections). Removal of sulfate esters was carried out by treatment with HCl and thereafter with Ba(OH)2 in paraffin sections or before embedding (ultrathin sections). Elimination of O-linked oligosaccharides (β-elimination) was carried out by incubation with NaOH in paraffin sections or before embedding. Hydrolysis of N-linked oligosaccharides was done by treatment of both section types with endob-acetylglu-cosaminidase F/peptide N-glycosidase F (endo-F) (Boehringer). Hydrolysis of terminal glucose residues was done in both section types by treatment with glucose oxidase (Sigma).
Labeling intensity and cellular localization of histochemical reactions to lectins in the epithelium of the human epididymis a

aGlcNAc,
bStaining decreased with endo-F digestion.
cStaining decreased with β-elimination.
dStaining increases with desialization.
eStaining increased with endo-F digestion.
f No staining with endo-F digestion.
gNo staining with β-elimination.
hStaining increased with desulfation.
The control procedures were substitution of conjugated and unconjugated lectins by their buffers and preincubation of each lectin with its hapten-sugar inhibitor (Sigma).
Comparison of surgical and autopsy specimens showed no histochemical changes. The results of lectin histochemistry are summarized in Table 1 and illustrated in Figures 1–10. The exact location and intensity of lectin labeling in epididymal cells were evaluated by electron microscopy. No labeling was found in control sections for all lectins and treatments used.
Lectin reactions in the epididymis could be related to structural oligosaccharides (microvilli and cytosol) and/or glycoprotein secretion (electron-dense vesicles and luminal content). The decreased WGA reactions after endo-F digestion and β-elimination suggest that terminal GlcNAc residues are in both N- and O-linked oligosaccharides. WGA receptors and GlcNAc residues have been reported over the sperm head (Liu et al. 1991). The increased WGA reaction after desialization in the cauda epididymidis suggests addition of Neu5Ac in this region. Sialoglycoconjugates have been found in the human sperm tail (Focarelli et al. 1995).
SNA labeling was absent in the lumen, weak in principal cells, and more intense in mitochondria-rich cells. Because this reactivity was maintained with β-elimination, the labeled residues are probably present in both N- and O-linked oligosaccharides. The disappearance of MAA labeling after β-elimination in the cells and lumen of the ductus epididymidis suggests secretion of Neu5Acα2,3Galβ1,4GlcNAc in O-linked glycoproteins.
Chemical treatments for PNA suggest the presence of three different chains (Galβ1,3GalNAc, Neu5AcGalβ1, 3GalNAc, and SO4-Galβ1,3GalNAc) in O-linked oligosaccharides. Sulfate has been implicated in gamete adhesion, induction of the acrosome reaction, and sperm-egg interactions (LoLeggio et al. 1994). Reactions to DSA suggest O-linked oligosaccharides with Galβ1, 4GlcNAc sequences without sialic acid of sulfate groups. AAA probably binds fucose residues that areα1, 6-linked to glycoproteins (Yamashita et al. 1985). Chemical treatments used here indicate that these residues are in O-linked oligosaccharides. GNA appears to bind terminal mannoses in high-mannose N-linked oligosaccharides, whereas ConA would label trimannosyl cores in complex carbohydrates (Brewer and Bhattacharyya 1988). GNA reactivity could be located in lysosomes, whereas ConA reaction would be associated with secreted and/or absorbed glycoproteins. DBA, SBA, and HPA preferentially bind nonreducing Gal- NAc residues (Spicer and Schulte 1992). The increase in HPA and SBA labeling with desialization and desulfation suggests the presence of Neu5AcGalNAc and SO4-GalNAc residues.
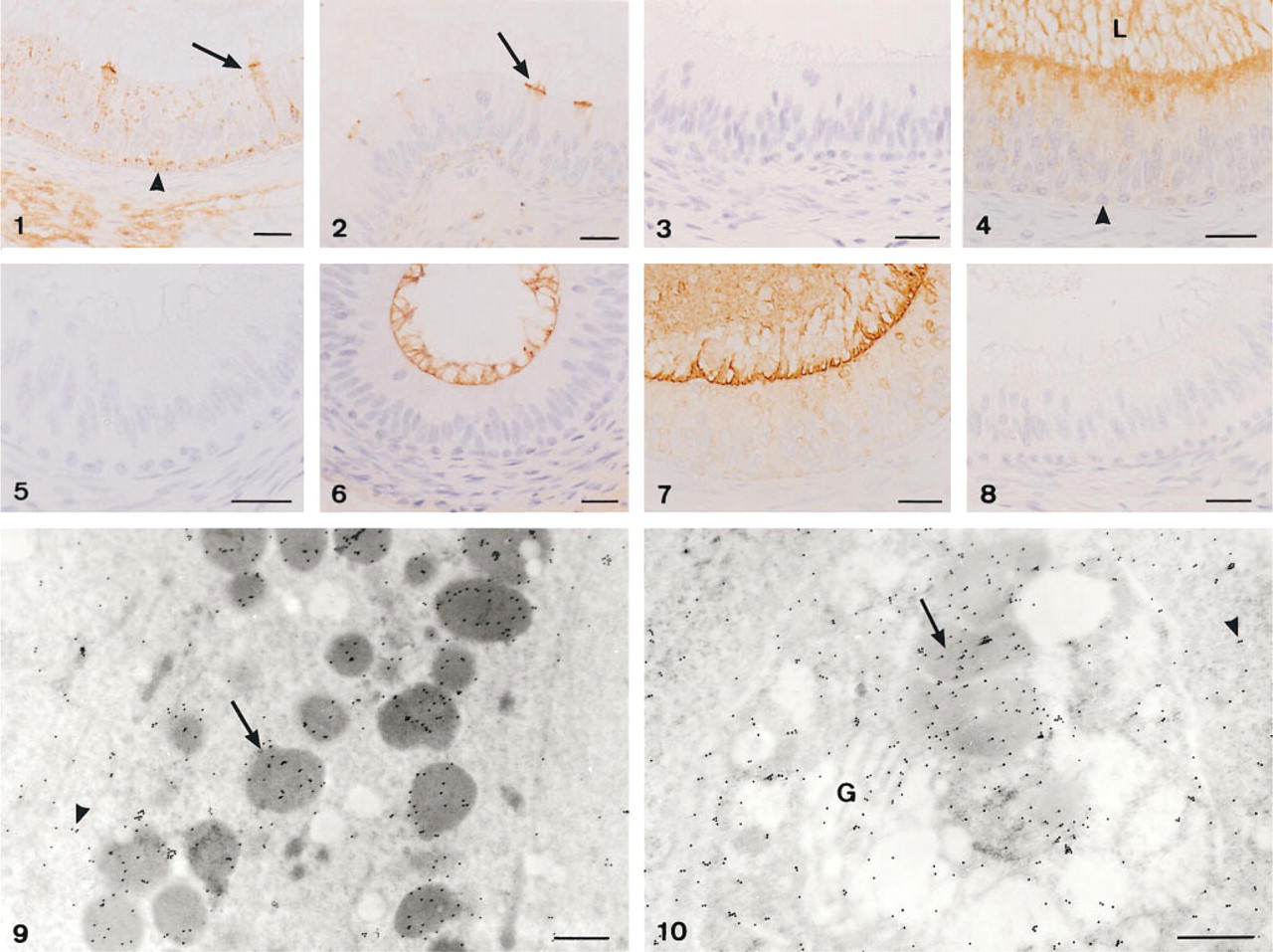
Lectin labeling of the human ductus epididymidis (corpus).
