Abstract
Affordable and physiologically relevant three-dimensional (3D) cell-based assays used in high-throughput screening (HTS) are on the rise in early drug discovery. These technologies have been aided by the recent adaptation of novel microplate treatments and spheroid culturing techniques. One such technology involves the use of nanoparticle (NanoShuttle-PL) labeled cells and custom magnetic drives to assist in cell aggregation to ensure rapid 3D structure formation after the cells have been dispensed into microtiter plates. Transitioning this technology from a low-throughput manual benchtop application, as previously published by our lab, into a robotically enabled format achieves orders of magnitude greater throughput but required the development of specialized support hardware. This effort included in-house development, fabrication, and testing of ancillary devices that assist robotic handing and high-precision placement of microtiter plates into an incubator embedded with magnetic drives. Utilizing a “rapid prototyping” approach facilitated by cloud-based computer-aided design software, we built the necessary components using hobby-grade 3D printers with turnaround times that rival those of traditional manufacturing/development practices at a substantially reduced cost. This approach culminated in a first-in-class HTS-compatible 3D system in which we have coupled 3D bioprinting to a fully automated HTS robotic platform utilizing our novel magnetic incubator shelf assemblies.
Introduction
Efforts to develop high-throughput screening (HTS) methodologies using cultured cells as three-dimensional (3D) spheroids and organoids have gained momentum for drug discovery because they are more physiologically relevant than traditional monolayer assays and recently have become more cost effective and less complex for large-scale screening campaigns.1–4 Technologies that enable cell culture in 3D are referred to as 3D bioprinting and include photolithography, magnetic bioprinting, stereolithography, and direct cell extrusion.5–10 The most critical, unmet challenge of 3D bioprinting has been scaling production in order to meet the needs of traditional HTS, which requires hundreds of thousands of wells to be tested for a given campaign. 11 Of the 3D methods currently available, magnetic bioprinting using cell-repellent surface treatment plates from Greiner Bio-One (Monroe, NC) and ultra-low-attachment (ULA) microcavity plates from Corning Inc (Corning, NY). has proven most promising in its utility to meet the scalability needs of HTS.12–20
3D magnetic bioprinting has important advantages for HTS, but also presents hurdles in its integration. The largest challenge is ensuring that magnetic drives are fabricated and installed with sufficient precision for proper alignment of the microtiter plate wells with the magnets found on the drives, all the while remaining as minimally invasive to the system hardware and software components as possible. Enabling this procedure in an HTS environment requires fabricating and installing multiple embedded magnetic drives into the plate incubator, all with acceptable tolerances to minimize drive-to-plate placement variability. Error in this capacity has greater significance for the 1536-well format, where ~2 mm wells must align with smaller-diameter magnets requiring critical co-alignment in the
In this paper, we are focused on the validation and testing of the Greiner Bio-One m3D and cell-repellent microplate technology to enable 3D bioprinting for HTS, thereby extending previously published techniques for 384-well and 1536-well offline nonrobotic formats to a 1536-well robotic format.21,22 NanoShuttle-PL employs a nanoparticle assembly technique consisting of gold, iron oxide, and poly-

(
Additional details regarding the assay biology, usage, and validation of m3D technology compared with a traditional 2D monolayer variant of the assay in an offline nonrobotic capacity can be found in a previously published manuscript from our lab. 22 Herein, we describe the validation and testing of the m3D and cell-repellent microplate technology in 1536-well format to enable 3D bioprinting and prove its utility for fully automated large-scale spheroid and organoid production to support HTS campaigns.
Materials and Methods
Custom Fabrication with CAD
Rapid prototyping of needed hardware was readily facilitated through 3D printing of custom thermoplastic parts, greatly mitigating problems and reducing lengthy turnaround times that traditional methods in engineering experience with fabrication, testing, and redesign.23,24 To facilitate the rapid prototyping and testing of custom incubator shelves, we fabricated various magnetic shelf designs using fused deposition modeling 3D printers with polylactic acid (PLA) thermoplastic. A RepRapPro (Foxham, England) Tricolor Mendel was used to fabricate parts for 384- and 1536-well first-generation prototypes (

(

(
Modification of Shelves
Over the course of this effort, we developed three designs in a sequential fashion with each attempting to address limitations of the previous design. The first- and third-generation designs involved the repurposing of existing magnetic drive assemblies manufactured by n3D Biosciences (Houston, TX) that were initially provided for nonrobotic assay use, while the second-generation design required individual magnetic cylinders to be purchased and inserted directly into the prototype shelf. The 3D cell culture system components utilized include flat-bottom, cell-repellent, black-wall, clear-bottom 384-well and 1536-well plates (specialized version of parts 781976 and 789979, respectively; Greiner Bio-One); 384- and 1536-well m3D magnetic spheroid drives (specialized versions of parts 781835 and 789830, respectively; Greiner Bio-One); and NanoShuttle-PL (part 657846; Greiner Bio-One). Note that Greiner Bio-One m3D spheroid drives were referred to as n3D Bioscience drives prior to acquisition of the technology by Greiner.
HTS System Overview
The facility at Scripps Florida uses a GNF Systems (San Diego, CA) HTS platform to execute hundreds of biochemical and cell-based assays in either 384- or 1536-well plate format.
28
This platform consists of three integrated, environmentally controllable microtiter plate incubators, two dispensers, a pintool transfer station, a centrifuge, and a suite of plate readers. Each of the robotically accessible incubators contains a rotating carousel composed of 18 segmented plate hotel stacks around the incubator’s circumference, with each hotel containing 27 shelves for plate placement. Any hotel stack can be readily removed from the carousel. The hotel stacks can be further disassembled to gain access to the individual shelf positions where the microtiter plates reside during incubation. By removing and fully disassembling a shelving unit, seen in
Figure 1D
, custom magnetic drive shelves can be adapted and installed to create a modified hotel shelf, as seen in
Figure 3A
, and its final installation onto the carousel (

GNF shelf stack installed into GNF robotic carousel.
Performing the Screening Assay
The assay we used to test the function of the first-generation, second-generation, and third-generation magnetic drivers was initially developed in the 384-well format and then miniaturized to the 1536-well format. The protocol that we used for the assay required incubating the assay plates on the magnetic drivers in the following steps: (1) Add NanoShuttle-PL to cells in a flask (0.6 mL per T175) and incubate the cells overnight at 37 °C, 5% CO2, 95% relative humidity (RH); (2) harvest the cells and seed 1250 cells in 5 µL of culture medium to a 1536-well Greiner cell-repellent plate; (3) put the plate atop the spheroid drive and incubate the plate on the drive for 4 h; (4) incubate the cells for 24 h at 37 °C, 5% CO2, 95% RH; (5) pintool transfer 10 nL of compounds; (6) incubate cells for an additional 3 days at 37 °C, 5% CO2, 95% RH; (7) image spheroids using high-content screening, and (8) dispense 5 µL of CellTiter-Glo 3D reagent, shake for 10 min, incubate for 60 min at room temperature, and read on ViewLux. This assay is described in greater detail in a previous manuscript from our lab, which discusses the initial efforts to develop this assay and the results of preliminary offline screening in the 1536-well format against an internally curated approved drug library of ~3300 compounds. 22 Concentration–response curves (CRCs) and IC50 values of pharmacological control compounds were used as a guide for assay optimization and validation for drug screening.
Results and Discussion
First-Generation Design
Our initial design efforts to enable automation of the m3D technology involved adapting the existing lab-bench 384-well magnetic drives onto a custom 3D printed HTS incubator shelf. To make the magnetic drives robotically compatible, we removed the side alignment tabs needed for plate alignment when used with nonrobotic assays (
The first-generation HTS incubator shelf design for the 384-well m3D magnetic drive is seen with and without the magnetic drive installed in
Figure 1F
and
Second-Generation Design
After this first-generation design was tested and found to be functional, we addressed the primary limitation of the initial design: alternating empty slots in the plate hotel resulting in a loss of HTS capacity due to the height requirements of the initial shelf design. The second-generation design addressed this limitation by incorporating the magnets directly into the incubator shelves, as seen in
The second-generation 384-well drive used stock magnets that were 1/16-inch by ¼-inch cylindrical neodymium magnets from Apex Magnets (Petersburg, WV) at a cost of $8.99 per 50 magnets, not including shipping. 29 For the 1536-well drive, custom-manufactured 3/64-inch by ¼-inch cylindrical neodymium magnets were ordered from SM Magnetics (Pelham, AL) at a cost of $1.05 per magnet, not including shipping. 30 These custom magnets were ordered with a tolerance of 0.05 mm, nickel plated, and magnetized through the length of the magnet. Due to the high cost of the magnets for the 1536-well prototype, only 250 magnets were ordered to provide a sufficient quantity to partially populate a 1536-well driver and validate the design.
We found that a limitation of the second-generation design was the need for magnets to be inserted into custom incubator shelving, requiring a mechanical jig to facilitate loading the magnets into the prepositioned holes without breaking during the press fit. To address this limitation, we designed a magnetic drive jig that sits over the top of the modified shelf during the assembly process to ensure magnets were perpendicular, matched in height, and not damaged during installation. These magnetic installation jigs can be seen to the left of the drives in
Third-Generation Design
The second-generation drives worked well with robotic testing; however, we found the 1536-well magnets to be expensive and the assembly of both the 384- and 1536-well drive shelves to be laborious. A third prototype, seen in
Figure 3A
, was created that reused preexisting drives obtained from Greiner Bio-One and n3D Biosciences. This design also eliminated the Z-height clearance issue of the first-generation design by installing a set of winged adapters on the sides of the original drive plate. One of the challenges with this third design was ensuring the proper front-to-back alignment of the adapters from drive to drive. To address this constraint, a
At each phase of development, the magnetic drive shelves were installed into the robotic incubator (
One of the primary benefits of the magnetic 3D bioprinting approach is that it requires no significant changes to the HTS platform, nor does it interrupt standard operation apart from the addition of magnetic drives installed within the HTS incubators. Once the drives are installed, the 3D bioprinting assays can commence using established HTS routines with no need of additional support hardware or customized software to complete the adaption.
Screening Assay
To demonstrate the robustness and high-throughput capabilities of 3D spheroid screening, a large-scale screen comprising 151,977 compounds was run using the third-generation m3D magnetic drives (
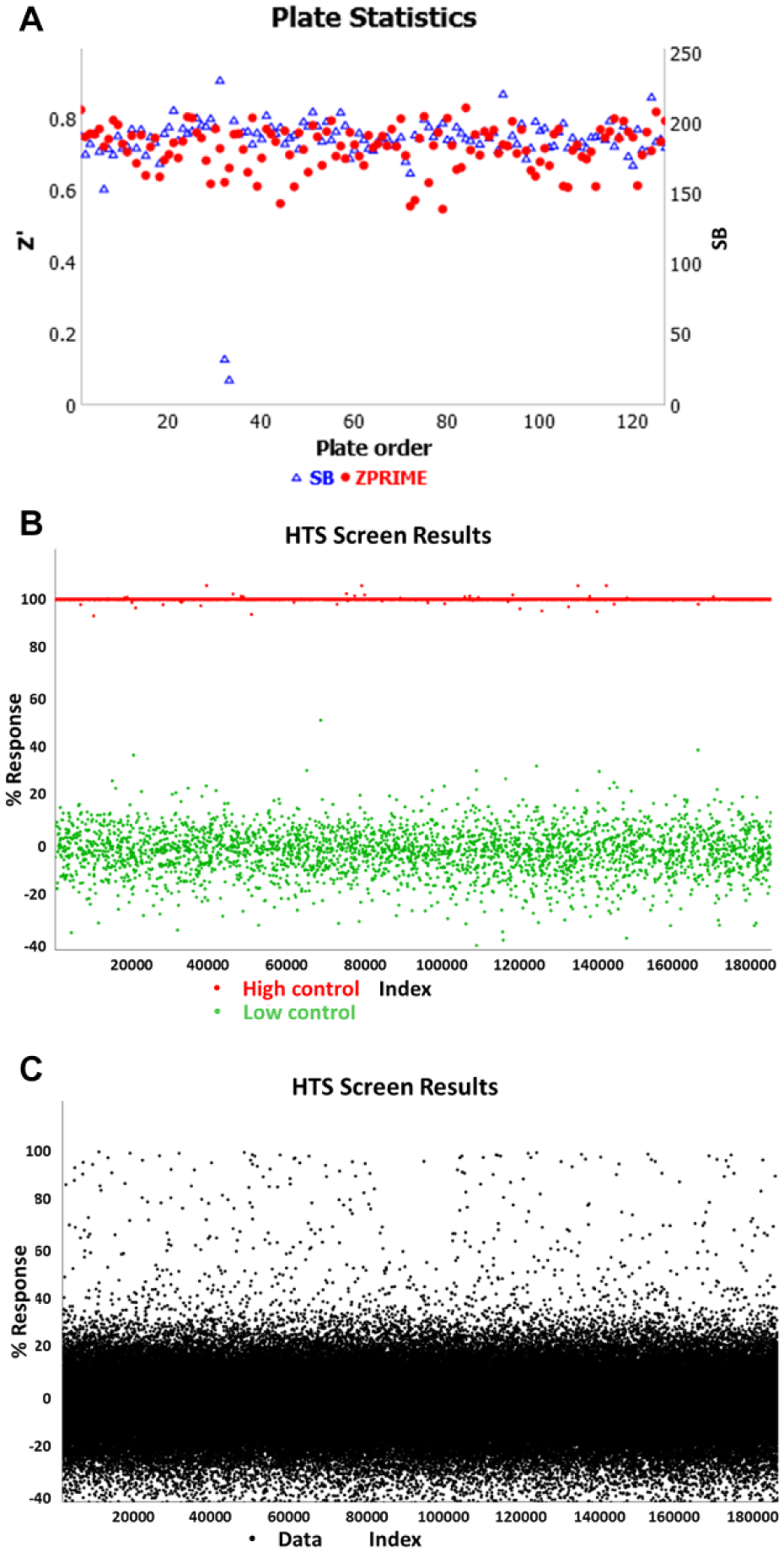
(
CRC of control compounds in 1536-well offline nonrobotic screening validation results were compared against 1536-well online robotic screening validation results and can be seen in
Figure 6A
and
CRC Values of Control Compounds Used for Robotic Validation.
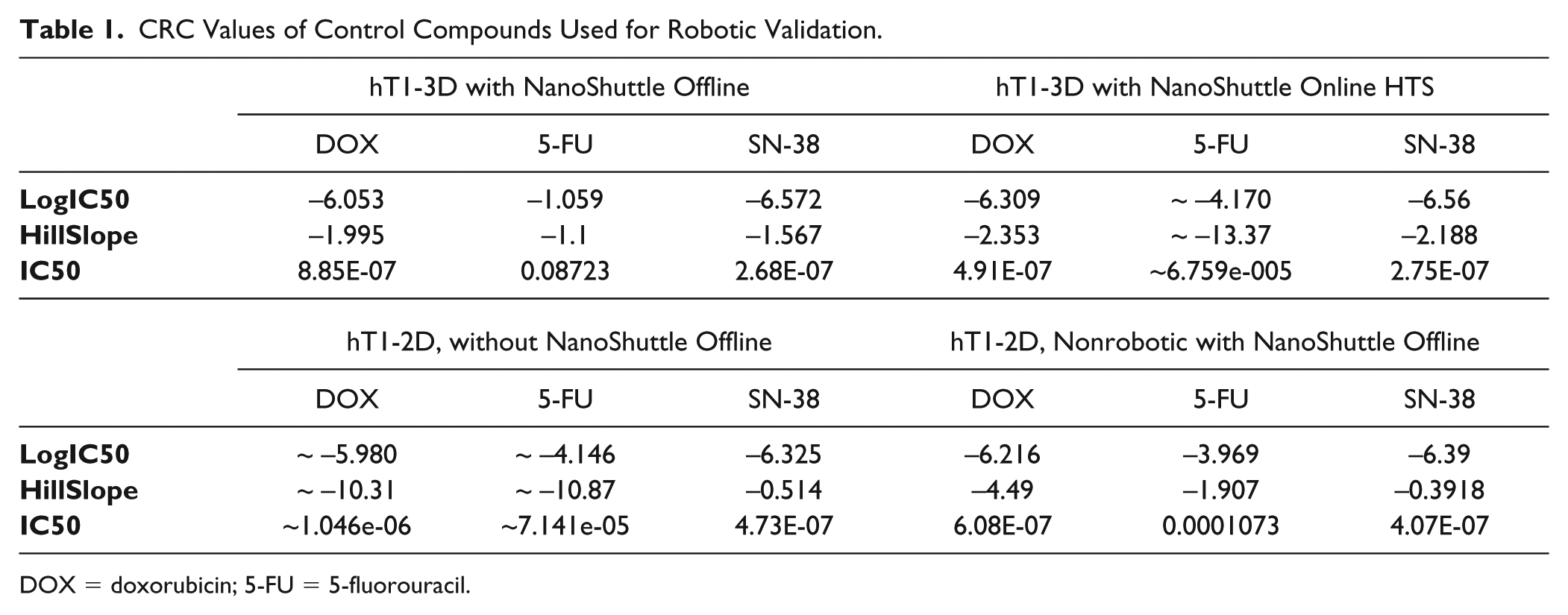
DOX = doxorubicin; 5-FU = 5-fluorouracil.
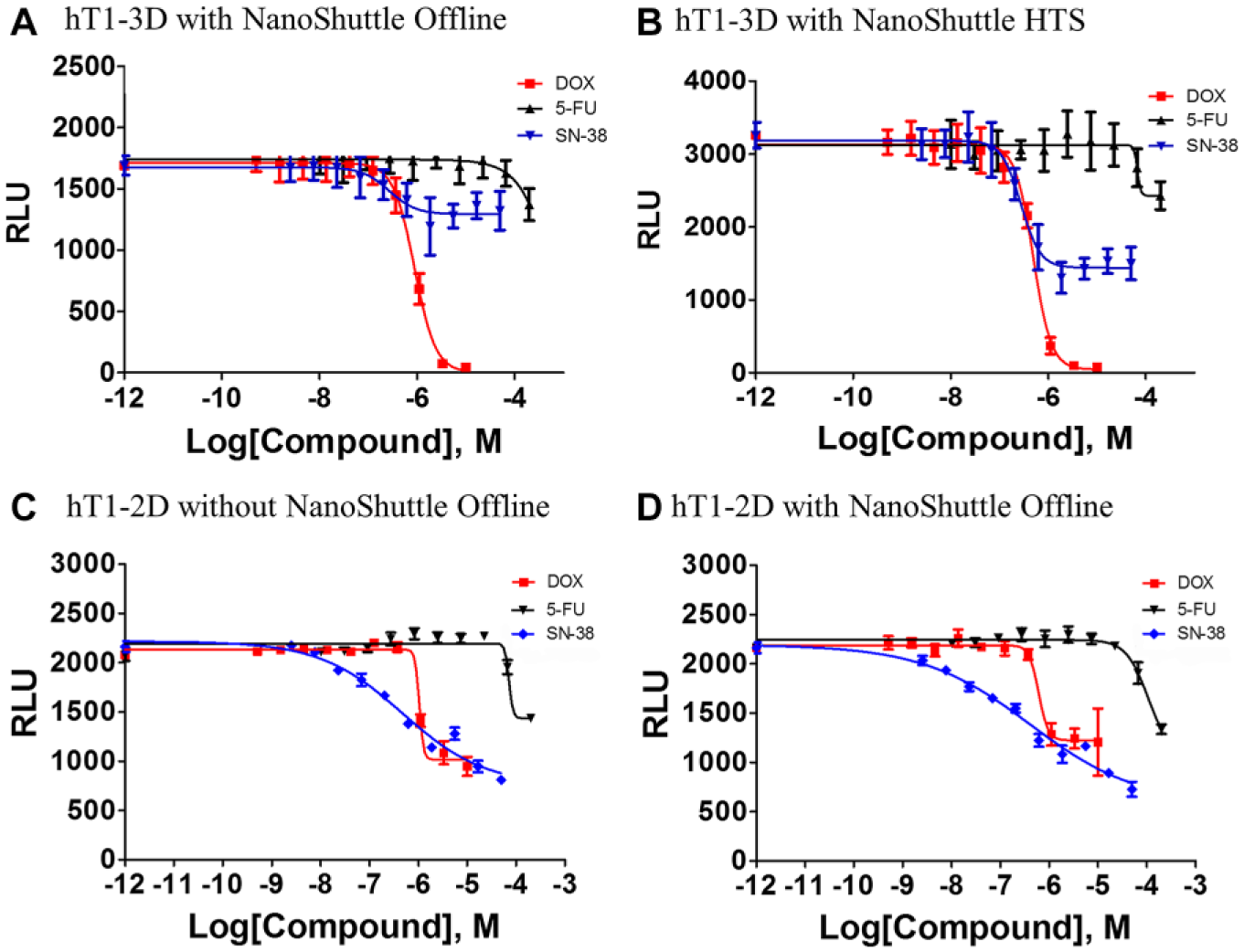
(
For the pilot screen, spheroids were grown from hT1 cells and then quantified using a CellTiter-Glo 3D viability assay. The hT1 cells were derived from patient pancreatic cancer cells intended for use in primary cancer cell research.
34
The pilot screen statistics were monitored with an average Z′ factor of 0.72, a signal-to-background ratio of 188.13, and a hit rate of 0.48%. The ability of 3D spheroid formation for hT1 pancreatic cancer cells is different compared with other cells lines like hT29, which is extremely compact with a well-defined surface. 3D formation structures for hT1 cells have been confirmed by Z-stack analysis using confocal microscopy. In addition, the cells were cultured in Corning spheroid plates to test side by side with the 3D bioprinting technology, which confirms that the spheroid formation is independent of the technology used. This was done to address concerns that the PLL might interfere with endogenous cell aggregation behavior and resulted in confirmation that the PLL did not interfere, as confirmed by previous publications.12,21,35-38 Spheroids produced were found to be uniform as validated previously
22
and by the tight reproducibility of the IC50 values of known anticancer drugs such as doxorubicin. This was assessed using a CRC plate that was included in each robotic HTS run for the 150K initiative, which determined doxorubicin to have an average IC50 value of 398 nM with only a variance of ~13% (
The 150K screen efforts described in this paper have demonstrated that (1) 3D bioprinting of spheroids is feasible in a 1536-well format; (2) full robotic automation and plate handling can be employed for spheroid production, drug dosing, and plate readout; and (3) large-scale HTS screening can be implemented using spheroids in a cost-effective fashion relative to other 3D technologies. When compared with traditional 2D bioassays, bioprinting techniques using conventional 1536-well microplates were found to be threefold higher in cost. However, this compares favorably against bioprinting in specialized microcavity microplates, which were found to be five- to sixfold higher in cost.
Footnotes
Acknowledgements
We would like to acknowledge Line Deluca for her help with compound management as well as Herve Tiriac and David Tuveson at CHSL for initial provision of the primary pancreas cancer hT1 cells.
Authors’ Note
Spicer and Scampavia codirect the screening center at Scripps and this work was supported by the R33 Grant under the PI, Spicer. In addition, this work was overseen and directed by both investigators with multiple authors under their individual supervision.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under award number R33CA206949. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
